Abscisic Acid Affects Phenolic Acid Content to Increase Tolerance to UV-B Stress in Rhododendron chrysanthum Pall.
Abstract
1. Introduction
2. Results
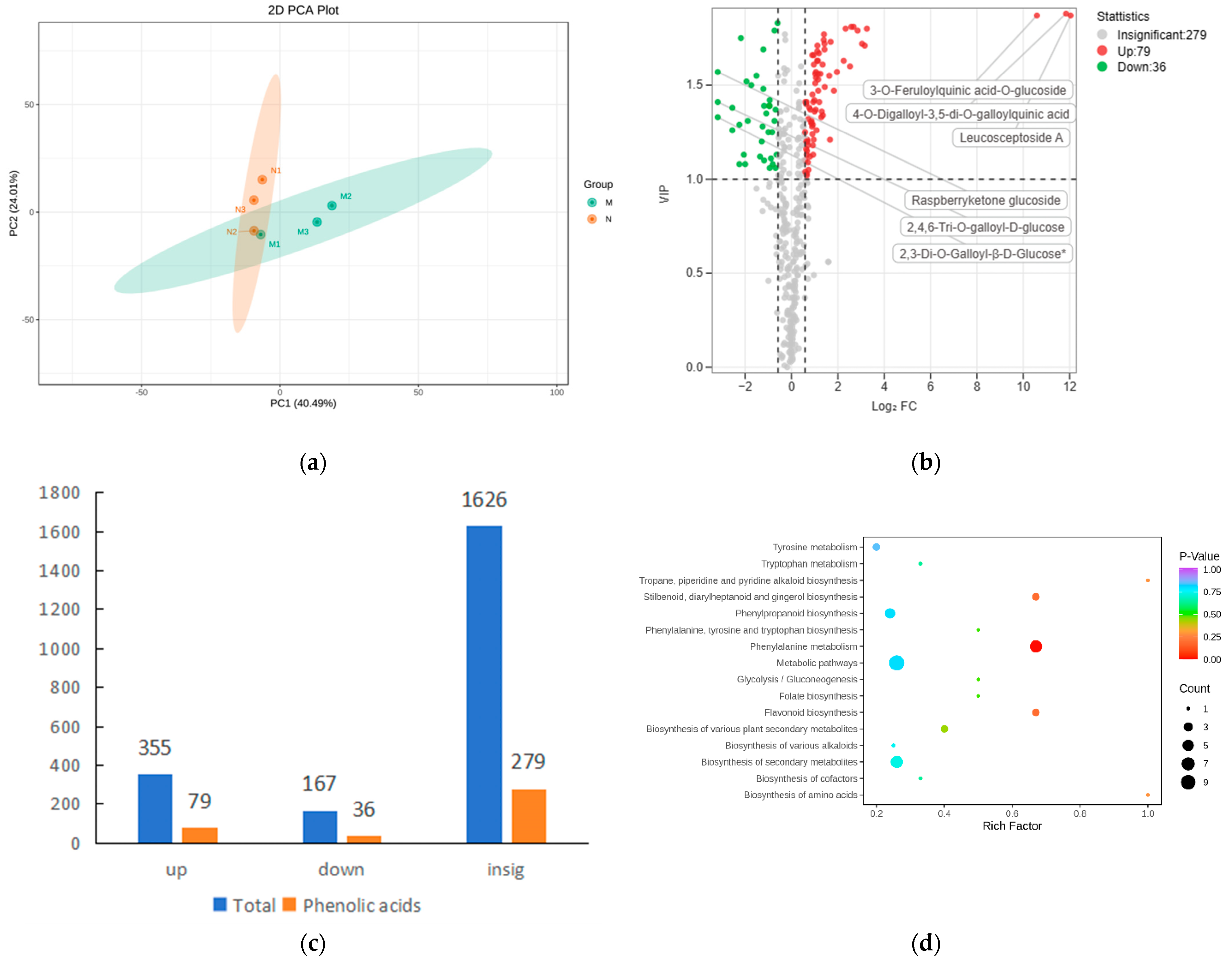
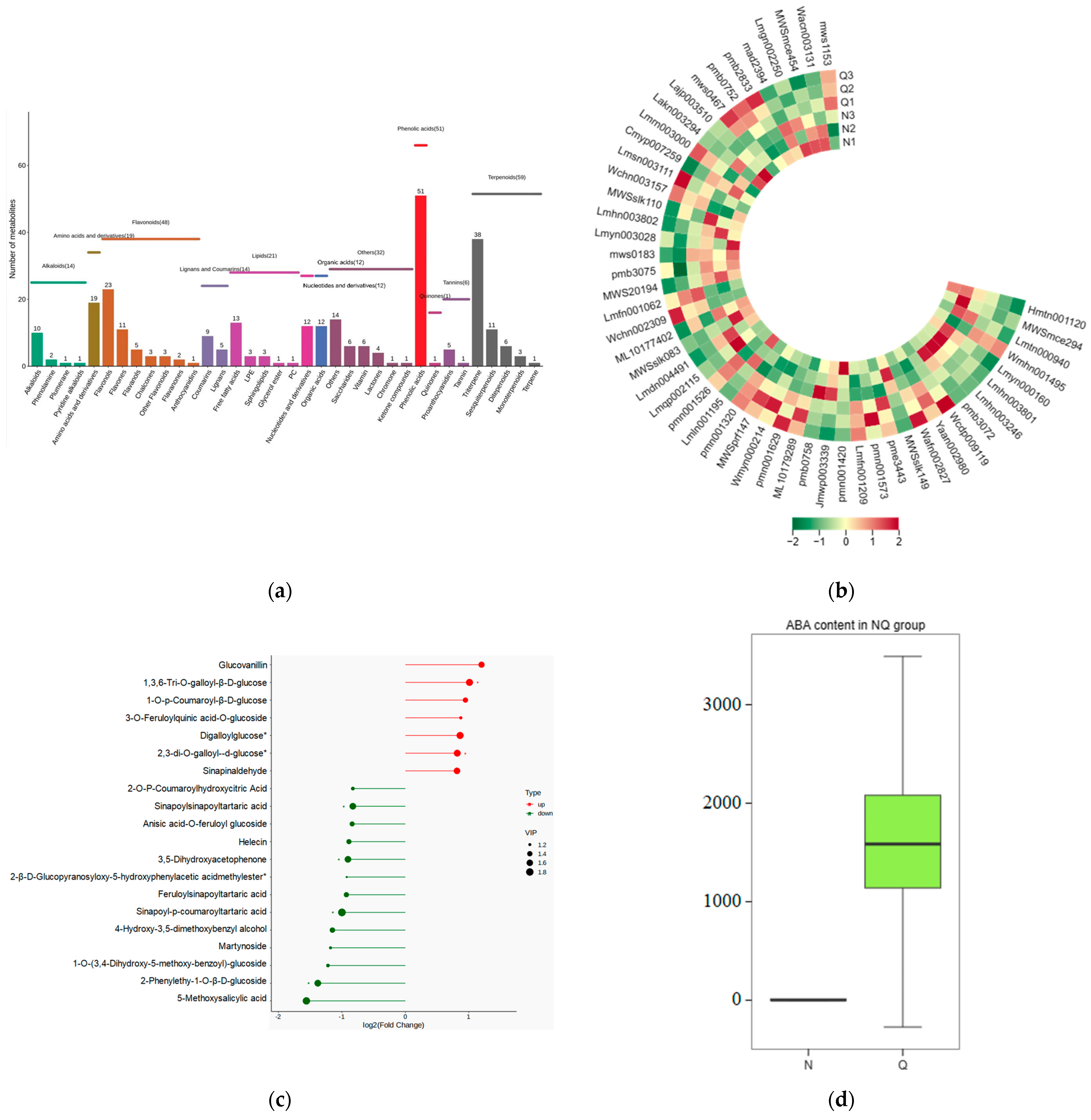
2.1. Large Amounts of Secondary Metabolites Were Produced in the Leaves of R. chrysanthum after UV-B Stress
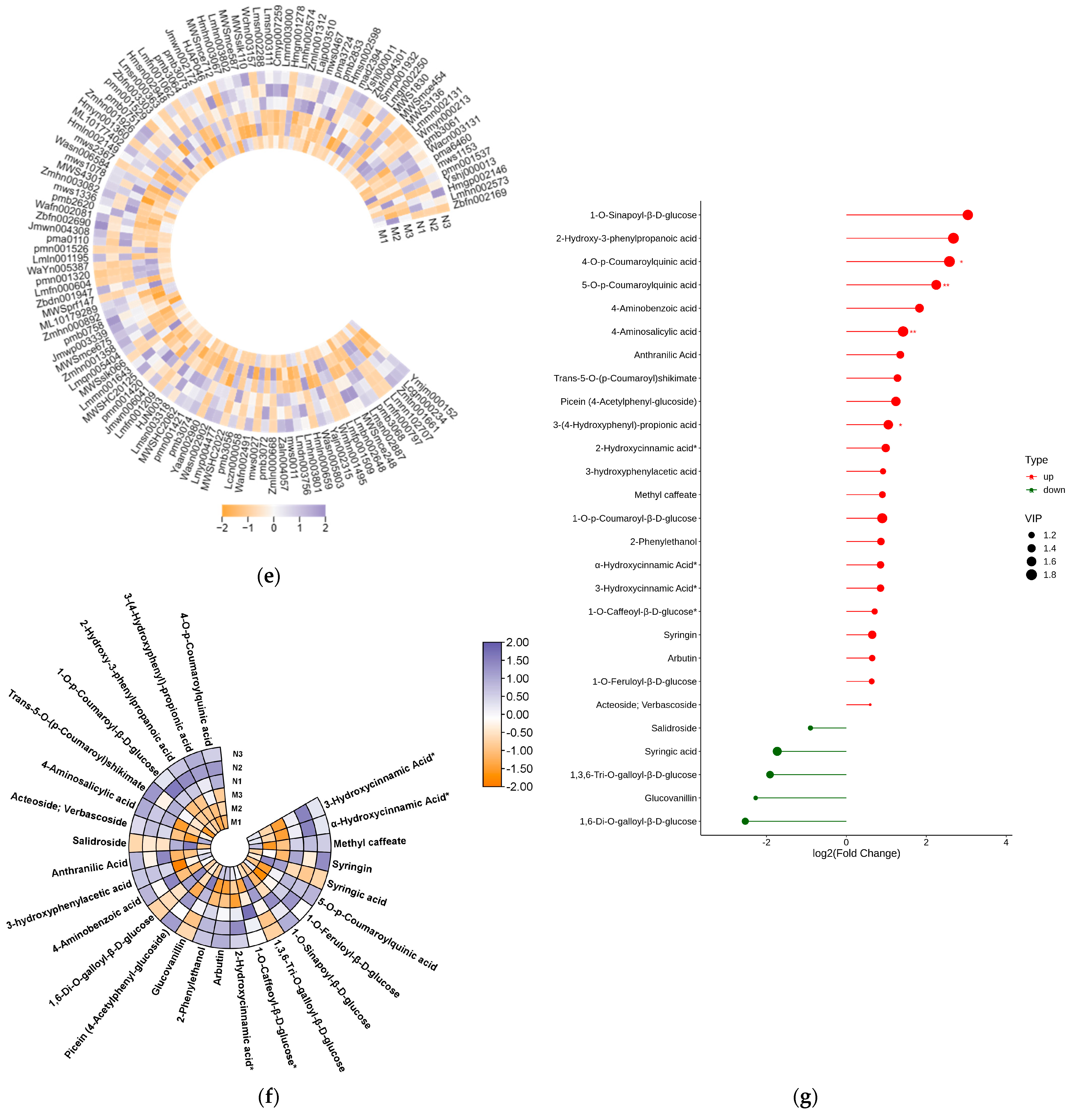
2.2. Significant Changes in Phenolic Acids in Leaves after UV-B Stress
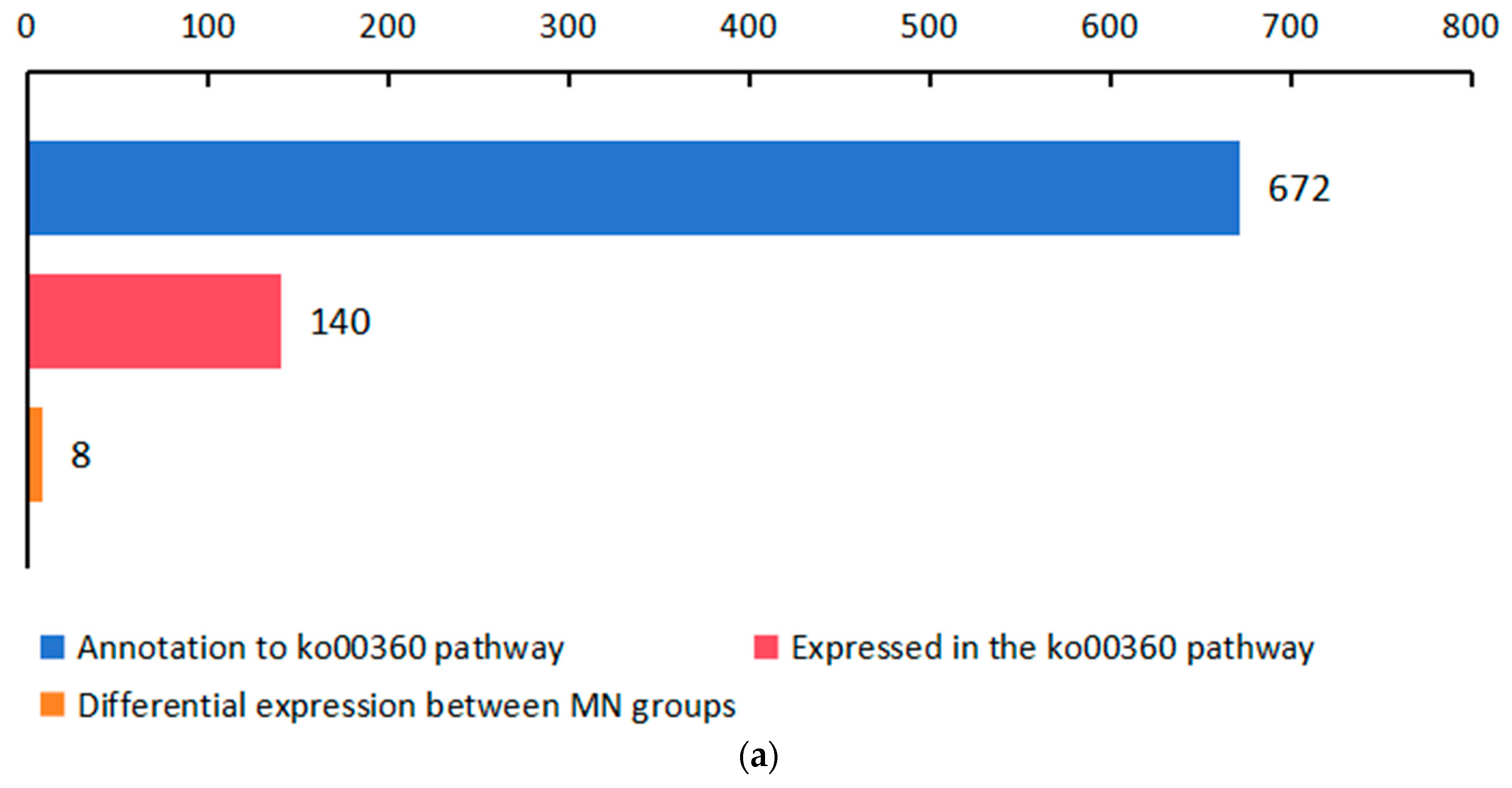
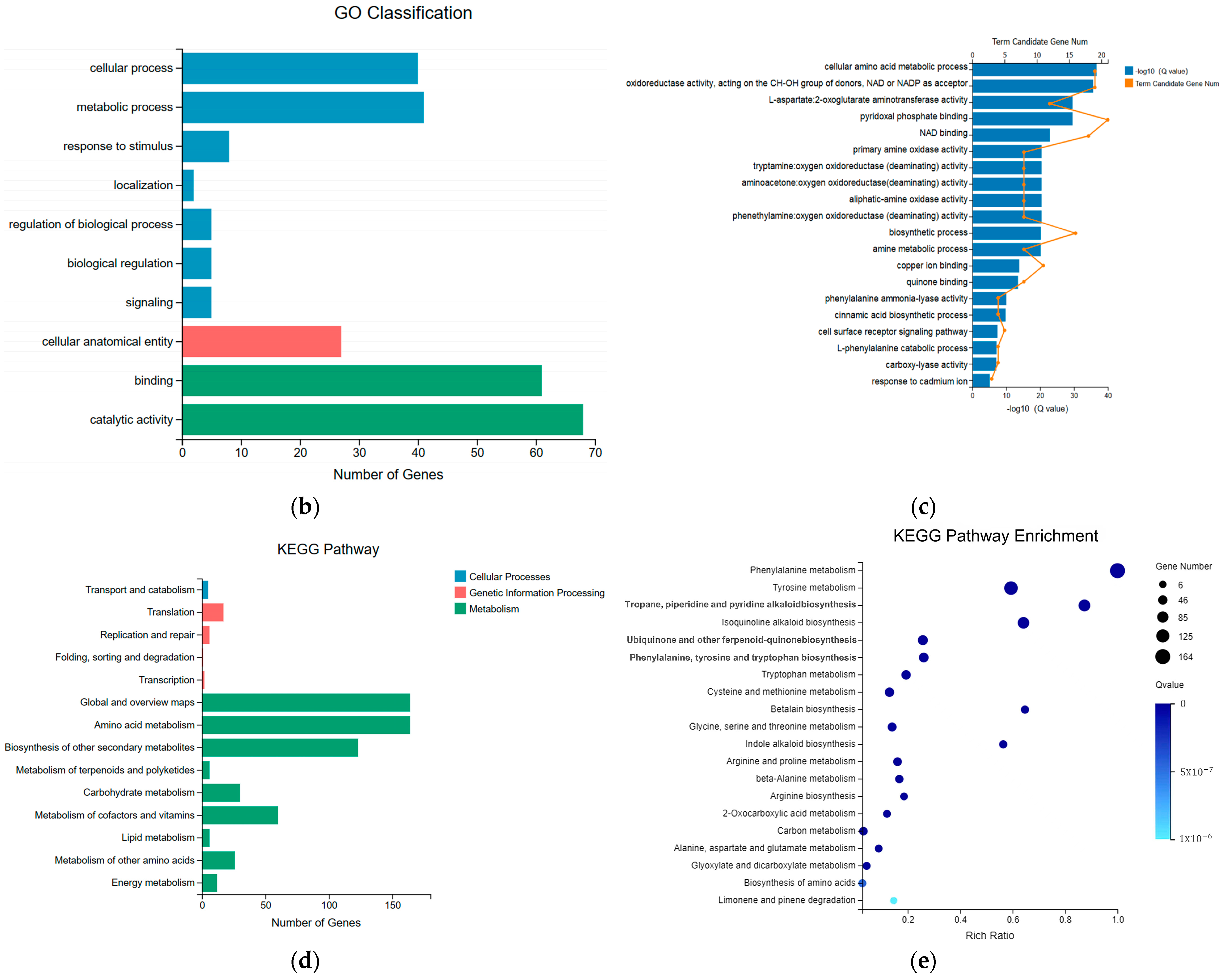
2.3. Significant Changes in Genes Related to Phenolic Acid Synthesis in Leaves after UV-B Stress
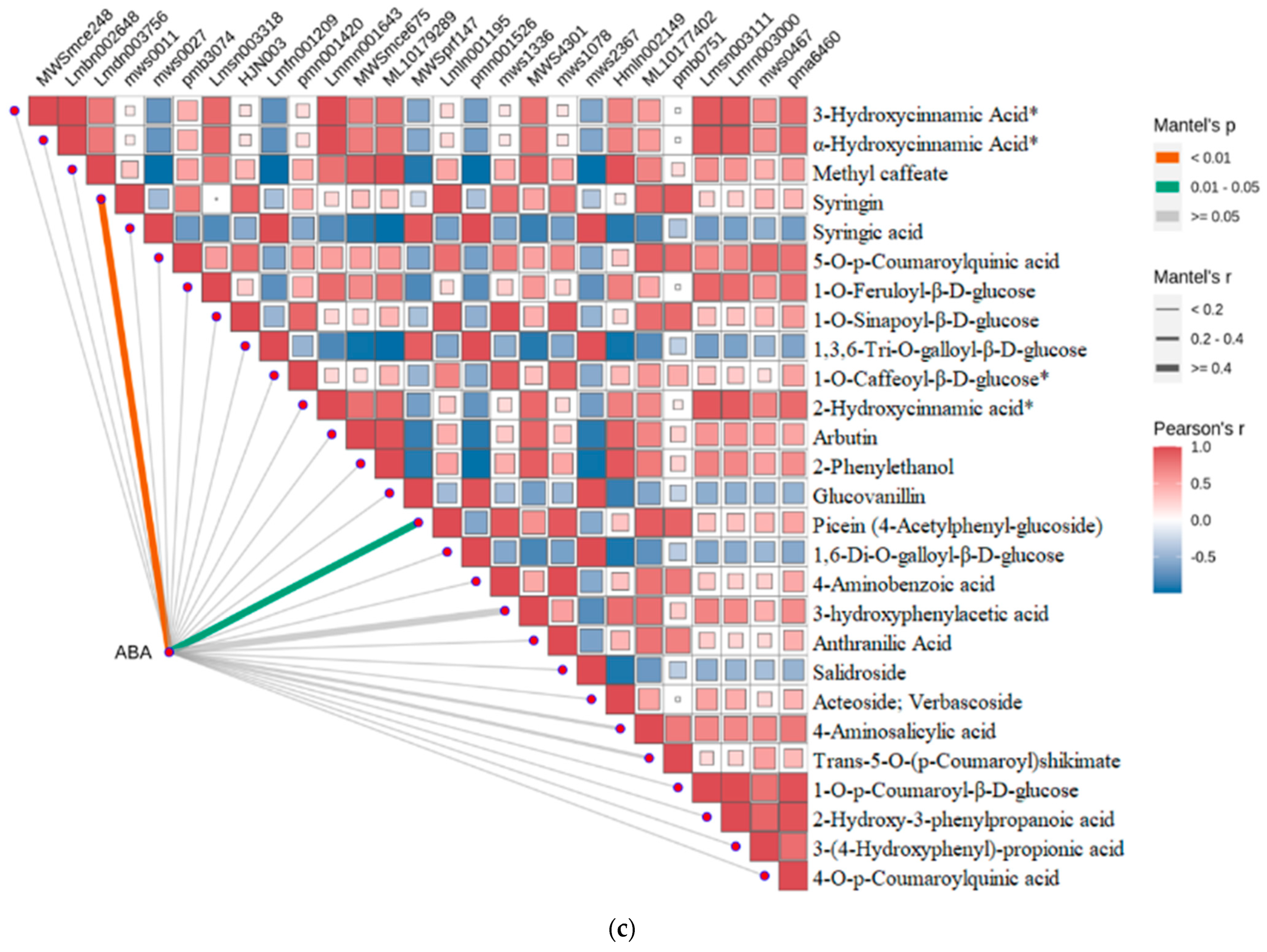
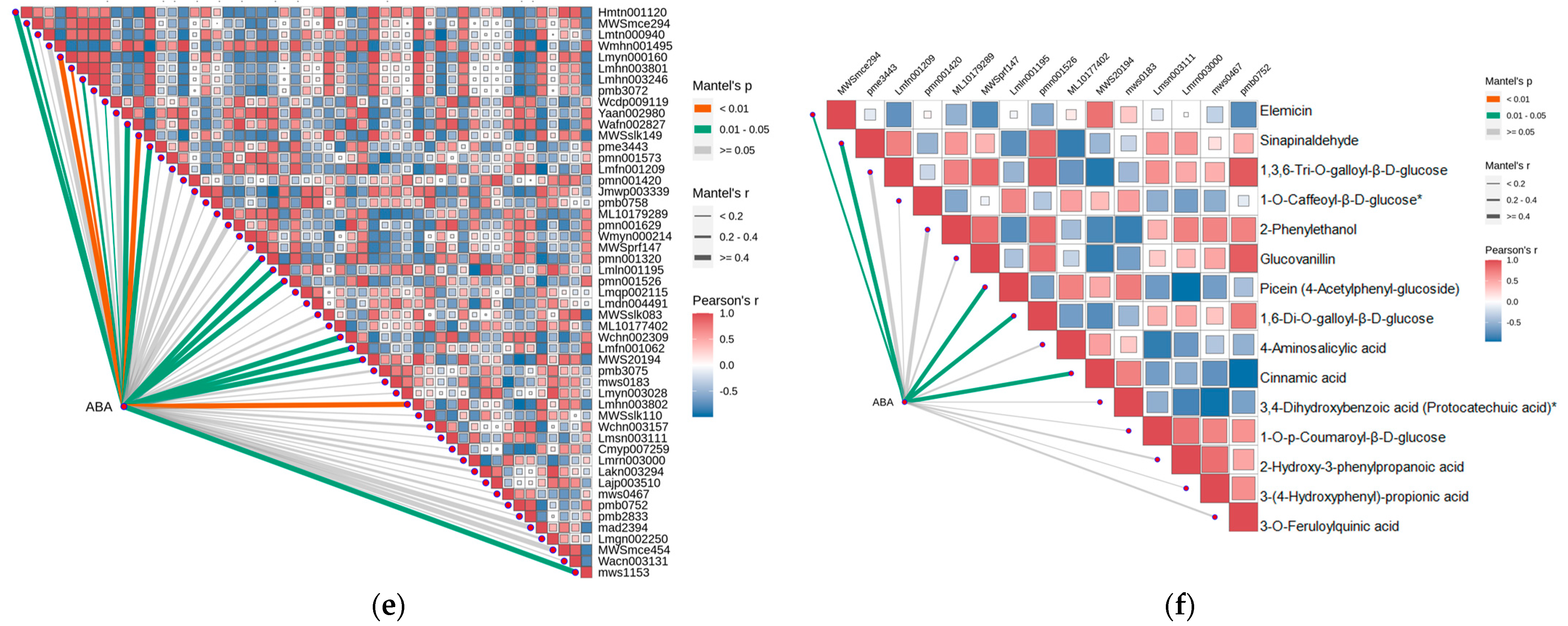
2.4. Strong Correlation between Phenolic Acid and Abscisic Acid in Leaves of R. chrysanthum
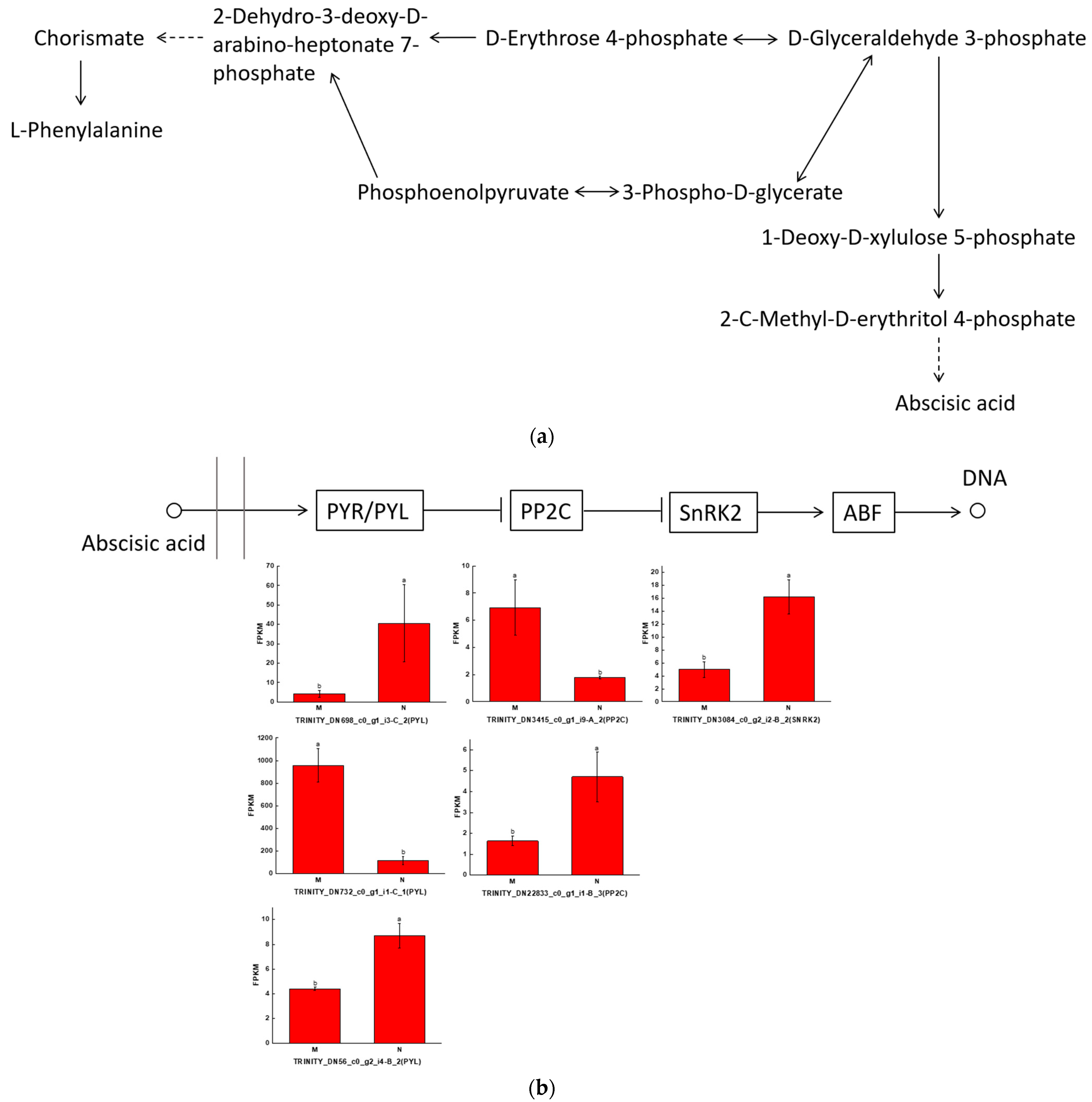
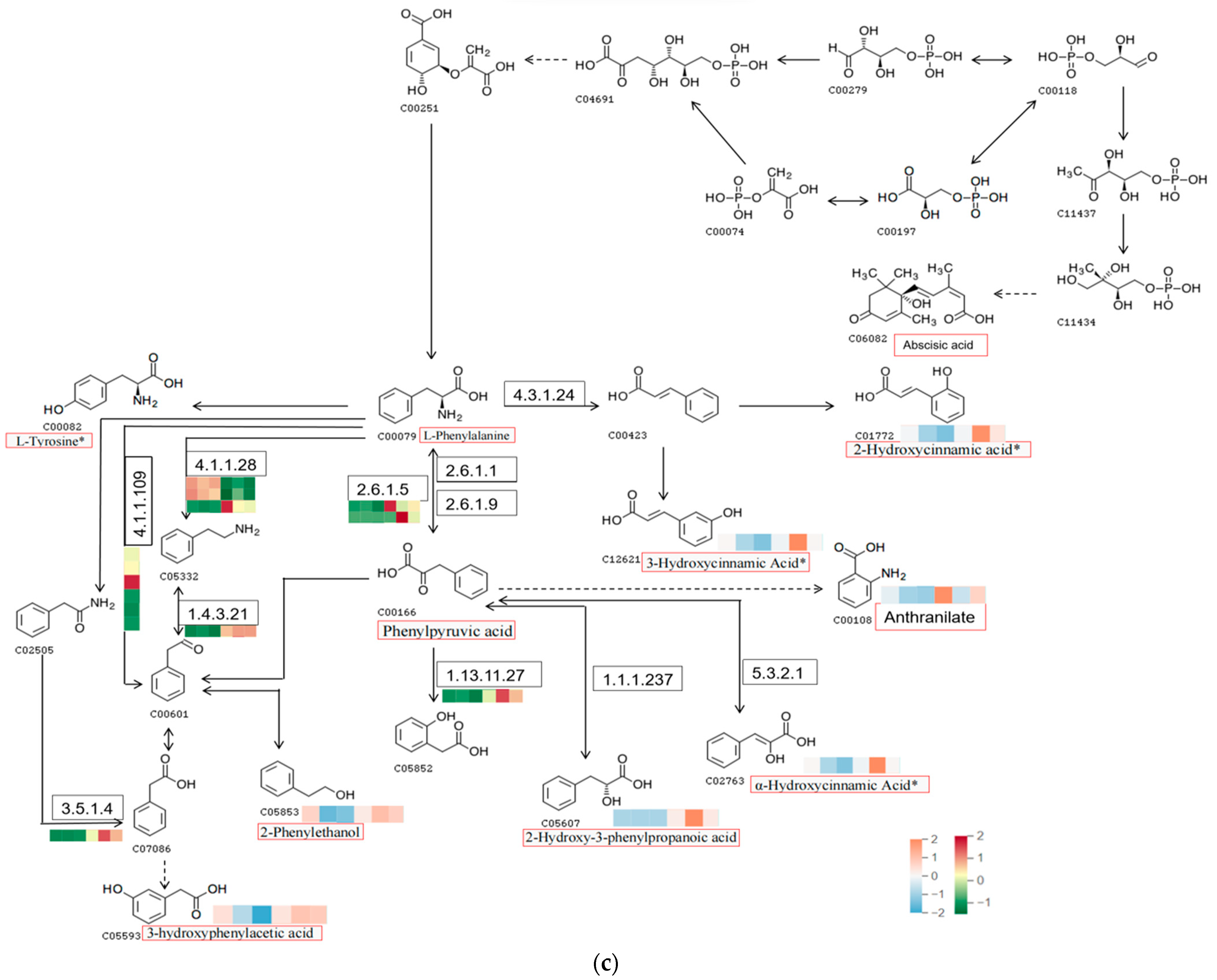
2.5. Integrating Transcriptomic and Metabolomic Analyses to Map Phenolic Acid Synthesis Pathways
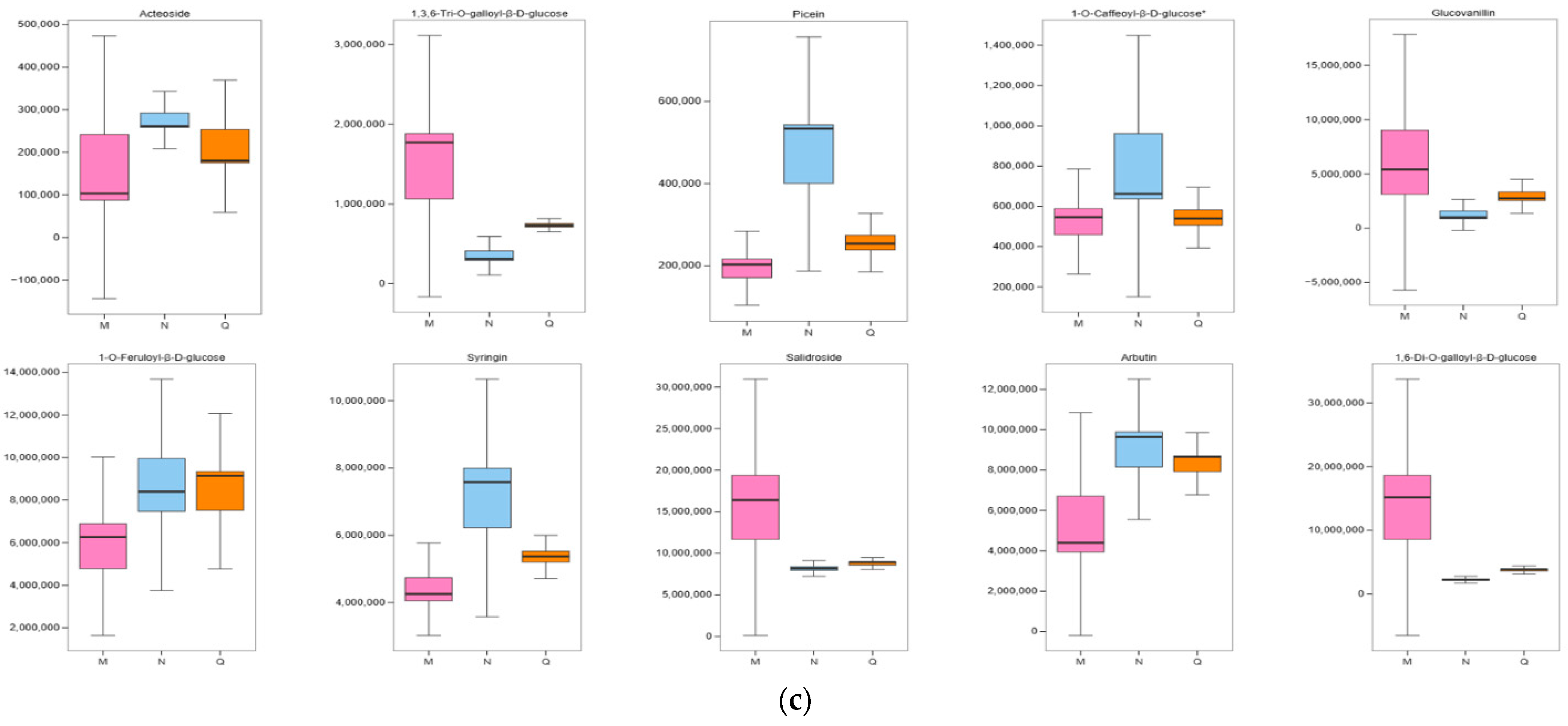
2.6. Significant Changes in Phenolic Acids in Externally Applied Abscisic Acid in R. chrysanthum
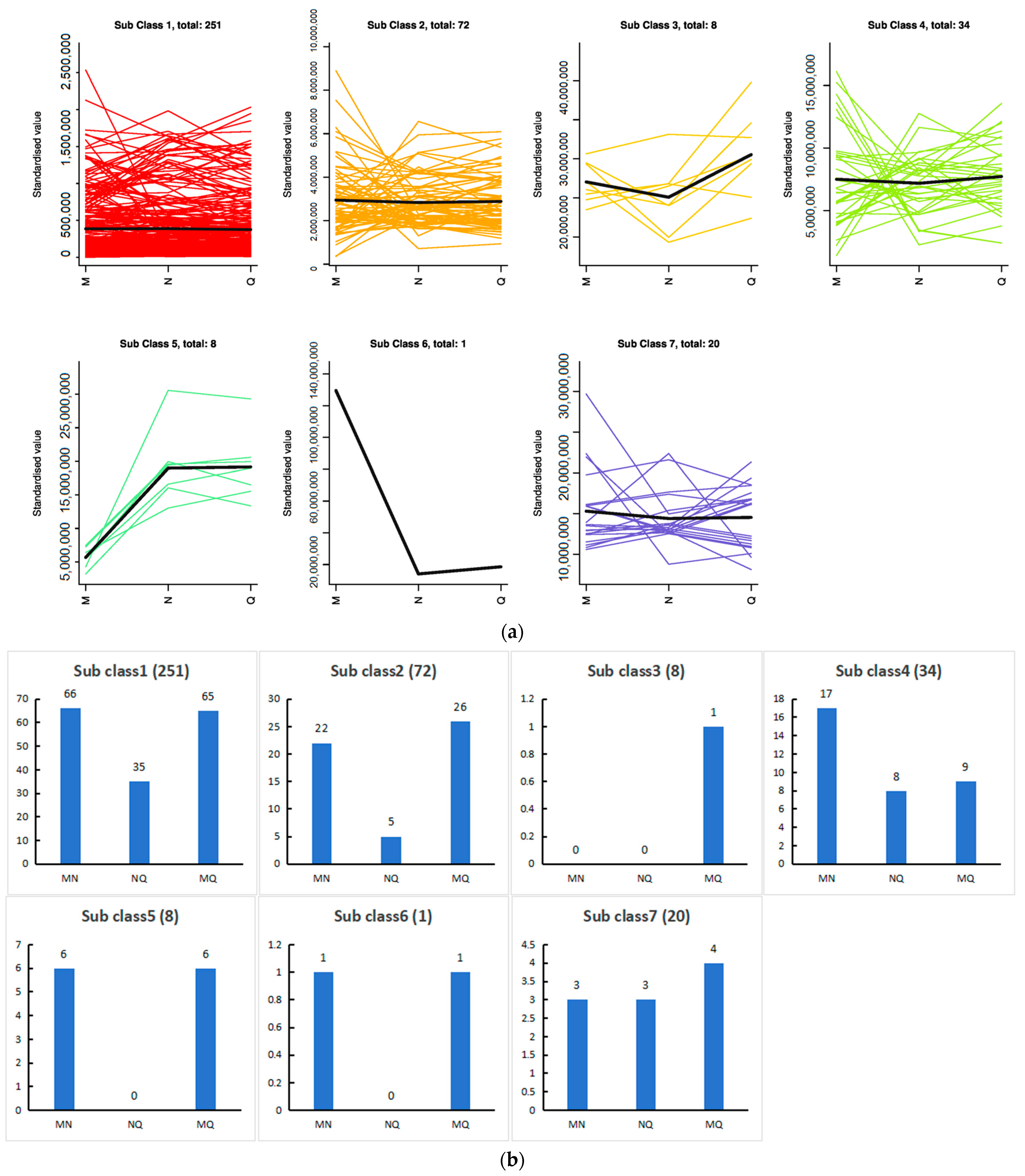
2.7. K-Means Analysis of the Three Treatment Groups Revealed That Phenolic Acids Were Affected by ABA
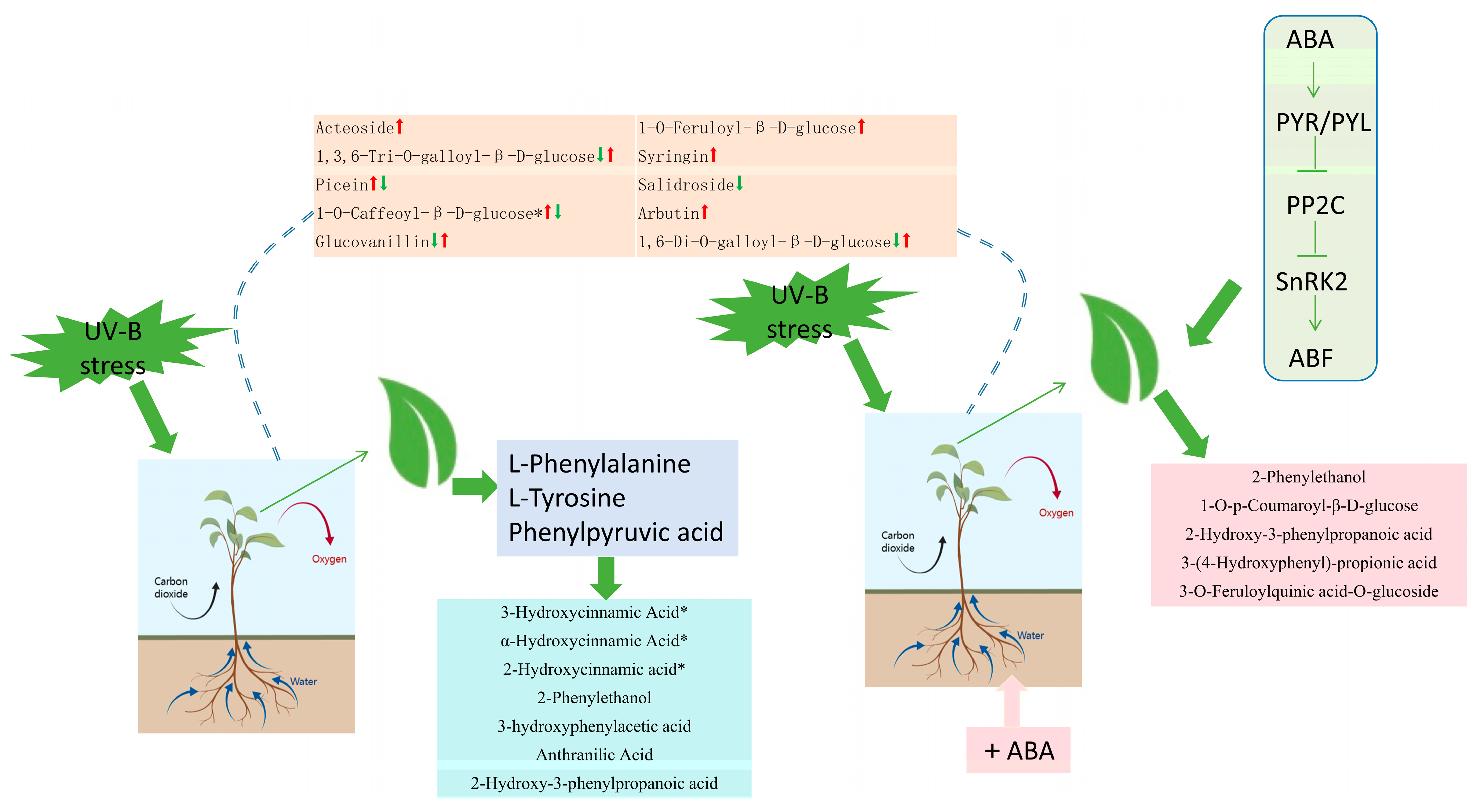
2.8. Hypothetical Modeling of Phenolic Acid Response of R. chrysanthum Leaves under UV-B Stress
3. Discussion
3.1. Phenolic Acid Enhances UV-B Radiation Resistance in R. chrysanthum
3.2. Abscisic Acid Plays an Important Role in UV-B Radiation Resistance in R. chrysanthum
3.3. External Application of Abscisic Acid Gives Greater Resistance to Radiation in R. chrysanthum
3.4. Abscisic Acid Can Influence Phenolic Acid Production to Promote the Resistance of R. chrysanthum to UV-B Stress
4. Materials and Methods
4.1. Plant Materials and Treatments
4.2. Widely Targeted Metabolomics Assays
4.3. RNA-Seq Analysis
4.4. De Novo Assemble
4.5. Gene Annotation
4.6. Screening for Differential Metabolites and Differentially Expressed Genes
4.7. Statistical Analysis for Bioinformatics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| R. chrysanthum | Rhododendron chrysanthum Pall. |
| ABA | abscisic acid |
| PAR | 400–700 nm light required by plants for photosynthesis |
| UV-B | Ultraviolet radiation B |
