Effects of Milling and Cooking Processes on the Deoxynivalenol Content in Wheat
Abstract
:1. Introduction
2. Effects of primary processing; cleaning, scouring, density segregation and milling on the retention of DON
2.1. Effects of selection processes prior to milling on the removal of DON
2.2. Effects of dry milling and DON distribution for each milling fraction
3. Effects of secondary processing; cooking with heating on the retention of DON
4. DON metabolites and reaction products with matrix
5. Discussion
- Development of effective selection method for the discrimination of visually uninfected kernels from DON containing ones, since elimination of DON during processing is not an easy task.
- Validation of analytical method for the assessment of the retention of DON in food production chain. There are many reports with analytical values cited in this article, but little of them describe validation data such as method recovery for each matrix, limit of detection, and intermediate precision.
- Multiple analyses of DON, NIV and ZON, which often co-exist in scabby wheat. Gas chromatography (GC) coupled with mass spectrometry (GC-MS), high-performance liquid chromatography (HPLC), and HPLC coupled with tandem mass spectrometry (LC-MS/MS) will be applicable for the multiple analyses of Fusarium mycotoxins [70].
- Elucidation of small amounts of degraded DON and conjugated DON metabolites with their toxicological effects. Toxic effects in food are not always correlated with DON content, since there are some degradation products with different toxicological effects. The chemical fate of DON during thermal cooking is also not well understood and it is unclear that reduced concentrations of DON results from decomposition of DON or from their chemical binding to compounds (sugar, protein, and others) in food matrices. Very recently, several studies have suggested that DON may occur in a bound or conjugated form. DON-3G has gained considerable interest because of its universal existence in cereals. Therefore, it might be necessary to investigate total DON in cereals including DON-3G or masked mycotoxin [71, 72]. Recent advance on the analytical instruments such as LC-MSMS and LC- time of flight (TOF)-MS will help the elucidation of small amounts of degraded DON and conjugated DON metabolites. The development of the analytical equipment combined with a practical bioassay system which can predict biological effects will greatly contribute to the toxicological study of DON in humans.
6. Conclusions
Acknowledgments
References and Notes
- Creppy, EE. Update of survey, regulation and toxic effects of mycotoxins in Europe. Toxicol. Lett 2002, 127, 19–28. [Google Scholar]
- McLean, M. The phytotoxicity of Fusarium metabolites: An update since 1989. Mycopathologia 1996, 133, 163–179. [Google Scholar]
- Seitz, LM; Bechtel, DB. Chemical, physical and microscopical studies of scab-infected hard red winter wheat. J. Agric. Food Chem 1985, 33, 373–179. [Google Scholar]
- Gutleb, AC; Morrison, E; Murk, AJ. Cytotoxicity assays for mycotoxins produced by Fusarium strains: a review. Environ. Toxicol. Pharmacol 2002, 1, 309–320. [Google Scholar]
- Lee, US; Jang, HS; Tanaka, T; Toyasaki, N; Sugiura, Y; Oh, YJ; Cho, CM; Ueno, Y. Mycological survey of Korean cereals and production of mycotoxins by Fusarium isolates. Appl. Environ. Microbiol 1986, 52, 1258–1260. [Google Scholar]
- Osborne, LE; Stein, JM. Epidemiology of Fusarium head blight on small-grain cereals. Int. J. Food Microbiol 2007, 119, 103–108. [Google Scholar]
- Mirocha, CJ; Pathre, SV; Schauerhamer, B; Christensen, CM. Natural occurrence of Fusarium toxins in feedstuff. Appl. Environ. Microbiol 1976, 32, 553–556. [Google Scholar]
- Morgavi, DP; Riley, RT. An historical overview of field disease outbreaks known or suspected to be caused by consumption of feeds contaminated with Fusarium toxins. Anim. Feed Sci. Technol 2007, 137, 201–212. [Google Scholar]
- Ueno, Y; Ishii, K; Sato, N; Ohtsubo, K. Toxicological approaches to the metabolites of Fusaria. VI. Vomiting factor from moldy corn infected with Fusarium spp. Jpn. J. Exp. Med 1974, 44, 123–127. [Google Scholar]
