A Unified Pathogenesis of Allergic Diseases; The Protein–Homeostasis–System Hypothesis
Abstract
1. Introduction
2. A Brief History of Immunology and Allergy
3. Evolutionary Aspects of the Immune System: Immunological Memory and Cross-Reactivity
3.1. Immunological Memory
3.2. Cross-Reactivity
4. Limitations of Modern Immunology
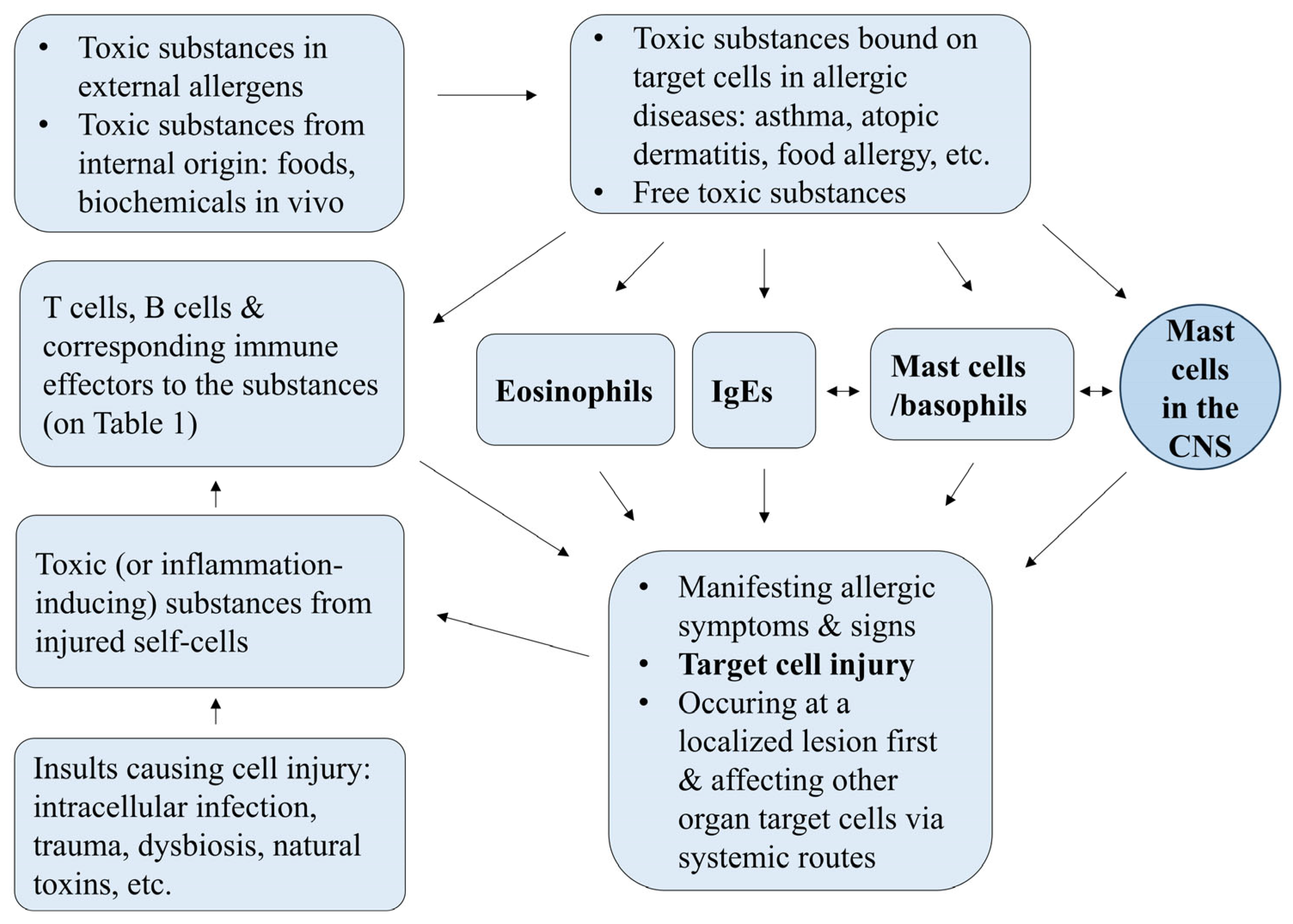
5. The PHS Hypothesis
5.1. Functional Consistency of Immune Components and the Foci of Etiological Substances
5.2. Target Cells and Receptor-Mediated Inflammation
5.3. Phenotype, Pathology, and Pathogenesis of Diseases
5.4. Integrated Immune Response and Lack of Specific Immune Effectors
5.5. PHS Perspective on Cancer and Genetic Disorders
6. Antigens, Allergens, and Etiological Substances
6.1. Antigens: Proteins/Peptides
6.2. Allergens
6.3. Pathogenic Substances Within Cells
7. Immune Components in Allergy Under the PHS Hypothesis
7.1. T Cells and MHCs
7.2. B Cells and IgEs
7.3. Innate Immune Cells and Eosinophils
7.4. Mast Cells, the Mast Cell-Associated Network, and Psychological System
8. Pathologic Findings in Allergic Diseases
9. Microbiota in Allergic and Autoimmune Diseases
10. Pathophysiology of Allergic and Autoimmune Diseases Under the PHS Hypothesis
10.1. Asthma
10.2. Atopic Dermatitis
10.3. Other Allergic, Chronic Inflammatory and Autoimmune Diseases
11. Treatment
12. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | atopic dermatitis |
| APC | antigen presenting cell |
| ARDS | acute respiratory distress syndrome |
| BAL | bronchial alveolar lavage |
| BCR | B cell receptor |
| CNS | Central nervous system |
| COPD | chronic obstructive pulmonary disease |
| CS | Corticosteroid |
| DAMPs | Damage (danger)-associated molecular patterns |
| IVIG | Intravenous immunoglobulin |
| JIA | Juvenile idiopathic arthritis |
| KD | Kawasaki disease |
| MCAS | Mast cell activation syndrome |
| MIS-C | Multisystem inflammatory syndrome in children |
| MHC | Major histocompatibility complex |
| NK | Natural killer |
| PAMPs | Pathogen-associated molecular patterns |
| PHS | Protein–homeostasis–system |
| PRRs | Pattern recognizing receptors |
| RA | Rheumatoid arthritis |
| SLE | systemic lupus erythematosus |
| TCR | T cell receptor |
| TLRs | Toll-like receptors |
| TNF-α | Tumor necrosis factor-α |
References
- Zheng, J.; Jin, Y.J.; Wang, C.H.; Feng, C.; Lai, X.Y.; Hua, S.Q.; Tai, J.H. Global, regional, and national epidemiology of allergic diseases in children from 1990 to 2021: Findings from the Global Burden of Disease Study 2021. BMC Pulm. Med. 2025, 25, 54. [Google Scholar] [CrossRef]
- Cao, F.; He, Y.S.; Wang, Y.; Zha, C.K.; Lu, J.M.; Tao, L.M.; Jiang, Z.X.; Pan, H.F. Global burden and cross-country inequalities in autoimmune diseases from 1990 to 2019. Autoimmun. Rev. 2023, 22, 103326. [Google Scholar] [CrossRef]
- Lundbäck, B.; Backman, H.; Lötvall, J.; Rönmark, E. Is asthma prevalence still increasing? Expert Rev. Respir. Med. 2016, 10, 39–51. [Google Scholar] [CrossRef]
- Rosslenbroich, B. Outline of a concept for organismic systems biology. Semin. Cancer Biol. 2011, 21, 156–164. [Google Scholar] [CrossRef]
- Lee, K.Y.; Rhim, J.W.; Kang, J. Kawasaki disease: Laboratory findings and an immunopathogenesis on the premise of a “protein homeostasis system”. Yonsei Med. J. 2012, 53, 262–275. [Google Scholar] [CrossRef]
- Lee, K.Y. A common immunopathogenesis mechanism for infectious diseases: The protein-homeostasis-system hypothesis. Infect. Chemother. 2015, 47, 12–26. [Google Scholar] [CrossRef]
- Lee, K.Y. A unified pathogenesis for kidney diseases, including genetic diseases and cancers, by the protein-homeostasis-system hypothesis. Kidney Res. Clin. Pract. 2017, 36, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Rhim, J.W.; Kang, J.H. Hyperactive immune cells (T cells) may be responsible for acute lung injury in influenza virus infections: A need for early immune-modulators for severe cases. Med. Hypotheses 2011, 76, 64–69. [Google Scholar] [CrossRef]
- Lee, K.Y.; Rhim, J.W.; Kang, J.H. Immunopathogenesis of COVID-19 and early immunomodulators. Clin. Exp. Pediatr. 2020, 63, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Rhim, J.W.; Kang, J.H.; Lee, K.Y. Etiological and pathophysiological enigmas of severe coronavirus disease 2019, multisystem inflammatory syndrome in children, and Kawasaki disease. Clin. Exp. Pediatr. 2022, 65, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y. Pneumonia, Acute Respiratory Distress Syndrome, and Early Immune-Modulator Therapy. Int. J. Mol. Sci. 2017, 18, 388. [Google Scholar] [CrossRef]
- Lee, K.Y. Common immunopathogenesis of central nervous system diseases: The protein-homeostasis-system hypothesis. Cell Biosci. 2022, 12, 184. [Google Scholar] [CrossRef] [PubMed]
- Rhim, J.W.; Lee, K.Y. An Alternate Perspective on Cancer Immunology: The Protein-Homeostasis-System Hypothesis. Preprints 2024. [Google Scholar] [CrossRef]
- Kapri, A.; Pant, S.; Gupta, N.; Paliwal, S.; Nain, S. Asthma history, current situation, an overview of its control history, challenges, and ongoing management programs: An updated review. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2023, 93, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Pulgaron, E.R.; Delamater, A.M. Obesity and type 2 diabetes in children: Epidemiology and treatment. Curr. Diab. Rep. 2014, 14, 508. [Google Scholar] [CrossRef]
- Le Fanu, J. The Rise and Fall of Modern Medicine; Little, Brown & Co.: London, UK, 1999. [Google Scholar]
