A Comprehensive Review of Fortification, Bioavailability, and Health Benefits of Folate
Abstract
1. Introduction
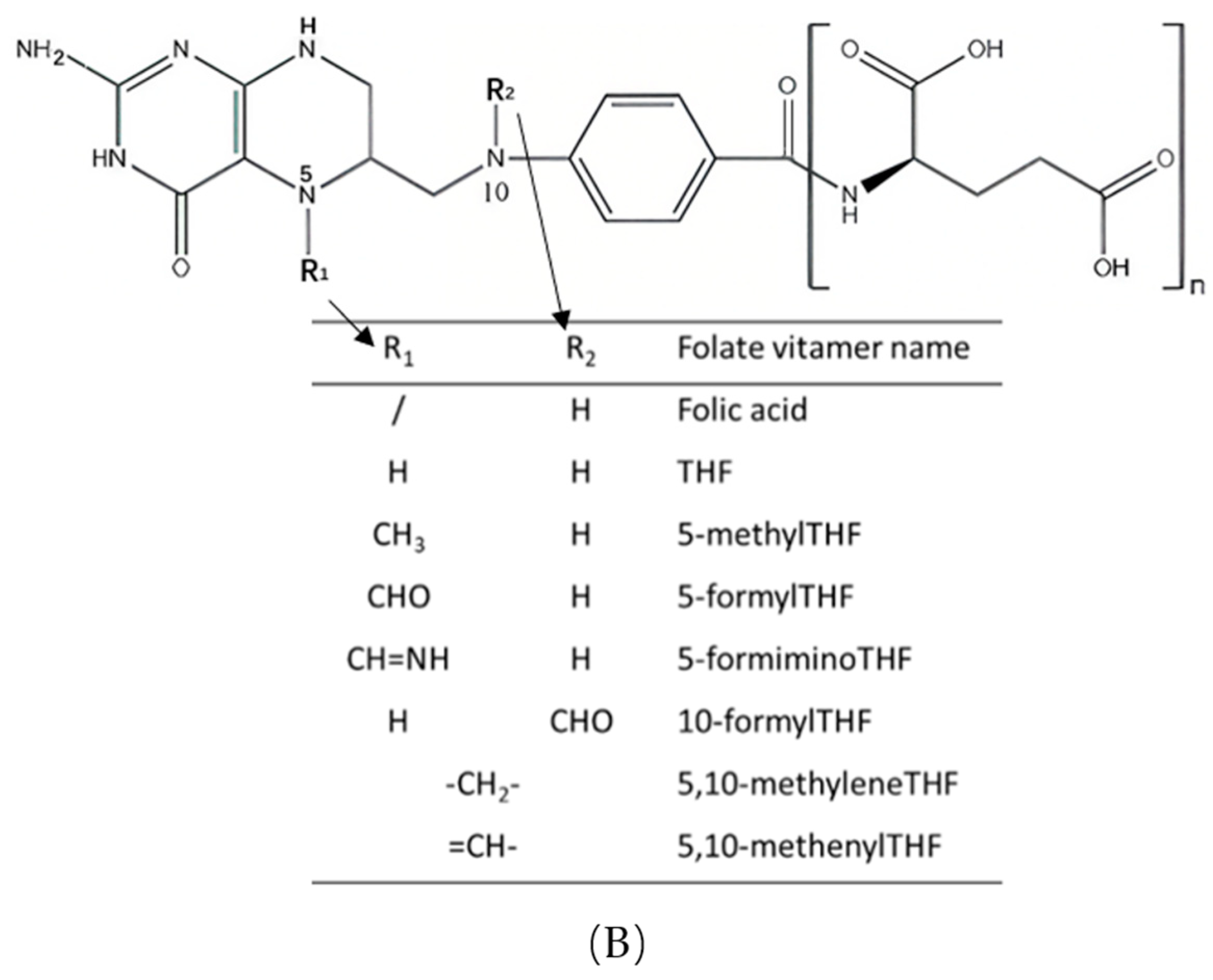
2. Structure and Dietary Sources of Folate
3. Folate Fortification
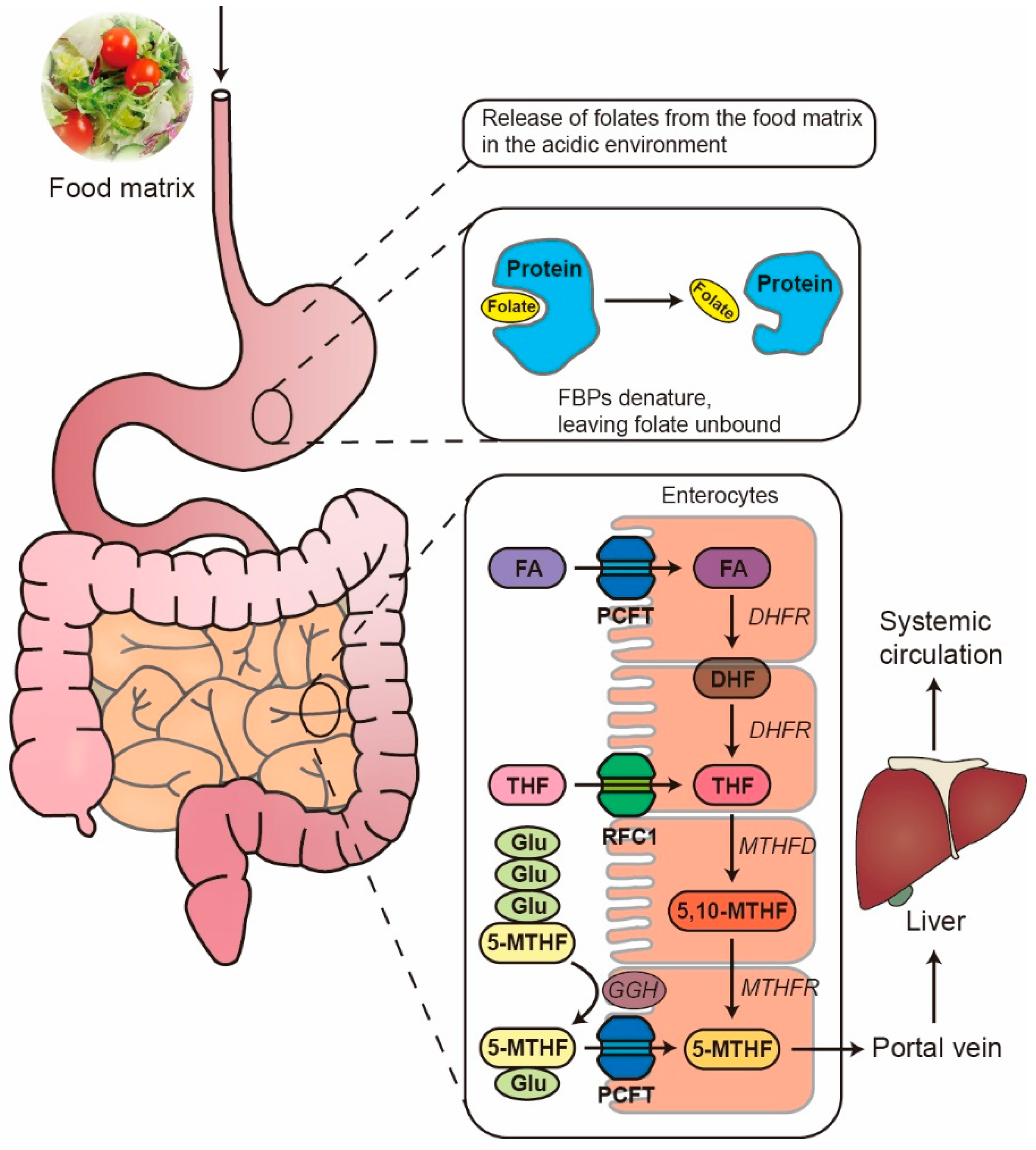
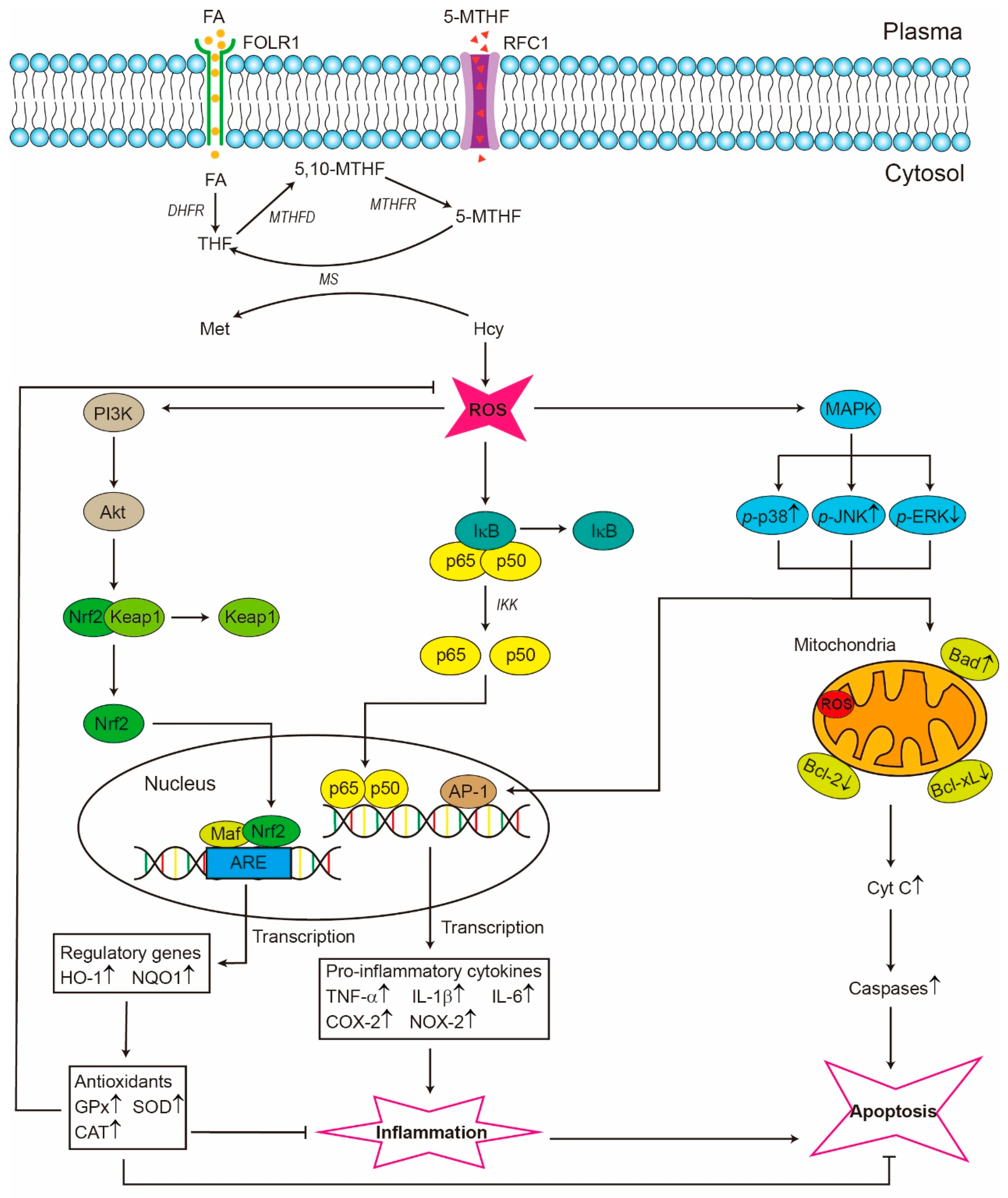
4. Bioaccessibility and Bioavailability of Folate
5. Health Benefits of Folate
5.1. Cardiovascular Protection
5.2. Hematopoiesis Protection
5.3. Gut Homeostasis Maintenance
5.4. Immune Response Enhancement
5.5. Neuroprotection
5.6. Anti-Cancer Activity
5.7. Neural Tube Protection
5.8. Danger of Excess Folate and Recommendations for FA Intake
| Model/object | Dosage and Duration | Outcomes | Reference |
|---|---|---|---|
| Cardiovascular protection | |||
| SHRs | 2 mg/kg diet for 4 weeks | Systolic BP ↓; plasma Hcy ↓; liver TG ↓; glucose tolerance ↑; insulin resistant ↑; oxidative stress (SOD ↑; GPx ↑; GSH ↑; GSSG ↑; TBARS ↓) | [76] |
| Ang II-infused mice | 0.015 g/L in drinking water | Plasma Hcy ↓; systolic BP ↓; oxidative stress (ROS production ↓); renal function (renal cortical blood flow ↑; vascular density ↑; Nox2/Nox4 ↓; eNOS ↑; ADMA ↑; VEGF ↓; Col IV ↓) | [80] |
| SHRs | 0.4 mg/kg/d for 8 weeks | Hcy ↓; oxidative stress (SOD ↑; MDA ↓); inflammation (IL-6 ↓; NF-κB p65/Rela ↓) | [77] |
| SHRs | 0.4 mg/kg/d for 8 weeks | Plasma Hcy ↓; oxidative stress (serum SOD ↑; serum MDA ↓); renal function (NOX2/NOX4 ↓; UACR ↓; GFR ↑; glomerular sclerosis index ↓) | [78] |
| Ang II-infused mice | 0.006% wt/wt for 3 weeks | Systolic BP ↓; cardiac hypertrophy (calcineurin ↓; NFAT ↓; heart wt/body wt ↓; contractility ↓; myocyte area ↓; ANF ↓; BNP ↓); inflammation (CD68+ area ↓; IL-6 ↓; IL-1b ↓; TNF-α ↓, NF-κB ↓; TGF-β ↓); fibrosis (a-SMA ↓; Col I ↓; Col III ↓) | [81] |
| LDLR−/− mice with high-fat diet | 75 μg/kg/d for 16 weeks | TG ↓; TC ↓; LDL ↓; VLDL ↓; HDL ↑; oxidative stress (serum SOD ↑; serum MDA ↓; serum GPx ↑); inflammation (IL-6 ↓; IL-1β ↓; TNF-a ↓); atherosclerotic lesion area ↓; VSMC dedifferentiation (a-SMA ↑; OPN ↓) | [82] |
| ApoE−/− mice | 0.006% wt/wt (combined with aerobic exercise) for 8 weeks | Plasma Hcy ↓; atherosclerosis (aortic root plaque area/burden ↓; plasma MCP-1 ↓) | [83] |
| BDL rats | 5 and 10 mg/kg for 4 weeks | Serum Hcy ↓; oxidative stress (GSH: GSSG ↑); TG ↓; TC ↓; HDL ↑; LDL ↓ | [84] |
| Pregnancy-induced hypertension in rats | 8 mg/kg diet (combined with 120 mg of DHA, 180 mg of EPA, and 50 mg of VB12) for 20 d | Dam: Systolic BP ↓; plasma Hcy ↓; oxidative stress (plasma MDA ↓); inflammation (placental TNF-α↓); placental AA ↓ Offspring: oxidative stress (liver MDA ↓; liver protein carbonyl ↓); liver DHA ↑ | [85] |
| SHRs | 0.4 mg/kg/d (combined with losartan at 25 mg/kg/d) for 12 weeks | More smooth and intact cellular membrane of endothelial cells | [86] |
| Mice with triple-transgenic late-stage Alzheimer’s disease | 12 mg/kg/d for 3 months | Cardiac apoptosis (apoptotic cells ↓; fas-ligand ↓; FADD ↓; BAK ↓; cytochrome-c ↓; cleaved-caspase-3 ↓); p-IGF1-receptor ↑; p-PI3K ↑; p-AMPKα ↑; sirtuin 1 ↑ | [9] |
| Hematopoiesis protection | |||
| Mice | 2 mg/kg for 6 months | Higher hematopoietic reconstitution potential and numbers of circulating lymphocytes | [91] |
| Zebrafish model of congenital sideroblastic anemia | 100 mM glycine with 1 mM sodium for 44 h | Higher hemoglobin levels | [88] |
| Human colon epithelial cells | Presence or absence of FA (4 mg/L) for up to 14 d | Folate deficiency increases uracil misincorporation two- to threefold in these cells Folate-deficient human colonocytes exposed to hydrogen peroxide or methyl methane sulfonate, an alkylating agent, are unable to repair DNA strand breakage as efficiently as folate-sufficient cells | [87] |
| 240 participants with MCI | FA alone, vitamin B12 alone, FA plus vitamin B12, or control without treatment daily for 6 months | Post hoc Turkey tests found that FA and vitamin B12 supplementation was significantly more effective than FA alone for all endpoints | [8] |
| C2C12 myoblast cells | Vitamin C at 100 µM | Compared to untreated cells, treatment of C2C12 cells with AA at 100 µM resulted in enhanced concentrations of FA (2.5-fold) and 5-MTHF (10-fold increase) | [93] |
| Patients undergoing hemodialysis | ESA consisted of darbepoetin alfa (injection at doses of 10, 20, 30, and 40 μg) HIF-PHI included roxadustat (tablets at doses of 20, 50, and 100 mg) | Zinc supplementation improves ERI during darbepoetin alfa administration. The use of roxadustat stabilizes HIF-1α, HIF-2α, and HIF-3α | [94] |