- Desjardins, AE; Hohn, TM; McCormick, SP. Trichothecenes biosynthesis in Fusarium species: Chemistry, genetics, and significance. Microbiol. Rev 1993, 57, 595–604. [Google Scholar]
- Ehrlich, K; Daigle, KW. Protein synthesis inhibition by 8-oxo-12,13-epoxytrichothecenes. Biochim. Biophys. Acta 1987, 923, 206–213. [Google Scholar]
- Rotter, BA; Prelusky, DB; Pestka, JJ. Toxicology of deoxynivalenol (Vomitoxin). J. Toxicol. Environ. Health 1996, 48, 1–34. [Google Scholar]
- Dong, W; Pestka, JJ. Persistent dysregulation of IgA production and IgA nephropathy in the B6C3F1 mouse following withdrawal of dietary vomitoxin (deoxynivalenol). Fundam. Appl. Toxicol 1993, 20, 38–47. [Google Scholar]
- Tryphonas, H; Iverson, F; So, Y; Nera, EA; Mcguire, PF; O'Grady, L; Clayson, DB; Scott, PM. Effects of deoxynivalenol (vomitoxin) on the humoral and cellular immunity of mice. Toxicol. Lett 1986, 30, 137–150. [Google Scholar]
- Pestka, JJ; Bondy, GS. Alteration of immune function following dietary mycotoxin exposure. Can. J. Physiol. Pharmacol 1990, 66, 1009–1016. [Google Scholar]
- Cole, RJ; Cox, RH. Handbook of toxic fungal metabolites; Academic Press: New York, USA, 1981; pp. 152–263. [Google Scholar]
- Forsell, JH; Jensen, R; Tai, J-H; Witt, M; Lin, WS; Pestka, JJ. Comparison of acute toxicities of deoxynivalenol (vomitoxin) and 15-acetyldeoxynivalenol in the B6C3F1 mouse. Food Chem. Toxicol 1987, 25, 155–162. [Google Scholar]
- Rocha, O; Ansari, K; Doohan, FM. Effects of trichothecene mycotoxins on eukaryotic cells. Food Addit. Contam 2005, 22, 369–378. [Google Scholar]
- Pestka, JJ; Smolinski, AT. Deoxynivalenol: toxicology and potential effects on humans. J. Toxicol. Environ. Health B Crit. Rev 2005, 8, 39–69. [Google Scholar]
- Sundstøl Eriksen, G; Pettersson, H; Lundh, T. Comparative cytotoxicity of deoxynivalenol, nivalenol, their acetylated derivatives and de-epoxy metabolites. Food Chem. Toxicol 2004, 42, 619–624. [Google Scholar]
- Canady, RA; Coker, RD; Egan, SK; Krska, R; Kuiper-Goodman, T; Olsen, M; Pestka, J; Resnik, S; Schlatter, J. Deoxynivalenol. WHO/IPCS safety evaluation of certain mycotoxins in food. WHO Food Addit. Ser 2001, 47, 419–555. [Google Scholar]
- Poapolathep, A; Ohtsuka, R; Kiatipattanasakul, W; Ishigami, N; Nakayama, H; Doi, K. Nivalenol-induced apoptosis in thymus, spleen and Peyer's patches of mice. Exp. Toxicol. Pathol 2002, 53, 441–446. [Google Scholar]
- European Commission, Scientific Committee on Food. Opinion of the Scientific Committee on Food on Fusarium toxins, Part 6: Group evaluation of T-2 toxin, HT-2 toxin, nivalenol and deoxynivalenol. 2002.
- Yuen, GY; Schoneweis, SD. Strategies for managing Fusarium head blight and deoxynivalenol accumulation in wheat. Int. J. Food Microbiol 2007, 119, 126–130. [Google Scholar]
- Scott, PM; Kanhere, SR; Lau, P-Y; Dexter, JE; Greenhalgh, R. Effects of Experimental flour milling and breadbaking on retention of deoxynivalenol (vomitoxin) in hard red spring wheat. Cereal Chem 1983, 60, 421–424. [Google Scholar]
- Young, JC; Fulcher, RG; Hayhoe, JH; Scott, PM; Dexter, JE. Effect of milling and baking on deoxynivalenol (vomitoxin) content of eastern Canadian wheats. In J. Agric. Food Chem.; 1984; Volume 32, pp. 659–664. [Google Scholar]
- Seitz, LM; Yamazaki, WT; Clements, RL; Mohr, HE; Andrews, L. Distribution of deoxynivalenol in soft wheat mill streams. Cereal Chem 1985, 62, 467–469. [Google Scholar]
- Abbas, HK; Mirocha, CJ; Pawlosky, RJ; Pusch, DJ. Effect of cleaning, milling, and baking on deoxynivalenol in wheat. Appl. Environ. Microbiol 1985, 50, 482–486. [Google Scholar]
- Seitz, LM; Eustace, WD; Mohr, HE; Shogren, MD; Yamazaki, WT. Cleaning, milling and baking tests with hard red winter wheat containing deoxynivalenol. Cereal Chem 1986, 63, 146–150. [Google Scholar]
- Nowicki, TW; Gaba, DG; Dexter, JE; Matsuo, RR; Clear, RM. Retention of the Fusarium mycotoxin deoxynivalenol in wheat during processing and cooking of spaghetti and noodles. J. Cereal Sci 1988, 8, 189–202. [Google Scholar]
- Tkachuk, R; Dexter, JE; Tipples, KH; Nowicki, TW. Removal by specific gravity table of tombstone kernels and associated trichothecene from wheat infected with Fusarium head blight. Cereal Chem 1991, 68, 428–431. [Google Scholar]
