The Influence of Gut Microbial Species on Diabetes Mellitus
Abstract
1. Introduction
1.1. Diabetes Mellitus (DM)
1.2. Gut Microbiome and Diabetes
1.3. Gut Microbial Profile in Diabetes
2. Search Strategy and Selection Criteria
3. Diabetes Management Using Microbial Species
4. The Influence of Specific Microbial Species on Diabetes
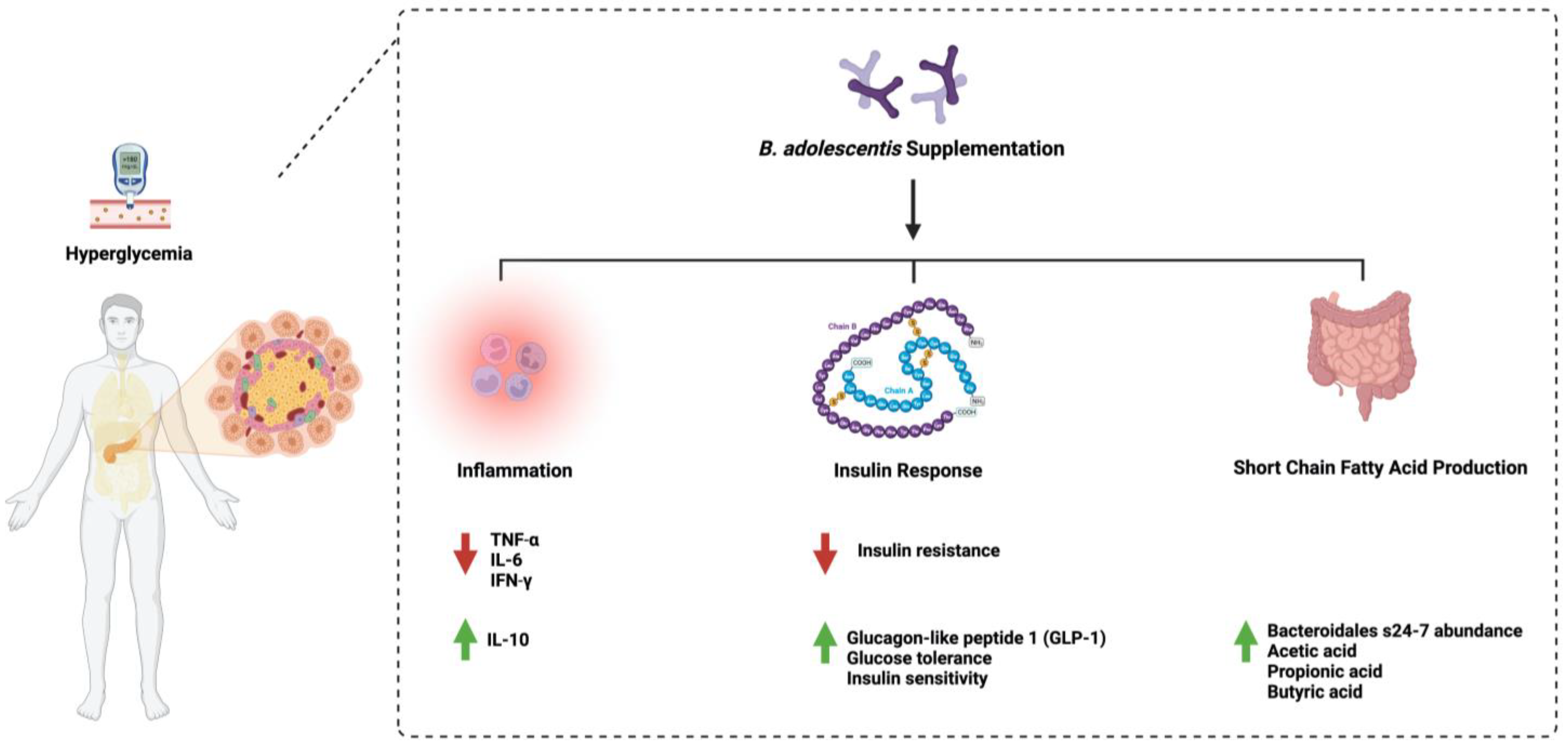
4.1. Bifidobacterium adolescentis
4.2. Bifidobacterium bifidum
4.3. Lactobacillus rhamnosus
5. Discussion
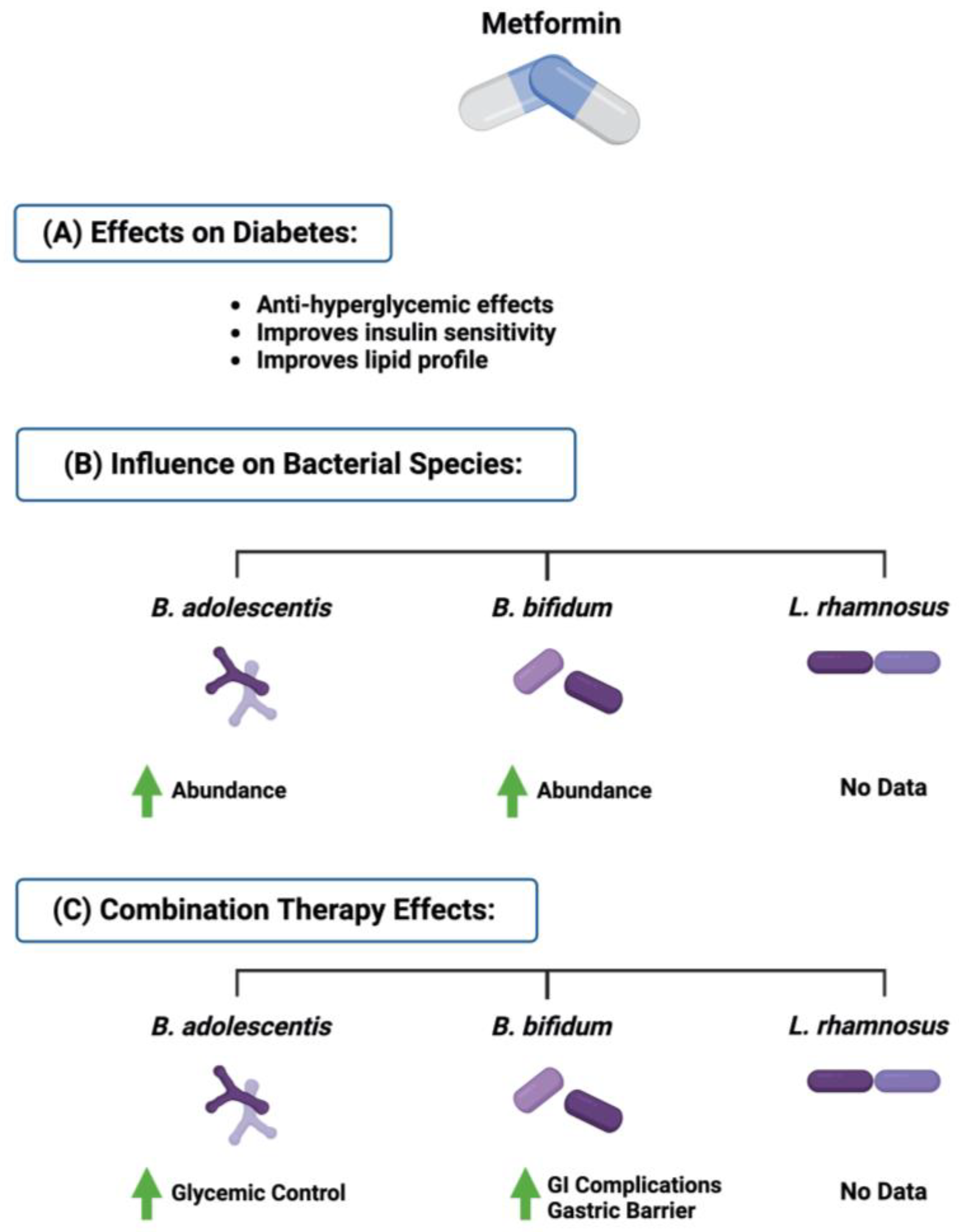
5.1. The Influence of Combination Therapy on Diabetes
5.2. The Influence of Flavonoids on Species Abundance
5.3. Fecal Microbiota Transplantation and Diabetes
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CEs | carbohydrate esterases |
| CRC | colorectal cancer |
| DM | diabetes mellitus |
| FMT | fecal microbiota transplantation |
| G6pc | glucose-6-phosphatase |
| GHs | glycosyl hydrolases |
| GI | gastrointestinal |
| GLP-1 | glucagon-like peptide 1 |
| GLUT | glucose transporter |
| GTs | glycosyl transferases |
| HbA1c | hemoglobin A1C (glycated hemoglobin) |
| HFD | high-fat diet |
| IL-6 | interleukin 6 |
| LDL | light density lipoprotein |
| NF-B | nuclear Factor kappa-light-chain-enhancer of activated B cells |
| PFM | probiotic fermented milk |
| SCFA | short chain fatty acid |
| STZ | streptozotocin |
| TC | total cholesterol |
| TG | triglyceride |
| TNF | tumor necrosis factor |
References
- Zhou, C.; Byard, R.W. An Analysis of The Morbidity and Mortality of Diabetes Mellitus in a Forensic Context. J. Forensic Sci. 2018, 63, 1149–1154. [Google Scholar] [CrossRef] [PubMed]
- Kharroubi, A.T.; Darwish, H.M. Diabetes mellitus: The epidemic of the century. World J. Diabetes 2015, 6, 850–867. [Google Scholar] [CrossRef] [PubMed]
- Banday, M.Z.; Sameer, A.S.; Nissar, S. Pathophysiology of diabetes: An overview. Avicenna J. Med. 2020, 10, 174–188. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Magliano, D.J.; Zimmet, P.Z. The worldwide epidemiology of type 2 diabetes mellitus-present and future perspectives. Nat. Rev. Endocrinol. 2011, 8, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Al-Ishaq, R.K.; Mazurakova, A.; Kubatka, P.; Busselberg, D. Flavonoids’ Dual Benefits in Gastrointestinal Cancer and Diabetes: A Potential Treatment on the Horizon? Cancers 2020, 14, 6073. [Google Scholar] [CrossRef]
- Reyes, J.; Tripp-Reimer, T.; Parker, E.; Muller, B.; Laroche, H. Factors Influencing Diabetes Self-Management Among Medically Underserved Patients with Type II Diabetes. Glob. Qual. Nurs. Res. 2017, 4, 3097. [Google Scholar] [CrossRef]
- Ramachandran, A. Know the signs and symptoms of diabetes. Indian J. Med. Res. 2014, 140, 579–581. [Google Scholar]
- Blaslov, K.; Naranda, F.S.; Kruljac, I.; Renar, I.P. Treatment approach to type 2 diabetes: Past, present and future. World J. Diabetes 2018, 9, 209–219. [Google Scholar] [CrossRef]
- Al Bander, Z.; Nitert, M.D.; Mousa, A.; Naderpoor, N. The Gut Microbiota and Inflammation: An Overview. Int. J. Environ. Res. Public Health 2020, 17, 7618. [Google Scholar] [CrossRef]
- Singh, R.K.; Chang, H.W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Liao, W. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017, 15, 73. [Google Scholar] [CrossRef]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef]