| ROS | Reactive oxygen species |
| PSII | Photosystem II |
| UPLC-MS/MS | Ultra-performance liquid chromatography—tandem mass spectrometry |
| PCA | Principal component analysis |
| FC | Fold Change |
| VIP | Variable Importance in Projection |
| KEGG | Kyoto encyclopedia of genes and genomes |
| GO | Gene Ontology |
| DEGs | Differentially expressed genes |
| EBR | 24-epibrassinolide |
References
- Piofczyk, T.; Jeena, G.; Pecinka, A. Arabidopsis thaliana natural variation reveals connections between UV radiation stress and plant pathogen-like defense responses. Plant Physiol. Biochem. 2015, 93, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Takshak, S.; Agrawal, S.B. Defense potential of secondary metabolites in medicinal plants under UV-B stress. J. Photochem. Photobiol. B 2019, 193, 51–88. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Dong, Y.; Huang, X. Plant responses to UV-B radiation: Signaling, acclimation and stress tolerance. Stress. Biol. 2022, 2, 51. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Liu, H. How plants protect themselves from ultraviolet-B radiation stress. Plant Physiol. 2021, 187, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Luis, J.C.; Pérez, R.M.; González, F.V. UV-B radiation effects on foliar concentrations of rosmarinic and carnosic acids in rosemary plants. Food Chem. 2007, 101, 1211–1215. [Google Scholar] [CrossRef]
- Moura, D.J.; Richter, M.F.; Boeira, J.M.; Pêgas Henriques, J.A.; Saffi, J. Antioxidant properties of beta-carboline alkaloids are related to their antimutagenic and antigenotoxic activities. Mutagenesis 2007, 22, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Landry, L.G.; Chapple, C.C.; Last, R.L. Arabidopsis mutants lacking phenolic sunscreens exhibit enhanced ultraviolet-B injury and oxidative damage. Plant Physiol. 1995, 109, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Jianzhou, C.; Xueli, H.; Binbin, L.; Jingmin, L.; Zhaowei, Y. Effects of selenium on agronomical characters of winter wheat exposed to enhanced ultraviolet-B. Ecotoxicol. Environ. Saf. 2013, 92, 320–326. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, M.; Cao, K.; Xu, H.; Zhou, X. UV-B Irradiation to Amino Acids and Carbohydrate Metabolism in Rhododendron chrysanthum Leaves by Coupling Deep Transcriptome and Metabolome Analysis. Plants 2022, 11, 2730. [Google Scholar] [CrossRef]
