First In Vitro Characterization of Salinomycinic Acid-Containing Two-Line Ferrihydrite Composites with Pronounced Antitumor Activity as MRI Contrast Agents
Abstract
1. Introduction
2. Results
2.1. ICP-MS Analysis
2.2. Dynamic Light Scattering Analysis (DLS)
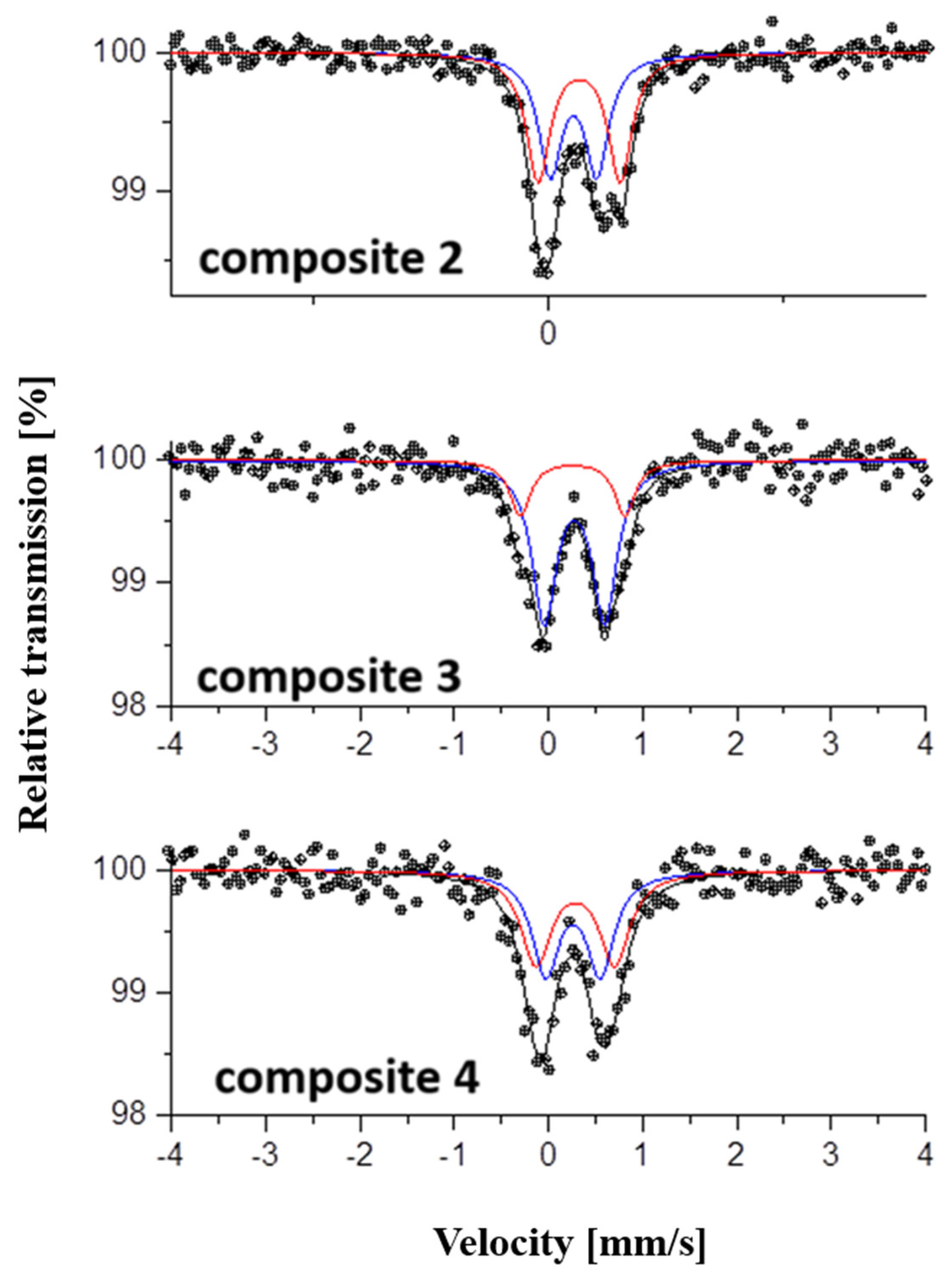
2.3. Mössbauer Spectroscopy
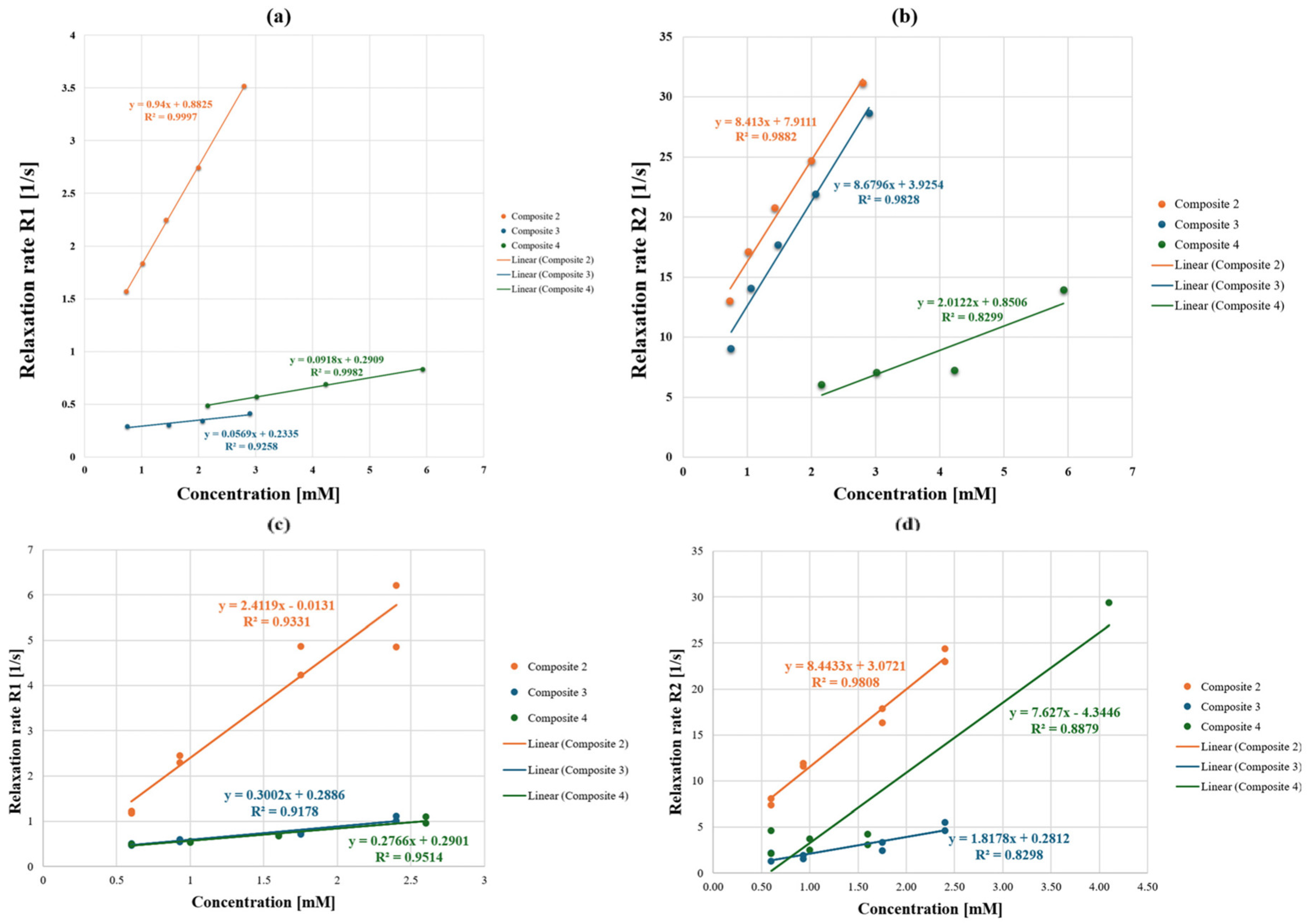
2.4. In Vitro MRI Relaxivity
2.5. Cytotoxicity Studies
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Synthesis
4.3. Determination of the Fe Content by Inductively Coupled Plasma Mass Spectrometry (ICP-MS)
4.4. Dynamic Light Scattering (DLS)
4.5. Mössbauer Spectroscopy
4.6. In Vitro Relaxivity Measurements
4.7. Cytotoxicity Tests
4.7.1. Cell Culture
4.7.2. MTT Assay
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATR-FTIR | Reflectance-Fourier transform spectroscopy |
| CAs | Contrast agents |
| CSC | Cancer stem cells |
| EPR | Paramagnetic resonance spectroscopy |
| ICP-MS | Inductively coupled plasma mass spectrometry |
| MRI | Magnetic resonance imaging |
| PP | Polypropylene |
References
- Uzal-Varela, R.; Lucio-Martínez, F.; Nucera, A.; Botta, M.; Esteban-Gómez, D.; Valencia, L.; Rodríguez-Rodríguez, A.; Platas-Iglesias, C. A systematic investigation of the NMR relaxation properties of Fe(III)-EDTA derivatives and their potential as MRI contrast agents. Inorg. Chem. Front. 2023, 10, 1633–1649. [Google Scholar] [CrossRef]
- Cineus, R.; Abozeid, S.M.; Sokolow, G.E.; Spernyak, J.A.; Morrow, J.R. Fe(III) T1 MRI Probes Containing Phenolate or Hydroxypyridine-Appended Triamine Chelates and a Coordination Site for Bound Water. Inorg. Chem. 2023, 62, 16513–16522. [Google Scholar] [CrossRef]
- Chen, S.; An, L.; Yang, S. Low-Molecular-Weight Fe(III) Complexes for MRI Contrast Agents. Molecules 2022, 27, 4573. [Google Scholar] [CrossRef] [PubMed]
- Kras, E.A.; Cineus, R.; Crawley, M.R.; Morrow, J.R. Macrocyclic complexes of Fe(III) with mixed hydroxypropyl and phenolate or amide pendants as T1 MRI probes. Dalton Trans. 2024, 53, 4154–4164. [Google Scholar] [CrossRef] [PubMed]
