Retinoic Acid-Induced Transglutaminase 2 Expression Reduces Sensitivity to Cisplatin in the Hormone-Positive MCF-7 Breast Cancer Cell Model
Abstract
1. Introduction
2. Results
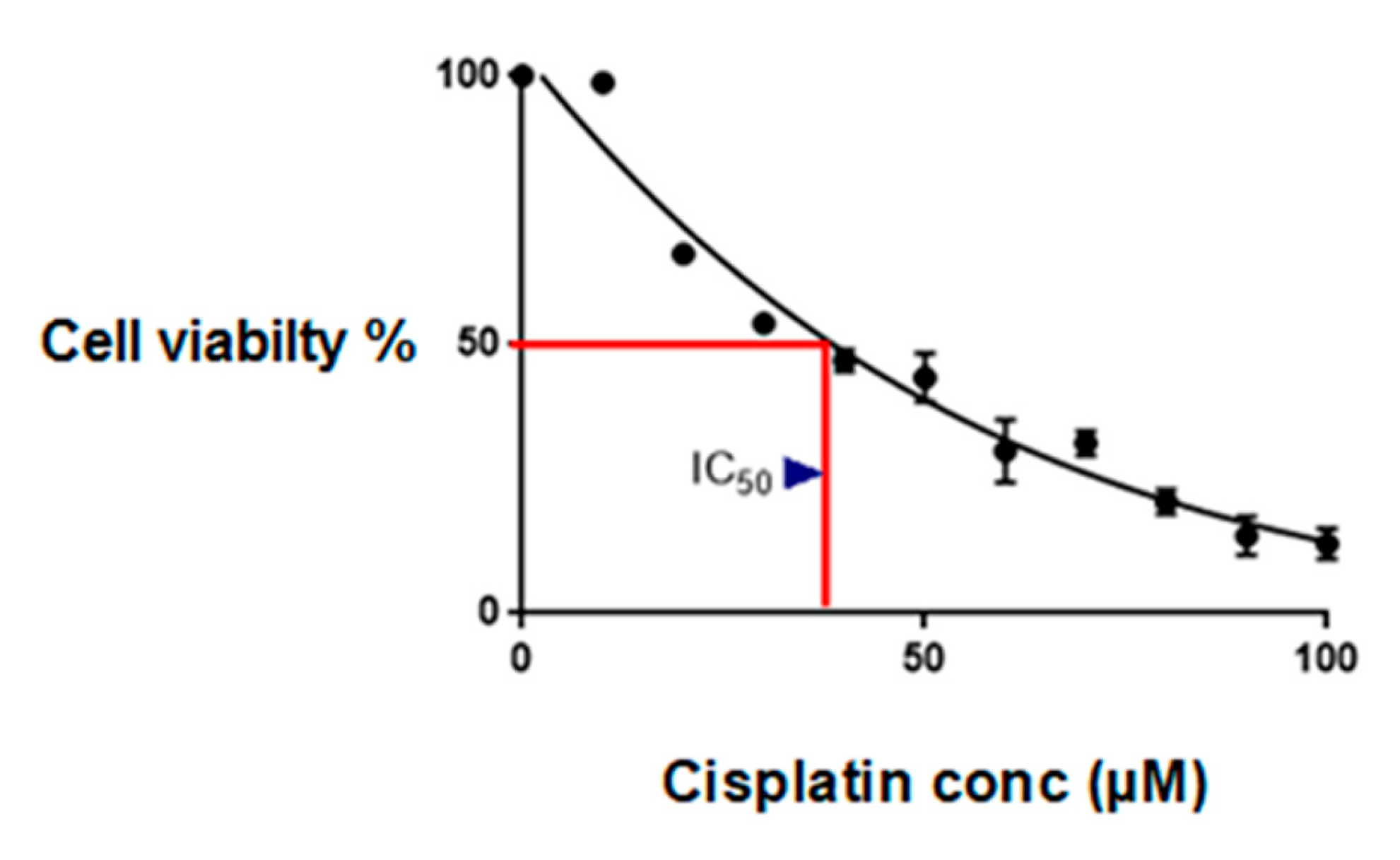
2.1. Establishment of the Inhibitory Concentration of Cisplatin
2.1.1. Establishment of Cisplatin-Surviving MCF-7 Cells (cMCF-7)
2.1.2. TG2 Levels Are Increased in Cisplatin-Surviving Cells
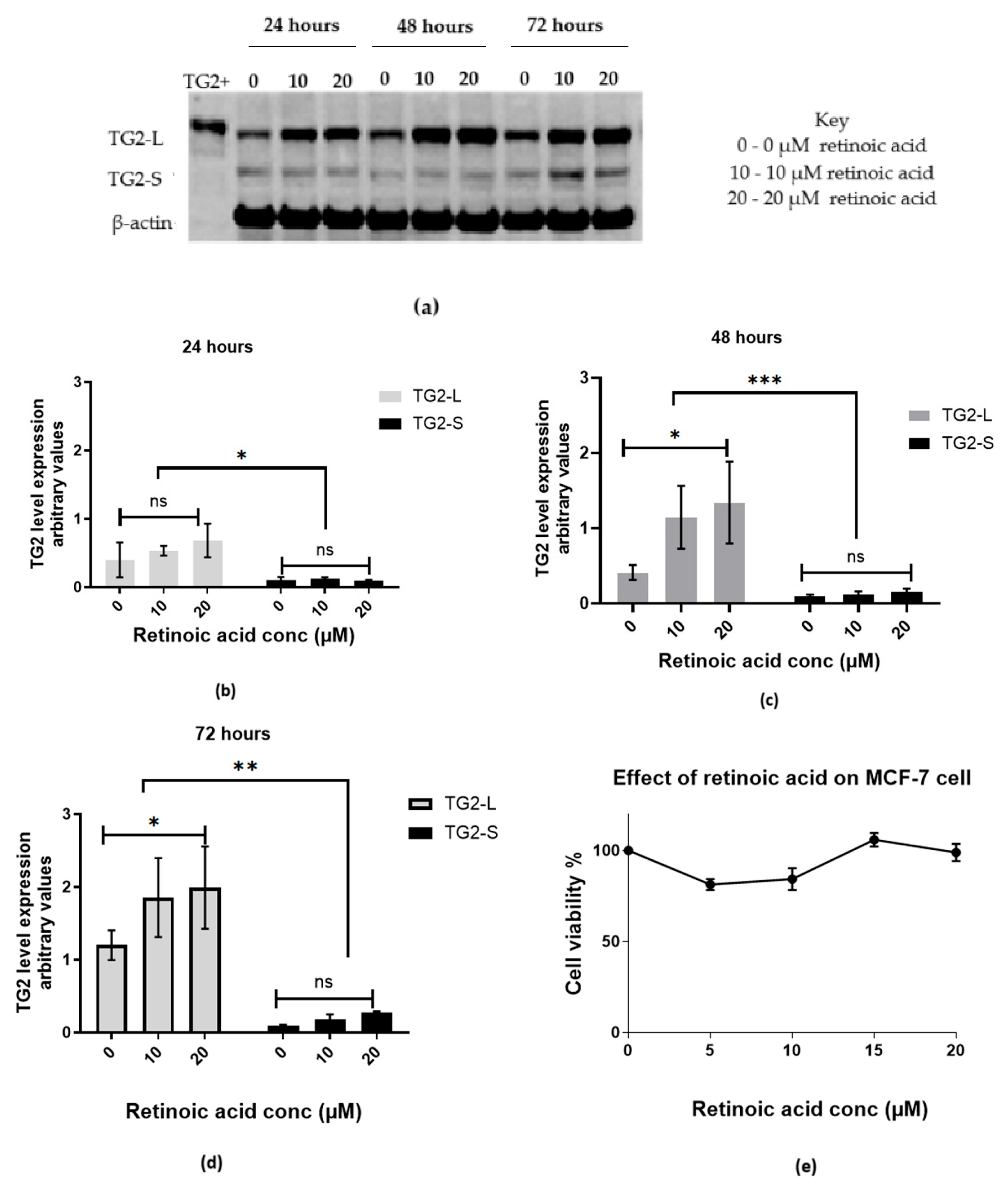
2.2. Retinoic Acid Increases TG2-L Expression
2.2.1. Retinoic Acid Increases TG2-L Levels and Reduces Cisplatin Cytotoxicity in MCF-7 Wild-Type Cells
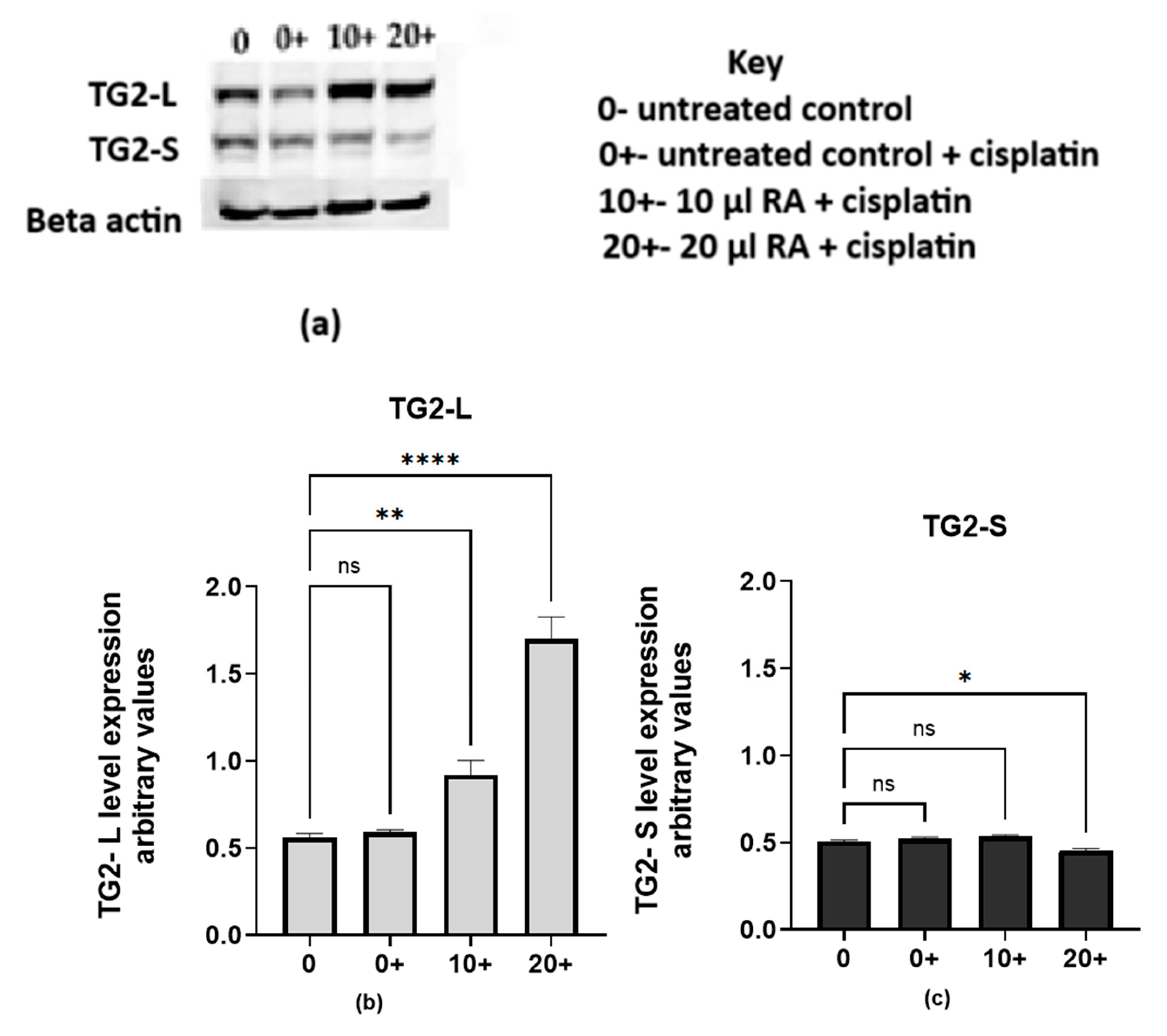
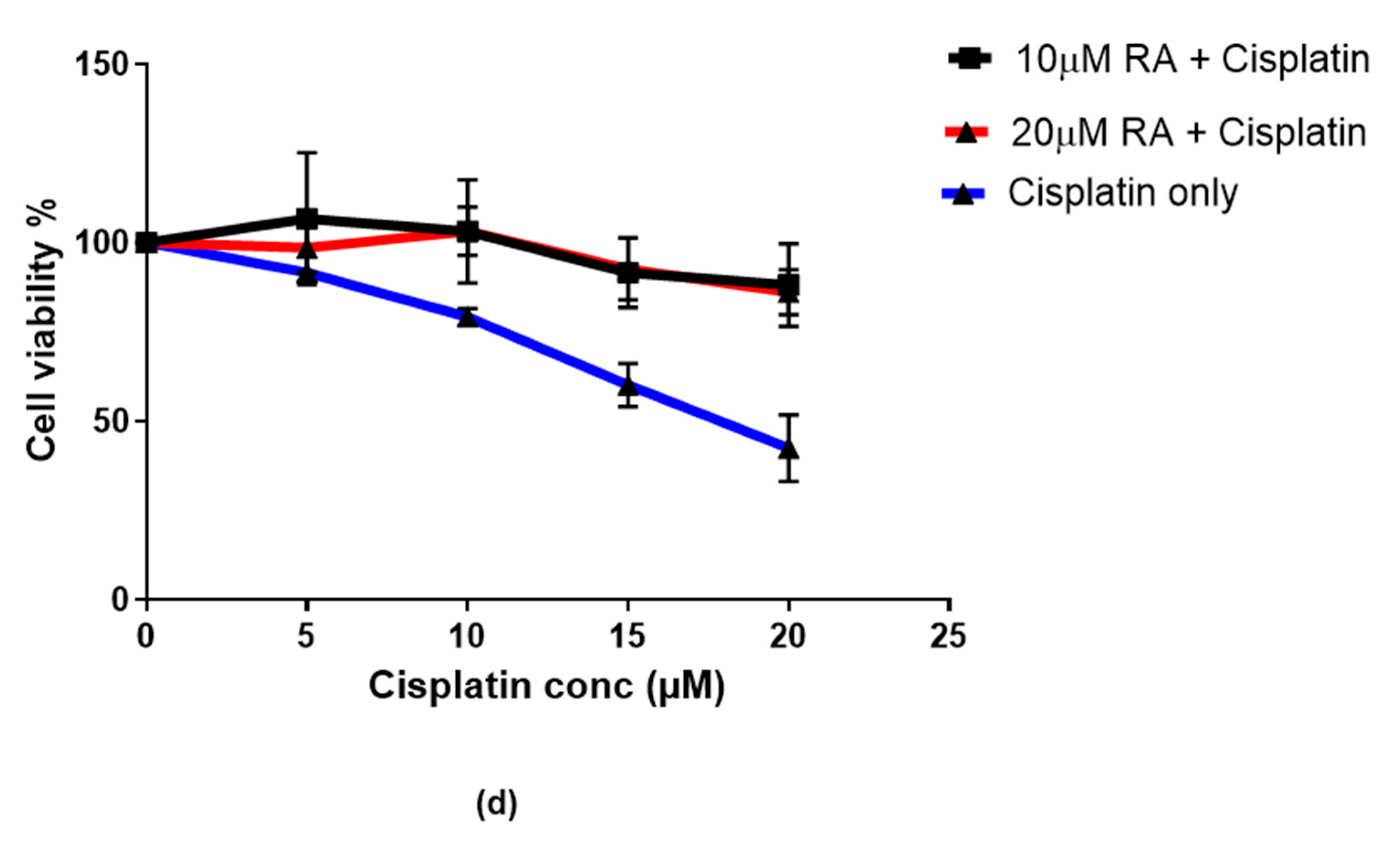
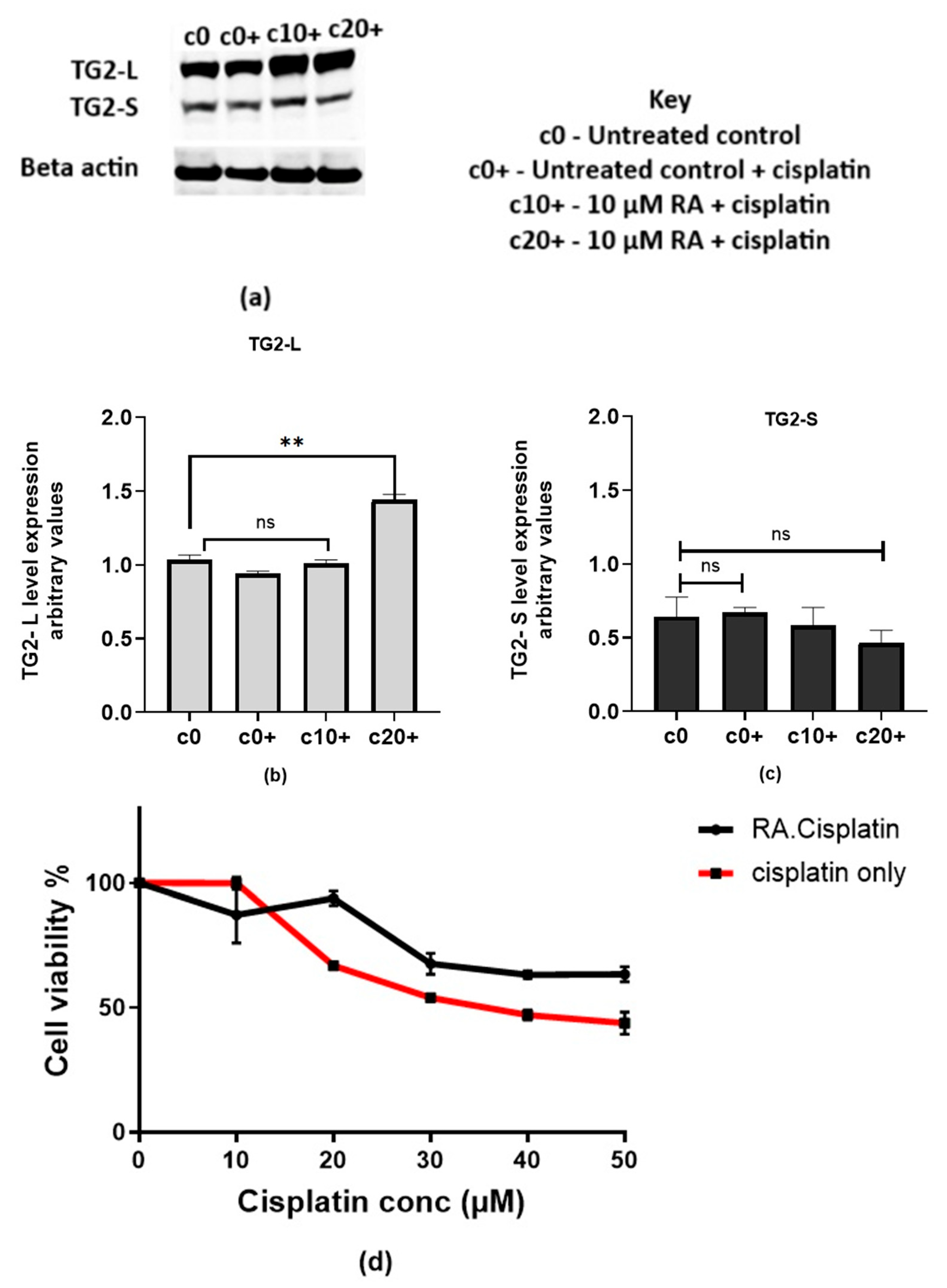
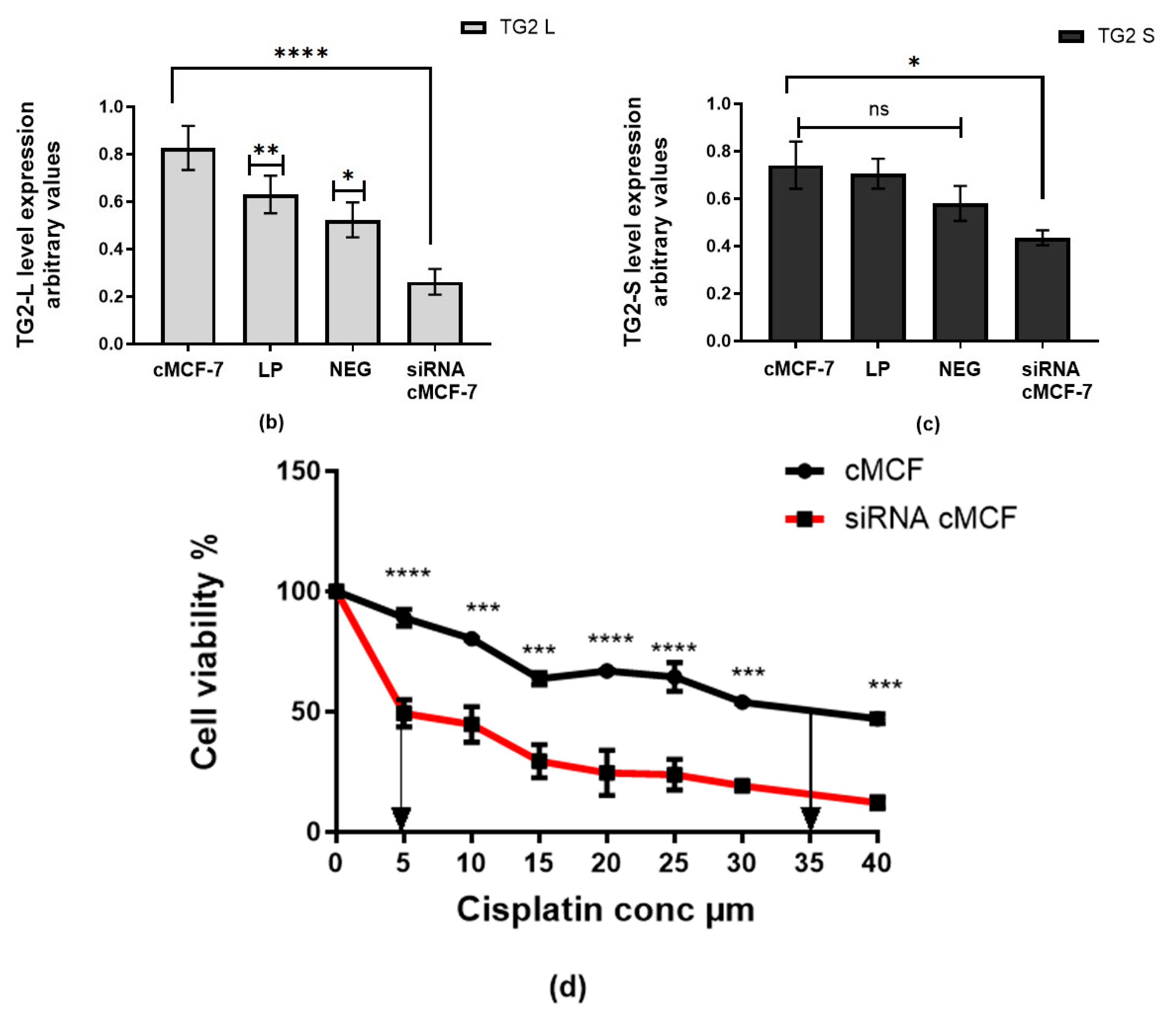
2.2.2. Retinoic Acid Increases TG2-L Levels and Reduces Cisplatin Cytotoxicity in Cisplatin-Surviving Cells (cMCF-7)
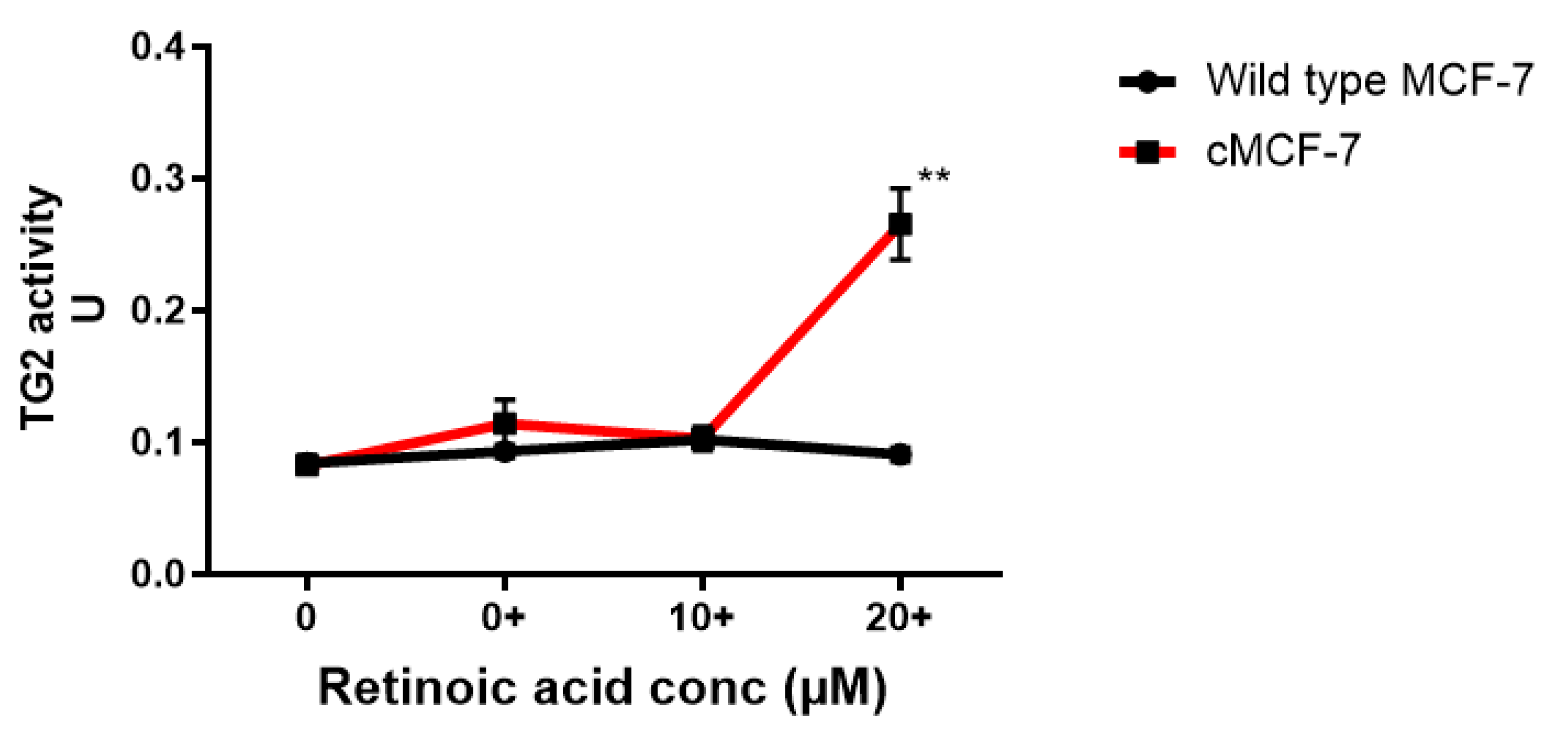
2.3. TG2 Enzyme Activity
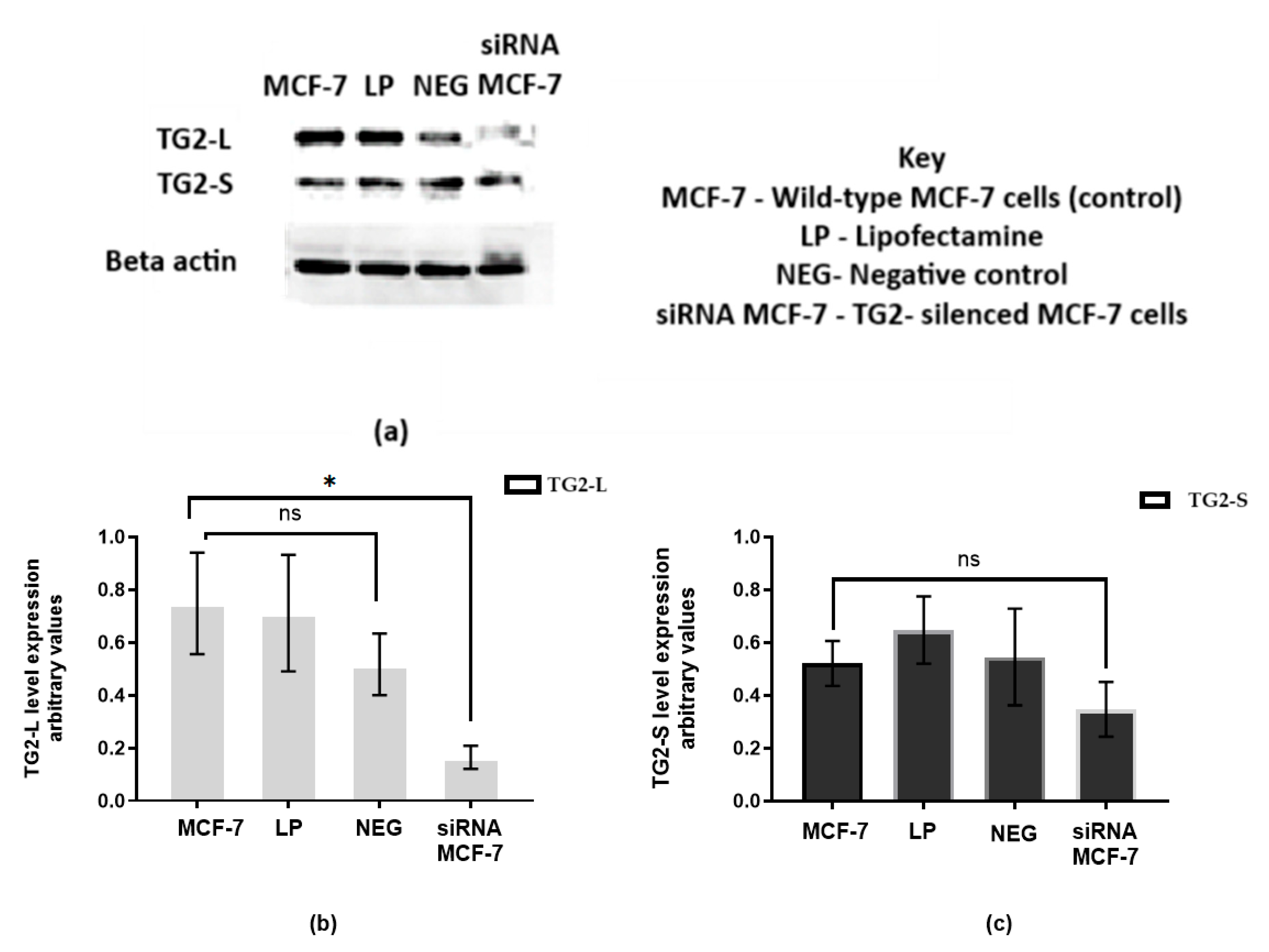
2.4. Transfection with TG2-Specific siRNA Increases Cisplatin Cytotoxicity in Wild-Type MCF-7 Cells
3. Discussion
Limitations
4. Materials and Methods
4.1. Pharmacologic Agents
4.2. Cell Culture
4.3. Development of a Cisplatin-Surviving MCF-7 Sub-Line
4.4. Cell Viability Assay/IC50 Toxicity Assay
4.5. Wound-Healing “Scratch” Assay
4.6. Treatment with Retinoic Acid and/or Cisplatin
4.7. Tissue Transglutaminase Enzyme Activity
4.8. TG2 Inhibition by siRNA
4.9. Apoptosis Analysis
4.10. Western Blot Analysis
