1. Introduction
Quercetin, 2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4
H-chromen-4-one, is one of the secondary metabolites of plants, representative of the flavonoid family [
1]. It has several biological activities, including antioxidant, anti-proliferative, anti-diabetic, vasodilating, anti-inflammatory and others. These health-promoting properties are usually limited by its fast metabolism, which affect often the critical points of the molecule’s bioactivity (i.e., OH groups) [
2]. Several chemical modifications have been proposed to increase bioavailability and limit metabolism. In this context, it is well known how the glycosylation and mostly acylation reactions are used to improve flavonoids properties [
3]. In recent years regioselective syntheses emerged as very important chemical processes; various researchers have proposed enzymatic reactions as very selective and high yielding methods, although often in the case of flavonoids glycosides [
4]. Recently, two studies [
5,
6] reported the enzymatic synthesis of cinnamate and oleate esters of quercetin, demonstrating how in particular reaction’s conditions it is possible to obtain a mixture of three main products, 4′-monoester, 3′,4′-diester and 7,3′,4′-triester. In this short paper, an ecofriendly, bio-catalytic process has been proposed to synthesize the C-3-oleate of quercetin in only one step. Using pancreatic porcine lipase (PPL), quercetin-3-oleate was obtained in very high yield, as unique product, and recycling of the enzyme was achieved five times. This study demonstrates the regioselectivity of PPL towards the C3-hydroxyl group.
2. Results
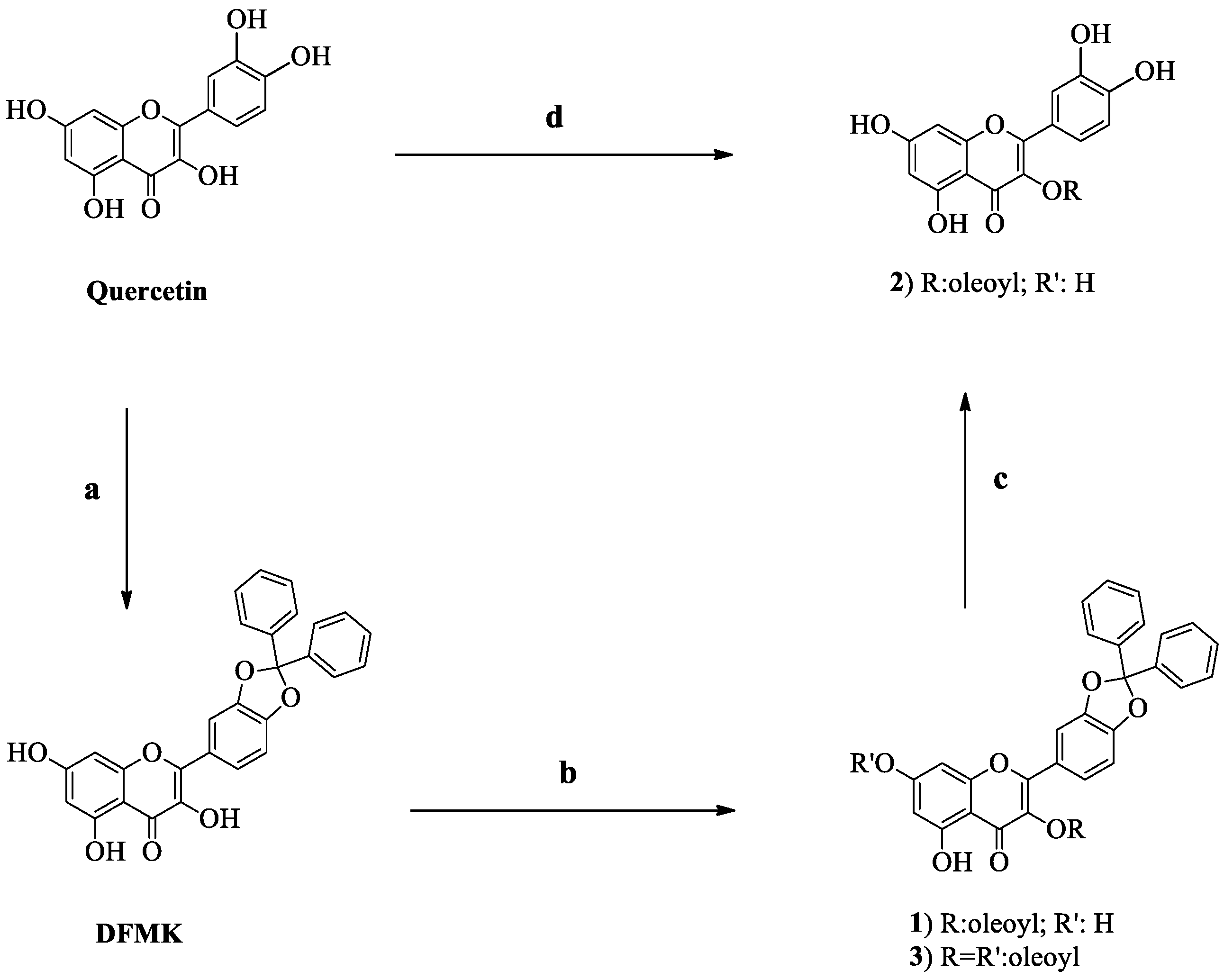
In a previous paper, a semisynthetic derivative of quercetin, 2-(2,2-diphenylbenzo[
d][1,3] dioxol-5-yl)-5,7-dihydroxy-4-oxo-4
H-chromen-3-yl oleate, was synthesized according to Steglich conditions and validated as a potential insulin secretagogue agent G-protein-coupled receptor 40 ligand [
3]. In this context, 2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4-oxo-4
H-chromen-3-yl oleate, quercetin-3-oleate, was synthesized avoiding the preventive catechol protection but considering several literature observations [
