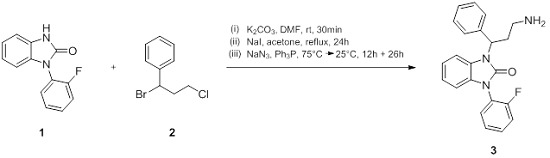
1-(3-Amino-1-phenylpropyl)-3-(2-fluorophenyl)-1,3-dihydro-2H-benzimidazol-2-one
1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. 1-(3-Amino-1-phenylpropyl)-3-(2-fluorophenyl)-1,3-dihydro-2H-benzimidazol-2-one
- (i)
- To a solution of 1 (1.00 g, 4.38 mmol) in DMF (5 mL) was added K2CO3 (1.21 g, 8.76 mmol) and the resulting mixture was stirred for 30 min at 25 °C. After 30 min, 2 (1.53 g, 6.57 mmol) was added and stirring was continued overnight. Ethyl acetate (5 mL) and water (5 mL) were added to the mixture and the aqueous layer was extracted several times with ethyl acetate (10 mL). The combined organic layers were washed with brine, dried over MgSO4, and evaporated to dryness. Purification of the resulting product was carried out by column chromatography (silica gel 60) with petroleum ether/ethyl acetate 9:1 to afford an orange resin.
- (ii)
- The resulting orange resin (0.84 g, 2.20 mmol) was dissolved in acetone (7 mL) and NaI (0.66 g, 4.40 mmol) was added. The mixture was refluxed for 24 h, after which the formed precipitate was filtered and concentrated prior to purification via column chromatography (silica gel 60) with petroleum ether/ethyl acetate 9:1, to give the intermediate product as yellow crystals.
- (iii)
- The obtained product (0.15 g, 0.32 mmol) was dissolved in DMF (3 mL) and heated to 75 °C upon addition of NaN3 (0.05 g, 0.65 mmol). After 12 h the reaction was quenched with H2O (10 mL) and the resulting mixture was extracted with ethyl acetate (3 × 30 mL). The organic layer was washed with brine (3 × 10 mL), dried over Na2SO4, filtered, and concentrated under reduced pressure. The crude product was purified via column chromatography (silica gel 60, petroleum ether/ethyl acetate 8.5:1.5) and was introduced in the subsequent reaction step.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Author Contributions
Conflicts of Interest
References
- Sung, U.; Apparsundram, S.; Galli, A.; Kahlig, K.M.; Savchenko, V.; Schroeter, S.; Quick, M.W.; Blakely, R.D. A regulated interaction of syntaxin 1A with the antidepressant-sensitive norepinephrine transporter establishes catecholamine clearance capacity. J. Neurosci. 2003, 23, 1697–1709. [Google Scholar] [PubMed]
- Kim, C.H.; Hahn, M.K.; Joung, Y.; Anderson, S.L.; Steele, A.H.; Mazei-Robinson, M.S.; Gizer, I.; Teicher, M.H.; Cohen, B.M.; Robertson, D.; et al. A polymorphism in the norepinephrine transporter gene alters promoter activity and is associated with attention-deficit hyperactivity disorder. Proc. Natl. Acad. Sci. USA 2006, 103, 19164–19169. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M.K.; Robertson, D.; Blakely, R.D. A mutation in the human norepinephrine transporter gene (SLC6A2) associated with orthostatic intolerance disrupts surface expression of mutant and wild-type transporters. J. Neurosci. 2003, 23, 4470–4478. [Google Scholar] [PubMed]
- Mirbolooki, M.R.; Upadhyay, S.K.; Constantinescu, C.C.; Pan, M.L.; Mukherjee, J. Adrenergic pathway activation enhances brown adipose tissue metabolism: A [18F]FDG PET/CT study in mice. Nucl. Med. Biol. 2014, 41, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.L.; Fan, X.; Yeckel, C.W.; Weinzimmer, D.; Mulnix, T.; Gallezot, J.D.; Carson, R.E.; Sherwin, R.S.; Ding, Y.S. Ex vivo and in vivo Evaluation of the Norepinephrine Transporter Ligand [11C]MRB for Brown Adipose Tissue Imaging. Nucl. Med. Biol. 2012, 39, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Wadsak, W.; Mitterhauser, M. Basics and principles of radiopharmaceuticals for PET/CT. Eur. J. Radiol. 2010, 73, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Rami-Mark, C; Zhang, M.R.; Mitterhauser, M.; Lanzenberger, R.; Hacker, M.; Wadsak, W. [18F]FMeNER-D2: Reliable fully-automated synthesis for visualization of the norepinephrine transporter. Nucl. Med. Biol. 2013, 40, 1049–1054. [Google Scholar]
- Vanicek, T.; Spies, M.; Rami-Mark, C.; Savli, M.; Höflich, A.; Kranz, G.S.; Hahn, A.; Kutzelnigg, A.; Traub-Weidinger, T.; Mitterhauser, M.; et al. The norepinephrine transporter in attention-deficit/hyperactivity disorder investigated with positron emission tomography. JAMA Psychiatry 2014, 71, 1340–1349. [Google Scholar] [CrossRef] [PubMed]
- Mark, C.; Bornatowicz, B.; Mitterhauser, M.; Hendl, M.; Nics, L.; Haeusler, D.; Lanzenberger, R.; Berger, M.L.; Spreitzer, H.; Wadsak, W. Development and automation of a novel NET-PET tracer: [C-11]Me@APPI. Nucl. Med. Biol. 2013, 40, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Neudorfer, C.; Seddik, A.; Shanab, K.; Jurik, A.; Rami-Mark, C.; Holzer, W.; Ecker, G.; Mitterhauser, M.; Wadsak, W.; Spreitzer, H. Synthesis and in Silico Evaluation of Novel Compounds for PET-Based Investigations of the Norepinephrine Transporter. Molecules 2015, 20, 1712–1730. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neudorfer, C.; Eberherr, N.; Shanab, K.; Holzer, W.; Rami-Mark, C.; Mitterhauser, M.; Wadsak, W.; Spreitzer, H. 1-(3-Amino-1-phenylpropyl)-3-(2-fluorophenyl)-1,3-dihydro-2H-benzimidazol-2-one. Molbank 2015, 2015, M867. https://doi.org/10.3390/M867
Neudorfer C, Eberherr N, Shanab K, Holzer W, Rami-Mark C, Mitterhauser M, Wadsak W, Spreitzer H. 1-(3-Amino-1-phenylpropyl)-3-(2-fluorophenyl)-1,3-dihydro-2H-benzimidazol-2-one. Molbank. 2015; 2015(3):M867. https://doi.org/10.3390/M867
Chicago/Turabian StyleNeudorfer, Catharina, Nadine Eberherr, Karem Shanab, Wolfgang Holzer, Christina Rami-Mark, Markus Mitterhauser, Wolfgang Wadsak, and Helmut Spreitzer. 2015. "1-(3-Amino-1-phenylpropyl)-3-(2-fluorophenyl)-1,3-dihydro-2H-benzimidazol-2-one" Molbank 2015, no. 3: M867. https://doi.org/10.3390/M867
APA StyleNeudorfer, C., Eberherr, N., Shanab, K., Holzer, W., Rami-Mark, C., Mitterhauser, M., Wadsak, W., & Spreitzer, H. (2015). 1-(3-Amino-1-phenylpropyl)-3-(2-fluorophenyl)-1,3-dihydro-2H-benzimidazol-2-one. Molbank, 2015(3), M867. https://doi.org/10.3390/M867