Colonic Epithelial Permeability to Ions Is Restored after Vedolizumab Treatment and May Predict Clinical Response in Inflammatory Bowel Disease Patients
Abstract
1. Introduction
2. Results
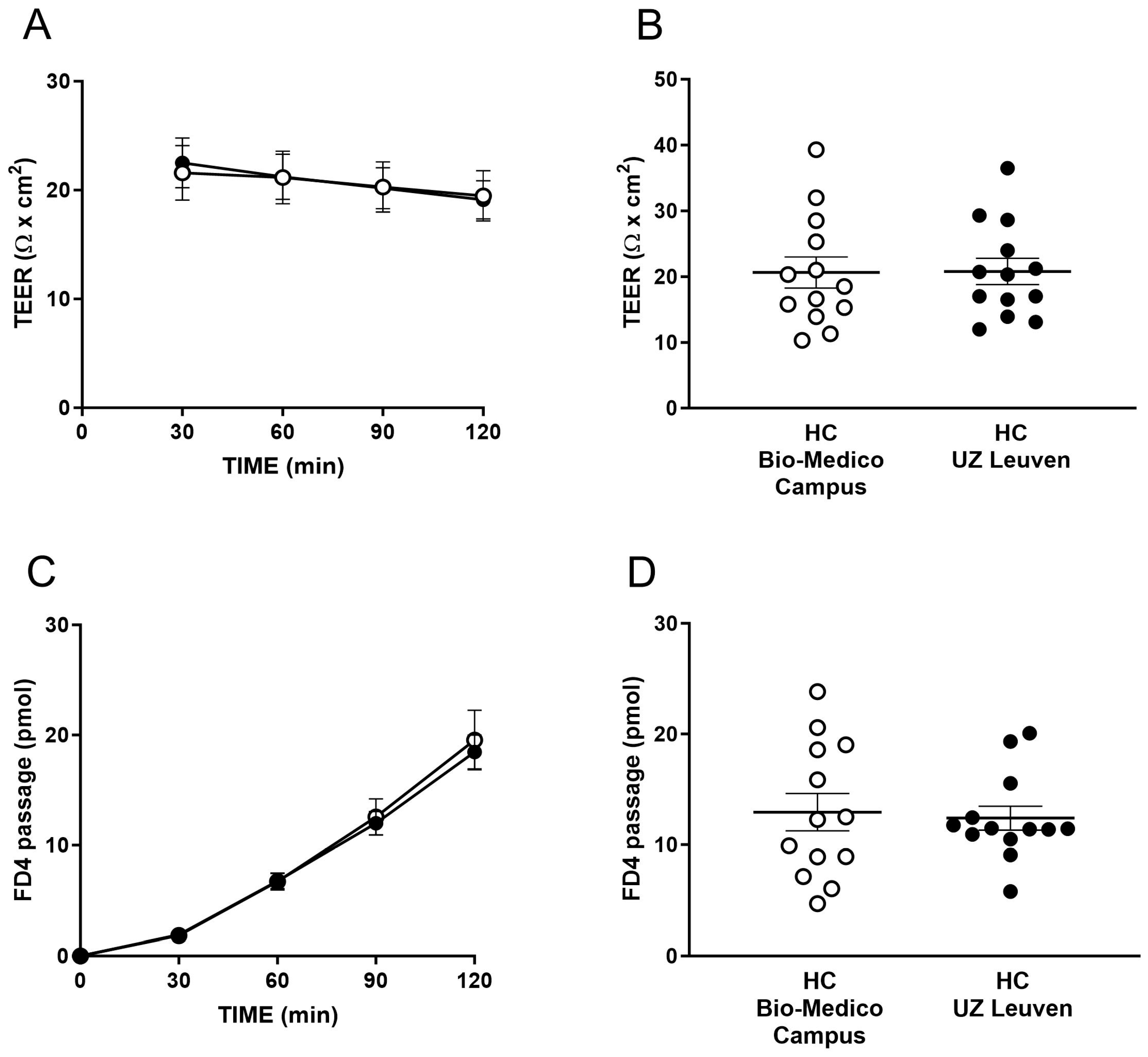
2.1. Study Population
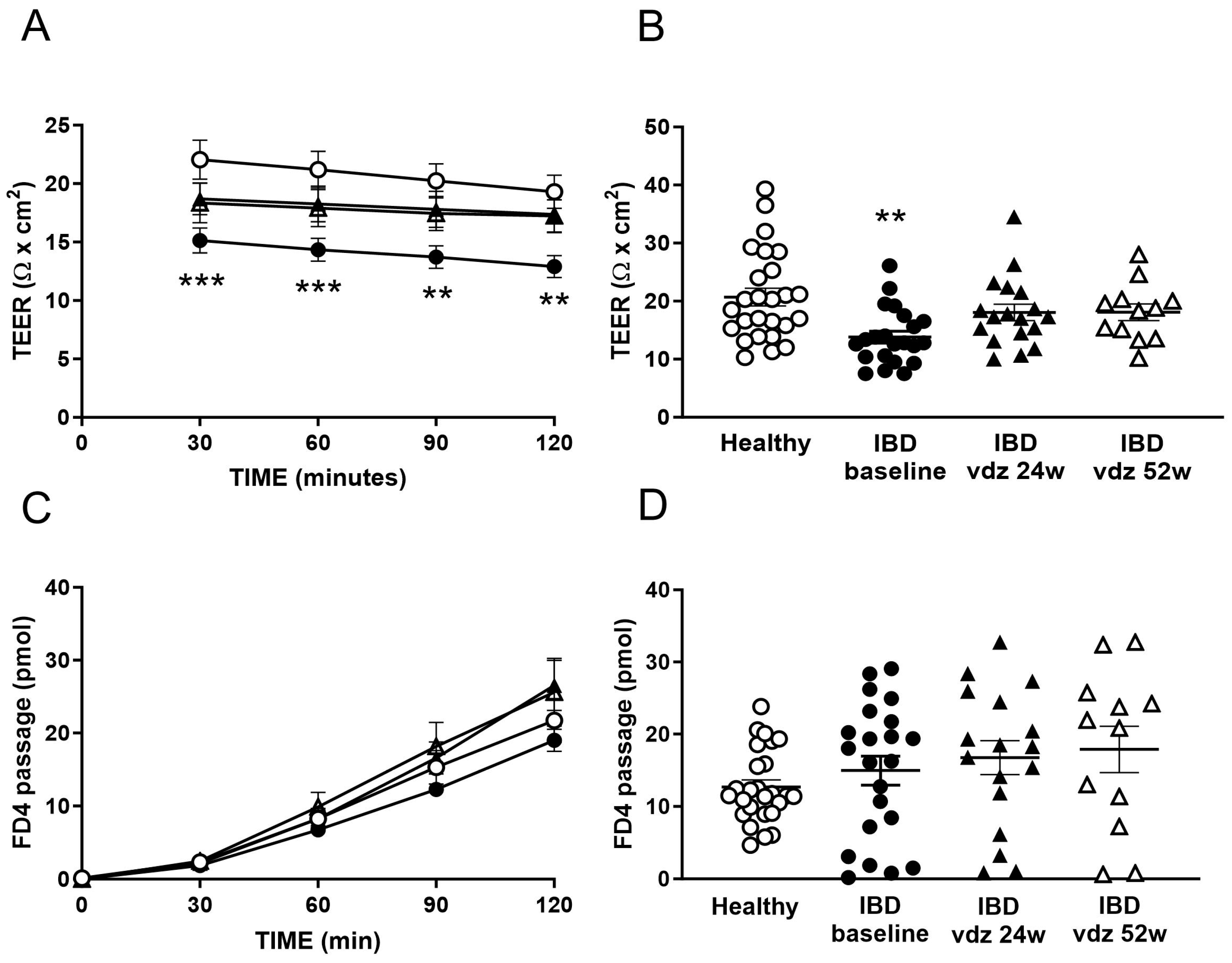
2.2. The Colonic Epithelium of IBD Patients Presented an Increased Permeability to Ions
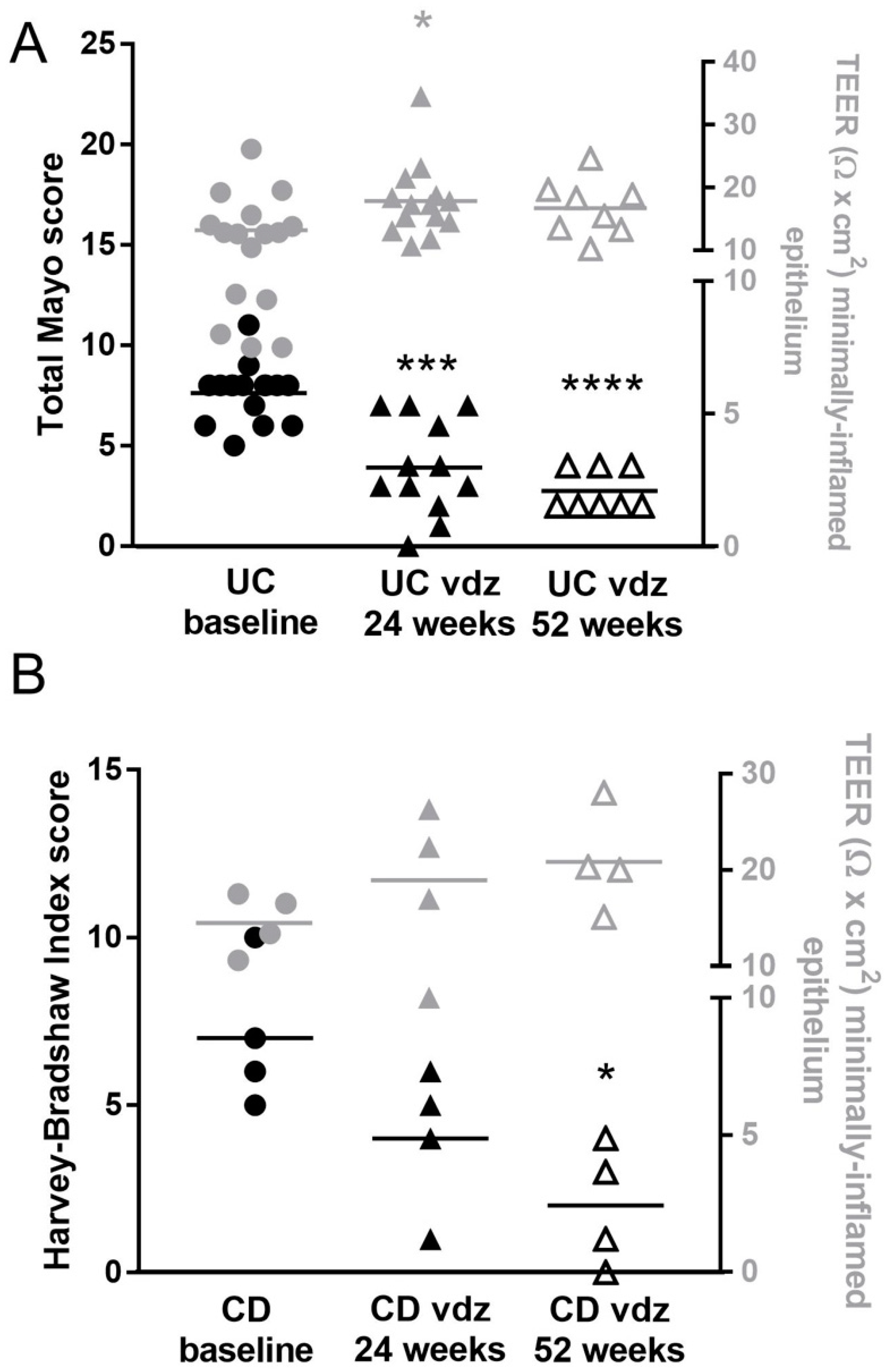
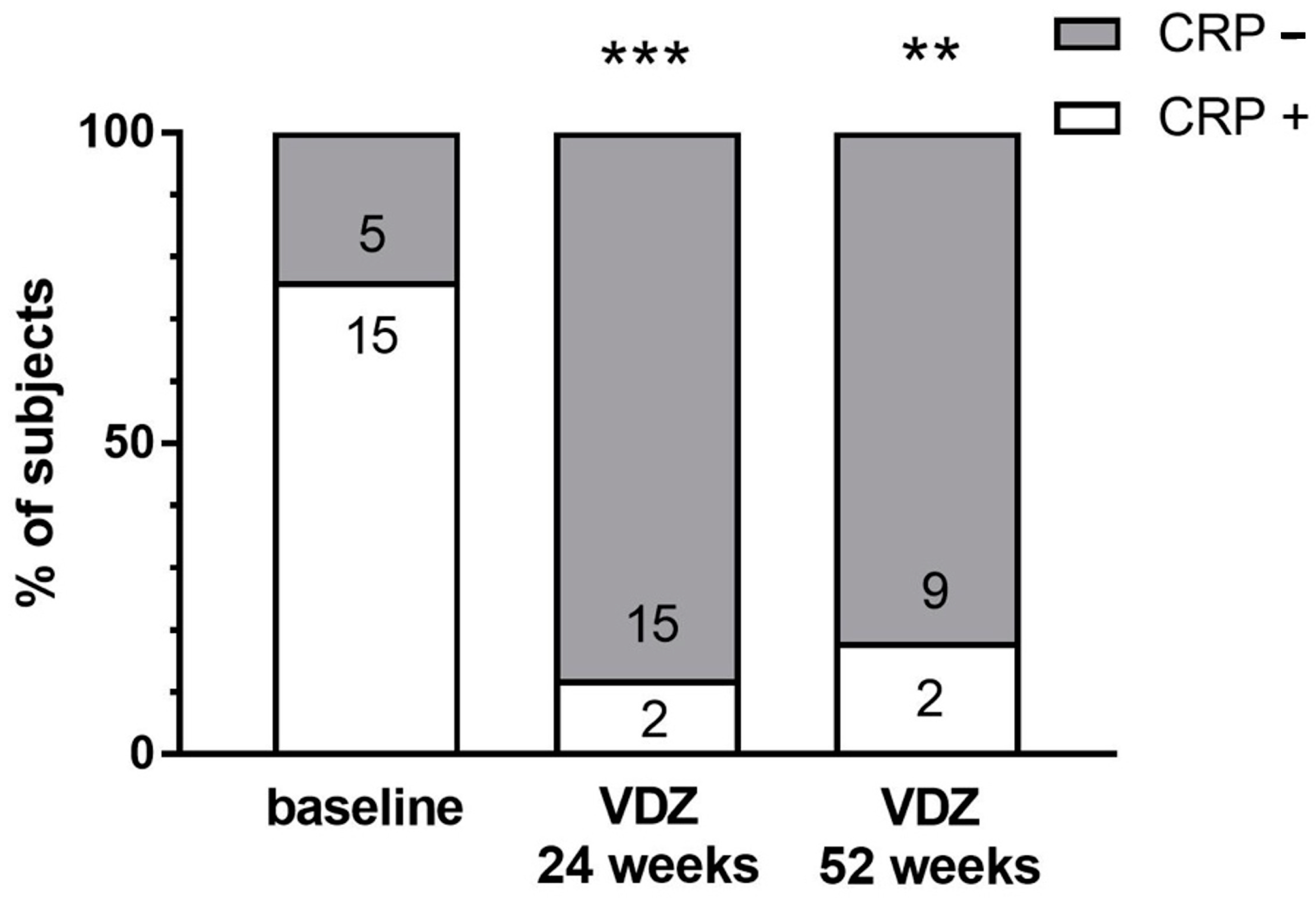
2.3. Clinical Outcomes of Vedolizumab Therapy
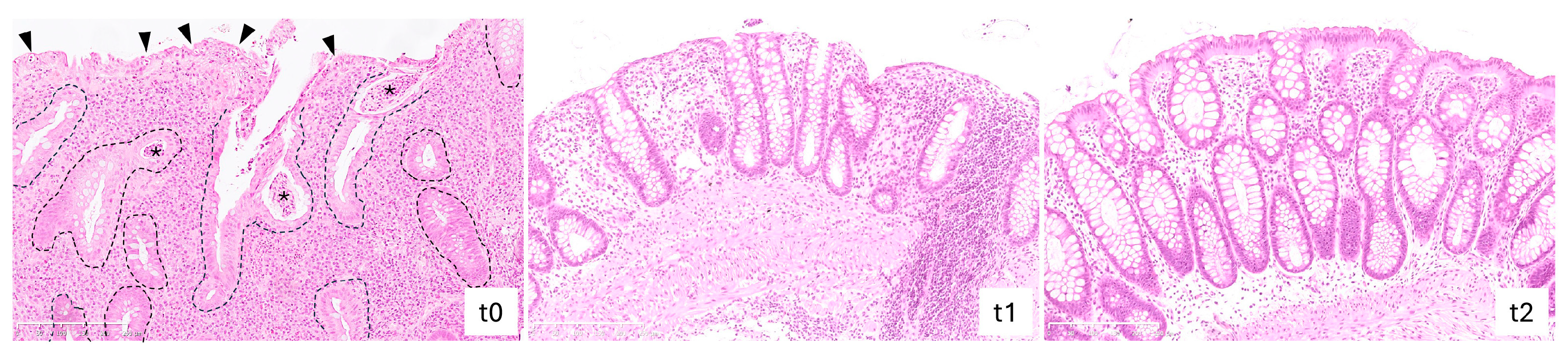
2.4. Vedolizumab Treatment Reduced Histological Grading of Inflammatory Activity in IBD
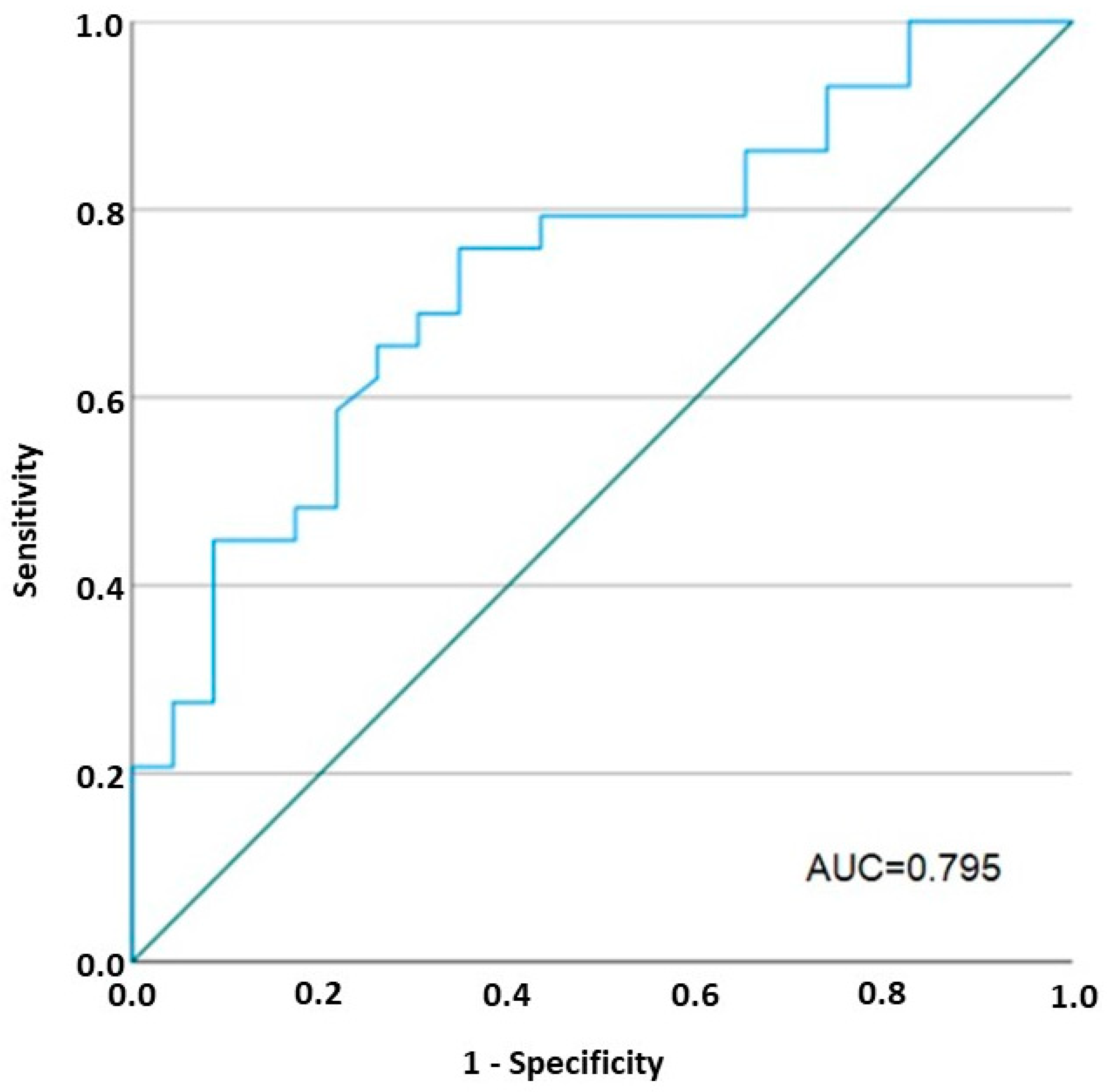
2.5. Vedolizumab Restored the Increased Permeability to Ions in IBD
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Clinical Evaluation
4.3. Collection of Colonic Biopsies
4.4. Histological Evaluation
4.5. Assessment of the Epithelial Barrier Function
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fiocchi, C. Inflammatory bowel disease pathogenesis: Where are we? J. Gastroenterol. Hepatol. 2015, 30 (Suppl. S1), 12–18. [Google Scholar] [CrossRef]
- Pastorelli, L.; De Salvo, C.; Mercado, J.R.; Vecchi, M.; Pizarro, T.T. Central role of the gut epithelial barrier in the pathogenesis of chronic intestinal inflammation: Lessons learned from animal models and human genetics. Front. Immunol. 2013, 4, 280. [Google Scholar] [CrossRef]
- Pearson, A.D.; Eastham, E.J.; Laker, M.F.; Craft, A.W.; Nelson, R. Intestinal permeability in children with Crohn’s disease and coeliac disease. Br. Med. J. (Clin. Res. Ed.) 1982, 285, 20–21. [Google Scholar] [CrossRef]
- Barmeyer, C.; Fromm, M.; Schulzke, J.D. Active and passive involvement of claudins in the pathophysiology of intestinal inflammatory diseases. Pflugers Arch. 2017, 469, 15–26. [Google Scholar] [CrossRef]