- Smith, G.M. Vis Medicatrix Naturæ: Inaugural Address at the Annual Meeting of the Bath and Bristol Branch of the British Medical Association, 26 May 1909. Bristol. Med. Chir. J. 1909, 27, 321–336. [Google Scholar] [PubMed]
- Carlsson, F.; Råberg, L. The germ theory revisited: A noncentric view on infection outcome. Proc. Natl. Acad. Sci. USA 2024, 121, e2319605121. [Google Scholar] [CrossRef]
- Doherty, M.; Robertson, M.J. Some early trends in immunology. Trends Immunol. 2004, 25, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Debelic, M. Immunology and allergy: The twins. J. Investig. Allergol. Clin. Immunol. 1991, 1, 231–234. [Google Scholar]
- Igea, J.M. The history of the idea of allergy. Allergy 2013, 68, 966–973. [Google Scholar] [CrossRef]
- Shulman, S.T. Clemens von Pirquet: A remarkable life and career. J. Pediatric Infect. Dis. Soc. 2017, 6, 376–379. [Google Scholar] [CrossRef][Green Version]
- Jamieson, M. Imagining ‘reactivity’: Allergy within the history of immunology. Stud. Hist. Philos. Biol. Biomed. Sci. 2010, 41, 356–366. [Google Scholar] [CrossRef]
- Ring, J. Terminology of allergic phenomena. Chem. Immunol. Allergy 2014, 100, 46–52. [Google Scholar] [CrossRef]
- Ishizaka, T. IgE and mechanisms of IgE-mediated hypersensitivity. Ann. Allergy 1982, 48, 313–319. [Google Scholar] [PubMed]
- Ribatti, D. The discovery of immunoglobulin E. Immunol. Lett. 2016, 171, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.G.; Irvin, C.G. Mechanisms of airway hyper-responsiveness in asthma: The past, present and yet to come. Clin. Exp. Allergy 2015, 45, 706–719. [Google Scholar] [CrossRef]
- Sampson, H.A. Food allergy: Past, present and future. Allergol. Int. 2016, 65, 363–369. [Google Scholar] [CrossRef]
- Koonin, E.V.; Makarova, K.S.; Wolf, Y.I. Evolutionary genomics of defense systems in Archaea and Bacteria. Annu. Rev. Microbiol. 2017, 71, 233–261. [Google Scholar] [CrossRef] [PubMed]
- Muthamilarasan, M.; Prasad, M. Plant innate immunity: An updated insight into defense mechanism. J. Biosci. 2013, 38, 433–449. [Google Scholar] [CrossRef]
- Cooper, D.; Eleftherianos, I. Memory and specificity in the insect immune system: Current perspectives and future challenges. Front. Immunol. 2017, 8, 539. [Google Scholar] [CrossRef]
- Kumar, V.; Stewart, J.H. Pattern-recognition receptors and immunometabolic reprogramming: What we know and what to explore. J. Innate Immun. 2024, 16, 295–323. [Google Scholar] [CrossRef] [PubMed]
- Netea, M.G.; Joosten, L.A.; Latz, E.; Mills, K.H.; Natoli, G.; Stunnenberg, H.G.; O’Neill, L.A.; Xavier, R.J. Trained immunity: A program of innate immune memory in health and disease. Science 2016, 352, aaf1098. [Google Scholar] [CrossRef] [PubMed]
- Ochando, J.; Mulder, W.J.M.; Madsen, J.C.; Netea, M.G.; Duivenvoorden, R. Trained immunity—Basic concepts and contributions to immunopathology. Nat. Rev. Nephrol. 2023, 19, 23–37. [Google Scholar] [CrossRef]
- Ziogas, A.; Bruno, M.; van der Meel, R.; Mulder, W.J.M.; Netea, M.G. Trained immunity: Target for prophylaxis and therapy. Cell Host Microbe 2023, 31, 1776–1791. [Google Scholar] [CrossRef] [PubMed]
- Proal, A.D.; Marshall, T.G. Re-framing the theory of autoimmunity in the era of the microbiome: Persistent pathogens, auto-antibodies, and molecular mimicry. Discov. Med. 2018, 25, 299–308. [Google Scholar] [PubMed]
- Popescu, F.D. Cross-reactivity between aeroallergens and food allergens. World J. Methodol. 2015, 5, 31–50. [Google Scholar] [CrossRef]
- Tyagi, N.; Farnell, E.J.; Fitzsimmons, C.M.; Ryan, S.; Tukahebwa, E.; Maizels, R.M.; Dunne, D.W.; Thornton, J.M.; Furnham, N. Comparisons of allergenic and metazoan parasite proteins: Allergy, the price of immunity. PLoS Comput. Biol. 2015, 11, e1004546. [Google Scholar] [CrossRef]
- Bacher, P.; Hohnstein, T.; Beerbaum, E.; Röcker, M.; Blango, M.G.; Kaufmann, S.; Röhmel, J.; Eschenhagen, P.; Grehn, C.; Seidel, K.; et al. Human anti-fungal Th17 immunity and pathology rely on cross-reactivity against Candida albicans. Cell 2019, 176, 1340–1355.e15. [Google Scholar] [CrossRef]
- Kuhn, T.S. The Structure of Scientific Revolutions, 3rd ed.; The University of Chicago Press: Chicago, IL, USA, 1999. [Google Scholar]
- Matzinger, P. The danger model: A renewed sense of self. Science 2002, 296, 301–305. [Google Scholar] [CrossRef]
- Sun, L.; Su, Y.; Jiao, A.; Wang, X.; Zhang, B. T cells in health and disease. Signal Transduct. Target. Ther. 2023, 8, 235. [Google Scholar] [CrossRef]
- Jansen, K.; Cevhertas, L.; Ma, S.; Satitsuksanoa, P.; Akdis, M.; van de Veen, W. Regulatory B cells, A to Z. Allergy 2021, 76, 2699–2715. [Google Scholar] [CrossRef]
- Cramer, D.V. Natural antibodies and the host immune responses to xenografts. Xenotransplantation 2000, 7, 83–92. [Google Scholar] [CrossRef]
- Funes, S.C.; Rios, M.; Escobar-Vera, J.; Kalergis, A.M. Implications of macrophage polarization in autoimmunity. Immunology 2018, 154, 186–195. [Google Scholar] [CrossRef]
- Mihele, D.M.; Nistor, P.A.; Bruma, G.; Mitran, C.I.; Mitran, M.I.; Condrat, C.E.; Tovaru, M.; Tampa, M.; Georgescu, S.R. Mast cell activation syndrome update—A dermatological perspective. J. Pers. Med. 2023, 13, 1116. [Google Scholar] [CrossRef]
- Golstein, P.; Griffiths, G.M. An early history of T cell-mediated cytotoxicity. Nat. Rev. Immunol. 2018, 18, 527–535. [Google Scholar] [CrossRef]
- Liu, S.; Galat, V.; Galat, Y.; Lee, Y.K.A.; Wainwright, D.; Wu, J. NK cell-based cancer immunotherapy: From basic biology to clinical development. J. Hematol. Oncol. 2021, 14, 7. [Google Scholar] [CrossRef]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer immunoediting: From immunosurveillance to tumor escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef]
