Stromules: An Incident or Formalized Pathway for Molecules Transfer Between Organelles?
Abstract
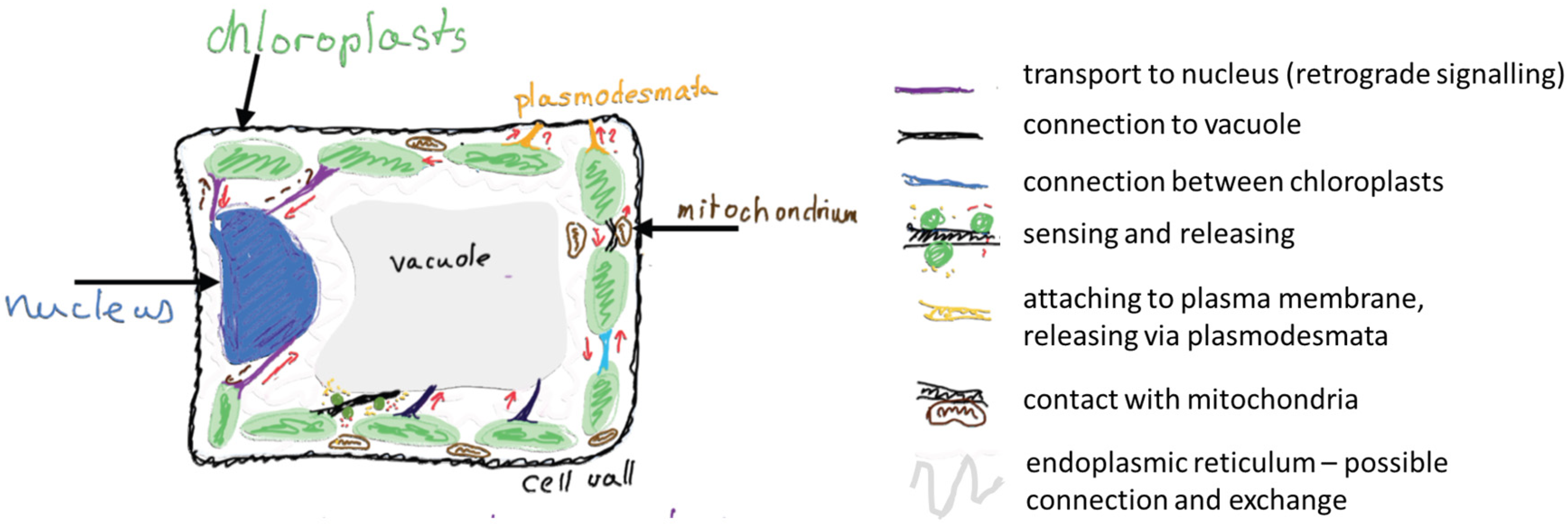
1. Introduction
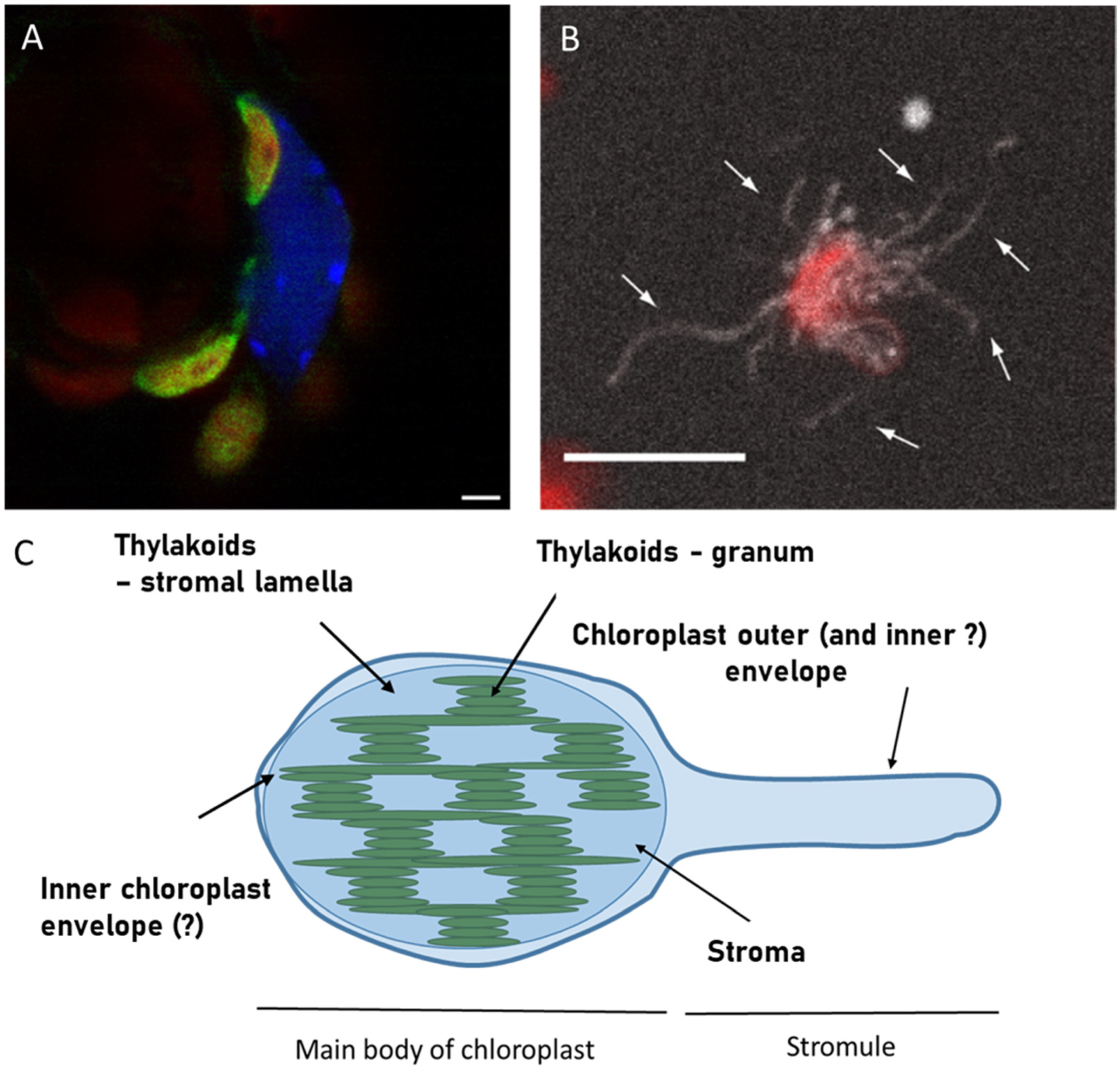
2. How They Began
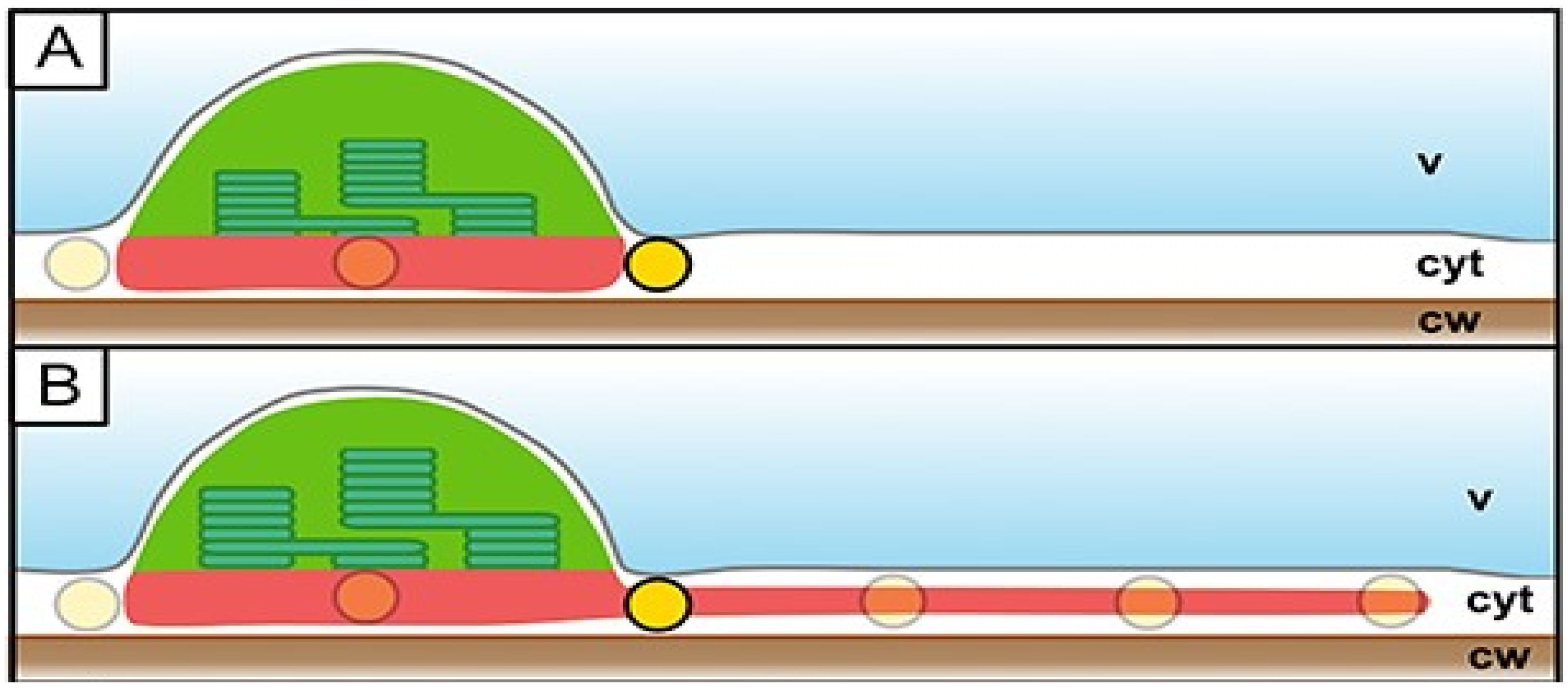
3. The Stromules and Chloroplast Envelope Membranes
4. Are Tubular Pathways Necessary?
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Waters, M.T.; Fray, R.G.; Pyke, K.A. Stromule formation is dependent upon plastid size, plastid differentiation status and the density of plastids within the cell. Plant J. 2004, 39, 655–667. [Google Scholar] [CrossRef]
- Gray, J.; Sullivan, J.; Hibberd, J.; Hansen, M. Stromules: Mobile protrusions and interconnections between plastids. Plant Biol. 2001, 3, 223–233. [Google Scholar] [CrossRef]
- Hanson, M.R.; Hines, K.M. Stromules: Probing formation and function. Plant Physiol. 2018, 176, 128–137. [Google Scholar] [CrossRef]
- Natesan, S.K.A.; Sullivan, J.A.; Gray, J.C. Stromules: A characteristic cell-specific feature of plastid morphology. J. Exp. Bot. 2005, 56, 787–797. [Google Scholar] [CrossRef]
- Kwok, E.; Hanson, M. Stromules and the dynamic nature of plastid morphology. J. Microsc. 2004, 214, 124–137. [Google Scholar] [CrossRef]
- Hanson, M.R.; Conklin, P.L. Stromules, functional extensions of plastids within the plant cell. Curr. Opin. Plant Biol. 2020, 58, 25–32. [Google Scholar] [CrossRef]
- Mullineaux, P.M.; Exposito-Rodriguez, M.; Laissue, P.P.; Smirnoff, N.; Park, E. Spatial chloroplast-to-nucleus signalling involving plastid–nuclear complexes and stromules. Philos. Trans. R. Soc. B 2020, 375, 20190405. [Google Scholar] [CrossRef]
- Jung, S.; Woo, J.; Park, E. Talk to your neighbors in an emergency: Stromule-mediated chloroplast-nucleus communication in plant immunity. Curr. Opin. Plant Biol. 2024, 79, 102529. [Google Scholar] [CrossRef]
- Caplan, J.L.; Kumar, A.S.; Park, E.; Padmanabhan, M.S.; Hoban, K.; Modla, S.; Czymmek, K.; Dinesh-Kumar, S.P. Chloroplast stromules function during innate immunity. Dev. Cell 2015, 34, 45–57. [Google Scholar] [CrossRef]
- Huang, J.; Taylor, J.P.; Chen, J.-G.; Uhrig, J.F.; Schnell, D.J.; Nakagawa, T.; Korth, K.L.; Jones, A.M. The plastid protein THYLAKOID FORMATION1 and the plasma membrane G-protein GPA1 interact in a novel sugar-signaling mechanism in Arabidopsis. Plant Cell 2006, 18, 1226–1238. [Google Scholar] [CrossRef]
- Michaud, M.; Jouhet, J. Lipid trafficking at membrane contact sites during plant development and stress response. Front. Plant Sci. 2019, 10, 2. [Google Scholar] [CrossRef]