- Kjaergaard, S.; Damm, M.M.B.; Chang, J.; Riis, L.B.; Rasmussen, H.B.; Hytting-Andreasen, R.; Krug, S.M.; Schulzke, J.D.; Bindslev, N.; Hansen, M.B. Altered Structural Expression and Enzymatic Activity Parameters in Quiescent Ulcerative Colitis: Are These Potential Normalization Criteria? Int. J. Mol. Sci. 2020, 21, 1887. [Google Scholar] [CrossRef]
- Lee, S.H. Intestinal permeability regulation by tight junction: Implication on inflammatory bowel diseases. Intest. Res. 2015, 13, 11–18. [Google Scholar] [CrossRef]
- Schmitz, H.; Barmeyer, C.; Fromm, M.; Runkel, N.; Foss, H.D.; Bentzel, C.J.; Riecken, E.O.; Schulzke, J.D. Altered tight junction structure contributes to the impaired epithelial barrier function in ulcerative colitis. Gastroenterology 1999, 116, 301–309. [Google Scholar] [CrossRef]
- Gori, M.; Altomare, A.; Cocca, S.; Solida, E.; Ribolsi, M.; Carotti, S.; Rainer, A.; Francesconi, M.; Morini, S.; Cicala, M.; et al. Palmitic Acid Affects Intestinal Epithelial Barrier Integrity and Permeability In Vitro. Antioxidants 2020, 9, 417. [Google Scholar] [CrossRef]
- Hering, N.A.; Fromm, M.; Schulzke, J.D. Determinants of colonic barrier function in inflammatory bowel disease and potential therapeutics. J. Physiol. 2012, 590, 1035–1044. [Google Scholar] [CrossRef]
- Laukoetter, M.G.; Nava, P.; Nusrat, A. Role of the intestinal barrier in inflammatory bowel disease. World J. Gastroenterol. 2008, 14, 401–407. [Google Scholar] [CrossRef]
- Sandle, G.I.; Higgs, N.; Crowe, P.; Marsh, M.N.; Venkatesan, S.; Peters, T.J. Cellular basis for defective electrolyte transport in inflamed human colon. Gastroenterology 1990, 99, 97–105. [Google Scholar] [CrossRef]