- Coimbra, S.; Rocha, S.; Sousa, N.R.; Catarino, C.; Belo, L.; Bronze-da-Rocha, E.; Valente, M.J.; Santos-Silva, A. Toxicity Mechanisms of Gadolinium and Gadolinium-Based Contrast Agents-A Review. Int. J. Mol. Sci. 2024, 25, 4071. [Google Scholar] [CrossRef]
- Trapasso, G.; Chiesa, S.; Freitas, R.; Pereira, E. What do we know about the ecotoxicological implications of the rare earth element gadolinium in aquatic ecosystems? Sci. Total Environ. 2021, 781, 146273. [Google Scholar] [CrossRef]
- Snyder, E.M.; Asik, D.; Abozeid, S.M.; Burgio, A.; Bateman, G.; Turowski, S.G.; Spernyak, J.A.; Morrow, J.R. A Class of FeIII Macrocyclic Complexes with Alcohol Donor Groups as Effective T1 MRI Contrast Agents. Angew. Chem. Int. Ed. 2020, 59, 2414–2419. [Google Scholar] [CrossRef]
- Schneppensieper, T.; Seibig, S.; Zahl, A.; Tregloan, P.; van Eldik, R. Influence of chelate effects on the water-exchange mechanism of polyaminecarboxylate complexes of iron(III). Inorg. Chem. 2001, 40, 3670–3676. [Google Scholar] [CrossRef]
- Boehm-Sturm, P.; Haeckel, A.; Hauptmann, R.; Mueller, S.; Kuhl, C.K.; Schellenberger, E.A. Low-Molecular-Weight Iron Chelates May Be an Alternative to Gadolinium-based Contrast Agents for T1-weighted Contrast-enhanced MR Imaging. Radiology 2018, 286, 537–546. [Google Scholar] [CrossRef]
- Wang, H.; Jordan, V.C.; Ramsay, I.A.; Sojoodi, M.; Fuchs, B.C.; Tanabe, K.K.; Caravan, P.; Gale, E.M. Molecular Magnetic Resonance Imaging Using a Redox-Active Iron Complex. J. Am. Chem. Soc. 2019, 141, 5916–5925. [Google Scholar] [CrossRef]
- Wang, H.; Cleary, M.B.; Lewis, L.C.; Bacon, J.W.; Caravan, P.; Shafaat, H.S.; Gale, E.M. Enzyme Control Over Ferric Iron Magnetostructural Properties. Angew. Chem. Int. Ed. 2022, 61, e202114019. [Google Scholar] [CrossRef]
- Wang, H.; Wong, A.; Lewis, L.C.; Nemeth, G.R.; Jordan, V.C.; Bacon, J.W.; Caravan, P.; Shafaat, H.S.; Gale, E.M. Rational Ligand Design Enables pH Control over Aqueous Iron Magnetostructural Dynamics and Relaxometric Properties. Inorg. Chem. 2020, 59, 17712–17721. [Google Scholar] [CrossRef]
