The Role of the Gap Junction Protein Connexin in Adrenal Gland Tumorigenesis
Abstract
1. Introduction to Connexins
2. Connexins—Tumor Suppressor or Tumor Promoter?
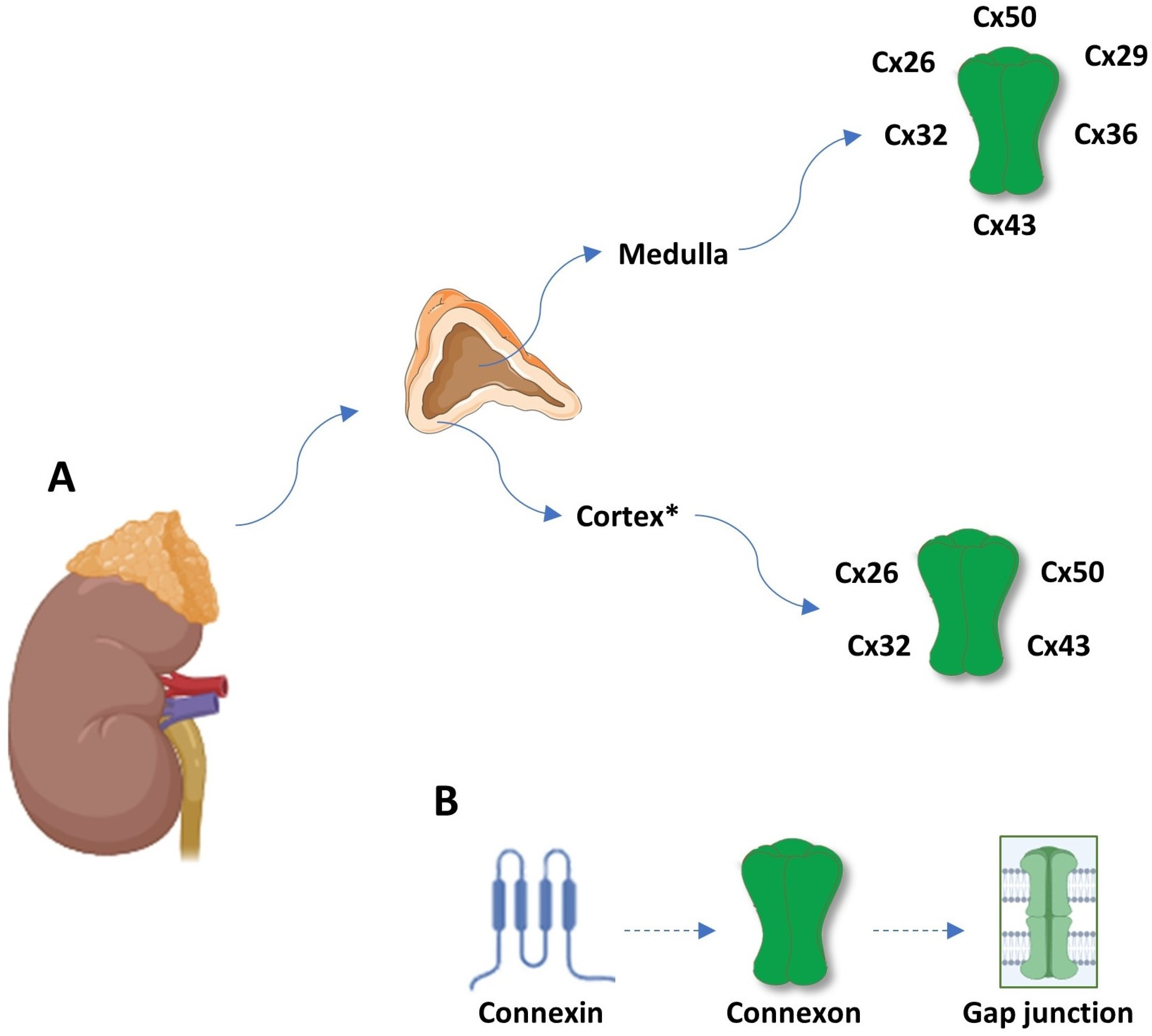
3. Gap Junction-Mediated Intercellular Communication in the Adrenal Gland
4. Gap Junction Intercellular Communication in Adrenal Gland Tumorigenesis
5. Dysregulation of Connexins in Cancer: Therapeutic Opportunities and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Kutova, O.M.; Pospelov, A.D.; Balalaeva, I.V. The Multifaceted Role of Connexins in Tumor Microenvironment Initiation and Maintenance. Biology 2023, 12, 204. [Google Scholar] [CrossRef] [PubMed]
- Nalewajska, M.; Marchelek-Myśliwiec, M.; Opara-Bajerowicz, M.; Dziedziejko, V.; Pawlik, A. Connexins-Therapeutic Targets in Cancers. Int. J. Mol. Sci. 2020, 21, 9119. [Google Scholar] [CrossRef] [PubMed]
- Vinken, M.; Vanhaecke, T.; Papeleu, P.; Snykers, S.; Henkens, T.; Rogiers, V. Connexins and their channels in cell growth and cell death. Cell Signal 2006, 18, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.M.; Gilula, N.B. The gap junction communication channel. Cell 1996, 84, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.S.; Axelsen, L.N.; Sorgen, P.L.; Verma, V.; Delmar, M.; Holstein-Rathlou, N.H. Gap junctions. Compr. Physiol. 2012, 2, 1981–2035. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Xu, C.; Wang, S.; Zhang, Y.; Li, W. The Role of Connexin Hemichannels in Inflammatory Diseases. Biology 2022, 11, 237. [Google Scholar] [CrossRef]
- Defamie, N.; Mesnil, M. The modulation of gap-junctional intercellular communication by lipid rafts. Biochim. Biophys. Acta 2012, 1818, 1866–1869. [Google Scholar] [CrossRef]
- Rodríguez-Sinovas, A.; Sánchez, J.A.; Valls-Lacalle, L.; Consegal, M.; Ferreira-González, I. Connexins in the Heart: Regulation, Function and Involvement in Cardiac Disease. Int. J. Mol. Sci. 2021, 22, 4413. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Oite, T.; Kitamura, M. Gap junctional intercellular communication in the juxtaglomerular apparatus. Am. J. Physiol. Ren. Physiol. 2009, 296, 939–946. [Google Scholar] [CrossRef]
- Grek, C.L.; Rhett, J.M.; Bruce, J.S.; Ghatnekar, G.S.; Yeh, E.S. Connexin 43, breast cancer tumor suppressor: Missed connections? Cancer Lett. 2016, 374, 117–126. [Google Scholar] [CrossRef]
- Su, V.; Lau, A.F. Connexins: Mechanisms regulating protein levels and intercellular communication. FEBS Lett. 2014, 588, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Aasen, T. Connexins: Junctional and non-junctional modulators of proliferation. Cell Tissue Res. 2015, 360, 685–699. [Google Scholar] [CrossRef] [PubMed]
- Graham, S.V.; Jiang, J.X.; Mesnil, M. Connexins and Pannexins: Important Players in Tumorigenesis, Metastasis and Potential Therapeutics. Int. J. Mol. Sci. 2018, 19, 1645. [Google Scholar] [CrossRef] [PubMed]
