The Role of MICA/B Molecules and the NKG2D Receptor in the Interaction Between NK-92 Cells and JEG-3 Cells
Abstract
1. Introduction
2. Results
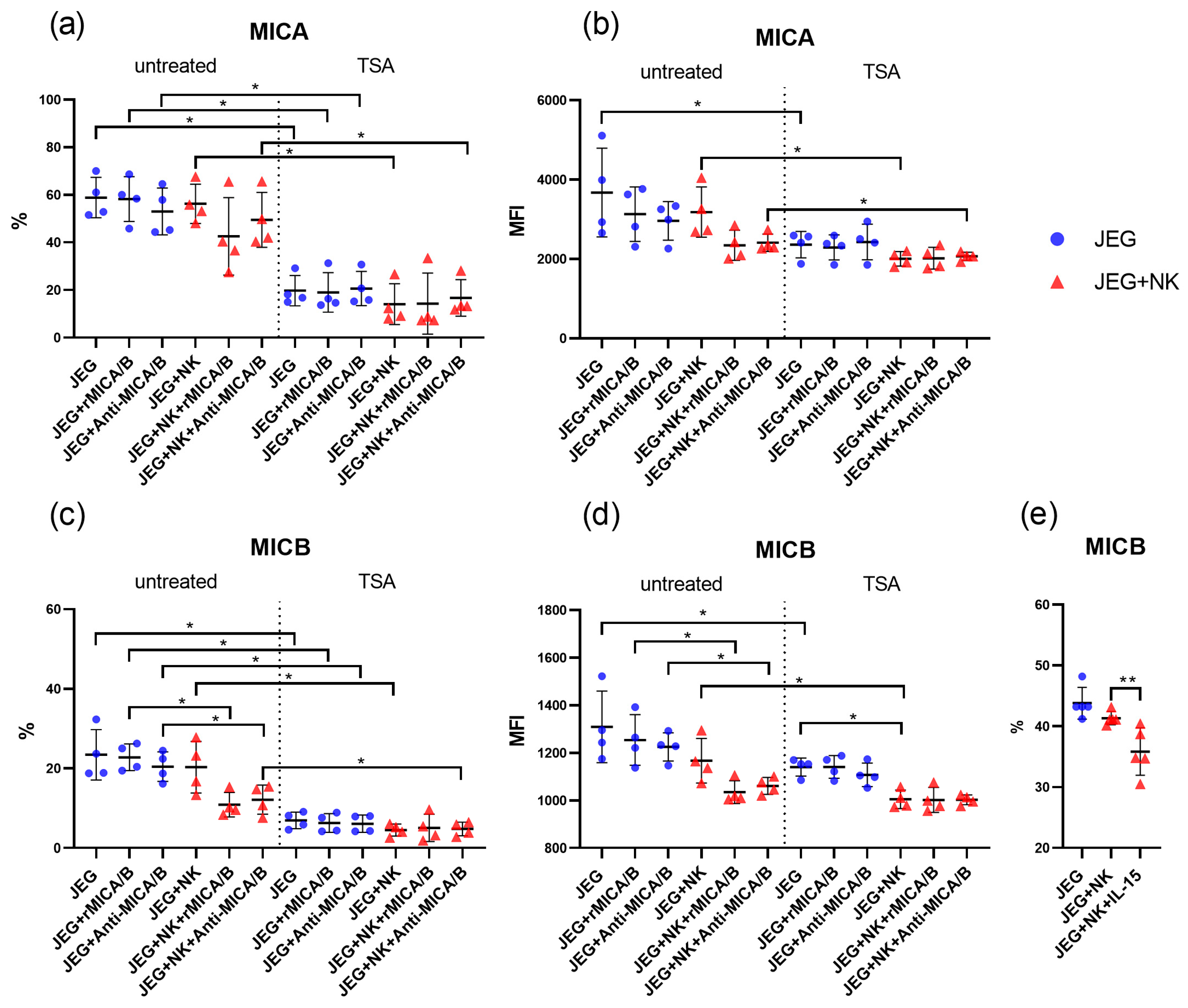
2.1. TSA as a Potential Cause for Decline in MIC-Expressing Cells
2.2. Downregulation of MICB in JEG-3 Cells by NK Cells in the Presence of rMICA/B or Anti-MICA/B
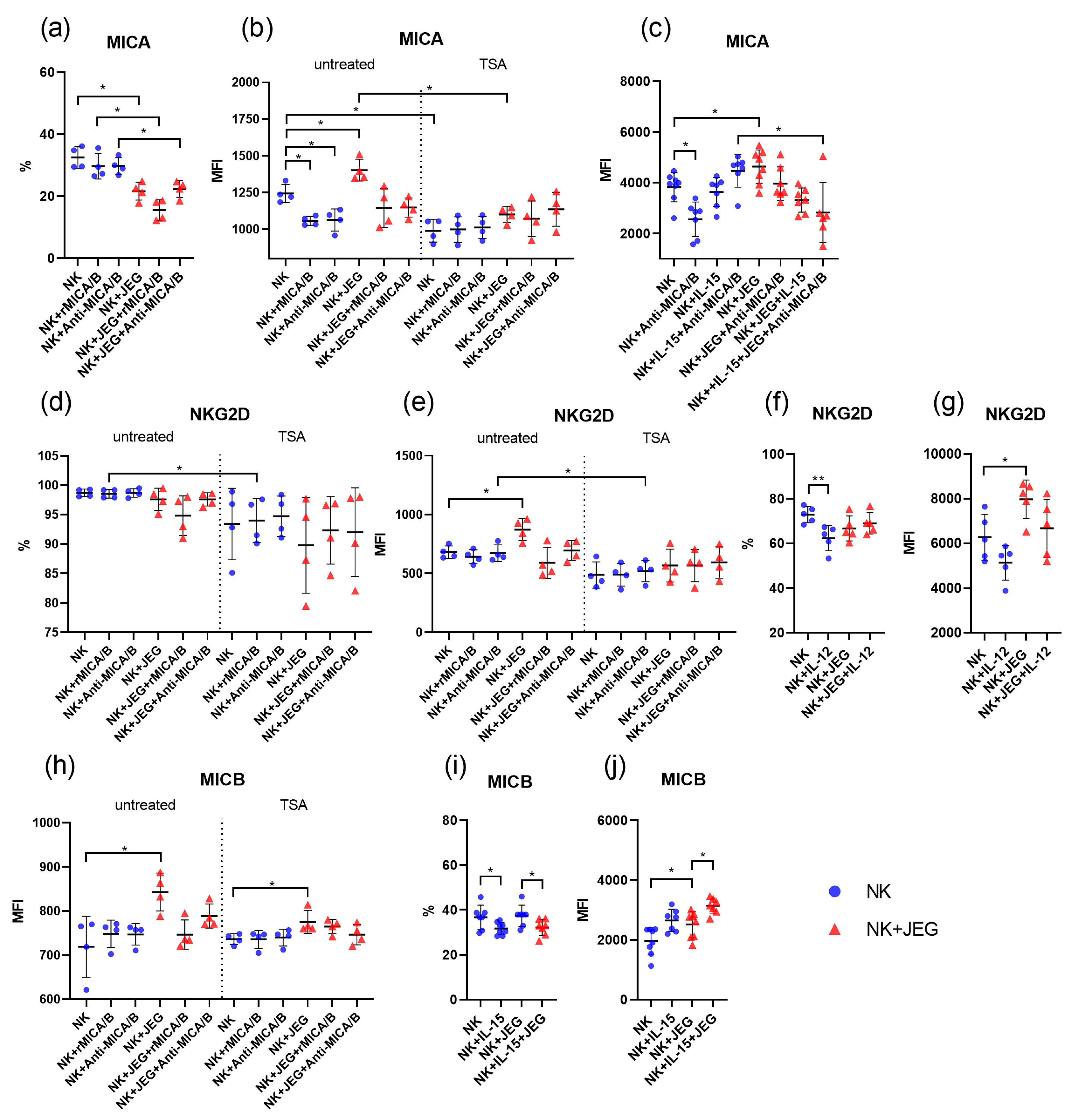
2.3. The Effect of TSA on MICA Expression and NK Cell Phenotype Modulation by IL-12, rMICA/B, and Anti-MICA/B
2.4. JEG-3 Cells’ Effects on NK Phenotype
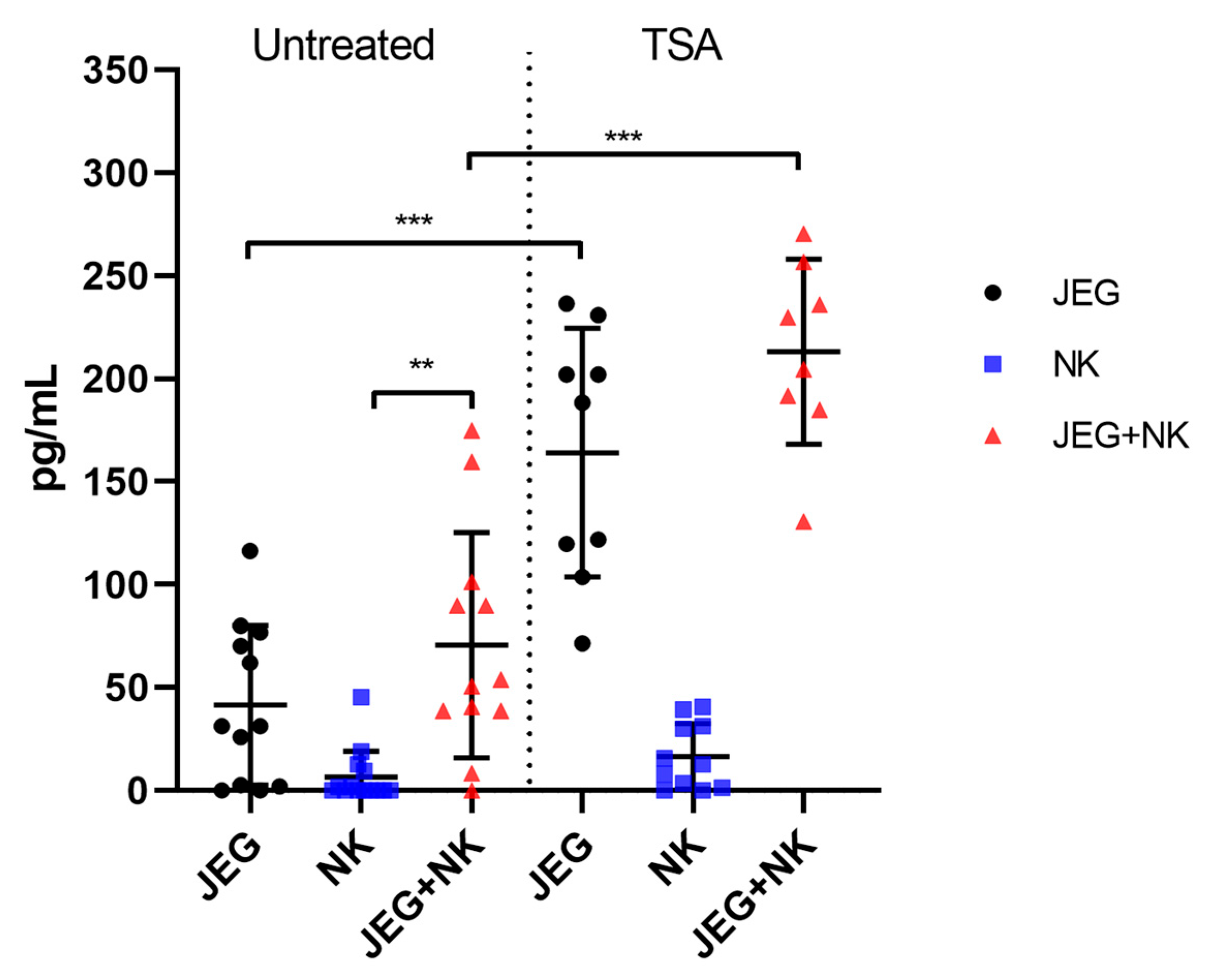
2.5. sMICB Secretion by JEG-3 Cells Stimulated with TSA
2.6. TSA Enhanced the Death Rates of K-562 and JEG-3 Cells, but This Effect Was Not Observed in the Presence of NK Cells or PBMCs
2.7. Enhanced NK Cell Cytotoxicity Against JEG-3 Cells with rMICA/B or Anti-MICA/B
2.8. IL-12-Activated NK/PBMC Cytotoxicity and JEG-3 Survival in the Presence of Anti-MICA/B
2.9. TSA-Mediated Protection of Target Cells from IL-12-Activated NK/PBMC Cytotoxicity in the Presence of rMICA/B
3. Discussion
4. Materials and Methods
4.1. Cell Lines
4.2. Inductors
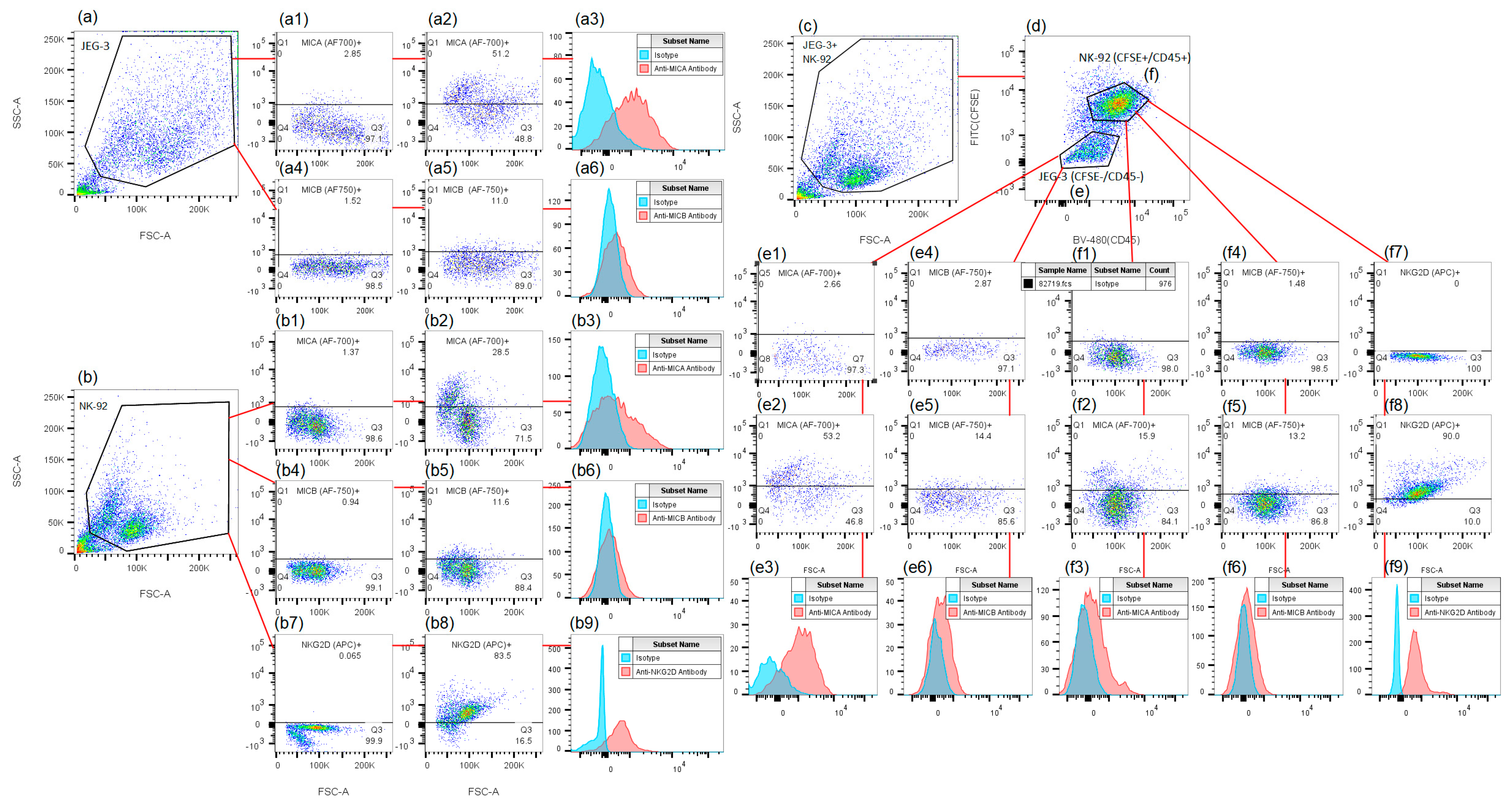
4.3. Assessment of the Phenotype of JEG-3 and NK-92 Cells Following Mono- and Co-Culture in the Presence of Inducers
4.4. Assessment of the Cytotoxic Activity of NK-92 Cells Against JEG-3 and K-562 Cells and PBMCs Against K-562 Cells
4.5. Enzyme-Linked Immunosorbent Assay
4.6. Quantitative RT-qPCR
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| NK cells | natural killer cells |
| MICs | MICA/B proteins |
| TSA | trichostatin A |
| sMICs | soluble MICA/B proteins |
| HDACi | histone deacetylase inhibitor |
| rMICA/B | recombinant MICA/B proteins |
| Anti-MICA/B | antibodies to MICA/B proteins |
| PBMCs | peripheral blood mononuclear cells |
References