- Mathur, J.; Kroeker, O.F.; Lobbezoo, M.; Mathur, N. The ER is a common mediator for the behavior and interactions of other organelles. Front. Plant Sci. 2022, 13, 846970. [Google Scholar] [CrossRef]
- Mathur, J. Organelle extensions in plant cells. Plant Physiol. 2021, 185, 593–607. [Google Scholar] [CrossRef]
- Schattat, M.H.; Barton, K.A.; Mathur, J. The myth of interconnected plastids and related phenomena. Protoplasma 2015, 252, 359–371. [Google Scholar] [CrossRef]
- Baillie, A.L.; Falz, A.-L.; Müller-Schüssele, S.J.; Sparkes, I. It started with a kiss: Monitoring organelle interactions and identifying membrane contact site components in plants. Front. Plant Sci. 2020, 11, 517. [Google Scholar] [CrossRef] [PubMed]
- Brunkard, J.O.; Runkel, A.M.; Zambryski, P. Visualizing stromule frequency with fluorescence microscopy. J. Vis. Exp. 2016, 54692. [Google Scholar] [CrossRef]
- Banas, A.; Grzyb, J.; Zglobicki, P.; Pacak, A.; Mysliwa-Kurdziel, B.; Leja, K.; Kozieradzka-Kiszkurno, M.; Klodawska, K.; Konieczny, R.; Pilarska, M.; et al. GERALT, A Cryptochrome/Photolyase Family Protein, Is Essential for Young Chloroplast Development and Function with Its Importance Decreasing in Older Plants. Plant Cell Physiol. 2025; accepted. [Google Scholar] [CrossRef]
- Ho, J.; Theg, S.M. The formation of stromules In Vitro from chloroplasts isolated from Nicotiana benthamiana. PLoS ONE 2016, 11, e0146489. [Google Scholar] [CrossRef][Green Version]
- Kunjumon, T.K.; Ghosh, P.P.; Currie, L.M.; Mathur, J. Proximity driven plastid–nucleus relationships are facilitated by tandem plastid–ER dynamics. J. Exp. Bot. 2024, 75, 6275–6294. [Google Scholar] [CrossRef] [PubMed]
- Pyke, K.A.; Howells, C.A. Plastid and stromule morphogenesis in tomato. Ann. Bot. 2002, 90, 559–566. [Google Scholar] [CrossRef]
- Kwok, E.Y.; Hanson, M.R. In vivo analysis of interactions between GFP-labeled microfilaments and plastid stromules. BMC Plant Biol. 2004, 4, 2. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barton, K.A.; Schattat, M.H.; Jakob, T.; Hause, G.; Wilhelm, C.; Mckenna, J.F.; Máthé, C.; Runions, J.; Van Damme, D.; Mathur, J. Epidermal pavement cells of Arabidopsis have chloroplasts. Am. Soc. Plant Biol. 2016, 171, 723–726. [Google Scholar]
- Erickson, J.L.; Prautsch, J.; Reynvoet, F.; Niemeyer, F.; Hause, G.; Johnston, I.G.; Schattat, M.H. Stromule geometry allows optimal spatial regulation of organelle interactions in the quasi-2D cytoplasm. Plant Cell Physiol. 2024, 65, 618–630. [Google Scholar] [CrossRef]