- Liu, M.; Sun, Q.; Cao, K.; Xu, H.; Zhou, X. Acetylated Proteomics of UV-B Stress-Responsive in Photosystem II of Rhododendron chrysanthum. Cells 2023, 12, 478. [Google Scholar] [CrossRef]
- Sehrawat, R.; Rathee, P.; Akkol, E.K.; Khatkar, S.; Lather, A.; Redhu, N.; Khatkar, A. Phenolic Acids—Versatile Natural Moiety with Numerous Biological Applications. Curr. Top. Med. Chem. 2022, 22, 1472–1484. [Google Scholar] [CrossRef] [PubMed]
- Goufo, P.; Trindade, H. Rice antioxidants: Phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, γ-oryzanol, and phytic acid. Food Sci. Nutr. 2014, 2, 75–104. [Google Scholar] [CrossRef] [PubMed]
- Bogucka-Kocka, A.; Zidorn, C.; Kasprzycka, M.; Szymczak, G.; Szewczyk, K. Phenolic acid content, antioxidant and cytotoxic activities of four Kalanchoë species. Saudi J. Biol. Sci. 2018, 25, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Waadt, R.; Seller, C.A.; Hsu, P.K.; Takahashi, Y.; Munemasa, S.; Schroeder, J.I. Plant hormone regulation of abiotic stress responses. Nat. Rev. Mol. Cell Biol. 2022, 23, 680–694. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Partida, R.; Rosario, S.M.; Lozano-Juste, J. An Update on Crop ABA Receptors. Plants 2021, 10, 1087. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Liu, J.H.; Mohapatra, S.; Hill, R.D.; Mohapatra, S.S. Characterization of a gene family encoding abscisic acid- and environmental stress-inducible proteins of alfalfa. J. Biol. Chem. 1992, 267, 15367–15374. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yang, A.; Yin, H.; Zhang, J. Influence of water stress on endogenous hormone contents and cell damage of maize seedlings. J. Integr. Plant Biol. 2008, 50, 427–434. [Google Scholar] [CrossRef]
- Haverroth, E.J.; Oliveira, L.A.; Andrade, M.T.; Taggart, M.; McAdam, S.A.M.; Zsögön, A.; Thompson, A.J.; Martins, S.C.V.; Cardoso, A.A. Abscisic acid acts essentially on stomata, not on the xylem, to improve drought resistance in tomato. Plant Cell Environ. 2023, 46, 3229–3241. [Google Scholar] [CrossRef]
- Singh, N.K.; Larosa, P.C.; Handa, A.K.; Hasegawa, P.M.; Bressan, R.A. Hormonal regulation of protein synthesis associated with salt tolerance in plant cells. Proc. Natl. Acad. Sci. USA 1987, 84, 739–743. [Google Scholar] [CrossRef]