- Baranyai, Z.; Carniato, F.; Nucera, A.; Horváth, D.; Tei, L.; Platas-Iglesias, C.; Botta, M. Defining the conditions for the development of the emerging class of FeIII-based MRI contrast agents. Chem. Sci. 2021, 12, 11138–11145. [Google Scholar] [CrossRef]
- Wang, R.; An, L.; He, J.; Li, M.; Jiao, J.; Yang, S. A class of water-soluble Fe(III) coordination complexes as T1-weighted MRI contrast agents. J. Mater. Chem. B 2021, 9, 1787–1791. [Google Scholar] [CrossRef]
- Bales, B.C.; Grimmond, B.; Johnson, B.F.; Luttrell, M.T.; Meyer, D.E.; Polyanskaya, T.; Rishel, M.J.; Roberts, J. Fe-HBED Analogs: A Promising Class of Iron-Chelate Contrast Agents for Magnetic Resonance Imaging. Contrast Media Mol. Imaging 2019, 2019, 8356931. [Google Scholar] [CrossRef]
- Kras, E.A.; Snyder, E.M.; Sokolow, G.E.; Morrow, J.R. Distinct Coordination Chemistry of Fe(III)-Based MRI Probes. Acc. Chem. Res. 2022, 55, 1435–1444. [Google Scholar] [CrossRef]
- Petkov, N.; Tadjer, A.; Encheva, E.; Cherkezova-Zheleva, Z.; Paneva, D.; Stoyanova, R.; Kukeva, R.; Dorkov, P.; Pantcheva, I. Experimental and DFT Study of Monensinate and Salinomycinate Complexes Containing {Fe3(µ3–O)}7+ Core. Molecules 2024, 29, 364. [Google Scholar] [CrossRef]
- Ivanova, J.; Kukeva, R.; Stoyanova, R.; Zhivkova, T.; Abudalleh, A.; Dyakova, L.; Alexandrova, R.; Pashkunova-Martic, I.; Theiner, J.; Dorkov, P.; et al. New Iron(III)-Containing Composite of Salinomycinic Acid with Antitumor Activity—Synthesis and Characterization. Inorganics 2024, 12, 206. [Google Scholar] [CrossRef]
- Gupta, P.B.; Onder, T.T.; Jiang, G.; Tao, K.; Kuperwasser, C.; Weinberg, R.A.; Lander, E.S. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 2009, 138, 645–659. [Google Scholar] [CrossRef]
- Mao, Z.; Wu, Y.; Zhou, J.; Xing, C. Salinomycin reduces epithelial-mesenchymal transition-mediated multidrug resistance by modifying long noncoding RNA HOTTIP expression in gastric cancer cells. Anti-Cancer Drugs 2019, 30, 892–899. [Google Scholar] [CrossRef]
- Serttas, R.; Erdogan, S. Pretreatment of prostate cancer cells with salinomycin and Wnt inhibitor increases the efficacy of cabazitaxel by inducing apoptosis and decreasing cancer stem cells. Med. Oncol. 2023, 40, 194. [Google Scholar] [CrossRef]
