Rodent Models of Lung Disease: A Road Map for Translational Research
Abstract
1. Introduction
2. LPS Model of ALI
2.1. Biochemical and Morphological Features
2.2. Synergistic Effect Between LPS and Cigarette Smoke
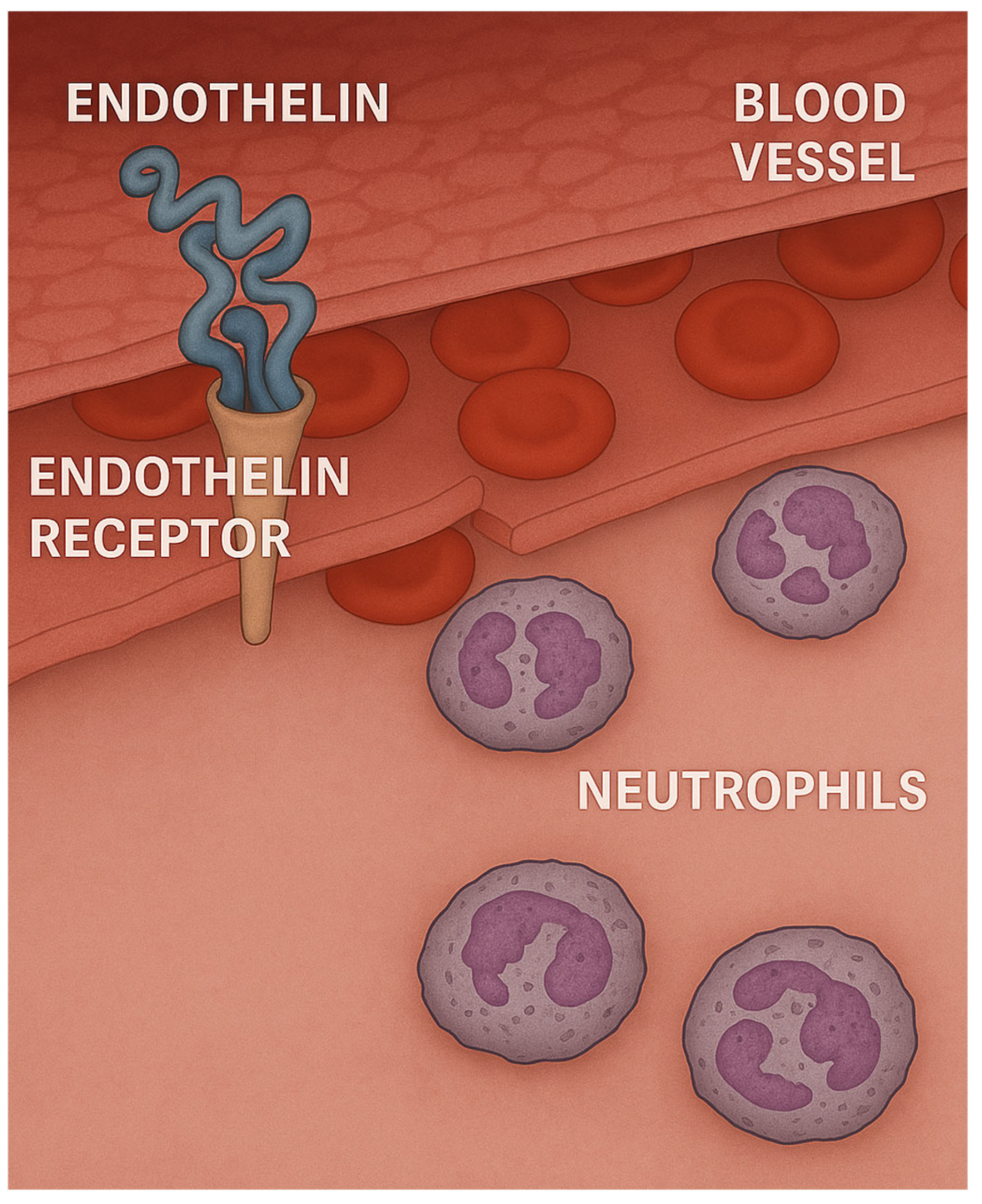
2.3. Role of Endothelin in LPS-Induced Lung Injury
3. Elastase-Induced Pulmonary Emphysema
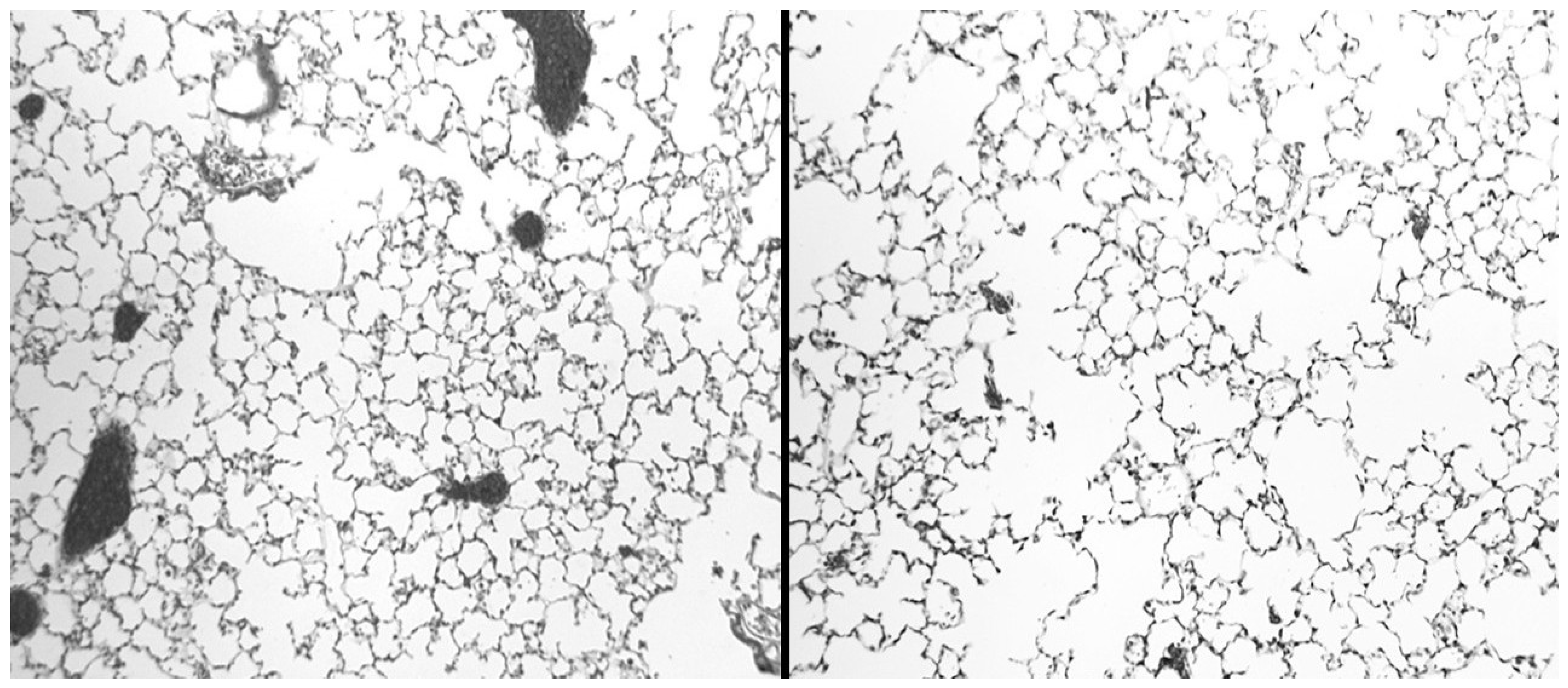
3.1. Biochemical and Morphological Features
3.2. The Ratio of Free to Total Desmosine as a Biomarker for Pulmonary Emphysema
3.3. Combining Elastase-Induced Emphysema with LPS
4. Cigarette Smoke-Induced Pulmonary Emphysema
4.1. Biochemical and Morphological Features
4.2. The Effect of Aerosolized Hyaluronan on Airspace Enlargement
5. Bleomycin Model of Pulmonary Fibrosis
5.1. Biochemical and Morphological Features
5.2. The Effect of Endothelin Inhibition
6. Analyzing the Microenvironment of Rodent Lung Disease Models
7. Animal Model Alternatives
8. Strategies for Addressing the Limitations of Animal Models
9. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Singh, V.K.; Seed, T.M. How necessary are animal models for modern drug discovery? Expert Opin. Drug Discov. 2021, 16, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- Tanner, L.; Single, A.B. Animal models reflecting chronic obstructive pulmonary disease and related respiratory disorders: Translating pre-clinical data into clinical relevance. J. Innate Immun. 2020, 12, 203–225. Available online: https://karger.com/jin/article/12/3/203/180041 (accessed on 24 May 2025). [CrossRef] [PubMed]
- Craig, J.M.; Scott, A.L.; Mitzner, W. Immune-mediated inflammation in the pathogenesis of emphysema: Insights from mouse models. Cell Tissue Res. 2017, 367, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Domscheit, H.; Hegeman, M.A.; Carvalho, N.; Spieth, P.M. Molecular dynamics of lipopolysaccharide-induced lung injury in rodents. Front. Physiol. 2020, 11, 36. [Google Scholar] [CrossRef]
- Jenkins, R.G.; Moore, B.B.; Chambers, R.C.; Eickelberg, O.; Fernandez, I.E.; Hogaboam, C.M.; Kolls, J.K.; Kolb, M.; Lawson, W.E.; Lloyd, C.M.; et al. An official American Thoracic Society workshop report: Use of animal models for the preclinical assessment of potential therapies for pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 2017, 57, P1–P10. [Google Scholar] [CrossRef]
- Tu, X.; Donovan, C.; Kim, R.Y.; Wark, P.A.B. Asthma-COPD overlap: Current understanding and the utility of experimental models. Eur. Respir. Rev. 2021, 30, 190185. Available online: https://publications.ersnet.org/content/errev/30/159/190185.abstract (accessed on 20 May 2025). [CrossRef]
- Baron, R.M.; Choi, A.J.; Owen, C.A.; Choi, A.M. Genetically manipulated mouse models of lung disease: Potential and pitfalls. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2012, 302, L485–L497. [Google Scholar] [CrossRef]
- Aeffner, F.; Bolon, B.; Davis, I.C. Mouse models of acute respiratory distress syndrome: A review of analytical approaches, pathologic features, and common measurements. Toxicol. Pathol. 2015, 43, 1074–1092. [Google Scholar] [CrossRef]
- Galvão, I.; Kim, R.Y.; Shen, S.; Budden, K.F.; Vieira, A.T.; Hansbro, P.M. Emerging therapeutic targets and preclinical models for severe asthma. Expert Opin. Ther. Targets 2020, 24, 845–857. [Google Scholar] [CrossRef]
- Bos, L.D.J.; Ware, L.B. Acute respiratory distress syndrome: Causes, pathophysiology, and phenotypes. Lancet 2022, 399, 2319–2332. Available online: https://www.thelancet.com/article/S0140-6736(22)01485-4/abstract (accessed on 25 May 2025). [CrossRef]
- Mokra, D. Acute lung injury—From pathophysiology to treatment. Physiol. Res. 2020, 69 (Suppl. 3), S353. [Google Scholar] [CrossRef]
- Chen, H.; Bai, C.; Wang, X. The value of the lipopolysaccharide-induced acute lung injury model in respiratory medicine. Expert Rev. Respir. Med. 2010, 4, 773–783. [Google Scholar] [CrossRef]
- Marrocco, A.; Ortiz, L.A. Role of metabolic reprogramming in proinflammatory cytokine secretion from LPS or silica-activated macrophages. Front. Immunol. 2022, 13, 936167. [Google Scholar] [CrossRef] [PubMed]
- Ciesielska, A.; Matyjek, M.; Kwiatkowska, K. TLR4 and CD14 trafficking and its influence on LPS-induced pro-inflammatory signaling. Cell. Mol. Life Sci. 2021, 78, 1233–1261. [Google Scholar] [CrossRef] [PubMed]