- Forslund, K.; Hildebrand, F.; Nielsen, T.; Falony, G.; Le Chatelier, E.; Sunagawa, S.; Pedersen, O. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature 2015, 528, 262–266. [Google Scholar] [CrossRef]
- Chen, Z.; Radjabzadeh, D.; Chen, L.; Kurilshikov, A.; Kavousi, M.; Ahmadizar, F.; Voortman, T. Association of Insulin Resistance and Type 2 DiabetesWith Gut Microbial Diversity: A Microbiome-Wide Analysis from Population Studies. JAMA Netw. Open 2021, 4, e2118811. [Google Scholar] [CrossRef]
- Creely, S.J.; McTernan, P.G.; Kusminski, C.M. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E740–E747. [Google Scholar] [CrossRef]
- Cani, P.D.; Amar, J.; Iglesias, M.A. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef]
- Moreno-Navarrete, J.M.; Manco, M.; Ibáñez, J. Metabolic endotoxemia and saturated fat contribute to circulating NGAL concentrations in subjects with insulin resistance. Int. J. Obes. 2010, 34, 240–249. [Google Scholar] [CrossRef]
- Tolhurst, G.; Heffron, H.; Lam, Y.S.; Parker, H.E.; Habib, A.M.; Diakogiannaki, E.; Gribble, F.M. Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes 2020, 61, 364–371. [Google Scholar] [CrossRef]
- Polidori, I.; Marullo, L.; Ialongo, C.; Tomassetti, F.; Colombo, R.; di Gaudio, F.; Pieri, M. Characterization of Gut Microbiota Composition in Type 2 Diabetes Patients: A Population-Based Study. Int. J. Environ. Res. Public Health 2022, 19, 15913. [Google Scholar] [CrossRef]
- Gerard, C.; Vidal, H. Impact of Gut Microbiota on Host Glycemic Control. Front. Endocrinol. 2019, 10, 29. [Google Scholar] [CrossRef]
- Martin, A.M.; Yabut, J.M.; Choo, J.M.; Page, A.J.; Sun, E.W.; Jessup, C.F.; Keating, D.J. The gut microbiome regulates host glucose homeostasis via peripheral serotonin. Proc. Natl. Acad. Sci. USA 2019, 116, 19802–19804. [Google Scholar] [CrossRef]
- Larsen, N.; Vogensen, F.K.; van den Berg, F.W.; Nielsen, D.S.; Andreasen, A.S.; Pedersen, B.K.; Jakobsen, M. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS ONE 2010, 5, e9085. [Google Scholar] [CrossRef] [PubMed]
- Howard, E.J.; Lam, T.K.T.; Duca, F.A. The Gut Microbiome: Connecting Diet, Glucose Homeostasis, and Disease. Annu. Rev. Med. 2022, 73, 469–481. [Google Scholar] [CrossRef] [PubMed]
- Delzenne, N.M.; Cani, P.D. Gut microbiota and the pathogenesis of insulin resistance. Curr. Diabetes Rep. 2011, 11, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Li, W.Z.; Stirling, K.; Yang, J.J.; Zhang, L. Gut microbiota and diabetes: From correlation to causality and mechanism. World J. Diabetes 2020, 11, 293–308. [Google Scholar] [CrossRef]
- Zhang, L.; Chu, J.; Hao, W.; Zhang, J.; Li, H.; Yang, C.; Wang, H. Gut Microbiota and Type 2 Diabetes Mellitus: Association, Mechanism, and Translational Applications. Mediat. Inflamm. 2021, 2021, 5110276. [Google Scholar] [CrossRef]
- Karlsson, F.H.; Tremaroli, V.; Nookaew, I.; Bergstrom, G.; Behre, C.J.; Fagerberg, B.; Backhed, F. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature 2013, 498, 99–103. [Google Scholar] [CrossRef]
- Wachsmuth, H.R.; Weninger, S.N.; Duca, F.A. Role of the gut-brain axis in energy and glucose metabolism. Exp. Mol. Med. 2022, 54, 377–392. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R. Microbiota and diabetes: An evolving relationship. Gut 2014, 63, 1513–1521. [Google Scholar] [CrossRef]
- Fijan, S. Microorganisms with claimed probiotic properties: An overview of recent literature. Int. J. Environ. Res. Public Health 2014, 11, 4745–4767. [Google Scholar] [CrossRef]
