Evaluation of Flathead Grey Mullets (Mugil cephalus) Immunization and Long-Term Protection against Vibrio harveyi Infection Using Three Different Vaccine Preparations
Abstract
1. Introduction
2. Results
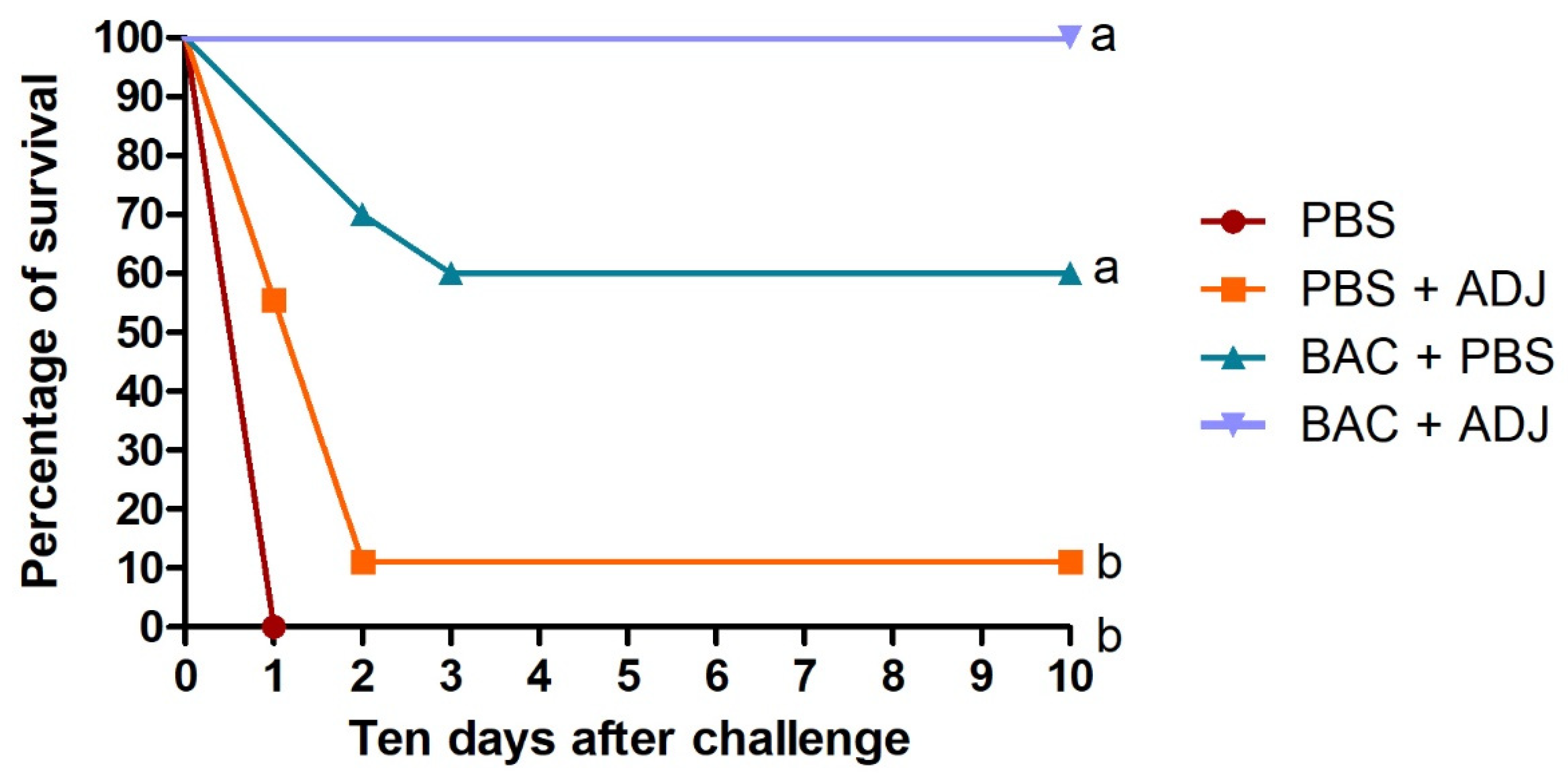
2.1. Fish Mortality Rates Post Challenges with Vibrio harveyi and Bacteriology Analysis
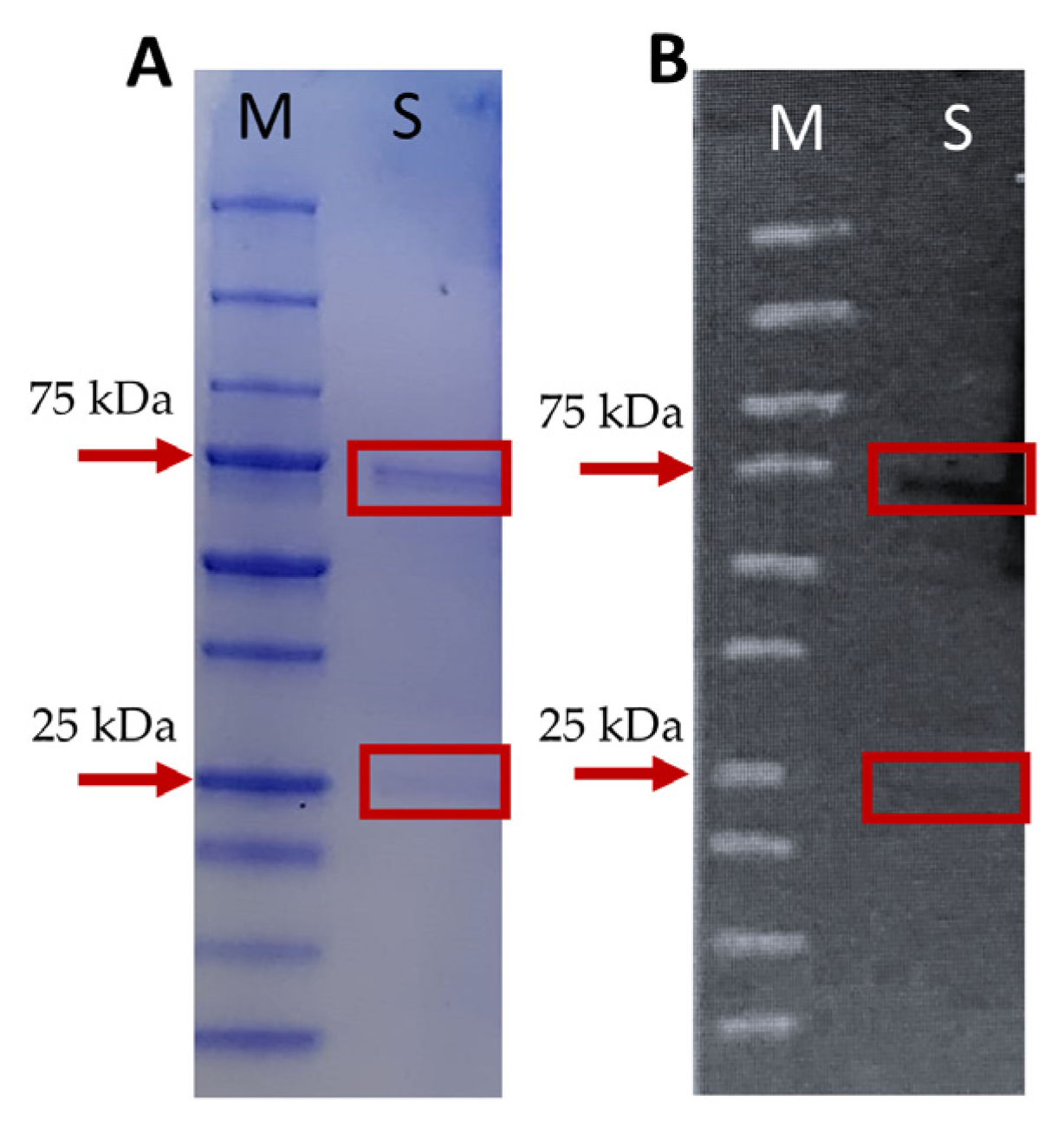
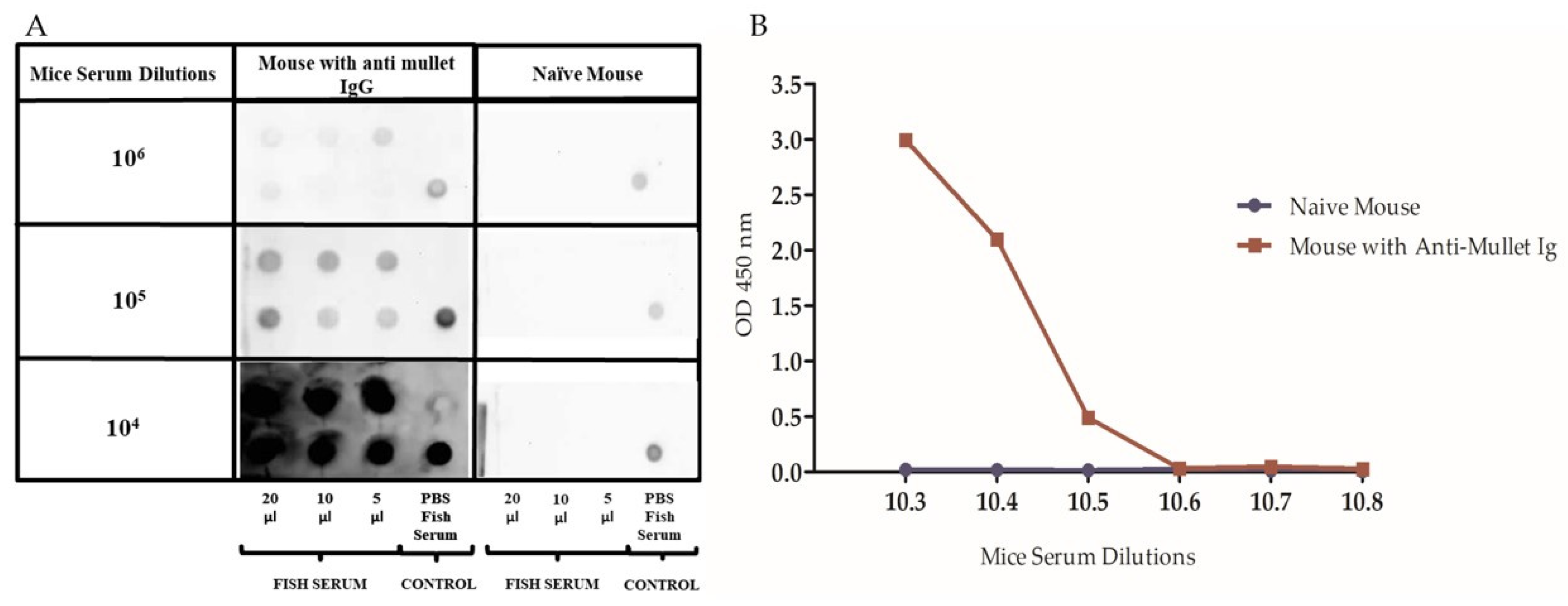
2.2. Production and Standardization of Mice Anti-Mullet Antibodies
2.3. ELISA Analysis of Mullet Anti-Vibrio harveyi Antibodies in Immunized Fish
2.4. ELISA Analysis to Detect Cross Reactivity against Other Vibrio spp.
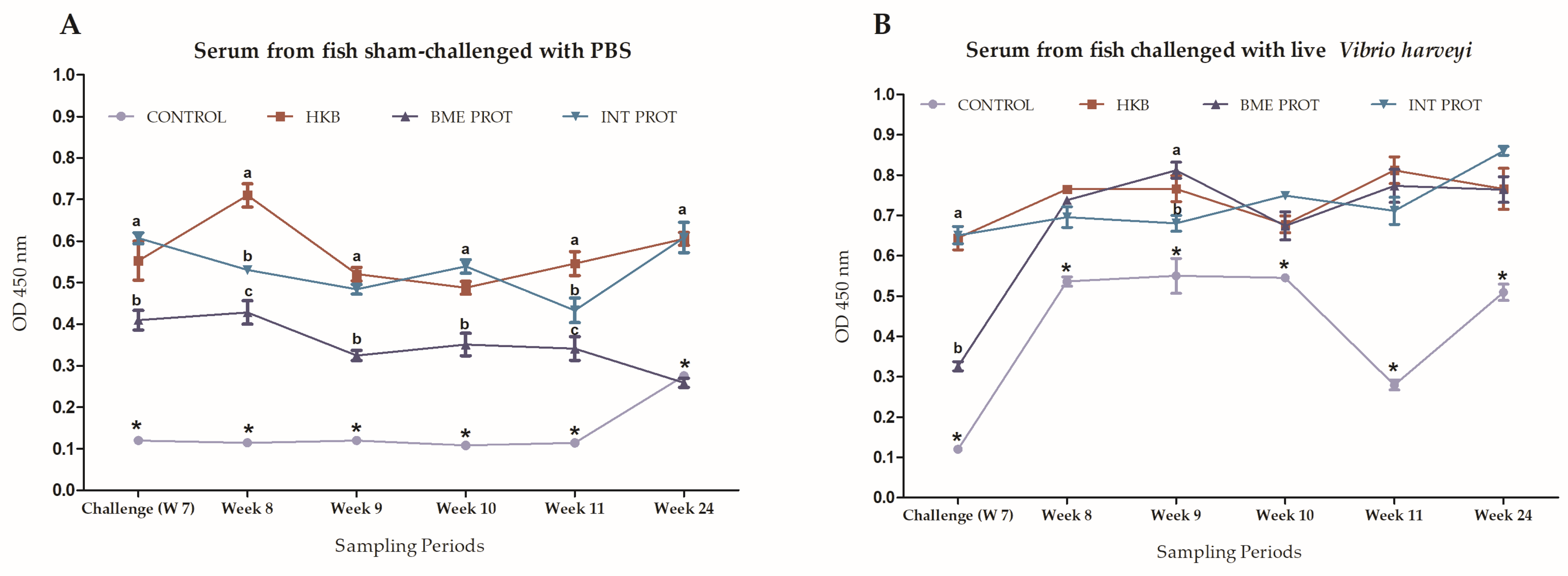
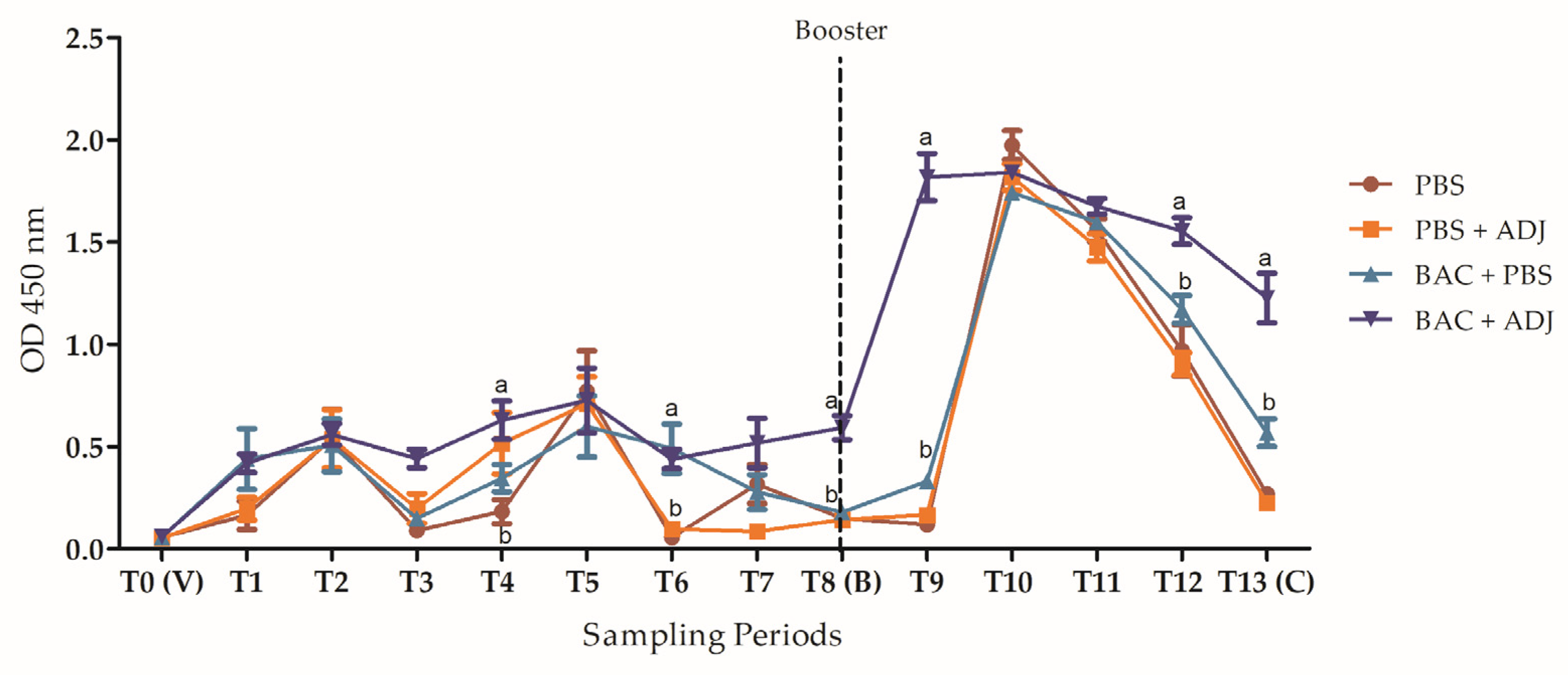
2.5. Evaluation of Heath-Killed Bacteria’s Long-Term Protection, and Antibody Production
3. Discussion
4. Materials and Methods
4.1. Preparation of Three Different Vaccines
4.1.1. Isolation and Culture of Vibrio harveyi Strain RS2016
4.1.2. Heat-Killed Bacteria (HKB) Preparation
4.1.3. Denatured Membrane Proteins (BME) Preparation
4.1.4. Internal Proteins Preparation
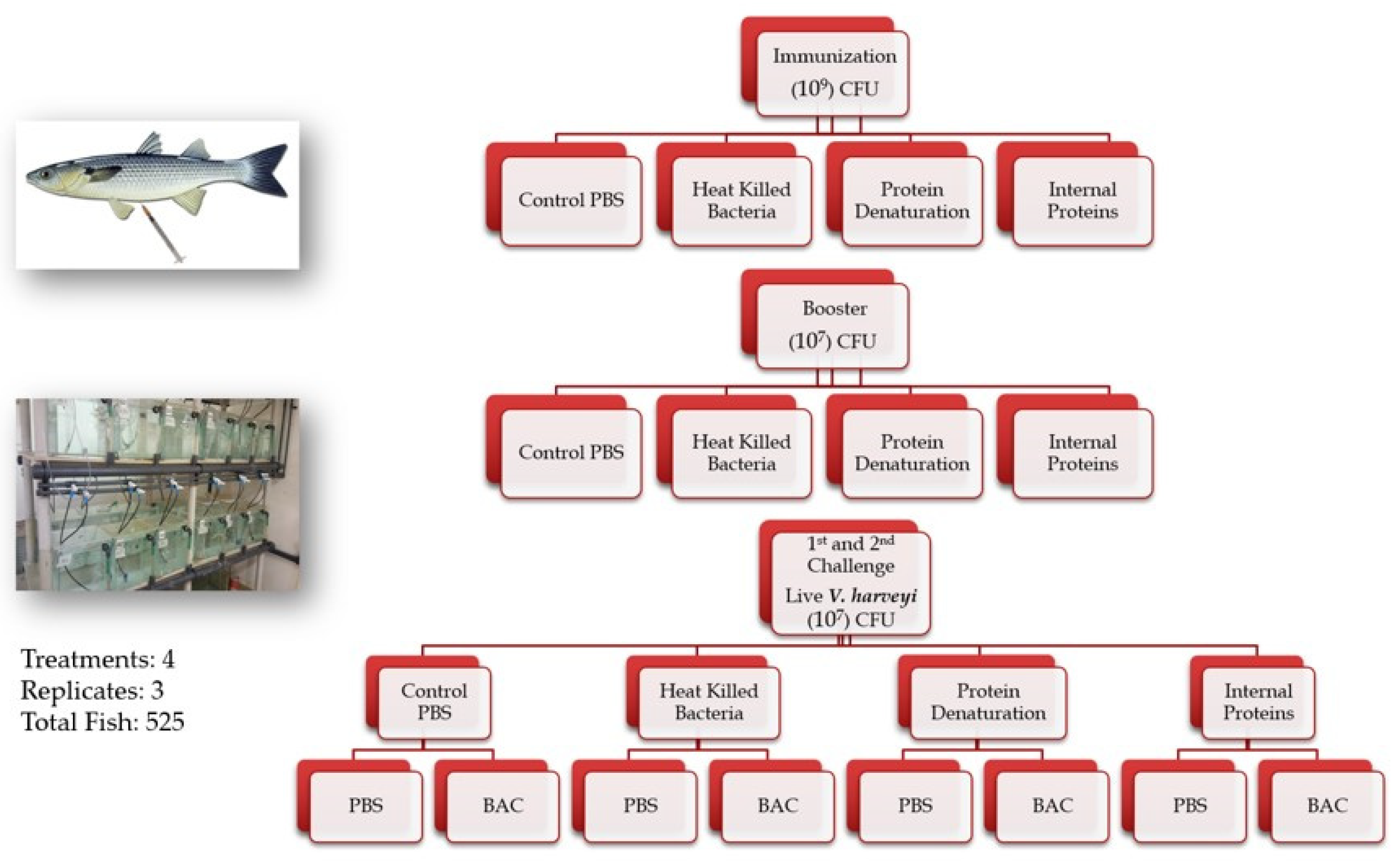
4.2. Immunization and Challenge Infections
4.2.1. Fish
4.2.2. Experimental Design