- Płóciennikowska, A.; Hromada, A. Co-operation of TLR4 and raft proteins in LPS-induced pro-inflammatory signaling. Cell. Mol. Life Sci. 2015, 72, 557–581. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Lucas, R.; Fulton, D.J.; Verin, A.D. Mechanisms of pulmonary endothelial barrier dysfunction in acute lung injury and acute respiratory distress syndrome. Chin. Med. J. Pulm. Crit. Care Med. 2024, 2, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Park, I.; Kim, M.; Choe, K.; Song, E.; Seo, H.; Lee, S. Neutrophils disturb pulmonary microcirculation in sepsis-induced acute lung injury. Eur. Respir. J. 2019, 53, 1800786. Available online: https://publications.ersnet.org/content/erj/53/3/1800786.abstract (accessed on 27 May 2025). [CrossRef] [PubMed]
- Zhou, X.; Dai, Q.; Huang, X. Neutrophils in acute lung injury. Front. Biosci. (Landmark Ed.) 2012, 17, 107–115. Available online: https://article.imrpress.com/bri/Landmark/articles/pdf/Landmark4051.pdf (accessed on 28 May 2025). [CrossRef]
- Millar, M.W.; Fazal, F.; Rahman, A. Therapeutic targeting of NF-κB in acute lung injury: A double-edged sword. Cells 2022, 11, 3317. [Google Scholar] [CrossRef]
- Brass, D.M.; Hollingsworth, J.W.; Cinque, M.; Schwartz, D.A. Chronic LPS inhalation causes emphysema-like changes in mouse lung that are associated with apoptosis. Am. J. Respir. Cell Mol. Biol. 2008, 39, 340–348. [Google Scholar] [CrossRef]
- Chimenti, L.; Morales-Quinteros, L.; Puig, F.; Camprubi-Rimblas, M.; Guillamat-Prats, R.; Gómez, M.N.; Tijero, J.; Blanch, L.; Matute-Bello, G.; Artigas, A. Comparison of direct and indirect models of early induced acute lung injury. Intensive Care Med. Exp. 2020, 8, 62. [Google Scholar] [CrossRef] [PubMed]
- Matute-Bello, G.; Frevert, C.W.; Martin, T.R. Animal models of acute lung injury. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2008, 295, L379–L399. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.; Xu, S.; Lam, T.Y.W.; Liao, W.; Wong, W.F.; Ge, R. ISM1 suppresses LPS-induced acute lung injury and post-injury lung fibrosis in mice. Mol. Med. 2022, 28, 72. [Google Scholar] [CrossRef] [PubMed]
- van der Vaart, H.; Postma, D.S.; Timens, W.; Ten Hacken, N.H. Acute effects of cigarette smoke on inflammation and oxidative stress: A review. Thorax 2004, 59, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Dauchet, L.; Hulo, S.; Cherot-Kornobis, N.; Matran, R.; Amouyel, P.; Edmé, J.L.; Giovannelli, J. Short-term exposure to air pollution: Associations with lung function and inflammatory markers in non-smoking, healthy adults. Environ. Int. 2018, 121, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Alexander, L.E.C.; Shin, S.; Hwang, J.H. Inflammatory diseases of the lung induced by conventional cigarette smoke: A review. Chest 2015, 148, 1307–1322. [Google Scholar] [CrossRef]
- Lin, L.; Li, J.; Song, Q.; Cheng, W.; Chen, P. The role of HMGB1/RAGE/TLR4 signaling pathways in cigarette smoke-induced inflammation in chronic obstructive pulmonary disease. Immun. Inflamm. Dis. 2022, 10, e711. [Google Scholar] [CrossRef]
- Lugg, S.T.; Scott, A.; Parekh, D.; Naidu, B.; Thickett, D.R. Cigarette smoke exposure and alveolar macrophages: Mechanisms for lung disease. Thorax 2022, 77, 94–101. [Google Scholar] [CrossRef]
- Seo, Y.S.; Park, J.M.; Kim, J.H.; Lee, M.Y. Cigarette smoke-induced reactive oxygen species formation: A concise review. Antioxidants 2023, 12, 1732. [Google Scholar] [CrossRef]
- Strzelak, A.; Ratajczak, A.; Adamiec, A.; Feleszko, W. Tobacco smoke induces and alters immune responses in the lung triggering inflammation, allergy, asthma and other lung diseases: A mechanistic review. Int. J. Environ. Res. Public Health 2018, 15, 1033. [Google Scholar] [CrossRef]
- Kulkarni, G.S.; Nadkarni, P.P.; Cerreta, J.M.; Ma, S.; Cantor, J.O. Short-term cigarette smoke exposure potentiates endotoxin-induced pulmonary inflammation. Exp. Lung Res. 2007, 33, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, D.K.; Hirata, F.; Rishi, A.K.; Gairola, C.G. Cigarette smoke, inflammation, and lung injury: A mechanistic perspective. J. Toxicol. Environ. Health 2009, 12 Pt B, 45–64. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Yeo, C.D.; Lee, H.Y.; Rhee, C.K.; Kim, I.K.; Lee, D.G.; Lee, S.H.; Kim, J.W. Inhibition of neutrophil elastase contributes to attenuation of lipopolysaccharide-induced acute lung injury during neutropenia recovery in mice. J. Anesth. 2017, 31, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Okeke, E.B.; Louttit, C.; Fry, C.; Najafabadi, A.H.; Han, K.; Nemzek, J.; Moon, J.J. Inhibition of neutrophil elastase prevents neutrophil extracellular trap formation and rescues mice from endotoxic shock. Biomaterials 2020, 238, 119836. [Google Scholar] [CrossRef] [PubMed]