- Sirnes, S.; Lind, G.E.; Bruun, J.; Fykerud, T.A.; Mesnil, M.; Lothe, R.A.; Rivedal, E.; Kolberg, M.; Leithe, E. Connexins in colorectal cancer pathogenesis. Int. J. Cancer 2015, 137, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ableser, M.J.; Penuela, S.; Lee, J.; Shao, Q.; Laird, D.W. Connexin43 reduces melanoma growth within a keratinocyte microenvironment and during tumorigenesis in vivo. J. Biol. Chem. 2014, 289, 1592–1603. [Google Scholar] [CrossRef] [PubMed]
- Mizdrak, I.; Mizdrak, M.; Racetin, A.; Bošković, B.; Benzon, B.; Durdov, M.G.; Vukojević, K.; Filipović, N. Expression of Connexins 37, 40 and 45, Pannexin 1 and Vimentin in Laryngeal Squamous Cell Carcinomas. Genes 2023, 14, 446. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wang, Z.; He, X.; Jiang, Y.; Pan, R.; Chen, S.; Chen, Y.; Han, Y.; Yu, H.; Zhang, T. GJA1 reverses arsenic-induced EMT via modulating MAPK/ERK signaling pathway. Toxicol. Appl. Pharmacol. 2022, 450, 116138. [Google Scholar] [CrossRef] [PubMed]
- Orellana, V.P.; Tittarelli, A.; Retamal, M.A. Connexins in melanoma: Potential role of Cx46 in its aggressiveness. Pigment. Cell Melanoma Res. 2021, 34, 853–868. [Google Scholar] [CrossRef] [PubMed]
- Stoletov, K.; Strnadel, J.; Zardouzian, E.; Momiyama, M.; Park, F.D.; Kelber, J.A.; Pizzo, D.P.; Hoffman, R.; VandenBerg, S.R.; Klemke, R.L. Role of connexins in metastatic breast cancer and melanoma brain colonization. J. Cell Sci. 2013, 126, 904–913. [Google Scholar] [CrossRef]
- Saito-Katsuragi, M.; Asada, H.; Niizeki, H.; Katoh, F.; Masuzawa, M.; Tsutsumi, M.; Kuniyasu, H.; Ito, A.; Nojima, H.; Miyagawaet, S. Role for connexin 26 in metastasis of human malignant melanoma. Cancer 2007, 110, 1162–1172. [Google Scholar] [CrossRef]
- Bell, C.L.; Murray, S.A. Adrenocortical Gap Junctions and Their Functions. Front. Endocrinol. 2016, 7, 82. [Google Scholar] [CrossRef] [PubMed]
- Hodson, D.J.; Legros, C.; Desarménien, M.G.; Guérineau, N.C. Roles of connexins and pannexins in (neuro)endocrine physiology. Cell Mol. Life Sci. 2015, 72, 2911–2928. [Google Scholar] [CrossRef] [PubMed]
- Friend, D.S.; Gilula, N.B. A distinctive cell contact in the rat adrenal cortex. J. Cell Biol. 1972, 53, 148–163. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.A.; Oyoyo, U.A.; Pharrams, S.Y.; Kumar, N.M.; Gilula, N.B. Characterization of gap junction expression in the adrenal gland. Endocr. Res. 1995, 21, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.A.; Williams, S.Y.; Dillard, C.Y.; Narayanan, S.K.; McCauley, J. Relationship of cytoskeletal filaments to annular gap junction expression in human adrenal cortical tumor cells in culture. Exp. Cell Res. 1997, 234, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.A.; Pharrams, S.Y. Comparison of gap junction expression in the adrenal gland. Microsc. Res. Tech. 1997, 36, 510–519. [Google Scholar] [CrossRef]
- Willenberg, H.S.; Schott, M.; Saeger, W.; Tries, A.; Scherbaum, W.A.; Bornstein, S.R. Expression of connexins in chromaffin cells of normal human adrenals and in benign and malignant pheochromocytomas. Ann. N. Y. Acad. Sci. 2006, 1073, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.A.; Davis, K.; Fishman, L.M.; Bornstein, S.R. Alpha1 Connexin 43 gap junctions are decreased in human adrenocortical tumors. J. Clin. Endocrinol. Metab. 2000, 85, 890–895. [Google Scholar] [CrossRef] [PubMed]