- Rogozynski, N.P.; Dixon, B. The Th1/Th2 paradigm: A misrepresentation of helper T cell plasticity. Immunol. Lett. 2024, 268, 106870. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, M.G.; Stanfield, R.L.; Wilson, I.A. How TCRs bind MHCs, peptides, and coreceptors. Annu. Rev. Immunol. 2006, 24, 419–466. [Google Scholar] [CrossRef] [PubMed]
- Akshata, K.R.; Ranganath, V.; Nichani, A.S. Thesis, antithesis, and synthesis in periodontal and systemic interlink. J. Indian Soc. Periodontol. 2012, 16, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Weille, F.L.; Vang, R.R. Sinusitis as focus of infection in uveitis, keratitis, and retrobulbar neuritis. AMA Arch. Otolaryngol. 1953, 58, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Konkel, J.E.; O’Boyle, C.; Krishnan, S. Distal consequences of oral inflammation. Front. Immunol. 2019, 10, 1403. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.U.; Kim, E.K.; Youn, Y.S.; Rhim, J.W.; Lee, K.Y. Outbreaks of mumps: An observational study over two decades in a single hospital in Korea. Korean J. Pediatr. 2014, 57, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Yee, K.T.; Pitts, M.; Tongyoo, P.; Rojnuckarin, P.; Wilkinson, M.C. Snake venom metalloproteinases and their peptide inhibitors from Myanmar Russell’s viper venom. Toxins 2016, 9, 15. [Google Scholar] [CrossRef]
- Munawar, A.; Ali, S.A.; Akrem, A.; Betzel, C. Snake venom peptides: Tools of biodiscovery. Toxins 2018, 10, 474. [Google Scholar] [CrossRef]
- Durandy, A.; Kracker, S. Immunoglobulin class-switch recombination deficiencies. Arthritis Res. Ther. 2012, 14, 218. [Google Scholar] [CrossRef]
- Eggel, A.; Pennington, L.F.; Jardetzky, T.S. Therapeutic monoclonal antibodies in allergy: Targeting IgE, cytokine, and alarmin pathways. Immunol. Rev. 2024, 328, 387–411. [Google Scholar] [CrossRef]
- Kosten, T.R.; Domingo, C.B. Can you vaccinate against substance abuse? Expert Opin. Biol. Ther. 2013, 13, 1093–1097. [Google Scholar] [CrossRef]
- Satoh, M.; Tanaka, S.; Chan, E.K. The uses and misuses of multiplex autoantibody assays in systemic autoimmune rheumatic diseases. Front. Immunol. 2015, 6, 181. [Google Scholar] [CrossRef]
- Cockx, M.; Van Hoovels, L.; De Langhe, E.; Lenaerts, J.; Thevissen, K.; Persy, B.; Bonroy, C.; Vercammen, M.; Bossuyt, X. La-boratory evaluation of anti-dsDNA antibodies. Clin. Chim. Acta 2022, 528, 34–43. [Google Scholar] [CrossRef]
- Raptopoulou, A.; Sidiropoulos, P.; Katsouraki, M.; Boumpas, D.T. Anti-citrulline antibodies in the diagnosis and prognosis of rheumatoid arthritis: Evolving concepts. Crit. Rev. Clin. Lab. Sci. 2007, 44, 339–363. [Google Scholar] [CrossRef]
- Carmel, N.N.; Rotman-Pikielny, P.; Lavrov, A.; Levy, Y. Vitamin D antibodies in systemic sclerosis patients: Findings and clinical correlations. Isr. Med. Assoc. J. 2015, 17, 80–84. [Google Scholar]
- Palm, N.W.; Rosenstein, R.K.; Medzhitov, R. Allergic host defences. Nature 2012, 484, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.; Starkl, P.; Marichal, T.; Galli, S.J. Testing the ‘toxin hypothesis of allergy’: Mast cells, IgE, and innate and acquired immune responses to venoms. Curr. Opin. Immunol. 2015, 36, 80–87. [Google Scholar] [CrossRef]
- Smith-Norowitz, T.A.; Wong, D.; Kusonruksa, M.; Norowitz, K.B.; Joks, R.; Durkin, H.G.; Bluth, M.H. Long term persistence of IgE anti-influenza virus antibodies in pediatric and adult serum post vaccination with influenza virus vaccine. Int. J. Med. Sci. 2011, 8, 239–244. [Google Scholar] [CrossRef]
- Sicherer, S.H.; Sampson, H.A. Food allergy: A review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J. Allergy Clin. Immunol. 2018, 141, 41–58. [Google Scholar] [CrossRef]
- Khatri, K.; O’Malley, A.; Linn, C.; Kowal, K.; Chruszcz, M. Role of small molecule ligands in IgE-mediated allergy. Curr. Allergy Asthma Rep. 2023, 23, 497–508. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, J.; Chanez, P.; Chanal, I.; Michel, F.B. Comparison between RAST and Pharmacia CAP system: A new automated specific IgE assay. J. Allergy Clin. Immunol. 1990, 85, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- Seth, D.; Poowutikul, P.; Pansare, M.; Kamat, D. Food allergy: A review. Pediatr. Ann. 2020, 49, e50–e58. [Google Scholar] [CrossRef]
- Manfredo Vieira, S.; Hiltensperger, M.; Kumar, V.; Zegarra-Ruiz, D.; Dehner, C.; Khan, N.; Costa, F.R.C.; Tiniakou, E.; Greiling, T.; Ruff, W.; et al. Translocation of a gut pathobiont drives autoimmunity in mice and humans. Science 2018, 359, 1156–1161, https://doi.org/10.1126/science.aar7201, Erratum in Science 2018, 360, eaat9922. [Google Scholar] [CrossRef]
- Yang, S.-N.; Hsieh, C.-C.; Kuo, H.-F.; Lee, M.-S.; Huang, M.-Y.; Kuo, C.-H.; Hung, C.-H. The effects of environmental toxins on allergic inflammation. Allergy Asthma Immunol. Res. 2014, 6, 478–484. [Google Scholar] [CrossRef]
- Trivedi, V.; Apala, D.R.; Iyer, V.N. Occupational asthma: Diagnostic challenges and management dilemmas. Curr. Opin. Pulm. Med. 2017, 23, 177–183. [Google Scholar] [CrossRef]
- van den Anker, J.; Reed, M.D.; Allegaert, K.; Kearns, G.L. Developmental Changes in Pharmacokinetics and Pharmacodynamics. J. Clin. Pharmacol. 2018, 58 (Suppl. S10), S10–S25. [Google Scholar] [CrossRef]
- Jackson, M. “Divine stramonium”: The rise and fall of smoking for asthma. Med. Hist. 2010, 54, 171–194. [Google Scholar] [CrossRef]
