1-(4-Hexylbenzoyl)-3-methylthiourea
Abstract
1. Introduction
2. Results
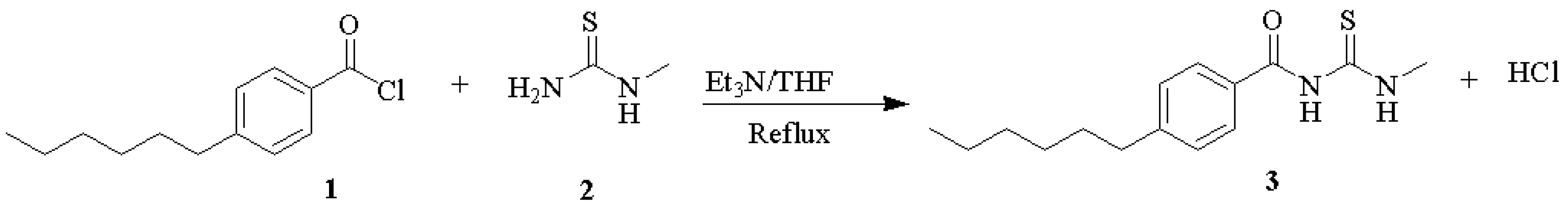
2.1. 1-(4-Hexylbenzoyl)-3-methylthiourea
2.2. Cytotoxic Activity
3. Materials and Methods
3.1. 1-(4-Hexylbenzoyl)-3-Methylthiourea
3.2. Cytotoxic Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lan, J.; Huang, L.; Lou, H.; Chen, C.; Liu, T. Design, and synthesis of novel C14-urea-tetrandrine derivatives with potent anti-cancer activity. Eur. J. Med. Chem. 2018, 143, 1968–1980. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.; Zhong, Y.; Mai, X.; Liao, J.; Liu, C.; Feng, H. Synthesis and Anticancer Evaluation of Benzyloxyurea Derivatives. Chem. Pharm. Bull. 2014, 62, 898–905. [Google Scholar] [CrossRef] [PubMed]
- North, E.J.; Scherman, M.S.; Bruhn, D.F.; Scarborough, J.S.; Maddox, M.M.; Jones, V. Design, synthesis and anti-tuberculosis activity of 1-adamantyl-3-heteroaryl ureas with improved in vitro pharmacokinetic properties. Bioorg. Med. Chem. 2013, 19, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.; North, E.J.; Hurdle, J.G.; Morisseau, C.; Scarborough, J.S.; Sun, D.; Korduláková, J.; Scherman, M.S.; Jones, V.; Grzegorzewicz, A.; et al. The Structure Activity Relationship of Urea Derivatives as Anti-Tuberculosis Agents. Bioorg. Med. Chem. 2011, 19, 5585–5595. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, B.D.; Skinner-Adams, T.S.; Andrews, K.T.; Coster, M.J.; Edstein, M.D.; Mackenzie, D.; Charman, S.A.; Koltun, M.; Blundell, S.; Campbell, A.; et al. Biomolecular Chemistry Synthesis and antimalarial evaluation of amide and natural product scaffold. Org. Biomol. Chem. 2015, 13, 1558–1570. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, J.N.; León, C.; Rodrigues, J.; Gamboa de Domínguez, N.; Gut, J.; Rosenthal, P.J. Synthesis and Evaluation of New Antimalarial Phenylurenyl Chalcone Derivatives. J. Med. Chem. 2005, 48, 3654–3658. [Google Scholar] [CrossRef] [PubMed]
- Sartoril, E.; Camyl, F.; Teulonl, J.M.; Cambordez, F.; Meignen, J.; Hertzz, F. Synthesis and analgesic activities of urea derivatives of a-amino-N-pyridyl benzene propanamide. Eur. J. Med. Chem. 1994, 29, 431–4399. [Google Scholar] [CrossRef]
- Manjula, S.N.; Noolvi, N.M.; Parihar, K.V.; Reddy, S.A.M.; Ramani, V.; Gadad, A.K. European Journal of Medicinal Chemistry Synthesis and antitumor activity of optically active thiourea and their 2-aminobenzothiazole derivatives: A novel class of anticancer agents. Eur. J. Med. Chem. 2009, 44, 2923–2929. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Hu, Y.; Yang, Y.S.; Zhang, F.; Zhang, Y.B.; Wang, X.L.; Tang, J.F.; Zhong, W.Q.; Zhu, H.L. Design, modification and 3D QSAR studies of novel naphthalin-containing pyrazoline derivatives with/without thiourea skeleton as anticancer agents. Bioorg. Med. Chem. 2013, 21, 1050–1063. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Chen, J.; He, Z.; Sun, W.; Xu, W. Design, synthesis and biological activities of thiourea containing sorafenib analogs as antitumor agents. Bioorg. Med. Chem. 2012, 20, 2923–2929. [Google Scholar] [CrossRef] [PubMed]
- Karakuş, S.; Küçükgüzel, Ş.G.; Küçükgüzel, İ.; De Clercq, E.; Pannecouque, C.; Andrei, G.; Snoeck, R.; Sahin, F.; Bayrak, Ö.F. Synthesis, antiviral and anticancer activity of some novel thioureas derived from N-(4-nitro-2-phenoxyphenyl)-methanesulfonamide. Eur. J. Med. Chem. 2009, 44, 3591–3595. [Google Scholar] [CrossRef] [PubMed]
- Pingaew, R.; Prachayasittikul, V.; Anuwongcharoen, N.; Prachayasittikul, S.; Ruchirawat, S.; Prachayasittikul, V. Synthesis and molecular docking of N, N′-disubstituted thiourea derivatives as novel aromatase inhibitors. Bioorg. Chem. 2018, 79, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Sett, P.P.; Ranken, R.; Robinson, D.E.; Osgood, S.A.; Risen, L.M.; Rodgers, E.L.; Migawa, M.T.; Jefferson, E.A.; Swayze, E.E. Aryl urea analogs with broad-spectrum antibacterial activity. Bioorg. Med. Chem. Lett. 2004, 14, 5569–5572. [Google Scholar] [CrossRef] [PubMed]
- Upadhayaya, R.S.; Kulkarni, G.M.; Vasireddy, N.R.; Vandavasi, J.K.; Dixit, S.S.; Sharma, V.; Chattapadhayaya, J. Design, synthesis and biological evaluation of novel triazole, urea and thiourea derivatives of quinoline against Mycobacterium tuberculosis. Bioorg. Med. Chem. 2009, 17, 4681–4692. [Google Scholar] [CrossRef] [PubMed]
- Vega-Pérez, J.M.; Periñán, I.; Argandoña, M.; Vega-Holm, M.; Palo-Nieto, C.; Burgos-Morón, E.; López-Lázaro, M.; Vargas, C.; Nieto, J.J.; Iglesias-Guerra, F. Isoprenyl-thiourea and urea derivatives as new farnesyl diphosphate analogs: Synthesis and in vitro antimicrobial and cytotoxic activities. Eur. J. Med. Chem. 2012, 58, 591–612. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Singh, N.; Saleem, K. Synthesis, characterization and in vitro antibacterial activity of thiourea and urea derivatives of stereoids. Eur. J. Med. Chem. 2008, 43, 2272–2277. [Google Scholar] [CrossRef] [PubMed]
- Binzet, G.; Gumus, I.; Dogen, A.; Flörke, U.; Kulcu, N.; Arslan, H. Nickel(II) and Copper(II) Complexes of N,N-Dialkyl-N′-3-Chlorobenzoylthiourea: Synthesis, Characterization, Crystal Structures, Hirshfeld Surfaces and Antimicrobial Activity. J. Mol. Struct. 2018, 1161, 519–529. [Google Scholar] [CrossRef]