- Colomer, C.; Martin, A.O.; Desarménien, M.G.; Guérineau, N.C. Gap junction-mediated intercellular communication in the adrenal medulla: An additional ingredient of stimulus-secretion coupling regulation. Biochim. Biophys. Acta 2012, 1818, 1937–1951. [Google Scholar] [CrossRef]
- Murray, S.; Davis, K.; Gay, V. ACTH and adrenocortical gap junctions. Microsc. Res. Tech. 2003, 61, 240–246. [Google Scholar] [CrossRef]
- Murray, S.A.; Taylor, F. Dibutyryl cyclic AMP modulation of gap junctions in SW-13 human adrenal cortical tumor cells. Am. J. Anat. 1988, 181, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Oyoyo, U.A.; Shah, U.S.; Murray, S.A. The role of alpha1 (connexin-43) gap junction expression in adrenal cortical cell function. Endocrinology 1997, 138, 5385–5397. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.J.L.; Chan, L. Stability and Turnover of the ACTH Receptor Complex. Front. Endocrinol. 2019, 10, 491. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, C.; Lalli, E. Impact of ACTH Signaling on Transcriptional Regulation of Steroidogenic Genes. Front. Endocrinol. 2016, 7, 24. [Google Scholar] [CrossRef]
- Murray, S.A.; Nickel, B.M.; Gay, V.L. Endocytosis of connexin protein in adrenal cells. Endocr. Res. 2004, 30, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.O.; Mathieu, M.N.; Chevillard, C.; Guérineau, N.C. Gap junctions mediate electrical signaling and ensuing cytosolic Ca2+ increases between chromaffin cells in adrenal slices: A role in catecholamine release. J. Neurosci. 2001, 21, 5397–5405. [Google Scholar] [CrossRef] [PubMed]
- Else, T.; Kim, A.C.; Sabolch, A.; Raymond, V.M.; Kandathil, A.; Caoili, E.M.; Jolly, S.; Miller, B.S.; Giordano, T.J.; Hammer, G.D. Adrenocortical carcinoma. Endocr. Rev. 2014, 35, 282–326. [Google Scholar] [CrossRef]
- Chandrasekar, T.; Goldberg, H.; Klaassen, Z.; Wallis, C.J.D.; Woon, D.T.S.; Herrera-Caceres, J.O.; Kulkarni, G.S.; Fleshner, N.E. The who, when, and why of primary adrenal malignancies: Insights into the epidemiology of a rare clinical entity. Cancer 2019, 125, 1050–1059. [Google Scholar] [CrossRef] [PubMed]
- Mizdrak, M.; Tičinović Kurir, T.; Božić, J. The Role of Biomarkers in Adrenocortical Carcinoma: A Review of Current Evidence and Future Perspectives. Biomedicines 2021, 9, 174. [Google Scholar] [CrossRef]
- Aygun, N.; Uludag, M. Pheochromocytoma and Paraganglioma: From Epidemiology to Clinical Findings. Sisli Etfal Hastan. Tip. Bul. 2020, 54, 159–168. [Google Scholar] [CrossRef]
- Kamibayashi, Y.; Oyamada, Y.; Mori, M.; Oyamada, M. Aberrant expression of gap junction proteins (connexins) is associated with tumor progression during multistage mouse skin carcinogenesis in vivo. Carcinogenesis 1995, 16, 1287–1297. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.K.; An, J.H.; Jeon, K.H.; Choi, S.H.; Cho, Y.M.; Jang, H.C.; Chung, J.H.; Lee, C.H.; Lim, S. Two cases of ectopic adrenocorticotropic hormone syndrome with olfactory neuroblastoma and literature review. Endocr. J. 2008, 55, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Miekus, K.; Czernik, M.; Sroka, J.; Czyz, J.; Madeja, Z. Contact stimulation of prostate cancer cell migration: The role of gap junctional coupling and migration stimulated by heterotypic cell-to-cell contacts in determination of the metastatic phenotype of Dunning rat prostate cancer cells. Biol. Cell 2005, 97, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Nwagwu, C.; Le, D.M.; Yong, V.W.; Song, H.; Couldwell, W.T. Increased invasive capacity of connexin43-overexpressing malignant glioma cells. J. Neurosurg. 2003, 99, 1039–1046. [Google Scholar] [CrossRef]
- Wu, X.; Azizan, E.A.B.; Goodchild, E.; Garg, S.; Hagiyama, M.; Cabrera, C.P.; Fernandes-Rosa, F.L.; Boulkroun, S.; Kuan, J.L.; Tiang, Z.; et al. Somatic mutations of CADM1 in aldosterone-producing adenomas and gap junction-dependent regulation of aldosterone production. Nat. Genet. 2023, 55, 1009–1021. [Google Scholar] [CrossRef] [PubMed]
- Nunes, B.; Pópulo, H.; Lopes, J.M.; Reis, M.; Nascimento, G.; Nascimento, A.G.; Fernandes, J.; Faria, M.; de Carvalho, D.P.; Soares, P.; et al. Connexin Expression in Pituitary Adenomas and the Effects of Overexpression of Connexin 43 in Pituitary Tumor Cell Lines. Genes 2022, 13, 674. [Google Scholar] [CrossRef] [PubMed]
- Bišćanin, A.; Ljubičić, N.; Boban, M.; Baličević, D.; Pavić, I.; Bišćanin, M.M.; Budimir, I.; Dorosulic, Z.; Duvnjak, M. CX43 Expression in Colonic Adenomas and Surrounding Mucosa Is a Marker of Malignant Potential. Anticancer Res. 2016, 36, 5437–5442. [Google Scholar] [CrossRef]
- Avanzo, J.L.; Mesnil, M.; Hernandez-Blazquez, F.J.; da Silva, T.C.; Fukumasu, H.; Mori, C.M.; Yamasaki, H.; Dagli, M.L. Altered expression of connexins in urethane-induced mouse lung adenomas. Life Sci. 2006, 79, 2202–2208. [Google Scholar] [CrossRef] [PubMed]
- Husøy, T.; Knutsen, H.K.; Cruciani, V.; Olstørn, H.B.; Mikalsen, S.O.; Løberg, E.M.; Alexander, J. Connexin 43 is overexpressed in Apc(Min/+)-mice adenomas and colocalises with COX-2 in myofibroblasts. Int. J. Cancer 2005, 116, 351–358. [Google Scholar] [CrossRef]
- Haddad, N.F.; Teodoro, A.J.; Leite de Oliveira, F.; Soares, N.; de Mattos, R.M.; Hecht, F.; Dezonne, R.S.; Vairo, L.; Goldenberg, R.C.; Gomes, F.C.; et al. Lycopene and beta-carotene induce growth inhibition and proapoptotic effects on ACTH-secreting pituitary adenoma cells. PLoS ONE 2013, 8, e62773. [Google Scholar] [CrossRef]
- Maulik, M.; Vasan, L.; Bose, A.; Dutta Chowdhury, S.; Sengupta, N.; Das Sarma, J. Amyloid-β regulates gap junction protein connexin 43 trafficking in cultured primary astrocytes. J. Biol. Chem. 2020, 295, 15097–15111. [Google Scholar] [CrossRef]
- Koulakoff, A.; Mei, X.; Orellana, J.A.; Sáez, J.C.; Giaume, C. Glial connexin expression and function in the context of Alzheimer’s disease. Biochim. Biophys. Acta 2012, 1818, 2048–2057. [Google Scholar] [CrossRef] [PubMed]
- Mei, X.; Ezan, P.; Giaume, C.; Koulakoff, A. Astroglial connexin immunoreactivity is specifically altered at β-amyloid plaques in β-amyloid precursor protein/presenilin1 mice. Neuroscience 2010, 171, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Yi, C.; Koulakoff, A.; Giaume, C. Astroglial Connexins as a Therapeutic Target for Alzheimer’s Disease. Curr. Pharm. Des. 2017, 23, 4958–4968. [Google Scholar] [CrossRef]