- Trigo-Stockli, DM. Effect of processing on deoxynivalenol and other trichothecenes. Adv. Exp. Med. Biol 2002, 504, 181–188. [Google Scholar]
- Huff, WE; Hagler, WM, Jr. Density segregation of corn and wheat naturally contaminated with aflatoxin, deoxynivalenol and zearalenone. J. Food Prot 1985, 48, 416–420. [Google Scholar]
- Kos, G; Lohninger, H; Krska, R. Development of a method for the determination of Fusarium fungi on corn using mid-infrared spectroscopy with attenuated total reflection and chemometrics. Anal. Chem 2003, 75, 1211–1217. [Google Scholar]
- Hart, LP; Braselton, WE, Jr. Distribution of vomitoxin in dry milled fractions of wheat infected with Gibberella zeae. J. Agric. Food Chem 1983, 31, 657–659. [Google Scholar]
- Scott, PM; Kanhere, SR; Dexter, JE; Brennan, PW; Trenholm, HL. Distribution of the trichothecene mycotoxin deoxynivalenol (vomitoxin) during the milling of naturally contaminated hard red spring wheat and its fate in baked products. Food Addit. Contam 1984, 1, 313–323. [Google Scholar]
- Tanaka, T; Hasegawa, A; Yamamoto, S; Matsuki, Y; Ueno, Y. Residues of Fusarium mycotoxins, nivalenol, deoxynivalenol and zearalenone, in wheat and processed food after milling and baking. J. Food Hyg. Soc. Japan 1986, 27, 653–655. [Google Scholar]
- Lee, U-S; Jang, H-S; Tanaka, T; Oh, Y-J; Cho, C-M; Ueno, Y. Effect of milling on decontamination of Fusarium mycotoxins nivalenol, deoxynivalenol, and zearalenone in Korean wheat. J. Agric. Food Chem 1987, 35, 126–129. [Google Scholar]
- Dexter, JE; Clear, RM; Preston, KR. Fusarium head blight: effect on the milling and baking of some Canadian wheats. Cereal Chem 1996, 73, 695–701. [Google Scholar]
- Trigo-Stockli, DM; Deyoe, CW; Satumbaga, RF; Pedersen, JR. Distribution of deoxynivalenol and zearalenone in milled fractions of wheat. Cereal Chem 1996, 73, 388–391. [Google Scholar]
- Dexter, JE; Marchylo, BA; Clear, RM; Clarke, JM. Effect of Fusarium head blight on semolina milling and pasta-making quality of durum wheat. Cereal Chem 1997, 74, 519–524. [Google Scholar]
- Hart, LP; Casper, H; Schabenberger, O; Ng, P. Comparison of gas chromatography-electron capture and enzyme-linked immunosorbent assay for deoxynivalenol in milled fractions of naturally contaminated wheat. J. Food Prot 1998, 61, 1695–1697. [Google Scholar]
- Manthey, FA; Wolf-Hall, CE; Yalla, S; Vijayakumar, C; Carlson, D. Microbial loads, mycotoxins, and quality of durum wheat from the 2001 harvest of the northern plains region of the United States. J. Food Prot 2004, 67, 772–780. [Google Scholar]
- Visconti, A; Haidukowski, EM; Pascale, M; Silvestri, M. Reduction of deoxynivalenol during durum wheat processing and spaghetti cooking. Toxicol. Lett 2004, 153, 181–189. [Google Scholar]
- Hazel, CM; Patel, S. Influence of processing on trichothecene levels. Toxicol. Lett 2004, 153, 51–59. [Google Scholar]
- Tanaka, K; Hara, N; Goto, T; Manabe, M. Reduction of mycotoxins contamination by processing grain. In Proc. Int. Symp. Mycotoxicol; 1999; Volume 1999, pp. 95–100. [Google Scholar]
- Palpacelli, V; Beco, L; Ciani, M. Vomitoxin and zearalenone content of soft wheat flour milled by different methods. J. Food Prot 2007, 70, 509–513. [Google Scholar]
- Lancova, K; Hajslova, J; Kostelanska, M; Kohoutkova, J; Nedelnik, J; Moravcova, H; Vanova, M. Fate of trichothecene mycotoxins during the processing: milling and baking. Food Addit. Contam 2008, 25, 650–659. [Google Scholar]
- Gärtner, BH; Munich, M; Kleijer, G; Mascher, F. Characterisation of kernel resistance against Fusarium infection in spring wheat by baking quality and mycotoxin assessments. Eur. J. Plant Pathol 2008, 120, 61–68. [Google Scholar]
- El-Banna, AA; Lau, P-Y; Scott, PM. Fate of mycotoxins during processing of foodstuffs II-Deoxynivalenol (vomitoxin) during making of Egyptian bread. J. Food Prot 1983, 46, 484–486. [Google Scholar]
- Boyacioglu, D; Hettiarachchy, NS; D’appolonia, BL. Additives affect deoxynivalenol (vomitoxin) flour during breadbaking. J. Food Sci 1993, 58, 416–418. [Google Scholar]
- Accerbi, M; Rinaldi, VE; Ng, PK. Utilization of highly deoxynivalenol-contaminated wheat via extrusion processing. J. Food Prot 1999, 62, 1485–1487. [Google Scholar]
