Emerging Therapies for the Treatment of Atherosclerotic Cardiovascular Disease: From Bench to Bedside
Abstract
1. Introduction
2. Contemporary View on the Pathophysiology of Atherosclerosis
3. Emerging Therapies in Atherosclerosis
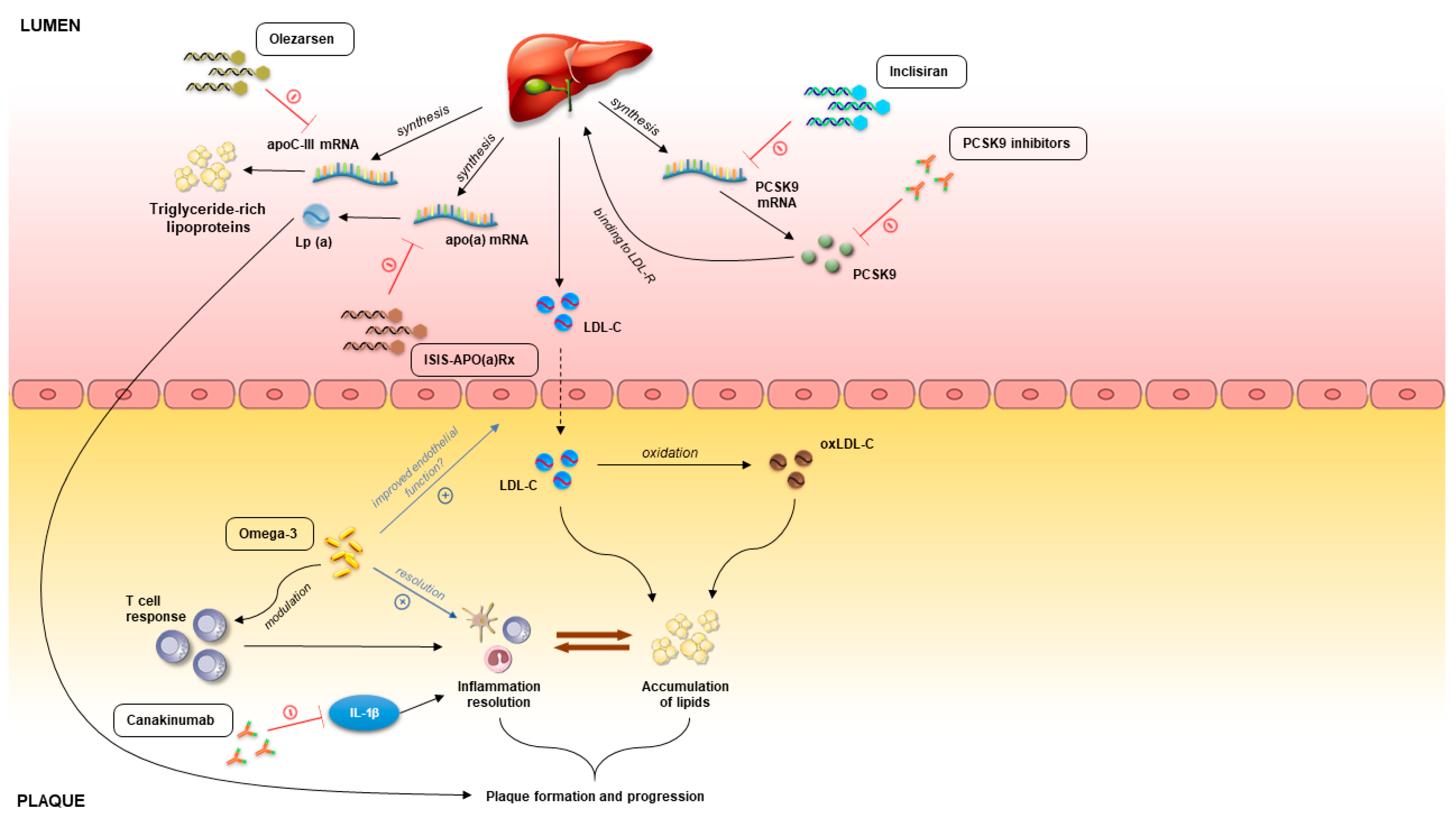
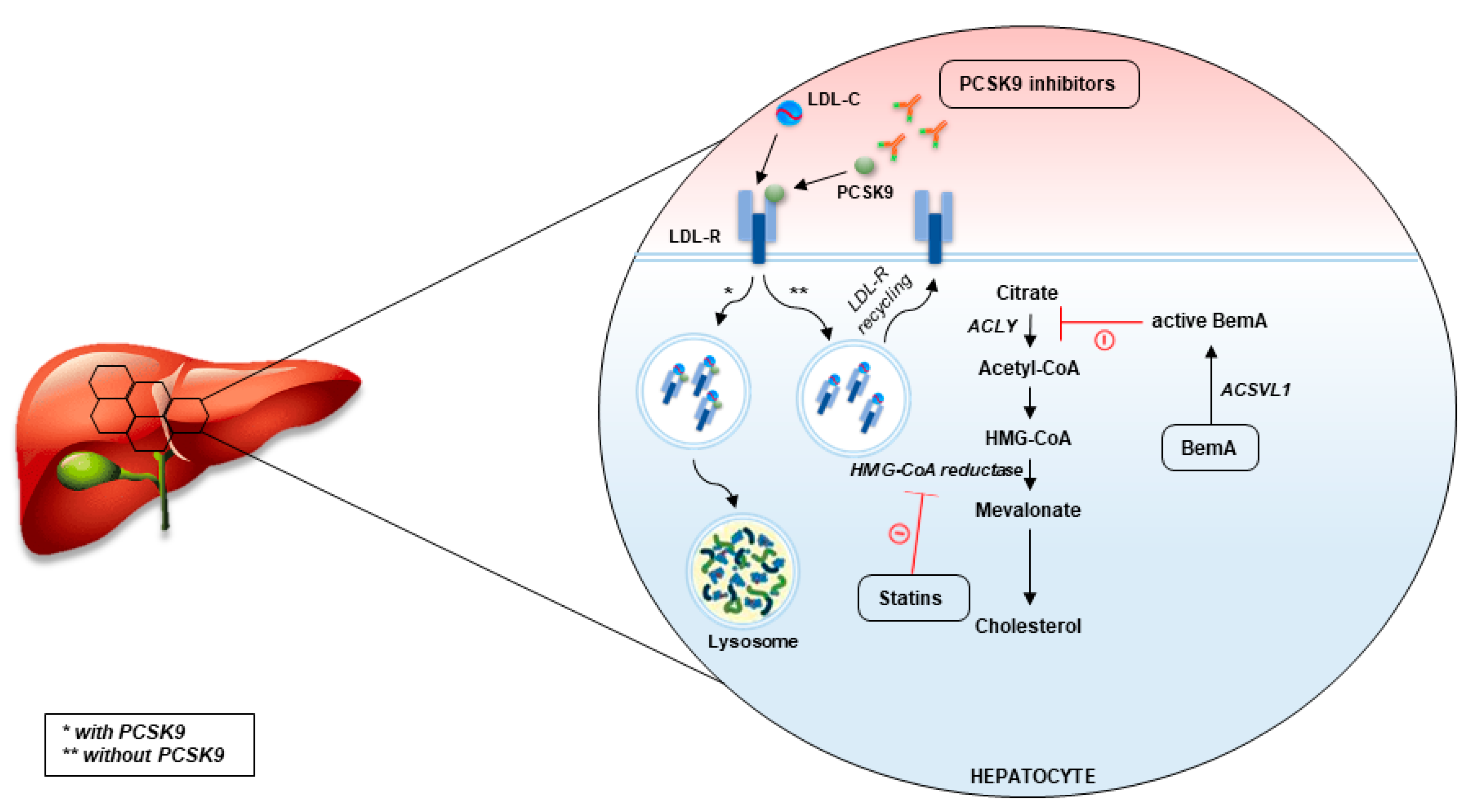
3.1. PCSK9 as Therapeutic Target
3.2. Omega-3 Fatty Acids
3.3. Bempedoic Acid
3.4. Pemafibrate
3.5. RNA-Based Therapies
3.6. Therapies Based on Apolipoprotein A-I
3.7. Immunomodulatory Therapies
4. Future Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Libby, P.; Sukhova, G.; Lee, R.T.; Liao, J.K. Molecular biology of atherosclerosis. Int. J. Cardiol. 1997, 62 (Suppl. 2), S23–S29. [Google Scholar] [CrossRef] [PubMed]
- Kumric, M.; Borovac, J.A.; Martinovic, D.; Kurir, T.T.; Bozic, J. Circulating Biomarkers Reflecting Destabilization Mechanisms of Coronary Artery Plaques: Are We Looking for the Impossible? Biomolecules 2021, 11, 881. [Google Scholar] [CrossRef] [PubMed]
- Yurdagul, A.; Finney, A.C.; Woolard, M.D.; Orr, A.W. The arterial microenvironment: The where and why of atherosclerosis. Biochem. J. 2016, 473, 1281–1295. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.A.; Mensah, G.A.; Johnson, C.O.; Addolorato, G.; Ammirati, E.; Baddour, L.M.; Barengo, N.C.; Beaton, A.Z.; Benjamin, E.J.; Benziger, C.P.; et al. Global Burden of Cardiovascular Diseases and Risk Factors, 1990–2019: Update From the GBD 2019 Study. J. Am. Coll. Cardiol. 2020, 76, 2982–3021. [Google Scholar] [CrossRef] [PubMed]
- Herrington, W.; Lacey, B.; Sherliker, P.; Armitage, J.; Lewington, S. Epidemiology of Atherosclerosis and the Potential to Reduce the Global Burden of Atherothrombotic Disease. Circ. Res. 2016, 118, 535–546. [Google Scholar] [CrossRef]
- Torzewski, M. The Initial Human Atherosclerotic Lesion and Lipoprotein Modification—A Deep Connection. Int. J. Mol. Sci. 2021, 22, 11488. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef]