- Zeissig, S.; Burgel, N.; Gunzel, D.; Richter, J.; Mankertz, J.; Wahnschaffe, U.; Kroesen, A.J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn’s disease. Gut 2007, 56, 61–72. [Google Scholar] [CrossRef]
- Hu, J.E.; Weiss, F.; Bojarski, C.; Branchi, F.; Schulzke, J.D.; Fromm, M.; Krug, S.M. Expression of tricellular tight junction proteins and the paracellular macromolecule barrier are recovered in remission of ulcerative colitis. BMC Gastroenterol. 2021, 21, 141. [Google Scholar] [CrossRef] [PubMed]
- Soderholm, J.D.; Peterson, K.H.; Olaison, G.; Franzen, L.E.; Westrom, B.; Magnusson, K.E.; Sjodahl, R. Epithelial permeability to proteins in the noninflamed ileum of Crohn’s disease? Gastroenterology 1999, 117, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Yakymenko, O.; Schoultz, I.; Gullberg, E.; Strom, M.; Almer, S.; Wallon, C.; Wang, A.; Keita, A.V.; Campbell, B.J.; McKay, D.M.; et al. Infliximab restores colonic barrier to adherent-invasive E. coli in Crohn’s disease via effects on epithelial lipid rafts. Scand. J. Gastroenterol. 2018, 53, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Katinios, G.; Casado-Bedmar, M.; Walter, S.A.; Vicario, M.; Gonzalez-Castro, A.M.; Bednarska, O.; Soderholm, J.D.; Hjortswang, H.; Keita, A.V. Increased Colonic Epithelial Permeability and Mucosal Eosinophilia in Ulcerative Colitis in Remission Compared With Irritable Bowel Syndrome and Health. Inflamm. Bowel Dis. 2020, 26, 974–984. [Google Scholar] [CrossRef]
- Wallon, C.; Persborn, M.; Jonsson, M.; Wang, A.; Phan, V.; Lampinen, M.; Vicario, M.; Santos, J.; Sherman, P.M.; Carlson, M.; et al. Eosinophils express muscarinic receptors and corticotropin-releasing factor to disrupt the mucosal barrier in ulcerative colitis. Gastroenterology 2011, 140, 1597–1607. [Google Scholar] [CrossRef]