- Nagy, J.I.; Li, W.; Hertzberg, E.L.; Marotta, C.A. Elevated connexin43 immunoreactivity at sites of amyloid plaques in Alzheimer’s disease. Brain Res. 1996, 717, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Kajiwara, Y.; Wang, E.; Wang, M.; Sin, W.C.; Brennand, K.J.; Schadt, E.; Naus, C.C.; Buxbaum, J.; Zhang, B. GJA1 (connexin43) is a key regulator of Alzheimer’s disease pathogenesis. Acta Neuropathol. Commun. 2018, 6, 144. [Google Scholar] [CrossRef]
- Oloris, S.C.; Mesnil, M.; Reis, V.N.; Sakai, M.; Matsuzaki, P.; Fonseca Ede, S.; da Silva, T.C.; Avanzo, J.L.; Sinhorini, I.L.; Guerra, J.L.; et al. Hepatic granulomas induced by Schistosoma mansoni in mice deficient for connexin 43 present lower cell proliferation and higher collagen content. Life Sci. 2007, 80, 1228–1235. [Google Scholar] [CrossRef]
- Wu, J.; Gao, M.; Rice, S.G.; Tsang, C.; Beggs, J.; Turner, D.; Li, G.; Yang, B.; Xia, K.; Gao, F.; et al. Gap Junctions Contribute to Ictal/Interictal Genesis in Human Hypothalamic Hamartomas. EBioMedicine 2016, 8, 96–102. [Google Scholar] [CrossRef]
- Chu, P.H.; Yeh, H.I.; Jung, S.M.; Chien, L.Y.; Cheng, N.F.; Wu, H.H.; Chu, J.J.; Hsueh, C.; Lee, Y.S. Irregular connexin43 expressed in a rare cardiac hamartoma containing adipose tissue in the crista terminalis. Virchows Arch. 2004, 444, 383–386. [Google Scholar] [CrossRef]
- Frings, V.G.; Goebeler, M.; Schilling, B.; Kneitz, H. Aberrant cytoplasmic connexin43 expression as a helpful marker in vascular neoplasms. J. Cutan. Pathol. 2021, 48, 1335–1341. [Google Scholar] [CrossRef]
- Simon, A.M.; McWhorter, A.R. Vascular abnormalities in mice lacking the endothelial gap junction proteins connexin37 and connexin40. Dev. Biol. 2002, 251, 206–220. [Google Scholar] [CrossRef]
- Korkiamäki, T.; Ylä-Outinen, H.; Koivunen, J.; Karvonen, S.L.; Peltonen, J. Altered calcium-mediated cell signaling in keratinocytes cultured from patients with neurofibromatosis type 1. Am. J. Pathol. 2002, 160, 1981–1990. [Google Scholar] [CrossRef] [PubMed]
- Mambetisaeva, E.T.; Gire, V.; Evans, W.H. Multiple connexin expression in peripheral nerve, Schwann cells, and Schwannoma cells. J. Neurosci. Res. 1999, 57, 166–175. [Google Scholar] [CrossRef]
- Butler, J.; Dale, N. X-linked Charcot Marie Tooth mutations alter CO2 sensitivity of connexin32 hemichannels. Front. Cell Neurosci. 2023, 17, 1330983. [Google Scholar] [CrossRef]
- Kiszner, G.; Balla, P.; Wichmann, B.; Barna, G.; Baghy, K.; Nemeth, I.B.; Varga, E.; Furi, I.; Toth, B.; Krenacs, T. Exploring Differential Connexin Expression across Melanocytic Tumor Progression Involving the Tumor Microenvironment. Cancers 2019, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Scatolini, M.; Patel, A.; Grosso, E.; Mello-Grand, M.; Ostano, P.; Coppo, R.; Vitiello, M.; Venesio, T.; Zaccagna, A.; Pisacane, A.; et al. GJB5 association with BRAF mutation and survival in cutaneous malignant melanoma. Br. J. Dermatol. 2022, 186, 117–128. [Google Scholar] [CrossRef] [PubMed]
