Septic Hyperinflammation—Is There a Role for Extracorporeal Blood Purification Techniques?
Abstract
1. Introduction
1.1. What Is Septic Hyperinflammation, and Why Should It Be Treated?
- Antimicrobial therapy: Prompt initiation of broad-spectrum antibiotics, tailored based on suspected sources of infection and local microbial resistance patterns.
- Source control: Identification and management of the infectious source, such as drainage of abscesses or removal of infected devices.
- Fluid resuscitation: Administration of intravenous fluids to restore hemodynamic stability.
- Supportive care: Oxygen supplementation and use of vasopressors if necessary to maintain adequate blood pressure.
- Continuation and adjustment of antimicrobial therapy based on microbiological findings and clinical response.
- Enhanced supportive care, including mechanical ventilation for respiratory failure and renal replacement therapy for acute kidney injury.
- Glycemic control and nutritional support as adjunctive treatments.
- Aggressive hemodynamic support with fluids and multiple vasopressors to maintain systemic perfusion.
- In refractory shock: adjunctive hydrocortisone (200 mg/d).
- Consideration of inotropic support for myocardial dysfunction.
- Blood purification techniques: Introduced in this stage for patients with refractory shock and/or significant organ dysfunction, aimed at removing excessively elevated inflammatory mediators and toxins. Blood purification techniques are considered adjunctive therapies and are typically reserved for cases where conventional treatments fail to stabilize the patient or when there is evidence of overwhelming inflammation contributing to organ dysfunction [6]. Their use is guided by the severity of sepsis, the patient’s response to initial treatments, and the presence of complications such as severe metabolic derangements or refractory shock.
1.2. Immune Response Mechanisms in Sepsis: From Recognition to Regulation
1.3. Neutrophils in Sepsis: Roles in Defense, Hyperinflammation, and Organ Damage
1.4. Endothelial Dysfunction and Thromboinflammation in Hyperinflammatory Diseases
1.5. Complement System Activation and Immunothrombosis in Sepsis and Systemic Inflammation
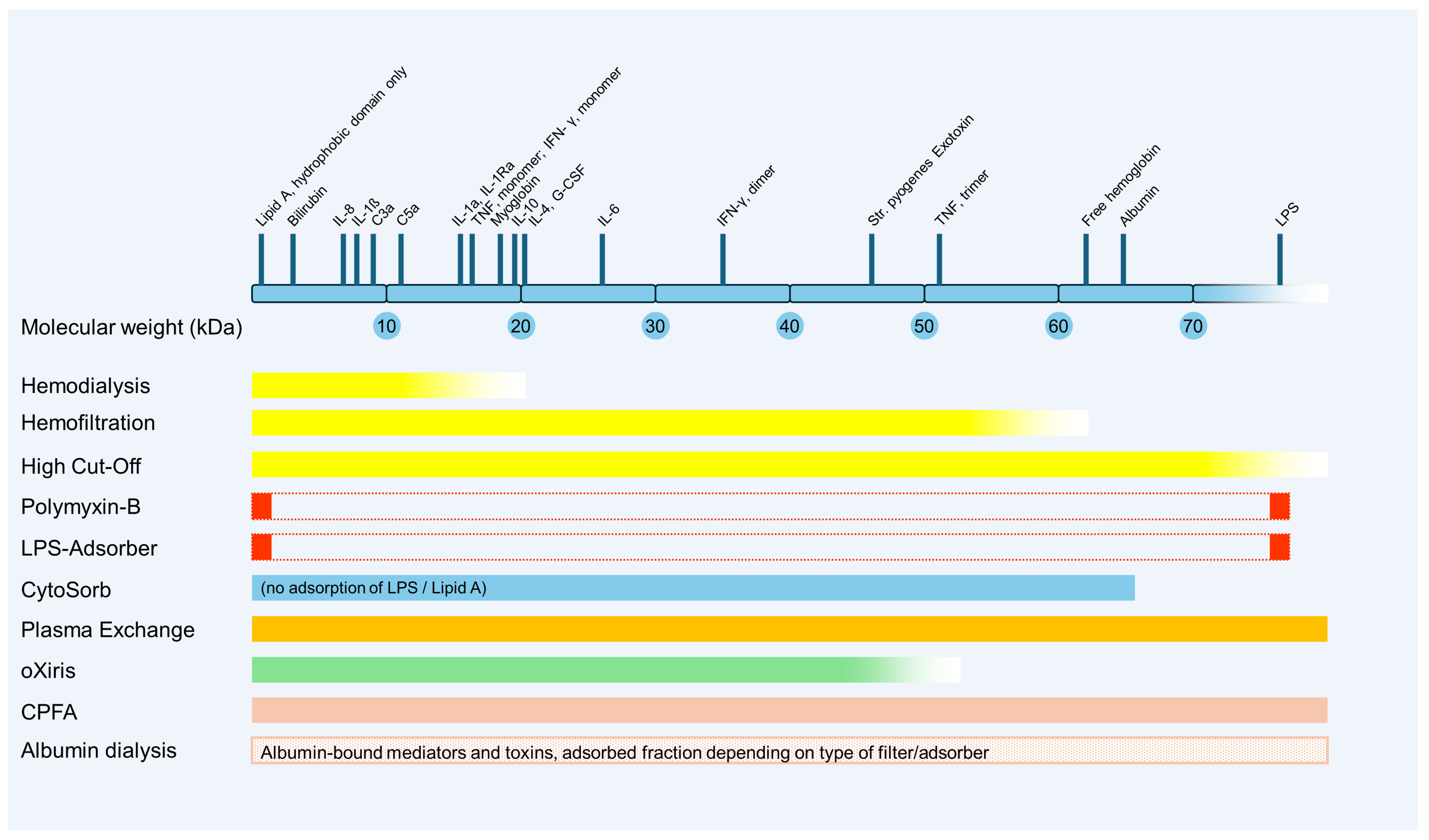
2. Renal Replacement Therapies (RRTs)
2.1. High-Cut-Off Membranes
2.2. High-Volume Hemofiltration
3. Adsorption
3.1. Polymyxin B-Immobilized Fiber Columns (Specific Hemoadsorption)
3.2. LPS Adsorber
3.3. CytoSorb® (Unspecific Hemadsorption)
4. Therapeutic Plasma Exchange (TPE)
5. Combination Methods
5.1. oXiris®
5.2. Coupled Plasma Filtration Adsorption (CPFA)
6. Albumin Dialysis
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACLF | Acute on chronic liver failure |
| ADAMTS13 | A disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 |
| ADP | Adenosine diphosphate |
| AKI | Acute kidney injury |
| ALF | Acute liver failure |
| APC | Antigen-presenting cells |
| CPFA | Coupled plasma filtration adsorption |
| CRRT | Continuous renal replacement therapy |
| DAMP | Pathogen-associated molecular patterns |
| DIC | Disseminated intravascular coagulopathy |
| DNA | Deoxyribonucleic acid |
| EAA | Endotoxin activity assay |
| ECLS | Extracorporeal liver support |
| ECMO | Extracorporeal membrane oxygenation |
| FFP | Fresh frozen plasma |
| FPSA | Fractional plasma separation and adsorption |
| HCO | High cut-off |
| HE | Hepatic encephalopathy |
| HVHF | High-volume hemofiltration |
| ICU | Intensive care unit |
| IFN | Interferon |
| IL | Interleukin |
| IL-1ra | Interleukin-1 receptor antagonist |
| KDIGO | Kidney Disease Improving Global Outcomes |
| LPS | Lipopolysaccharide |
| MARS | Molecular adsorbent recirculating system |
| NET | Neutrophil extracellular traps |
| NF-κB | Nuclear factor-kappa-light-chain-enhancer of activated B cells |
| NOS | Newcastle–Ottawa scale |
| PAMP | Pathogen-associated molecular patterns |
| PEI | Polyethyleneimine |
| PMX | Polymyxin B-immobilized fiber columns |