- Danese, S.; Panes, J. Development of drugs to target interactions between leukocytes and endothelial cells and treatment algorithms for inflammatory bowel diseases. Gastroenterology 2014, 147, 981–989. [Google Scholar] [CrossRef]
- Mosli, M.H.; Feagan, B.G. Vedolizumab for Crohn’s disease. Expert. Opin. Biol. Ther. 2013, 13, 455–463. [Google Scholar] [CrossRef]
- Bryant, R.V.; Sandborn, W.J.; Travis, S.P. Introducing vedolizumab to clinical practice: Who, when, and how? J. Crohns Colitis 2015, 9, 356–366. [Google Scholar] [CrossRef]
- Satsangi, J.; Silverberg, M.S.; Vermeire, S.; Colombel, J.F. The Montreal classification of inflammatory bowel disease: Controversies, consensus, and implications. Gut 2006, 55, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.S.; Camilleri, M.; Eckert, D.J.; Busciglio, I.; Burton, D.D.; Ryks, M.; Wong, B.S.; Lamsam, J.; Singh, R.; Zinsmeister, A.R. Urine sugars for in vivo gut permeability: Validation and comparisons in irritable bowel syndrome-diarrhea and controls. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, G919–G928. [Google Scholar] [CrossRef] [PubMed]
- Thomson, A.; Smart, K.; Somerville, M.S.; Lauder, S.N.; Appanna, G.; Horwood, J.; Sunder Raj, L.; Srivastava, B.; Durai, D.; Scurr, M.J.; et al. The Ussing chamber system for measuring intestinal permeability in health and disease. BMC Gastroenterol. 2019, 19, 98. [Google Scholar] [CrossRef] [PubMed]
- Vanuytsel, T.; Tack, J.; Farre, R. The Role of Intestinal Permeability in Gastrointestinal Disorders and Current Methods of Evaluation. Front. Nutr. 2021, 8, 717925. [Google Scholar] [CrossRef] [PubMed]