- Klose, J.; Trefz, S.; Wagner, T.; Steffen, L.; Preißendörfer Charrier, A.; Radhakrishnan, P.; Volz, C.; Schmidt, T.; Ulrich, A.; Dieter, S.M.; et al. Salinomycin: Anti-tumor activity in a pre-clinical colorectal cancer model. PLoS ONE 2019, 14, e0211916. [Google Scholar] [CrossRef]
- Zhang, X.; Lou, Y.; Zheng, X.; Wang, H.; Sun, J.; Dong, Q.; Han, B. Wnt blockers inhibit the proliferation of lung cancer stem cells. Drug Des. Devel. Ther. 2015, 9, 2399–2407. [Google Scholar] [CrossRef]
- Roulston, G.D.; Burt, C.L.; Kettyle, L.M.; Matchett, K.B.; Keenan, H.L.; Mulgrew, N.M.; Ramsey, J.M.; Dougan, C.; McKiernan, J.; Grishagin, I.V.; et al. Low-dose salinomycin induces anti-leukemic responses in AML and MLL. Oncotarget 2016, 7, 73448–73461. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, J.; Pantcheva, I.N.; Zhorova, R.; Momekov, G.; Simova, S.; Stoyanova, R.; Zhecheva, E.; Ivanova, S.; Mitewa, M. Synthesis, spectral properties, antibacterial and antitumor activity of salinomycin complexes with Co(II), Ni(II), Cu(II) and Zn(II) transition metal ions. J. Chem. Chem. Eng. 2012, 6, 551–562. [Google Scholar]
- Momekova, D.; Momekov, G.; Ivanova, J.; Pantcheva, I.; Drakalska, E.; Stoyanov, N.; Guenova, M.; Michova, A.; Balashev, K.; Arpadjan, S.; et al. Sterically stabilized liposomes as a platform for salinomycin metal coordination compounds: Physicochemical characterization and in vitro evaluation. J. Drug Deliv. Sci. Technol. 2013, 23, 215–223. [Google Scholar] [CrossRef]
- Pantcheva, I.; Petkov, N.; Simova, S.; Zhorova, R.; Dorkov, P. 4 Alkaline-earth metal(II) complexes of salinomycin—Spectral properties and antibacterial activity. In Volume 2 Biochemical and Environmental Applications; Ramasami, P., Ed.; De Gruyter: Berlin, Germany; Boston, MA, USA, 2022; pp. 65–78. [Google Scholar] [CrossRef]
- Pashkunova-Martic, I.; Kukeva, R.; Stoyanova, R.; Pantcheva, I.; Dorkov, P.; Friske, J.; Hejl, M.; Jakupec, M.; Hohagen, M.; Legin, A.; et al. Novel Salinomycin-Based Paramagnetic Complexes—First Evaluation of Their Potential Theranostic Properties. Pharmaceutics 2022, 14, 2319. [Google Scholar] [CrossRef]
