Influence of N-Acetylglucosamine and Melatonin Interaction in Modeling the Photosynthetic Component and Metabolomics of Cucumber under Salinity Stress
Abstract
1. Introduction
2. Results
2.1. Evaluation of Morphological Attributes of Cucumber Plants
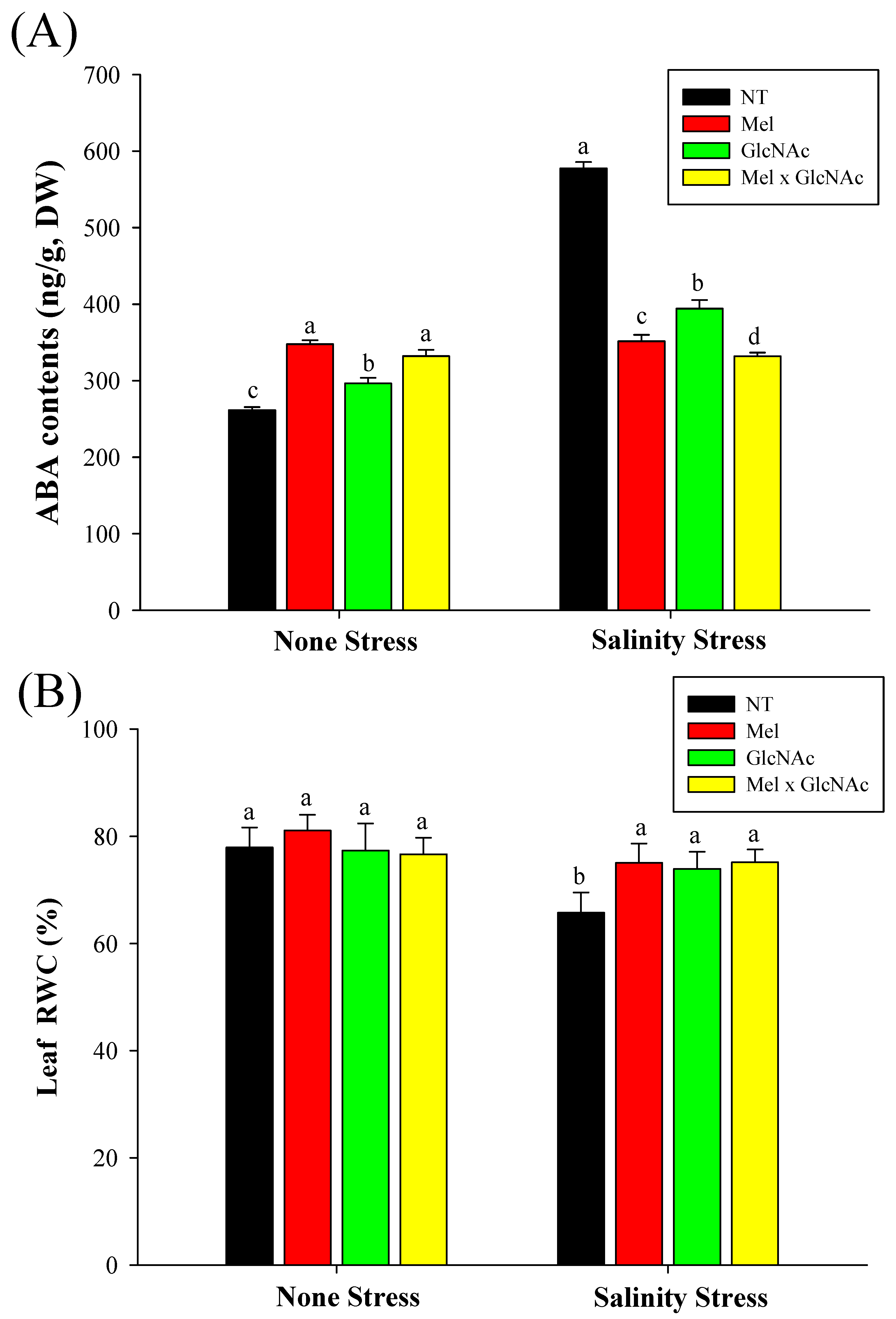
2.2. Extraction and Quantification of Abscisic Acid and Measurement of Leaf Water Content
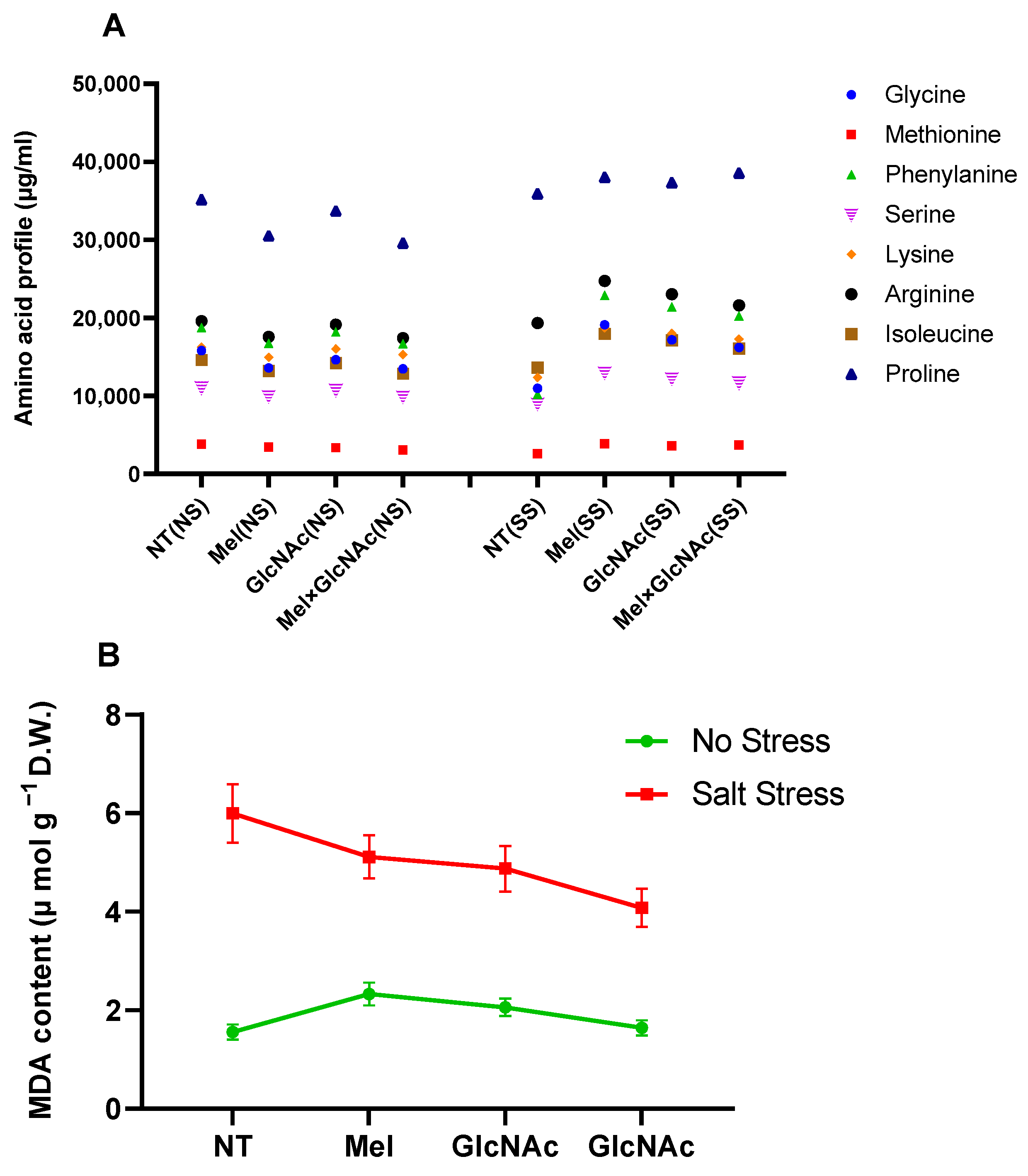
2.3. Quantification of Amino Acids and MDA Content in Cucumber Shoots
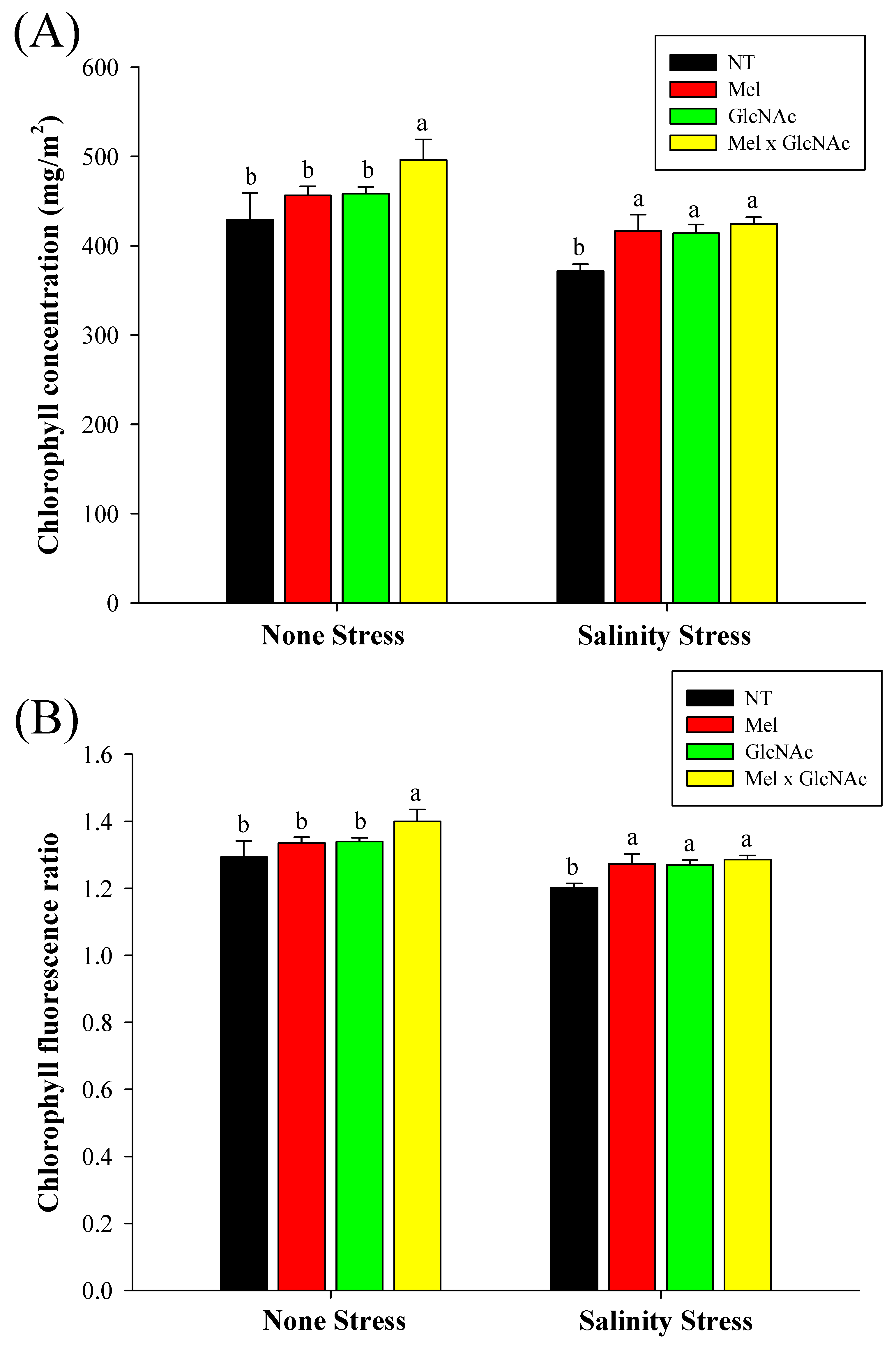
2.4. Determination of Chlorophyll, Chlorophyll Fluorescence Ratio, and Relative Water Content
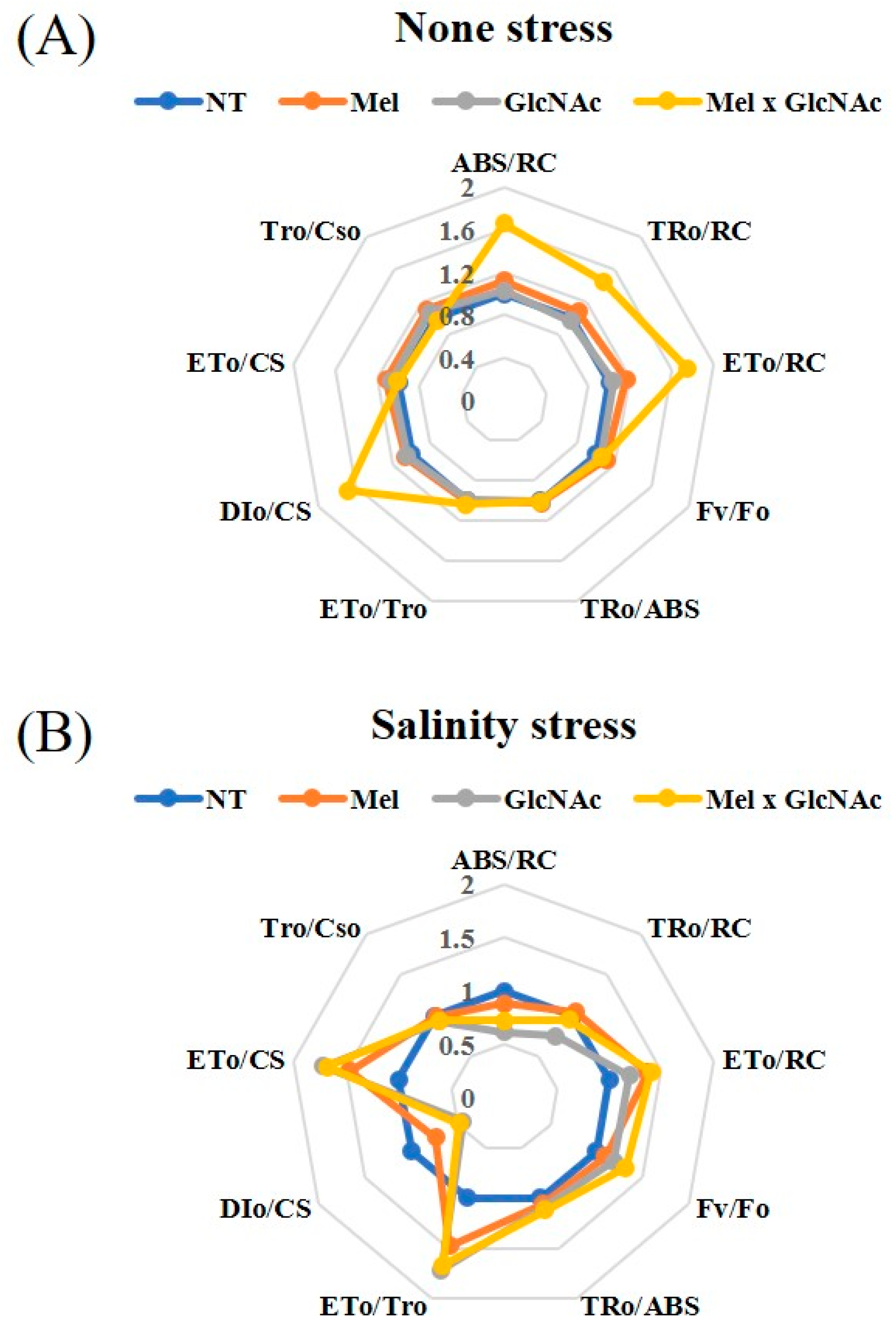
2.5. OJIP Curve and Test Parameters
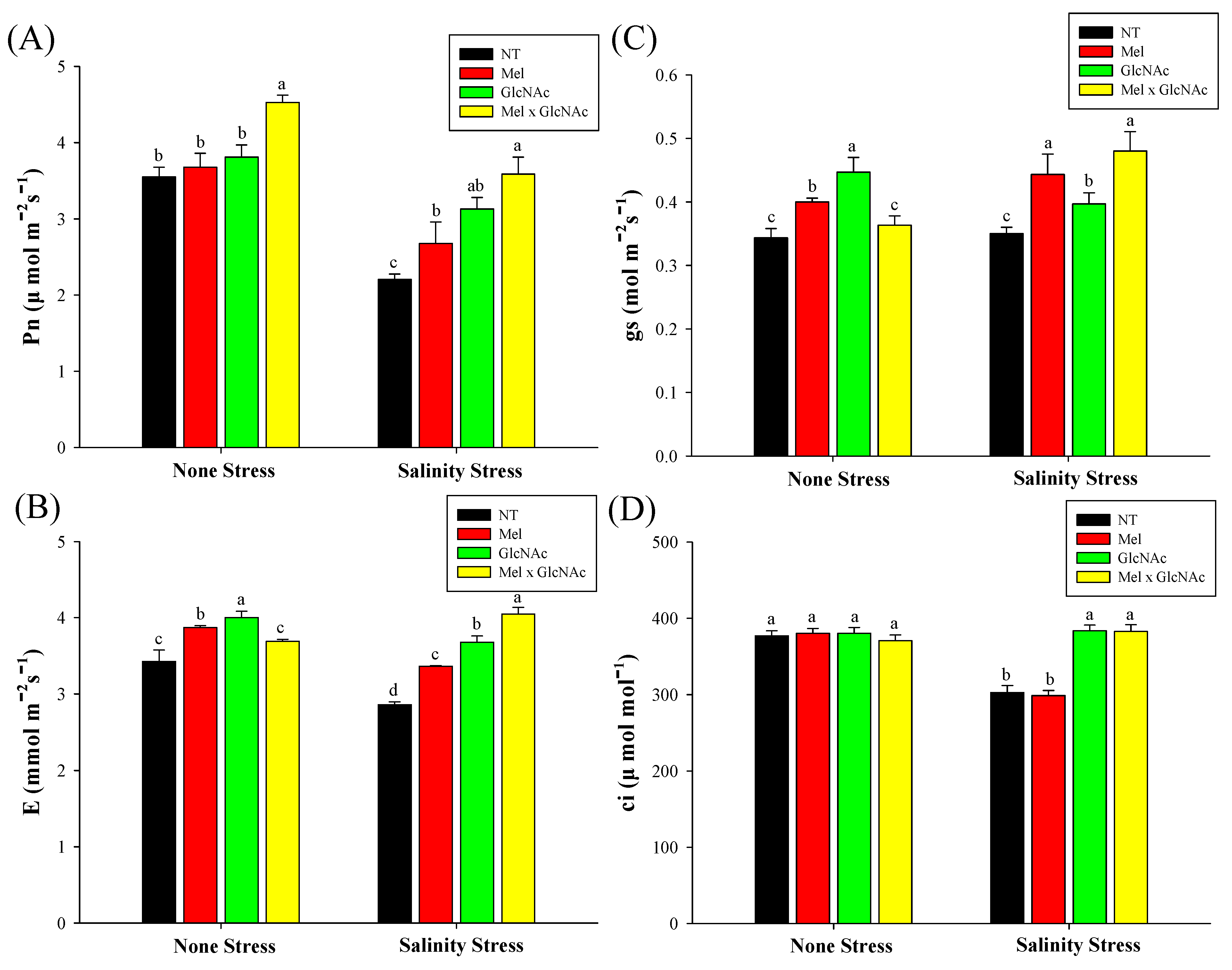
2.6. Measurement of Photosynthetic Rate (Pn), Stomatal Conductance (Gs), Transpiration Rate (E), and Substomatal CO2 Concentration
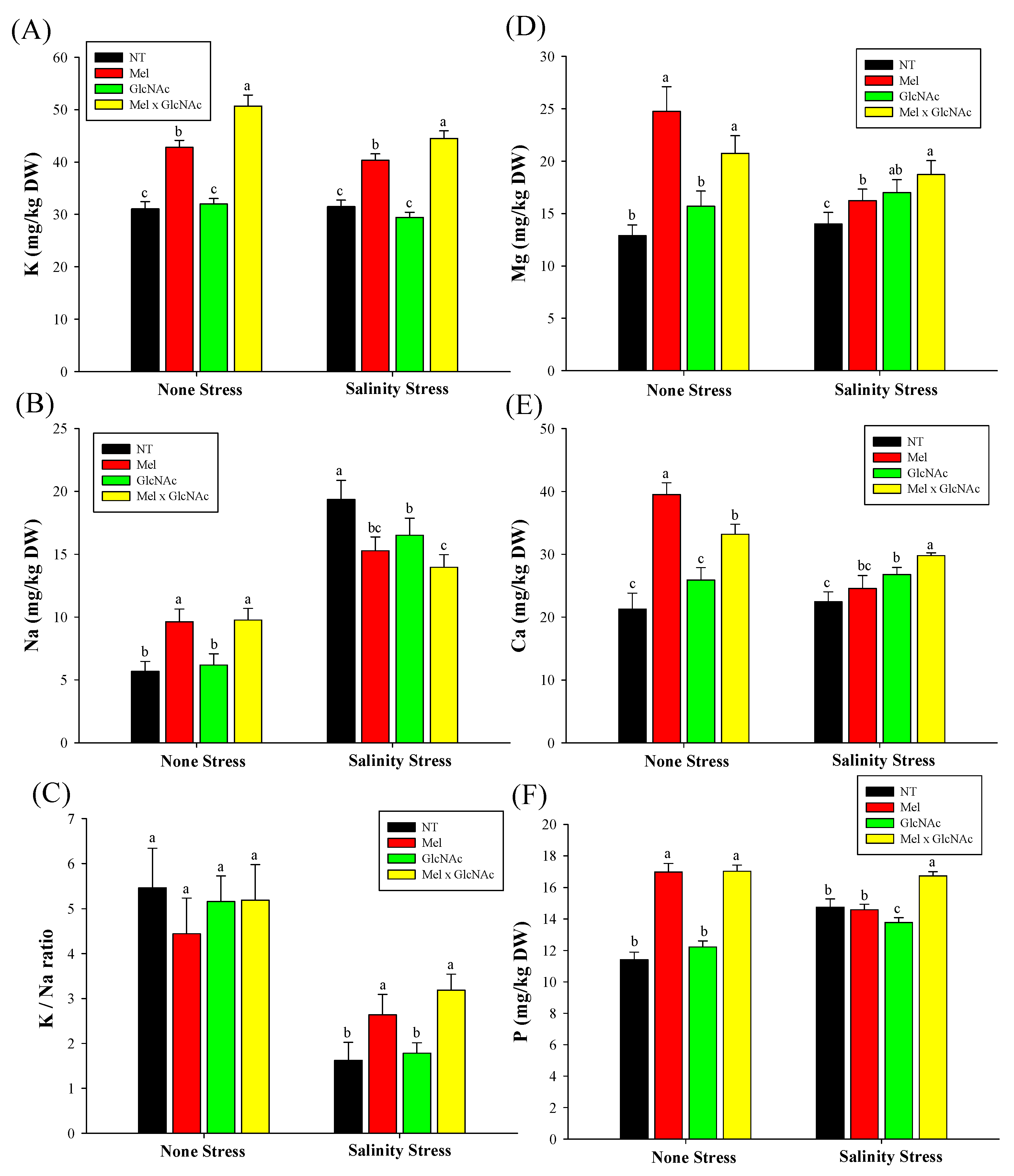
2.7. Quantification of K+, Mg+, Na+, Ca++ and P Content in Cucumber Shoots
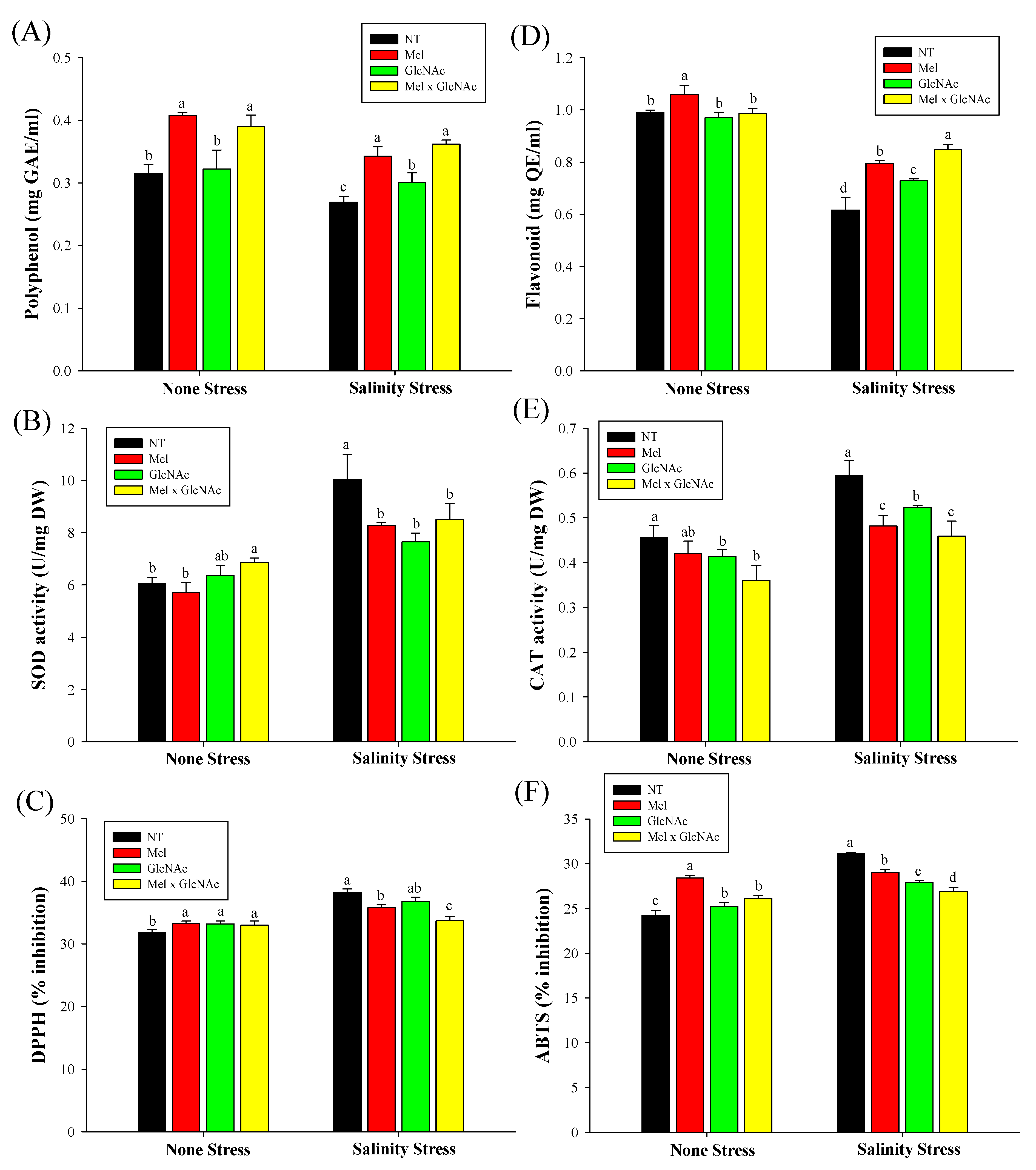
2.8. Analysis of Antioxidants and Their Related Activity
3. Discussion
4. Material and Methods
4.1. Plant Experiments
4.2. Measurement of Chlorophyll Fluorescence
4.3. Quantification of K/Mg/Na/Ca/P
4.4. Analysis of Antioxidant Activity and Enzymes
4.4.1. SOD and CAT Assay and Quantification of Total Flavonoids and Phenolic Content
4.4.2. Measurement of Radical Scavenging Activity (DPPH, and ABTS)
4.5. Extraction and Quantification of Abscisic Acid
4.6. Analysis of Amino Acids Profile in Cucumber Shoots
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, H.; Li, Y.; Zhu, J.-K. Developing naturally stress-resistant crops for a sustainable agriculture. Nat. Plants 2018, 4, 989–996. [Google Scholar] [CrossRef]
- Kumar, G.; Basu, S.; Singla-Pareek, S.L.; Pareek, A. Unraveling the contribution of OsSOS2 in conferring salinity and drought tolerance in a high-yielding rice. Physiol. Plant. 2022, 174, e13638. [Google Scholar] [CrossRef]
- Yadav, S.; Irfan, M.; Ahmad, A.; Hayat, S. Causes of salinity and plant manifestations to salt stress: A review. J. Environ. Biol. 2011, 32, 667. [Google Scholar]
- Van Zelm, E.; Zhang, Y.; Testerink, C. Salt tolerance mechanisms of plants. Annu. Rev. Plant Biol. 2020, 71, 403–433. [Google Scholar] [CrossRef]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef]
- Deinlein, U.; Stephan, A.B.; Horie, T.; Luo, W.; Xu, G.; Schroeder, J.I. Plant salt-tolerance mechanisms. Trends Plant Sci. 2014, 19, 371–379. [Google Scholar] [CrossRef]