- Rath, T.; Atreya, R.; Bodenschatz, J.; Uter, W.; Geppert, C.E.; Vitali, F.; Fischer, S.; Waldner, M.J.; Colombel, J.F.; Hartmann, A.; et al. Intestinal Barrier Healing Is Superior to Endoscopic and Histologic Remission for Predicting Major Adverse Outcomes in Inflammatory Bowel Disease: The Prospective ERIca Trial. Gastroenterology 2022, 64, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Leong, R.W.; Wasinger, V.C.; Ip, M.; Yang, M.; Phan, T.G. Impaired Intestinal Permeability Contributes to Ongoing Bowel Symptoms in Patients With Inflammatory Bowel Disease and Mucosal Healing. Gastroenterology 2017, 153, 723–731.e1. [Google Scholar] [CrossRef] [PubMed]
- Karstensen, J.G.; Saftoiu, A.; Brynskov, J.; Hendel, J.; Klausen, P.; Cartana, T.; Klausen, T.W.; Riis, L.B.; Vilmann, P. Confocal laser endomicroscopy: A novel method for prediction of relapse in Crohn’s disease. Endoscopy 2016, 48, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Kiesslich, R.; Duckworth, C.A.; Moussata, D.; Gloeckner, A.; Lim, L.G.; Goetz, M.; Pritchard, D.M.; Galle, P.R.; Neurath, M.F.; Watson, A.J. Local barrier dysfunction identified by confocal laser endomicroscopy predicts relapse in inflammatory bowel disease. Gut 2012, 61, 1146–1153. [Google Scholar] [CrossRef] [PubMed]
- Neurath, M.F. Confocal laser endomicroscopy for functional barrier imaging in Crohn’s disease. Endoscopy 2016, 48, 319–320. [Google Scholar] [CrossRef]
- Rusticeanu, M.; Zimmer, V.; Lammert, F. Visualising and quantifying intestinal permeability -where do we stand. Ann. Hepatol. 2021, 23, 100266. [Google Scholar] [CrossRef]
- Turcotte, J.F.; Wong, K.; Mah, S.J.; Dieleman, L.A.; Kao, D.; Kroeker, K.; Claggett, B.; Saltzman, J.R.; Wine, E.; Fedorak, R.N.; et al. Increased epithelial gaps in the small intestine are predictive of hospitalization and surgery in patients with inflammatory bowel disease. Clin. Transl. Gastroenterol. 2012, 3, e19. [Google Scholar] [CrossRef] [PubMed]