- Shi, Q.; Li, Y.; Bo, S.; Li, X.; Zhao, P.; Liu, Q.; Yang, Z.; Cong, H.; Deng, H.; Chen, M.; et al. Discovery of a (19)F MRI sensitive salinomycin derivative with high cytotoxicity towards cancer cells. Chem. Commun. 2016, 52, 5136–5139. [Google Scholar] [CrossRef]
- Sokolov, I.L.; Cherkasov, V.R.; Vasilyeva, A.V.; Bragina, V.A.; Nikitin, M.P. Paramagnetic colloidal ferrihydrite nanoparticles for MRI contrasting. Colloids Surf. A Physicochem. Eng. Asp. 2018, 539, 46–52. [Google Scholar] [CrossRef]
- Donglin, L.; Jiaojiao, L.; Chengbin, W.; Lu, A.; Jiaomin, L.; Qiwei, T.; Shiping, Y. Ultrasmall Fe@Fe3O4 nanoparticles as T1–T2 dual-mode MRI contrast agents for targeted tumor imaging. Nanomed. Nanotechnol. Biol. Med. 2021, 32, 102335. [Google Scholar] [CrossRef]
- Varbanov, H.P.; Göschl, S.; Heffeter, P.; Theiner, S.; Roller, A.; Jensen, F.; Jakupec, M.A.; Berger, W.; Galanski, M.S.; Keppler, B.K. A novel class of bis- and tris-chelate diam(m)inebis(dicarboxylato)platinum(IV) complexes as potential anticancer prodrugs. J. Med. Chem. 2014, 57, 6751–6764. [Google Scholar] [CrossRef]
- Murad, E. The Mössbauer spectrum of “well”-crystallized ferrihydrite. J. Magn. Magn. Mater. 1988, 74, 153–157. [Google Scholar] [CrossRef]
- Paschkunova-Martic, I.; Kremser, C.; Mistlberger, K.; Shcherbakova, N.; Dietrich, H.; Talasz, H.; Zou, Y.; Hugl, B.; Galanski, M.S.; Sölder, E.; et al. Design, synthesis, physical and chemical characterisation, and biological interactions of lectin-targeted latex nanoparticles bearing Gd-DTPA chelates: An exploration of magnetic resonance molecular imaging (MRMI). Histochem. Cell Biol. 2005, 123, 283–301. [Google Scholar] [CrossRef] [PubMed]
- Strbak, O.; Balejcikova, L.; Kmetova, M.; Gombos, J.; Kovac, J.; Dobrota, D.; Kopcansky, P. Longitudinal and Transverse Relaxivity Analysis of Native Ferritin and Magnetoferritin at 7 T MRI. Int. J. Mol. Sci. 2021, 22, 8487. [Google Scholar] [CrossRef]
- Lu, K.; Zhang, R.; Wang, H.; Li, C.; Yang, Z.; Xu, K.; Cao, X.; Wang, N.; Cai, W.; Zeng, J.; et al. PEGylated Ultrasmall Iron Oxide Nanoparticles as MRI Contrast Agents for Vascular Imaging and Real-Time Monitoring. ACS Nano 2025, 19, 3519–3530. [Google Scholar] [CrossRef]