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| RA | Retinoic Acid |
| RARs | Retinoic Acid Receptors |
| RXRs | Retinoid X Receptors |
| RAREs | Retinoic Acid Response Elements |
| atRA | All-Trans Retinoic Acid |
| TG2 | Tissue Transglutaminase 2 |
| TG2-L | Tissue Transglutaminase 2, Long Isoform |
| TG2-S | Tissue Transglutaminase 2, Short Isoform |
| Ca2+ | Calcium Ions |
| GTP | Guanosine Triphosphate |
| MCF-7 | Michigan Cancer Foundation-7 (a breast cancer cell line) |
| ERα | Oestrogen Receptor Alpha |
| NF-κB | Nuclear Factor Kappa-Light-Chain-Enhancer of Activated B Cells |
| PPARs | Peroxisome Proliferator-Activated Receptors |
| AML | Acute Myeloid Leukaemia |
| CCK-6 | Cell Counting Kit-6 |
| IC50 | Half Maximal Inhibitory Concentration |
| cMCF-7 | Cisplatin-surviving MCF-7 Cells |
| AA9 | A TG2 Inhibitor |
| DMEM | Dulbecco’s Modified Eagle Medium |
| FBS | Foetal Bovine Serum |
| ATCC | American Type Culture Collection |
| CO2 | Carbon Dioxide |
| MTT | Methylthiazolyldiphenyl-tetrazolium Bromide |
| ZOE™ | Fluorescent Cell Imager (Brand) |
| FITC | Fluorescein Isothiocyanate |
| PI | Propidium Iodide |
| FACS | Fluorescence-Activated Cell Sorting |
| FSC | Forward Scatter |
| SSC | Side Scatter |
| RIPA | Radioimmunoprecipitation Assay Buffer |
| TBST | TBS-Tween 20 |
| HRP | Horseradish Peroxidase |
| ECL | Enhanced Chemiluminescence |
References
- Schubert, M.; Germain, P. Retinoic acid and retinoid X receptors. Cells 2023, 12, 864. [Google Scholar] [CrossRef] [PubMed]