- Farre, R.; Vicario, M. Abnormal Barrier Function in Gastrointestinal Disorders. Handb. Exp. Pharmacol. 2017, 239, 193–217. [Google Scholar] [CrossRef]
- Amasheh, M.; Fromm, A.; Krug, S.M.; Amasheh, S.; Andres, S.; Zeitz, M.; Fromm, M.; Schulzke, J.D. TNFalpha-induced and berberine-antagonized tight junction barrier impairment via tyrosine kinase, Akt and NFkappaB signaling. J. Cell Sci. 2010, 123, 4145–4155. [Google Scholar] [CrossRef] [PubMed]
- Feagan, B.G.; Rutgeerts, P.; Sands, B.E.; Hanauer, S.; Colombel, J.F.; Sandborn, W.J.; Van Assche, G.; Axler, J.; Kim, H.J.; Danese, S.; et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N. Engl. J. Med. 2013, 369, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, A.; Chanez-Paredes, S.D.; Haest, X.; Turner, J.R. Paracellular permeability and tight junction regulation in gut health and disease. Nat. Rev. Gastroenterol. Hepatol. 2023, 20, 417–432. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Feagan, B.G.; Rutgeerts, P.; Hanauer, S.; Colombel, J.F.; Sands, B.E.; Lukas, M.; Fedorak, R.N.; Lee, S.; Bressler, B.; et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease. N. Engl. J. Med. 2013, 369, 711–721. [Google Scholar] [CrossRef]
- Takatsu, N.; Hisabe, T.; Higashi, D.; Ueki, T.; Matsui, T. Vedolizumab in the Treatment of Ulcerative Colitis: An Evidence-Based Review of Safety, Efficacy, and Place of Therapy. Core Evid. 2020, 15, 7–20. [Google Scholar] [CrossRef]
- Zeissig, S.; Bojarski, C.; Buergel, N.; Mankertz, J.; Zeitz, M.; Fromm, M.; Schulzke, J.D. Downregulation of epithelial apoptosis and barrier repair in active Crohn’s disease by tumour necrosis factor alpha antibody treatment. Gut 2004, 53, 1295–1302. [Google Scholar] [CrossRef]
