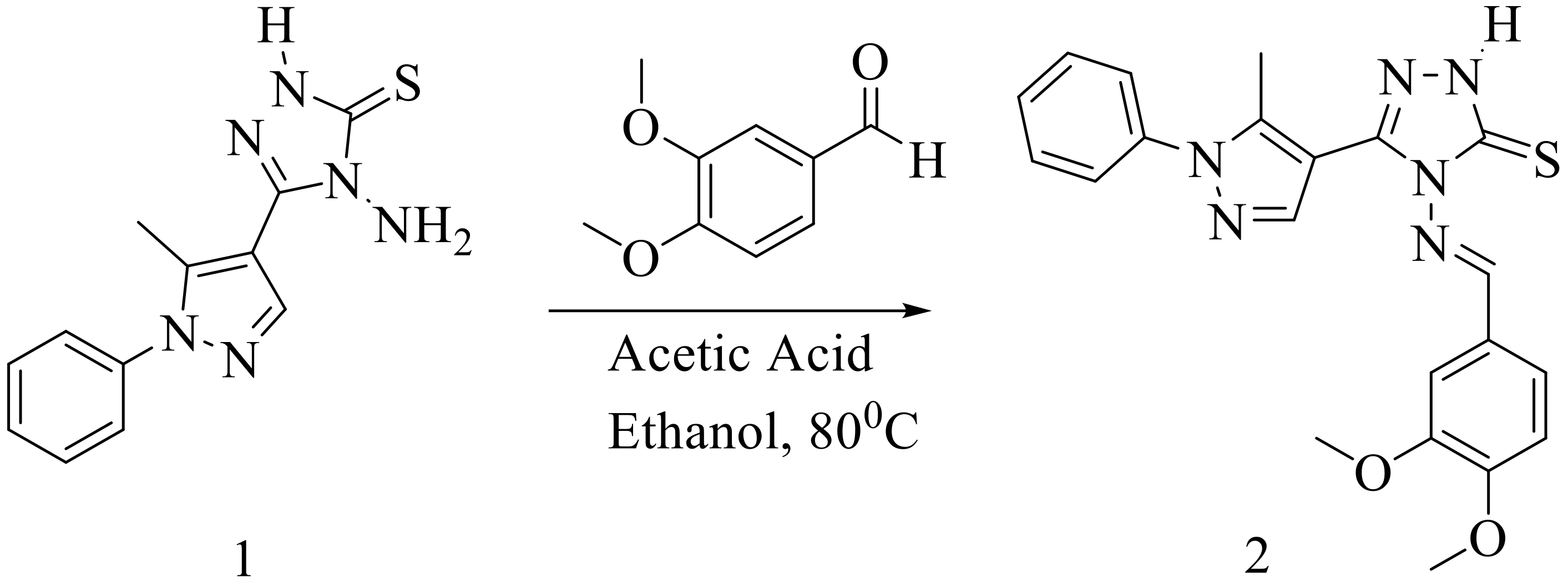
4-[(3,4-Dimethoxybenzylidene)amino]-5-(5-methyl-1-phenyl-1H-pyrazol-4-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione
Abstract
1. Introduction
2. Results
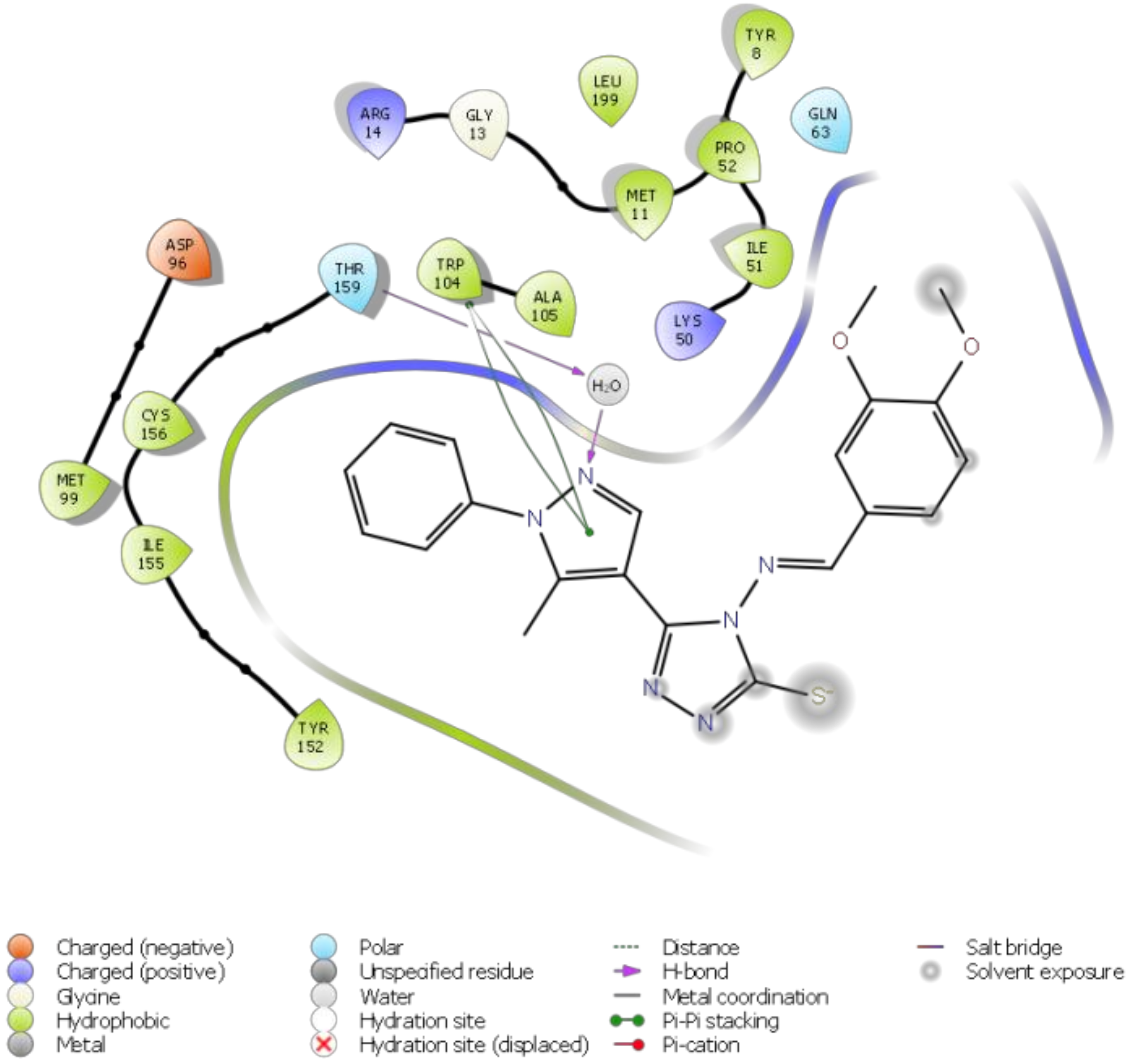
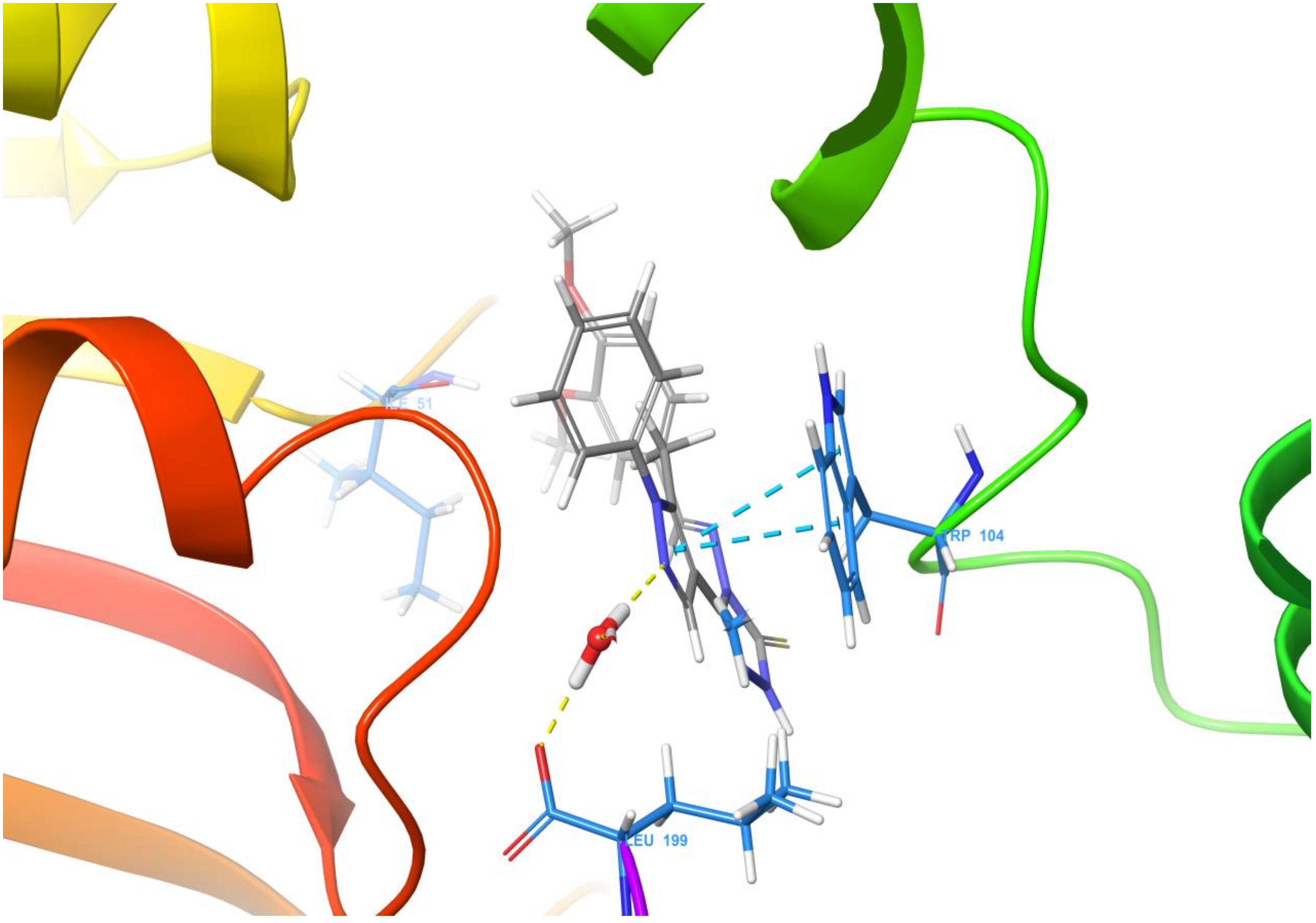
3. Molecular Docking
4. Discussion
5. Materials and Methods
4-[(3,4-Dimethoxybenzylidene)amino]-5-(5-methyl-1-phenyl-1H-pyrazol-4-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione (2)
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sharma, P.K. A review: Antimicrobial agents based on nitrogen and sulfur-containing heterocycles. Asian. J. Pharm. Clin. Res. 2017, 10, 47–49. [Google Scholar] [CrossRef]
- Bandgar, B.P.; Patil, S.A.; Gacche, R.N.; Korbad, B.L.; Hote, B.S.; Kinkar, S.N.; Jalde, S.S. Synthesis and biological evaluation of nitrogen-containing chalcones as possible anti-inflammatory and antioxidant agents. Bioorg. Med. Chem. Lett. 2010, 20, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Ruddarraju, R.R.; Murugulla, A.C.; Kotla, R.; Tirumalasetty, M.C.B.; Wudayagiri, R.; Donthabakthuni, S.; Maroju, R. Design, synthesis, anticancer activity and docking studies of theophylline containing 1,2,3-triazoles with variant amide derivatives. Med. Chem.Commun. 