- Vilhais-Neto, G.C.; Pourquié, O. Retinoic acid. Curr. Biol. 2008, 18, R191–R192. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Fan, Y.; Lin, L.; Kellems, R.E.; Xia, Y. Tissue transglutaminase: A multifunctional and multisite regulator in health and disease. Physiol. Rev. 2024, 104, 281–325. [Google Scholar] [CrossRef] [PubMed]
- Tatsukawa, H.; Hitomi, K. Role of transglutaminase 2 in cell death, survival, and fibrosis. Cells 2021, 10, 1842. [Google Scholar] [CrossRef]
- Ciulla, M.G.; Marchini, A.; Gazzola, J.; Forouharshad, M.; Pugliese, R.; Gelain, F. In situ transglutaminase cross-linking improves mechanical properties of self-assembling peptides for biomedical applications. ACS Appl. Bio Mater. 2024, 7, 1723–1734. [Google Scholar] [CrossRef]
- Ivashchenko, S.D.; Shulga, D.A.; Ivashchenko, V.D.; Zinovev, E.V.; Vlasov, A.V. In silico studies of the open form of human tissue transglutaminase. Sci. Rep. 2024, 10, 15981. [Google Scholar] [CrossRef]
- Liu, J.; Mouradian, M.M. Pathogenetic contributions and therapeutic implications of transglutaminase 2 in neurodegenerative diseases. Int. J. Mol. Sci. 2024, 25, 2364. [Google Scholar] [CrossRef]
- Phatak, V.M.; Croft, S.M.; Rameshaiah Setty, S.G.; Scarpellini, A.; Hughes, D.C.; Rees, R.; McArdle, S.; Verderio, E.A.M. Expression of transglutaminase-2 isoforms in normal human tissues and cancer cell lines: Dysregulation of alternative splicing in cancer. Amino Acids 2013, 44, 33–44. [Google Scholar] [CrossRef]
