Ethyl({[acryloyl(furan-2-ylmethyl)amino]acetyl}amino)acetate
Abstract
:1. Introduction
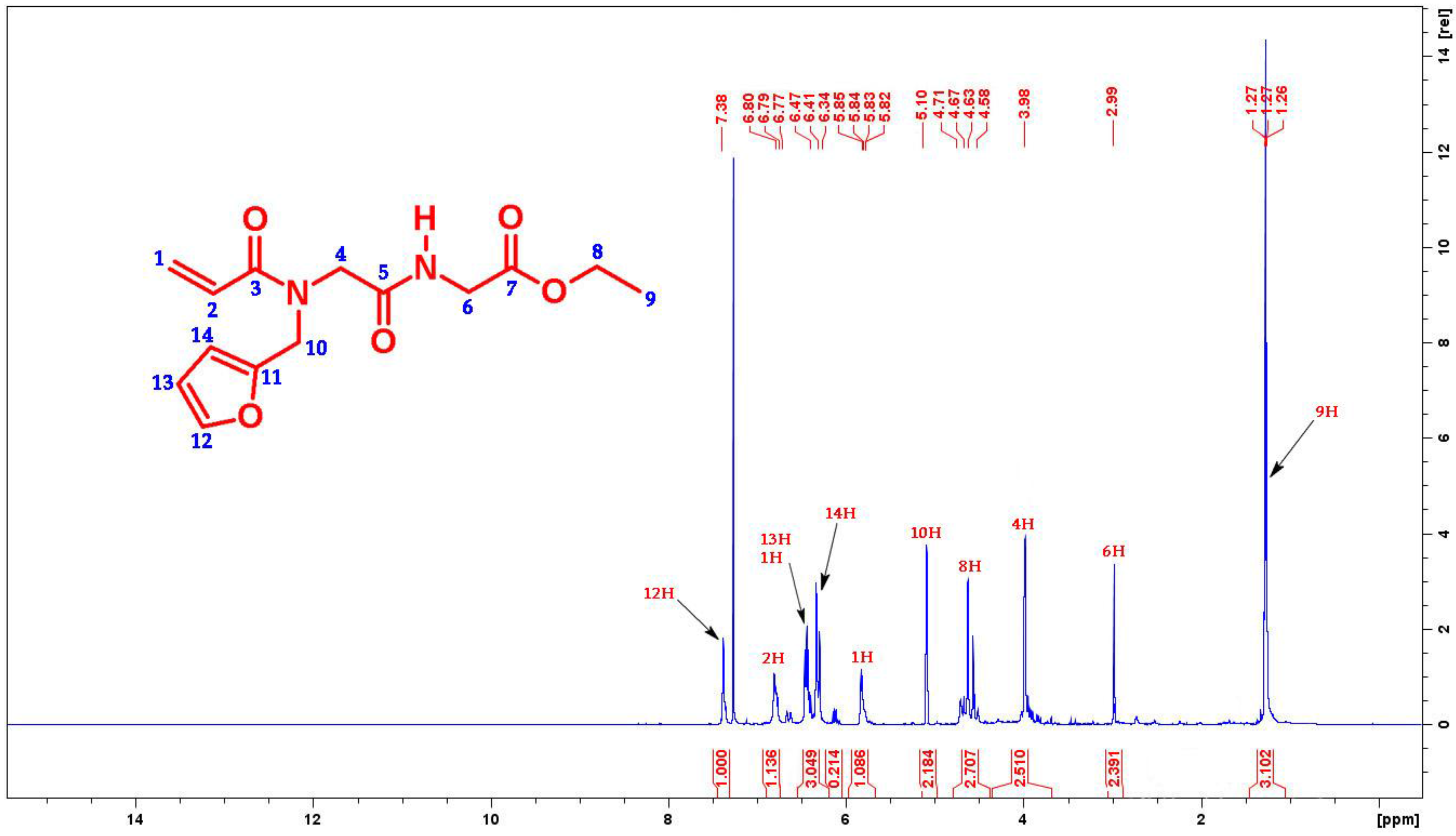
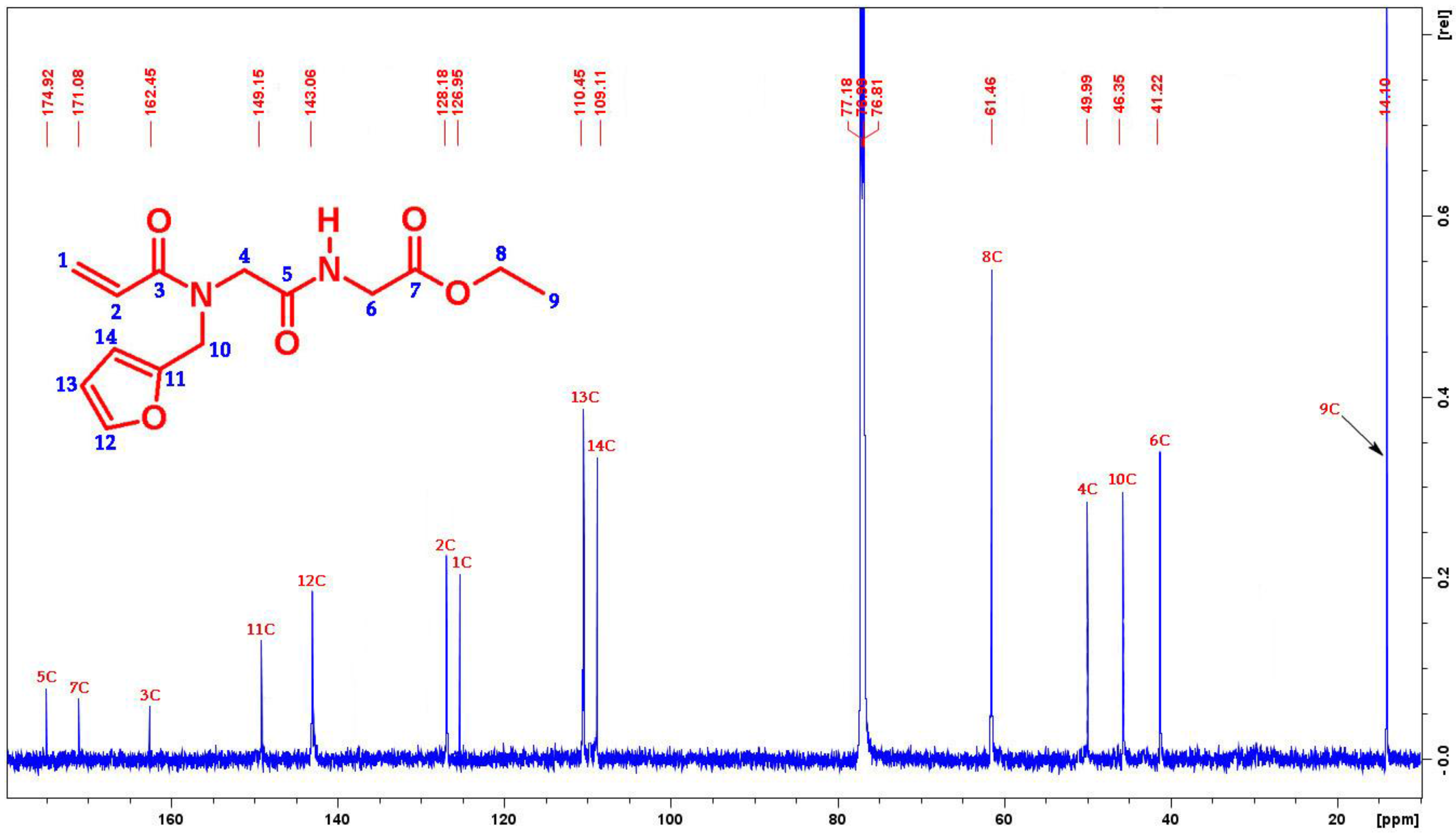
2. Results and Discussion
3. Experimental Section
3.1. Materials
3.2. Instrumentaion
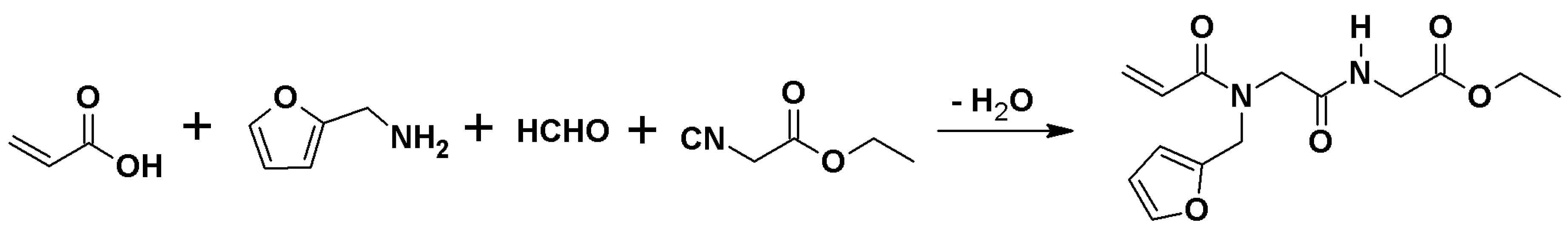
3.3. Synthesis of Ethyl({[acryloyl(furan-2-ylmethyl)amino]acetyl}amino)acetate
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gutteridge, A.; Thornton, J.M. Understanding nature’s catalytic toolkit. Trends Biochem. Sci. 2005, 30, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Reddington, S.C.; Howarth, M. Secrets of a covalent interaction for biomaterials and biotechnology: SpyTag and SpyCatcher. Curr. Opin. Chem. Biol. 2015, 29, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Simon, R.J.; Kania, R.S.; Zuckermann, R.N.; Huebner, V.D.; Jewell, D.A.; Banville, S.; Ng, S.; Wang, L.; Rosenberg, S.; Marlowe, C.K.; et al. Peptoids: A modular approach to drug discovery. Proc. Natl. Acad. Sci. USA 1992, 89, 9367–9371. [Google Scholar] [CrossRef] [PubMed]