- Chow, W.S.; Kim, E.-H.; Horton, P.; Anderson, J.M. Granal stacking of thylakoid membranes in higher plant chloroplasts: The physicochemical forces at work and the functional consequences that ensue. Photochem. Photobiol. Sci. 2005, 4, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Itoh, R.D. Tubular extensions of plant organelles and their implications on retrograde signaling. J. Biol. Res.-Boll. Soc. Ital. Biol. Sper. 2023, 96, 11724. [Google Scholar] [CrossRef]
- Brunkard, J.O.; Runkel, A.M.; Zambryski, P.C. Chloroplasts extend stromules independently and in response to internal redox signals. Proc. Natl. Acad. Sci. USA 2015, 112, 10044–10049. [Google Scholar] [CrossRef]
- Schattat, M.H.; Klösgen, R.B. Induction of stromule formation by extracellular sucrose and glucose in epidermal leaf tissue of Arabidopsis thaliana. BMC Plant Biol. 2011, 11, 115. [Google Scholar] [CrossRef]
- Delfosse, K.; Wozny, M.R.; Barton, K.A.; Mathur, N.; Griffiths, N.; Mathur, J. Plastid envelope-localized proteins exhibit a stochastic spatiotemporal relationship to stromules. Front. Plant Sci. 2018, 9, 754. [Google Scholar] [CrossRef]
- Gray, J.C.; Hansen, M.R.; Shaw, D.J.; Graham, K.; Dale, R.; Smallman, P.; Natesan, S.K.; Newell, C.A. Plastid stromules are induced by stress treatments acting through abscisic acid. Plant J. 2012, 69, 387–398. [Google Scholar] [CrossRef]
- Vismans, G.; van der Meer, T.; Langevoort, O.; Schreuder, M.; Bouwmeester, H.; Peisker, H.; Dörman, P.; Ketelaar, T.; van der Krol, A. Low-phosphate induction of plastidal stromules is dependent on strigolactones but not on the canonical strigolactone signaling component MAX2. Plant Physiol. 2016, 172, 2235–2244. [Google Scholar] [CrossRef]
- Ge, Z.; Jing, Y.; Zhu, J.; Yang, L.-E.; Lu, S.; Deng, Y. APE1 localizes to chloroplast stromules and interacts with ATI1 in Arabidopsis. Plant Signal. Behav. 2025, 20, 2511830. [Google Scholar] [CrossRef]
- Shimahara, Y.; Kutsuna, N.; Hasezawa, S.; Kojo, K.H. Quantitative evaluation of stromule frequency at hourly intervals in Arabidopsis stomatal guard cell chloroplasts. Cytologia 2019, 84, 31–35. [Google Scholar] [CrossRef]
- Meier, N.D.; Seward, K.; Caplan, J.L.; Dinesh-Kumar, S.P. Calponin homology domain containing kinesin, KIS1, regulates chloroplast stromule formation and immunity. Sci. Adv. 2023, 9, eadi7407. [Google Scholar] [CrossRef]