- Simionescu, M. Implications of Early Structural-Functional Changes in the Endothelium for Vascular Disease. Arter. Thromb. Vasc. Biol. 2007, 27, 266–274. [Google Scholar] [CrossRef]
- Kowara, M.; Cudnoch-Jedrzejewska, A. Pathophysiology of Atherosclerotic Plaque Development-Contemporary Experience and New Directions in Research. Int. J. Mol. Sci. 2021, 22, 3513. [Google Scholar] [CrossRef]
- Goldstein, J.L.; Brown, M.S. A Century of Cholesterol and Coronaries: From Plaques to Genes to Statins. Cell 2015, 161, 161–172. [Google Scholar] [CrossRef]
- Goldberg, I.J.; Sharma, G.; Fisher, E.A. Atherosclerosis: Making a U Turn. Annu. Rev. Med. 2020, 71, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.S.; Goldstein, J.L. Lowering LDL--Not Only How Low, But How Long? Science 2006, 311, 1721–1723. [Google Scholar] [CrossRef] [PubMed]
- Gidding, S.S.; Allen, N. Cholesterol and Atherosclerotic Cardiovascular Disease: A Lifelong Problem. J. Am. Heart Assoc. 2019, 8, e012924. [Google Scholar] [CrossRef]
- Roberts, W.C. Evaluating Lipid-Lowering Trials in the Twenty-First Century. Am. J. Cardiol. 2009, 103, 1325–1328. [Google Scholar] [CrossRef]
- Kim, K.; Lee, C.J.; Shim, C.Y.; Kim, J.-S.; Kim, B.-K.; Park, S.; Chang, H.-J.; Hong, G.-R.; Ko, Y.-G.; Kang, S.-M.; et al. Statin and clinical outcomes of primary prevention in individuals aged >75 years: The SCOPE-75 study. Atherosclerosis 2019, 284, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Roberts, W.C. Quantitative Extent of Atherosclerotic Plaque in the Major Epicardial Coronary Arteries in Patients with Fatal Coronary Heart Disease, in Coronary Endarterectomy Specimens, in Aorta-Coronary Saphenous Venous Conduits, and Means to Prevent the Plaques: A Review after Studying the Coronary Arteries for 50 Years. Am. J. Cardiol. 2018, 121, 1413–1435. [Google Scholar] [CrossRef]
- Guijarro, C.; Cosín-Sales, J. Colesterol LDL y aterosclerosis: Evidencias. Clínica Investig. Arterioscler. 2021, 33, 25–32. [Google Scholar] [CrossRef]
- Ketelhuth, D.F.; Hansson, G.K. Adaptive Response of T and B Cells in Atherosclerosis. Circ. Res. 2016, 118, 668–678. [Google Scholar] [CrossRef]