- Shah, N.; Swami, O. Role of probiotics in diabetes: A review of their rationale and efficacy. EMJ Diabetes 2017, 5, 104–110. [Google Scholar] [CrossRef]
- Zhang, Q. Effect of probiotics on glucose metabolism in patients with type 2 diabetes mellitus: A meta-analysis of randomized controlled trials. Medicina 2016, 52, 28–34. [Google Scholar] [CrossRef]
- Ejtahed, H.S.; Mohtadi-Nia, J.; Homayouni-Rad, A.; Niafar, M.; Asghari-Jafarabadi, M.; Mofid, V. Probiotic yogurt improves antioxidant status in type 2 diabetic patients. Nutrition 2012, 28, 539–543. [Google Scholar] [CrossRef]
- Mykhal’chyshyn, H.P. Effect of probiotics on proinflammatory cytokines level in patients with type 2 diabetes and nonalcoholic fatty liver disease. Likars’ ka Sprava 2013, 56–62. [Google Scholar]
- Iatcu, C.O.; Steen, A.; Covasa, M. Gut Microbiota and Complications of Type-2 Diabetes. Nutrients 2021, 14, 166. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Y.; Wu, L.; Qin, L.; Liu, T. The effects of probiotic administration on patients with prediabetes: A meta-analysis and systematic review. J. Transl. Med. 2022, 20, 498. [Google Scholar] [CrossRef]
- Amar, J.; Chabo, C.; Waget, A.; Klopp, P.; Vachoux, C.; Bermúdez-Humarán, L.G.; Smirnova, N.; Bergé, M.; Sulpice, T.; Lahtinen, S.; et al. Intestinal mucosal adherence and translocation of commensal bacteria at the early onset of type 2 diabetes: Molecular mechanisms and probiotic treatment. EMBO Mol. Med. 2011, 3, 559–572. [Google Scholar] [CrossRef]
- Yun, S.; Park, H.; Kang, J. Effect of Lactobacillus gasseri BNR17 on blood glucose levels and body weight in a mouse model of type 2 diabetes. J. Appl. Microbiol. 2009, 107, 1681–1686. [Google Scholar] [CrossRef]
- Turroni, F.; Peano, C.; Pass, D.A.; Foroni, E.; Severgnini, M.; Claesson, M.J.; Ventura, M. Diversity of bifidobacteria within the infant gut microbiota. PLoS ONE 2012, 7, e36957. [Google Scholar] [CrossRef]
- Picard, C.; Fioramonti, J.; Francois, A.; Robinson, T.; Neant, F.; Matuchansky, C. Review article: Bifidobacteria as probiotic agents—Physiological effects and clinical benefits. Aliment. Pharmacol. Ther. 2005, 22, 495–512. [Google Scholar] [CrossRef]
- Turroni, F.; van Sinderen, D.; Ventura, M. Genomics and ecological overview of the genus Bifidobacterium. Int. J. Food Microbiol. 2011, 149, 37–44. [Google Scholar] [CrossRef]
- Jung, D.H.; Chung, W.H.; Seo, D.H.; Kim, Y.J.; Nam, Y.D.; Park, C.S. Complete genome sequence of Bifidobacterium adolescentis P2P3, a human gut bacterium possessing strong resistant starch-degrading activity. 3 Biotech 2020, 10, 31. [Google Scholar] [CrossRef] [PubMed]
- Sedighi, M.; Razavi, S.; Navab-Moghadam, F.; Khamseh, M.E.; Alaei-Shahmiri, F.; Mehrtash, A.; Amirmozafari, N. Comparison of gut microbiota in adult patients with type 2 diabetes and healthy individuals. Microb. Pathog. 2017, 111, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Moroti, C.; Souza Magri, L.F.; de Rezende Costa, M.; Cavallini, D.C.; Sivieri, K. Effect of the consumption of a new symbiotic shake on glycemia and cholesterol levels in elderly people with type 2 diabetes mellitus. Lipids Health Dis. 2012, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.; Si, Q.; Lin, G.; Zhu, M.; Lu, J.; Zhang, H.; Chen, W. Bifidobacterium adolescentis Is Effective in Relieving Type 2 Diabetes and May Be Related to Its Dominant Core Genome and Gut Microbiota Modulation Capacity. Nutrients 2022, 14, 2479. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, R.; Li, X.F.; Wang, R.L. Bifidobacterium adolescentis supplementation ameliorates visceral fat accumulation and insulin sensitivity in an experimental model of the metabolic syndrome. Br. J. Nutr. 2012, 107, 1429–1434. [Google Scholar] [CrossRef]