- Jayaraj, G.G.; Hipp, M.S.; Hartl, F.U. Functional modules of the proteostasis network. Cold Spring Harb. Perspect. Biol. 2020, 12, a033951. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Lee, J.S. Immunoglobulin G has a role for systemic protein modulation in vivo: A new concept of protein homeostasis. Med. Hypotheses 2006, 67, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Hommen, F.; Bilican, S.; Vilchez, D. Protein clearance strategies for disease intervention. J. Neural Transm. 2022, 129, 141–172. [Google Scholar] [CrossRef]
- Morozov, A.V.; Karpov, V.L. Proteasomes and several aspects of their heterogeneity relevant to cancer. Front. Oncol. 2019, 9, 761. [Google Scholar] [CrossRef]
- Türker, F.; Cook, E.K.; Margolis, S.S. The proteasome and its role in the nervous system. Cell Chem. Biol. 2021, 28, 903–917. [Google Scholar] [CrossRef]
- Dasgupta, S.; Yang, C.; Castro, L.M.; Tashima, A.K.; Ferro, E.S.; Moir, R.D.; Willis, I.M.; Fricker, L.D. Analysis of the yeast pep-tidome and comparison with the human peptidome. PLoS ONE 2016, 11, e0163312. [Google Scholar] [CrossRef] [PubMed]
- Lyapina, I.; Ivanov, V.; Fesenko, I. Peptidome: Chaos or inevitability. Int. J. Mol. Sci. 2021, 22, 13128. [Google Scholar] [CrossRef]
- Trost, B.; Lucchese, G.; Stufano, A.; Bickis, M.; Kusalik, A.; Kanduc, D. No human protein is exempt from bacterial motifs, not even one. Self Nonself 2010, 1, 328–334. [Google Scholar] [CrossRef]
- Fricker, L.D. Proteasome inhibitor drugs. Annu. Rev. Pharmacol. Toxicol. 2020, 60, 457–476. [Google Scholar] [CrossRef]
- Ogulur, I.; Pat, Y.; Ardicli, O.; Barletta, E.; Cevhertas, L.; Fernandez-Santamaria, R.; Huang, M.; Imam, M.B.; Koch, J.; Ma, S.; et al. Advances and highlights in biomarkers of allergic diseases. Allergy 2021, 76, 3659–3686. [Google Scholar] [CrossRef]
- Muluk, N.B.; Altın, F.; Cingi, C. Role of superantigens in allergic inflammation: Their relationship to allergic rhinitis, chronic rhinosinusitis, asthma, and atopic dermatitis. Am. J. Rhinol. Allergy 2018, 32, 502–517. [Google Scholar] [CrossRef]
- O’Flynn, K.; Linch, D.C.; Tatham, P.E. The effect of mitogenic lectins and monoclonal antibodies on intracellular free calcium concentration in human T-lymphocytes. Biochem. J. 1984, 219, 661–666. [Google Scholar] [CrossRef]
- Wu, C.; Jiang, M.L.; Pang, T.; Zhang, C.J. T cell subsets and immune homeostasis. Methods Mol. Biol. 2024, 2782, 39–63. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hafez, M.; Shimada, M.; Lee, P.Y.; Johnson, R.J.; Garin, E.H. Idiopathic nephrotic syndrome and atopy: Is there a common link? Am. J. Kidney Dis. 2009, 54, 945–953. [Google Scholar] [CrossRef] [PubMed]
- Bluth, M.H.; Robin, J.; Ruditsky, M.; Norowitz, K.B.; Chice, S.; Pytlak, E.; Nowakowski, M.; Durkin, H.G.; Smith-Norowitz, T.A. IgE anti-Borrelia burgdorferi components (p18, p31, p34, p41, p45, p60) and increased blood CD8⁺CD60⁺ T cells in children with Lyme disease. Scand. J. Immunol. 2007, 65, 376–382. [Google Scholar] [CrossRef]
- Maurer, M.; Altrichter, S.; Schmetzer, O.; Scheffel, J.; Church, M.K.; Metz, M. Immunoglobulin E-mediated autoimmunity. Front. Immunol. 2018, 9, 689. [Google Scholar] [CrossRef] [PubMed]
- Magen, E.; Schlesinger, M.; David, M.; Ben-Zion, I.; Vardy, D. Selective IgE deficiency, immune dysregulation, and autoimmunity. Allergy Asthma Proc. 2014, 35, e27–e33. [Google Scholar] [CrossRef]
- Ferastraoaru, D.; Gross, R.; Rosenstreich, D. Increased malignancy incidence in IgE deficient patients not due to concomitant common variable immunodeficiency. Ann. Allergy Asthma Immunol. 2017, 119, 267–273. [Google Scholar] [CrossRef]
- Eguiluz-Gracia, I.; Pérez-Sánchez, N.; Bogas, G.; Campo, P.; Rondón, C. How to diagnose and treat local allergic rhinitis: A challenge for clinicians. J. Clin. Med. 2019, 8, 1062. [Google Scholar] [CrossRef] [PubMed]
- Trampert, D.C.; Hubers, L.M.; van de Graaf, S.F.J.; Beuers, U. On the role of IgG4 in inflammatory conditions: Lessons for IgG4-related disease. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 1401–1409. [Google Scholar] [CrossRef] [PubMed]
- Culver, E.L.; Vermeulen, E.; Makuch, M.; van Leeuwen, A.; Sadler, R.; Cargill, T.; Klenerman, P.; Aalberse, R.C.; van Ham, S.M.; Barnes, E.; et al. Increased IgG4 responses to multiple food and animal antigens indicate a polyclonal expansion and differentiation of pre-existing B cells in IgG4-related disease. Ann. Rheum. Dis. 2015, 74, 944–947. [Google Scholar] [CrossRef] [PubMed]
- Liew, P.X.; Kubes, P. The neutrophil’s role during health and disease. Physiol. Rev. 2019, 99, 1223–1248. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, H.F.; Dyer, K.D.; Foster, P.S. Eosinophils: Changing perspectives in health and disease. Nat. Rev. Immunol. 2013, 13, 9–22. [Google Scholar] [CrossRef]
- Poto, R.; Loffredo, S.; Marone, G.; Di Salvatore, A.; de Paulis, A.; Schroeder, J.T.; Varricchi, G. Basophils beyond allergic and parasitic diseases. Front. Immunol. 2023, 14, 1190034. [Google Scholar] [CrossRef] [PubMed]
- Kabashima, K.; Nakashima, C.; Nonomura, Y.; Otsuka, A.; Cardamone, C.; Parente, R.; De Feo, G.; Triggiani, M. Biomarkers for evaluation of mast cell and basophil activation. Immunol. Rev. 2018, 282, 114–120. [Google Scholar] [CrossRef]
- Velnar, T.; Bailey, T.; Smrkolj, V. The wound healing process: An overview of the cellular and molecular mechanisms. J. Int. Med. Res. 2009, 37, 1528–1542. [Google Scholar] [CrossRef]