- Schnoor, M.; Vadillo, E.; Guerrero, I.M. The extravasation cascade revisited from a neutrophil perspective. Curr. Opin. Hematol. 2021, 19, 119–128. [Google Scholar] [CrossRef]
- Bohr, A.; Tsapis, N.; Foged, C.; Andreana, I.; Yang, M.; Fattal, E. Treatment of acute lung inflammation by pulmonary delivery of anti-TNF-α siRNA with PAMAM dendrimers in a murine model. Eur. J. Pharm. Biopharm. 2020, 156, 114–120. [Google Scholar] [CrossRef]
- Fabricio, A.S.; Rae, G.A.; D’Orléans-Juste, P.; Souza, G.E. Endothelin-1 as a central mediator of LPS-induced fever in rats. Brain Res. 2005, 1066, 92–100. [Google Scholar] [CrossRef]
- Kowalczyk, A.; Kleniewska, P.; Kolodziejczyk, M.; Skibska, B.; Goraca, A. The role of endothelin-1 and endothelin receptor antagonists in inflammatory response and sepsis. Arch. Immunol. Et Ther. Exp. 2015, 63, 41–52. [Google Scholar] [CrossRef]
- Freeman, B.D.; Machado, F.S.; Tanowitz, H.B.; Desruisseaux, M.S. Endothelin-1 and its role in the pathogenesis of infectious diseases. Life Sci. 2014, 118, 110–119. [Google Scholar] [CrossRef]
- Zarpelon, A.C.; Pinto, L.G.; Cunha, T.M.; Vieira, S.M.; Carregaro, V.; Souza, G.R.; Silva, J.S.; Ferreira, S.H.; Cunha, F.Q.; Verri, W.A., Jr. Endothelin-1 induces neutrophil recruitment in adaptive inflammation via TNFα and CXCL1/CXCR2 in mice. Can. J. Physiol. Pharmacol. 2012, 90, 187–199. [Google Scholar] [CrossRef]
- Patel, S.; Liu, X.; Liu, M.; Stephani, R.; Patel, H.; Cantor, J. HJP272, a novel endothelin receptor antagonist, attenuates lipopolysaccharide-induced acute lung injury in hamsters. Lung 2014, 192, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Bhavsar, T.M.; Cerreta, J.M.; Liu, M.; Reznik, S.E.; Cantor, J.O. Phosphoramidon, an endothelin-converting enzyme inhibitor, attenuates lipopolysaccharide-induced acute lung injury. Exp. Lung Res. 2008, 34, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Schulz, H.; Dale, G.E.; Karimi-Nejad, Y.; Oefner, C. Structure of human endothelin-converting enzyme I complexed with phosphoramidon. J. Mol. Biol. 2009, 385, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Sauvageau, S.; Thorin, E.; Caron, A.; Dupuis, J. Endothelin-1-induced pulmonary vasoreactivity is regulated by ETA and ETB receptor interactions. J. Vasc. Res. 2007, 44, 375–384. Available online: https://karger.com/jvr/article/44/5/375/183677 (accessed on 22 May 2025). [CrossRef] [PubMed]
- Karakioulaki, M.; Papakonstantinou, E.; Stolz, D. Extracellular matrix remodelling in COPD. Eur. Respir. Rev. 2020, 29, 190124. [Google Scholar] [CrossRef]
- Heinz, A. Elastases and elastokines: Elastin degradation and its significance in health and disease. Crit. Rev. Biochem. Mol. Biol. 2020, 55, 252–273. [Google Scholar] [CrossRef]
- Kozel, B.A.; Mecham, R.P. Elastic fiber ultrastructure and assembly. Matrix Biol. 2019, 84, 31–40. [Google Scholar] [CrossRef]
- Trębacz, H.; Barzycka, A. Mechanical properties and functions of elastin: An overview. Biomolecules 2023, 13, 574. [Google Scholar] [CrossRef]
- Soskel, N.T.; Sandberg, L.B. Pulmonary Emphysema: From animal models to human diseases. In Connective Tissue Disease; Taylor & Francis: Boca Raton, FL, USA, 2021; Available online: https://www.taylorfrancis.com/chapters/edit/10.1201/9781003210016-22/pulmonary-emphysema-norman-soskel-lawrence-sandberg (accessed on 25 May 2025).
- Joshi, I.; Devine, A.J.; Joshi, R.; Smith, N.J.; Varisco, B.M. A titratable murine model of progressive emphysema using tracheal porcine pancreatic elastase. Sci. Rep. 2023, 13, 15259. [Google Scholar] [CrossRef]
- Oliveira, M.V.; Abreu, S.C.; Padilha, G.A.; Rocha, N.N.; Maia, L.A.; Takiya, C.M.; Xisto, D.G.; Suki, B.; Silva, P.L.; Rocco, P.R. Characterization of a mouse model of emphysema induced by multiple instillations of low-dose elastase. Front. Physiol. 2016, 7, 457. [Google Scholar] [CrossRef]
- Cantor, J. Desmosine as a biomarker for the emergent properties of pulmonary emphysema. Front. Med. 2023, 10, 1322283. [Google Scholar] [CrossRef] [PubMed]