- Zhu, J.-K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ruiz, J.; Arnao, M.B. Relationship of Melatonin and Salicylic Acid in Biotic/Abiotic Plant Stress Responses. Agronomy 2018, 8, 33. [Google Scholar] [CrossRef]
- Back, K.; Tan, D.X.; Reiter, R.J. Melatonin biosynthesis in plants: Multiple pathways catalyze tryptophan to melatonin in the cytoplasm or chloroplasts. J. Pineal Res. 2016, 61, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.E.; Mao, J.J.; Sun, L.Q.; Huang, B.; Ding, C.B.; Gu, Y.; Liao, J.Q.; Hu, C.; Zhang, Z.W.; Yuan, S. Exogenous melatonin enhances salt stress tolerance in maize seedlings by improving antioxidant and photosynthetic capacity. Physiol. Plant. 2018, 164, 349–363. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Shi, Z.; Zhang, X.; Zheng, S.; Wang, J.; Mo, J. Alleviating effects of exogenous melatonin on salt stress in cucumber. Sci. Hortic. 2020, 262, 109070. [Google Scholar] [CrossRef]
- Tan, D.-X.; Reiter, R.J. An evolutionary view of melatonin synthesis and metabolism related to its biological functions in plants. J. Exp. Bot. 2020, 71, 4677–4689. [Google Scholar] [CrossRef]
- Kumirska, J.; Czerwicka, M.; Kaczyński, Z.; Bychowska, A.; Brzozowski, K.; Thöming, J.; Stepnowski, P. Application of Spectroscopic Methods for Structural Analysis of Chitin and Chitosan. Mar. Drugs 2010, 8, 1567–1636. [Google Scholar] [CrossRef] [PubMed]
- Arakane, Y.; Baguinon, M.C.; Jasrapuria, S.; Chaudhari, S.; Doyungan, A.; Kramer, K.J.; Muthukrishnan, S.; Beeman, R.W. Both UDP N-acetylglucosamine pyrophosphorylases of Tribolium castaneum are critical for molting, survival and fecundity. Insect Biochem. Mol. Biol. 2011, 41, 42–50. [Google Scholar] [CrossRef]
- Yoo, J.Y.; Ko, K.S.; Vu, B.N.; Lee, Y.E.; Yoon, S.H.; Pham, T.T.; Kim, J.-Y.; Lim, J.-M.; Kang, Y.J.; Hong, J.C. N-acetylglucosaminyltransferase II is involved in plant growth and development under stress conditions. Front. Plant Sci. 2021, 12, 761064. [Google Scholar] [CrossRef]
- Ansari, S.; Kumar, V.; Bhatt, D.N.; Irfan, M.; Datta, A. N-Acetylglucosamine Sensing and Metabolic Engineering for Attenuating Human and Plant Pathogens. Bioengineering 2022, 9, 64. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Zhang, H.; Qin, H.; Li, K.; Liu, X.; Wang, W.; Ye, M.; Yin, H. Protein O-GlcNAcylation impairment caused by N-acetylglucosamine phosphate mutase deficiency leads to growth variations in Arabidopsis thaliana. Plant J. 2023, 114, 613–635. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, A.; Aneefi, A.G.; Sisuvanh, H.; Singkham, S.; Pius, M.V.; Akter, F.; Kwon, E.-H.; Kang, S.-M.; Woo, Y.-J.; Yun, B.-W.; et al. Dynamics of Humic Acid, Silicon, and Biochar under Heavy Metal, Drought, and Salinity with Special Reference to Phytohormones, Antioxidants, and Melatonin Synthesis in Rice. Int. J. Mol. Sci. 2023, 24, 17369. [Google Scholar] [CrossRef]
- Sharma, V.; Sharma, L.; Sandhu, K.S. Cucumber (Cucumis sativus L.). In Antioxidants in Vegetables and Nuts-Properties and Health Benefits; Springer: Berlin/Heidelberg, Germany, 2020; pp. 333–340. [Google Scholar]
- Campos, C.N.S.; Teixeira, G.C.M.; Prado, R.d.M.; Caione, G.; da Silva Júnior, G.B.; David, C.H.O.d.; Sales, A.C.; Roque, C.G.; Teodoro, P.E. Macronutrient deficiency in cucumber plants: Impacts in nutrition, growth and symptoms. J. Plant Nutr. 2021, 44, 2609–2626. [Google Scholar] [CrossRef]
- Strauss, A.J.; Krüger, G.H.J.; Strasser, R.J.; Van Heerden, P.D.R. Ranking of dark chilling tolerance in soybean genotypes probed by the chlorophyll a fluorescence transient OJIP. Environ. Exp. Bot. 2006, 56, 147–157. [Google Scholar] [CrossRef]
- Lima-Moro, A.; Bertoli, S.C.; Braga-Reis, I.; Moro, E.; Ziliani, R.R.; Spolaor, B.O.; de Freitas, Í.R.; dos Santos, B.L. Photosynthetic activity and OJIP fluorescence with the application of a nutritional solution. Acta Physiol. Plant. 2022, 44, 67. [Google Scholar] [CrossRef]
- Strasser, B.J. Measuring fast fluorescence transients to address environmental questions: The JIP test. In Photosynthesis: From Light to Biosphere; Springer: Berlin/Heidelberg, Germany, 1995; pp. 977–980. [Google Scholar]
- Cao, Y.; Pan, Y.; Wang, M.; Liu, T.; Meng, X.; Guo, S. The effects of different nitrogen forms on chlorophyll fluorescence and photosystem II in Lonicera japonica. J. Plant Growth Regul. 2023, 42, 4106–4117. [Google Scholar] [CrossRef]
- Luo, Q.; Yu, B.; Liu, Y. Differential sensitivity to chloride and sodium ions in seedlings of Glycine max and G. soja under NaCl stress. J. Plant Physiol. 2005, 162, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Newman, I.; Zhou, M.; Mendham, N.; Zhang, G.; Shabala, S. Screening plants for salt tolerance by measuring K+ flux: A case study for barley. Plant Cell Environ. 2005, 28, 1230–1246. [Google Scholar] [CrossRef]
- Larbi, A.; Kchaou, H.; Gaaliche, B.; Gargouri, K.; Boulal, H.; Morales, F. Supplementary potassium and calcium improves salt tolerance in olive plants. Sci. Hortic. 2020, 260, 108912. [Google Scholar] [CrossRef]