- Sugita-Konishi, Y; Park, BJ; Kobayashi-Hattori, K; Tanaka, T; Chonan, T; Yoshikawa, K; Kumagai, S. Effect of cooking process on the deoxynivalenol content and its subsequent cytotoxicity in wheat products. Biosci. Biotechnol. Biochem 2006, 70, 1764–1768. [Google Scholar]
- Samar, MM; Neira, MS; Resnik, SL; Pacin, A. Effect of fermentation on naturally occurring deoxynivalenol (DON) in Argentinean bread processing technology. Food Addit. Contam 2001, 18, 1004–1010. [Google Scholar]
- Samar, MM; Resnik, SL; Gonzalez, HHL; Pacin, AM; Castillo, MD. Deoxynivalenol reduction during the frying process of turnover pie coveres. Food Control 2007, 18, 1295–1299. [Google Scholar]
- Bretz, M; Beyer, M; Cramer, B; Knecht, A; Humpf, HU. Thermal degradation of the Fusarium mycotoxin deoxynivalenol. J. Agric. Food Chem 2006, 54, 6445–6451. [Google Scholar]
- Ragab, WS; Drusch, S; Schnieder, F; Beyer, M. Fate of deoxynivalenol in contaminated wheat grain during preparation of Egyptian 'balila'. Int. J. Food Sci. Nutr 2007, 58, 169–177. [Google Scholar]
- Castells, M; Marin, S; Sanchis, V; Ramos, AJ. Fate of mycotoxins in cereals during extrusion cooking: A review. Food Addit. Contam 2005, 22, 150–157. [Google Scholar]
- Scudamore, KA; Guy, RC; Kelleher, B; MacDonald, SJ. Fate of the fusarium mycotoxins, deoxynivalenol, nivalenol and zearalenone, during extrusion of wholemeal wheat grain. Food Addit. Contam 2008, 25, 331–337. [Google Scholar]
- Cenkowski, S; Pronyk, C; Zmidzinska, D; Muir, WE. Decontamination of food products with superheated steam. J. Food Eng 2007, 83, 68–75. [Google Scholar]
- Greenhalgh, R; Gilbert, J; King, RR; Blackwell, BA; Startin, JR; Shepherd, MJ. Synthesis, characterization, and occurrence in bread and cereal products of an isomer of 4-deoxynivalenol (vomitoxin). J. Agric. Food Chem 1984, 32, 1416–1420. [Google Scholar]
- Yoshizawa, T; Cote, LM; Swanson, SP; Buck, WB. Confirmation of DOM-1, a de-epoxidation metabolite of deoxynivalenol, in biological fluids of lactating cows. Agric. Biol. Chem 1986, 50, 227–229. [Google Scholar]
- He, P; Young, LG; Forsberg, C. Microbial transformation of deoxynivalenol (vomitoxin). Appl. Environ. Microbiol 1992, 58, 3857–3863. [Google Scholar]
- Völkl, A; Vogler, B; Schollenberger, M; Karlovsky, P. Microbial detoxification of mycotoxin deoxynivalenol. J. Basic Microbiol 2004, 44, 147–156. [Google Scholar]
- Shima, J; Takase, S; Takahashi, Y; Iwai, Y; Fujimoto, H; Yamazaki, M; Ochi, K. Novel detoxification of the trichothecene mycotoxin deoxynivalenol by a soil bacterium isolated by enrichment culture. Appl. Environ. Microbiol 1997, 63, 3825–3830. [Google Scholar]
- Wu, X; Murphy, P; Cunnick, J; Hendrich, S. Synthesis and characterization of deoxynivalenol glucuronide: Its comparative immunotoxicity with deoxynivalenol. Food Chem. Toxicol 2007, 45, 1846–1855. [Google Scholar]
- Sewald, N; Lepschy von Gleissenthall, J; Schuster, M; Müller, G; Aplin, RT. Structure elucidation of a plant metabolite of 4-desoxynivalenol. Tetrahedron Asymmetr 1992, 3, 953–960. [Google Scholar]
- Berthiller, F; Dall'Asta, C; Schuhmacher, R; Lemmens, M; Adam, G; Krska, R. Masked mycotoxins: determination of a deoxynivalenol glucoside in artificially and naturally contaminated wheat by liquid chromatography-tandem mass spectrometry. J. Agric. Food Chem 2005, 53, 3421–3425. [Google Scholar]
- Abbas, HK; Mirocha, CJ; Rosiles, R; Carvajal, M. Decomposition of zearalenone and deoxynivalenol in the process of making tortillas from corn. Cereal Chem 1988, 65, 15–19. [Google Scholar]
- Kushiro, M; Tanaka, K; Miyazaki, S; Nagata, T. Advances of liquid chromatographic determination of fumonisins; potential mycotoxins for humans. Curr. Pharm. Anal 2006, 2, 289–297. [Google Scholar]
- Liu, Y; Walker, F; Hoeglinger, B; Buchenauer, H. Solvolysis procedures for the determination of bound residues of the mycotoxin deoxynivalenol in fusarium species infected grain of two winter wheat cultivars preinfected with barley yellow dwarf virus. J. Agric. Food Chem 2005, 53, 6864–6869. [Google Scholar]
- Zhou, B; Li, Y; Gillespie, J; He, GQ; Horsley, R; Schwarz, P. Doehlert matrix design for optimization of the determination of bound deoxynivalenol in barley grain with trifluoroacetic acid (TFA). J. Agric. Food Chem 2007, 55, 10141–10149. [Google Scholar]