- Zaltron, E.; Vianello, F.; Ruzza, A.; Palazzo, A.; Brillo, V.; Celotti, I.; Scavezzon, M.; Rossin, F.; Leanza, L.; Severin, F. The Role of Transglutaminase 2 in Cancer: An Update. Int. J. Mol. Sci. 2024, 25, 2797. [Google Scholar] [CrossRef]
- Antonyak, M.A.; Jansen, J.M.; Miller, A.M.; Ly, T.K.; Endo, M.; Cerione, R.A. Two isoforms of tissue transglutaminase mediate opposing cellular fates. Proc. Natl. Acad. Sci. USA 2006, 103, 18609–18614. [Google Scholar] [CrossRef]
- Currò, M.; Nadia, F.; Maria, L.G.; Angela, S.M.; Tiziana, R.; Salvatore, A.; Pietro, I.; Daniela, C.; Carmelo, R.; Riccardo, I. Hypoxia-dependent expression of TG2 isoforms in neuroblastoma cells as consequence of different MYCN amplification status. Int. J. Mol. Sci. 2020, 4, 1364. [Google Scholar] [CrossRef]
- Rudlong, J.; Anson, C.; Gail, V.W. The role of transglutaminase 2 in mediating glial cell function and pathophysiology in the central nervous system. Anal. Biochem. 2020, 591, 113556. [Google Scholar] [CrossRef]
- Chiocca, E.A.; Davies, P.J.; Stein, J.P. The molecular basis of retinoic acid action. Transcriptional regulation of tissue transglutaminase gene expression in macrophages. J. Biol. Chem. 1988, 263, 11584–11589. [Google Scholar] [CrossRef] [PubMed]
- Piacentini, M.; Ceru, M.P.; Dini, L.; Di Rao, M.; Piredda, L.; Thomazy, V.; Davies, P.J.; Fesus, L. In vivo and in vitro induction of ‘tissue’transglutaminase in rat hepatocytes by retinoic acid. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 1992, 1135, 171–179. [Google Scholar] [CrossRef]