4.2.3. Samples Collection and Analyses
4.3. Evaluation of Long-Term Protection and Antibody Production of the Heat-Killed Bacteria with Adjuvant Preparation
4.4. Production of Mice Anti Mullets Antibodies
4.4.1. Mice Immunization with Mullet Igs
4.4.2. Gel Electrophoresis and Western Blot Analysis
4.4.3. Dot–Blot Assay
4.5. Multilayer ELISA
4.5.1. Standardization of the Mice Anti-Mullet Serum for the ELISA
4.5.2. ELISA Analysis for Anti-Vibrio Harveyi Antibodies in Mullet Serum
4.5.3. ELISA Analysis to Detect Cross-Reactivity against Other Vibrio spp.
4.6. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ruby, E.G.; Morin, J.G. Luminous Enteric Bacteria of the marine fishes: A study of their distribution, densities, and dispersion. J. Appl. Environ. Microbiol. 1979, 38, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, A.; Venugopalan, V.K. Response of enteric luminous bacteria to environmental conditions in the gut of the fish. J. Appl. Bacteriol. 1989, 66, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.C.; Lee, K.K.; Yii, K.C.; Kou, G.H.; Chen, S.N. Isolation of Vibrio harveyi from diseased kuruma prawns Penaeus japonicus. Curr. Microbiol. 1996, 33, 129–132. [Google Scholar] [CrossRef]