- King, T.J.; Lampe, P.D. Mice deficient for the gap junction protein Connexin32 exhibit increased radiation-induced tumorigenesis associated with elevated mitogen-activated protein kinase (p44/Erk1, p42/Erk2) activation. Carcinogenesis 2004, 25, 669–680. [Google Scholar] [CrossRef]
- Kojima, T.; Srinivas, M.; Fort, A.; Hopperstad, M.; Urban, M.; Hertzberg, E.L.; Mochizuki, Y.; Spray, D.C. TPA induced expression and function of human connexin 26 by post-translational mechanisms in stably transfected neuroblastoma cells. Cell Struct. Funct. 1999, 24, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Carystinos, G.D.; Alaoui-Jamali, M.A.; Phipps, J.; Yen, L.; Batist, G. Upregulation of gap junctional intercellular communication and connexin 43 expression by cyclic-AMP and all-trans-retinoic acid is associated with glutathione depletion and chemosensitivity in neuroblastoma cells. Cancer Chemother. Pharmacol. 2001, 47, 126–132. [Google Scholar] [CrossRef]
- Kim, I.S.; Ganesan, P.; Choi, D.K. Cx43 Mediates Resistance against MPP⁺-Induced Apoptosis in SH-SY5Y Neuroblastoma Cells via Modulating the Mitochondrial Apoptosis Pathway. Int. J. Mol. Sci. 2016, 17, 1819. [Google Scholar] [CrossRef]
- Arnold, J.M.; Phipps, M.W.; Chen, J.; Phipps, J. Cellular sublocalization of Cx43 and the establishment of functional coupling in IMR-32 neuroblastoma cells. Mol. Carcinog. 2005, 42, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Zoidl, G.; Meier, C.; Petrasch-Parwez, E.; Zoidl, C.; Habbes, H.W.; Kremer, M.; Srinivas, M.; Spray, D.C.; Dermietzel, R. Evidence for a role of the N-terminal domain in subcellular localization of the neuronal connexin36 (Cx36). J. Neurosci. Res. 2002, 69, 448–465. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.J.; Li, H.; Zhou, F.H.; Zhang, J.J.; Wang, L.X. Connexin 36 is expressed and associated with zonula occludens-1 protein in PC-12 cells. Gen. Physiol. Biophys. 2007, 26, 33–39. [Google Scholar] [PubMed]
- Potolicchio, I.; Cigliola, V.; Velazquez-Garcia, S.; Klee, P.; Valjevac, A.; Kapic, D.; Cosovic, E.; Lepara, O.; Hadzovic-Dzuvo, A.; Mornjacovic, Z.; et al. Connexin-dependent signaling in neuro-hormonal systems. Biochim. Biophys. Acta 2012, 1818, 1919–1936. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.A.; Nickel, B.M.; Gay, V.L. Gap junctions as modulators of adrenal cortical cell proliferation and steroidogenesis. Mol. Cell. Endocrinol. 2009, 300, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Kandouz, M.; Batist, G. Gap junctions and connexins as therapeutic targets in cancer. Expert. Opin. Ther. Targets 2010, 14, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Bonacquisti, E.E.; Nguyen, J. Connexin 43 (Cx43) in cancer: Implications for therapeutic approaches via gap junctions. Cancer Lett. 2019, 442, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.C.; Verswyvel, H.; Smits, E.; Cordeiro, R.M.; Bogaerts, A.; Lin, A. The pro- and anti-tumoral properties of gap junctions in cancer and their role in therapeutic strategies. Redox Biol. 2022, 57, 102503. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, S.; Zhao, Z.; Zhang, C.; Yang, X.; Wang, Y. Connexin 43 enhances paclitaxel cytotoxicity in colorectal cancer cell lines. Exp. Ther. Med. 2017, 14, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.P.; Hossain, M.Z.; Huang, R.; Gano, J.; Fan, Y.; Boynton, A.L. Connexin 43 (cx43) enhances chemotherapy-induced apoptosis in human glioblastoma cells. Int. J. Cancer 2001, 92, 130–138. [Google Scholar] [CrossRef]