- Meshram, D.D.; Pike, C.V.S.; Coussons, P.J. Inhibition of Transglutaminase 2 activity increases cisplatin cytotoxicity in a model of human hepatocarcinoma chemotherapy. Eur. J. Pharmacol. 2017, 815, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Livingston, M.J.; Safirstein, R.; Dong, Z. Cisplatin nephrotoxicity: New insights and therapeutic implications. Nat. Rev. Nephrol. 2023, 19, 53–72. [Google Scholar] [CrossRef]
- Hartmann, J.T.; Fels, L.M.; Knop, S.; Stolte, H.; Kanz, L.; Bokemeyer, C. A randomized trial comparing the nephrotoxicity of cisplatin/ifosfamide-based combination chemotherapy with or without amifostine in patients with solid tumors. Investig. New Drugs 2000, 18, 281–289. [Google Scholar] [CrossRef]
- Gao, Y.; Chen, D.L.; Zhou, M.; Zheng, Z.S.; He, M.F.; Huang, S.; Liao, X.Z.; Zhang, J.X. Cordycepin enhances the chemosensitivity of esophageal cancer cells to cisplatin by inducing the activation of AMPK and suppressing the AKT signaling pathway. Cell Death Dis. 2020, 11, 866. [Google Scholar] [CrossRef]
- Zoń, A.; Bednarek, I. Cisplatin in ovarian cancer treatment—Known limitations in therapy force new solutions. Int. J. Mol. Sci. 2023, 24, 7585. [Google Scholar] [CrossRef]
- Romani, A.M. Cisplatin in cancer treatment. Biochem. Pharmacol. 2022, 206, 115323. [Google Scholar] [CrossRef]
- Lorusso, D.; Petrelli, F.; Coinu, A.; Raspagliesi, F.; Barni, S. A systematic review comparing cisplatin and carboplatin plus paclitaxel-based chemotherapy for recurrent or metastatic cervical cancer. Gynecol. Oncol. 2014, 133, 117–123. [Google Scholar] [CrossRef]