| PRR | Pattern recognition receptor |
| RCT | Randomized clinical trial |
| SIC | Sepsis induced coagulopathy |
| SPAD | Single-pass albumin dialysis |
| SOFA | Sequential organ failure assessment |
| TAMOF | Thrombocytopenia-associated multiple organ failure |
| TF | Tissue factor |
| TFPI | Tissue factor pathway inhibitor |
| TLR | Toll-like receptor |
| TMA | Thrombocytopenic microangiopathy |
| TNF | Tumor necrosis factor |
| TPE | Therapeutic plasma exchange |
| TTP | Thrombotic thrombocytopenic purpura |
| VHVHF | Very-high-volume hemofiltration |
| vWF | von Willebrand factor |
| vWFCP | von Willebrand factor-cleaving protease |
References
- Rudd, K.E.; Johnson, S.C.; Agesa, K.M.; Shackelford, K.A.; Tsoi, D.; Kievlan, D.R.; Colombara, D.V.; Ikuta, K.S.; Kissoon, N.; Finfer, S.; et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: Analysis for the Global Burden of Disease Study. Lancet 2020, 395, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.L.; Jones, G.; David, S.; Olariu, E.; Cadwell, K.K. Frequency and mortality of septic shock in Europe and North America: A systematic review and meta-analysis. Crit. Care 2019, 23, 196. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Tisoncik, J.R.; Korth, M.J.; Simmons, C.P.; Farrar, J.; Martin, T.R.; Katze, M.G. Into the eye of the cytokine storm. Microbiol. Mol. Biol. Rev. 2012, 76, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.M.; Rhodes, A.; Phillips, G.S.; Townsend, S.R.; Schorr, C.A.; Beale, R.; Osborn, T.; Lemeshow, S.; Chiche, J.D.; Artigas, A.; et al. Surviving Sepsis Campaign: Association between performance metrics and outcomes in a 7.5-year study. Crit. Care Med. 2015, 43, 3–12. [Google Scholar] [CrossRef]
- Kovzel, V.A.; Davydova, L.A.; Karzin, A.V.; Tsarenko, S.V.; Baturova, V.Y.; Polupan, A.A.; Gutnikov, A.I. Methods of Extracorporeal Hemocorrection in Sepsis (Review). Gen. Reanimatol. 2023, 19, 68–82. [Google Scholar] [CrossRef]
- Chousterman, B.G.; Swirski, F.K.; Weber, G.F. Cytokine storm and sepsis disease pathogenesis. Semin. Immunopathol. 2017, 39, 517–528. [Google Scholar] [CrossRef]
- Kemball, C.C.; Alirezaei, M.; Whitton, J.L. Type B coxsackieviruses and their interactions with the innate and adaptive immune systems. Future Microbiol. 2010, 5, 1329–1347. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Moldawer, L.L.; Opal, S.M.; Reinhart, K.; Turnbull, I.R.; Vincent, J.L. Sepsis and septic shock. Nat. Rev. Dis. Primers 2016, 2, 16045. [Google Scholar] [CrossRef] [PubMed]
- Tamayo, E.; Fernandez, A.; Almansa, R.; Carrasco, E.; Heredia, M.; Lajo, C.; Goncalves, L.; Gomez-Herreras, J.I.; de Lejarazu, R.O.; Bermejo-Martin, J.F. Pro- and anti-inflammatory responses are regulated simultaneously from the first moments of septic shock. Eur. Cytokine Netw. 2011, 22, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Andaluz-Ojeda, D.; Bobillo, F.; Iglesias, V.; Almansa, R.; Rico, L.; Gandia, F.; Resino, S.; Tamayo, E.; de Lejarazu, R.O.; Bermejo-Martin, J.F. A combined score of pro- and anti-inflammatory interleukins improves mortality prediction in severe sepsis. Cytokine 2012, 57, 332–336. [Google Scholar] [CrossRef]
- Chaudhry, H.; Zhou, J.; Zhong, Y.; Ali, M.M.; McGuire, F.; Nagarkatti, P.S.; Nagarkatti, M. Role of cytokines as a double-edged sword in sepsis. In Vivo 2013, 27, 669–684. [Google Scholar]
- Tang, B.M.; Huang, S.J.; McLean, A.S. Genome-wide transcription profiling of human sepsis: A systematic review. Crit. Care 2010, 14, R237. [Google Scholar] [CrossRef] [PubMed]
- Drifte, G.; Dunn-Siegrist, I.; Tissieres, P.; Pugin, J. Innate immune functions of immature neutrophils in patients with sepsis and severe systemic inflammatory response syndrome. Crit. Care Med. 2013, 41, 820–832. [Google Scholar] [CrossRef]
- Nierhaus, A.; Linssen, J.; Wichmann, D.; Braune, S.; Kluge, S. Use of a weighted, automated analysis of the differential blood count to differentiate sepsis from non-infectious systemic inflammation: The intensive care infection score (ICIS). Inflamm. Allergy Drug Targets 2012, 11, 109–115. [Google Scholar] [CrossRef]