- Savage, Z.; Duggan, C.; Toufexi, A.; Pandey, P.; Liang, Y.; Segretin, M.E.; Yuen, L.H.; Gaboriau, D.C.; Leary, A.Y.; Tumtas, Y.; et al. Chloroplasts alter their morphology and accumulate at the pathogen interface during infection by Phytophthora infestans. Plant J. 2021, 107, 1771–1787. [Google Scholar] [CrossRef]
- Krenz, B.; Guo, T.W.; Kleinow, T. Stromuling when stressed! Acta Soc. Bot. Pol. 2014, 83, 325–329. [Google Scholar] [CrossRef]
- Holzinger, A.; Buchner, O.; Lütz, C.; Hanson, M. Temperature-sensitive formation of chloroplast protrusions and stromules in mesophyll cells of Arabidopsis thaliana. Protoplasma 2007, 230, 23–30. [Google Scholar] [CrossRef]
- Erickson, J.L.; Schattat, M.H. Shaping plastid stromules—Principles of in vitro membrane tubulation applied in planta. Curr. Opin. Plant Biol. 2018, 46, 48–54. [Google Scholar] [CrossRef]
- Wang, X.; Li, S.; Wang, H.; Shui, W.; Hu, J. Quantitative proteomics reveal proteins enriched in tubular endoplasmic reticulum of Saccharomyces cerevisiae. eLife 2017, 6, e23816. [Google Scholar] [CrossRef] [PubMed]
- Breeze, E.; Dzimitrowicz, N.; Kriechbaumer, V.; Brooks, R.; Botchway, S.W.; Brady, J.P.; Hawes, C.; Dixon, A.M.; Schnell, J.R.; Fricker, M.D.; et al. A C-terminal amphipathic helix is necessary for the in vivo tubule-shaping function of a plant reticulon. Proc. Natl. Acad. Sci. USA 2016, 113, 10902–10907. [Google Scholar] [CrossRef] [PubMed]
- Dultz, E.; Wojtynek, M.; Medalia, O.; Onischenko, E. The nuclear pore complex: Birth, life, and death of a cellular behemoth. Cells 2022, 11, 1456. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Xiang, Y.; Hu, J. Molecular basis of Climp63-mediated ER lumen spacing. J. Cell Sci. 2023, 136, jcs260976. [Google Scholar] [CrossRef]
- Kumar, A.S.; Park, E.; Nedo, A.; Alqarni, A.; Ren, L.; Hoban, K.; Modla, S.; McDonald, J.H.; Kambhamettu, C.; Dinesh-Kumar, S.P.; et al. Stromule extension along microtubules coordinated with actin-mediated anchoring guides perinuclear chloroplast movement during innate immunity. eLife 2018, 7, e23625. [Google Scholar] [CrossRef]
- Oikawa, K.; Yamasato, A.; Kong, S.-G.; Kasahara, M.; Nakai, M.; Takahashi, F.; Ogura, Y.; Kagawa, T.; Wada, M. Chloroplast outer envelope protein CHUP1 is essential for chloroplast anchorage to the plasma membrane and chloroplast movement. Plant Physiol. 2008, 148, 829–842. [Google Scholar] [CrossRef]
- Nedo, A.O.; Liang, H.; Sriram, J.; Razzak, M.A.; Lee, J.Y.; Kambhamettu, C.; Dinesh-Kumar, S.P.; Caplan, J.L. CHUP1 restricts chloroplast movement and effector-triggered immunity in epidermal cells. New Phytol. 2024, 244, 1864–1881. [Google Scholar] [CrossRef]