- Alvarez, J.D.; Austin, B.; Alvarez, A.M.; Reyes, H. Vibrio harveyi: A pathogen of penaeid shrimps and fish in Venezuela. J. Fish Dis. 1998, 21, 313–316. [Google Scholar] [CrossRef]
- Hispano, C.; Nebra, Y.; Blanch, A.R. Isolation of Vibrio harveyi from an ocular lesion in the short sunfish (Mola mola). Bull. Eur. Assoc. Fish Pathol. 1997, 17, 104–107. [Google Scholar]
- Kraxberger-Beatty, T.; McGarey, D.J.; Grier, H.J.; Lim, D.V. Vibrio harveyi, an opportunistic pathogen of common snook, Centropomus undecimalis (Bloch), held in captivity. J. Fish Dis. 1990, 13, 557–560. [Google Scholar] [CrossRef]
- Ishimaru, K.; Muroga, K. Taxonomical re-examination of two pathogenic Vibrio species isolated from milkfish and swimming crab. Fish Pathol. 1997, 32, 59–64. [Google Scholar] [CrossRef]
- Vera, P.; Navas, J.I.; Quintero, M.C. Experimental study of the virulence of three species of Vibrio bacteria in Penaeus japonicas (Bate 1881) juveniles. Aquaculture 1992, 107, 119–123. [Google Scholar] [CrossRef]
- Jun, L.I.; Huai-Shu, X. Isolation and biological characteristics of Vibrio harveyi affecting hatchery reared Penaeus chinensis larvae. Chin. J. Oceanol. Limnol. 1998, 29, 353–361. [Google Scholar]