- Venkataraman, G.M.; Suciu, D.; Groh, V.; Boss, J.M.; Spies, T. Promoter region architecture and transcriptional regulation of the genes for the MHC class I-related chain A and B ligands of NKG2D. J. Immunol. 2007, 178, 961–969. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, N.; Yu, Y.; Zhou, L.; Niu, C.; Liu, Y.; Tian, H.; Lv, Z.; Han, F.; Cui, J. Prognostic value of MICA/B in cancers: A systematic review and meta-analysis. Oncotarget 2017, 8, 96384–96395. [Google Scholar] [CrossRef]
- Torn, C.; Gupta, M.; Sanjeevi, C.B.; Aberg, A.; Frid, A.; Landin-Olsson, M. Different HLA-DR-DQ and MHC class I chain-related gene A (MICA) genotypes in autoimmune and nonautoimmune gestational diabetes in a Swedish population. Hum. Immunol. 2004, 65, 1443–1450. [Google Scholar] [CrossRef]
- Vinnars, M.T.; Bjork, E.; Nagaev, I.; Ottander, U.; Bremme, K.; Holmlund, U.; Sverremark-Ekstrom, E.; Mincheva-Nilsson, L. Enhanced Th1 and inflammatory mRNA responses upregulate NK cell cytotoxicity and NKG2D ligand expression in human pre-eclamptic placenta and target it for NK cell attack. Am. J. Reprod. Immunol. 2018, 80, e12969. [Google Scholar] [CrossRef]
- Apps, R.; Gardner, L.; Traherne, J.; Male, V.; Moffett, A. Natural-killer cell ligands at the maternal-fetal interface: UL-16 binding proteins, MHC class-I chain related molecules, HLA-F and CD48. Hum. Reprod. 2008, 23, 2535–2548. [Google Scholar] [CrossRef]
- Bauer, S.; Groh, V.; Wu, J.; Steinle, A.; Phillips, J.H.; Lanier, L.L.; Spies, T. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 1999, 285, 727–729. [Google Scholar] [CrossRef]
- Ogasawara, K.; Lanier, L.L. NKG2D in NK and T cell-mediated immunity. J. Clin. Immunol. 2005, 25, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Dhar, P.; Wu, J.D. NKG2D and its ligands in cancer. Curr. Opin. Immunol. 2018, 51, 55–61. [Google Scholar] [CrossRef]