- Sah, S.K.; Reddy, K.R.; Li, J. Abscisic Acid and Abiotic Stress Tolerance in Crop Plants. Front. Plant Sci. 2016, 7, 571. [Google Scholar] [CrossRef]
- Mohapatra, S.S.; Poole, R.J.; Dhindsa, R.S. Abscisic Acid-regulated gene expression in relation to freezing tolerance in alfalfa. Plant Physiol. 1988, 87, 468–473. [Google Scholar] [CrossRef]
- Dong, Z.; Chen, Y. Transcriptomics: Advances and approaches. Sci. China Life Sci. 2013, 56, 960–967. [Google Scholar] [CrossRef]
- Tyagi, P.; Singh, D.; Mathur, S.; Singh, A.; Ranjan, R. Upcoming progress of transcriptomics studies on plants: An overview. Front. Plant Sci. 2022, 13, 1030890. [Google Scholar] [CrossRef] [PubMed]
- Loke, K.K.; Rahnamaie-Tajadod, R.; Yeoh, C.C.; Goh, H.H.; Mohamed-Hussein, Z.A.; Zainal, Z.; Ismail, I.; Mohd Noor, N. Transcriptome analysis of Polygonum minus reveals candidate genes involved in important secondary metabolic pathways of phenylpropanoids and flavonoids. PeerJ 2017, 5, e2938. [Google Scholar] [CrossRef]
- Zou, S.; Wu, J.; Shahid, M.Q.; He, Y.; Lin, S.; Liu, Z.; Yang, X. Identification of key taste components in loquat using widely targeted metabolomics. Food Chem. 2020, 323, 126822. [Google Scholar] [CrossRef] [PubMed]
- Cuperlovic-Culf, M.; Belacel, N.; Culf, A. Integrated analysis of transcriptomics and metabolomics profiles. Expert. Opin. Med. Diagn. 2008, 2, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, Y.; Zhu, J.K. Developing naturally stress-resistant crops for a sustainable agriculture. Nat. Plants 2018, 4, 989–996. [Google Scholar] [CrossRef]
- Ferguson, J.N. Climate change and abiotic stress mechanisms in plants. Emerg. Top. Life Sci. 2019, 3, 165–181. [Google Scholar] [CrossRef]
- Holmes, M.G.; Keiller, D.R. Effects of pubescence and waxes on the reflectance of leaves in the ultraviolet and photosynthetic wavebands: A comparison of a range of species. Plant Cell Environ. 2002, 25, 85–93. [Google Scholar] [CrossRef]
- Gong, F.; Yu, W.; Zeng, Q.; Dong, J.; Cao, K.; Xu, H.; Zhou, X. Rhododendron chrysanthum’s Primary Metabolites Are Converted to Phenolics More Quickly When Exposed to UV-B Radiation. Biomolecules 2023, 13, 1700. [Google Scholar] [CrossRef]
- Qu, J.; Chen, T.; Yao, M.; Wang, Y.; Xiao, W.; Li, B. ABC transporter and its application in synthetic biology. Sheng Wu Gong. Cheng Xue Bao 2020, 36, 1754–1766. [Google Scholar] [CrossRef] [PubMed]