- Woodrow, J.S.; Sheats, M.K.; Cooper, B.; Bayless, R. Asthma: The use of animal models and their translational utility. Cells 2023, 12, 1091. [Google Scholar] [CrossRef] [PubMed]
- Savage, J.H.; Courneya, J.-P.; Sterba, P.M.; Macglashan, D.W.; Saini, S.S.; Wood, R.A. Kinetics of mast cell, basophil, and oral food challenge responses in omalizumab-treated adults with peanut allergy. J. Allergy Clin. Immunol. 2012, 130, 1123–1129.e2. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.F.; Alpan, O.; Hoffmann, H.J. Basophil activation test: Mechanisms and considerations for use in clinical trials and clinical practice. Allergy 2021, 76, 2420–2432. [Google Scholar] [CrossRef] [PubMed]
- Butterworth, A.E. The eosinophil and its role in immunity to helminth infection. Curr. Top. Microbiol. Immunol. 1977, 77, 127–168. [Google Scholar] [CrossRef]
- Sil, A.; Bhanja, D.B.; Chandra, A.; Biswas, S.K. Loeffler’s syndrome and multifocal cutaneous larva migrans: Case report of an uncommon occurrence and review of the literature. Sultan Qaboos Univ. Med. J. 2023, 23, 104–108. [Google Scholar] [CrossRef]
- Gipsman, A.I.; Feld, L.; Johnson, B.; Needleman, J.P.; Boas, H.; Lin, N.; DePasquale, B.; Pogoriler, J.; McDowell, K.M.; Piccione, J.C. Eosinophilic plastic bronchitis: Case series and review of the literature. Pediatr. Pulmonol. 2023, 58, 3023–3031. [Google Scholar] [CrossRef]
- Marasco, G.; Visaggi, P.; Vassallo, M.; Fiocca, M.; Cremon, C.; Barbaro, M.R.; De Bortoli, N.; Bellini, M.; Stanghellini, V.; Savarino, E.V.; et al. Current and Novel Therapies for Eosinophilic Gastrointestinal Diseases. Int. J. Mol. Sci. 2023, 24, 15165. [Google Scholar] [CrossRef]
- Cafone, J.; Capucilli, P.; Hill, D.A.; Spergel, J.M. Eosinophilic esophagitis during sublingual and oral allergen immunotherapy. Curr. Opin. Allergy Clin. Immunol. 2019, 19, 350–357. [Google Scholar] [CrossRef]
- Hendriksen, E.; van Bergeijk, D.; Oosting, R.S.; Redegeld, F.A. Mast cells in neuroinflammation and brain disorders. Neurosci. Biobehav. Rev. 2017, 79, 119–133. [Google Scholar] [CrossRef]
- Cardamone, C.; Parente, R.; De Feo, G.; Triggiani, M. Mast cells as effector cells of innate immunity and regulators of adaptive immunity. Immunol. Lett. 2016, 178, 10–14. [Google Scholar] [CrossRef]
- Weller, K.; Foitzik, K.; Paus, R.; Syska, W.; Maurer, M. Mast cells are required for normal healing of skin wounds in mice. FASEB J. 2006, 20, 2366–2368. [Google Scholar] [CrossRef]
- Crivellato, E.; Ribatti, D. The mast cell: An evolutionary perspective. Biol. Rev. Camb. Philos. Soc. 2010, 85, 347–360. [Google Scholar] [CrossRef]
- Vitte, J.; Vibhushan, S.; Bratti, M.; Montero-Hernandez, J.E.; Blank, U. Allergy, anaphylaxis, and nonallergic hypersensitivity: IgE, mast cells, and beyond. Med. Princ. Pract. 2022, 31, 501–515. [Google Scholar] [CrossRef]
- Theoharides, T.; Kalogerometros, D. The critical role of mast cells in allergy and inflammation. Ann. N. Y. Acad. Sci. 2006, 1088, 78–99. [Google Scholar] [CrossRef]
- Góralczyk-Bińkowska, A.; Szmajda-Krygier, D.; Kozłowska, E. The microbiota-gut-brain axis in psychiatric disorders. Int. J. Mol. Sci. 2022, 23, 11245. [Google Scholar] [CrossRef]
- Farup, P.G.; Ueland, T.; Rudi, K.; Lydersen, S.; Hestad, K. Functional bowel disorders are associated with a central immune activation. Gastroenterol. Res. Pract. 2017, 2017, 1642912. [Google Scholar] [CrossRef] [PubMed]
- Dave, N.D.; Xiang, L.; Rehm, K.E.; Marshall, G.D., Jr. Stress and allergic diseases. Immunol. Allergy Clin. N. Am. 2011, 31, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Fang, F.; Tomasson, G.; Arnberg, F.K.; Mataix-Cols, D.; de la Cruz, L.F.; Almqvist, C.; Fall, K.; Valdimarsdóttir, U.A. Association of stress-related disorders with subsequent autoimmune disease. JAMA 2018, 319, 2388–2400. [Google Scholar] [CrossRef]
- Costa-Pinto, F.A.; Basso, A.S.; Britto, L.R.G.; Malucelli, B.E.; Russo, M. Avoidance behavior and neural correlates of allergen exposure in a murine model of asthma. Brain Behav. Immun. 2005, 19, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Manalai, P.; Hamilton, R.G.; Langenberg, P.; Kosisky, S.E.; Lapidus, M.; Sleemi, A.; Scrandis, B.; Cabassa, J.A.; Rogers, C.A.; Regenold, W.T.; et al. Pollen-specific immunoglobulin E positivity is associated with worsening of depression scores in bipolar disorder patients during high pollen season. Bipolar Disord. 2012, 14, 90–98. [Google Scholar] [CrossRef]
- Patterson, A.M.; Yildiz, V.O.; Klatt, M.D.; Malarkey, W.B. Perceived stress predicts allergy flares. Ann. Allergy Asthma Immunol. 2014, 112, 317–321. [Google Scholar] [CrossRef]
- Matatia, P.R.; Christian, E.; Sokol, C.L. Sensory sentinels: Neuroimmune detection and food allergy. Immunol. Rev. 2024, 326, 83–101. [Google Scholar] [CrossRef] [PubMed]
- Aich, A.; Afrin, L.B.; Gupta, K. Mast cell-mediated mechanisms of nociception. Int. J. Mol. Sci. 2015, 16, 29069–29092. [Google Scholar] [CrossRef]
- Castells, M.; Giannetti, M.P.; Hamilton, M.J.; Novak, P.; Pozdnyakova, O.; Nicoloro-SantaBarbara, J.; Jennings, S.V.; Francomano, C.; Kim, B.; Glover, S.C.; et al. Mast cell activation syndrome: Current understanding and research needs. J. Allergy Clin. Immunol. 2024, 154, 255–263. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Valent, P.; Akin, C. Mast cells, mastocytosis, and related disorders. N. Engl. J. Med. 2015, 373, 163–172. [Google Scholar] [CrossRef]
- Jones, M.K.; Nair, A.; Gupta, M. Mast cells in neurodegenerative disease. Front. Cell. Neurosci. 2019, 13, 171. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, G. Mast cell and autoimmune diseases. Mediators Inflamm. 2015, 2015, 246126. [Google Scholar] [CrossRef]