2016, 8, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Khanage, S.G.; Raju, A.; Mohite, P.B.; Pandhare, R.B. Analgesic Activity of Some 1,2,4-triazole Heterocycles Clubbed with Pyrazole, Tetrazole, Isoxazole and Pyrimidine. Adv. Pharm. Bull. 2013, 3, 13–18. [Google Scholar]
- Fernandes, G.F.S.; Chin, C.M.; Santos, J.L. Advances in Drug Discovery of New Antitubercular Multidrug-Resistant Compounds. Pharmaceuticals 2017, 10, 51. [Google Scholar] [CrossRef] [PubMed]
- Shalini, K.; Kumar, N.; Drabu, S.; Sharma, P.K. Advances in synthetic approach to and antifungal activity of triazoles. Beilstein J. Org. Chem. 2011, 7, 668–677. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Chen, Q.; Yang, G. A review on recent developments of indole-containing antiviral agents. Eur. J. Med. Chem. 2015, 89, 421–441. [Google Scholar] [CrossRef] [PubMed]
- Bhat, K.S.; Poojary, B.; Prasad, D.J.; Naik, P.; Holla, B.S. Synthesis and antitumor activity studies of some new fused 1,2,4-triazole derivatives carrying 2,4-dichloro-5-fluorophenyl moiety. Eur. J. Med. Chem. 2009, 44, 5066–5070. [Google Scholar] [CrossRef] [PubMed]