- Federico, C.; Sun, J.; Muz, B.; Alhallak, K.; Cosper, P.F.; Muhammad, N.; Jeske, A.; Hinger, A.; Markovina, S.; Grigsby, P.; et al. Localized delivery of cisplatin to cervical cancer improves its therapeutic efficacy and minimizes its side effect profile. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Silvestre, A.; Garcia-Venzor, A.; Ceballos-Cancino, G.; Sánchez-López, J.M.; Vazquez-Santillan, K.; Mendoza-Almanza, G.; Lizarraga, F.; Melendez-Zajgla, J.; Maldonado, V. Transcriptomic changes in cisplatin-resistant MCF-7 cells. Int. J. Mol. Sci. 2024, 25, 3820. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Guo, S.; Yang, C.; Sun, L.; Zong, B.; Li, K.; Liu, L.; Tu, G.; Liu, M.; Liu, S. Although c-MYC contributes to tamoxifen resistance, it improves cisplatin sensitivity in ER-positive breast cancer. Int. J. Oncol. 2020, 56, 932–944. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, P.; Hassiotou, F.; Blancafort, P.; Filgueira, L. Cisplatin induces differentiation of breast cancer cells. Front. Oncol. 2013, 3, 134. [Google Scholar] [CrossRef]
- National Cancer Institute. Neoadjuvant Platinum Chemotherapy in Treating Patients with Estrogen Receptor Positive and/or Progesterone Receptor Positive Breast Cancer. My Cancer Genome. 2014. Available online: https://www.mycancergenome.org/content/clinical_trials/NCT02221999/ (accessed on 10 July 2025).