- Ku, S.; Park, M.S.; Ji, G.E.; You, H.J. Review on Bifidobacterium bifidum BGN4: Functionality and Nutraceutical Applications as a Probiotic Microorganism. Int. J. Mol. Sci. 2016, 17, 1544. [Google Scholar] [CrossRef]
- Tannock, G.W.; Lawley, B.; Munro, K.; Gowri Pathmanathan, S.; Zhou, S.J.; Makrides, M.; Hodgkinson, A.J. Comparison of the compositions of the stool microbiotas of infants fed goat milk formula, cow milk-based formula, or breast milk. Appl. Environ. Microbiol. 2013, 79, 3040–3048. [Google Scholar] [CrossRef]
- Turroni, F.; Duranti, S.; Milani, C.; Lugli, G.A.; van Sinderen, D.; Ventura, M. Bifidobacterium bifidum: A Key Member of the Early Human Gut Microbiota. Microorganisms 2019, 7, 544. [Google Scholar] [CrossRef]
- Turroni, F.; Bottacini, F.; Foroni, E.; Mulder, I.; Kim, J.H.; Zomer, A.; Ventura, M. Genome analysis of Bifidobacterium bifidum PRL2010 reveals metabolic pathways for host-derived glycan foraging. Proc. Natl. Acad. Sci. USA 2010, 107, 19514–19519. [Google Scholar] [CrossRef]
- Sharma, P.; Bhardwaj, P.; Singh, R. Administration of Lactobacillus casei and Bifidobacterium bifidum Ameliorated Hyperglycemia, Dyslipidemia, and Oxidative Stress in Diabetic Rats. Int. J. Prev. Med. 2016, 7, 102. [Google Scholar]
- Raygan, F.; Rezavandi, Z.; Bahmani, F.; Ostadmohammadi, V.; Mansournia, M.A.; Tajabadi-Ebrahimi, M.; Asemi, Z. The effects of probiotic supplementation on metabolic status in type 2 diabetic patients with coronary heart disease. Diabetol. Metab. Syndr. 2018, 10, 51. [Google Scholar] [CrossRef]
- Le, T.K.; Hosaka, T.; Le, T.T.; Nguyen, T.G.; Tran, Q.B.; Le, T.H.; Pham, X.D. Oral administration of Bifidobacterium spp. improves insulin resistance, induces adiponectin, and prevents inflammatory adipokine expressions. Biomed. Res. 2014, 35, 303–310. [Google Scholar] [CrossRef]
- Capurso, L. Thirty Years of Lactobacillus rhamnosus GG: A Review. J. Clin. Gastroenterol. 2019, 53 (Suppl. 1), S1–S41. [Google Scholar] [CrossRef]
- Park, K.Y.; Kim, B.; Hyun, C.K. Lactobacillus rhamnosus GG improves glucose tolerance through alleviating ER stress and suppressing macrophage activation in db/db mice. J. Clin. Biochem. Nutr. 2015, 56, 240–246. [Google Scholar] [CrossRef]
- Han, M.; Liao, W.; Dong, Y.; Bai, C.; Gai, Z. Lacticaseibacillus rhamnosus Hao9 exerts antidiabetic effects by regulating gut microbiome, glucagon metabolism, and insulin levels in type 2 diabetic mice. Front. Nutr. 2022, 9, 1081778. [Google Scholar] [CrossRef]
- Farida, E.; Nuraida, L.; Giriwono, P.E.; Jenie, B.S.L. Lactobacillus rhamnosus Reduces Blood Glucose Level through Downregulation of Gluconeogenesis Gene Expression in Streptozotocin-Induced Diabetic Rats. Int. J. Food Sci. 2020, 2020, 6108575. [Google Scholar] [CrossRef]
- Yan, F.; Li, N.; Yue, Y.; Wang, C.; Zhao, L.; Evivie, S.E.; Huo, G. Screening for Potential Novel Probiotics with Dipeptidyl Peptidase IV-Inhibiting Activity for Type 2 Diabetes Attenuation in vitro and in vivo. Front. Microbiol. 2019, 10, 2855. [Google Scholar] [CrossRef]