- Karegoudar, P.; Prasad, D.J.; Ashok, M.; Mahalinga, M.; Poojary, P.; Holla, B.S. Synthesis, antimicrobial and anti-inflammatory activities of some 1,2,4-triazolo[3,4-b][1,3,4]thiadiazoles and 1,2,4-triazolo[3,4-b][1,3,4]thiadiazines bearing trichlorophenyl moiety. Eur. J. Med. Chem. 2008, 43, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Hohwy, M.; Spadola, L.; Lundquist, B.; Hawtin, P.; Dahmen, J.; Groth-Clausen, I.; Nilsson, E.; Persdotter, S.; Wachenfeldt, K.V.; Folmer, R.H.A.; et al. Novel Prostaglandin D Synthase Inhibitors Generated by Fragment-Based Drug Design. J. Med. Chem. 2008, 51, 2178–2186. [Google Scholar] [CrossRef] [PubMed]
- Viveka, S.; Viveka, S.; Dinesha; Shama, P.; Nagaraja, G.K.; Deepa, N.; Sreenivasa, M.Y. Design, synthesis, and pharmacological studies of some new Mannich bases and S-alkylated analogs of pyrazole integrated 1,3,4-oxadiazole. Res. Chem. Intermed. 2016, 42, 2597–2617. [Google Scholar] [CrossRef]