- Zuckermann, R.N. Peptoid Origins. Biopolymers (PeptSci) 2011, 96, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Zuckermann, R.N.; Kodadek, T. Peptoids as potential therapeutics. Curr. Opin. Mol. Ther. 2009, 11, 299–307. [Google Scholar] [PubMed]
- Fosgerau, K.; Hoffmann, T. Peptide therapeutics: current status and future directions. Drug Discov. Today 2015, 20, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Mangunuru, H.P.R.; Yang, H.; Wang, G. Synthesis of peptoid based small molecular gelators by a multiple component reaction. Chem. Commun. 2013, 49, 4489–4491. [Google Scholar] [CrossRef] [PubMed]
- Mitra, R.N.; Das, D.; Roy, S.; Das, P.K. Structure and Properties of Low Molecular Weight Amphiphilic Peptide Hydrogelators. J. Phys. Chem. B 2007, 111, 14107–14113. [Google Scholar] [CrossRef] [PubMed]
- Worthington, P.; Pochan, D.J.; Langhans, S.A. Peptide Hydrogels—Versatile Matrices for 3D Cell Culture in Cancer Medicine. Front. Oncol. 2015, 5, 92. [Google Scholar] [CrossRef] [PubMed]
- Tibbitt, M.W.; Anseth, K.S. Hydrogels as Extracellular Matrix Mimics for 3D Cell Culture. Biotechnol. Bioeng. 2009, 103, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Biswas, G.; Moon, H.J.; Boratyński, P.; Jeong, B.; Kwon, Y.-U. Structural sensitivity of peptoid-based low molecular mass organogelator. Mater. Des. 2016, 108, 659–665. [Google Scholar] [CrossRef]