- Bootorabi, F.; Saadat, F.; Falak, R.; Manouchehri, H.; Changizi, R.; Mohammadi, H.; Khorramizadeh, M.R. Gut micobiota alteration by Lactobacillus rhamnosus reduces proinflammatory cytokines and glucose level in the adult model of Zebrafish. BMC Res. Notes 2021, 14, 302. [Google Scholar] [CrossRef]
- Yadav, R.; Dey, D.K.; Vij, R.; Meena, S.; Kapila, R.; Kapila, S. Evaluation of antidiabetic attributes of Lactobacillus rhamnosus MTCC: 5957, Lactobacillus rhamnosus MTCC: 5897 and Lactobacillus fermentum MTCC: 5898 in streptozotocin induced diabetic rats. Microb. Pathog. 2018, 125, 454–462. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, R.K.; Malhotra, S.; Pothuraju, R.; Shandilya, U.K. Lactobacillus rhamnosus NCDC17 ameliorates type-2 diabetes by improving gut function, oxidative stress and inflammation in high-fat-diet fed and streptozotocintreated rats. Benef. Microbes 2017, 8, 243–255. [Google Scholar] [CrossRef]
- Rodriguez, J.; Hiel, S.; Delzenne, N.M. Metformin: Old friend, new ways of action-implication of the gut microbiome? Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Rojas, L.B.; Gomes, M.B. Metformin: An old but still the best treatment for type 2 diabetes. Diabetol. Metab. Syndr. 2013, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- McCreight, L.J.; Bailey, C.J.; Pearson, E.R. Metformin and the gastrointestinal tract. Diabetologia 2016, 59, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Ermolenko, E.; Simanenkova, A.; Voropaeva, L.; Lavrenova, N.; Kotyleva, M.; Minasian, S.; Karonova, T. Metformin Influence on the Intestinal Microbiota and Organism of Rats with Metabolic Syndrome. Int. J. Mol. Sci. 2022, 23, 6837. [Google Scholar] [CrossRef]
- Wu, H.; Esteve, E.; Tremaroli, V.; Khan, M.T.; Caesar, R.; Manneras-Holm, L.; Backhed, F. Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat. Med. 2017, 23, 850–858. [Google Scholar] [CrossRef]
- Whang, A.; Nagpal, R.; Yadav, H. Bi-directional drug-microbiome interactions of antidiabetics. Ebiomedicine 2019, 39, 591–602. [Google Scholar] [CrossRef]
- Makizaki, Y.; Maeda, A.; Yamamoto, M.; Tamura, S.; Tanaka, Y.; Nakajima, S.; Ohno, H. Bifidobacterium bifidum G9-1 ameliorates soft feces induced by metformin without affecting its antihyperglycemic action. Biosci. Microbiot. Food Health 2020, 39, 145–151. [Google Scholar] [CrossRef]
- Hata, S.; Nakajima, H.; Hashimoto, Y.; Miyoshi, T.; Hosomi, Y.; Okamura, T.; Fukui, M. Effects of probiotic Bifidobacterium bifidum G9-1 on the gastrointestinal symptoms of patients with type 2 diabetes mellitus treated with metformin: An open-label, single-arm, exploratory research trial. J. Diabetes Investig. 2022, 13, 489–500. [Google Scholar] [CrossRef]
- Al-Ishaq, R.K.; Abotaleb, M.; Kubatka, P.; Kajo, K.; Busselberg, D. Flavonoids and Their Antidiabetic Effects: Cellular Mechanisms and Effects to Improve Blood Sugar Levels. Biomolecules 2019, 9, 430. [Google Scholar] [CrossRef]
- Al-Ishaq, R.K.; Overy, A.J.; Busselberg, D. Phytochemicals and Gastrointestinal Cancer: Cellular Mechanisms and Effects to Change Cancer Progression. Biomolecules 2020, 10, 105. [Google Scholar] [CrossRef]
- Kubatka, P.; Mazurakova, A.; Samec, M.; Koklesova, L.; Zhai, K.; Al-Ishaq, R.; Busselberg, D.; Golubnitschaja, O. Flavonoids against non-physiologic inflammation attributed to cancer initiation, development, and progression-3PM pathways. EPMA J. 2021, 12, 559–587. [Google Scholar] [CrossRef]