- Miller, F.G.; Rosenstein, D.L. The nature and power of the placebo effect. J. Clin. Epidemiol. 2006, 59, 331–335. [Google Scholar] [CrossRef]
- Björkman, B.; Lund, I.; Arnér, S.; Hydén, L.C. Phantom phenomena—Their perceived qualities and consequences from the patient’s perspective. Scand. J. Pain 2012, 3, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lu, X.; Bi, Y.; Hu, L. Pavlov’s pain: The effect of classical conditioning on pain perception and its clinical implications. Curr. Pain Headache Rep. 2019, 23, 19. [Google Scholar] [CrossRef] [PubMed]
- van Egmond, J.J. Beyond secondary gain. Am. J. Psychoanal. 2005, 65, 167–177. [Google Scholar] [CrossRef]
- Diaz, P.; Gonzalez, M.C.; Galleguillos, F.R.; Ancic, P.; Cromwell, O.; Shepherd, D.; Durham, S.R.; Gleich, G.J.; Kay, A.B. Leu-kocytes and mediators in bronchoalveolar lavage during allergen-induced late-phase asthmatic reactions. Am. Rev. Respir. Dis. 1989, 139, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Chupp, G. Phenotypes and endotypes of adult asthma: Moving toward precision medicine. J. Allergy Clin. Immunol. 2019, 144, 1–12. [Google Scholar] [CrossRef]
- Fainardi, V.; Esposito, S.; Chetta, A.; Pisi, G. Asthma phenotypes and endotypes in childhood. Minerva Med. 2022, 113, 94–105. [Google Scholar] [CrossRef]
- Rampelotto, P.H. Extremophiles and extreme environments. Life 2013, 3, 482–485. [Google Scholar] [CrossRef]
- Gilbert, J.A.; Stephens, B. Microbiology of the built environment. Nat. Rev. Microbiol. 2018, 16, 661–670. [Google Scholar] [CrossRef]
- Chung, H.; Pamp, S.J.; Hill, J.A.; Surana, N.K.; Edelman, S.M.; Troy, E.B.; Reading, N.C.; Villablanca, E.J.; Wang, S.; Mora, J.R.; et al. Gut immune maturation depends on colonization with a host-specific microbiota. Cell 2012, 149, 1578–1593. [Google Scholar] [CrossRef] [PubMed]
- Saeed, N.K.; Al-Beltagi, M.; Bediwy, A.S.; El-Sawaf, Y.; Toema, O. Gut microbiota in various childhood disorders: Implication and indications. World J. Gastroenterol. 2022, 28, 1875–1901. [Google Scholar] [CrossRef] [PubMed]
- Dethlefsen, L.; Huse, S.; Sogin, M.L.; Relman, D.A. The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16S rRNA sequencing. PLoS Biol. 2008, 6, e280. [Google Scholar] [CrossRef]
- Perkin, M.R.; Strachan, D.P. The hygiene hypothesis for allergy—Conception and evolution. Front. Allergy 2022, 3, 1051368. [Google Scholar] [CrossRef]
- Shreiner, A.; Huffnagle, G.B.; Noverr, M.C. The “microflora hypothesis” of allergic diseases. Adv. Exp. Med. Biol. 2008, 635, 113–134. [Google Scholar] [CrossRef] [PubMed]
- Akdis, C.A. Does the epithelial barrier hypothesis explain the increase in allergy, autoimmunity and other chronic conditions? Nat. Rev. Immunol. 2021, 21, 739–751. [Google Scholar] [CrossRef]
- Buchanan, W.W. Rheumatoid arthritis: Another New World disease? Semin. Arthritis Rheum. 1994, 23, 289–294. [Google Scholar] [CrossRef]
- Kawasaki, T.; Naoe, S. History of Kawasaki disease. Clin. Exp. Nephrol. 2014, 18, 301–304. [Google Scholar] [CrossRef]
- Burns, J.C.; Kushner, H.I.; Bastian, J.F.; Shike, H.; Shimizu, C.; Matsubara, T.; Turner, C.L. Kawasaki disease: A brief history. Pediatrics 2000, 106, e27. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Han, J.W.; Lee, J.S. Kawasaki disease may be a hyperimmune reaction of genetically susceptible children to variants of normal environmental flora. Med. Hypotheses 2007, 69, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Rhim, J.W.; Kang, H.M.; Han, J.W.; Lee, K.Y. A presumed etiology of Kawasaki disease based on epidemiological comparison with infectious or immune-mediated diseases. Front. Pediatr. 2019, 7, 202. [Google Scholar] [CrossRef]
- Hooks, K.B.; O’Malley, M.A. Dysbiosis and its discontents. mBio 2017, 8, e01492-17. [Google Scholar] [CrossRef]
- NIH Human Microbiome Portfolio Analysis Team. A review of 10 years of human microbiome research activities at the US National Institutes of Health, Fiscal Years 2007–2016. Microbiome 2019, 7, 31. [Google Scholar] [CrossRef]
- Mattick, J.S.; Dziadek, M.A.; Terrill, B.N.; Kaplan, W.; Spigelman, A.D.; Bowling, F.G.; Dinger, M.E. The impact of genomics on the future of medicine and health. Med. J. Aust. 2014, 201, 17–20. [Google Scholar] [CrossRef]
- Nayfach, S.; Shi, Z.J.; Seshadri, R.; Pollard, K.S.; Kyrpides, N. New insights from uncultivated genomes of the global human gut microbiome. Nature 2019, 568, 505–510. [Google Scholar] [CrossRef]
- Lagier, J.C.; Hugon, P.; Khelaifia, S.; Fournier, P.E.; La Scola, B.; Raoult, D. The rebirth of culture in microbiology through the example of culturomics to study human gut microbiota. Clin. Microbiol. Rev. 2015, 28, 237–264. [Google Scholar] [CrossRef] [PubMed]
- Castillo, D.J.; Rifkin, R.F.; Cowan, D.A.; Potgieter, M. The healthy human blood microbiome: Fact or fiction? Front. Cell Infect. Microbiol. 2019, 9, 148. [Google Scholar] [CrossRef]
- Hammad, D.B.M.; Liyanapathirana, V.; Tonge, D.P. Molecular characterisation of the synovial fluid microbiome in rheumatoid arthritis patients and healthy control subjects. PLoS ONE 2019, 14, e0225110. [Google Scholar] [CrossRef]
- Kim, S.Y.; Yi, D.Y. Components of human breast milk: From macronutrient to microbiome and microRNA. Clin. Exp. Pediatr. 2020, 63, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Kil, H.R.; Yu, J.W.; Lee, S.C.; Rhim, J.W.; Lee, K.Y. Changes in clinical and laboratory features of Kawasaki disease noted over time in Daejeon, Korea. Pediatr. Rheumatol. Online J. 2017, 15, 60. [Google Scholar] [CrossRef]
