1,1′-{1,4-Phenylene bis[3-(6-chloro-2-methyl-4-phenylquinolin-3-yl)-4,5-dihydro-1H-pyrazole-5,1-diyl]}dibutan-1-one
Abstract
:1. Introduction
2. Experimental Section
2.1. General Methods
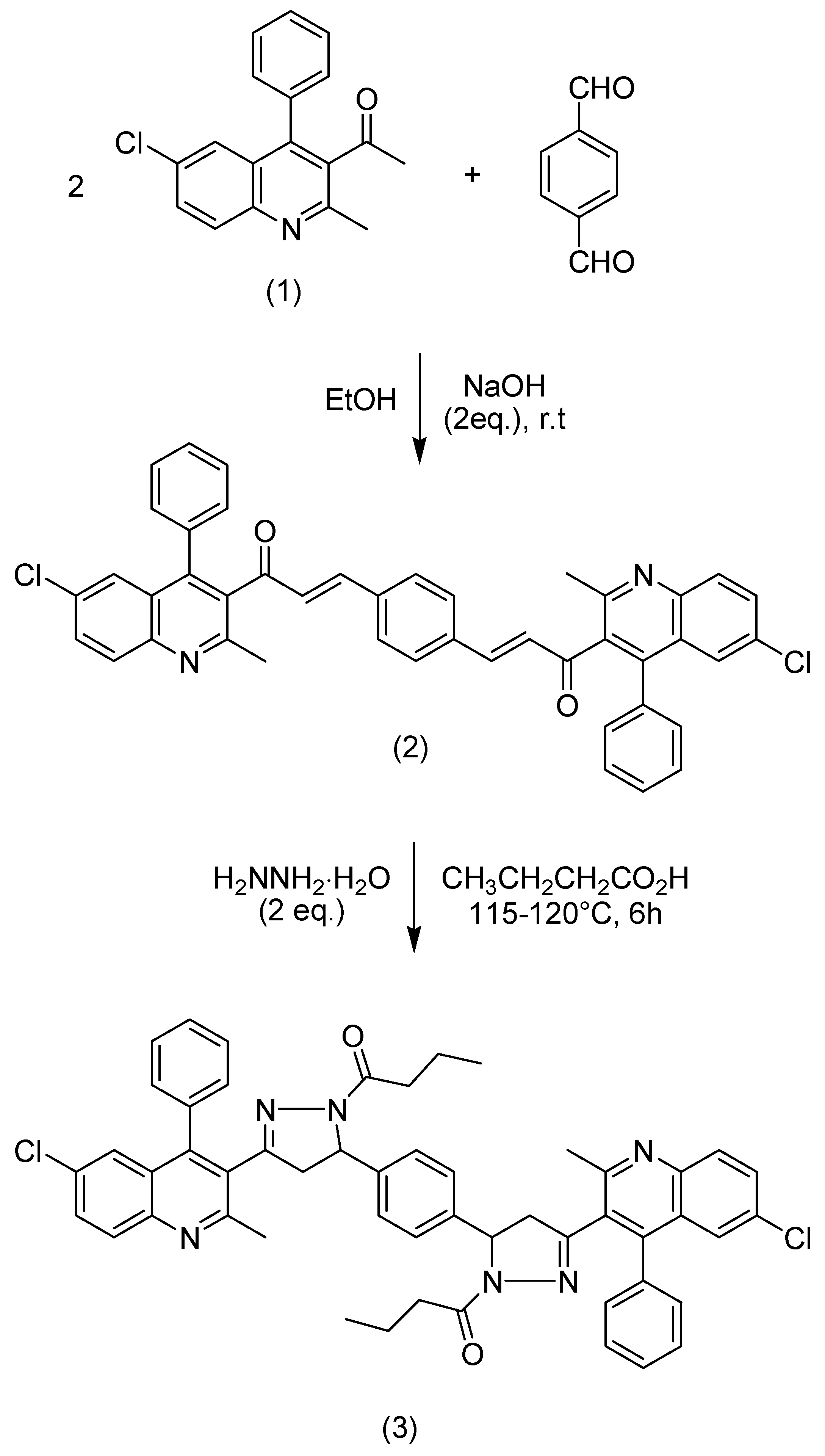
2.2. Synthesis of 1,1′-{1,4-phenylene bis[3-(6-chloro-2-methyl-4-phenylquinolin-3-yl)-4,5-dihydro-1H-pyrazole-5,1-diyl]}dibutan-1-one (3)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Acknowledgments
Author Contributions
Conflicts of Interest
References
- Levy, S.; Azoulay, S. Stories About the Origin of Quinquina and Quinidine. J. Cardiovas. Electrophysiol. 1994, 5, 635–636. [Google Scholar] [CrossRef]
- Roma, G.; Braccio, M.D.; Grossi, G.; Mattioli, F.; Ghia, H. 1,8-Naphthyridines IV. 9-Substituted N,N-dialkyl-5-(alkylamino or cycloalkylamino) [1,2,4]triazolo[4,3-a][1,8]naphthyridine-6-carboxamides, new compounds with anti-aggressive and potent anti-inflammatory activities. Eur. J. Med. Chem. 2000, 35, 1021–1035. [Google Scholar] [CrossRef]
- Phan, L.T.; Jian, T.; Chen, Z.; Qiu, Y.L.; Wang, Z.; Beach, T.; Polemeropoulos, A.; Or, Y.S. Synthesis and Antibacterial Activity of a Novel Class of 4′-Substituted 16-Membered Ring Macrolides Derived from Tylosin. J. Med. Chem. 2004, 47, 2965–2968. [Google Scholar] [CrossRef] [PubMed]
- Vargas, L.Y.; Castelli, M.V.; Kouznetsov, V.V.; Urbina, J.M.; Lopez, S.N.; Sortino, M.; Enriz, R.D.; Ribas, J.C.; Zacchino, S. In vitro antifungal activity of new series of homoallylamines and related compounds with inhibitory properties of the synthesis of fungal cell wall polymers. Bioorg. Med. Chem. 2003, 11, 1531–1550. [Google Scholar] [CrossRef]
- Dassonneville, L.; Lansiaux, A.; Wattelet, A.; Wattez, N.; Mahieu, C.; Van Miert, S.; Pieters, L.; Bailly, C. Cytotoxicity and cell cycle effects of the plant alkaloids cryptolepine and neocryptolepine: Relation to drug-induced apoptosis. Eur. J. Pharmacol. 2000, 409, 9–18. [Google Scholar] [CrossRef]