7], considering that
Candida antartica Lipase B
®—the most used acylating enzyme—was not able to produce esters of quercetin [
8]. In a typical optimized experiment, one equivalent of quercetin, and one equivalent of oleic acid were added to PPL and acetone. Temperature was maintained at 37 °C, with an agitation rate of 130 rpm. The desired product was obtained in 88% isolated yield after 48 h incubation. (
Scheme 1) Compound
2 was fully characterized by IR,
1H-NMR and
13C-spectroscopies and elemental analysis.
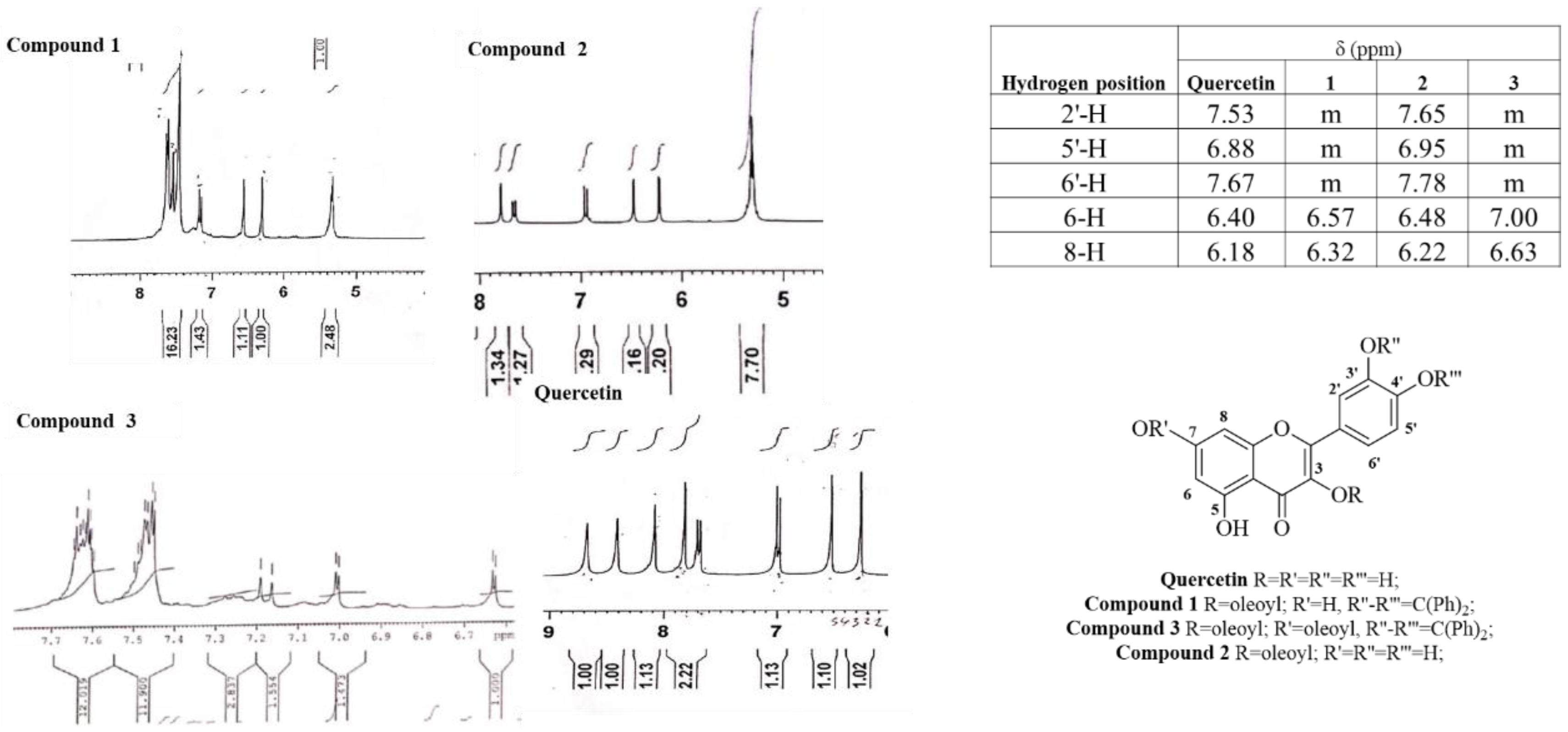
The exact acylation site was determined by comparing the chemical shifts between pure quercetin and its acylated analogues just published (Compounds
1 and
3 in
Figure 1) [
3]. The chemicals shifts were found to be slightly shifted to downfield or upfield values [
6]. For examples, substitutions at 3 and 7 hydroxyls to form Compound
3 moved the chemical shifts of the neighbouring protons (6 and 8) to higher field. The presence of diphenyl methyl ketal backbone moved the chemical shifts of the 2′ and 5′ protons to lower and higher field, respectively. The mono-substitution of quercetin-3-oleate did not move the chemical shifts of 6,8 protons in a significant manner, confirming the 7-OH free and at the same time, 2′ and 5′ protons of catechol did not move. To confirm the 3-acylated position, Compound
2 was also obtained by cleavage of the diphenylmethylketal in Compound
1. The resulted compound showed an
1H-NMR spectrum that matches perfectly with the spectrum of final compound obtained by lipase catalysis. Based on literature data, it is not convenient to consider significant the presence of signals related to OH phenol groups, known as mobile hydrogens [
9] and visible only in particular experimental conditions. In fact, in our qNMR (quantitative NMR) analysis (with maleic acid as trace NMR) they became visible. Overall, the most useful practice is the attribution based exclusively on the chemical shifts of the critical hydrogens like C-8, C-2′, C-5′.
3. Materials and Methods