- Pascual, M.B.; El-Azaz, J.; de la Torre, F.N.; Cañas, R.A.; Avila, C.; Cánovas, F.M. Biosynthesis and Metabolic Fate of Phenylalanine in Conifers. Front. Plant Sci. 2016, 7, 1030. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.N.; Wang, W.; Yao, G.D.; Jiang, X.F.; Zhang, Y.; Song, S.J. Antioxidant phenolic acids from the leaves of Armeniaca sibirica. J. Asian Nat. Prod. Res. 2018, 20, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Liu, W.; Mu, Y.P.; Zhang, H.; Wang, X.N.; Zhao, C.Q.; Chen, J.M.; Liu, P. Pharmacological Effects of Salvianolic Acid B Against Oxidative Damage. Front. Pharmacol. 2020, 11, 572373. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.N.; Bahler, B.D.; Hofmann, G.A.; Mattern, M.R.; Johnson, R.K.; Kingston, D.G. Phenylethanoid glycosides from Digitalis purpurea and Penstemonlinarioides with PKCalpha-inhibitory activity. J. Nat. Prod. 1998, 61, 1410–1412. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, J.; Nam, Y.J.; Kim, H.J.; No, K.T. Isolindleyin exerts anti-melanogenic effects in human epidermal melanocytes via direct binding to tyrosinase. Biochem. Biophys. Res. Commun. 2021, 534, 802–807. [Google Scholar] [CrossRef]
- Hayashi, K.; Kinoshita, T. Plant signaling: Abscisic acid receptor hole-in-one. Nat. Chem. Biol. 2014, 10, 414–415. [Google Scholar] [CrossRef]
- Sun, Q.; Zhou, X.; Yang, L.; Xu, H.; Zhou, X. Integration of Phosphoproteomics and Transcriptome Studies Reveals ABA Signaling Pathways Regulate UV-B Tolerance in Rhododendron chrysanthum Leaves. Genes 2023, 14, 1153. [Google Scholar] [CrossRef]
- Kushiro, T.; Okamoto, M.; Nakabayashi, K.; Yamagishi, K.; Kitamura, S.; Asami, T.; Hirai, N.; Koshiba, T.; Kamiya, Y.; Nambara, E. The Arabidopsis cytochrome P450 CYP707A encodes ABA 8’-hydroxylases: Key enzymes in ABA catabolism. EMBO J. 2004, 23, 1647–1656. [Google Scholar] [CrossRef]
- Yu, W.; Zhao, R.; Wang, L.; Zhang, S.; Li, R.; Sheng, J.; Shen, L. ABA signaling rather than ABA metabolism is involved in trehalose-induced drought tolerance in tomato plants. Planta 2019, 250, 643–655. [Google Scholar] [CrossRef]