- Moustafa-Farag, M.; Almoneafy, A.; Mahmoud, A.; Elkelish, A.; Arnao, M.B.; Li, L.; Ai, S. Melatonin and Its Protective Role against Biotic Stress Impacts on Plants. Biomolecules 2020, 10, 54. [Google Scholar] [CrossRef]
- Kwon, E.-H.; Adhikari, A.; Imran, M.; Lee, D.-S.; Lee, C.-Y.; Kang, S.-M.; Lee, I.-J. Exogenous SA Applications Alleviate Salinity Stress via Physiological and Biochemical changes in St John’s Wort Plants. Plants 2023, 12, 310. [Google Scholar] [CrossRef]
- Postiglione, A.E.; Muday, G.K. The role of ROS homeostasis in ABA-induced guard cell signaling. Front. Plant Sci. 2020, 11, 968. [Google Scholar] [CrossRef]
- Zhang, H.J.; Zhang, N.A.; Yang, R.C.; Wang, L.; Sun, Q.Q.; Li, D.B.; Cao, Y.Y.; Weeda, S.; Zhao, B.; Ren, S. Melatonin promotes seed germination under high salinity by regulating antioxidant systems, ABA and GA4 interaction in cucumber (Cucumis sativus L.). J. Pineal Res. 2014, 57, 269–279. [Google Scholar] [CrossRef]
- Sarropoulou, V.; Dimassi-Theriou, K.; Therios, I.; Koukourikou-Petridou, M. Melatonin enhances root regeneration, photosynthetic pigments, biomass, total carbohydrates and proline content in the cherry rootstock PHL-C (Prunus avium × Prunus cerasus). Plant Physiol. Biochem. 2012, 61, 162–168. [Google Scholar] [CrossRef]
- Dawood, M.G.; El-Awadi, M.E. Alleviation of salinity stress on Vicia faba L. plants via seed priming with melatonin. Acta Biológica Colomb. 2015, 20, 223–235. [Google Scholar] [CrossRef]
- Han, Q.-H.; Huang, B.; Ding, C.-B.; Zhang, Z.-W.; Chen, Y.-E.; Hu, C.; Zhou, L.-J.; Huang, Y.; Liao, J.-Q.; Yuan, S. Effects of melatonin on anti-oxidative systems and photosystem II in cold-stressed rice seedlings. Front. Plant Sci. 2017, 8, 785. [Google Scholar] [CrossRef]
- Luis Castañares, J.; Alberto Bouzo, C. Effect of Exogenous Melatonin on Seed Germination and Seedling Growth in Melon (Cucumis melo L.) Under Salt Stress. Hortic. Plant J. 2019, 5, 79–87. [Google Scholar] [CrossRef]
- Lam, E.; Kato, N.; Lawton, M. Programmed cell death, mitochondria and the plant hypersensitive response. Nature 2001, 411, 848–853. [Google Scholar] [CrossRef]
- Mansoor, S.; Ali Wani, O.; Lone, J.K.; Manhas, S.; Kour, N.; Alam, P.; Ahmad, A.; Ahmad, P. Reactive Oxygen Species in Plants: From Source to Sink. Antioxidants 2022, 11, 225. [Google Scholar] [CrossRef]