| Gut homeostasis maintenance | |||
| Piglets | 3–18 mg/kg for 14 d | Caecum (pH ↓; acetic acid ↑; Firmicutes ↑; Lactobacillus reuteri ↑; Lactobacillus salivarius ↑; Lactobacillus mucosae ↑); colon (IBA ↑; BA ↑; IVA ↑; acetic acid/propionic acid ↑; total SCFAs ↑) | [12] |
| Mice with DSS-induced colitis | 0.071 mg/kg for 7 d | Plasma Hcy ↓; inflammation (p-p38 ↓; p-cPLA-2 ↓; COX-2 ↓; PGE2 ↓; IL-17 ↓; RORgt ↓) | [109] |
| Male veterans for colonoscopy procedures | ≥227 μg vs. <227 μg DFE/d | Firmicutes (Dialister ↑; Roseburia ↑; Faecalibacterium ↑); Verrucomicrobia (Akkermansia ↑); Bacteroidota (Bacteroides ↓; Alistipes ↑; Odoribacter ↑; Parabacteroides ↑) | [110] |
| Mice fed with high-fat diet | 5 mg/kg for 25 weeks | Body weight ↓; Ace index ↑; Firmicutes ↓ (Streptococcus ↑; Colidextribacter ↓; Allobaculum ↑; Lactococcus ↑; Oscillibacter ↓; Roseburia ↓; Tuzzerella ↓; Ileibacterium ↑); Bacteroidota ↑ (Rikenella ↑); Desulfobacterota (Desulfovibrio ↓; Bilophila ↓) | [111] |
| Rats fed with high-purine diet | 4 mg/kg for 8 weeks | Uric acid ↓; Firmicutes ↓ (Lactobacillus ↑; Clostridium ↓; Romboutsia ↓; Blautia ↑; Ruminococcus ↓); Actinobacteria ↑ (Collinsella ↑); Desulfobacterota ↓; Bacteroidota ↑ (Bacteroides ↑) | [112] |
| Broiler chicken | 13 mg/kg for 4 weeks | Abdominal fat percentage ↓; Firmicutes ↓ (Clostridium ↑; Oscillospira ↑; Ruminococcus ↑; Dehalobacterium ↑); Bacteroidota ↑ (Alistipes ↑; Parabacteroides ↑); cecal (acetic acid ↑; propionic acid ↑; IBA ↑); adipocyte proliferation and differentiation genes (IGF1 ↓; EGF ↓; TGF-β ↓; C/EBPα ↓; FABP-4 ↓; PCNA ↓) | [99] |
| Human fecal slurry culture | 0.5 mL of fecal suspension into 100 μL of 1 mg/mL FA or 5-MTHF for 24 h fermentation | FA: Ace index ↑; Shannon index ↓; acetic acid ↓; Firmicutes ↓ (Lactobacillus ↑; Pediococcus ↑); Proteobacteria ↑; Actinobacteriota ↑ (Bifidobacterium ↑); Bacteroidota ↓ (Bacteroides ↓) 5-MTHF: acetic acid ↓; Shannon index ↓; Firmicutes ↑ (Lactobacillus ↑; Pediococcus ↑); Actinobacteriota ↑ (Bifidobacterium ↑); Bacteroidota ↓ (Bacteroides ↓) | [102] |
| Immune response enhancement | |||
| Broiler chicken | 1.5 mg/kg diet for 35 d | Thyroid hormones (IGF-1 ↑; triiodothyronine ↑; thyroxin ↑); oxidative stress (HSP70 ↑; TAC ↑; catalase ↑; SOD ↑); immune response (H/L ↓; antibody titration against NDV ↑) | [115] |
| Fish (Epinephelus malabaricus) | 0–10 mg/kg diet for 8 weeks | Oxidative stress (TBARS ↓; SOD ↑; superoxide anion production ↑); immune response (lysozyme ↑) | [116] |
| BALB/c mice | 4 μg/mL in drinking water | Colon immune response (Foxp3+ CD4+ ↑; IFN-g+ CD4+ ↑; IL-10 ↑; FR4+ CD4+ ↑; Bcl-2/GAPDH ↑; Bcl-xL/GAPDH ↑) | [118] |
| Female BALB/c mice | Control diet for 8 weeks | Immune response (small intestinal Foxp3+ CD4+ ↑) | [119] |
| Broiler chickens with high-energy diet | 2.2–15 ppm for 6 weeks | Ceca weight ↓; bursa weight ↓ | [114] |
| Castrated weanling piglets | 0–15 mg/kg diet for 24 d | Immune response (serum IFN-g ↑; CD3+ CD4+/CD3+ CD8+ ↓) | [13] |
| Fertilized broilers eggs | 0–150 μg at embryonic age 11 d | Broiler chicks: one-carbon metabolism (MTHFR ↑; MTRR ↑); immune response (plasma lysozyme activity ↑; plasma IgG ↑; plasma IgM ↑; splenic IL-2 ↑; splenic IL-4 ↑; splenic IL-6 ↓) | [117] |
| Neuroprotection | |||
| Patients with mild cognitive impairment | 400 μg/d for 24 months | Hcy ↓; neurological test scores (full scale IQ ↑; verbal IQ ↑; information ↑; digit span ↑); Ab-related biomarkers (Ab-42 ↓; APP-mRNA ↓) | [8] |
| Rat cortical neuron cultures exposed to Ab31-35 | 40 mg/mL (combined with 27 mg/mL of genistein) | Neuron viability ↑; MMP ↑; DNA damage (comet cells ↓; DNA migration length ↓); apoptosis (Bax ↓; Bcl-2 ↑; caspase-3 ↓; tumor p53 ↓) | [126] |
| Wistar rats with Hcy-induced cerebellar damage | 0.011 mmol/g for 3 weeks | Plasma Hcy ↓; cortical Hcy ↓; motor coordination impairment ↓; oxidative stress (cerebellar MDA ↓; cerebellar GPx ↑) | [128] |
| ICR mice | 2 mg/kg for 25 weeks | Hcy ↓; blood glucose ↓; serum insulin ↑; serum TG ↓; serum VLDL-cholesterol ↓; cognitive behaviors (open field test (peripheral time ↓; grooming ↓); elevated plus maze (open arm distance ↑; open/total arm entries ↑; open/total arm distance ↑); Morris water maze (latency ↓)) | [123] |