- Asaf, S.; Khan, A.L.; Khan, M.A.; Imran, Q.M.; Yun, B.-W.; Lee, I.-J. Osmoprotective functions conferred to soybean plants via inoculation with Sphingomonas sp. LK11 and exogenous trehalose. Microbiol. Res. 2017, 205, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Boneh, U.; Biton, I.; Schwartz, A.; Ben-Ari, G. Characterization of the ABA signal transduction pathway in Vitis vinifera. Plant Sci. 2012, 187, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Jędrejek, D.; Kontek, B.; Lis, B.; Stochmal, A.; Olas, B. Evaluation of antioxidant activity of phenolic fractions from the leaves and petals of dandelion in human plasma treated with H2O2 and H2O2/Fe. Chem. Biol. Interact. 2017, 262, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wang, X.; Zhang, J.; Li, N.; Wu, R.; Wang, T.; Ding, W. Comparative metabolomic analysis of different-colored hawthorn berries (Crataegus pinnatifida) provides a new interpretation of color trait and antioxidant activity. LWT 2022, 163, 113623. [Google Scholar] [CrossRef]
- Ye, N.; Jia, L.; Zhang, J. ABA signal in rice under stress conditions. Rice 2012, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Zhang, Y.; Liu, Y.; Jian, S.; Deng, S. A novel ABA-induced transcript factor from Millettia pinnata, MpAITR1, enhances salt and drought tolerance through ABA signaling in transgenic Arabidopsis. J. Plant Physiol. 2023, 288, 154060. [Google Scholar] [CrossRef] [PubMed]
- Nagao, M.; Minami, A.; Arakawa, K.; Fujikawa, S.; Takezawa, D. Rapid degradation of starch in chloroplasts and concomitant accumulation of soluble sugars associated with ABA-induced freezing tolerance in the moss Physcomitrella patens. J. Plant Physiol. 2005, 162, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Habibpourmehraban, F.; Wu, Y.; Masoomi-Aladizgeh, F.; Amirkhani, A.; Atwell, B.J.; Haynes, P.A. Pre-Treatment of Rice Plants with ABA Makes Them More Tolerant to Multiple Abiotic Stress. Int. J. Mol. Sci. 2023, 24, 9628. [Google Scholar] [CrossRef]
- Zhang, Y.; Xiao, Y.; Zhang, Y.; Dong, Y.; Liu, Y.; Liu, L.; Wan, S.; He, J.; Yu, Y. Accumulation of Galactinol and ABA Is Involved in Exogenous EBR-Induced Drought Tolerance in Tea Plants. J. Agric. Food Chem. 2022, 70, 13391–13403. [Google Scholar] [CrossRef]
- Lyu, J.; Wang, C.; Liang, D.Y.; Liu, L.; Pandey, L.K.; Xu, H.W.; Zhou, X.F. Sensitivity of wild and domesticated Rhododendron chrysanthum to different light regime (UVA, UVB, and PAR). Photosynthetica 2019, 57, 841–849. [Google Scholar] [CrossRef]