- Del Río, L.A.; Corpas, F.J.; Sandalio, L.M.; Palma, J.M.; Gómez, M.; Barroso, J.B. Reactive oxygen species, antioxidant systems and nitric oxide in peroxisomes. J. Exp. Bot. 2002, 53, 1255–1272. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Dumanović, J.; Nepovimova, E.; Natić, M.; Kuča, K.; Jaćević, V. The significance of reactive oxygen species and antioxidant defense system in plants: A concise overview. Front. Plant Sci. 2021, 11, 552969. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, K.; Raina, M.; Sanfratello, G.M.; Pandey, P.; Singh, A.; Rajwanshi, R.; Negi, N.P.; Rustagi, A.; Khushboo; Kumar, D. Exogenous Melatonin Counteracts Salinity and Cadmium Stress via Photosynthetic Machinery and Antioxidant Modulation in Solanum lycopersicum L. J. Plant Growth Regul. 2023, 42, 6332–6348. [Google Scholar] [CrossRef]
- Trchounian, A.; Petrosyan, M.; Sahakyan, N. Plant cell redox homeostasis and reactive oxygen species. In Redox State as a Central Regulator of Plant-Cell Stress Responses; Springer: Berlin/Heidelberg, Germany, 2016; pp. 25–50. [Google Scholar]
- Kiani, R.; Arzani, A.; Mirmohammady Maibody, S.A.M. Polyphenols, flavonoids, and antioxidant activity involved in salt tolerance in wheat, Aegilops cylindrica and their amphidiploids. Front. Plant Sci. 2021, 12, 646221. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Lu, J.; Meng, S.; Liu, Y.; Mostafa, I.; Qi, M.; Li, T. Exogenous melatonin improves salt tolerance in tomato by regulating photosynthetic electron flux and the ascorbate–glutathione cycle. J. Plant Interact. 2019, 14, 453–463. [Google Scholar] [CrossRef]
- Touchette, B.W.; Adams, E.C.; Laimbeer, P. Age-specific responses to elevated salinity in the coastal marsh plant black needlerush (Juncus roemerianus Scheele) as determined through polyphasic chlorophyll a fluorescence transients (OJIP). Mar. Biol. 2012, 159, 2137–2147. [Google Scholar] [CrossRef]
- Strasser, R.J.; Tsimilli-Michael, M.; Srivastava, A. Analysis of the chlorophyll a fluorescence transient. In Chlorophyll a Fluorescence: A Signature of Photosynthesis; Springer: Berlin/Heidelberg, Germany, 2004; pp. 321–362. [Google Scholar]
- Ayyaz, A.; Amir, M.; Umer, S.; Iqbal, M.; Bano, H.; Gul, H.S.; Noor, Y.; Javed, M.; Athar, H.R.; Zafar, Z.U. Melatonin induced changes in photosynthetic efficiency as probed by OJIP associated with improved chromium stress tolerance in canola (Brassica napus L.). Heliyon 2020, 6, e04364. [Google Scholar] [CrossRef]