- Gupta, M.; Blumenthal, C.; Chatterjee, S.; Bandyopadhyay, D.; Jain, V.; Lavie, C.J.; Virani, S.S.; Ray, K.K.; Aronow, W.S.; Ghosh, R.K. Novel emerging therapies in atherosclerosis targeting lipid metabolism. Expert Opin. Investig. Drugs 2020, 29, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. The changing landscape of atherosclerosis. Nature 2021, 592, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G. Triglyceride-Rich Lipoproteins and Atherosclerotic Cardiovascular Disease. Circ. Res. 2016, 118, 547–563. [Google Scholar] [CrossRef]
- Ridker, P.M.; Koenig, W.; Kastelein, J.J.; Mach, F.; Lüscher, T.F. Has the time finally come to measure hsCRP universally in primary and secondary cardiovascular prevention? Eur. Heart J. 2018, 39, 4109–4111. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. A Test in Context. J. Am. Coll. Cardiol. 2016, 67, 712–723. [Google Scholar] [CrossRef] [PubMed]
- Quillard, T.; Araújo, H.A.; Franck, G.; Shvartz, E.; Sukhova, G.; Libby, P. TLR2 and neutrophils potentiate endothelial stress, apoptosis and detachment: Implications for superficial erosion. Eur. Heart J. 2015, 36, 1394–1404. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. Inflammation in Atherosclerosis-No Longer a Theory. Clin Chem. 2021, 67, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Fatkhullina, A.R.; Peshkova, I.O.; Koltsova, E.K. The role of cytokines in the development of atherosclerosis. Biochem. Moscow 2016, 81, 1358–1370. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, P.; Jaiswal, S.; Kathiresan, S.; Siddhartha, J. Clonal Hematopoiesis. Circ. Genom. Precis. Med. 2018, 11, e001926. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, S.; Natarajan, P.; Silver, A.J.; Gibson, C.J.; Bick, A.G.; Shvartz, E.; McConkey, M.; Gupta, N.; Gabriel, S.; Ardissino, D.; et al. Clonal Hematopoiesis and Risk of Atherosclerotic Cardiovascular Disease. N. Engl. J. Med. 2017, 377, 111–121. [Google Scholar] [CrossRef]