- Sharma, N.; Trinidad, C.V.; Trembath, A.P.; Markiewicz, M.A. NKG2D Signaling between Human NK Cells Enhances TACE-Mediated TNF-alpha Release. J. Immunol. 2017, 199, 2865–2872. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Morris, D.L.; Willcox, B.E.; Steinle, A.; Spies, T.; Strong, R.K. Complex structure of the activating immunoreceptor NKG2D and its MHC class I-like ligand MICA. Nat. Immunol. 2001, 2, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.B.; Cella, M.; Giurisato, E.; Fujikawa, K.; Miletic, A.V.; Kloeppel, T.; Brim, K.; Takai, T.; Shaw, A.S.; Colonna, M.; et al. Vav1 controls DAP10-mediated natural cytotoxicity by regulating actin and microtubule dynamics. J. Immunol. 2006, 177, 2349–2355. [Google Scholar] [CrossRef]
- Watzl, C.; Urlaub, D. Molecular mechanisms of natural killer cell regulation. Front. Biosci. 2012, 17, 1418–1432. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hedlund, M.; Stenqvist, A.C.; Nagaeva, O.; Kjellberg, L.; Wulff, M.; Baranov, V.; Mincheva-Nilsson, L. Human placenta expresses and secretes NKG2D ligands via exosomes that down-modulate the cognate receptor expression: Evidence for immunosuppressive function. J. Immunol. 2009, 183, 340–351. [Google Scholar] [CrossRef]
- Ashiru, O.; Boutet, P.; Fernandez-Messina, L.; Aguera-Gonzalez, S.; Skepper, J.N.; Vales-Gomez, M.; Reyburn, H.T. Natural killer cell cytotoxicity is suppressed by exposure to the human NKG2D ligand MICA*008 that is shed by tumor cells in exosomes. Cancer Res. 2010, 70, 481–489. [Google Scholar] [CrossRef]
- Boutet, P.; Aguera-Gonzalez, S.; Atkinson, S.; Pennington, C.J.; Edwards, D.R.; Murphy, G.; Reyburn, H.T.; Vales-Gomez, M. Cutting edge: The metalloproteinase ADAM17/TNF-alpha-converting enzyme regulates proteolytic shedding of the MHC class I-related chain B protein. J. Immunol. 2009, 182, 49–53. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Y.; Zeng, X.; Ma, X.J.; Zhao, Y.; Qiao, J.; Cao, B.; Li, Y.X.; Ji, L.; Wang, Y.L. Self-control of HGF regulation on human trophoblast cell invasion via enhancing c-Met receptor shedding by ADAM10 and ADAM17. J. Clin. Endocrinol. Metab. 2012, 97, E1390–E1401. [Google Scholar] [CrossRef]