| Patients with newly diagnosed Alzheimer’s disease | 1.25 mg/d for 6 months | MMSE ↑; inflammation (TNF-α ↓; TNF-α-mRNA ↓; SAM ↑); Ab-related biomarkers (Ab-40 ↓; PS1-mRNA ↓;) | [130] |
| Mouse neuroblastoma N2a cells expressing human APP695 | 2.8–20 mmol/L | Increasing DNA methylation by down-regulating the mRNA expression of genes in JAK-STAT and LTD pathways | [127] |
| APP/PS1 mice | 2.1 mg/kg diet + 600 mg/kg for 60 d | Increasing DNA methylation by down-regulating the mRNA expression of genes in JAK-STAT and LTD pathways | [127] |
| Human SH-SY5Y cells with Al-maltolate | 10 mmol/L for 3 d | Cell viability ↑; miRNA-19 ↑; apoptosis (PTEN ↓; p-AKT ↑; p53 ↓; Bax ↓; Bcl-2 ↑; cleaved-caspase 9 ↓; cleaved-caspase 3 ↓) | [124] |
| Primary rat astrocyte culture | 0–40 mmol/L for 12 d | Cell proliferation ↑; apoptosis ↓; Hcy ↓; ROS production ↓; telomeric DNA oxidative damage ↓; telomere length ↑ | [125] |
| Wistar rats with cyclophosphamide-induced NTD | 0.7 mg/kg (combined with 160 mg/kg of soy isoflavone) for 14/20 d | DNA damage (comet cells ↓; DNA migration length ↓); oxidative stress (SOD ↑; MDA ↓; NO ↓); apoptosis (p53 ↓; Bax ↓; Bcl-2 ↑) | [129] |
| Anti-Cancer activity | |||
| 200 OSCC cases, 200 OPL cases, and 200 control cases | <24.43, 24.43–29.14, 29.14–36.24, and >36.24 mg/L | OPL: risk ↓; p16 methylation ↓; p53 methylation ↓ OSCC: risk ↓; p16 methylation ↓; p53 methylation ↓ | [10] |
| MCF-7 cells | 4 or 8 mg/L | MCF-7 cell viability ↓; caspase-dependent apoptosis ↑; tumor suppressor genes (PTEN ↑; RARB2 ↑; APC ↑); DNMT1 ↑ | [11] |
| HT-29 and SW480 cells | 0, 100, 10,000 ng/mL | Micronucleus score ↓; comet tail DNA ↓ | [139] |
| Elderly Chileans | <45.3 nmol/L and ≥45.3 nmol/L | Gene methylation (p16 ↑; MLH1 ↑; MGMT ↑) | [136] |
| Neural tube protection | |||
| Mouse embryo stem cells | 50 mg/L | Neural tube closure-related genes (Cdx2 ↑; Nestin ↑; Pax6 ↑; Gata4 ↑) | [146] |
| Women at their first antenatal visit | 400 mg/d at 4–12 weeks before last menstrual period | Achieved optimal RBC folate levels (≥906 nmol/L) | [145] |
| ICR mice with diabetes-induced NTD | 3 mg/kg/d with n-3 PUFA diet until sacrifice | Hcy ↓; NTD incidence ↓; apoptosis (apoptotic cells ↓; p53 ↓; Bax ↓); Pax3 methylation ↓; one-carbon metabolism (MTHFR ↑; MTR ↓; MAT ↓; Dnmt3b ↓; SAHH ↓; CBS ↑) | [151] |
| ICR mice with diabetic pregnancy | 10 mg/kg for 7/8/10 d | NTD incidence ↓; Pax3 ↓; oxidative stress (ROS production ↓; MDA ↓; H2O2 ↓) | [148] |
| Wistar rats with cyclophosphamide-induced NTD | 0.7 mg/kg (combined with 160 mg/kg of soy isoflavone) for 20 d | NTD incidence ↓ | [129] |
| ICR mice with LPS-induced NTD | 3 mg/kg/d from GD 8 to GD 12 | NTD incidence ↓; inflammation (p-JNK ↑; p-IkB ↓; NF-κB p65 ↓; TNF-α ↓; IL-1β ↓; IL-6 ↓); oxidative stress (GSH ↓) | [147] |
| CD-1 mice with valproic acid-induced NTD | 3 × 4 mg/kg/d from GD 5 to GD 10 | Exencephaly ↓; apoptosis (p53 ↓; NF-κB ↑; Pim-1 ↑; c-Myb ↑; Bax/Bcl2 ↓) | [149] |
| Life Stage | Age | Males (µg/day) | Females (µg/day) |
|---|---|---|---|
| Infants | 0–6 months | 65 | 65 |
| Infants | 7–12 months | 80 | 80 |
| Children | 1–3 years | 150 | 150 |
| Children | 4–8 years | 200 | 200 |
| Children | 9–13 years | 300 | 300 |
| Adolescents | 14–18 years | 400 | 400 |
| Adults | >18 years | 400 | 400 |
| Pregnancy | - | - | 600 |
| Lactation | - | - | 500 |
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Abbasi, I.H.R.; Abbasi, F.; Wang, L.; Abd El Hack, M.E.; Swelum, A.A.; Hao, R.; Yao, J.; Cao, Y. Folate promotes S-adenosyl methionine reactions and the microbial methylation cycle and boosts ruminants production and reproduction. AMB. Express. 2018, 8, 65. [Google Scholar] [CrossRef]
- Sisay, B.G.; Tamirat, H.; Sandalinas, F.; Joy, E.J.M.; Zerfu, D.; Belay, A.; Mlambo, L.; Lark, M.; Ander, E.L.; Gashu, D. Folate deficiency is spatially dependent and associated with local farming systems among women in Ethiopia. Curr. Dev. Nutr. 2022, 6, nzac088. [Google Scholar] [CrossRef]
- Linda, A.A.; Kyallo, F.; Okoth, J.K.; Kahenya, P.; Makokha, A.; Sila, D.; Mwai, J. Food fortification: The level of awareness among Kenyan consumers. J. Nutr. Metab. 2020, 2020, 8486129. [Google Scholar] [CrossRef]
- FDA. Folate and Folic Acid on the Nutrition and Supplement Facts Labels. Available online: https://www.fda.gov/food/nutrition-facts-label/folate-and-folic-acid-nutrition-and-supplement-facts-labels#:~:text=For%20folate%2C%20the%20DV%20is,consume%20500%20mcg%20DFE%20daily (accessed on 4 August 2025).