- Fraga, C.G.; Clowers, B.H.; Moore, R.J.; Zink, E.M. Signature-discovery approach for sample matching of a nerve-agent precursor using liquid chromatography-mass spectrometry, XCMS, and chemometrics. Anal. Chem. 2010, 82, 4165–4173. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 2011, 12, 323. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]



| Metabolite Categories | Class I | Class II | Number of Metabolites |
|---|---|---|---|
| Primary metabolites (505) | Amino acids and derivatives (188) | Amino acids and derivatives | 188 |
| Nucleotides and derivatives (75) | Nucleotides and derivatives | 75 | |
| Organic acids (108) | Organic acids | 108 | |
| Lipids (134) | Glycerol ester | 14 | |
| PC | 2 | ||
| Sphingolipids | 8 | ||
| LPC | 12 | ||
| LPE | 24 | ||
| Free fatty acids | 74 | ||
| Secondary metabolites (1372) | Alkaloids (117) | Pyridine alkaloids | 6 |
| Phenolamine | 17 | ||
| Quinoline alkaloids | 7 | ||
| Alkaloids | 61 | ||
| Isoquinoline alkaloids | 5 | ||
| Plumerane | 18 | ||
| Piperidine alkaloids | 3 | ||
| Flavonoids (487) | Anthocyanidins | 5 | |
| Chalcones | 15 | ||
| Flavanols | 38 | ||
| Flavanones | 46 | ||
| Flavanonols | 10 | ||
| Flavones | 200 | ||
| Flavonols | 139 | ||
| Isoflavones | 12 | ||
| Other Flavonoids | 22 | ||
| Phenolic acids (394) | Phenolic acids | 394 | |
| Quinones (17) | Anthraquinone | 10 | |
| Quinones | 7 | ||
| Lignans and Coumarins (91) | Lignans | 49 | |
| Coumarins | 42 | ||
| Tannins (37) | Tannin | 20 | |
| Proanthocyanidins | 17 | ||
| Terpenoids (227) | Sesquiterpenoids | 58 | |
| Monoterpenoids | 44 | ||
| Ditepenoids | 34 | ||
| Triterpene | 88 | ||
| Terpene | 3 | ||
| Steroids (2) | Steroid | 2 | |
| Others (271) | Others (271) | Alcohol compounds | 10 |
| Lactones | 10 | ||
| Others | 96 | ||
| Aldehyde compounds | 14 | ||
| Chromone | 11 | ||
| Saccharides | 79 | ||
| Ketone compounds | 17 | ||
| Vitamin | 29 | ||
| Stilbene | 5 |
| Compounds | Structure | MN Fold_Change | NQ Fold_Change |
|---|---|---|---|
| 2-phenylethanol | 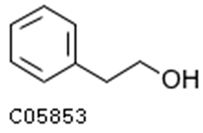 | 1.82 | 1.65 |
| 1-o-p-coumaroyl-β-d-glucose |  | 1.86 | 1.93 |
| 2-hydroxy-3-phenylpropanoic acid | 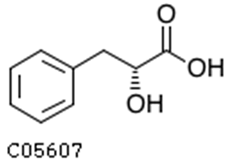 | 6.39 | 1.61 |
| 3-(4-hydroxyphenyl)-propionic acid | 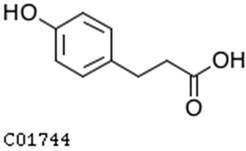 | 2.07 | 1.59 |