- Groh, V.; Wu, J.; Yee, C.; Spies, T. Tumour-derived soluble MIC ligands impair expression of NKG2D and T-cell activation. Nature 2002, 419, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Ferrari de Andrade, L.; Tay, R.E.; Pan, D.; Luoma, A.M.; Ito, Y.; Badrinath, S.; Tsoucas, D.; Franz, B.; May, K.F., Jr.; Harvey, C.J.; et al. Antibody-mediated inhibition of MICA and MICB shedding promotes NK cell-driven tumor immunity. Science 2018, 359, 1537–1542. [Google Scholar] [CrossRef]
- Kato, N.; Tanaka, J.; Sugita, J.; Toubai, T.; Miura, Y.; Ibata, M.; Syono, Y.; Ota, S.; Kondo, T.; Asaka, M.; et al. Regulation of the expression of MHC class I-related chain A, B (MICA, MICB) via chromatin remodeling and its impact on the susceptibility of leukemic cells to the cytotoxicity of NKG2D-expressing cells. Leukemia 2007, 21, 2103–2108. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Lavender, H.; Soilleux, E.J.; O’Callaghan, C.A. NF-kappaB regulates MICA gene transcription in endothelial cell through a genetically inhibitable control site. J. Biol. Chem. 2012, 287, 4299–4310. [Google Scholar] [CrossRef]
- Langhans, B.; Ahrendt, M.; Nattermann, J.; Sauerbruch, T.; Spengler, U. Comparative study of NK cell-mediated cytotoxicity using radioactive and flow cytometric cytotoxicity assays. J. Immunol. Methods 2005, 306, 161–168. [Google Scholar] [CrossRef]
- Jurisic, V.; Spuzic, I.; Konjevic, G. A comparison of the NK cell cytotoxicity with effects of TNF-alpha against K-562 cells, determined by LDH release assay. Cancer Lett. 1999, 138, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Drvar, V.; Curko-Cofek, B.; Karleusa, L.; Aralica, M.; Rogoznica, M.; Kehler, T.; Legovic, D.; Rukavina, D.; Laskarin, G. Granulysin expression and granulysin-mediated apoptosis in the peripheral blood of osteoarthritis patients. Biomed. Rep. 2022, 16, 44. [Google Scholar] [CrossRef]