The lipase from porcine pancreas (PPL), Type II, 100–500 units/mg protein (using olive oil (30 min incubation)), 30–90 units/mg protein (using triacetin) and Quercetin dehydrate ≥98% (HPLC), powder and Oleic acid ≥99% (GC) were purchased from Merck (Darmstad, Germania). Acetone was purchased from Levanchimica Srl (Bari, Italy). The selective C-3 position of acylation was determined by NMR. 1H and 13C-NMR spectra were recorded on a Bruker Advance 300 spectrometer (Bruker, Milano, Italy) at 300 and 75 MHz, respectively. Acetone-d6, and DMSO-d6 were used as solvents. IR spectrum was recorded on a PerkinElmer machine 10.4.00 (PerkinElmer, Milano, Italy).
Synthetic procedure for the preparation of quercetin-3-oleate [2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4-oxo-4H-chromen-3-yl-octadec-9-enoate]: In a round-bottom flask, to a solution of quercetin (100 mg, 0.330 mmol) and oleic acid (93.46 mg, 0.330 mmol) in 30 mL of acetone were added 260 mg of PPL. Temperature was maintained at 37 °C, with an agitation rate of 130 rpm. After 48 h, the reaction was monitored under UV light, after elution of a thin-layer chromatography (TLC) on silica gel 60 F254 plates, using a solvent mixture system of chloroform:diethyl ether (50:50). The crude mixture was then filtered, washed with a cold solution of NaHCO3 and extracted with diethyl ether, to afford the corresponding oleate. Compound 2: Quercetin-3-oleate [2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4-oxo-4H-chromen-3-yl-octadec-9-enoate] was obtained in 88% yield as a yellow resin; 1H-NMR (300 MHz, DMSO-d6): δ 7.78 (d, 1H, J = 1.9 Hz), 7.64 (dd, 1H, J = 1.7 Hz, 8.5 Hz), 6.98 (d, 1H, J = 8.5 Hz), 6.50 (d, 1H, J = 1.3 Hz), 6.28 (d, 1H, J = 1.4 Hz), 5.42–5.38 (m, 2H), 2.29 (t, 2H, J = 7.3 Hz), 2.05 (m, 4H), 1.57 (m, 2H), 1.34–1.24 (m, 20H), 0.94 (m, 3H). 13C-NMR (75 MHz, DMSO-d6): δ 176.2, 174.8, 164.3, 161.1, 156.5, 148.1, 147.2, 145.4, 136.1, 130.0, 129.9, 122.4, 120.3, 115.9, 115.4, 103.4, 98.5, 93.7, 34.0, 31.7, 29.5, 29.4, 29.2, 29.1, 29.0, 28.9, 28.9, 26.9, 24.9, 22.5, 14.4, 14.2. Elemental Analysis for C33H42O8 found C, 69.81; H, 7.60. IR: ν = 3275.59 (m), 2921.96 (s), 2852.57 (s), 1708.89 (s), 1462.81 (m), 1349.03 (s), 1301.90 (m) cm−1.