- Schmelzer, C.E.; Duca, L. Elastic fibers: Formation, function, and fate during aging and disease. FEBS J. 2022, 289, 3704–3730. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ma, S.; Liu, S.; Liu, M.; Turino, G.; Cantor, J. The ratio of free to bound desmosine and isodesmosine may reflect emphysematous changes in COPD. Lung 2015, 193, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Cantor, J.O.; Ma, S.; Turino, G.M. What percolation theory can tell us about COPD. Med. Hypotheses 2013, 81, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Ko, F.W.; Chan, K.P.; Hui, D.S.; Goddard, J.R.; Shaw, J.G.; Reid, D.W.; Yang, I.A. Acute exacerbation of COPD. Respirology 2016, 21, 1152–1165. [Google Scholar] [CrossRef]
- Mehraban, S.; Gu, G.; Ma, S.; Liu, X.; Turino, G.; Cantor, J. The proinflammatory activity of structurally altered elastic fibers. Am. J. Respir. Cell Mol. Biol. 2020, 63, 699–706. [Google Scholar] [CrossRef]
- De Oliveira, M.V.; Rocha, N.N.; Santos, R.S.; Rocco, M.R.M.; de Magalhães, R.F.; Silva, J.D.; Souza, S.A.L.; Capelozzi, V.L.; Pelosi, P.; Silva, P.L.; et al. Endotoxin-Induced emphysema exacerbation: A novel model of chronic obstructive pulmonary disease exacerbations causing cardiopulmonary impairment and diaphragm dysfunction. Front. Physiol. 2019, 10, 664. [Google Scholar] [CrossRef]
- Kobayashi, S.; Fujinawa, R.; Ota, F.; Kobayashi, S.; Angata, T.; Ueno, M.; Maeno, T.; Kitazume, S.; Yoshida, K.; Ishii, T.; et al. A single dose of lipopolysaccharide into mice with emphysema mimics human chronic obstructive pulmonary disease exacerbation as assessed by micro-computed tomography. Am. J. Respir. Cell Mol. Biol. 2013, 49, 971–977. [Google Scholar] [CrossRef]
- Upadhyay, P.; Wu, C.W.; Pham, A.; Zeki, A.A.; Royer, C.M.; Kodavanti, U.P.; Takeuchi, M.; Bayram, H.; Pinkerton, K.E. Animal models and mechanisms of tobacco smoke-induced chronic obstructive pulmonary disease (COPD). J. Toxicol. Environ. Health 2023, 26 Pt B, 275–305. [Google Scholar] [CrossRef]
- Wright, J.L.; Churg, A. Animal models of cigarette smoke-induced chronic obstructive pulmonary disease. Expert Rev. Respir. Med. 2010, 4, 723–734. [Google Scholar] [CrossRef] [PubMed]
- Churg, A.; Cosio, M.; Wright, J.L. Mechanisms of cigarette smoke-induced COPD: Insights from animal models. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2008, 294, L612–L631. [Google Scholar] [CrossRef] [PubMed]
- Serré, J.; Tanjeko, A.T.; Mathyssen, C.; Vanherwegen, A.S.; Heigl, T.; Janssen, R.; Verbeken, E.; Maes, K.; Vanaudenaerde, B.; Janssens, W.; et al. Enhanced lung inflammatory response in whole-body compared to nose-only cigarette smoke-exposed mice. Respir. Res. 2021, 22, 86. [Google Scholar] [CrossRef] [PubMed]
- Kogel, U.; Wong, E.T.; Szostak, J.; Tan, W.T.; Lucci, F.; Leroy, P.; Titz, B.; Xiang, Y.; Low, T.; Wong, S.K.; et al. Impact of whole-body versus nose-only inhalation exposure systems on systemic, respiratory, and cardiovascular endpoints in a 2-month cigarette smoke exposure study in the ApoE−/− mouse model. J. Appl. Toxicol. 2021, 41, 1598–1619. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Cho, Y.; Oh, G.J.; Park, H.B.; Yang, M.J.; Park, C.M.; Kim, Y.H.; Choi, K.C.; Go, R.E.; Kim, M.S. Repeated intratracheal instillation of whole-cigarette smoke condensate to assess lung damage in a rat model. Environ. Toxicol. 2024, 39, 2304–2315. [Google Scholar] [CrossRef]
- Yao, H.; Edirisinghe, I.; Rajendrasozhan, S.; Yang, S.R.; Caito, S.; Adenuga, D.; Rahman, I. Cigarette smoke-mediated inflammatory and oxidative responses are strain-dependent in mice. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2008, 294, L1174–L1186. [Google Scholar] [CrossRef]
- Tsuji, H.; Fujimoto, H.; Lee, K.M.; Renne, R.; Iwanaga, A.; Okubo, C.; Onami, S.; Nomura, A.K.; Nishino, T.; Yoshimura, H. Characterization of biochemical, functional and structural changes in mice respiratory organs chronically exposed to cigarette smoke. Inhal. Toxicol. 2015, 27, 342–353. [Google Scholar] [CrossRef]
- Wick, M.R. Pathologic features of smoking-related lung diseases, with emphasis on smoking-related interstitial fibrosis and a consideration of differential diagnoses. Semin. Diagn. Pathol. 2018, 35, 315–323. [Google Scholar] [CrossRef]
- Cantor, J.O.; Cerreta, J.M.; Ochoa, M.; Ma, S.; Liu, M.; Turino, G.M. Therapeutic effects of hyaluronan on smoke-induced elastic fiber injury: Does delayed treatment affect efficacy? Lung 2011, 189, 51–56. [Google Scholar] [CrossRef]
- Morse, D.; Rosas, I.O. Tobacco smoke–induced lung fibrosis and emphysema. Annu. Rev. Physiol. 2014, 76, 493–513. [Google Scholar] [CrossRef]