- Al-Ishaq, R.K.; Liskova, A.; Kubatka, P.; Busselberg, D. Enzymatic Metabolism of Flavonoids by Gut Microbiota and Its Impact on Gastrointestinal Cancer. Cancers 2021, 13, 3934. [Google Scholar] [CrossRef]
- Al-Ishaq, R.K.; Koklesova, L.; Kubatka, P.; Busselberg, D. Immunomodulation by Gut Microbiome on Gastrointestinal Cancers: Focusing on Colorectal Cancer. Cancers 2022, 14, 2140. [Google Scholar] [CrossRef]
- Pan, L.; Ye, H.; Pi, X.; Liu, W.; Wang, Z.; Zhang, Y.; Zheng, J. Effects of several flavonoids on human gut microbiota and its metabolism by in vitro simulated fermentation. Front. Microbiol. 2023, 14, 1092729. [Google Scholar] [CrossRef]
- Kawabata, K.; Sugiyama, Y.; Sakano, T.; Ohigashi, H. Flavonols enhanced production of anti-inflammatory substance(s) by Bifidobacterium adolescentis: Prebiotic actions of galangin, quercetin, and fisetin. Biofactors 2013, 39, 422–429. [Google Scholar] [CrossRef]
- Parkar, S.G.; Stevenson, D.E.; Skinner, M.A. The potential influence of fruit polyphenols on colonic microflora and human gut health. Int. J. Food Microbiol. 2008, 124, 295–298. [Google Scholar] [CrossRef]
- Wang, L.; Gao, M.; Kang, G.; Huang, H. The Potential Role of Phytonutrients Flavonoids Influencing Gut Microbiota in the Prophylaxis and Treatment of Inflammatory Bowel Disease. Front. Nutr. 2021, 8, 798038. [Google Scholar] [CrossRef]
- Vindigni, S.M.; Surawicz, C.M. Fecal Microbiota Transplantation. Gastroenterol. Clin. N. Am. 2017, 46, 171–185. [Google Scholar] [CrossRef]
- Park, S.Y.; Seo, G.S. Fecal Microbiota Transplantation: Is It Safe? Clin. Endosc. 2021, 54, 157–160. [Google Scholar] [CrossRef]
- Wang, S.; Xu, M.; Wang, W.; Cao, X.; Piao, M.; Khan, S.; Wang, B. Systematic Review: Adverse Events of Fecal Microbiota Transplantation. PLoS ONE 2016, 11, e0161174. [Google Scholar] [CrossRef]
- de Groot, P.; Nikolic, T.; Pellegrini, S.; Sordi, V.; Imangaliyev, S.; Rampanelli, E.; Nieuwdorp, M. Faecal microbiota transplantation halts progression of human new-onset type 1 diabetes in a randomised controlled trial. Gut 2021, 70, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.T.; Ye, X.L.; Yong, H.J.; Song, B.; Zheng, X.L.; Cui, B.T.; Ding, D.F. Fecal microbiota transplantation relieve painful diabetic neuropathy: A case report. Medicine 2018, 97, e13543. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.C.; Jing, X.B.; Chen, X.; Chen, L.Z.; Zhang, S.H.; Cai, X.B. Fecal microbiota transplantation treatment for type 1 diabetes mellitus with malnutrition: A case report. Ther. Adv. Chronic Dis. 2022, 13, 7449. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Hong, Z.; Zhou, T.; Jian, Y.; Xu, M.; Zhang, X.; Wang, J. Health improvements of type 2 diabetic patients through diet and diet plus fecal microbiota transplantation. Sci. Rep. 2022, 12, 1152. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Lu, Y.; Yan, Y.; Tian, S.; Zheng, D.; Leng, D.; Bai, Y. Promising Treatment for Type 2 Diabetes: Fecal Microbiota Transplantation Reverses Insulin Resistance and Impaired Islets. Front. Cell. Infect. Microbiol. 2019, 9, 455. [Google Scholar] [CrossRef]



| Bacterial Species | Targeted Metabolites/Proteins/Genes/Pathway | Mode of Administration | Number of Strain Tested/Concentration | Dietary Intervention | Follow-Up Period | Mechanism of Action | Methods of Testing | Model Used | References | |
|---|---|---|---|---|---|---|---|---|---|---|
| In Vivo | In Vitro | |||||||||