- Nierhaus, A.; Klatte, S.; Linssen, J.; Eismann, N.M.; Wichmann, D.; Hedke, J.; Braune, S.A.; Kluge, S. Revisiting the white blood cell count: Immature granulocytes count as a diagnostic marker to discriminate between SIRS and sepsis—A prospective, observational study. BMC Immunol. 2013, 14, 8. [Google Scholar] [CrossRef]
- Zucoloto, A.Z.; Jenne, C.N. Platelet-Neutrophil Interplay: Insights Into Neutrophil Extracellular Trap (NET)-Driven Coagulation in Infection. Front. Cardiovasc. Med. 2019, 6, 85. [Google Scholar] [CrossRef] [PubMed]
- Cox, L.E.; Walstein, K.; Vollger, L.; Reuner, F.; Bick, A.; Dotsch, A.; Engler, A.; Peters, J.; von Kockritz-Blickwede, M.; Schafer, S.T. Neutrophil extracellular trap formation and nuclease activity in septic patients. BMC Anesthesiol. 2020, 20, 15. [Google Scholar] [CrossRef] [PubMed]
- Ortmann, W.; Kolaczkowska, E. Age is the work of art? Impact of neutrophil and organism age on neutrophil extracellular trap formation. Cell Tissue Res. 2018, 371, 473–488. [Google Scholar] [CrossRef] [PubMed]
- Camicia, G.; Pozner, R.; de Larranaga, G. Neutrophil extracellular traps in sepsis. Shock 2014, 42, 286–294. [Google Scholar] [CrossRef]
- Kaplan, M.J.; Radic, M. Neutrophil extracellular traps: Double-edged swords of innate immunity. J. Immunol. 2012, 189, 2689–2695. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Kobayashi, S.D.; Quinn, M.T.; Deleo, F.R. A NET Outcome. Front. Immunol. 2012, 3, 365. [Google Scholar] [CrossRef] [PubMed]
- Daix, T.; Guerin, E.; Tavernier, E.; Mercier, E.; Gissot, V.; Herault, O.; Mira, J.P.; Dumas, F.; Chapuis, N.; Guitton, C.; et al. Multicentric Standardized Flow Cytometry Routine Assessment of Patients With Sepsis to Predict Clinical Worsening. Chest 2018, 154, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Keshari, R.S.; Jyoti, A.; Dubey, M.; Kothari, N.; Kohli, M.; Bogra, J.; Barthwal, M.K.; Dikshit, M. Cytokines induced neutrophil extracellular traps formation: Implication for the inflammatory disease condition. PLoS ONE 2012, 7, e48111. [Google Scholar] [CrossRef] [PubMed]
- Czaikoski, P.G.; Mota, J.M.; Nascimento, D.C.; Sonego, F.; Castanheira, F.V.; Melo, P.H.; Scortegagna, G.T.; Silva, R.L.; Barroso-Sousa, R.; Souto, F.O.; et al. Neutrophil Extracellular Traps Induce Organ Damage during Experimental and Clinical Sepsis. PLoS ONE 2016, 11, e0148142. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Sarmiento, J.; Salazar-Pelaez, L.M.; Carcillo, J.A. The Endothelial Glycocalyx: A Fundamental Determinant of Vascular Permeability in Sepsis. Pediatr. Crit. Care Med. 2020, 21, e291–e300. [Google Scholar] [CrossRef]
- Milusev, A.; Rieben, R.; Sorvillo, N. The Endothelial Glycocalyx: A Possible Therapeutic Target in Cardiovascular Disorders. Front. Cardiovasc. Med. 2022, 9, 897087. [Google Scholar] [CrossRef]
- Swystun, L.L.; Liaw, P.C. The role of leukocytes in thrombosis. Blood 2016, 128, 753–762. [Google Scholar] [CrossRef]
- Ince, C.; Mayeux, P.R.; Nguyen, T.; Gomez, H.; Kellum, J.A.; Ospina-Tascon, G.A.; Hernandez, G.; Murray, P.; De Backer, D.; Workgroup, A.X. The Endothelium in Sepsis. Shock 2016, 45, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Grover, S.P.; Mackman, N. Tissue Factor: An Essential Mediator of Hemostasis and Trigger of Thrombosis. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 709–725. [Google Scholar] [CrossRef] [PubMed]
- Denk, S.; Taylor, R.P.; Wiegner, R.; Cook, E.M.; Lindorfer, M.A.; Pfeiffer, K.; Paschke, S.; Eiseler, T.; Weiss, M.; Barth, E.; et al. Complement C5a-Induced Changes in Neutrophil Morphology During Inflammation. Scand. J. Immunol. 2017, 86, 143–155. [Google Scholar] [CrossRef]
- Ricklin, D.; Hajishengallis, G.; Yang, K.; Lambris, J.D. Complement: A key system for immune surveillance and homeostasis. Nat. Immunol. 2010, 11, 785–797. [Google Scholar] [CrossRef]