- Maaser, C.; Sturm, A.; Vavricka, S.R.; Kucharzik, T.; Fiorino, G.; Annese, V.; Calabrese, E.; Baumgart, D.C.; Bettenworth, D.; Borralho Nunes, P.; et al. ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J. Crohns Colitis 2019, 13, 144–164. [Google Scholar] [CrossRef]
- Altomare, A.; Gori, M.; Cocca, S.; Carotti, S.; Francesconi, M.; Ribolsi, M.; Emerenziani, S.; Perrone, G.; Morini, S.; Cicala, M.; et al. Impaired Colonic Contractility and Intestinal Permeability in Symptomatic Uncomplicated Diverticular Disease. J. Neurogastroenterol. Motil. 2021, 27, 292–301. [Google Scholar] [CrossRef]
- Vanheel, H.; Vicario, M.; Vanuytsel, T.; Van Oudenhove, L.; Martinez, C.; Keita, A.V.; Pardon, N.; Santos, J.; Soderholm, J.D.; Tack, J.; et al. Impaired duodenal mucosal integrity and low-grade inflammation in functional dyspepsia. Gut 2014, 63, 262–271. [Google Scholar] [CrossRef] [PubMed]

| UC | CD | Healthy Controls | |
|---|---|---|---|
| Age, media (range) | 48 (22–81) | 47 (25–76) | 51 (41–69) |
| Gender | 7 women, 12 men | 2 women, 2 men | 13 women, 13 men |
| Current smokers, n (%) | 4 (21%) | 0 (0%) | 6 (23%) |
| Disease duration (mean months) | 32 (6–91) | 58 (52–61) | N/A |
| Location and behavior * | E2 11/19 (42.1%) E3 8/19 (57.9%) | L2 B1 2/4 (50%) L3 B1 2/4 (50%) | N/A |
| MES, median (range) | 2.5 (2–3) | N/A | N/A |
| Total Mayo Score, median (range) | 7.57 (5–11) | N/A | N/A |
| HBI score, median (range) | N/A | 7 (5–10) | N/A |
| SES-CD, median (range) | N/A | 7 (4–16) | N/A |