- Dhar, S.; Kolishetti, N.; Lippard, S.J.; Farokhzad, O.C. Targeted delivery of a cisplatin prodrug for safer and more effective prostate cancer therapy in vivo. Proc. Natl. Acad. Sci. USA 2011, 108, 1850–1855. [Google Scholar] [CrossRef]
- Zhou, Z.; Shi, P.; Wang, C.; Sun, Y.; Gao, C. Recent updates in nanoscale delivery systems of platinum (IV) antitumor prodrugs. Coord. Chem. Rev. 2024, 508, 215774. [Google Scholar] [CrossRef]
- Cao, L.; Petrusca, D.N.; Satpathy, M.; Nakshatri, H.; Petrache, I.; Matei, D. Tissue transglutaminase protects epithelial ovarian cancer cells from cisplatin-induced apoptosis by promoting cell survival signaling. Carcinogenesis 2008, 29, 1893–1900. [Google Scholar] [CrossRef]
- Park, K.-S.; Kim, H.-K.; Lee, J.-H.; Choi, Y.-B.; Park, S.-Y.; Yang, S.-H.; Kim, S.-Y.; Hong, K.-M. Transglutaminase 2 as a cisplatin resistance marker in non-small cell lung cancer. J. Cancer Res. Clin. Oncol. 2010, 136, 493–502. [Google Scholar] [CrossRef]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Retinoic acids in the treatment of most lethal solid cancers. J. Clin. Med. 2020, 9, 360. [Google Scholar] [CrossRef]
- Sengupta, S.; Ray, S.; Chattopadhyay, N.; Biswas, N.; Chatterjee, A. Effect of retinoic acid on integrin receptors of B16F10 melanoma cells. J. Exp. Clin. Cancer Res. 2000, 19, 81–87. [Google Scholar]
- Cui, J.; Gong, M.; He, Y.; Li, Q.; He, T.; Bi, Y. All-trans retinoic acid inhibits proliferation, migration, invasion and induces differentiation of hepa1-6 cells through reversing EMT in vitro. Int. J. Oncol. 2016, 48, 349–357. [Google Scholar] [CrossRef]
- Zuo, L.; Yang, X.; Lu, M.; Hu, R.; Zhu, H.; Zhang, S.; Zhou, Q.; Chen, F.; Gui, S.; Wang, Y. All-trans retinoic acid inhibits human colorectal cancer cells RKO migration via downregulating myosin light chain kinase expression through MAPK signaling pathway. Nutr. Cancer 2016, 68, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- Budisan, L.; Gulei, D.; Zanoaga, O.M.; Irimie, A.I.; Chira, S.; Braicu, C.; Gherman, C.D.; Berindan-Neagoe, I. Dietary intervention by phytochemicals and their role in modulating coding and non-coding genes in cancer. Int. J. Mol. Sci. 2017, 18, 1178. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, A.; Ghorbani, A. Cancer therapy with phytochemicals: Evidence from clinical studies. Avicenna J. Phytomed. 2015, 5, 84. [Google Scholar]
- Chen, S.; Hu, Q.; Tao, X.; Xia, J.; Wu, T.; Cheng, B.; Wang, J. Retinoids in cancer chemoprevention and therapy: Meta-analysis of randomized controlled trials. Front. Genet. 2022, 13, 1065320. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.C.; Hsu, S.L.; Lin, H.; Yang, T.Y. Retinoic acid and cancer treatment. Biomedicine 2014, 4, 22. [Google Scholar] [CrossRef]
- Schug, T.T.; Berry, D.C.; Shaw, N.S.; Travis, S.N.; Noy, N. Opposing effects of retinoic acid on cell growth result from alternate activation of two different nuclear receptors. Cell 2007, 129, 723–733. [Google Scholar] [CrossRef]
- Bushue, N.; Wan, Y.J. Retinoid pathway and cancer therapeutics. Adv. Drug Deliv. Rev. 2010, 62, 1285–1298. [Google Scholar] [CrossRef]
- Tang, X.H.; Gudas, L.J. Retinoids, retinoic acid receptors, and cancer. Annu. Rev. Pathol. Mech. Dis. 2011, 6, 345–364. [Google Scholar] [CrossRef]
- Ross-Innes, C.S.; Stark, R.; Holmes, K.A.; Schmidt, D.; Spyrou, C.; Russell, R.; Massie, C.E.; Vowler, S.L.; Eldridge, M.; Carroll, J.S. Cooperative interaction between retinoic acid receptor-α and estrogen receptor in breast cancer. Genes Dev. 2010, 24, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.D. Transglutaminase 2 and NF-κB: An odd couple that shapes breast cancer phenotype. Breast Cancer Res. Treat. 2013, 137, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.Y.; Sun, P.M. Estrogen receptor-associated receptor α and peroxisome proliferator-activated receptor γ in metabolism and disease. Mol. Med. Rep. 2021, 23, 156. [Google Scholar] [CrossRef] [PubMed]
- Gallo, M.; Ferrari, E.; Terrazzan, A.; Brugnoli, F.; Spisni, A.; Taccioli, C.; Aguiari, G.; Trentini, A.; Volinia, S.; Keillor, J.W.; et al. Metabolic characterisation of transglutaminase 2 inhibitor effects in breast cancer cell lines. Fed. Eur. Biochem. Soc. 2023, 290, 5411–5433. [Google Scholar] [CrossRef]
- Schuppan, D.; Mäki, M.; Lundin, K.E.; Isola, J.; Friesing-Sosnik, T.; Taavela, J.; Popp, A.; Koskenpato, J.; Langhorst, J.; Hovde, Ø.; et al. A randomized trial of a transglutaminase 2 inhibitor for celiac disease. N. Engl. J. Med. 2021, 385, 35–45. [Google Scholar] [CrossRef]
- Jacamo, R.O.; Zhang, Q.; McQueen, T.; Davis, R.E.; Konopleva, M.; Andreeff, M. Silencing TG2 in Acute Myeloid Leukemia. Possible Role of TG2 in Microenvironment-Mediated Chemoresistance. Blood 2016, 128, 5087. [Google Scholar] [CrossRef]