- Cantor, J.O.; Cerreta, J.M.; Keller, S.; Turino, G.M. Modulation of Airspace Enlargement in Elastase-Induced Emphysema by Intratracheal Instalment of Hyaluronidase and Hyaluronic Acid. Exp. Lung Res. 1995, 21, 423–436. [Google Scholar] [CrossRef]
- Cantor, J.O.; Cerreta, J.M.; Armand, G.; Turino, G.M. Further investigation of the use of intratracheally administered hyaluronic acid to ameliorate elastase-induced emphysema. Exp. Lung Res. 1997, 23, 229–244. [Google Scholar] [CrossRef]
- Cantor, J.O.; Cerreta, J.M.; Ochoa, M.; Ma, S.; Chow, T.; Grunig, G.; Turino, G.M. Aerosolized hyaluronan limits airspace enlargement in a mouse model of cigarette smoke–induced pulmonary emphysema. Exp. Lung Res. 2005, 31, 417–430. [Google Scholar] [CrossRef]
- Cantor, J.O.; Ma, S.; Liu, X.; Campos, M.A.; Strange, C.; Stocks, J.M.; Devine, M.S.; El Bayadi, S.G.; Lipchik, R.J.; Sandhaus, R.A.; et al. A 28-day clinical trial of aerosolized hyaluronan in alpha-1 antiprotease deficiency COPD using desmosine as a surrogate marker for drug efficacy. Respir. Med. 2021, 182, 106402. [Google Scholar] [CrossRef] [PubMed]
- Della Latta, V.; Cecchettini, A.; Del Ry, S.; Morales, M.A. Bleomycin in the setting of lung fibrosis induction: From biological mechanisms to counteractions. Pharmacol. Res. 2015, 97, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Gul, A.; Yang, F.; Xie, C.; Du, W.; Mohammadtursun, N.; Wang, B.; Le, J.; Dong, J. Pulmonary fibrosis model of mice induced by different administration methods of bleomycin. BMC Pulm. Med. 2023, 23, 91. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Kuninaka, Y.; Mukaida, N.; Kondo, T. Immune mechanisms of pulmonary fibrosis with bleomycin. Int. J. Mol. Sci. 2023, 24, 3149. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Qin, Y.; Fu, X.; Yang, F.; Huo, F.; Liang, X.; Wang, S.; Cui, H.; Lin, P.; Zhou, G.; et al. Inhibition of ferroptosis and iron accumulation alleviates pulmonary fibrosis in a bleomycin model. Redox Biol. 2022, 57, 102509. [Google Scholar] [CrossRef]
- Scotton, C.J.; Chambers, R.C. Bleomycin revisited: Towards a more representative model of IPF? Am. J. Physiol.-Lung Cell. Mol. Physiol. 2010, 299, L439–L441. [Google Scholar] [CrossRef]
- Liu, X.; Khadtare, N.; Patel, H.; Stephani, R.; Cantor, J. Time-dependent effects of HJP272, an endothelin receptor antagonist, in bleomycin-induced pulmonary fibrosis. Pulm. Pharmacol. Ther. 2017, 45, 164–169. [Google Scholar] [CrossRef]
- Raghu, G.; Behr, J.; Brown, K.K.; Egan, J.J.; Kawut, S.M.; Flaherty, K.R.; Martinez, F.J.; Nathan, S.D.; Wells, A.U.; Collard, H.R.; et al. Treatment of idiopathic pulmonary fibrosis with ambrisentan: A parallel, randomized trial. Ann. Intern. Med. 2013, 158, 641–649. [Google Scholar] [CrossRef]
- Li, H.F.; Wang, J.X.; Xie, Z.F.; Li, L.H.; Li, B.; Huang, F.F.; Li, J.; Zhou, X.L. Bosentan and ambrisentan in the treatment of idiopathic pulmonary fibrosis: A meta-analysis. Eur. Rev. Med. Pharmacol. Sci. 2024, 28, 1183–1193. [Google Scholar]
- Li, S.; Pan, Y.L.; Xin, W.; Yan, C. The potential benefit of endothelin receptor antagonists’ therapy in idiopathic pulmonary fibrosis: A meta-analysis of results from randomized controlled trials. Medicine 2022, 101, e29981. [Google Scholar] [CrossRef] [PubMed]
- Hardie, W.D.; Glasser, S.W.; Hagood, J.S. Emerging concepts in the pathogenesis of lung fibrosis. Am. J. Pathol. 2009, 175, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Suki, B.; Bates, J.H. Lung tissue mechanics as an emergent phenomenon. J. Appl. Physiol. 2011, 110, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, R.R.; Lü, L.; Hu, M.B.; Xu, S.; Zhang, Y.C. Percolation on complex networks: Theory and application. Phys. Rep. 2021, 907, 1–68. [Google Scholar] [CrossRef]
- Oliveira, C.L.; Bates, J.H.; Suki, B. A network model of correlated growth of tissue stiffening in pulmonary fibrosis. New J. Phys. 2014, 16, 065022. [Google Scholar] [CrossRef]
- Habermann, A.C.; Gutierrez, A.J.; Bui, L.T.; Yahn, S.L.; Winters, N.I.; Calvi, C.L.; Kropski, J.A. Single-cell RNA sequencing reveals profibrotic roles of distinct epithelial and mesenchymal lineages in pulmonary fibrosis. Sci. Adv. 2020, 6, eaba1972. [Google Scholar] [CrossRef]
- Adams, T.S.; Schupp, J.C.; Poli, S.; Ayaub, E.A.; Neumark, N.; Ahangari, F.; Chu, S.G.; Raby, B.A.; DeIuliis, G.; Januszyk, M.; et al. Single-cell RNA-seq reveals ectopic and aberrant lung-resident cell populations in idiopathic pulmonary fibrosis. Sci. Adv. 2020, 6, eaba1983. [Google Scholar] [CrossRef]