- Huang, R.F.; Hsu, Y.C.; Lin, H.L.; Yang, F.L. Folate depletion and elevated plasma homocysteine promote oxidative stress in rat livers. J. Nutr. 2001, 131, 33–38. [Google Scholar] [CrossRef]
- Huang, R.F.; Yaong, H.C.; Chen, S.C.; Lu, Y.F. In vitro folate supplementation alleviates oxidative stress, mitochondria-associated death signalling and apoptosis induced by 7-ketocholesterol. Br. J. Nutr. 2004, 92, 887–894. [Google Scholar] [CrossRef]
- Solini, A.; Santini, E.; Ferrannini, E. Effect of short-term folic acid supplementation on insulin sensitivity and inflammatory markers in overweight subjects. Int. J. Obes. 2006, 30, 1197–1202. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Li, Q.; Zhou, X.; Zhao, J.; Song, A.; Li, W.; Liu, H.; Xu, W.; Huang, G. Effects of folic acid supplementation on cognitive function and Aβ-related biomarkers in mild cognitive impairment: A randomized controlled trial. Eur. J. Nutr. 2019, 58, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Ju, D.T.; Huang, R.F.S.; Tsai, B.C.K.; Su, Y.C.; Chiu, P.L.; Chang, Y.M.; Padma, V.V.; Ho, T.J.; Yao, C.H.; Kuo, W.W.; et al. Folic acid and folinic acid protect hearts of aging triple-transgenic Alzheimer’s disease mice via IGF1R/PI3K/AKT and SIRT1/AMPK pathways. Neurotoxic Res. 2023, 41, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Pan, D.; Su, M.; Huang, G.; Sun, G. Moderately high folate level may offset the effects of aberrant DNA methylation of P16 and P53 genes in esophageal squamous cell carcinoma and precancerous lesions. Genes Nutr. 2020, 15, 18. [Google Scholar] [CrossRef]
- Lubecka-Pietruszewska, K.; Kaufman-Szymczyk, A.; Stefanska, B.; Fabianowska-Majewska, K. Folic acid enforces DNA methylation-mediated transcriptional silencing of PTEN, APC and RARbeta2 tumour suppressor genes in breast cancer. Biochem. Biophys. Res. Commun. 2013, 430, 623–628. [Google Scholar] [CrossRef]
- Wang, L.; Zou, L.; Li, J.; Yang, H.; Yin, Y. Effect of dietary folate level on organ weight, digesta pH, short-chain fatty acid concentration, and intestinal microbiota of weaned piglets. J. Anim. Sci. 2021, 99, skab015. [Google Scholar] [CrossRef]
- Gao, Q.; Chen, D.; Ding, X.; Xu, Z.; Wu, A.; Zhang, K. Effects of dietary folic acid supplementation on growth performance and immune parameters in weanling piglets. Agriculture 2023, 13, 2271. [Google Scholar] [CrossRef]
- Gazzali, A.M.; Lobry, M.; Colombeau, L.; Acherar, S.; Azaïs, H.; Mordon, S.; Arnoux, P.; Baros, F.; Vanderesse, R.; Frochot, C. Stability of folic acid under several parameters. Eur. J. Pharm. Sci. 2016, 93, 419–430. [Google Scholar] [CrossRef]
- Liu, F.; Edelmann, M.; Piironen, V.; Li, Y.; Liu, X.; Yan, J.; Li, L.; Kariluoto, S. How food matrices modulate folate bioaccessibility: A comprehensive overview of recent advances and challenges. Compr. Rev. Food Sci. Food. Saf. 2024, 23, e13328. [Google Scholar] [CrossRef]
- Hirsch, S.; de la Maza, P.; Barrera, G.; Gattás, V.; Petermann, M.; Bunout, D. The Chilean flour folic acid fortification program reduces serum homocysteine levels and masks vitamin B-12 deficiency in elderly people. J. Nutr. 2002, 132, 289–291. [Google Scholar] [CrossRef]
- Lu, C.; Liu, Y.; Li, J.; Liu, L.; Du, G. Engineering of Biosynthesis Pathway and NADPH Supply for Improved L-5-Methyltetrahydrofolate Production by Lactococcus lactis. J. Microbiol. Biotechnol. 2019, 31, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Edelmann, M.; Piironen, V.; Kariluoto, S. 5-methyltetrahydrofolate is a crucial factor in determining the bioaccessibility of folate in bread. J. Agric. Food Chem. 2022, 70, 13379–13390. [Google Scholar] [CrossRef]
- Zavala-Castillo, K.A.; Flores-Ramirez, N.; Vasquez-Garcia, S.R.; Martinez-Flores, H.E.; Fernandez-Quiroz, D. Folic acid in carboxymethylcellulose/polyethylene oxide electrospun nanofibers: Preparation, release and stability. J. Sci. Food Agric. 2024, 104, 9014–9022. [Google Scholar] [CrossRef] [PubMed]
- Entine, J.; Felipe, M.S.S.; Groenewald, J.H.; Kershen, D.L.; Lema, M.; McHughen, A.; Nepomuceno, A.L.; Ohsawa, R.; Ordonio, R.L.; Parrott, W.A.; et al. Regulatory approaches for genome edited agricultural plants in select countries and jurisdictions around the world. Transgenic Res. 2021, 30, 551–584. [Google Scholar] [CrossRef] [PubMed]
- Fulgoni, V.L., 3rd; Agarwal, S. Nutritional impact of adding a serving of mushrooms on usual intakes and nutrient adequacy using National Health and Nutrition Examination Survey 2011–2016 data. Food Sci. Nutr. 2021, 9, 1504–1511. [Google Scholar] [CrossRef]
- Edelmann, M.; Aalto, S.; Chamlagain, B.; Kariluoto, S.; Piironen, V. Riboflavin, niacin, folate and vitamin B12 in commercial microalgae powders. J. Food Compos. Anal. 2019, 82, 103226. [Google Scholar] [CrossRef]
- Mattila, P.; Könkö, K.; Eurola, M.; Pihlava, J.M.; Astola, J.; Vahteristo, L.; Hietaniemi, V.; Kumpulainen, J.; Valtonen, M.; Piironen, V. Contents of vitamins, mineral elements, and some phenolic compounds in cultivated mushrooms. J. Agric. Food Chem. 2001, 49, 2343–2348. [Google Scholar] [CrossRef] [PubMed]
- Phillips, K.M.; Ruggio, D.M.; Haytowitz, D.B. Folate composition of 10 types of mushrooms determined by liquid chromatography-mass spectrometry. Food Chem. 2011, 129, 630–636. [Google Scholar] [CrossRef]
- Brown, M.R.; Mular, M.; Miller, I.; Farmer, C.; Trenerry, C. The vitamin content of microalgae used in aquaculture. J. Appl. Phycol. 1999, 11, 247–255. [Google Scholar] [CrossRef]
- McKillop, D.J.; Pentieva, K.; Daly, D.; McPartlin, J.M.; Hughes, J.; Strain, J.J.; Scott, J.M.; McNulty, H. The effect of different cooking methods on folate retention in various foods that are amongst the major contributors to folate intake in the UK diet. Br. J. Nutr. 2002, 88, 681–688. [Google Scholar] [CrossRef]
- Del Mondo, A.; Smerilli, A.; Sané, E.; Sansone, C.; Brunet, C. Challenging microalgal vitamins for human health. Microb. Cell Fact. 2020, 19, 201. [Google Scholar] [CrossRef]
- Honein, M.A.; Paulozzi, L.J.; Mathews, T.J.; Erickson, J.D.; Wong, L.Y.C. Impact of folic acid fortification of the US food supply on the occurrence of neural tube defects. JAMA 2001, 285, 2981–2986. [Google Scholar] [CrossRef]
- Sayed, A.R.; Bourne, D.; Pattinson, R.; Nixon, J.; Henderson, B. Decline in the prevalence of neural tube defects following folic acid fortification and its cost-benefit in South Africa. Birth Defects Res. Clin. Mol. Hepatol. Teratol. 2008, 82, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Ray, J.G.; Meier, C.; Vermeulen, M.J.; Boss, S.; Wyatt, P.R.; Cole, D.E.C. Association of neural tube defects and folic acid food fortification in Canada. Lancet 2002, 360, 2047–2048. [Google Scholar] [CrossRef]
- He, Y.; Li, C.; Yang, M.; Wang, C.; Guo, H.; Liu, J.; Liu, H. Transcriptome Analysis Reveals the Mechanisms of Accumulation and Conversion of Folate Derivatives during Germination of Quinoa (Chenopodium quinoa Willd). Seeds. J. Agric. Food Chem. 2024, 72, 3800–3813. [Google Scholar] [CrossRef] [PubMed]
- Sallam, S.M.; Shawky, E.; El Sohafy, S.M. Determination of the effect of germination on the folate content of the seeds of some legumes using HPTLC-mass spectrometry-multivariate image analysis. Food Chem. 2021, 362, 130206. [Google Scholar] [CrossRef]
- Hefni, M.; Witthöft, C.M. Effect of germination and subsequent oven-drying on folate-content in different wheat and rye cultivars. J. Cereal Sci. 2012, 56, 374–378. [Google Scholar] [CrossRef]
- Mahara, F.A.; Nuraida, L.; Lioe, H.N.; Nurjanah, S. Hypothetical regulation of folate biosynthesis and strategies for folate overproduction in lactic acid bacteria. Prev. Nutr. Food Sci. 2023, 28, 386–400. [Google Scholar] [CrossRef]
- Rizo, J.; Guillén, D.; Díaz-Ruiz, G.; Wacher, C.; Encarnación, S.; Sánchez, S.; Rodríguez-Sanoja, R. Metaproteomic Insights Into the Microbial Community in Pozol. Front. Nutr. 2021, 8, 714814. [Google Scholar] [CrossRef] [PubMed]