- Huang, S.Y.; Chiang, C.H.; Chen, F.P.; Yu, C.L. The alteration of placental-derived soluble MHC class I chain-related protein A and B during pregnancy. Acta Obstet. Gynecol. Scand. 2011, 90, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Marlin, R.; Duriez, M.; Berkane, N.; de Truchis, C.; Madec, Y.; Rey-Cuille, M.A.; Cummings, J.S.; Cannou, C.; Quillay, H.; Barre-Sinoussi, F.; et al. Dynamic shift from CD85j/ILT-2 to NKG2D NK receptor expression pattern on human decidual NK during the first trimester of pregnancy. PLoS ONE 2012, 7, e30017. [Google Scholar] [CrossRef]
- Molinero, L.L.; Domaica, C.I.; Fuertes, M.B.; Girart, M.V.; Rossi, L.E.; Zwirner, N.W. Intracellular expression of MICA in activated CD4 T lymphocytes and protection from NK cell-mediated MICA-dependent cytotoxicity. Hum. Immunol. 2006, 67, 170–182. [Google Scholar] [CrossRef]
- Molinero, L.L.; Fuertes, M.B.; Rabinovich, G.A.; Fainboim, L.; Zwirner, N.W. Activation-induced expression of MICA on T lymphocytes involves engagement of CD3 and CD28. J. Leukoc. Biol. 2002, 71, 791–797. [Google Scholar] [CrossRef]
- Molinero, L.L.; Fuertes, M.B.; Girart, M.V.; Fainboim, L.; Rabinovich, G.A.; Costas, M.A.; Zwirner, N.W. NF-kappa B regulates expression of the MHC class I-related chain A gene in activated T lymphocytes. J. Immunol. 2004, 173, 5583–5590. [Google Scholar] [CrossRef]
- Stolbovaya, A.Y.; Pinevich, A.A.; Gryazeva, I.V.; Krutetskaya, I.Y.; Zharinov, G.M.; Morozova, M.A.; Kneev, A.Y.; Terekhina, L.A.; Ishchuk, S.A.; Shashkova, O.A.; et al. Detection and Quantification of Polymorphic MICA and MICB Molecules in Immunoassays: Initial Insights. HLA 2025, 105, e70039. [Google Scholar] [CrossRef] [PubMed]