- Engelmann, B.; Massberg, S. Thrombosis as an intravascular effector of innate immunity. Nat. Reviews. Immunol. 2013, 13, 34–45. [Google Scholar] [CrossRef]
- Delvaeye, M.; Conway, E.M. Coagulation and innate immune responses: Can we view them separately? Blood 2009, 114, 2367–2374. [Google Scholar] [CrossRef]
- Oehmcke-Hecht, S.; Kohler, J. Interaction of the Human Contact System with Pathogens-An Update. Front. Immunol. 2018, 9, 312. [Google Scholar] [CrossRef]
- Taylor, F.B., Jr.; Toh, C.H.; Hoots, W.K.; Wada, H.; Levi, M.; Scientific Subcommittee on Disseminated Intravascular Coagulation of the International Society on Thrombosis and Haemostasis. Towards definition, clinical and laboratory criteria, and a scoring system for disseminated intravascular coagulation. Thromb. Haemost. 2001, 86, 1327–1330. [Google Scholar] [CrossRef] [PubMed]
- Iba, T.; Levi, M.; Levy, J.H. Sepsis-Induced Coagulopathy and Disseminated Intravascular Coagulation. Semin. Thromb. Hemost. 2020, 46, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Ankawi, G.; Neri, M.; Zhang, J.; Breglia, A.; Ricci, Z.; Ronco, C. Extracorporeal techniques for the treatment of critically ill patients with sepsis beyond conventional blood purification therapy: The promises and the pitfalls. Crit. Care 2018, 22, 262. [Google Scholar] [CrossRef] [PubMed]
- Yin, F.; Zhang, F.; Liu, S.; Ning, B. The therapeutic effect of high-volume hemofiltration on sepsis: A systematic review and meta-analysis. Ann. Transl. Med. 2020, 8, 488. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, C.; Mao, Z.; Qi, S.; Song, R.; Zhou, F. Effectiveness of polymyxin B-immobilized hemoperfusion against sepsis and septic shock: A systematic review and meta-analysis. J. Crit. Care 2021, 63, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Lipcsey, M.; Tenhunen, J.; Pischke, S.E.; Kuitunen, A.; Flaatten, H.; De Geer, L.; Sjolin, J.; Frithiof, R.; Chew, M.S.; Bendel, S.; et al. Endotoxin Removal in Septic Shock with the Alteco LPS Adsorber Was Safe But Showed no Benefit Compared to Placebo in the Double-Blind Randomized Controlled Trial-the Asset Study. Shock 2020, 54, 224–231. [Google Scholar] [CrossRef]
- Becker, S.; Lang, H.; Vollmer Barbosa, C.; Tian, Z.; Melk, A.; Schmidt, B.M.W. Efficacy of CytoSorb(R): A systematic review and meta-analysis. Crit. Care 2023, 27, 215. [Google Scholar] [CrossRef]
- Wang, G.; He, Y.; Guo, Q.; Zhao, Y.; He, J.; Chen, Y.; Chen, W.; Zhou, Y.; Peng, Z.; Deng, K.; et al. Continuous renal replacement therapy with the adsorptive oXiris filter may be associated with the lower 28-day mortality in sepsis: A systematic review and meta-analysis. Crit. Care 2023, 27, 275. [Google Scholar] [CrossRef]
- Li, Y.; Li, H.; Guo, J.; Wang, Y.; Zhang, D. Coupled plasma filtration adsorption for the treatment of sepsis or septic shock: A systematic review and meta-analysis. BMC Infect. Dis. 2022, 22, 714. [Google Scholar] [CrossRef] [PubMed]
- Kuklin, V.; Sovershaev, M.; Bjerner, J.; Keith, P.; Scott, L.K.; Thomas, O.M.T.; Szpirt, W.; Rock, G.; Stegmayr, B. Influence of therapeutic plasma exchange treatment on short-term mortality of critically ill adult patients with sepsis-induced organ dysfunction: A systematic review and meta-analysis. Crit. Care 2024, 28, 12. [Google Scholar] [CrossRef]
- Morgera, S.; Slowinski, T.; Melzer, C.; Sobottke, V.; Vargas-Hein, O.; Volk, T.; Zuckermann-Becker, H.; Wegner, B.; Muller, J.M.; Baumann, G.; et al. Renal replacement therapy with high-cutoff hemofilters: Impact of convection and diffusion on cytokine clearances and protein status. Am. J. Kidney Dis. 2004, 43, 444–453. [Google Scholar] [CrossRef]
- Atari, R.; Peck, L.; Visvanathan, K.; Skinner, N.; Eastwood, G.; Bellomo, R.; Storr, M.; Goehl, H. High cut-off hemofiltration versus standard hemofiltration: Effect on plasma cytokines. Int. J. Artif. Organs 2016, 39, 479–486. [Google Scholar] [CrossRef]