- Sauler, M.; McDonough, J.E.; Adams, T.S.; Kothapalli, N.; Barnthaler, T.; Werder, R.B.; Schupp, J.C.; Nouws, J.; Robertson, M.J.; Coarfa, C.; et al. Characterization of the COPD alveolar niche using single-cell RNA sequencing. Nat. Commun. 2022, 13, 494. [Google Scholar] [CrossRef]
- Van de Sande, B.; Lee, J.S.; Mutasa-Gottgens, E.; Naughton, B.; Bacon, W.; Manning, J.; Wang, Y.; Pollard, J.; Mendez, M.; Hill, J.; et al. Applications of single-cell RNA sequencing in drug discovery and development. Nat. Rev. Drug Discov. 2023, 22, 496–520. [Google Scholar] [CrossRef]
- Tang, W.; Li, M.; Teng, F.; Cui, J.; Dong, J.; Wang, W. Single-cell RNA-sequencing in asthma research. Front. Immunol. 2022, 13, 988573. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Coyle, J.P.; Xiong, R.; Wang, Y.; Heflich, R.H.; Ren, B.; Gwinn, W.M.; Hayden, P.; Rojanasakul, L. Invited review: Human air-liquid-interface organotypic airway tissue models derived from primary tracheobronchial epithelial cells—Overview and perspectives. Vitr. Cell. Dev. Biol.-Anim. 2021, 57, 104–132. [Google Scholar] [CrossRef] [PubMed]
- Kühl, L.; Graichen, P.; von Daacke, N.; Mende, A.; Wygrecka, M.; Potaczek, D.P.; Miethe, S.; Garn, H. Human lung organoids—A novel experimental and precision medicine approach. Cells 2023, 12, 2067. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.Y.; Jiang, Y.X.; Yang, Y.C.; Liu, J.Y.; Huo, C.; Ji, X.L.; Qu, Y.Q. Cigarette smoke extract induces pyroptosis in human bronchial epithelial cells through the ROS/NLRP3/caspase-1 pathway. Life Sci. 2021, 269, 119090. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.; Freund, D.; Gomperts, B.N. Three-dimensional models of the lung: Past, present and future: A mini review. Biochem. Soc. Trans. 2022, 50, 1045–1056. [Google Scholar] [CrossRef] [PubMed]
- Alhaque, S.; Themis, M.; Al-Jamal, K.T. Three-dimensional cell culture: From evolution to revolution. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170216. [Google Scholar] [CrossRef] [PubMed]
- Suki, B.; Parameswaran, H. Computational modeling helps uncover mechanisms related to the progression of emphysema. Drug Discov. Today Dis. Models 2015, 15, 9–15. [Google Scholar] [CrossRef][Green Version]
- Suki, B.; Bates, J.H. Pulmonary Fibrosis. In Mathematical Modeling of the Healthy and Diseased Lung: Linking Structure, Biomechanics, and Mechanobiology; Springer Nature: Cham, Switzerland, 2025; pp. 193–224. [Google Scholar]
- Fröhlich, E. Replacement strategies for animal studies in inhalation testing. Sci 2021, 3, 45. [Google Scholar] [CrossRef]
- McGonigle, P.; Ruggeri, B. Animal models of human disease: Challenges in enabling translation. Biochem. Pharmacol. 2014, 87, 162–171. [Google Scholar] [CrossRef]
- Nair, A.; Morsy, M.A.; Jacob, S. Dose translation between laboratory animals and human in preclinical and clinical phases of drug development. Drug Dev. Res. 2018, 79, 373–382. [Google Scholar] [CrossRef]
- Wang, K.; Liao, Y.; Li, X.; Wang, R.; Zeng, Z.; Cheng, M.; Gao, L.; Xu, D.; Wen, F.; Wang, T. Inhibition of neutrophil elastase prevents cigarette smoke exposure-induced formation of neutrophil extracellular traps and improves lung function in a mouse model of chronic obstructive pulmonary disease. Int. Immunopharmacol. 2023, 114, 109537. [Google Scholar] [CrossRef]
- Liang, C.C.; Zhao, J.; Zhang, Y.Q.; Wang, L.; Chen, H. Research progress on the effect of neutrophil elastase and its inhibitors in respiratory diseases. J. Respir. Med. 2025, 12, 45–58. [Google Scholar] [CrossRef]
- Cooper, T.K.; Meyerholz, D.K.; Beck, A.P.; Delaney, M.A.; Piersigilli, A.; Southard, T.L.; Brayton, C.F. Research-Relevant Conditions and Pathology of Laboratory Mice, Rats, Gerbils, Guinea Pigs, Hamsters, Naked Mole Rats, and Rabbits. ILAR J. 2021, 62, 77–132. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Borzone, G.; Liberona, L.; Olmos, P.; Sáez, C.; Meneses, M.; Reyes, T.; Moreno, R.; Lisboa, C. Rat and hamster species differences in susceptibility to elastase-induced pulmonary emphysema relate to differences in elastase inhibitory capacity. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2007, 293, R1342–R1349. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bartalesi, B.; Cavarra, E.; Fineschi, S.; Lucattelli, M.; Lunghi, B.; Martorana, P.A.; Lungarella, G. Different lung responses to cigarette smoke in two strains of mice sensitive to oxidants. Eur. Respir. J. 2005, 25, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Khadtare, N.; Patel, H.; Stephani, R.; Cantor, J. Transient blockade of endothelin-1 mitigates amiodarone-induced pulmonary fibrosis. Lung 2018, 196, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Bhavsar, T.; Liu, X.J.; Patel, H.; Stephani, R.; Cantor, J.O. Preferential recruitment of neutrophils by endothelin-1 in acute lung inflammation induced by lipopolysaccharide or cigarette smoke. Int. J. Chron. Obs. Pulmon Dis. 2008, 3, 477–481. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]