- Guo, Y.Y.; Li, H.J.; Liu, J.; Bai, Y.W.; Xue, J.Q.; Zhang, R.H. Melatonin Alleviates Drought-Induced Damage of Photosynthetic Apparatus in Maize Seedlings. Russ. J. Plant Physiol. 2020, 67, 312–322. [Google Scholar] [CrossRef]
- Alyammahi, O.; Gururani, M.A. Chlorophyll-a Fluorescence Analysis Reveals Differential Response of Photosynthetic Machinery in Melatonin-Treated Oat Plants Exposed to Osmotic Stress. Agronomy 2020, 10, 1520. [Google Scholar] [CrossRef]
- Varghese, N.; Alyammahi, O.; Nasreddine, S.; Alhassani, A.; Gururani, M.A. Melatonin Positively Influences the Photosynthetic Machinery and Antioxidant System of Avena sativa during Salinity Stress. Plants 2019, 8, 610. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.Y.; Liu, J.L.; Wang, W.X.; Sun, Y. Exogenous melatonin improves growth and photosynthetic capacity of cucumber under salinity-induced stress. Photosynthetica 2016, 54, 19–27. [Google Scholar] [CrossRef]
- Nagashima, Y.; von Schaewen, A.; Koiwa, H. Function of N-glycosylation in plants. Plant Sci. 2018, 274, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, Q.; Wei, L.; Shi, Y.; Li, T.; Hu, K.; Liu, S.; Zhong, H.; Liao, J.; Li, Y. UDP-N-acetylglucosamine pyrophosphorylase 2 (UAP2) and 1 (UAP1) perform synergetic functions for leaf survival in rice. Front. Plant Sci. 2021, 12, 685102. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-H.; Shen, H.-L.; Chou, S.-J.; Sato, Y.; Cheng, W.-H. Interference of Arabidopsis N-Acetylglucosamine-1-P Uridylyltransferase Expression Impairs Protein N-Glycosylation and Induces ABA-Mediated Salt Sensitivity During Seed Germination and Early Seedling Development. Front. Plant Sci. 2022, 13, 903272. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Li, S.; Fan, C.; Cui, K.; Tan, H.; Qiao, L.; Lu, L. N-Acetylglucosamine promotes tomato plant growth by shaping the community structure and metabolism of the rhizosphere microbiome. Microbiol. Spectr. 2022, 10, e00358-22. [Google Scholar] [CrossRef]
- Bian, F.; Xiao, Q.; Hao, G.; Sun, Y.; Lu, W.; Zhai, H. Effect of root-applied melatonin on endogenous melatonin and chlorophyll fluorescence characteristics in grapevine under NaCl stress. Sci. Agric. Sin. 2018, 51, 952–963. [Google Scholar]
- Shomali, A.; Aliniaeifard, S.; Didaran, F.; Lotfi, M.; Mohammadian, M.; Seif, M.; Strobel, W.R.; Sierka, E.; Kalaji, H.M. Synergistic Effects of Melatonin and Gamma-Aminobutyric Acid on Protection of Photosynthesis System in Response to Multiple Abiotic Stressors. Cells 2021, 10, 1631. [Google Scholar] [CrossRef]