- Honore, P.M.; Jacobs, R.; Boer, W.; Joannes-Boyau, O.; De Regt, J.; De Waele, E.; Van Gorp, V.; Collin, V.; Spapen, H.D. New insights regarding rationale, therapeutic target and dose of hemofiltration and hybrid therapies in septic acute kidney injury. Blood Purif. 2012, 33, 44–51. [Google Scholar] [CrossRef]
- Villa, G.; Neri, M.; Bellomo, R.; Cerda, J.; De Gaudio, A.R.; De Rosa, S.; Garzotto, F.; Honore, P.M.; Kellum, J.; Lorenzin, A.; et al. Nomenclature for renal replacement therapy and blood purification techniques in critically ill patients: Practical applications. Crit. Care 2016, 20, 283. [Google Scholar] [CrossRef] [PubMed]
- Joannes-Boyau, O.; Honore, P.M.; Perez, P.; Bagshaw, S.M.; Grand, H.; Canivet, J.L.; Dewitte, A.; Flamens, C.; Pujol, W.; Grandoulier, A.S.; et al. High-volume versus standard-volume haemofiltration for septic shock patients with acute kidney injury (IVOIRE study): A multicentre randomized controlled trial. Intensive Care Med. 2013, 39, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Network, V.N.A.R.F.T.; Palevsky, P.M.; Zhang, J.H.; O’Connor, T.Z.; Chertow, G.M.; Crowley, S.T.; Choudhury, D.; Finkel, K.; Kellum, J.A.; Paganini, E.; et al. Intensity of renal support in critically ill patients with acute kidney injury. N. Engl. J. Med. 2008, 359, 7–20. [Google Scholar] [CrossRef]
- Investigators, R.R.T.S.; Bellomo, R.; Cass, A.; Cole, L.; Finfer, S.; Gallagher, M.; Lo, S.; McArthur, C.; McGuinness, S.; Myburgh, J.; et al. Intensity of continuous renal-replacement therapy in critically ill patients. N. Engl. J. Med. 2009, 361, 1627–1638. [Google Scholar] [CrossRef]
- Zhang, P.; Yang, Y.; Lv, R.; Zhang, Y.; Xie, W.; Chen, J. Effect of the intensity of continuous renal replacement therapy in patients with sepsis and acute kidney injury: A single-center randomized clinical trial. Nephrol. Dial. Transplant. 2012, 27, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Junhai, Z.; Beibei, C.; Jing, Y.; Li, L. Effect of High-Volume Hemofiltration in Critically Ill Patients: A Systematic Review and Meta-Analysis. Med. Sci. Monit. 2019, 25, 3964–3975. [Google Scholar] [CrossRef] [PubMed]
- Yaroustovsky, M.; Plyushch, M.; Popov, D.; Samsonova, N.; Abramyan, M.; Popok, Z.; Krotenko, N. Prognostic value of endotoxin activity assay in patients with severe sepsis after cardiac surgery. J. Inflamm. 2013, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Kamohara, H.; Suda, S.; Nagura, T.; Tomino, M.; Sugi, M.; Wajima, Z. Comparative Evaluation of Endotoxin Activity Level and Various Biomarkers for Infection and Outcome of ICU-Admitted Patients. Biomedicines 2019, 7, 47. [Google Scholar] [CrossRef]
- Vincent, J.L.; Laterre, P.F.; Cohen, J.; Burchardi, H.; Bruining, H.; Lerma, F.A.; Wittebole, X.; De Backer, D.; Brett, S.; Marzo, D.; et al. A pilot-controlled study of a polymyxin B-immobilized hemoperfusion cartridge in patients with severe sepsis secondary to intra-abdominal infection. Shock 2005, 23, 400–405. [Google Scholar] [CrossRef]
- Cruz, D.N.; Antonelli, M.; Fumagalli, R.; Foltran, F.; Brienza, N.; Donati, A.; Malcangi, V.; Petrini, F.; Volta, G.; Bobbio Pallavicini, F.M.; et al. Early use of polymyxin B hemoperfusion in abdominal septic shock: The EUPHAS randomized controlled trial. JAMA 2009, 301, 2445–2452. [Google Scholar] [CrossRef]
- Payen, D.M.; Guilhot, J.; Launey, Y.; Lukaszewicz, A.C.; Kaaki, M.; Veber, B.; Pottecher, J.; Joannes-Boyau, O.; Martin-Lefevre, L.; Jabaudon, M.; et al. Early use of polymyxin B hemoperfusion in patients with septic shock due to peritonitis: A multicenter randomized control trial. Intensive Care Med. 2015, 41, 975–984. [Google Scholar] [CrossRef]
- Klein, D.J.; Foster, D.; Schorr, C.A.; Kazempour, K.; Walker, P.M.; Dellinger, R.P. The EUPHRATES trial (Evaluating the Use of Polymyxin B Hemoperfusion in a Randomized controlled trial of Adults Treated for Endotoxemia and Septic shock): Study protocol for a randomized controlled trial. Trials 2014, 15, 218. [Google Scholar] [CrossRef]