Abbreviations:
| DON | deoxynivalenol |
| FHB | Fusarium head blight |
| ZON | zearalenone |
| NIV | nivalenol |
| DON-3G | DON-3-glucoside |
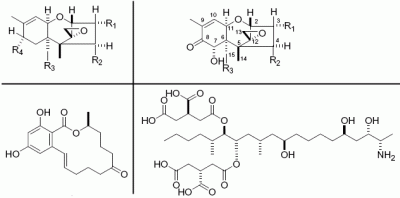
| Type B trichothecene | Molecular formula | Toxicity | Comments | Ref. |
|---|---|---|---|---|
| deoxynivalenol (DON) | C15H20O6 | emesis and feed refusal in swine, IgA nephropathy in mice, alter humoral immunity in mice, inhibition of RNA and DNA synthesis in human cell lines, activation of MAPK in human cell lines, activation of cytokines in murine and human cells, LD50 in ddS strain of male mice dosed IP was 70 mg/kg; females, 76.7 mg/kg, IC50 for the inhibition of DNA synthesis was 1.50 mM in mouse 3T3 cells, LD50 values for DON were estimated to be 78 mg/kg (oral) and 49 mg/kg (i.p.) in the B6C3F1 mouse | Major mycotoxin in scabby cereals | [4, 12–21] |
| nivalenol (NIV) | C15H20O7 | LD50 in mice dosed IP was 5 mg/kg, induced apoptosis in thymus, spleen and Peyer's patches of mice, IC50 for the inhibition of DNA synthesis was 1.19 mM in mouse 3T3 cells | Another major type B trichothecene co-occurred with DON | [16, 18–20, 22] |
| 3-acetyldeoxynivalenol | C17H22O7 | LD50 in ddS strain of male mice dosed IP was 49.5 mg/kg; females, 49.9 mg/kg, IC50 for the inhibition of DNA synthesis was 14.4 mM in mouse 3T3 cells | biosynthetic precursor of DON in fungi | [16, 20] |
| 15-acetyldeoxynivalenol | C17H22O7 | IC50 for the inhibition of DNA synthesis was 1.51 mM in mouse 3T3 cells, LD50 values for 15-acetylDON were 34 mg/kg (oral) and 113 mg/kg (i.p.). | biosynthetic precursor of DON in fungi | [20] |
| Fusarenon-X | C17H22O8 | LD50 in ddS strain of male mice dosed IP was 3.3 mg/kg, IC50 for the inhibition of DNA synthesis was 0.72 mM in mouse 3T3 cells | [16, 20] |
| Wheat | Mycotoxin | Treatment | Effect | Ref. |
|---|---|---|---|---|
| Hard Red Spring (Canada) | DON (7.1 mg/kg) | cleaning (Carter dockage tester) | DON in cleaned wheat was 4.6 mg/kg, while DON in dockage was 16.7 mg/kg | [25] |
| Soft White Winter(Canada) | DON | cleaning (Carter dockage tester) | cleaning did not reduce DON level | [26] |
| Soft wheat (USA) | DON | cleaning (combination of screening and air flow) | DON content was decreased by 16% and screening had 4.7 fold higher DON contents than cleaned wheat | [27] |
| wheat (USA) | DON (7.9–9.6 mg/kg) | cleaning | The percent reduction found in the cleaned wheat ranged from 6 to 19%. | [28] |
| Hard Red Winter (USA) | DON (0.64–5.1 mg/kg) | cleaning (Superior cylinder separators) followed by washing (Smico wheat washer) | normal, single cleaning obtained the cleaning efficiency ranging from 48% to 86% depending on DON concentration. | [29] |
| Canadian Western Red Spring, variety Sinton (Canada) | DON (12.5 mg/kg) | scouring | scouring reduced DON level by 22% | [30] |
| Canadian Western Amber Durum, variety Coulter(Canada) | DON (9.6 mg/kg) | scouring | without scouring did not affect DON level | [30] |
| Canadian Eastern White Winter(Canada) | DON | cleaning (Carter C-989 dockage tester) followed by fractionating (SY 300 gravity separator, Spiroll Kipp Kelly Inc.) | DON is highest in the least dense fractions | [31] |
| wheat naturally contaminated with DON and ZON | DON (2.4 mg/kg) | density segregation by soaking in water and 30% sucrose | removing wheat buoyant in water and then 30% sucrose decreased the DON present by 96%, and reduced the DON content by 96% | [33] |
| wheat naturally contaminated with DON and ZON | DON (0.6 mg/kg) | density segregation by soaking in water and 30% sucrose | removing wheat buoyant in water and then 30% sucrose decreased the DON present by 68%, and reduced the DON content by 67% | [33] |
| Wheat | Mycotoxin | Treatment | Effect | Ref. |
|---|---|---|---|---|
| Hard Red Spring (Canada) | DON (4.6 mg/kg) | milling (Allis-Chalmers laboratory mill) | DON in 75% yield straight-grade flour was 4.1mg/kg, while 4.6, 6.9, 8.0 ppm in bran, shorts, and feed flour (red dog), respectively | [25] |