- Bielenica, A.; Drzewiecka-Antonik, A.; Rejmak, P.; Stefańska, J.; Koliński, M.; Kmiecik, S.; Lesyng, B.; Włodarczyk, M.; Pietrzyk, P.; Struga, M. Synthesis, Structural and Antimicrobial Studies of Type II Topoisomerase-Targeted Copper(II) Complexes of 1,3-Disubstituted Thiourea Ligands. J. Inorg. Biochem. 2018, 182, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Burmistrov, V.; Morisseau, C.; Pitushkin, D.; Karlov, D.; Fayzullin, R.R.; Butov, G.M.; Hammock, B.D. Adamantyl Thioureas as Soluble Epoxide Hydrolase Inhibitors. Bioorg. Med. Chem. Lett. 2018, 28, 2302–2313. [Google Scholar] [CrossRef] [PubMed]
- Burgeson, J.R.; Moore, A.L.; Boutilier, J.K.; Cerruti, N.R.; Gharaibeh, D.N.; Lovejoy, C.E.; Amberg, S.M.; Hruby, D.E.; Tyavanagimatt, S.R.; Allen, R.D.; et al. Analysis of a series of acylthiourea derivatives possessing broad-spectrum antiviral. Bioorg. Med. Chem Lett. 2012, 22, 4263–4272. [Google Scholar] [CrossRef] [PubMed]
- Ruswanto; Miftah, A.M.; Tjahjono, D.H.; Siswandono. Synthesis and in vitro Cytotoxicity of 1-Benzoyl-3-methyl thiourea Derivatives. Procedia Chem. 2015, 17, 157–161. [Google Scholar]
- Gumus, I.; Solmaz, U.; Binzet, G.; Keskin, E.; Arslan, B.; Arslan, H. Hirshfeld Surface Analyses and Crystal Structures of Supramolecular Self-Assembly Thiourea Derivatives Directed by Non-Covalent Interactions. J. Mol. Struct. 2018, 1157, 78–88. [Google Scholar] [CrossRef]
- Ansari, M.F.; Idrees, D.; Hassan, M.I.; Ahmad, K.; Avecilla, F.; Azam, A. Design, synthesis and biological evaluation of novel pyridine-thiazolidinone derivatives as anticancer agents: Targeting human carbonic anhydrase IX. Eur. J. Med. Chem. 2018, 144, 544–556. [Google Scholar] [CrossRef] [PubMed]
| Compound | IC50 (μM) | |||
|---|---|---|---|---|
| T47D | MCF-7 | WiDr | HeLa | |
| 1-(4-Hexylbenzoyl)-3-methylthiourea | 179 | 390 | 433 | 412 |
| Hydroxyurea (HU) | 2724 | 2829 | 1803 | 5632 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruswanto, R.; Mardianingrum, R.; Lestari, T.; Nofianti, T.; Siswandono, S. 1-(4-Hexylbenzoyl)-3-methylthiourea. Molbank 2018, 2018, M1005. https://doi.org/10.3390/M1005
Ruswanto R, Mardianingrum R, Lestari T, Nofianti T, Siswandono S. 1-(4-Hexylbenzoyl)-3-methylthiourea. Molbank. 2018; 2018(3):M1005. https://doi.org/10.3390/M1005
Chicago/Turabian StyleRuswanto, Ruswanto, Richa Mardianingrum, Tresna Lestari, Tita Nofianti, and Siswandono Siswandono. 2018. "1-(4-Hexylbenzoyl)-3-methylthiourea" Molbank 2018, no. 3: M1005. https://doi.org/10.3390/M1005
APA StyleRuswanto, R., Mardianingrum, R., Lestari, T., Nofianti, T., & Siswandono, S. (2018). 1-(4-Hexylbenzoyl)-3-methylthiourea. Molbank, 2018(3), M1005. https://doi.org/10.3390/M1005