- Pinciroli, R.; Checchi, S.; Bottiroli, M.; Monti, G.; Casella, G.; Fumagalli, R. Endotoxin Activity Assay for the Detection of Whole Blood Endotoxemia in Critically Ill Patients. J. Vis. Exp. 2019. [Google Scholar] [CrossRef]
- Romaschin, A.D.; Klein, D.J.; Marshall, J.C. Bench-to-bedside review: Clinical experience with the endotoxin activity assay. Crit. Care 2012, 16, 248. [Google Scholar] [CrossRef]
- Dellinger, R.P.; Bagshaw, S.M.; Antonelli, M.; Foster, D.M.; Klein, D.J.; Marshall, J.C.; Palevsky, P.M.; Weisberg, L.S.; Schorr, C.A.; Trzeciak, S.; et al. Effect of Targeted Polymyxin B Hemoperfusion on 28-Day Mortality in Patients With Septic Shock and Elevated Endotoxin Level: The EUPHRATES Randomized Clinical Trial. JAMA 2018, 320, 1455–1463. [Google Scholar] [CrossRef]
- Klein, D.J.; Foster, D.; Walker, P.M.; Bagshaw, S.M.; Mekonnen, H.; Antonelli, M. Polymyxin B hemoperfusion in endotoxemic septic shock patients without extreme endotoxemia: A post hoc analysis of the EUPHRATES trial. Intensive Care Med. 2018, 44, 2205–2212. [Google Scholar] [CrossRef]
- Shoji, H.; Opal, S.M. Therapeutic Rationale for Endotoxin Removal with Polymyxin B Immobilized Fiber Column (PMX) for Septic Shock. Int. J. Mol. Sci. 2021, 22, 2228. [Google Scholar] [CrossRef]
- Schädler, D.; Porzelius, C.; Jörres, A.; Marx, G.; Meier-Hellmann, A.; Putensen, C.; Quintel, M.; Spies, C.; Engel, C.; Weiler, N.; et al. A multicenter randomized controlled study of an extracorporeal cytokine hemoadsorption device in septic patients. Crit. Care 2013, 17, P62. [Google Scholar] [CrossRef]
- Kogelmann, K.; Jarczak, D.; Scheller, M.; Druner, M. Hemoadsorption by CytoSorb in septic patients: A case series. Crit. Care 2017, 21, 74. [Google Scholar] [CrossRef]
- Friesecke, S.; Stecher, S.S.; Gross, S.; Felix, S.B.; Nierhaus, A. Extracorporeal cytokine elimination as rescue therapy in refractory septic shock: A prospective single-center study. J. Artif. Organs 2017, 20, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Hawchar, F.; Laszlo, I.; Oveges, N.; Trasy, D.; Ondrik, Z.; Molnar, Z. Extracorporeal cytokine adsorption in septic shock: A proof of concept randomized, controlled pilot study. J. Crit. Care 2019, 49, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Schädler, D.; Pausch, C.; Heise, D.; Meier-Hellmann, A.; Brederlau, J.; Weiler, N.; Marx, G.; Putensen, C.; Spies, C.; Jörres, A.; et al. The effect of a novel extracorporeal cytokine hemoadsorption device on IL-6 elimination in septic patients: A randomized controlled trial. PLoS ONE 2017, 12, e0187015. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, W.P.; Duran, S.; Kuijper, M.; Ince, C. Hemoadsorption with CytoSorb shows a decreased observed versus expected 28-day all-cause mortality in ICU patients with septic shock: A propensity-score-weighted retrospective study. Crit. Care 2019, 23, 317. [Google Scholar] [CrossRef]
- Kogelmann, K.; Hubner, T.; Schwameis, F.; Druner, M.; Scheller, M.; Jarczak, D. First Evaluation of a New Dynamic Scoring System Intended to Support Prescription of Adjuvant CytoSorb Hemoadsorption Therapy in Patients with Septic Shock. J. Clin. Med. 2021, 10, 2939. [Google Scholar] [CrossRef]
- David, S.; Stahl, K. To remove and replace-a role for plasma exchange in counterbalancing the host response in sepsis. Crit. Care 2019, 23, 14. [Google Scholar] [CrossRef]
- Bockmeyer, C.L.; Claus, R.A.; Budde, U.; Kentouche, K.; Schneppenheim, R.; Losche, W.; Reinhart, K.; Brunkhorst, F.M. Inflammation-associated ADAMTS13 deficiency promotes formation of ultra-large von Willebrand factor. Haematologica 2008, 93, 137–140. [Google Scholar] [CrossRef]
- Azfar, M.F.; Khan, M.F.; Habib, S.S.; Aseri, Z.A.; Zubaidi, A.M.; Aguila, D.O.; Suriya, M.O.; Ullah, H. Prognostic value of ADAMTS13 in patients with severe sepsis and septic shock. Clin. Investig. Med. 2017, 40, E49–E58. [Google Scholar] [CrossRef] [PubMed]