- Nigam, M.; Rush, B.; Patel, J.; Castillo, R.; Dhar, P. Aza-Michael Reaction for an Undergraduate Organic Chemistry Laboratory. J. Chem. Educ. 2016, 93, 753–756. [Google Scholar] [CrossRef]
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganesh, S.D.; Saha, N.; Zuckermann, R.N.; Sáha, P. Ethyl({[acryloyl(furan-2-ylmethyl)amino]acetyl}amino)acetate. Molbank 2017, 2017, M925. https://doi.org/10.3390/M925
Ganesh SD, Saha N, Zuckermann RN, Sáha P. Ethyl({[acryloyl(furan-2-ylmethyl)amino]acetyl}amino)acetate. Molbank. 2017; 2017(1):M925. https://doi.org/10.3390/M925
Chicago/Turabian StyleGanesh, Shimoga D., Nabanita Saha, Ronald N. Zuckermann, and Petr Sáha. 2017. "Ethyl({[acryloyl(furan-2-ylmethyl)amino]acetyl}amino)acetate" Molbank 2017, no. 1: M925. https://doi.org/10.3390/M925
APA StyleGanesh, S. D., Saha, N., Zuckermann, R. N., & Sáha, P. (2017). Ethyl({[acryloyl(furan-2-ylmethyl)amino]acetyl}amino)acetate. Molbank, 2017(1), M925. https://doi.org/10.3390/M925