- Fuster, J.J.; MacLauchlan, S.; Zuriaga, M.A.; Polackal, M.N.; Ostriker, A.C.; Chakraborty, R.; Wu, C.-L.; Sano, S.; Muralidharan, S.; Rius, C.; et al. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 2017, 355, 842–847. [Google Scholar] [CrossRef]
- Fidler, T.P.; Xue, C.; Yalcinkaya, M.; Hardaway, B.; Abramowicz, S.; Xiao, T.; Liu, W.; Thomas, D.G.; Hajebrahimi, M.A.; Pircher, J.; et al. The AIM2 inflammasome exacerbates atherosclerosis in clonal haematopoiesis. Nature 2021, 592, 296–301. [Google Scholar] [CrossRef]
- Schloss, M.J.; Swirski, F.; Nahrendorf, M. Modifiable Cardiovascular Risk, Hematopoiesis, and Innate Immunity. Circ. Res. 2020, 126, 1242–1259. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Nahrendorf, M.; Swirski, F.K. Leukocytes Link Local and Systemic Inflammation in Ischemic Cardiovascular Disease. J. Am. Coll. Cardiol. 2016, 67, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Hoermann, G.; Greiner, G.; Griesmacher, A.; Valent, P. Clonal Hematopoiesis of Indeterminate Potential: A Multidisciplinary Challenge in Personalized Hematology. J. Pers. Med. 2020, 10, 94. [Google Scholar] [CrossRef] [PubMed]
- Joy, T.R.; A Hegele, R. Narrative review: Statin-related myopathy. Ann. Intern. Med. 2009, 150, 858–868. [Google Scholar] [CrossRef]
- Fernandez, G.; Spatz, E.S.; Jablecki, C.; Phillips, P.S. Statin myopathy: A common dilemma not reflected in clinical trials. Clevel. Clin. J. Med. 2011, 78, 393–403. [Google Scholar] [CrossRef]
- Krähenbühl, S.; Pavik-Mezzour, I.; von Eckardstein, A. Unmet Needs in LDL-C Lowering: When Statins Won’t Do! Drugs 2016, 76, 1175–1190. [Google Scholar] [CrossRef]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the primary prevention of cardiovascular disease: A report of the american college of cardiology/American heart association task force on clinical practice guidelines. Circulation 2019, 140, e596–e646. [Google Scholar] [CrossRef]
- Clader, J.W. The Discovery of Ezetimibe: A View from Outside the Receptor. J. Med. Chem. 2003, 47, 1–9. [Google Scholar] [CrossRef]