- Hashem, M.; El-Barbary, M. Vibrio harveyi infection in Arabian surgeon fish (Acanthurus sohal) of Red Sea at Hurghada Egypt. Egypt. J. Aquat. Res. 2013, 39, 199–203. [Google Scholar] [CrossRef]
- Liu, P.C.; Lee, K.K.; Chen, S.N. Pathogenicity of different isolates of Vibrio harveyi in tiger prawn: Penaeus monodon. Lett. Appl. Microbiol. 1996, 22, 413–416. [Google Scholar]
- Montero, A.B.; Austin, B. Characterization of extracellular products from an isolate of Vibrio Harveyi recovered from diseased post-larval Penaeus vannamei (Bonne). J. Fish Dis. 1999, 22, 377–386. [Google Scholar] [CrossRef]
- Austin, B.A.; Pride, C.; Rhodie, G.A. Association of a bacteriophage with virulence in Vibrio harveyi. J. Fish Dis. 2003, 26, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Zhang, X.H.; Chen, J.; Chi, Z.; Sun, B.; Li, Y.; Austin, B. Overexpression, purification, characterization, and pathogenicity of Vibrio harveyi Hemolysin VHH. Infect. Immun. 2006, 74, 6001–6005. [Google Scholar] [CrossRef]
- Darshanee-Ruwandeepika, H.A.; Prasad-Jayaweera, T.S.; Bhowmick, P.P.; Karunasagar, I.; Bossier, P.; Defroit, T. Pathogenesis, Virulence regulation of vibrios belonging to the harveyi clade. Rev. Aquac. 2012, 4, 59–74. [Google Scholar] [CrossRef]