- Kaiser, W.M.; Stepper, W.; Urbach, W. Photosynthesis of isolated chloroplasts and protoplasts under osmotic stress: Reversible swelling of chloroplasts by hypotonic treatment and its effect on photosynthesis. Planta 1981, 151, 375–380. [Google Scholar] [CrossRef]
- Block, M.A.; Albrieux, C.; Maréchal, E. Purification of Chloroplast Envelope, Thylakoids, and Stroma from Angiosperm Leaves. In Plastids: Methods and Protocols; Springer: Berlin/Heidelberg, Germany, 2024; pp. 137–149. [Google Scholar]
- Schuldiner, S.; Avron, M. Anion permeability of chloroplasts. Eur. J. Biochem. 1971, 19, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.J.; Reski, R. Mitochondrial dynamics and the ER: The plant perspective. Front. Cell Dev. Biol. 2015, 3, 78. [Google Scholar] [CrossRef]
- Wright, Z.J.; Bartel, B. Peroxisomes form intralumenal vesicles with roles in fatty acid catabolism and protein compartmentalization in Arabidopsis. Nat. Commun. 2020, 11, 6221. [Google Scholar] [CrossRef] [PubMed]
- Chustecki, J.M.; Gibbs, D.J.; Bassel, G.W.; Johnston, I.G. Network analysis of Arabidopsis mitochondrial dynamics reveals a resolved tradeoff between physical distribution and social connectivity. Cell Syst. 2021, 12, 419–431.e4. [Google Scholar] [CrossRef] [PubMed]
- Austin, J.R.; Frost, E.; Vidi, P.-A.; Kessler, F.; Staehelin, L.A. Plastoglobules are lipoprotein subcompartments of the chloroplast that are permanently coupled to thylakoid membranes and contain biosynthetic enzymes. Plant Cell 2006, 18, 1693–1703. [Google Scholar] [CrossRef]
- Rottet, S.; Besagni, C.; Kessler, F. The role of plastoglobules in thylakoid lipid remodeling during plant development. Biochim. Biophys. Acta (BBA)-Bioenerg. 2015, 1847, 889–899. [Google Scholar] [CrossRef]
- Ying, S. Get the Ball Rolling: Update and Perspective on the Role of Chloroplast Plastoglobule-associated Protein under Abiotic Stress. J. Exp. Bot. 2025. [Google Scholar] [CrossRef]
- Coulon, D.; Nacir, H.; Bahammou, D.; Jouhet, J.; Bessoule, J.-J.; Fouillen, L.; Bréhélin, C. Roles of plastoglobules and lipid droplets in leaf neutral lipid accumulation during senescence and nitrogen deprivation. J. Exp. Bot. 2024, 75, 6542–6562. [Google Scholar] [CrossRef]
- Block, M.A.; Douce, R.; Joyard, J.; Rolland, N. Chloroplast envelope membranes: A dynamic interface between plastids and the cytosol. Photosynth. Res. 2007, 92, 225–244. [Google Scholar] [CrossRef]