- Rimmer, E.; Houston, B.L.; Kumar, A.; Abou-Setta, A.M.; Friesen, C.; Marshall, J.C.; Rock, G.; Turgeon, A.F.; Cook, D.J.; Houston, D.S.; et al. The efficacy and safety of plasma exchange in patients with sepsis and septic shock: A systematic review and meta-analysis. Crit. Care 2014, 18, 699. [Google Scholar] [CrossRef] [PubMed]
- Knaup, H.; Stahl, K.; Schmidt, B.M.W.; Idowu, T.O.; Busch, M.; Wiesner, O.; Welte, T.; Haller, H.; Kielstein, J.T.; Hoeper, M.M.; et al. Early therapeutic plasma exchange in septic shock: A prospective open-label nonrandomized pilot study focusing on safety, hemodynamics, vascular barrier function, and biologic markers. Crit. Care 2018, 22, 285. [Google Scholar] [CrossRef] [PubMed]
- Stahl, K.; Wand, P.; Seeliger, B.; Wendel-Garcia, P.D.; Schmidt, J.J.; Schmidt, B.M.W.; Sauer, A.; Lehmann, F.; Budde, U.; Busch, M.; et al. Clinical and biochemical endpoints and predictors of response to plasma exchange in septic shock: Results from a randomized controlled trial. Crit. Care 2022, 26, 134. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, X.Y.; Guo, S.Y.; Jiang, J.; Wang, G.; Weng, Y.B. An inquiry into the treatment of sepsis using plasma exchange therapy: A systematic review and meta-analysis. Int. Wound J. 2023, 20, 1979–1986. [Google Scholar] [CrossRef]
- Broman, M.E.; Hansson, F.; Vincent, J.L.; Bodelsson, M. Endotoxin and cytokine reducing properties of the oXiris membrane in patients with septic shock: A randomized crossover double-blind study. PLoS ONE 2019, 14, e0220444. [Google Scholar] [CrossRef] [PubMed]
- Malard, B.; Lambert, C.; Kellum, J.A. In vitro comparison of the adsorption of inflammatory mediators by blood purification devices. Intensive Care Med. Exp. 2018, 6, 12. [Google Scholar] [CrossRef] [PubMed]
- Monard, C.; Rimmele, T.; Ronco, C. Extracorporeal Blood Purification Therapies for Sepsis. Blood Purif. 2019, 47 (Suppl. S3), 1–14. [Google Scholar] [CrossRef]
- Schwindenhammer, V.; Girardot, T.; Chaulier, K.; Gregoire, A.; Monard, C.; Huriaux, L.; Illinger, J.; Leray, V.; Uberti, T.; Crozon-Clauzel, J.; et al. oXiris(R) Use in Septic Shock: Experience of Two French Centres. Blood Purif. 2019, 47 (Suppl. S3), 1–7. [Google Scholar] [CrossRef]
- Tetta, C.; Cavaillon, J.M.; Schulze, M.; Ronco, C.; Ghezzi, P.M.; Camussi, G.; Serra, A.M.; Curti, F.; Lonnemann, G. Removal of cytokines and activated complement components in an experimental model of continuous plasma filtration coupled with sorbent adsorption. Nephrol. Dial. Transplant. 1998, 13, 1458–1464. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garbero, E.; Livigni, S.; Ferrari, F.; Finazzi, S.; Langer, M.; Malacarne, P.; Meca, M.C.C.; Mosca, S.; Olivieri, C.; Pozzato, M.; et al. High dose coupled plasma filtration and adsorption in septic shock patients. Results of the COMPACT-2: A multicentre, adaptive, randomised clinical trial. Intensive Care Med. 2021, 47, 1303–1311. [Google Scholar] [CrossRef]
- Gimenez-Esparza, C.; Portillo-Requena, C.; Colomina-Climent, F.; Allegue-Gallego, J.M.; Galindo-Martinez, M.; Molla-Jimenez, C.; Anton-Pascual, J.L.; Marmol-Peis, E.; Dolera-Moreno, C.; Rodriguez-Serra, M.; et al. The premature closure of ROMPA clinical trial: Mortality reduction in septic shock by plasma adsorption. BMJ Open 2019, 9, e030139. [Google Scholar] [CrossRef]
- Alcaraz-Quiles, J.; Casulleras, M.; Oettl, K.; Titos, E.; Flores-Costa, R.; Duran-Guell, M.; Lopez-Vicario, C.; Pavesi, M.; Stauber, R.E.; Arroyo, V.; et al. Oxidized Albumin Triggers a Cytokine Storm in Leukocytes Through P38 Mitogen-Activated Protein Kinase: Role in Systemic Inflammation in Decompensated Cirrhosis. Hepatology 2018, 68, 1937–1952. [Google Scholar] [CrossRef] [PubMed]
- Casulleras, M.; Flores-Costa, R.; Duran-Guell, M.; Alcaraz-Quiles, J.; Sanz, S.; Titos, E.; Lopez-Vicario, C.; Fernandez, J.; Horrillo, R.; Costa, M.; et al. Albumin internalizes and inhibits endosomal TLR signaling in leukocytes from patients with decompensated cirrhosis. Sci. Transl. Med. 2020, 12, eaax5135. [Google Scholar] [CrossRef]