- Klumkrathok, K.; Jumnainsong, A.; Leelayuwat, C. Allelic MHC class I chain related B (MICB) molecules affect the binding to the human cytomegalovirus (HCMV) unique long 16 (UL16) protein: Implications for immune surveillance. J. Microbiol. 2013, 51, 241–246. [Google Scholar] [CrossRef]
- Carapito, R.; Bahram, S. Genetics, genomics, and evolutionary biology of NKG2D ligands. Immunol. Rev. 2015, 267, 88–116. [Google Scholar] [CrossRef]
- Du, C.; Bevers, J., 3rd; Cook, R.; Lombana, T.N.; Rajasekaran, K.; Matsumoto, M.; Spiess, C.; Kim, J.M.; Ye, Z. MICA immune complex formed with alpha 3 domain-specific antibody activates human NK cells in a Fc-dependent manner. J. Immunother. Cancer 2019, 7, 207. [Google Scholar] [CrossRef]
- Gong, J.H.; Maki, G.; Klingemann, H.G. Characterization of a human cell line (NK-92) with phenotypical and functional characteristics of activated natural killer cells. Leukemia 1994, 8, 652–658. [Google Scholar]
- Wu, J.D.; Higgins, L.M.; Steinle, A.; Cosman, D.; Haugk, K.; Plymate, S.R. Prevalent expression of the immunostimulatory MHC class I chain-related molecule is counteracted by shedding in prostate cancer. J. Clin. Investig. 2004, 114, 560–568. [Google Scholar] [CrossRef]
- Dhar, P.; Basher, F.; Ji, Z.; Huang, L.; Qin, S.; Wainwright, D.A.; Robinson, J.; Hagler, S.; Zhou, J.; MacKay, S.; et al. Tumor-derived NKG2D ligand sMIC reprograms NK cells to an inflammatory phenotype through CBM signalosome activation. Commun. Biol. 2021, 4, 905. [Google Scholar] [CrossRef] [PubMed]
- Hakam, M.S.; Miranda-Sayago, J.M.; Hayrabedyan, S.; Todorova, K.; Spencer, P.S.; Jabeen, A.; Barnea, E.R.; Fernandez, N. Preimplantation Factor (PIF) Promotes HLA-G, -E, -F, -C Expression in JEG-3 Choriocarcinoma Cells and Endogenous Progesterone Activity. Cell Physiol. Biochem. 2017, 43, 2277–2296. [Google Scholar] [CrossRef] [PubMed]