- Singh, H.; Kumar, D.; Soni, V. Impact of mercury on photosynthetic performance of Lemna minor: A chlorophyll fluorescence analysis. Sci. Rep. 2023, 13, 12181. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, A.; Kwon, E.-H.; Khan, M.A.; Shaffique, S.; Kang, S.-M.; Lee, I.-J. Enhanced use of chemical fertilizers and mitigation of heavy metal toxicity using biochar and the soil fungus Bipolaris maydis AF7 in rice: Genomic and metabolomic perspectives. Ecotoxicol. Environ. Saf. 2024, 271, 115938. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar] [CrossRef]
- Cao, J.; Xia, X.; Dai, X.; Xiao, J.; Wang, Q.; Andrae-Marobela, K.; Okatch, H. Flavonoids profiles, antioxidant, acetylcholinesterase inhibition activities of extract from Dryoathyrium boryanum (Willd.) Ching. Food Chem. Toxicol. 2013, 55, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Gulati, V.; Harding, I.H.; Palombo, E.A. Enzyme inhibitory and antioxidant activities of traditional medicinal plants: Potential application in the management of hyperglycemia. BMC Complement. Altern. Med. 2012, 12, 77. [Google Scholar] [CrossRef]
- Saeed, N.; Khan, M.R.; Shabbir, M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement. Altern. Med. 2012, 12, 221. [Google Scholar] [CrossRef]
- Kang, D.J.; Seo, Y.J.; Lee, J.D.; Ishii, R.; Kim, K.U.; Shin, D.H.; Park, S.K.; Jang, S.W.; Lee, I.J. Jasmonic acid differentially affects growth, ion uptake and abscisic acid concentration in salt-tolerant and salt-sensitive rice cultivars. J. Agron. Crop Sci. 2005, 191, 273–282. [Google Scholar] [CrossRef]
- Bhatta, D.; Adhikari, A.; Kang, S.-M.; Kwon, E.-H.; Jan, R.; Kim, K.-M.; Lee, I.-J. Hormones and the antioxidant transduction pathway and gene expression, mediated by Serratia marcescens DB1, lessen the lethality of heavy metals (As, Ni, and Cr) in Oryza sativa L. Ecotoxicol. Environ. Saf. 2023, 263, 115377. [Google Scholar] [CrossRef] [PubMed]

| SL (cm) | RL (cm) | SFW (g) | RFW (g) | SDW (g) | RDW (g) | SD (mm) | |
|---|---|---|---|---|---|---|---|
| No Stress (NS) | |||||||
| NT | 23.9 ± 0.59 b | 22.9 ± 0.22 b | 13.16 ± 0.35 b | 5.20 ± 0.13 b | 1.68 ± 0.05 c | 0.40 ± 0.02 c | 7.12 ± 0.03 b |
| Mel | 26.2 ± 0.30 a | 24.5 ± 0.44 ab | 16.33 ± 0.29 a | 5.88 ± 0.15 ab | 1.89 ± 0.03 b | 0.48 ± 0.01 b | 7.22 ± 0.10 b |
| GlcNAc | 27.0 ± 0.54 a | 24.8 ± 0.29 ab | 16.82 ± 0.30 a | 5.91 ± 0.16 ab | 1.84 ± 0.04 b | 0.46 ± 0.02 b | 7.79 ± 0.07 a |
| Mel × GlcNAc | 28.7 ± 0.20 a | 26.9 ± 0.33 a | 17.30 ± 0.17 a | 6.64 ± 0.20 a | 1.96 ± 0.02 a | 0.52 ± 0.02 a | 7.96 ± 0.03 a |