| Previous anti-inflammatory medication | Naïve to anti-TNFα (n = 5) Refractory to anti-TNFα (n = 14) | Naïve to anti-TNFα (n = 3) Refractory to anti-TNFα (n = 1) | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cicala, M.; Gori, M.; Balestrieri, P.; Altomare, A.; Tullio, A.; Di Cola, S.; Dejongh, S.; Graziani, M.G.; Pagnini, C.; Carotti, S.; et al. Colonic Epithelial Permeability to Ions Is Restored after Vedolizumab Treatment and May Predict Clinical Response in Inflammatory Bowel Disease Patients. Int. J. Mol. Sci. 2024, 25, 5817. https://doi.org/10.3390/ijms25115817
Cicala M, Gori M, Balestrieri P, Altomare A, Tullio A, Di Cola S, Dejongh S, Graziani MG, Pagnini C, Carotti S, et al. Colonic Epithelial Permeability to Ions Is Restored after Vedolizumab Treatment and May Predict Clinical Response in Inflammatory Bowel Disease Patients. International Journal of Molecular Sciences. 2024; 25(11):5817. https://doi.org/10.3390/ijms25115817
Chicago/Turabian StyleCicala, Michele, Manuele Gori, Paola Balestrieri, Annamaria Altomare, Alessandro Tullio, Simone Di Cola, Sander Dejongh, Maria Giovanna Graziani, Cristiano Pagnini, Simone Carotti, and et al. 2024. "Colonic Epithelial Permeability to Ions Is Restored after Vedolizumab Treatment and May Predict Clinical Response in Inflammatory Bowel Disease Patients" International Journal of Molecular Sciences 25, no. 11: 5817. https://doi.org/10.3390/ijms25115817
APA StyleCicala, M., Gori, M., Balestrieri, P., Altomare, A., Tullio, A., Di Cola, S., Dejongh, S., Graziani, M. G., Pagnini, C., Carotti, S., Perrone, G., Ribolsi, M., Fiorani, M., Guarino, M. P. L., & Farré, R. (2024). Colonic Epithelial Permeability to Ions Is Restored after Vedolizumab Treatment and May Predict Clinical Response in Inflammatory Bowel Disease Patients. International Journal of Molecular Sciences, 25(11), 5817. https://doi.org/10.3390/ijms25115817