| Soft White Winter (Canada) | DON (0.42–0.62 mg/kg) | milling (industrially milled and pilot-milled) | milling led to a fractionation of DON, with increased levels in the outer kernel (bran, 0.98 mg/kg, for example) portions and decreased levels in the inner flour portions (break flour, 0.28 mg/kg) | [26] |
| soft Wheat, variety Pike, Hart, Stadler, Oasis, Pioneer, Mcnair, Arthur (USA) | DON (0.03–3.35 mg/kg) | dry milling (Miag Multomat mill) | DON was found in all fractions, which included straight-grade flour, four break flours, six reduction flours, break and reduction shorts, red dog, and bran. Mean DON concentration in straight-grade flour was about 90% of that in cleaned wheat. | [27] |
| wheat (USA) | DON (7.9–9.6 mg/kg) | milling (Bühler automatic laboratory mill Model MLU-202) | DON was found throughout all the milling fractions. The greatest (21 mg/kg) concentration of DON was found in the bran, and the smallest (1 mg/kg) was found in the break flour. | [28] |
| Hard Red Winter (USA) | DON (0.64–5.1 mg/kg) | milling (Miag Multomat Mill) | all mill fractions contained DON with lower concentration in flours and higher concentration in offals compared to cleaned wheat, DON concentration of straight-grade flour was 75% of cleaned wheat | [29] |
| Canadian Western Red Spring, variety Sinton (Canada) | DON (12.5 mg/kg) | milling (Bühler laboratory mill Model MLU-202) | mould counts highest in bran, 29% of DON was retained in flour | [30] |
| Canadian Western Amber Durum, variety Coulter (Canada) | DON (9.6 mg/kg) | milling (Bühler laboratory mill Model MLU-202) | mould counts highest in shorts, 52% of DON was retained in semolina | [30] |
| White Winter (Canada) | milling (Allis-Chalmers laboratory mill) | [31] | ||
| soft wheats; Red Winter (USA), White Winter (Canada) | DON (2.0 mg/kg) | dry milling | DON was distributed throughout all fractions of the milled grains; bran,red dogs and shorts: 5.2 mg/kg, straight-grade flour: 4.5 mg/kgwhile total DON recovered was 4.7 mg/kg | [35] |
| Hard Red Spring (Canada) | DON (7.5 mg/kg, 1.4 mg/kg) | milling by experimental mill (Allis-Chalmers mill) and commercial-scale pilot mill | no appreciable losses of DON occurred during the milling, DON concentrations raised up to two-fold in shorts and feed flour fractions | [36] |
| wheat (Japan) | DON (0.17mg/kg), co-contaminated with NIV and ZON | milling (Bühler experimental mill) | ca 60% of DON was remained after milling, DON content in bran was 2.7 times of that in the original wheat | [37] |
| wheat (Korea) | DON (0.068, 0.31 mg/kg), co-contaminated with NIV and ZON | milling (Bühler test mill) | 24–48% reduction of DON in flour fractions intended for human consumption | [38] |
| Hard Red Spring, cultivar Glenlea, Grandin, Roblin, Taber (Canada) | DON (0.6–2.0 mg/kg) | milling (Allis-Chalmers five-stand mill) | reduction ratio of DON during milling was cultivar specific, DON level in flour was about half of wheat in Glenlea | [39] |
| Hard Red Winter (USA) | DON, ZON (<1.0 - >5.0 mg/kg) | milling (Bühler mill) | DON levels was highest in the bran (3.4 mg/kg) and lowest in the flour (1.5 mg/kg) | [40] |
| durum, cultivar Reva, DT 673, AC Melita, Durex, Hercules, DT 666, Kyle, Plenty, DT 369, Duraking (Canada) | DON (0.6–3.7 mg/kg) | milling (Allis-Chalmers five-stand mill) | about half of DON was retained in semolina of all samples | [41] |
| durum, cultivar Duilio, Bracco, Orobel (Italy) | DON (0.3 to 13.1 mg/kg) | milling (Bühler automatic laboratory mill Model MLU-202) | the level of DON in cleaned wheat was 77% of that in uncleaned wheat. DON levels in the screenings, bran and fine middlings were 4.1–, 1.6- and 0.6-fold, respectively, relative to the uncleaned wheat. | [44] |
| wheat, cultivar Seto-komugi, Asakaze-komugi (Japan) | DON, NIV (0.15–0.55 mg/kg) | milling (Bühler test mill) | DON content in bran was 2.3–2.4 times of that in the original wheat | [46] |
| wheat, variety Bolero (Italy) | DON (undetermine d before milling) | traditional milling with a stone mill/modern milling with a roller mill | traditional milling reduced DON level to 0.17mg/kg, while DON level became 0.36 mg/kg by modern milling | [47] |