- Farghaly, A.; Clercq, E.D.; El-Kashef, H. Synthesis and antiviral activity of novel[1,2,4]triazolo[3,4-b][1,3,4]thiadiazoles, [1,2,4]triazolo[3,4-b] [1,3,4]thiadiazines and [1,2,4]triazolo[3,4-b][1,3,4] thiadiazepines. Arkivoc 2006, 137–151. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nayak, S.G.; Poojary, B. 4-[(3,4-Dimethoxybenzylidene)amino]-5-(5-methyl-1-phenyl-1H-pyrazol-4-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione. Molbank 2019, 2019, M1055. https://doi.org/10.3390/M1055
Nayak SG, Poojary B. 4-[(3,4-Dimethoxybenzylidene)amino]-5-(5-methyl-1-phenyl-1H-pyrazol-4-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione. Molbank. 2019; 2019(1):M1055. https://doi.org/10.3390/M1055
Chicago/Turabian StyleNayak, Soukhyarani Gopal, and Boja Poojary. 2019. "4-[(3,4-Dimethoxybenzylidene)amino]-5-(5-methyl-1-phenyl-1H-pyrazol-4-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione" Molbank 2019, no. 1: M1055. https://doi.org/10.3390/M1055
APA StyleNayak, S. G., & Poojary, B. (2019). 4-[(3,4-Dimethoxybenzylidene)amino]-5-(5-methyl-1-phenyl-1H-pyrazol-4-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione. Molbank, 2019(1), M1055. https://doi.org/10.3390/M1055