| Knockout Gene | Function | Phenotypic Outcomes | Disease Relevance | Common Applications | References |
|---|---|---|---|---|---|
| TGF-β | Fibrogenic signaling | Enhanced fibrosis; alveolar thickening | Pulmonary fibrosis | Studying ECM deposition and fibroblast activation | Calthorpe et al. Am J Physiol Lung Cell Mol Physiol. 2023;324(3):L285–L296. doi: 10.1152/ajplung.00106.2021 |
| Elastase | Proteolytic tissue degradation | Alveolar destruction; emphysematous changes | Emphysema | Role of elastase in pathogenesis of emphysema | Suzuki et al. Am J Physiol Lung Cell Mol Physiol. 2020;318(6):L1172–L1182 doi: 10.1152/ajplung.00214.2019 |
| IL-1 | Pro-inflammatory cytokine | Reduced inflammation and immune cell infiltration | COPD, acute inflammation | Analyzing cytokine cascade and immune modulation | Malireddi et al. Front Immunol. 2022; 13:1068230 doi: 10.3389/fimmu.2022.1068230 |
| MMP-12 | Extracellular matrix remodeling | Impaired elastin breakdown; altered alveolar repair | Emphysema | ECM turnover and elastase activity studies | Lagente et al. Expert Opin Ther Targets. 2009:287–95 doi: 10.1517/14728220902751632 |
| SP-A/SP-D | Surfactant proteins, innate immunity | Impaired host defense; surfactant dysfunction | Infection, alveolar injury | Surfactant biology and microbial clearance | Watson et al. Front Immunol. 2021 Jan 19;11:622598 doi: 10.3389/fimmu.2020.622598 |
| Caveolin-1 | Endothelial signaling and caveolae | Vascular leakage; exaggerated fibrosis response | Fibrosis, vascular remodeling | Vascular permeability and fibrosis progression | Fan et al. Front Pharmacol. 2024;15:1417834 doi: 10.3389/fphar.2024.1417834 |
| TNF-α | Cytokine mediator of inflammation | Blunted inflammatory response; altered apoptosis | Chronic inflammation | Drug response and immunomodulation studies | Redente et al. Am J Respir Cell Mol Biol. 2014;50(4):825–37 doi: 10.1165/rcmb.2013-0386OC |
| Stage | Scale | Key Mechanisms | Emergent Phenomena | Outcome in Lung Architecture |
|---|---|---|---|---|
| Initial Strain | Biomechanical | - Localized fiber overextension - Shear stress on fiber networks | Localized fiber fraying and interstitial rupture | Subtle weakening of septal integrity |
| Cellular Response | Cellular | - Epithelial-mesenchymal transition (EMT) - Fibroblast recruitment | Fibroblast recruitment and matrix remodeling | Early stiffening and remodeling of tissue zones |
| Molecular Escalation | Molecular | - TGF-β signaling - ROS accumulation - Piezo channel activation | Signal amplification and sustained matrix production | Loss of compliance and architecture |
| Feedback Intensification | Multiscale | - Altered mechanical load distribution - Time-dependent remodeling | Cyclic progression of strain and remodeling | Fibrotic stiffening begins maladaptive self-reinforcement |
| Structural Collapse | Biomechanical | - Elastic fiber tearing and fragmentation - Airspace dilation | Architectural failure and airspace dilation | Fibrotic consolidation and emphysematous expansion |
| Systemic Emergence | Systems-Level | - Cross-talk between immune, mechanical, and signaling systems | Disease-wide progression, system-wide “order” | Irregular lung patterning and irreversible damage |
| Category | Model | Advantages | Limitations | Rodent Species Other Sources | References |
|---|---|---|---|---|---|
| In Vivo –Chemical Induction | Bleomycin-induced fibrosis | Mimics human fibrosis; widely studied | Reduction in fibrosis over time | C57BL/6 mice, Sprague- Dawley rats; Syrian hamsters | Ishida et al. 2023 [77] Int J Mol Sci. 2023;24(4):3149 doi: 10.3390/ijms24043149 |
| Elastase-induced emphysema | Direct alveolar wall injury; dose-related | Limited inflammation; fast onset | C57BL/6 mice, Wistar rats; Syrian hamsters | Joshi et al. 2023 [50] Sci Rep 13, 2023; 15259 doi: 10.1038/s41598-023-41527-1 | |