- Pintaske, J.; Martirosian, P.; Graf, H.; Erb, G.; Lodemann, K.P.; Claussen, C.D.; Schick, F. Relaxivity of Gadopentetate Dimeglumine (Magnevist), Gadobutrol (Gadovist), and Gadobenate Dimeglumine (MultiHance) in human blood plasma at 0.2, 1.5, and 3 Tesla. Investig. Radiol. 2006, 41, 213–221. [Google Scholar] [CrossRef]


| Number | Tube | Compound | Fe Concentration, mg/L | Total Fe Concentration, mg/kg | Theoretical Fe Concentration, mg/kg |
|---|---|---|---|---|---|
| 1 | A2 | Composite 2 | 0.013 | 24,166 | 23,200 |
| 2 | A3 | Composite 3 | 0.016 | 21,730 | 23,200 |
| 3 | A4 | Composite 4 | 0.023 | 20,257 | 23,200 |
| Compound | Composition | Aver Size [nm] ± St Dev | Intensity [%] | ζ-Potential [mV] ± St Dev |
|---|---|---|---|---|
| Composite 2 | 338.9 ± 3.94 | 100 | 19.2 ± 0.700 | |
| Composite 3 | [(FeOOH)(C42H70O11)3].4H2O | 420.1 ± 25.21 | 100 | 22.0 ± 0.557 |
| Composite 4 | 369.8 ± 34.89 | 94.8 | 20.0 ± 0.751 | |
| 102.1 ± 1.78 | 5.2 |
| Sample | Components | IS, mm/s | QS, mm/s | FWHM, mm/s | A, % |
|---|---|---|---|---|---|
| Composite 2 | Db1-Fe(III) | 0.37 | 0.49 | 0.30 | 48 |
| Db2-Fe(III) | 0.43 | 0.87 | 0.30 | 52 | |
| Composite 3 | Db1-Fe(III) | 0.38 | 0.62 | 0.30 | 69 |
| Db2-Fe(III) | 0.37 | 1.06 | 0.30 | 31 | |
| Composite 4 | Db1-Fe(III) | 0.37 | 0.54 | 0.30 | 57 |
| Db2-Fe(III) | 0.38 | 0.92 | 0.30 | 43 |
| Complex | r1 [1/mM·s] MeOH/H2O | r2 [1/mM·s] MeOH/H2O | r2/r1 MeOH/H2O | Magnetic Field Strength, [T] |
|---|---|---|---|---|
| Composite 2 | 0.94/2.41 | 8.41/8.44 | 8.95/3.50 | 9.4 |
| Composite 3 | 0.06/0.30 | 8.68/1.82 | 144.67/6.07 | 9.4 |
| Composite 4 | 0.09/0.28 | 2.01/7.63 | 22.3/27.3 | 9.4 |
| PAA-Fe@Fe3O4 [31] | 0.72–4.00 | 13.8–27.8 | 4.5–38.6 | 0.5 |
| PEG-Fe@Fe3O4 [31] | 0.72–4.00 | 13.8–27.8 | 4.5–38.6 | 0.5 |
| DHCA-Fe@Fe3O4 [31] | 0.72–4.00 | 13.8–27.8 | 4.5–38.6 | 0.5 |
| Fe(L2) [7] | 1.1 | 1.4 | 1.16 | 1.4 |
| Fe(CDTA) [3] | 2.0 * | 2.2 * | 1.1 | 0.94 |
| Fe(HBED) [3] | 0.49 | 0.52 | 1.06 | 1.5 |
| Cell Line | A549 | SW480 | CH1/PA1 |
|---|---|---|---|
| Sample | |||