- Mosso, A.L.; LeBlanc, J.G.; Motta, C.; Castanheira, I.; Ribotta, P.; Samman, N. Effect of fermentation in nutritional, textural and sensorial parameters of vegan-spread products using a probiotic folate-producing Lactobacillus sakei strain. LWT 2020, 127, 109339. [Google Scholar] [CrossRef]
- Tamene, A.; Baye, K.; Humblot, C. Folate content of a staple food increased by fermentation of a cereal using selected folate-producing microorganisms. Heliyon 2022, 8, e09526. [Google Scholar] [CrossRef]
- Hjortmo, S.; Patring, J.; Jastrebova, J.; Andlid, T. Biofortification of folates in white wheat bread by selection of yeast strain and process. Int. J. Food Microbiol. 2008, 127, 32–36. [Google Scholar] [CrossRef]
- Meng, Z.; Yi, L.; Hu, Q.; Lin, Z.; Ramaswamy, H.S.; Wang, C. Optimized extraction and characterization of folates from date palm fruits and their tracking during fruits wine fermentation. Front. Nutr. 2021, 8, 699555. [Google Scholar] [CrossRef]
- Zhou, S.; Zhu, S.; Cui, S.; Hou, H.; Wu, H.; Hao, B.; Cai, L.; Xu, Z.; Liu, L.; Jiang, L.; et al. Transcriptional and post-transcriptional regulation of heading date in rice. New Phytol. 2021, 230, 943–956. [Google Scholar] [CrossRef]
- Shariatipour, N.; Heidari, B.; Tahmasebi, A.; Richards, C. Comparative genomic analysis of quantitative trait loci associated with micronutrient contents, grain quality, and agronomic traits in wheat (Triticum aestivum L.). Front. Plant Sci. 2021, 12, 709817. [Google Scholar] [CrossRef]
- Dong, W.; Cheng, Z.; Xu, J.L.; Zheng, T.; Wang, X.; Zhang, H.; Wang, J.; Wan, J. Identification of QTLs underlying folate content in milled rice. J. Integr. Agric. 2014, 13, 1827–1834. [Google Scholar] [CrossRef]
- Agyenim-Boateng, K.G.; Zhang, S.; Gu, R.; Zhang, S.; Qi, J.; Azam, M.; Ma, C.; Li, Y.; Feng, Y.; Liu, Y.; et al. Identification of quantitative trait loci and candidate genes for seed folate content in soybean. Theor. Appl. Genet. 2023, 136, 149. [Google Scholar] [CrossRef]
- Guo, W.; Lian, T.; Wang, B.; Guan, J.; Yuan, D.; Wang, H.; Azam, F.M.S.; Wan, X.; Wang, W.; Liang, Q.; et al. Genetic mapping of folate QTLs using a segregated population in maize. J. Integr. Plant Biol. 2019, 61, 675–690. [Google Scholar] [CrossRef]
- Chang, J.; Xie, C.; Wang, P.; Gu, Z.; Han, Y.; Yang, R. Red light enhances folate accumulation in wheat seedlings. J. Zhejiang Univ.Sci. B 2021, 22, 906–916. [Google Scholar] [CrossRef]
- Dong, W.; Cheng, Z.; Lei, C.; Wang, X.; Wang, J.; Wang, J.; Wu, F.; Zhang, X.; Guo, X.; Zhai, H.; et al. Overexpression of folate biosynthesis genes in rice (Oryza sativa L.) and evaluation of their impact on seed folate content. Plant Foods Hum. Nutr. 2014, 69, 379–385. [Google Scholar] [CrossRef]
- Blancquaert, D.; Storozhenko, S.; Van Daele, J.; Stove, C.; Visser, R.G.F.; Lambert, W.; Van Der Straeten, D. Enhancing pterin and para-aminobenzoate content is not sufficient to successfully biofortify potato tubers and Arabidopsis thaliana plants with folate. J. Exp. Bot. 2013, 64, 3899–3909. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H.; Min, Y.; Sanchez-Ortiz, E.; Huang, J.; Mireault, A.A.; Shelton, J.M.; Kim, J.; Mammen, P.P.A.; Bassel-Duby, R.; et al. Enhanced CRISPR-Cas9 correction of Duchenne muscular dystrophy in mice by a self-complementary AAV delivery system. Sci. Adv. 2020, 6, eaay6812. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, J.; Wei, P.; Zhang, B.; Gou, F.; Feng, Z.; Mao, Y.; Yang, L.; Zhang, H.; Xu, N.; et al. The CRISPR/Cas9 system produces specific and homozygous targeted gene editing in rice in one generation. Plant Biotechnol. J. 2014, 12, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Veillet, F.; Perrot, L.; Chauvin, L.; Kermarrec, M.P.; Guyon-Debast, A.; Chauvin, J.E.; Nogué, F.; Mazier, M. Transgene-free genome editing in tomato and potato plants using Agrobacterium-mediated delivery of a CRISPR/Cas9 cytidine base editor. Int. J. Mol. Sci. 2019, 20, 402. [Google Scholar] [CrossRef]
- Agulló, V.; Moreno, D.A.; Domínguez-Perles, R.; García-Viguera, C. Contribution of the diverse experimental models to unravelling the biological scope of dietary (poly)phenols. J. Sci. Food Agric. 2022, 102, 3940–3951. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Nile, S.H.; Keum, Y.S. Folates: Chemistry, analysis, occurrence, biofortification and bioavailability. Food Res. Int. 2016, 89, 1–13. [Google Scholar] [CrossRef]
- Brogna, B.; Amitrano, M.; Mangiacapra, S.; Gravino, E.; Capasso, P.M.F.; Gagliardi, G.; Musto, L.A. A floating thrombus detected by CT in the descending aorta in an obese patient: A possible genetic-environmental interaction. Radiol. Case. Rep. 2021, 16, 348–352. [Google Scholar] [CrossRef]
- De Re, V.; Cannizzaro, R.; Canzonieri, V.; Cecchin, E.; Caggiari, L.; De Mattia, E.; Pratesi, C.; De Paoli, P.; Toffoli, G. MTHFR polymorphisms in gastric cancer and in first-degree relatives of patients with gastric cancer. Tumour. Biol. 2010, 31, 23–32. [Google Scholar] [CrossRef]
- Tsang, B.L.; Devine, O.J.; Cordero, A.M.; Marchetta, C.M.; Mulinare, J.; Mersereau, P.; Guo, J.; Qi, Y.P.; Berry, R.J.; Rosenthal, J.; et al. Assessing the association between the methylenetetrahydrofolate reductase (MTHFR) 677C>T polymorphism and blood folate concentrations: A systematic review and meta-analysis of trials and observational studies. Am. J. Clin. Nutr. 2015, 101, 1286–1294. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, R.D.; Karmacharya, B.M.; Shrestha, A.; Timalsena, D.; Madhup, S.; Shahi, R.; Katuwal, N.; Shrestha, R.; Fitzpatrick, A.L.; Risal, P. Prevalence of MTHFR C677T polymorphism and its association with serum homocysteine and blood pressure among different ethnic groups: Insights from a cohort study of Nepal. BMC. Cardiovasc. Disord. 2025, 25, 235. [Google Scholar] [CrossRef]
- Huh, H.J.; Chi, H.S.; Shim, E.H.; Jang, S.; Park, C.J. Gene--nutrition interactions in coronary artery disease: Correlation between the MTHFR C677T polymorphism and folate and homocysteine status in a Korean population. Thromb. Res. 2006, 117, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Edelmann, M.; Piironen, V.; Kariluoto, S. The bioaccessibility of folate in breads and the stability of folate vitamers during in vitro digestion. Food Funct. 2022, 13, 3220–3233. [Google Scholar] [CrossRef]
- Ringling, C.; Rychlik, M. Simulation of food folate digestion and bioavailability of an oxidation product of 5-methyltetrahydrofolate. Nutrients 2017, 9, 969. [Google Scholar] [CrossRef] [PubMed]
- Arkbage, K.; Verwei, M.; Havenaar, R.; Witthoft, C. Bioaccessibility of folic acid and (6S)-5-methyltetrahydrofolate decreases after the addition of folate-binding protein to yogurt as studied in a dynamic in vitro gastrointestinal model. J. Nutr. 2003, 133, 3678–3683. [Google Scholar] [CrossRef]
- de Jong, R.J.; Verwei, M.; West, C.E.; van Vliet, T.; Siebelink, E.; van den Berg, H.; Castenmiller, J.J. Bioavailability of folic acid from fortified pasteurised and UHT-treated milk in humans. Eur. J. Clin. Nutr. 2005, 59, 906–913. [Google Scholar] [CrossRef]
- Liu, F.; Kariluoto, S.; Edelmann, M.; Piironen, V. Bioaccessibility of folate in faba bean, oat, rye and wheat matrices. Food Chem. 2021, 350, 129259. [Google Scholar] [CrossRef]
- Mo, H.; Kariluoto, S.; Piironen, V.; Zhu, Y.; Sanders, M.G.; Vincken, J.P.; Wolkers-Rooijackers, J.; Nout, M.J. Effect of soybean processing on content and bioaccessibility of folate, vitamin B12 and isoflavones in tofu and tempe. Food Chem. 2013, 141, 2418–2425. [Google Scholar] [CrossRef]
- Bationo, F.; Humblot, C.; Songré-Ouattara, L.T.; Hama-Ba, F.; Le Merrer, M.; Chapron, M.; Kariluoto, S.; Hemery, Y.M. Total folate in West African cereal-based fermented foods: Bioaccessibility and influence of processing. J. Food Compos. Anal. 2020, 85, 103309. [Google Scholar] [CrossRef]