- Vacca, P.; Cantoni, C.; Prato, C.; Fulcheri, E.; Moretta, A.; Moretta, L.; Mingari, M.C. Regulatory role of NKp44, NKp46, DNAM-1 and NKG2D receptors in the interaction between NK cells and trophoblast cells. Evidence for divergent functional profiles of decidual versus peripheral NK cells. Int. Immunol. 2008, 20, 1395–1405. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, J.; Niu, J.; Zhou, Z.; Zhang, J.; Tian, Z. Interleukin-12 improves cytotoxicity of natural killer cells via upregulated expression of NKG2D. Hum. Immunol. 2008, 69, 490–500. [Google Scholar] [CrossRef]
- Kohler, P.O.; Bridson, W.E. Isolation of hormone-producing clonal lines of human choriocarcinoma. J. Clin. Endocrinol. Metab. 1971, 32, 683–687. [Google Scholar] [CrossRef]
- Lozzio, B.B.; Lozzio, C.B. Properties and usefulness of the original K-562 human myelogenous leukemia cell line. Leuk. Res. 1979, 3, 363–370. [Google Scholar] [CrossRef]
- Shin, S.; Kim, M.; Lee, S.J.; Park, K.S.; Lee, C.H. Trichostatin A Sensitizes Hepatocellular Carcinoma Cells to Enhanced NK Cell-mediated Killing by Regulating Immune-related Genes. Cancer Genom. Proteom. 2017, 14, 349–362. [Google Scholar] [CrossRef]
- Flieger, D.; Gruber, R.; Schlimok, G.; Reiter, C.; Pantel, K.; Riethmuller, G. A novel non-radioactive cellular cytotoxicity test based on the differential assessment of living and killed target and effector cells. J. Immunol. Methods 1995, 180, 1–13. [Google Scholar] [CrossRef] [PubMed]


| Marker | Fluorochrome | Cat. No | Figure | Supplementary Figure |
|---|---|---|---|---|
| CD45 | BV480 | BD, Franklin Lakes, NJ, USA, 566115 | — | — |
| NKG2D | APC | BD, Franklin Lakes, NJ, USA, 558071 | Figure 2d,e | Figure S3(a1,a2) |
| NKG2D | APC | Thermo Fisher, Waltham, Ma, USA 17-5878-81 | Figure 2f,g | Figure S3(d1,d2) |
| MICA | AF700 | R&D, Minneapolis, MN, USA, FAB1300N | Figure 1a,b and Figure 2a,b | Figures S2(a1,a2) and S3(b1,b2) |