| 1. SalH [28] | 0.23 ± 0.06 | 1.1 ± 0.6 | 0.32 ± 0.12 |
| 2. SalNa [28] | 0.27 ± 0.02 | 0.88 ± 0.44 | 0.43 ± 0.11 |
| Composite 4: [(FeOOH)(C42H70O11)3].4H2O | 0.117 ± 0.062 | 0.53 ± 0.08 | 0.071 ± 0.004 |
| [Mn(Sal)2(H2O)2] [28] | 0.19 ± 0.11 | 0.52 ± 0.22 | 0.17 ± 0.05 |
| [Gd(Sal)3(H2O)3] [28] | 0.15 ± 0.12 | 0.36 ± 0.12 | 0.093 ± 0.025 |
| Cisplatin® [32] | 6.2 ± 1.2 | 3.3 ± 0.2 | 0.077 ± 0.006 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pashkunova-Martic, I.; Friske, J.; Paneva, D.; Cherkezova-Zheleva, Z.; Hejl, M.; Jakupec, M.; Braeuer, S.; Dorkov, P.; Keppler, B.K.; Helbich, T.H.; et al. First In Vitro Characterization of Salinomycinic Acid-Containing Two-Line Ferrihydrite Composites with Pronounced Antitumor Activity as MRI Contrast Agents. Int. J. Mol. Sci. 2025, 26, 8405. https://doi.org/10.3390/ijms26178405
Pashkunova-Martic I, Friske J, Paneva D, Cherkezova-Zheleva Z, Hejl M, Jakupec M, Braeuer S, Dorkov P, Keppler BK, Helbich TH, et al. First In Vitro Characterization of Salinomycinic Acid-Containing Two-Line Ferrihydrite Composites with Pronounced Antitumor Activity as MRI Contrast Agents. International Journal of Molecular Sciences. 2025; 26(17):8405. https://doi.org/10.3390/ijms26178405
Chicago/Turabian StylePashkunova-Martic, Irena, Joachim Friske, Daniela Paneva, Zara Cherkezova-Zheleva, Michaela Hejl, Michael Jakupec, Simone Braeuer, Peter Dorkov, Bernhard K. Keppler, Thomas H. Helbich, and et al. 2025. "First In Vitro Characterization of Salinomycinic Acid-Containing Two-Line Ferrihydrite Composites with Pronounced Antitumor Activity as MRI Contrast Agents" International Journal of Molecular Sciences 26, no. 17: 8405. https://doi.org/10.3390/ijms26178405
APA StylePashkunova-Martic, I., Friske, J., Paneva, D., Cherkezova-Zheleva, Z., Hejl, M., Jakupec, M., Braeuer, S., Dorkov, P., Keppler, B. K., Helbich, T. H., & Ivanova, J. (2025). First In Vitro Characterization of Salinomycinic Acid-Containing Two-Line Ferrihydrite Composites with Pronounced Antitumor Activity as MRI Contrast Agents. International Journal of Molecular Sciences, 26(17), 8405. https://doi.org/10.3390/ijms26178405