- Zhang, X.; He, X.; Austin, B. Vibrio harveyi: A serious pathogen of fish and invertebrates in mariculture. Mar. Life Sci. Technol. 2020, 2, 231–245. [Google Scholar] [CrossRef]
- Ruwandeepika, H.A.D. Expression of Virulence Genes of Vibrios Belonging to the harveyi Clade in the Brine Shrimp Artemia. Ph.D. Thesis, Ghent University, Ghent, Belgium, 2010. [Google Scholar]
- Novriadi, R. Vibriosis in aquaculture. Omni-Akuatika 2016, 12, 1–12. [Google Scholar] [CrossRef]
- Wei, Q. Social and economic impacts of aquatic animal health problems in aquaculture in China. In Primary Aquatic Animal Health Care in Rural, Small-Scale, Aquaculture Development; FAO Fish Technical Paper N° 406; Arthur, J.R., Phillips, M.J., Subasinghe, R.P., Reantaso, M.B., MacRae, I.H., Eds.; FAO: Rome, Italy, 2002; pp. 55–61. [Google Scholar]
- Tamaru, C.S.; Tamaru, C.C.T.; FitzGerald, W.J.; Ako, H.; Sato, V.T. Advantages in the culture of striped mullet. Am. Fish Soc. Symp. 2005, 46, 439–456. [Google Scholar]