- Schmuck, R.B.; Nawrot, G.H.; Fikatas, P.; Reutzel-Selke, A.; Pratschke, J.; Sauer, I.M. Single Pass Albumin Dialysis-A Dose-Finding Study to Define Optimal Albumin Concentration and Dialysate Flow. Artif. Organs 2017, 41, 153–161. [Google Scholar] [CrossRef]
- Stange, J.; Mitzner, S.R.; Risler, T.; Erley, C.M.; Lauchart, W.; Goehl, H.; Klammt, S.; Peszynski, P.; Freytag, J.; Hickstein, H.; et al. Molecular adsorbent recycling system (MARS): Clinical results of a new membrane-based blood purification system for bioartificial liver support. Artif. Organs 1999, 23, 319–330. [Google Scholar] [CrossRef]
- Rifai, K.; Ernst, T.; Kretschmer, U.; Bahr, M.J.; Schneider, A.; Hafer, C.; Haller, H.; Manns, M.P.; Fliser, D. Prometheus--a new extracorporeal system for the treatment of liver failure. J. Hepatol. 2003, 39, 984–990. [Google Scholar] [CrossRef] [PubMed]
- Huber, W.; Henschel, B.; Schmid, R.; Al-Chalabi, A. First clinical experience in 14 patients treated with ADVOS: A study on feasibility, safety and efficacy of a new type of albumin dialysis. BMC Gastroenterol. 2017, 17, 32. [Google Scholar] [CrossRef] [PubMed]
- Alshamsi, F.; Alshammari, K.; Belley-Cote, E.; Dionne, J.; Albrahim, T.; Albudoor, B.; Ismail, M.; Al-Judaibi, B.; Baw, B.; Subramanian, R.M.; et al. Extracorporeal liver support in patients with liver failure: A systematic review and meta-analysis of randomized trials. Intensive Care Med. 2020, 46, 1–16. [Google Scholar] [CrossRef] [PubMed]

| Method | Number and Type of Trials | Number of Patients (n) | Patient Status | Results | Reference |
|---|---|---|---|---|---|
| HCO | 4 RCT 3 OS | 215 | Sepsis or septic shock | No significant differences in hospital mortality or length of ICU stay. One trial was stopped prematurely for futility after enrolment of 81 patients. | [38] |
| HVHF Ultrafiltrate rate in intervention group >35 mL/kg/h | 5 RCT | 241 | Sepsis | Available evidence does not support effectiveness in terms of survival. HVHF may be effective in improving individual morbidity. | [39] |
| PMX-HP | 13 RCT | 1163 | Sepsis or septic shock | Therapy with PMX-HP may reduce mortality, compared to standard of care. Patients with less severe sepsis may benefit more. | [40] |
| LPS Adsorber | 1 RCT | 8 | Sepsis or septic shock | Terminated prematurely due to recruitment problems. | [41] |
| CytoSorb | 2 RCT 6 CS | 776 | Sepsis or septic shock | No significant mortality reduction. May be effective in improving ICU morbidity. | [42] |
| oXiris | 4 RCT 10 CS | 695 | Septic patients undergoing CRRT | Potential association with lower 28-day mortality, decreased norepinephrine dose and shorter ICU stay, no 90-day mortality benefit. | [43] |
| CPFA | 4 RCT 2 CS | 537 | Sepsis or septic shock | No all-cause mortality benefit. | [44] |
| Plasma exchange | 5 RCT 6 CS | 627 | Critically ill patients with sepsis-induced multiorgan dysfunction | Potential survival benefit compared to standard of care. | [45] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jarczak, D.; Kluge, S.; Nierhaus, A. Septic Hyperinflammation—Is There a Role for Extracorporeal Blood Purification Techniques? Int. J. Mol. Sci. 2024, 25, 3120. https://doi.org/10.3390/ijms25063120
Jarczak D, Kluge S, Nierhaus A. Septic Hyperinflammation—Is There a Role for Extracorporeal Blood Purification Techniques? International Journal of Molecular Sciences. 2024; 25(6):3120. https://doi.org/10.3390/ijms25063120
Chicago/Turabian StyleJarczak, Dominik, Stefan Kluge, and Axel Nierhaus. 2024. "Septic Hyperinflammation—Is There a Role for Extracorporeal Blood Purification Techniques?" International Journal of Molecular Sciences 25, no. 6: 3120. https://doi.org/10.3390/ijms25063120
APA StyleJarczak, D., Kluge, S., & Nierhaus, A. (2024). Septic Hyperinflammation—Is There a Role for Extracorporeal Blood Purification Techniques? International Journal of Molecular Sciences, 25(6), 3120. https://doi.org/10.3390/ijms25063120