| wheat (Czech Republic) | DON (0.09–2.99 mg/kg) | milling (Bühler automatic laboratory mill Model MLU-202) | the highest concentrations of DON were found in the bran, the lowest in the reduction flours. In Sulamit; uncleaned grain 108, dust 2689, cleaned grain 59, 1st-3rd break flour 42–64, 1st-3rd reduction flour 26–33, bran 117 (ng/g) | [48] |
| spring wheat,variety Grena, Carasso, Lana, Brusino, Toronit, Fiorina, Quarna, Nadro (Switzerland) | DON (undetermined before milling) | medium size sample mill (Bühler MLU202) | DON contents in break flour, reduction flour, shorts, bran were 16.2, 16.9, 84.4, 122.0 mg/kg respectively, in Greina variety, which showed the most severe FHB symptom | [49] |
| Wheat | Mycotoxin | Treatment | Effect | Ref. |
|---|---|---|---|---|
| Hard Red Spring (Canada) | DON (4.1 mg/kg) | baking (30 min at 205°C) | DON was not destroyed on making bread | [25] |
| Soft White Winter (Canada) | DON (0.28–0.44 mg/kg) | baking cookies by a standard commercial recipe | DON level was decreased up to 35% | [26] |
| wheat (USA) | DON (0.52–0.31 mg/kg) | baking by an approved method of the American Association of Cereal Chemists | DON was not destroyed, but the effect on its concentration in the samples analyzed varied, the reduction ranging from 19 to 69% | [28] |
| Hard Red Winter (USA) | DON (0.22–5.8 mg/kg) | baking (24 min at 215 °C) | decreased DON in bread from lower DON contaminated wheat, whereas increased DON in bread from higher (>1.6 mg/kg) DON contaminated wheat | [29] |
| wheat (Japan) | DON (0.17 mg/kg), co-contaminated with NIV and ZON | baking (30 min at 170 °C) | baking did not affect DON content | [37] |
| wheat (Czech Republic) | DON (0.09–2.99 mg/kg) | baking (14 min at 210 °C) | baking had no significant effect on DON levels | [48] |
| DON | baking Egyption bread (2 min at 350 °C) | DON was not reduced | [50] | |
| Hard Red Spring, cultivar Len (USA) | DON (3.13 mg/kg) | baking (20 min at 220 °C) | L-cysteine as a food additive was significantly reduced DON (38–46%) in bread, isoDON was formed | [51] |
| wheat, cultivar Haruyokoi (Japan) | DON (0.71 mg/kg) | baking (35 min at 160 °C) | the level of DON was not reduced while the cytotoxicity (in Swiss mouse 3T3 fibloblasts) was reduced slightly but significantly | [53] |
| wheat (Argentina) | DON (150 mg/kg) | baking French bread on a pilot scale | 41% of reduction of DON was observed during fermentation at 50°C | [54] |
| wheat (Argentina) | DON (150 mg/kg) | baking Vienna bread on a pilot scale | 56% of reduction of DON was observed during fermentation at 50°C | [54] |
| wheat (Argentina) | DON (1.2 mg/kg) | frying (15 min at 169 °C, 2.5 min at 205 °C, 1.0 min at 243 °C) | No significant reduction of DON was observed | [55] |
| Canadian Western Red Spring, variety Sinton (Canada) | DON (12.5 mg/kg) | Chinese noodles (75 g of dried noodles (containing 1% NaCl and 1% Kansui, 12% moisture) were boiled in 750 mL water for 10 min and drained for 5 min | total DON retention (DON in cooked noodles and DON in cooking water solids/DON in uncooked noodles) was 57–58%, reduction of DON level was 49% after boiling | [30] |
| Canadian Western Red Spring, variety Sinton (Canada) | DON (12.5 mg/kg) | Japanese noodles (75 g of dried noodles (containing 1% NaCl, 12% moisture) were boiled in 750 mL water for 10 min and drained for 5 min | total DON retention (DON in cooked noodles and DON in cooking water solids/DON in uncooked noodles) was 107–115%, reduction of DON level was 40% after boiling | [30] |
| Canadian Western Amber Durum, variety Coulter (Canada) | DON (9.6 mg/kg) | spaghetti (75 g) made by low temperature extrusion (39°C for 28 h) was boiled in 750 mL water for 10 min and drained for 5 min | total DON retention* was 87–92%, DON concentration in cooked spaghetti made of uncooked spaghetti (DON conc. 6.2 mg/kg) was 3.5 mg/kg | [30] |
| Canadian Western Amber Durum, variety Coulter (Canada) | DON (9.6 mg/kg) | spaghetti (75 g) made by high temperature extrusion (70°C for 12 h) was boiled in 750 mL water for 10 min and drained for 5 min | total DON retention* was 90–92%, DON concentration in cooked spaghetti made of uncooked spaghetti (DON conc. 6.1 mg/kg) was 3.4 mg/kg | [30] |