- Whitfield, A.K.; Panfili, J.; Durand, J.D. A global review of the cosmopolitan flathead grey mullet (Mugil cephalus), (Linnaeus 1758; Teleostei: Mugilidae), with emphasis on the biology, genetics, ecology, and fisheries aspects of this apparent species complex. Rev. Fish Biol. Fish. 2012, 22, 641–681. [Google Scholar] [CrossRef]
- Hung, C.M.; Shaw, D. The impact of upstream catch and global warming on the flathead grey mullet fishery in Taiwan: A non-cooperative game analysis. Mar. Resour. Econ. 2006, 21, 285–300. [Google Scholar] [CrossRef]
- Maitland, P.S.; Herdson, D. Key to the Marine and Freshwater Fishes of Britain and Ireland; Environmental Agency: Bristol, UK, 2009.
- Strem, R.I.; Ehrlich, R.; Shashar, N.; Sharon, G. First Description of Vibrio harveyi as the Causative Agent of Morbidity and Mortality in Farmed Flathead Grey Mullet (Mugil cephalus). Dis. Aquat. Org. 2023, in press. [Google Scholar] [CrossRef]
- Adams, A. Advantages in disease diagnosis, vaccine development and other emerging methods to control pathogens in aquaculture. New Technol. Aquac. 2009, 2009, 197–214. [Google Scholar] [CrossRef]
- Sudhagar, A.; Khan, F.; Prabu, L. Fish Vaccination: A Health Management Tool for Aquaculture. Available online: http://aquafind.com/articles/Vaccination.php (accessed on 23 February 2023).