- Wang, C.; Riedl, K.M.; Somerville, J.; Balasubramaniam, V.M.; Schwartz, S.J. Influence of high-pressure processing on the profile of polyglutamyl 5-methyltetrahydrofolate in selected vegetables. J. Agric. Food Chem. 2011, 59, 8709–8717. [Google Scholar] [CrossRef]
- Wang, C.; Riedl, K.M.; Schwartz, S.J. Fate of folates during vegetable juice processing—Deglutamylation and interconversion. Food Res. Int. 2013, 53, 440–448. [Google Scholar] [CrossRef]
- Munyaka, A.W.; Oey, I.; Verlinde, P.; Van Loey, A.; Hendrickx, M. Acidification, crushing and thermal treatments can influence the profile and stability of folate poly-γ-glutamates in broccoli (Brassica oleracea L. var. italica). Food Chem. 2009, 117, 568–575. [Google Scholar] [CrossRef]
- Garza-Aguilar, S.M.; García-Salinas, C.; Mejía-Ponce, P.M.; Licona-Cassani, C.; Ramos-Parra, P.A.; de la Garza, R.I.D. The complexity of folate polyglutamylation in plants: Postharvest ripening and ethylene modulate polyglutamylated profiles in climacteric fruits plus systematic analysis of the glutamyl tail-editing enzymes. Sci. Hortic. 2020, 273, 109588. [Google Scholar] [CrossRef]
- McKillop, D.J.; McNulty, H.; Scott, J.M.; McPartlin, J.M.; Strain, J.J.; Bradbury, I.; Girvan, J.; Hoey, L.; McCreedy, R.; Alexander, J.; et al. The rate of intestinal absorption of natural food folates is not related to the extent of folate conjugation. Am. J. Clin. Nutr. 2006, 84, 167–173. [Google Scholar] [CrossRef]
- Pamunuwa, G.; Anjalee, N.; Kukulewa, D.; Edirisinghe, C.; Shakoor, F.; Karunaratne, D.N. Tailoring of release properties of folic acid encapsulated nanoparticles via changing alginate and pectin composition in the matrix. Carbohydr. Polym. Technol. Appl. 2020, 1, 100008. [Google Scholar] [CrossRef]
- Mohammed, A.Y.; Dyab, A.K.F.; Taha, F.; Abd El-Mageed, A.I.A. Encapsulation of folic acid (vitamin B9) into sporopollenin microcapsules: Physico-chemical characterisation, in vitro controlled release and photoprotection study. Mater. Sci. Eng. C 2021, 128, 112271. [Google Scholar] [CrossRef]
- Osojnik Crnivec, I.G.; Istenic, K.; Skrt, M.; Poklar Ulrih, N. Thermal protection and pH-gated release of folic acid in microparticles and nanoparticles for food fortification. Food Funct. 2020, 11, 1467–1477. [Google Scholar] [CrossRef]
- Tu, W.; Yan, F.; Chao, B.; Ji, X.; Wang, L. Status of hyperhomocysteinemia in China: Results from the China Stroke High-risk Population Screening Program, 2018. Front. Med. 2021, 15, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Franco, C.; Sciatti, E.; Favero, G.; Bonomini, F.; Vizzardi, E.; Rezzani, R. Essential hypertension and oxidative stress: Novel future perspectives. Int. J. Mol. Sci. 2022, 23, 14489. [Google Scholar] [CrossRef] [PubMed]
- Bourgonje, A.R.; Abdulle, A.E.; Al-Rawas, A.M.; Al-Maqbali, M.; Al-Saleh, M.; Enriquez, M.B.; Al-Siyabi, S.; Al-Hashmi, K.; Al-Lawati, I.; Bulthuis, M.L.C.; et al. Systemic oxidative stress is increased in postmenopausal women and independently associates with homocysteine levels. Int. J. Mol. Sci. 2020, 21, 314. [Google Scholar] [CrossRef]
- Pravenec, M.; Kozich, V.; Krijt, J.; Sokolová, J.; Zídek, V.; Landa, V.; Simáková, M.; Mlejnek, P.; Silhavy, J.; Oliyarnyk, O.; et al. Folate deficiency is associated with oxidative stress, increased blood pressure, and insulin resistance in spontaneously hypertensive rats. Am. J. Hypertens. 2013, 26, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, Z.; Xing, C.; Gao, N.; Xu, R. Folate reverses NF-κB p65/Rela/IL-6 level induced by hyperhomocysteinemia in spontaneously hypertensive rats. Front. Pharmacol. 2021, 12, 651582. [Google Scholar] [CrossRef]
- Gao, N.; Zhang, Y.; Lei, L.; Li, L.; Cao, P.; Zhao, X.; Lin, L.; Xu, R. Low doses of folic acid can reduce hyperhomocysteinemia-induced glomerular injury in spontaneously hypertensive rats. Hypertens. Res. 2020, 43, 1182–1191. [Google Scholar] [CrossRef]
- Li, Q.; Xu, S.; Chen, X.; Zhang, X.; Li, X.; Lin, L.; Gao, D.; Wu, M.; Yang, S.; Cao, X.; et al. Folic acid supplement use and increased risk of gestational hypertension. Hypertension 2020, 76, 150–156. [Google Scholar] [CrossRef]
- Pushpakumar, S.B.; Kundu, S.; Metreveli, N.; Sen, U. Folic Acid Mitigates Angiotensin-II-Induced Blood Pressure and Renal Remodeling. PLoS ONE 2013, 8, e83813. [Google Scholar] [CrossRef]
- Deng, Y.; Li, Z.; An, X.; Fan, R.; Wang, Y.; Li, J.; Yang, X.; Liao, J.; Xia, Y. Hyperhomocysteinemia promotes cardiac hypertrophy in hypertension. Oxid. Med. Cell. Longev. 2022, 2022, 1486157. [Google Scholar] [CrossRef]
- Pan, S.; Liu, H.; Gao, F.; Luo, H.; Lin, H.; Meng, L.; Jiang, C.; Guo, Y.; Chi, J.; Guo, H. Folic acid delays development of atherosclerosis in low-density lipoprotein receptor-deficient mice. J. Cell. Mol. Med. 2018, 22, 3183–3191. [Google Scholar] [CrossRef]
- Zhong, X.; He, R.; You, S.; Liu, B.; Wang, X.; Mao, J. The roles of aerobic exercise and folate supplementation in hyperhomocysteinemia-accelerated atherosclerosis. Acta Cardiol. Sinica 2023, 39, 309–318. [Google Scholar] [CrossRef]
- Mohammadian, Z.; Eidi, A.; Mortazavi, P.; Tavangar, S.M.; Asghari, A. Effects of folic acid on dyslipidemia and serum Homocysteine in a rat model of cholestasis and hepatic fibrosis. Pol. J. Pathol. 2015, 66, 49–56. [Google Scholar] [CrossRef]
- Kemse, N.G.; Kale, A.A.; Joshi, S.R. A combined supplementation of omega-3 fatty acids and micronutrients (folic Acid, vitamin B) reduces oxidative stress markers in a rat model of pregnancy induced hypertension. PLoS ONE 2014, 9, e111902. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Yu, J.; Jia, B.; Zhao, F.; Tang, M.; Hu, L.; Lin, F. Effect of losartan with folic acid on plasma homocysteine and vascular ultrastructural changes in spontaneously hypertensive rats. Int. J. Clin. Exp. Path. 2015, 8, 12908–12914. [Google Scholar]
- Duthie, S.J.; Narayanan, S.; Brand, G.M.; Pirie, L.; Grant, G. Impact of folate deficiency on DNA stability. J. Nutr. 2002, 132, s2444–s2449. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Murray, J.P.; Prykhozhij, S.V.; Dufay, J.N.; Steele, S.L.; Gaston, D.; Nasrallah, G.K.; Coombs, A.J.; Liwski, R.S.; Fernandez, C.V.; Berman, J.N.; et al. Glycine and Folate Ameliorate Models of Congenital Sideroblastic Anemia. PLoS Genet. 2016, 12, e1005783. [Google Scholar] [CrossRef]
- Cerreto, G.M.; Pozzi, G.; Cortellazzi, S.; Pasini, L.M.; Di Martino, O.; Mirandola, P.; Carubbi, C.; Vitale, M.; Masselli, E. Folate metabolism in myelofibrosis: A missing key? Ann. Hematol. 2025, 104, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Geiger, H. Balance your folate or the yin and yang of folate in hematopoiesis. Haematologica 2017, 102, 1969–1970. [Google Scholar] [CrossRef]
- Henry, C.J.; Nemkov, T.; Casás-Selves, M.; Bilousova, G.; Zaberezhnyy, V.; Higa, K.C.; Serkova, N.J.; Hansen, K.C.; D’Alessandro, A.; DeGregori, J. Folate dietary insufficiency and folic acid supplementation similarly impair metabolism and compromise hematopoiesis. Haematologica 2017, 102, 1985–1994. [Google Scholar] [CrossRef]
- Koury, M.J.; Ponka, P. New insights into erythropoiesis: The roles of folate, vitamin B12, and iron. Annu. Rev. Nutr. 2004, 24, 105–131. [Google Scholar] [CrossRef]
- Magana, A.A.; Reed, R.L.; Koluda, R.; Miranda, C.L.; Maier, C.S.; Stevens, J.F. Vitamin C Activates the Folate-Mediated One-Carbon Cycle in C2C12 Myoblasts. Antioxidants 2020, 9. [Google Scholar] [CrossRef]
- Takahashi, A. Zinc Supplementation Enhances the Hematopoietic Activity of Erythropoiesis-Stimulating Agents but Not Hypoxia-Inducible Factor-Prolyl Hydroxylase Inhibitors. Nutrients 2024, 16, 520. [Google Scholar] [CrossRef]
- Gao, J.; Li, Y.B.; Wan, Y.; Hu, T.T.; Liu, L.T.; Yang, S.J.; Gong, Z.L.; Zeng, Q.; Wei, Y.; Yang, W.J.; et al. A novel postbiotic from Lactobacillus rhamnosus GG with a beneficial effect on intestinal barrier function. Front. Microbiol. 2019, 10, 477. [Google Scholar] [CrossRef] [PubMed]
- Pham, V.; Dold, S.; Rehman, A.; Bird, J.K.; Steinert, R.E. Vitamins, the gut microbiome and gastrointestinal health in humans. Nutr. Res. 2021, 95, 35–53. [Google Scholar] [CrossRef]