- Levai, A. Synthesis of pyrazolines by the reactions of α,β-enones with diazomethane and hydrazines. Chem. Heterocycl. Compd. 1997, 33, 647–659. [Google Scholar] [CrossRef]
- Özdemir, A.; Zitouni, T.G.; Kaplancıklı, Z.A.; Revial, G.; Güven, K. Synthesis and antimicrobial activity of 1-(4-aryl-2-thiazolyl)-3-(2-thienyl)-5-aryl-2-pyrazoline derivatives. Eur. J. Med. Chem. 2007, 42, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Shaharyar, M.; Siddiqui, A.A.; Ali, M.A.; Sriram, D.; Yogeeswari, P. Synthesis and in vitro antimycobacterial activity of N1-nicotinoyl-3-(4′-hydroxy-3′-methyl phenyl)-5-[(sub)phenyl]-2-pyrazolines. Bioorg. Med. Chem. Lett. 2006, 16, 3947–3949. [Google Scholar] [CrossRef] [PubMed]
- Manna, F.; Chimenti, F.; Bolasco, A.; Cenicola, M.L.; D’Amico, M.; Parrillo, C.; Rossi, F.; Marmo, E. Anti-inflammatory, analgesic and antipyretic N-acetyl-Δ2-pyrazolines and dihydrothienocoumarines. Eur. J. Med. Chem. 1992, 27, 633–639. [Google Scholar] [CrossRef]
- Bhat, A.R.; Athar, F.; Azam, A. Bis-pyrazolines: Synthesis, characterization and antiamoebic activity as inhibitors of growth of Entamoeba histolytica. Eur. J. Med. Chem. 2009, 44, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Manna, F.; Chimenti, F.; Bolasco, A.; Secci, D.; Bizzarri, B.; Befani, O.; Turini, P.; Mondovi, B.; Alcaro, S.; Tafi, A. Inhibition of amine oxidases activity by 1-acetyl-3,5-diphenyl-4,5-dihydro-(1H)-pyrazole derivatives. Bioorg. Med. Chem. Lett. 2002, 12, 3629–3633. [Google Scholar] [CrossRef]
- Levai, A.; Patonay, T.; Silva, A.M.S.; Pinto, D.C.G.A.; Cavaleiro, J.A.S. Synthesis of 3-aryl-5-styryl-2-pyrazolines by the reaction of (E,E)-cinnamylideneacetophenones with hydrazines and their oxidation into pyrazoles. J. Heterocycl. Chem. 2002, 39, 751–758. [Google Scholar] [CrossRef]
- Kedjadja, A.; Kolli, E.; Bouraiou, A.; Merdes, R. 1-[3-(2-Methyl-4-phenylquinolin-3-yl)-5-phenyl-4,5-dihydro-1H-pyrazol-1-yl]-propane-1-one. Molbank 2015, 2015, M863. [Google Scholar] [CrossRef]