- Rhim, J.W.; Lee, Y.T.; Kang, H.M.; Suh, J.S.; Lee, K.Y. Changes in clinical features in Henoch–Schönlein purpura during three decades: An observational study at a single hospital in Korea. Clin. Rheumatol. 2019, 38, 2811–2818. [Google Scholar] [CrossRef]
- Rhim, J.W.; Kang, H.M.; Yang, E.A.; Lee, K.Y. Epidemiological relationship between Mycoplasma pneumoniae pneumonia and recurrent wheezing episode in children: An observational study at a single hospital in Korea. BMJ Open 2019, 9, e026461. [Google Scholar] [CrossRef] [PubMed]
- Cunha, B.A.; Burillo, A.; Bouza, E. Legionnaires’ disease. Lancet 2016, 387, 376–385. [Google Scholar] [CrossRef]
- Plaza, P.I.; Gamarra-Toledo, V.; Euguí, J.R.; Lambertucci, S.A. Recent changes in patterns of mammal infection with highly pathogenic avian influenza A(H5N1) virus worldwide. Emerg. Infect. Dis. 2024, 30, 444–452. [Google Scholar] [CrossRef]
- Chisholm, R.H.; Campbell, P.T.; Wu, Y.; Tong, S.Y.C.; McVernon, J.; Geard, N. Implications of asymptomatic carriers for in-fectious disease transmission and control. R. Soc. Open Sci. 2018, 5, 172341. [Google Scholar] [CrossRef]
- Pretorius, E.; Akeredolu, O.-O.; Soma, P.; Kell, D.B. Major involvement of bacterial components in rheumatoid arthritis and its accompanying oxidative stress, systemic inflammation and hypercoagulability. Exp. Biol. Med. 2017, 242, 355–373. [Google Scholar] [CrossRef]
- Gianchecchi, E.; Fierabracci, A. Recent advances on microbiota involvement in the pathogenesis of autoimmunity. Int. J. Mol. Sci. 2019, 20, 283. [Google Scholar] [CrossRef]
- Earl, C.S.; An, S.; Ryan, R.P. The changing face of asthma and its relation with microbes. Trends Microbiol. 2015, 23, 408–418. [Google Scholar] [CrossRef]
- Hua, X.; Goedert, J.J.; Pu, A.; Yu, G.; Shi, J. Allergy associations with the adult fecal microbiota: Analysis of the American Gut Project. eBioMedicine 2016, 3, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Descotes, J.; Choquet-Kastylevsky, G. Gell and Coombs’s classification: Is it still valid? Toxicology 2001, 158, 43–49. [Google Scholar] [CrossRef]
- Giavina-Bianchi, P.; Vivolo Aun, M.; Takejima, P.; Kalil, J.; Agondi, R.C. United airway disease: Current perspectives. J. Asthma Allergy 2016, 9, 93–100. [Google Scholar] [CrossRef]
- Taniuchi, S.; Soejima, K.; Hatano, Y.; Takahashi, M.; Minami, H. Dual factors may be necessary for development of atopic march in early infancy. J. Nippon Med. Sch. 2018, 85, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Reed, C.E. The natural history of asthma. J. Allergy Clin. Immunol. 2006, 118, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Rajvanshi, N.; Kumar, P.; Goyal, J.P. Global Initiative for Asthma Guidelines 2024: An update. Indian Pediatr. 2024, 61, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.; Camiolo, M.; Fitzpatrick, A.; Gauthier, M.; Wenzel, S.E. Are we meeting the promise of endotypes and precision medicine in asthma? Physiol. Rev. 2020, 100, 983–1017. [Google Scholar] [CrossRef]
- Anonymous. Psychogenic factors in bronchial asthma. Br. Med. J. 1943, 1, 416. [Google Scholar] [CrossRef] [PubMed]
- Ledford, D.K.; Lockey, R.F. Asthma and comorbidities. Curr. Opin. Allergy Clin. Immunol. 2013, 13, 78–86. [Google Scholar] [CrossRef]
- Thomas, M.; Bruton, A.; Moffat, M.; Cleland, J. Asthma and psychological dysfunction. Prim. Care Respir. J. 2011, 20, 250–256. [Google Scholar] [CrossRef]
- Dalziel, S.R.; Haskell, L.; O’Brien, S.; Borland, M.L.; Plint, A.C.; Babl, F.E.; Oakley, E. Bronchiolitis. Lancet 2022, 400, 392–406. [Google Scholar] [CrossRef]
- Savin, I.A.; Zenkova, M.A.; Sen’kova, A.V. Bronchial Asthma, Airway Remodeling and Lung Fibrosis as Successive Steps of One Process. Int. J. Mol. Sci. 2023, 24, 16042. [Google Scholar] [CrossRef]
- Kwah, J.H.; Peters, A.T. Nasal polyps and rhinosinusitis. Allergy Asthma Proc. 2019, 40, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Schuler, C.F., IV; Billi, A.C.; Maverakis, E.; Tsoi, L.C.; Gudjonsson, J.E. Novel insights into atopic dermatitis. J. Allergy Clin. Immunol. 2023, 151, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Correa, J.; Rodriguez, A. Divergent Roles of Antiself Antibodies during Infection. Trends Immunol. 2018, 39, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Avrameas, S.; Alexopoulos, H.; Moutsopoulos, H.M. Natural Autoantibodies: An Undersugn Hero of the Immune System and Autoimmune Disorders—A Point of View. Front. Immunol. 2018, 9, 1320. [Google Scholar] [CrossRef]
- Tedeschi, A.; Asero, R. Asthma and autoimmunity: A complex but intriguing relation. Expert Rev. Clin. Immunol. 2008, 4, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Krauss-Etschmann, S.; Petersen, F.; Yu, X. Autoantibodies in Chronic Obstructive Pulmonary Disease. Front. Immunol. 2018, 9, 66. [Google Scholar] [CrossRef]
- Cojocaru, M.; Cojocaru, I.M.; Silosi, I.; Vrabie, C.D. Pulmonary manifestations of systemic autoimmune diseases. Maedica 2011, 6, 224–229. [Google Scholar]
- Mendell, M.J.; Mirer, A.G.; Cheung, K.; Tong, M.; Douwes, J. Respiratory and allergic health effects of dampness, mold, and dampness-related agents: A review of the epidemiologic evidence. Environ. Health Perspect. 2011, 119, 748–756. [Google Scholar] [CrossRef]
- Molyneaux, P.L.; Maher, T.M. The role of infection in the pathogenesis of idiopathic pulmonary fibrosis. Eur. Respir. Rev. 2013, 22, 376–381. [Google Scholar] [CrossRef]