- Hossain, K.S.; Amarasena, S.; Mayengbam, S. B citamins and their roles in gut health. Microorganisms 2022, 10, 1168. [Google Scholar] [CrossRef]
- Wang, T.; Chen, L.; Huang, P.; Yang, T.; Zhang, S.; Zhao, L.; Chen, L.; Ye, Z.; Luo, L.; Qin, J. Association of maternal gut microbiota and plasma metabolism with congenital heart disease in offspring: A multi-omic analysis. Sci. Rep. 2021, 11, 5339. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, J.; Liu, X.; Liu, R.; Wang, Y.; Huang, X.; Li, Y.; Liu, R.; Yang, X. Dietary folic acid addition reduces abdominal fat deposition mediated by alterations in gut microbiota and SCFA production in broilers. Anim. Nutr. 2023, 12, 54–62. [Google Scholar] [CrossRef]
- Peredo-Lovillo, A.; Romero-Luna, H.E.; Jiménez-Fernández, M. Health promoting microbial metabolites produced by gut microbiota after prebiotics metabolism. Food Res. Int. 2020, 136, 109473. [Google Scholar] [CrossRef]
- Sorbara, M.T.; Dubin, K.; Littmann, E.R.; Moody, T.U.; Fontana, E.; Seok, R.; Leiner, I.M.; Taur, Y.; Peled, J.U.; van den Brink, M.R.M.; et al. Inhibiting antibiotic-resistant Enterobacteriaceae by microbiota-mediated intracellular acidification. J. Exp. Med. 2019, 216, 84–98. [Google Scholar] [CrossRef]
- Zheng, X.; Xia, C.; Liu, M.; Wu, H.; Yan, J.; Zhang, Z.; Huang, Y.; Gu, Q.; Li, P. Role of folic acid in regulating gut microbiota and short-chain fatty acids based on an in vitro fermentation model. Appl. Microbiol. Biotechnol. 2024, 108, 10–15. [Google Scholar] [CrossRef]
- Mir, S.A.; Nagy-Szakal, D.; Dowd, S.E.; Szigeti, R.G.; Smith, C.W.; Kellermayer, R. Prenatal methyl-donor supplementation augments colitis in young adult mice. PLoS ONE 2013, 8, e73162. [Google Scholar] [CrossRef]
- Dziedzic, A.; Maciak, K.; Blizniewska-Kowalska, K.; Galecka, M.; Kobierecka, W.; Saluk, J. The power of psychobiotics in depression: A modern approach through the microbiota-gut-brain axis: A literature review. Nutrients 2024, 16, 1054. [Google Scholar] [CrossRef]
- Zhou, Y.; Cong, Y.; Liu, H. Folic acid ameliorates depression-like behaviour in a rat model of chronic unpredictable mild stress. BMC Neurosci. 2020, 21, 1. [Google Scholar] [CrossRef]
- Di Tommaso, N.; Gasbarrini, A.; Ponziani, F.R. Intestinal barrier in human health and disease. Int. J. Environ. Res. Public Health. 2021, 18, 12836. [Google Scholar] [CrossRef] [PubMed]
- Kayama, H.; Okumura, R.; Takeda, K. Interaction between the microbiota, epithelia, and immune cells in the intestine. Annu. Rev. Immunol. 2020, 38, 23–48. [Google Scholar] [CrossRef] [PubMed]
- Martel, J.; Chang, S.H.; Ko, Y.F.; Hwang, T.L.; Young, J.D.; Ojcius, D.M. Gut barrier disruption and chronic disease. Trends Endocrinol. Metab. 2022, 33, 247–265. [Google Scholar] [CrossRef]
- Zhu, S.; Li, J.; Bing, Y.; Yan, W.; Zhu, Y.; Xia, B.; Chen, M. Diet-induced hyperhomocysteinaemia increases intestinal inflammation in an animal model of colitis. J. Crohns Colitis 2015, 9, 708–719. [Google Scholar] [CrossRef] [PubMed]
- Gurwara, S.; Ajami, N.J.; Jang, A.; Hessel, F.C.; Chen, L.; Plew, S.; Wang, Z.; Graham, D.Y.; Hair, C.; White, D.L.; et al. Dietary nutrients involved in one-carbon metabolism and colonic mucosa-associated gut microbiome in individuals with an endoscopically normal colon. Nutrients 2019, 11, 613. [Google Scholar] [CrossRef]
- Chen, S.; Yang, M.; Wang, R.; Fan, X.; Tang, T.; Li, P.; Zhou, X.; Qi, K. Suppression of high-fat-diet-induced obesity in mice by dietary folic acid supplementation is linked to changes in gut microbiota. Eur. J. Nutr. 2022, 61, 2015–2031. [Google Scholar] [CrossRef]
- Sun, X.; Wen, J.; Guan, B.; Li, J.; Luo, J.; Li, J.; Wei, M.; Qiu, H. Folic acid and zinc improve hyperuricemia by altering the gut microbiota of rats with high-purine diet-induced hyperuricemia. Front. Microbiol. 2022, 13, 907952. [Google Scholar] [CrossRef]
- Margolis, K.G.; Cryan, J.F.; Mayer, E.A. The microbiota-gut-brain axis: From motility to mood. Gastroenterology 2021, 160, 1486–1501. [Google Scholar] [CrossRef]
- Akinyemi, F.T.; Adewole, D.I. Effect of dietary folic acid and energy density on immune response, gut morphology, and oxidative status in blood and breast muscle of broiler chickens. Can. J. Anim. Sci. 2022, 102, 243–254. [Google Scholar] [CrossRef]
- Gouda, A.; Amer, S.A.; Gabr, S.; Tolba, S.A. Effect of dietary supplemental ascorbic acid and folic acid on the growth performance, redox status, and immune status of broiler chickens under heat stress. Trop. Anim. Health Prod. 2020, 52, 2987–2996. [Google Scholar] [CrossRef]
- Lin, Y.H.; Lin, H.Y.; Shiau, S.Y. Dietary folic acid requirement of grouper, Epinephelus malabaricus, and its effects on non-specific immune responses. Aquaculture 2011, 317, 133–137. [Google Scholar] [CrossRef]
- Li, S.; Zhi, L.; Liu, Y.; Shen, J.; Liu, L.; Yao, J.; Yang, X. Effect of in ovo feeding of folic acid on the folate metabolism, immune function and epigenetic modification of immune effector molecules of broiler. Br. J. Nutr. 2016, 115, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, M.; Kayama, H.; Kusu, T.; Yamaguchi, T.; Kunisawa, J.; Kiyono, H.; Sakaguchi, S.; Takeda, K. Dietary folic acid promotes survival of Foxp3+ regulatory T cells in the colon. J. Immunol. 2012, 189, 2869–2878. [Google Scholar] [CrossRef]
- Kunisawa, J.; Hashimoto, E.; Ishikawa, I.; Kiyono, H. A pivotal role of vitamin B9 in the maintenance of regulatory T cells in vitro and in vivo. PLoS ONE 2012, 7, e32094. [Google Scholar] [CrossRef] [PubMed]
- Meadows, D.N.; Bahous, R.H.; Best, A.F.; Rozen, R. High dietary folate in mice alters immune response and reduces survival after malarial infection. PLoS ONE 2015, 10, e0143738. [Google Scholar] [CrossRef]
- Visentin, D.; Gobin, I.; Maglica, Z. Periodontal pathogens and their links to neuroinflammation and neurodegeneration. Microorganisms 2023, 11, 1832. [Google Scholar] [CrossRef] [PubMed]
- Leal, S.L.; Lockhart, S.N.; Maass, A.; Bell, R.K.; Jagust, W.J. Subthreshold amyloid predicts tau deposition in aging. J. Neurosci. 2018, 38, 4482–4489. [Google Scholar] [CrossRef]
- Zhao, M.; Yuan, M.M.; Yuan, L.; Huang, L.L.; Liao, J.H.; Yu, X.L.; Su, C.; Chen, Y.H.; Yang, Y.Y.; Yu, H.; et al. Chronic folate deficiency induces glucose and lipid metabolism disorders and subsequent cognitive dysfunction in mice. PLoS ONE 2018, 13, e0202910. [Google Scholar] [CrossRef]
- Zhu, M.; Li, B.; Ma, X.; Huang, C.; Wu, R.; Zhu, W.; Li, X.; Liang, Z.; Deng, F.; Zhu, J.; et al. Folic acid protected neural cells against aluminum-maltolate-induced apoptosis by preventing miR-19 downregulation. Neurochem. Res. 2016, 41, 2110–2118. [Google Scholar] [CrossRef]
- Li, W.; Ma, Y.; Li, Z.; Lv, X.; Wang, X.; Zhou, D.; Luo, S.; Wilson, J.X.; Huang, G. Folic acid decreases astrocyte apoptosis by preventing oxidative stress-induced telomere attrition. Int. J. Mol. Sci. 2020, 21, 62. [Google Scholar] [CrossRef]
- Yu, H.; Li, L.; Zhang, X.; Xiang, L.; Zhang, J.; Feng, J.; Xiao, R. Neuroprotective effects of genistein and folic acid on apoptosis of rat cultured cortical neurons induced by β-amyloid 31–35. Br. J. Nutr. 2009, 102, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, H.; Yu, M.; Zhang, X.; Zhang, Y.; Liu, H.; Wilson, J.; Huang, G. Folic acid alters methylation profile of JAK-STAT and long-term depression signaling pathways in Alzheimer’s disease models. Mol. Neurobiol. 2016, 53, 6548–6556. [Google Scholar] [CrossRef] [PubMed]
- Koohpeyma, H.; Goudarzi, I.; Salmani, M.E.; Lashkarbolouki, T.; Shabani, M. Postnatal administration of homocysteine induces cerebellar damage in rats: Protective effect of folic acid. Neurotoxic. Res. 2019, 35, 724–738. [Google Scholar] [CrossRef]
- Zhao, H.; Liang, J.; Li, X.; Yu, H.; Li, X.; Xiao, R. Folic acid and soybean isoflavone combined supplementation protects the post-neural tube closure defects of rodents induced by cyclophosphamide in vivo and in vitro. Neurotoxicology 2010, 31, 180–187. [Google Scholar] [CrossRef]