| MICA | RB705 | BD, Franklin Lakes, NJ, USA, 757415 | Figure 2c | Figures S2(c1,c2) and S3(e1,e2) |
| MICB | AF750 | R&D, Minneapolis, MN, USA, FAB1599S | Figure 1c,d and Figure 2h | Figures S2(b1,b2) and S3(c1,c2) |
| MICB | AF488 | R&D, Minneapolis, MN, USA, FAB1599G | Figure 1e and Figure 2i,j | Figures S2(d1,d2) and S3(f1,f2) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tyshchuk, E.; Denisova, E.; Grebenkina, P.; Pereviazkina, M.; Stolbovaya, A.; Smirnov, I.; Shashkova, O.; Gryazeva, I.; Terekhina, L.; Sokolov, D. The Role of MICA/B Molecules and the NKG2D Receptor in the Interaction Between NK-92 Cells and JEG-3 Cells. Int. J. Mol. Sci. 2025, 26, 8400. https://doi.org/10.3390/ijms26178400
Tyshchuk E, Denisova E, Grebenkina P, Pereviazkina M, Stolbovaya A, Smirnov I, Shashkova O, Gryazeva I, Terekhina L, Sokolov D. The Role of MICA/B Molecules and the NKG2D Receptor in the Interaction Between NK-92 Cells and JEG-3 Cells. International Journal of Molecular Sciences. 2025; 26(17):8400. https://doi.org/10.3390/ijms26178400
Chicago/Turabian StyleTyshchuk, Elizaveta, Elizaveta Denisova, Polina Grebenkina, Marina Pereviazkina, Anastasia Stolbovaya, Ilya Smirnov, Olga Shashkova, Irina Gryazeva, Lidiya Terekhina, and Dmitry Sokolov. 2025. "The Role of MICA/B Molecules and the NKG2D Receptor in the Interaction Between NK-92 Cells and JEG-3 Cells" International Journal of Molecular Sciences 26, no. 17: 8400. https://doi.org/10.3390/ijms26178400
APA StyleTyshchuk, E., Denisova, E., Grebenkina, P., Pereviazkina, M., Stolbovaya, A., Smirnov, I., Shashkova, O., Gryazeva, I., Terekhina, L., & Sokolov, D. (2025). The Role of MICA/B Molecules and the NKG2D Receptor in the Interaction Between NK-92 Cells and JEG-3 Cells. International Journal of Molecular Sciences, 26(17), 8400. https://doi.org/10.3390/ijms26178400