- Banaś, A.K.; Leja, K.; Zgłobicki, P.; Jedynak, P.; Kowalska, E.; Strzałka, W.; Grzyb, J.; Myśliwa-Kurdziel, B. De-etiolation is almost color blind: The study of photosynthesis awakening under blue and red light. Plant Cell Physiol. 2024, 65, 1993–2017. [Google Scholar] [CrossRef]
- Breuers, F.; Bräutigam, A.; Weber, A.P. The plastid outer envelope–a highly dynamic interface between plastid and cytoplasm. Front. Plant Sci. 2011, 2, 17899. [Google Scholar] [CrossRef]
- Jin, Z.; Wan, L.; Zhang, Y.; Li, X.; Cao, Y.; Liu, H.; Fan, S.; Cao, D.; Wang, Z.; Li, X.; et al. Structure of a TOC-TIC supercomplex spanning two chloroplast envelope membranes. Cell 2022, 185, 4788–4800.e4713. [Google Scholar] [CrossRef]
- van Wijk, K.J.; Adam, Z. Does the polyubiquitination pathway operate inside intact chloroplasts to remove proteins? Plant Cell 2024, 36, 2984–2989. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Tadini, L.; Peracchio, C.; Ferrari, R.; Pesaresi, P. GUN1, a jack-of-all-trades in chloroplast protein homeostasis and signaling. Front. Plant Sci. 2016, 7, 1427. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Yi, T.; Ha, S.-H. Diversity of plastid types and their interconversions. Front. Plant Sci. 2021, 12, 692024. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, C.; Gallage, N.J.; Hansen, C.C.; Møller, B.L.; Laursen, T. Dynamic metabolic solutions to the sessile life style of plants. Nat. Prod. Rep. 2018, 35, 1140–1155. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Du, J.; Wang, L.; Pan, Z.; Xu, Q.; Xiao, S.; Deng, X. A comprehensive analysis of chromoplast differentiation reveals complex protein changes associated with plastoglobule biogenesis and remodeling of protein systems in sweet orange flesh. Plant Physiol. 2015, 168, 1648–1665. [Google Scholar] [CrossRef]
- Eastmond, P.J.; Dennis, D.T.; Rawsthorne, S. Evidence that a malate/inorganic phosphate exchange translocator imports carbon across the leucoplast envelope for fatty acid synthesis in developing castor seed endosperm. Plant Physiol. 1997, 114, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.-C.; Li, H.-m. Identification of transit peptides that boost plastid protein import in different tissues and plant species. Nat. Plants 2025, 11, 1231–1232. [Google Scholar]