- Wang, G.W.; Jia, C.S.; Dong, Y.W. Benign and highly efficient synthesis of quinolines from 2-aminoarylketone or 2-aminoarylaldehyde and carbonyl compounds mediated by hydrochloric acid in water. Tetrahedron Lett. 2006, 47, 1059–1063. [Google Scholar] [CrossRef]
- Kotra, V.; Ganapaty, S.; Adapa, S.R. Synthesis of new series of quinolinylchalcones as anticancer and anti-inflammatory agents. Indian J. Chem. Sec. B 2010, 49, 1109–1116. [Google Scholar]
- Fun, H.K.; Loh, W.S.; Sarveswari, S.; Vijayakumar, V.; Reddy, B.P. 1-(6-Chloro-2-methyl-4-phenyl-3-quinolyl)ethanone. Acta Crystallogr. 2009, E65, o2688–o2689. [Google Scholar] [CrossRef] [PubMed]
- Kedjadja, A.; Merdes, R.; Bouacida, S.; Roisnel, T.; Belfaitah, A. (2E,2′E)-1,1′-Bis(6-chloro-2-methyl-4-phenylquinolin-3-yl)-3,3′-(1,4-phenylene)diprop-2-en-1-one ethyl acetate disolvate. Acta Crystallogr. 2013, E69, o37–o38. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kedjadja, A.; Kolli, E.; Bouraiou, A.; Merdes, R.; Haddour, N. 1,1′-{1,4-Phenylene bis[3-(6-chloro-2-methyl-4-phenylquinolin-3-yl)-4,5-dihydro-1H-pyrazole-5,1-diyl]}dibutan-1-one. Molbank 2015, 2015, M868. https://doi.org/10.3390/M868
Kedjadja A, Kolli E, Bouraiou A, Merdes R, Haddour N. 1,1′-{1,4-Phenylene bis[3-(6-chloro-2-methyl-4-phenylquinolin-3-yl)-4,5-dihydro-1H-pyrazole-5,1-diyl]}dibutan-1-one. Molbank. 2015; 2015(4):M868. https://doi.org/10.3390/M868
Chicago/Turabian StyleKedjadja, Allaoua, Elhadj Kolli, Abdelmalek Bouraiou, Rachid Merdes, and Naoufel Haddour. 2015. "1,1′-{1,4-Phenylene bis[3-(6-chloro-2-methyl-4-phenylquinolin-3-yl)-4,5-dihydro-1H-pyrazole-5,1-diyl]}dibutan-1-one" Molbank 2015, no. 4: M868. https://doi.org/10.3390/M868
APA StyleKedjadja, A., Kolli, E., Bouraiou, A., Merdes, R., & Haddour, N. (2015). 1,1′-{1,4-Phenylene bis[3-(6-chloro-2-methyl-4-phenylquinolin-3-yl)-4,5-dihydro-1H-pyrazole-5,1-diyl]}dibutan-1-one. Molbank, 2015(4), M868. https://doi.org/10.3390/M868