- He, W.; Sun, Z.; Liu, Z. Silencing of TGM2 reverses epithelial to mesenchymal transition and modulates the chemosensitivity of breast cancer to docetaxel. Exp. Ther. Med. 2015, 10, 1413–1418. [Google Scholar] [CrossRef]
- Tran, V.; Kim, R.; Maertens, M.; Hartung, T.; Maertens, A. Similarities and differences in gene expression networks between the breast cancer cell line Michigan Cancer Foundation-7 and invasive human breast cancer tissues. Front. Artif. Intell. 2021, 4, 674370. [Google Scholar] [CrossRef]
- Morgenstern, J.; Fleming, T.; Kliemank, E.; Brune, M.; Nawroth, P.; Fischer, A. Quantification of all-trans retinoic acid by liquid chromatography–tandem mass spectrometry and association with lipid profile in patients with type 2 diabetes. Metabolites 2021, 11, 60. [Google Scholar] [CrossRef]
- Bezerra, D.S.; de Melo, A.T.; de Oliveira, K.C.; de Araújo, K.Q.; Medeiros, M.S.; Santos, F.A.; Medeiros, J.F.; Lima, M.S.; da Silva, A.G.; Ribeiro, K.D.; et al. Breast Milk Retinol Levels after Vitamin A Supplementation at Different Postpartum Amounts and Intervals. Nutrients 2022, 14, 3570. [Google Scholar] [CrossRef]
- Caccamo, D.; Curro, M.; Cannavo, G.; Ientile, R. Tissue transglutaminase was up-regulated by EGF-retinoid interplay in epithelial carcinoma cells. Life Sci. 2004, 75, 2643–2652. [Google Scholar] [CrossRef]
- Kwon, M.-H.; Jung, J.-W.; Jung, S.-H.; Park, J.-Y.; Kim, Y.-M.; Ha, K.-S. Quantitative and rapid analysis of transglutaminase activity using protein arrays in mammalian cells. Mol. Cells 2009, 27, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Park, M.K.; Bae, H.C.; Yoon, H.J.; Kim, S.Y.; Lee, C.H. Transglutaminase-2 is involved in all-trans retinoic acid-induced invasion and matrix metalloproteinases expression of SH-SY5Y neuroblastoma cells via NF-κB pathway. Biomol. Ther. 2012, 20, 286. [Google Scholar] [CrossRef] [PubMed]
- Conserva, M.R.; Anelli, L.; Zagaria, A.; Specchia, G.; Albano, F. The pleiotropic role of retinoic Acid/Retinoic acid receptors signaling: From vitamin A metabolism to gene rearrangements in acute promyelocytic leukemia. Int. J. Mol. Sci. 2019, 20, 2921. [Google Scholar] [CrossRef] [PubMed]
- Maire, N.L.; Teyssier, N.; Balaguer, N.; Bourguet, N.; Germain, N. Regulation of RXR-RAR heterodimers by RXR- and RAR-Specific ligands and their combinations. Cells 2019, 8, 1392. [Google Scholar] [CrossRef]
- Petkovich, M.; Chambon, P. Retinoic acid receptors at 35 years. J. Mol. Endocrinol. 2022, 69, T13–T24. [Google Scholar] [CrossRef]
- Siddik, Z.H. Cisplatin: Mode of cytotoxic action and molecular basis of resistance. Oncogene 2003, 47, 7265–7279. [Google Scholar] [CrossRef]
- Srinivasan, B.; Lloyd, M.D. Dose–Response Curves and the Determination of IC50 and EC50 Values. J. Med. Chem. 2024, 67, 17931–17934. [Google Scholar] [CrossRef]
- Liang, C.C.; Park, A.Y.; Guan, J.L. In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef]
- Jambrovics, K.; Uray, I.P.; Keresztessy, Z.; Keillor, J.W.; Fésüs, L.; Balajthy, Z. Transglutaminase 2 programs differentiating acute promyelocytic leukemia cells in all-trans retinoic acid treatment to inflammatory stage through NF-κB activation. Haematologica 2018, 104, 505. [Google Scholar] [CrossRef]
- Blows, F.M.; Ali, H.R.; Cope, W.; Pharoah, P.D.P.; Pike, C.V.S.; Provenzano, E.; Coussons, P. Expression of transglutaminase-2 (TGM2) in the prognosis of female invasive breast cancer. Br. J. Cancer Rep. 2024, 2, 5. [Google Scholar] [CrossRef]
- Meshram, D.D.; Fanutti, C.; Pike, C.V.S.; Coussons, P.J. Membrane Association of the Short Transglutaminase Type 2 Splice Variant (TG2-S) Modulates Cisplatin Resistance in a Human Hepatocellular Carcinoma (HepG2) Cell Line. Curr. Issues Mol. Biol. 2024, 46, 4251–4270. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lawani-Luwaji, E.U.; Pike, C.V.S.; Coussons, P.J. Retinoic Acid-Induced Transglutaminase 2 Expression Reduces Sensitivity to Cisplatin in the Hormone-Positive MCF-7 Breast Cancer Cell Model. Int. J. Mol. Sci. 2025, 26, 8101. https://doi.org/10.3390/ijms26168101
Lawani-Luwaji EU, Pike CVS, Coussons PJ. Retinoic Acid-Induced Transglutaminase 2 Expression Reduces Sensitivity to Cisplatin in the Hormone-Positive MCF-7 Breast Cancer Cell Model. International Journal of Molecular Sciences. 2025; 26(16):8101. https://doi.org/10.3390/ijms26168101
Chicago/Turabian StyleLawani-Luwaji, Ebidor U., Claire V. S. Pike, and Peter J. Coussons. 2025. "Retinoic Acid-Induced Transglutaminase 2 Expression Reduces Sensitivity to Cisplatin in the Hormone-Positive MCF-7 Breast Cancer Cell Model" International Journal of Molecular Sciences 26, no. 16: 8101. https://doi.org/10.3390/ijms26168101
APA StyleLawani-Luwaji, E. U., Pike, C. V. S., & Coussons, P. J. (2025). Retinoic Acid-Induced Transglutaminase 2 Expression Reduces Sensitivity to Cisplatin in the Hormone-Positive MCF-7 Breast Cancer Cell Model. International Journal of Molecular Sciences, 26(16), 8101. https://doi.org/10.3390/ijms26168101