- Brownell, I.; Ramírez-Valle, F.; Sanchez, M.; Prystowsky, S. Evidence for mycobacteria in sarcoidosis. Am. J. Respir. Cell Mol. Biol. 2011, 45, 899–905. [Google Scholar] [CrossRef]
- Bernstein, J.A.; Llanos, J.P.; Hunter, G.; Martin, N.; Ambrose, C.S. Efficacy of Biologics in Patients with Allergic Severe Asthma, Overall and by Blood Eosinophil Count: A Literature Review. Adv. Ther. 2023, 40, 4721–4740. [Google Scholar] [CrossRef]
- McGregor, M.C.; Krings, J.G.; Nair, P.; Castro, M. Role of Biologics in Asthma. Am. J. Respir. Crit. Care Med. 2019, 199, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Armario-Hita, J.C.; Galán-Gutiérrez, M.; Dodero-Anillo, J.M.; Carrascosa, J.M.; Ruiz-Villaverde, R. Updated Review on Treatment of Atopic Dermatitis. J. Investig. Allergol. Clin. Immunol. 2023, 33, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Bleecker, E.R.; Menzies-Gow, A.N.; Price, D.B.; Bourdin, A.; Sweet, S.; Martin, A.L.; Alacqua, M.; Tran, T.N. Systematic Literature Review of Systemic Corticosteroid Use for Asthma Management. Am. J. Respir. Crit. Care Med. 2020, 201, 276–293. [Google Scholar] [CrossRef]
- Dahl, R. Systemic side effects of inhaled corticosteroids in patients with asthma. Respir. Med. 2006, 100, 1307–1317. [Google Scholar] [CrossRef] [PubMed]
- Marron, R.M.; Vega Sanchez, M.E. Asthma-COPD Overlap Syndrome. Chronic Obstr. Pulm. Dis. 2019, 6, 200–202. [Google Scholar] [CrossRef]
- Dhami, S.; Kakourou, A.; Asamoah, F.; Agache, I.; Lau, S.; Jutel, M.; Muraro, A.; Roberts, G.; Akdis, A.; Bonini, M.; et al. Allergen immunotherapy for allergic asthma: A systematic review and meta-analysis. Allergy 2017, 72, 1825–1848. [Google Scholar] [CrossRef]
- Dunn, R.M.; Busse, P.J.; Wechsler, M.E. Asthma in the elderly and late-onset adult asthma. Allergy 2018, 73, 284–294. [Google Scholar] [CrossRef]
- Kew, K.M.; Nashed, M.; Dulay, V.; Yorke, J. Cognitive behavioural therapy (CBT) for adults and adolescents with asthma. Cochrane Database Syst. Rev. 2016, 9, CD011818. [Google Scholar] [CrossRef]
- Konzett, V.; Aletaha, D. Management strategies in rheumatoid arthritis. Nat. Rev. Rheumatol. 2024, 20, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.A.; Kang, H.M.; Rhim, J.W.; Kang, J.H.; Lee, K.Y. Early Corticosteroid Therapy for Mycoplasma pneumoniae Pneumonia Irrespective of Used Antibiotics in Children. J. Clin. Med. 2019, 8, 726. [Google Scholar] [CrossRef] [PubMed]
| Immune Components and Effectors (or Events) | Main Functions |
|---|---|
| Adaptive immune system | |
| T cells | Control of pathogenic peptides targeted to host cells, including cancer cells, through production of cytokines (possibly peptides) and immune effectors within the cells in the MHC-restricted (TCR-associated) and non-MHC-restricted events. |
| B cells | Control of pathogenic proteins targeted to host cells, including cancer cells, by production of antibodies in the MHC-restricted (BCR-associated) and non-MHC-restricted events. |
| Innate immune system | |
| Natural killer cells | Control of transformed cells such as virus-infected cells and tumor cells through their recognizing receptors and immune effectors within the cells. |
| Tissue macrophage-linaeged cells | Antigen presentation to adaptive immune cells in the MHC-restricted immune responses. Possible control of communications between immune cells and affected organ cells including cancers cells in TMEs [13]. |
| Phagocytes (neutrophils and circulating monocyte/macrophages) | Control of large complex substances such as viruses, bacteria, parasites, and apoptotic & necrotic bodies associated with cell injury caused by infectin, trauma or other conditions. |
| Mast cells, basophils, eosionphils | These cells are activated by mainly external toxic substances that are recognized by receptors of the cells and control the substances through immune effetors within the cells and inflammation involved in these cells. |
| Unidentified innate immune components against small non-protein toxic materials | There are non-protein toxic or inflammation-inducing substances, including elements, monoamins, neuropeptides, LPS, RNAs, DNAs, chemicals and biochemicals. TLR-associated immune responses, natural antibodies, and immun proteins and/or peptides control these diverse substances. The immune proteins, including PrP gene products and amyloid proteins, control pathogenic monoamine metabolites or neuropeptides especially in CNS [12]. |
| Production of alternative proteins in genetic diseases and cancer | The systemic and intracellular PHS control in part insults from a protein deficiency or malfunctional protein in organ tissues or within a cell [7,12,13]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, Y.; Lee, K.-Y. A Unified Pathogenesis of Allergic Diseases; The Protein–Homeostasis–System Hypothesis. Int. J. Mol. Sci. 2025, 26, 8358. https://doi.org/10.3390/ijms26178358
Ko Y, Lee K-Y. A Unified Pathogenesis of Allergic Diseases; The Protein–Homeostasis–System Hypothesis. International Journal of Molecular Sciences. 2025; 26(17):8358. https://doi.org/10.3390/ijms26178358
Chicago/Turabian StyleKo, Younhee, and Kyung-Yil Lee. 2025. "A Unified Pathogenesis of Allergic Diseases; The Protein–Homeostasis–System Hypothesis" International Journal of Molecular Sciences 26, no. 17: 8358. https://doi.org/10.3390/ijms26178358
APA StyleKo, Y., & Lee, K.-Y. (2025). A Unified Pathogenesis of Allergic Diseases; The Protein–Homeostasis–System Hypothesis. International Journal of Molecular Sciences, 26(17), 8358. https://doi.org/10.3390/ijms26178358