| durum wheat | DON (0.3 to 13.1 mg/kg) | spaghetti (25 g, containing 1% NaCl, 11.5% moisture) was boiled in 100 mL water for 7 min and drained | total DON retention* was 88–114%, DON concentration in cooked spaghetti made of uncooked spaghetti (DON conc. 7.0 mg/kg) was 2.7 mg/kg | [44] |
| durum wheat | DON (0.3 to 13.1 mg/kg) | spaghetti (25 g, containing 1% NaCl, 11.5% moisture) was boiled in 125 mL water for 7 min and drained | total DON retention* was 74–89%, DON concentration in cooked spaghetti made of uncooked spaghetti (DON conc. 0.26 mg/kg) was 0.048 mg/kg | [44] |
| DON (7.3 mg/kg) | the effects of sodium bisulfite, extrusion cooking under high temperature and pressure | soaking treatment with SB solution (5% SO2 equivalent) lowered DON to 0.8 mg/kg and to 0.3 mg/kg with following extrusion process | [52] | |
| wheat, cultivar Hokushin (Japan) | DON (0.86 mg/kg) | Japanese noodles (50 g of dried noodles (containing 4% NaCl) were boiled in 1000 mL water for 10 min and dried at 40°C for 3 h | boiling process reduced both the level of DON and cytotoxicity (in Swiss mouse 3T3 fibloblasts) | [53] |
| no grain used | DON (100 mg) | cooking or baking with model heating (heat with 1 mg of model compounds: α-D-glucose as sugar model, for example) | heating under alkaline conditions decomposed DON partially and gave a mixture of norDON series | [56] |
| DON | preparation of Egyptian 'balila' | preparation by using Na2CO3 solution was effective to reduce DON level | [57] | |
| wheat | DON (1.0 mg/kg) | extrusion (140 °C –180 °C ) | No reduction of DON content by simple extrusion was observed | [59] |
| Hard Red Spring (Canada) | DON (15.8 mg/kg) | superheated steam | 48% of reduction of DON was occurred at 185 °C in the processing times of 6 min | [60] |
| Compound | Molecular formula | Toxicity | Comments | Ref. |
|---|---|---|---|---|
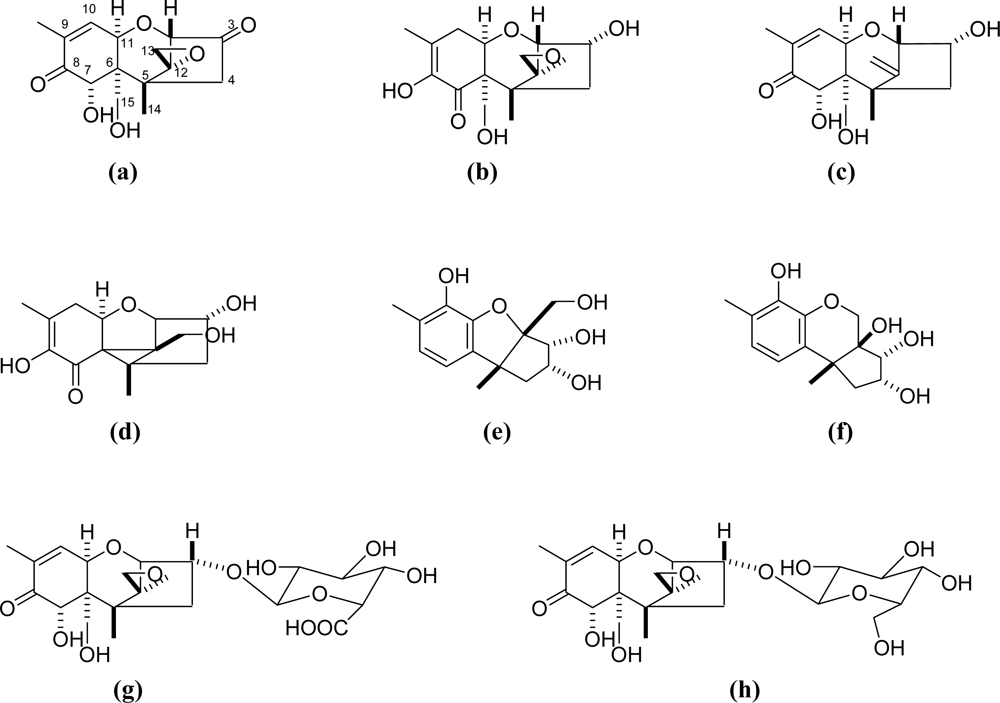
| 3-keto DON (a)* | C15H18O6 | no data available | transformed product of DON under aerobic conditions by microbial cultures | [64, 65] |
| isoDON (b) | C15H20O6 | no data available | found in bread | [51, 61] |
| de-epoxy DON (c) | C15H20O5 | IC50 for inhibition of DNA synthesis was 83.0 mM in mouse 3T3 cells | found in milk and serum in cows fed DON contaminated feed | [20, 62, 63] |
| norDON A (d) | C14H18O5 | less cytotoxic up to 100 mM in human kidney epithelial IHKE cells compared to DON (EC50=1.1 μm) | detected in 29–66% of the samples in mean concentrations ranging from 3 to 15 mg/kg during heating under alkaline conditions | [56] |
| norDON B (e) | C14H18O5 | less cytotoxic up to 100 mM in human kidney epithelial IHKE cells compared to DON (EC50=1.1 μm) | detected in 29–66% of the samples in mean concentrations ranging from 3 to 15 mg/kg during heating under alkaline conditions | [56] |
| norDON C (f) | C14H18O5 | less cytotoxic up to 100 mM in human kidney epithelial IHKE cells compared to DON (EC50=1.1 μm) | detected in 29–66% of the samples in mean concentrations ranging from 3 to 15 mg/kg during heating under alkaline conditions | [56] |
| DON-3-glucuronide (g) | C21H28O12 | no toxicity at up to 270 mM in human K562 erythroleukemia cell line, and significant difference in immunotoxicity compared to DON | detoxification product of DON in plasma or urine | [66] |
| DON-3-glucoside (DON-3G) (h) | C21H30O11 | no data available | in almost all investigated DON contaminated cereals also contain DON-3G | [67, 68] |
© 2008 by MDPI This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kushiro, M. Effects of Milling and Cooking Processes on the Deoxynivalenol Content in Wheat. Int. J. Mol. Sci. 2008, 9, 2127-2145. https://doi.org/10.3390/ijms9112127
Kushiro M. Effects of Milling and Cooking Processes on the Deoxynivalenol Content in Wheat. International Journal of Molecular Sciences. 2008; 9(11):2127-2145. https://doi.org/10.3390/ijms9112127
Chicago/Turabian StyleKushiro, Masayo. 2008. "Effects of Milling and Cooking Processes on the Deoxynivalenol Content in Wheat" International Journal of Molecular Sciences 9, no. 11: 2127-2145. https://doi.org/10.3390/ijms9112127
APA StyleKushiro, M. (2008). Effects of Milling and Cooking Processes on the Deoxynivalenol Content in Wheat. International Journal of Molecular Sciences, 9(11), 2127-2145. https://doi.org/10.3390/ijms9112127