| Salinity Stress (SS) | |||||||
| NT | 18.9 ± 0.43 b | 18.0 ± 0.34 c | 9.40 ± 0.57 c | 4.15 ± 0.37 c | 1.21 ± 0.06 c | 0.32 ± 0.02 c | 6.31 ± 0.09 c |
| Mel | 20.6 ± 0.70 b | 22.1 ± 0.41 b | 10.34 ± 0.37 b | 6.21 ± 0.40 a | 1.59 ± 0.07 b | 0.44 ± 0.02 b | 7.03 ± 0.06 b |
| GlcNAc | 20.8 ± 0.55 b | 22.8 ± 0.47 b | 11.41 ± 0.47 b | 5.76 ± 0.37 b | 1.49 ± 0.06 b | 0.45 ± 0.02 b | 7.06 ± 0.05 b |
| Mel × GlcNAc | 24.6 ± 0.44 a | 25.1 ± 0.38 a | 13.63 ± 0.26 a | 6.30 ± 0.29 a | 1.77 ± 0.02 a | 0.50 ± 0.03 a | 7.82 ± 0.14 a |
| ABS/RC | Absorption Flux per RC |
|---|---|
| TRo/RC | Trapping flux per RC |
| ETo/RC | Electron transport flux per RC |
| Fv/Fo | Ratio of variable fluorescence and minimum fluorescence |
| TRo/ABS | Maximum quantum yield of the apparatus primary photochemical |
| ETo/TRo | Electron movement efficiency |
| DIo/CS | Dissipated energy flux per CS (at t = 0) |
| ETo/CS | Electron transport flux (further than QA−) per RC |
| TRo/CSo | Trapping flux (leading to QA reduction) per Cso |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, S.-M.; Adhikari, A.; Kwon, E.-H.; Gam, H.-J.; Jeon, J.R.; Woo, J.-I.; Lee, I.-J. Influence of N-Acetylglucosamine and Melatonin Interaction in Modeling the Photosynthetic Component and Metabolomics of Cucumber under Salinity Stress. Int. J. Mol. Sci. 2024, 25, 2844. https://doi.org/10.3390/ijms25052844
Kang S-M, Adhikari A, Kwon E-H, Gam H-J, Jeon JR, Woo J-I, Lee I-J. Influence of N-Acetylglucosamine and Melatonin Interaction in Modeling the Photosynthetic Component and Metabolomics of Cucumber under Salinity Stress. International Journal of Molecular Sciences. 2024; 25(5):2844. https://doi.org/10.3390/ijms25052844
Chicago/Turabian StyleKang, Sang-Mo, Arjun Adhikari, Eun-Hae Kwon, Ho-Jun Gam, Jin Ryeol Jeon, Ji-In Woo, and In-Jung Lee. 2024. "Influence of N-Acetylglucosamine and Melatonin Interaction in Modeling the Photosynthetic Component and Metabolomics of Cucumber under Salinity Stress" International Journal of Molecular Sciences 25, no. 5: 2844. https://doi.org/10.3390/ijms25052844
APA StyleKang, S.-M., Adhikari, A., Kwon, E.-H., Gam, H.-J., Jeon, J. R., Woo, J.-I., & Lee, I.-J. (2024). Influence of N-Acetylglucosamine and Melatonin Interaction in Modeling the Photosynthetic Component and Metabolomics of Cucumber under Salinity Stress. International Journal of Molecular Sciences, 25(5), 2844. https://doi.org/10.3390/ijms25052844