- Ma, J.; Bruce, T.J.; Jones, E.M.; Cain, K.D. A review of fish vaccine development strategies: Conventional methods and modern biotechnological approaches. Microorganisms 2019, 7, 569. [Google Scholar] [CrossRef]
- Mohd-Aris, A.; Muhamed-Sofie, M.H.N.; Zamri-Saad, M.; Daud, H.M.; Ina-Salwany, M.Y. Live vaccines against bacterial fish diseases: A review. Vet. World 2019, 12, 1806–1815. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Hu, X.; Miao, L.; Chen, J. Current status and development prospects of aquatic vaccines. Front. Immunol. 2022, 13, 1040336. [Google Scholar] [CrossRef] [PubMed]
- Mondal, H.; Thomas, J. A review on the recent advances and application of vaccines against fish pathogens in aquaculture. Aquac. Int. 2022, 30, 1971–2000. [Google Scholar] [CrossRef]
- Sharon, G.; Nath, P.; Isakov, N.; Zilberg, D. Evaluation of guppy (Poecilia reticulata Peters) immunization against Tetrahymena sp. by enzyme-linked immunosorbent assay (ELISA). Vet. Parasitol. 2014, 205, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Svobodova, Z.; Jankovicova, B.; Horak, D.; Bilkova, Z. Dot-ELISA affinity test> an easy, low-cost method to estimate binding activity of monoclonal antibodies. J. Anal. Bioanal. Tech. 2013, 4, 168. [Google Scholar]
- Melingen, G.O.; Nilsen, F.; Wergeland, H.I. The serum antibody levels in Atlantic salmon (Salmo salar L.) after vaccination with Vibrio salmonicida at different times during the smolting and early post-smolt period. Fish Shellfish Immunol. 1995, 5, 223–235. [Google Scholar] [CrossRef]
- Crosbie, P.B.B.; Nowak, B.F. Immune responses of barramundi (Lates calcalifer; Block), after administration of an experimental Vibrio harveyi bacterin by intraperitoneal injection, anal intubation and immersion. J. Fish Dis. 2004, 27, 623–632. [Google Scholar] [CrossRef]
- Mohd-Aris, A.; Mohd-Zamri, S.; Mohd-Daud, H.; Mohd-Temizi, Y.; Ina-Salwany, M.Y. Vibrio harveyi protease deletion mutant as a live attenuated vaccine candidate against vibriosis and transcriptome profiling following vaccination for Epinephelus fuscoguttatus. Aquac. Int. 2019, 27, 125–140. [Google Scholar] [CrossRef]
- Trunk, T.; Khalil, H.S.; Leo, J.C. Bacterial autoaggregation. AIMS Microbiol. 2018, 4, 140–164. [Google Scholar] [CrossRef] [PubMed]
- Corno, G.; Coci, M.; Giardina, M. Antibiotics promote aggregation within aquatic bacterial communities. Front. Microbiol. 2014, 5, 297. [Google Scholar] [CrossRef] [PubMed]
- Kumra, A.V.; de Neergaard, T.; Sundwall, M.; Amjornsson, T.; Nordenfelt, P. A predictive model of antibody binding in the presence of IgG-interacting bacterial surface proteins. Front. Immunol. 2021, 12, 661. [Google Scholar]
- Hettiarachchi, M.; Pathirage, S.G.; Hettiarachchi, D.C. Isolation of the bacterium Vibrio harveyi from cultured shrimp, Penaeus monodon and production of vaccines against the bacterium. J. Natl. Sci. Found. Sri Lanka 2005, 33, 257–263. [Google Scholar] [CrossRef]
- Hu, Y.H.; Deng, T.; Sun, B.G.; Sun, L. Development and efficacy of an attenuated Vibrio harveyi vaccine candidate with cross-protective against Vibrio alginolyticus. Fish Shellfish Immunol. 2012, 32, 1155–1161. [Google Scholar] [CrossRef]
- Harikrishnan, R.; Kim, J.S.; Balasundaram, C.; Heo, M.S. Vaccination effect of liposomes entrapped whole-cell bacterial vaccine on immune response and disease protection in Epinephelus bruneus against Vibrio harveyi. Aquaculture 2012, 342, 69–74. [Google Scholar] [CrossRef]
- Xu, W.T.; Jiao, C.L.; Bao, P.C.; Liu, Q.; Wang, P.B.; Zhang, R.L.; Liu, X.H.; Zhang, Y.X. Efficacy of Monthanide ™ ISA763 A VG as aquatic adjuvant administered with an inactivated Vibrio harveyi vaccine in turbot (Scophthalmus maximus L.). Fish Shellfish Immunol. 2019, 84, 56–61. [Google Scholar] [CrossRef]
- Linh, N.V.; Dien, L.T.; Dong, H.T.; Khongdee, N.; Hoseinifar, S.H.; Musthafa, M.S.; Dawood, M.A.O.; Van Doan, H. Efficacy of different routes of formalin-killed vaccine administration on immunity and disease resistance of tilapia (Oreochromis niloticus) challenged with Streptococcus agalactiae. Fishes 2022, 7, 398. [Google Scholar] [CrossRef]
- Facciola, A.; Visalli, G.; Lagana, A.; Di Pietro, A. An Overview of Vaccine Adjuvants: Current Evidence and Future Perspectives. Vaccines 2022, 10, 819. [Google Scholar] [CrossRef]
- Firdaus-Nawi, M.; Yusoff, S.M.; Abdullah, S.Z.; Zamri-Saad, M. Efficacy of feed-based adjuvant vaccine against Streptococcus agalactiae in Oreochromis spp. in Malaysia. Aquac. Res. 2013, 45, 87–96. [Google Scholar] [CrossRef]