- Mesnil, M.; Piccoli, C.; Yamasaki, H. A tumor suppressor gene, Cx26, also mediates the bystander effect in HeLa cells. Cancer Res. 1997, 57, 2929–2932. [Google Scholar] [PubMed]
- Wang, Y.W.; Wang, Q.; Zhang, S.Z.; Zhang, Y.; Tao, L. Baicalein increases the cytotoxicity of cisplatin by enhancing gap junction intercellular communication. Mol. Med. Rep. 2014, 10, 515–521. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.Y.; Wang, Q.; Ge, H.; Tao, L. Propofol depresses cisplatin cytotoxicity via the inhibition of gap junctions. Mol. Med. Rep. 2016, 13, 4715–4720. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.X.; Penuela, S. Connexin and pannexin channels in cancer. BMC Cell Biol. 2016, 17, 12. [Google Scholar] [CrossRef] [PubMed]
- Yusubalieva, G.M.; Baklaushev, V.P.; Gurina, O.I.; Gulyaev, M.V.; Pirogo, Y.A.; Chekhonin, V.P. Antitumor effects of monoclonal antibodies to connexin 43 extracellular fragment in induced low-differentiated glioma. Bull. Exp. Biol. Med. 2012, 153, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Yulyana, Y.; Endaya, B.B.; Ng, W.H.; Guo, C.M.; Hui, K.M.; Lam, P.Y.; Ho, I.A. Carbenoxolone enhances TRAIL-induced apoptosis through the upregulation of death receptor 5 and inhibition of gap junction intercellular communication in human glioma. Stem Cells Dev. 2013, 22, 1870–1882. [Google Scholar] [CrossRef] [PubMed]
- Desarménien, M.G.; Jourdan, C.; Toutain, B.; Vessières, E.; Hormuzdi, S.G.; Guérineau, N.C. Gap junction signalling is a stress-regulated component of adrenal neuroendocrine stimulus-secretion coupling in vivo. Nat. Commun. 2013, 4, 2938. [Google Scholar] [CrossRef] [PubMed]
- Baklaushev, V.P.; Nukolova, N.N.; Khalansky, A.S.; Gurina, O.I.; Yusubalieva, G.M.; Grinenko, N.P.; Gubskiy, I.L.; Melnikov, P.A.; Kardashova, K.; Kabanov, A.V.; et al. Treatment of glioma by cisplatin-loaded nanogels conjugated with monoclonal antibodies against Cx43 and BSAT1. Drug Deliv. 2015, 22, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Martins-Marques, T.; Rodriguez-Sinovas, A.; Girao, H. Cellular crosstalk in cardioprotection: Where and when do reactive oxygen species play a role? Free Radic. Biol. Med. 2021, 169, 397–409. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, Y.; Wei, S.; Wang, X.; Zhang, J. Effects and Mechanisms of Non-Thermal Plasma-Mediated ROS and Its Applications in Animal Husbandry and Biomedicine. Int. J. Mol. Sci. 2023, 24, 15889. [Google Scholar] [CrossRef]
- Bagati, A.; Hutcherson, T.C.; Koch, Z.; Pechette, J.; Dianat, H.; Higley, C.; Chiu, L.; Song, Y.; Shah, J.; Chazen, E.; et al. Novel combination therapy for melanoma induces apoptosis via a gap junction positive feedback mechanism. Oncotarget 2020, 11, 3443–3458. [Google Scholar] [CrossRef] [PubMed]