- Chen, H.; Liu, S.; Ji, L.; Wu, T.; Ji, Y.; Zhou, Y.; Zheng, M.; Zhang, M.; Xu, W.; Huang, G. Folic acid supplementation mitigates Alzheimer’s disease by reducing inflammation: A randomized controlled trial. Mediat. Inflamm. 2016, 2016, 5912146. [Google Scholar] [CrossRef] [PubMed]
- Aisen, P.S.; Schneider, L.S.; Sano, M.; Diaz-Arrastia, R.; van Dyck, C.H.; Weiner, M.F.; Bottiglieri, T.; Jin, S.; Stokes, K.T.; Thomas, R.G.; et al. High-dose B vitamin supplementation and cognitive decline in Alzheimer disease—A randomized controlled trial. JAMA 2008, 300, 1774–1783. [Google Scholar] [CrossRef]
- Bailey, R.L.; Jun, S.; Murphy, L.; Green, R.; Gahche, J.J.; Dwyer, J.T.; Potischman, N.; McCabe, G.P.; Miller, J.W. High folic acid or folate combined with low vitamin B-12 status: Potential but inconsistent association with cognitive function in a nationally representative cross-sectional sample of US older adults participating in the NHANES. Am. J. Clin. Nutr. 2020, 112, 1547–1557. [Google Scholar] [CrossRef]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct. Target. Ther. 2018, 3, 7. [Google Scholar] [CrossRef]
- Perales, S.; Torres, C.; Jimenez-Luna, C.; Prados, J.; Martinez-Galan, J.; Sanchez-Manas, J.M.; Caba, O. Liquid biopsy approach to pancreatic cancer. World J. Gastrointest. Oncol. 2021, 13, 1263–1287. [Google Scholar] [CrossRef] [PubMed]
- Chiaradonna, F.; Ricciardiello, F.; Palorini, R. The nutrient-sensing hexosamine biosynthetic pathway as the hub of cancer metabolic rewiring. Cells 2018, 7, 53. [Google Scholar] [CrossRef]
- Sanchez, H.; Hossain, M.B.; Lera, L.; Hirsch, S.; Albala, C.; Uauy, R.; Broberg, K.; Ronco, A.M. High levels of circulating folate concentrations are associated with DNA methylation of tumor suppressor and repair genes p16, MLH1, and MGMT in elderly Chileans. Clin. Epigenet. 2017, 9, 74. [Google Scholar] [CrossRef] [PubMed]
- Taira, J.; Ogi, T. Nitric oxide modulation by folic acid fortification. Antioxidants 2020, 9, 393. [Google Scholar] [CrossRef]
- Asbaghi, O.; Ghanavati, M.; Ashtary-Larky, D.; Bagheri, R.; Kelishadi, M.R.; Nazarian, B.; Nordvall, M.; Wong, A.; Dutheil, F.; Suzuki, K.; et al. Effects of folic acid supplementation on oxidative stress Markers: A systematic review and meta-analysis of randomized controlled trials. Antioxidants 2021, 10, 871. [Google Scholar] [CrossRef]
- Zsigrai, S.; Kalmár, A.; Barták, B.K.; Nagy, Z.B.; Szigeti, K.A.; Valcz, G.; Kothalawala, W.; Dankó, T.; Sebestyén, A.; Barna, G.; et al. Folic acid treatment directly influences the genetic and epigenetic regulation along with the associated cellular maintenance processes of HT-29 and SW480 colorectal cancer cell lines. Cancers 2022, 14, 1820. [Google Scholar] [CrossRef] [PubMed]
- Stolzenberg-Solomon, R.Z.; Chang, S.C.; Leitzmann, M.F.; Johnson, K.A.; Johnson, C.; Buys, S.S.; Hoover, R.N.; Ziegler, R.G. Folate intake, alcohol use, and postmenopausal breast cancer risk in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Am. J. Clin. Nutr. 2006, 83, 895–904. [Google Scholar] [CrossRef]
- Chang, W.C.L.; Ghosh, J.; Cooper, H.S.; Vanderveer, L.; Schultz, B.; Zhou, Y.; Harvey, K.N.; Kaunga, E.; Devarajan, K.; Li, Y.; et al. Folic acid supplementation promotes hypomethylation in both the inflamed colonic mucosa and colitis-associated dysplasia. Cancers 2023, 15, 2949. [Google Scholar] [CrossRef]
- Huang, W.; Huang, T.; Liu, Y.; Fu, J.; Wei, X.; Liu, D.; Ma, W.; Gu, H.; Yuan, Z. Nuclear factor I-C disrupts cellular homeostasis between autophagy and apoptosis via miR-200b-Ambra1 in neural tube defects. Cell Death Dis. 2022, 13, 17. [Google Scholar] [CrossRef]
- Wu, Y.; Peng, S.S.; Finnell, R.H.; Zheng, Y.F. Organoids as a new model system to study neural tube defects. FASEB J. 2021, 35, e21545. [Google Scholar] [CrossRef]
- Zaganjor, I.; Sekkarie, A.; Tsang, B.L.; Williams, J.; Razzaghi, H.; Mulinare, J.; Sniezek, J.E.; Cannon, M.J.; Rosenthal, J. Describing the prevalence of neural tube defects worldwide: A systematic literature review. PLoS ONE 2016, 11, e0151586. [Google Scholar] [CrossRef] [PubMed]
- Cawley, S.; McCartney, D.; Woodside, J.V.; Sweeney, M.R.; McDonnell, R.; Molloy, A.M.; Turner, M.J. Optimization of folic acid supplementation in the prevention of neural tube defects. J. Public Health 2018, 40, 827–834. [Google Scholar] [CrossRef]
- Pei, P.; Cheng, X.; Yu, J.; Shen, J.; Li, X.; Wu, J.; Wang, S.; Zhang, T. Folate deficiency induced H2A ubiquitination to lead to downregulated expression of genes involved in neural tube defects. Epigenet. Chromatin. 2019, 12, 69. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Chen, Y.; Chen, X.; Dong, X.; Zhou, J.; Wang, H.; Wu, S.; Zhang, C.; Xu, D. Folic acid supplementation during pregnancy protects against lipopolysaccharide-induced neural tube defects in mice. Toxicol. Lett. 2014, 224, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Hu, Y.; Chen, X.; Li, K. Folic acid supplementation diminishes diabetes induced neural tube defects by recovering impaired embryo gene expression through its antioxidant activity. J. Pharm. Res. Int. 2021, 33, 258–271. [Google Scholar] [CrossRef]
- Dawson, J.E.; Raymond, A.M.; Winn, L.M. Folic acid and pantothenic acid protection against valproic acid-induced neural tube defects in CD-1 mice. Toxicol. Appl. Pharmacol. 2006, 211, 124–132. [Google Scholar] [CrossRef]
- Dolin, C.D.; Deierlein, A.L.; Evans, M.I. Folic acid supplementation to prevent recurrent neural tube defects: 4 milligrams is too much. Fetal Diagn. Ther. 2018, 44, 161–165. [Google Scholar] [CrossRef]
- Li, K.; Shao, X.; Li, H.; Kuang, X.; Song, X.; Wang, Y.; Zhu, S.; Li, D. Synergistic effects of folic acid and n-3 polyunsaturated fatty acid in preventing neural tube defects in diabetic mice. J. Agric. Food Chem. 2022, 70, 11281–11289. [Google Scholar] [CrossRef]
- Kancherla, V.; Tsang, B.; Wagh, K.; Dixon, M.; Oakley, G.P. Modeling shows high potential of folic acid-fortified salt to accelerate global prevention of major neural tube defects. Birth Defects Res. 2020, 112, 1461–1474. [Google Scholar] [CrossRef] [PubMed]
- Fardous, A.M.; Heydari, A.R. Uncovering the Hidden Dangers and Molecular Mechanisms of Excess Folate: A Narrative Review. Nutrients 2023, 15, 4699. [Google Scholar] [CrossRef]
- Selhub, J.; Rosenberg, I.H. Excessive folic acid intake and relation to adverse health outcome. Biochimie 2016, 126, 71–78. [Google Scholar] [CrossRef]
- Patel, K.R.; Sobczyńska-Malefora, A. The adverse effects of an excessive folic acid intake. Eur. J. Clin. Nutr. 2017, 71, 159–163. [Google Scholar] [CrossRef]
- Chan, Y.M.; Bailey, R.; O’Connor, D.L. Folate. Adv. Nutr. 2013, 4, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Suitor, C.W.; Bailey, L.B. Dietary folate equivalents: Interpretation and application. J. Am. Diet. Assoc. 2000, 100, 88–94. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Duan, H.; Ramaswamy, H.; Wang, C. A Comprehensive Review of Fortification, Bioavailability, and Health Benefits of Folate. Int. J. Mol. Sci. 2025, 26, 7703. https://doi.org/10.3390/ijms26167703
Li J, Duan H, Ramaswamy H, Wang C. A Comprehensive Review of Fortification, Bioavailability, and Health Benefits of Folate. International Journal of Molecular Sciences. 2025; 26(16):7703. https://doi.org/10.3390/ijms26167703
Chicago/Turabian StyleLi, Jiarun, Hanying Duan, Hosahalli Ramaswamy, and Chao Wang. 2025. "A Comprehensive Review of Fortification, Bioavailability, and Health Benefits of Folate" International Journal of Molecular Sciences 26, no. 16: 7703. https://doi.org/10.3390/ijms26167703
APA StyleLi, J., Duan, H., Ramaswamy, H., & Wang, C. (2025). A Comprehensive Review of Fortification, Bioavailability, and Health Benefits of Folate. International Journal of Molecular Sciences, 26(16), 7703. https://doi.org/10.3390/ijms26167703