| 3-o-feruloylquinic acid-o-glucoside | - | 1538.86 | 1.83 |
| Compounds | cpd_ID | Formula | Exact Mass | Mol Weight | Structure |
|---|---|---|---|---|---|
| acteoside | C10501 | C29H36O15 | 624.2054 | 624.5871 | 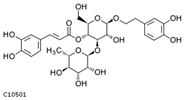 |
| 1,3,6-tri-o-galloyl-β-d-glucose | C17458 | C27H24O18 | 636.0963 | 636.4687 | 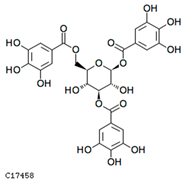 |
| picein | C10720 | C14H18O7 | 298.1053 | 298.2885 | 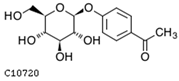 |
| 1-o-caffeoyl-β-d-glucose* | C10433 | C15H18O9 | 342.0951 | 342.298 |  |
| glucovanillin | C19808 | C14H18O8 | 314.1002 | 314.2879 |  |
| 1-o-feruloyl-β-d-glucose | C17759 | C16H20O9 | 356.1107 | 356.3246 | 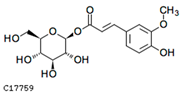 |
| syringin | C01533 | C17H24O9 | 372.142 | 372.3671 | 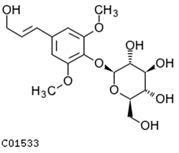 |
| salidroside | C06046 | C14H20O7 | 300.1209 | 300.3044 | 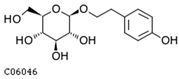 |
| arbutin | C06186 | C12H16O7 | 272.0896 | 272.2512 | 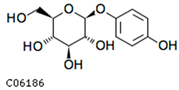 |
| 1,6-di-o-galloyl-β-d-glucose | C04101 | C20H20O14 | 484.0853 | 484.3644 | 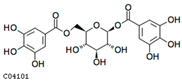 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, X.; Gong, F.; Dong, J.; Lin, X.; Cao, K.; Xu, H.; Zhou, X. Abscisic Acid Affects Phenolic Acid Content to Increase Tolerance to UV-B Stress in Rhododendron chrysanthum Pall. Int. J. Mol. Sci. 2024, 25, 1234. https://doi.org/10.3390/ijms25021234
Zhou X, Gong F, Dong J, Lin X, Cao K, Xu H, Zhou X. Abscisic Acid Affects Phenolic Acid Content to Increase Tolerance to UV-B Stress in Rhododendron chrysanthum Pall. International Journal of Molecular Sciences. 2024; 25(2):1234. https://doi.org/10.3390/ijms25021234
Chicago/Turabian StyleZhou, Xiangru, Fushuai Gong, Jiawei Dong, Xiaoru Lin, Kun Cao, Hongwei Xu, and Xiaofu Zhou. 2024. "Abscisic Acid Affects Phenolic Acid Content to Increase Tolerance to UV-B Stress in Rhododendron chrysanthum Pall." International Journal of Molecular Sciences 25, no. 2: 1234. https://doi.org/10.3390/ijms25021234
APA StyleZhou, X., Gong, F., Dong, J., Lin, X., Cao, K., Xu, H., & Zhou, X. (2024). Abscisic Acid Affects Phenolic Acid Content to Increase Tolerance to UV-B Stress in Rhododendron chrysanthum Pall. International Journal of Molecular Sciences, 25(2), 1234. https://doi.org/10.3390/ijms25021234