| Bifidobacterium adolescentis | Glucose Lipid metabolism Inflammatory markers | Oral gavage Intraperitoneally | 8 | High-fat diet | 12 weeks | -Alleviate insulin resistance -Restore gut microbiota homeostasis -Increase the abundance of SCFA-producing flora -Alleviate inflammation by reducing the concentration of TNF-a, IL-6, and IFN-Y | Biochemical analysis Histopathological analysis SCFA analysis Polymerase chain reaction | -C57BL/6J mice | -Pancreatic cells -Hepatic cells | [44] |
| Visceral fat accumulation Insulin sensitivity | Orally | 5 × 108 colony-forming units/mL of live B. adolescentis | High-fat diet | 12 weeks | -Supplementation of this bacteria improved diabetes and insulin sensitivity by increasing the production of glucagon-like peptide 1 (GLP-1) -A reduced visceral fat accumulation (liver steatosis and mesenteric fat) | Insulin sensitivity Quantitative reverse transcription PCR Histological analysis | -Male Wistar rats | -Hepatic cells | [45] | |
| Bifidobacteriumbifidum | Glucose Lipid metabolism Inflammatory markers | Orally | 1 × 107 colony-forming units/mL | NA | 28 days | -Administration of B. bifidum significantly reduced serum fasting blood glucose -It reduced the level of total cholesterol, triglycerides, low-density lipoproteins, and very low-density lipoproteins and enhanced the level of high-density lipoproteins -Reduced the activity of lipid peroxidation -Enhanced the activity of glutathione, superoxide dismutase, catalase, glutathione peroxidase, glutathione reductase, and glutathione-S-transferase | Glucose tolerance test Oxidative stress enzymatic assay | -Male Wistar rats | -Pancreatic cells -Hepatic cells | [50] |
| Glucose Lipid metabolism Inflammatory markers | Oral gavage | 1 × 109 colony-forming units/mL | High-fat diet | 5 weeks | -Administration of B. bifidum significantly reduced plasma glucose level -Treatment with B. bifidum increased the adiponectin mRNA level and decreased MCP-1 and IL-6 mRNA levels | Quantitative real-time PCR RNA extraction Glucose tolerance test ELISA Insulin tolerance test | -Swiss-Webster mice -C57BL/6J mice | -Adipose tissue | [52] | |
| Lactobacillus rhamnosus | Glucose Lipid metabolism | Orally | 1 × 109 colony-forming units/mL | Standard diet | 30 days | -Administration of L. rhamnosus significantly reduced serum fasting blood glucose -Improved glucose tolerance via downregulation of glucose-6-phosphatase (G6p) expression -Significantly reduced the level of total cholesterol -Lowered the risk of atherosclerosis by lowering the atherogenic index (AI) | Biochemical parameter analysis Glucose tolerance test Quantitative real-time PCR Gene expression analysis | -Sprague-Dawley rats | -Hepatic cells | [56] |
| Glucose Inflammatory markers | NA | 106 colony-forming units/mL | Fish commercial food | 10 days | -Reduced blood glucose level -Supplementation with L. rhamnosus resulted in a significant decrease in the expression levels of proinflammatory cytokines -Improved the villus length and width of the intestine | Histological staining Quantitative real-time PCR Immunohistochemistry | -Zebrafish | -Intestinal cells | [58] | |
| Glucose | Intraperitoneally | 1 × 109 colony-forming units/mL | High-fat diet | 12 weeks | -Administration of L. rhamnosus significantly reduced serum fasting blood glucose -Significantly improved glucose intolerance -Significantly reduced the level of HbA1c and GLP-1 | Glucose tolerance test Quantitative real-time PCR Lipid peroxidation inhibiting capacity ELISA Biochemical parameters | -Male C57BL/6J mice | [57] | ||