| LPS-induced acute lung injury | Strong immune response; acute model for ARDS | Limited chronic mimicry | BALB/c, C57BL/6 mice, Wistar rats; Syrian hamsters | Domscheit et al. 2020 [4] Front Physiol. 2020;11:36 doi: 10.3389/fphys.2020.00036 | |
| Cigarette smoke exposure | Closely mimics human emphysema; includes oxidative stress | Long exposure periods; delivery variability | C57BL/6 mice, BALB/c mice, Wistar rats | Yao et al. 2008 [66] Am J Physiol Lung Cell Mol Physiol. 2008;294(6):L1174-L1186. doi: 10.1152/ajplung.00439.2007 | |
| In Vitro–2D Cultures | Lung cell stretch | Reproducible; controlled mechanical insult | Limited cell diversity; lacks feedback | Primary cultures and cell lines from various species | Martin-Vicente et al. Am J Respir Cell Mol Biol.; 72(2):195–205 doi: 10.1165/rcmb.2023-0449OC |
| Air–liquid interface cultures | Preserves polarity; suitable for aerosol studies | Culture-sensitive; short lifespan | Primary cultures and cell lines from various species | Cao et al. 2021 [93] In Vitro Cell Dev Biol Anim. 2021;57:104-132. doi: 10.1007/s11626-020-00517-7 | |
| In Vitro–3D Systems | Lung organoids | Multicellular dynamics; native-like structure | Variable differentiation; perfusion limits | Humans and other species | Kühl et al. 2023 [94] Cells. 2023;12(16):2067. doi: 10.3390/cells12162067 |
| Lung-on-a-chip | Human relevance; mechanical/fluid integration | Technical complexity; limited throughput | Human cells | Francis et al. Drug Discov Today. 2022:9:2593–2602. doi: 10.1016/j.drudis.2022.06.004 | |
| Ex Vivo Systems | Isolated perfused lung | Preserves whole-organ architecture; real-time monitoring | Viability is short; setup intensive | Rats, mice, and other species | Eriksson et al. AAPS J. 2020;22(3):71 doi: 10.1208/s12248-020-0456-x |
| Precision-cut lung slices | Spatial structure retained; ECM studies | Static system; fresh tissue required | Rats, mice, and other species | Lam et al. Front Pharmacol. 2023;14:1162889. doi: 10.3389/fphar.2023.1162889 |
| Optimization Strategy | Description | Potential Benefits | Challenges |
|---|---|---|---|
| Combining toxins or insults | Use multiple agents (e.g., elastase + LPS, or bleomycin + cigarette smoke) to emulate complex pathologies. | Mimics multifactorial human disease; induces robust, heterogeneous injury. | Potential for confounding interactions. |
| Altering temporal sequence of insults | Adjust timing between exposures (e.g., preconditioning vs. simultaneous vs. sequential insults). | Reveals priming effects, injury progression, and recovery mechanisms. | Requires precise scheduling; may complicate interpretation of outcomes. |
| Delaying therapeutic intervention | Introduce treatment after disease is well-established (e.g., 7–21 days post-injury). | Reflects realistic clinical scenarios; allows evaluation of regeneration mechanisms. | Requires longer studies; potential for irreversible damage before treatment. |
| Comparative model analysis | Test multiple models (e.g., elastase vs. bleomycin; transgenic vs. toxin exposure) for same disease context. | Distills mechanistic relevance; improves translational accuracy. | Strain, time course, or injury severity may confound comparisons. |
| Stratifying by injury outcome | Focus on specific outcomes (e.g., fibrosis vs. emphysema), or pathological features. | Enhances precision and rigor; allows targeted mechanistic insights. | Requires extensive phenotyping; may exclude intermediate/combined outcomes. |
| Integrating genetic manipulation | Combine with transgenic or knockout models. | Clarifies gene-environment interactions; enables mechanistic dissection. | Genetic background may influence response; complex breeding required. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cantor, J. Rodent Models of Lung Disease: A Road Map for Translational Research. Int. J. Mol. Sci. 2025, 26, 8386. https://doi.org/10.3390/ijms26178386
Cantor J. Rodent Models of Lung Disease: A Road Map for Translational Research. International Journal of Molecular Sciences. 2025; 26(17):8386. https://doi.org/10.3390/ijms26178386
Chicago/Turabian StyleCantor, Jerome. 2025. "Rodent Models of Lung Disease: A Road Map for Translational Research" International Journal of Molecular Sciences 26, no. 17: 8386. https://doi.org/10.3390/ijms26178386
APA StyleCantor, J. (2025). Rodent Models of Lung Disease: A Road Map for Translational Research. International Journal of Molecular Sciences, 26(17), 8386. https://doi.org/10.3390/ijms26178386