- Abu Nor, N.; Zamri-Saad, M.; Md Yasin, I.S.; Salleh, A.; Mustaffa-Kamal, F.; Matori, M.F.; Azmai, M.N.A. Efficacy of whole cell inactivated Vibrio harveyi vaccine against vibriosis in a marine red tilapia (Oreochromis niloticus x O. mossambicus) model. Vaccines 2020, 8, 734. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, A.; Mursidi, F.-A.; Zamri-Saad, M.; Amal, M.N.A.; Annas, S.; Monir, M.S.; Loqman, M.; Hairudin, F.; Al-saari, N.; Ina-Salwany, M.Y. Laboratory and field assessment of oral Vibrio vaccine indicate the potential for protection against vibriosis in cultured marine fishes. Animals 2022, 12, 133. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.K.; Liu, P.C.; Kou, G.H.; Chen, S.C. Passive immunization of the tiger prawn, Penaeus monodon, using rabbit antisera to Vibrio harveyi. Lett. Appl. Microbiol. 1997, 25, 34–37. [Google Scholar] [CrossRef]
- Phianphak, K.; Rengpipat, S.; Rukpratanporn, S.; Longyant, S.; Chaivisuthangkura, P.; Sithigorngul, P. Production of monoclonal antibodies to detect Vibrio harveyi. Dis. Aquat. Org. 2005, 63, 161–168. [Google Scholar] [CrossRef]
- Wilkinson, D.; Harrison, R. Predicting the solubility of recombinant proteins in Escherichia coli. Natl. Biotechnol. 1991, 9, 443–448. [Google Scholar] [CrossRef]
- Shehadul, L.; Islam, M.; Arysomayajula, A.; Selvaganapathy, P.R. A review on macroscale and microscale cell lysis methods. Micromachines 2017, 8, 83. [Google Scholar] [CrossRef]
- Weisburg, W.G.; Barns, S.M.; Pelletier, D.A.; Lane, D.J. 16S ribosomal DNA amplification for phylogenetic study. J. Bacteriol. 1991, 173, 697–703. [Google Scholar] [CrossRef]
- Haldar, S.; Maharajan, A.; Chatterjee, S.; Hunter, S.A.; Chowdhury, N.; Hinenoya, A.; Asakura, M.; Yamasaki, S. Identification of Vibrio harveyi as the causative bacterium for a tail rot disease of sea bream (Sparus aurata) from research hatchery in Malta. Microbiol. Res. 2010, 165, 639–648. [Google Scholar] [CrossRef]
- Pang, L.; Zhang, X.H.; Zhong, Y.; Chen, J.; Li, Y.; Austin, B. Identification of Vibrio harveyi using PCR amplification of the ToxR gene. Lett. Appl. Microbiol. 2006, 43, 249–255. [Google Scholar] [CrossRef]
- Folitse, R.; Halvorson, D.; Sivanandan, V. A dot immunoblotting assay (Dot Blot ELISA) for early detection of Newcastle disease antibodies in chickens. Avian Dis. 1998, 42, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Sithigorngul, P.; Chauichuwong, P.; Sithigorngul, W.; Longyant, S.; Chaivisuthangkura, P.; Menasveta, P. Development of monoclonal antibody specific to yellow head virus (YHV) from Penaeus monodon. Dis. Aquat. Org. 2000, 42, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Zeng, Y.; Chu, C.; Wang, H. Development of a quick dot blot assay for the tittering of bovine ephemeral fever virus. BMC Vet. Res. 2019, 15, 313. [Google Scholar] [CrossRef] [PubMed]
- National Academy of Sciences. Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011; p. 246. [Google Scholar]



| Chronology | Number of Bleedings | Time (Weeks) |
|---|---|---|
| Immunization | Bleeding Time 0 (T0) | 0 |
| Bleeding Time 1 (T1) | 1 | |
| Bleeding Time 2 (T2) | 2 | |
| Booster | Bleeding Time 0 (T0B) | 4 |
| Bleeding Time 1 (T1B) | 5 | |
| Bleeding Time 2 (T2B) | 6 | |
| First Challenge | Bleeding time 0 (T0C) | 7 |
| Bleeding time 1 (T1C) | 8 | |
| Bleeding time 2 (T2C) | 9 | |
| Bleeding time 3 (T3C) | 10 | |
| Bleeding time 4 (T4C) | 11 | |
| Second Challenge | Bleeding time 5 (T5C) | 24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strem, R.; Meiri-Ashkenazi, I.; Segal, N.; Ehrlich, R.; Shashar, N.; Sharon, G. Evaluation of Flathead Grey Mullets (Mugil cephalus) Immunization and Long-Term Protection against Vibrio harveyi Infection Using Three Different Vaccine Preparations. Int. J. Mol. Sci. 2023, 24, 8277. https://doi.org/10.3390/ijms24098277
Strem R, Meiri-Ashkenazi I, Segal N, Ehrlich R, Shashar N, Sharon G. Evaluation of Flathead Grey Mullets (Mugil cephalus) Immunization and Long-Term Protection against Vibrio harveyi Infection Using Three Different Vaccine Preparations. International Journal of Molecular Sciences. 2023; 24(9):8277. https://doi.org/10.3390/ijms24098277
Chicago/Turabian StyleStrem, Rosa, Iris Meiri-Ashkenazi, Na’ama Segal, Roberto Ehrlich, Nadav Shashar, and Galit Sharon. 2023. "Evaluation of Flathead Grey Mullets (Mugil cephalus) Immunization and Long-Term Protection against Vibrio harveyi Infection Using Three Different Vaccine Preparations" International Journal of Molecular Sciences 24, no. 9: 8277. https://doi.org/10.3390/ijms24098277
APA StyleStrem, R., Meiri-Ashkenazi, I., Segal, N., Ehrlich, R., Shashar, N., & Sharon, G. (2023). Evaluation of Flathead Grey Mullets (Mugil cephalus) Immunization and Long-Term Protection against Vibrio harveyi Infection Using Three Different Vaccine Preparations. International Journal of Molecular Sciences, 24(9), 8277. https://doi.org/10.3390/ijms24098277