- Shah, M.; Soares, E.L.; Lima, M.L.; Pinheiro, C.B.; Soares, A.A.; Domont, G.B.; Nogueira, F.C.; Campos, F.A. Deep proteome analysis of gerontoplasts from the inner integument of developing seeds of Jatropha curcas. J. Proteom. 2016, 143, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, R.; Maekawa, M.; Kusano, M.; Tomita, K.; Kondo, H.; Nishimura, H.; Crofts, N.; Fujita, N.; Sakamoto, W. Amyloplast membrane protein SUBSTANDARD STARCH GRAIN6 controls starch grain size in rice endosperm. Plant Physiol. 2016, 170, 1445–1459. [Google Scholar] [CrossRef]
- Altamura, M.M.; Piacentini, D.; Della Rovere, F.; Fattorini, L.; Valletta, A.; Falasca, G. Transition dynamics in plastid interconversion in land plants. Plant Biosyst.-Int. J. Deal. All Asp. Plant Biol. 2024, 158, 894–924. [Google Scholar]
- Zhu, M.; Lin, J.; Ye, J.; Wang, R.; Yang, C.; Gong, J.; Liu, Y.; Deng, C.; Liu, P.; Chen, C.; et al. A comprehensive proteomic analysis of elaioplasts from citrus fruits reveals insights into elaioplast biogenesis and function. Hortic. Res. 2018, 5, 6. [Google Scholar] [CrossRef]
- John, A.; Keller, I.; Ebel, K.W.; Neuhaus, H.E. Two critical membranes: How does the chloroplast envelope affect plant acclimation properties? J. Exp. Bot. 2025, 76, 214–227. [Google Scholar] [CrossRef]
- Mamaeva, A.; Taliansky, M.; Filippova, A.; Love, A.J.; Golub, N.; Fesenko, I. The role of chloroplast protein remodeling in stress responses and shaping of the plant peptidome. New Phytol. 2020, 227, 1326–1334. [Google Scholar] [CrossRef]
- Jarvis, P.; López-Juez, E. Biogenesis and homeostasis of chloroplasts and other plastids. Nat. Rev. Mol. Cell Biol. 2013, 14, 787–802, Erratum in Nat. Rev. Mol. Cell Biol. 2014, 15, 147. [Google Scholar] [CrossRef]
- Tachibana, R.; Abe, S.; Marugami, M.; Yamagami, A.; Akema, R.; Ohashi, T.; Nishida, K.; Nosaki, S.; Miyakawa, T.; Tanokura, M.; et al. BPG4 regulates chloroplast development and homeostasis by suppressing GLK transcription factors and involving light and brassinosteroid signaling. Nat. Commun. 2024, 15, 370. [Google Scholar] [CrossRef]
- Sun, X.; Feng, P.; Xu, X.; Guo, H.; Ma, J.; Chi, W.; Lin, R.; Lu, C.; Zhang, L. A chloroplast envelope-bound PHD transcription factor mediates chloroplast signals to the nucleus. Nat. Commun. 2011, 2, 477. [Google Scholar] [CrossRef]
- Pan, T.; Liu, Y.; Hu, X.; Li, P.; Lin, C.; Tang, Y.; Tang, W.; Liu, Y.; Guo, L.; Kim, C.; et al. Stress-induced endocytosis from chloroplast inner envelope membrane is mediated by CHLOROPLAST VESICULATION but inhibited by GAPC. Cell Rep. 2023, 42, 113208. [Google Scholar] [CrossRef]