| Glucose Inflammatory markers | Intraperitoneally Orally | 1 × 108 colony-forming units/mL | Chow diet | 4 weeks | -Treatment with L. rhamnosus significantly improved glucose tolerance -It alleviated endoplasmic reticulum stress by modulating lipid metabolism in skeletal muscle -It alleviated macrophage markers expression F4/80 and CD11b | Glucose tolerance test Real-time PCR Western blot Immunofluorescence | -C57BL/KsJ db/db (db/db) mice | -Adipose tissue -Skeletal muscle | [54] | |
| Glucose | Intraperitoneally | 5 × 109 colony-forming units/mL | High-fat diet | 12 weeks | -Treatment with L. rhamnosus significantly reduced fasting blood glucose and insulin levels -It significantly decreased glucose-6-phosphatase and phosphoenolpyruvate carboxykinase expression in the livers -It reduced the serum concentrations of proinflammatory cytokines such as tumor necrosis factor alpha (TNFa), interleukin-1b (IL1b), and IL6 -Improved intestinal barrier function in diabetic mice | Glucose tolerance test Histopathological examination Biochemical analysis RNA isolation and RT-PCR analysis Colonic tight junction protein expression analysis | -Male C57BL/6J mice | -Hepatic tissues -Colon tissues | [55] | |
| Glucose Inflammatory markers | Oral gavage Intraperitoneally | NA | Probiotic fermented milk (PFM) | 6 weeks | -PFM significantly improved glucose metabolism (fasting blood glucose, glycated hemoglobin, serum insulin) -It also improved the serum inflammation status (tumor necrosis factor-α, and serum interleukin-6) -PFM has significantly reduced the mRNA expression of pepck and g6pase genes that code the key enzymes of gluconeogenesis pathway | Glucose tolerance test Histopathological examination Biochemical analysis | -Male Wistar rats | [59] | ||
| Glucose Inflammatory markers | Oral gavage Intraperitoneally | NA | High-fat diet | 6 weeks | -Treatment with L. rhamnosus improved oral glucose tolerance test -It improved the biochemical parameters such as fasting blood glucose, plasma insulin, glycosylated hemoglobin, free fatty acids, triglycerides, total cholesterol, low-density lipoprotein cholesterol, and high-density lipoprotein cholesterol -It also improved the expression of glucagon-like peptide-1-producing genes in the cecum -It reduced the expression of tumor necrosis factor-α and interleukin-6 | Glucose tolerance test Biochemical analysis | -Rats | [60] | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
AL-Ishaq, R.K.; Samuel, S.M.; Büsselberg, D. The Influence of Gut Microbial Species on Diabetes Mellitus. Int. J. Mol. Sci. 2023, 24, 8118. https://doi.org/10.3390/ijms24098118
AL-Ishaq RK, Samuel SM, Büsselberg D. The Influence of Gut Microbial Species on Diabetes Mellitus. International Journal of Molecular Sciences. 2023; 24(9):8118. https://doi.org/10.3390/ijms24098118
Chicago/Turabian StyleAL-Ishaq, Raghad Khalid, Samson Mathews Samuel, and Dietrich Büsselberg. 2023. "The Influence of Gut Microbial Species on Diabetes Mellitus" International Journal of Molecular Sciences 24, no. 9: 8118. https://doi.org/10.3390/ijms24098118
APA StyleAL-Ishaq, R. K., Samuel, S. M., & Büsselberg, D. (2023). The Influence of Gut Microbial Species on Diabetes Mellitus. International Journal of Molecular Sciences, 24(9), 8118. https://doi.org/10.3390/ijms24098118