- Solan, J.L.; Lampe, P.D. Src Regulation of Cx43 Phosphorylation and Gap Junction Turnover. Biomolecules 2020, 10, 1596. [Google Scholar] [CrossRef] [PubMed]
- Ming, J.; Zhou, Y.; Du, J.; Fan, S.; Pan, B.; Wang, Y.; Fan, L.; Jiang, J. Identification of miR-200a as a novel suppressor of connexin 43 in breast cancer cells. Biosci. Rep. 2015, 35, e00251. [Google Scholar] [CrossRef] [PubMed]
- Naus, C.C.; Giaume, C. Bridging the gap to therapeutic strategies based on connexin/pannexin biology. J. Transl. Med. 2016, 14, 330. [Google Scholar] [CrossRef]
- Schneider, M.; Potthoff, A.L.; Evert, B.O.; Dicks, M.; Ehrentraut, D.; Dolf, A.; Schmidt, E.N.C.; Schäfer, N.; Borger, V.; Pietsch, T.; et al. Inhibition of Intercellular Cytosolic Traffic via Gap Junctions Reinforces Lomustine-Induced Toxicity in Glioblastoma Ind. ependent of MGMT Promoter Methylation Status. Pharmaceuticals 2021, 14, 195. [Google Scholar] [CrossRef]

| Connexin Expression Pattern in the Adrenal Gland | Connexin Expression Pattern of Specific Lesion of the Other Site | References | |
|---|---|---|---|
| Adenoma |
|
| [28,45,46,47,48,49,50] |
| Amyloidosis | Unknown |
| [51,52,53,54,55,56] |
| Congenital adrenal hyperplasia | Unknown | - | - |
| Ganglioneuroma | - | Unknown | - |
| Granuloma | Unknown |
| [57] |
| Hamartoma | Unknown |
| [58,59] |
| Hemangioma | Unknown |
| [60,61] |
| Lipoma | Unknown | Unknown | - |
| Myelolipoma | Unknown | Unknown | - |
| Neurofibroma | Unknown |
| [62] |
| Pheochromocytoma |
| - | [27] |
| Connexin Expression Pattern in the Adrenal Gland | Connexin Expression Pattern of Specific Lesion of the Other Site | References | |
|---|---|---|---|
| Adrenocortical carcinoma |
| - | [27,28] |
| Angiosarcoma | Unknown |
| [60] |
| Leiomyosarcoma | Unknown | Unknown | - |
| Malignant schwannoma | Unknown |
| [63,64] |
| Malignant primary melanoma of adrenal gland | Unknown |
| [65,66] |
| Malignant pheochromocytoma |
| - | [27,67] |
| Neuroblastoma | Unknown |
| [68,69,70,71,72] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mizdrak, M.; Ticinovic Kurir, T.; Mizdrak, I.; Kumric, M.; Krnic, M.; Bozic, J. The Role of the Gap Junction Protein Connexin in Adrenal Gland Tumorigenesis. Int. J. Mol. Sci. 2024, 25, 5399. https://doi.org/10.3390/ijms25105399
Mizdrak M, Ticinovic Kurir T, Mizdrak I, Kumric M, Krnic M, Bozic J. The Role of the Gap Junction Protein Connexin in Adrenal Gland Tumorigenesis. International Journal of Molecular Sciences. 2024; 25(10):5399. https://doi.org/10.3390/ijms25105399
Chicago/Turabian StyleMizdrak, Maja, Tina Ticinovic Kurir, Ivan Mizdrak, Marko Kumric, Mladen Krnic, and Josko Bozic. 2024. "The Role of the Gap Junction Protein Connexin in Adrenal Gland Tumorigenesis" International Journal of Molecular Sciences 25, no. 10: 5399. https://doi.org/10.3390/ijms25105399
APA StyleMizdrak, M., Ticinovic Kurir, T., Mizdrak, I., Kumric, M., Krnic, M., & Bozic, J. (2024). The Role of the Gap Junction Protein Connexin in Adrenal Gland Tumorigenesis. International Journal of Molecular Sciences, 25(10), 5399. https://doi.org/10.3390/ijms25105399