- Hwang, Y.; Han, S.; Yoo, C.Y.; Hong, L.; You, C.; Le, B.H.; Shi, H.; Zhong, S.; Hoecker, U.; Chen, X.; et al. Anterograde signaling controls plastid transcription via sigma factors separately from nuclear photosynthesis genes. Nat. Commun. 2022, 13, 7440. [Google Scholar] [CrossRef]
- Richter, A.S.; Nägele, T.; Grimm, B.; Kaufmann, K.; Schroda, M.; Leister, D.; Kleine, T. Retrograde signaling in plants: A critical review focusing on the GUN pathway and beyond. Plant Commun. 2023, 4, 100511. [Google Scholar] [CrossRef]
- Hernández-Verdeja, T.; Strand, Å. Retrograde signals navigate the path to chloroplast development. Plant Physiol. 2018, 176, 967–976. [Google Scholar] [CrossRef]
- Ding, X.; Jimenez-Gongora, T.; Krenz, B.; Lozano-Duran, R. Chloroplast clustering around the nucleus is a general response to pathogen perception in Nicotiana benthamiana. Mol. Plant Pathol. 2019, 20, 1298–1306. [Google Scholar] [CrossRef] [PubMed]
- Kwok, E.Y.; Hanson, M.R. Plastids and stromules interact with the nucleus and cell membrane in vascular plants. Plant Cell Rep. 2004, 23, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Doan, T.M.; Senthilkumar, S.; Yoo, C.Y. Mechanism of nucleus-chloroplast communication by alternative promoter usage and stromules to establish photomorphogenesis in Arabidopsis. bioRxiv 2024. [Google Scholar] [CrossRef]
- Hanson, M.R.; Sattarzadeh, A. Trafficking of proteins through plastid stromules. Plant Cell 2013, 25, 2774–2782. [Google Scholar] [CrossRef] [PubMed]
- Köhler, R.H.; Schwille, P.; Webb, W.W.; Hanson, M.R. Active protein transport through plastid tubules: Velocity quantified by fluorescence correlation spectroscopy. J. Cell Sci. 2000, 113, 3921–3930. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grzyb, J. Stromules: An Incident or Formalized Pathway for Molecules Transfer Between Organelles? Int. J. Mol. Sci. 2025, 26, 10680. https://doi.org/10.3390/ijms262110680
Grzyb J. Stromules: An Incident or Formalized Pathway for Molecules Transfer Between Organelles? International Journal of Molecular Sciences. 2025; 26(21):10680. https://doi.org/10.3390/ijms262110680
Chicago/Turabian StyleGrzyb, Joanna. 2025. "Stromules: An Incident or Formalized Pathway for Molecules Transfer Between Organelles?" International Journal of Molecular Sciences 26, no. 21: 10680. https://doi.org/10.3390/ijms262110680
APA StyleGrzyb, J. (2025). Stromules: An Incident or Formalized Pathway for Molecules Transfer Between Organelles? International Journal of Molecular Sciences, 26(21), 10680. https://doi.org/10.3390/ijms262110680