- E Bays, H.; Moore, P.B.; A Drehobl, M.; Rosenblatt, S.; Toth, P.D.; A Dujovne, C.; Knopp, R.H.; Lipka, L.J.; LeBeaut, A.P.; Yang, B.; et al. Effectiveness and tolerability of ezetimibe in patients with primary hypercholesterolemia: Pooled analysis of two phase II studies. Clin. Ther. 2001, 23, 1209–1230. [Google Scholar] [CrossRef]
- Sawayama, Y.; Maeda, S.; Ohnishi, H.; Hayashi, S.; Hayashi, J. Efficacy and safety of ezetimibe for Japanese patients with dyslipidaemia: The ESSENTIAL Study. Clin Drug Investig. 2010, 30, 157–166. [Google Scholar] [CrossRef]
- Cannon, C.P.; Blazing, M.A.; Giugliano, R.P.; McCagg, A.; White, J.A.; Théroux, P.; Darius, H.; Lewis, B.S.; Ophuis, T.O.; Jukema, J.W.; et al. Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes. N. Engl. J. Med. 2015, 372, 2387–2397. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.S.; Anderson, R.G.; Goldstein, J.L. Recycling receptors: The round-trip itinerary of migrant membrane proteins. Cell 1983, 32, 663–667. [Google Scholar] [CrossRef] [PubMed]
- Seidah, N.G.; Awan, Z.; Chretien, M.; Mbikay, M. PCSK9: A key modulator of cardiovascular health. Circ. Res. 2014, 114, 1022–1036. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Soffer, G.; Pavlyha, M.; Ngai, C.; Thomas, T.; Holleran, S.; Ramakrishnan, R.; Karmally, W.; Nandakumar, R.; Fontanez, N.; Obunike, J.C.; et al. Effects of PCSK9 Inhibition with Alirocumab on Lipoprotein Metabolism in Healthy Humans. Circulation 2017, 135, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Leiter, L.A.; Teoh, H.; Kallend, D.; Wright, R.S.; Landmesser, U.; Wijngaard, P.L.; Kastelein, J.J.; Ray, K.K. Inclisiran Lowers LDL-C and PCSK9 Irrespective of Diabetes Status: The ORION-1 Randomized Clinical Trial. Diabetes Care 2018, 42, 173–176. [Google Scholar] [CrossRef]
- Ooi, T.C.; A Krysa, J.; Chaker, S.; Abujrad, H.; Mayne, J.; Henry, K.; Cousins, M.; Raymond, A.; Favreau, C.; Taljaard, M.; et al. The Effect of PCSK9 Loss-of-Function Variants on the Postprandial Lipid and ApoB-Lipoprotein Response. J. Clin. Endocrinol. Metab. 2017, 102, 3452–3460. [Google Scholar] [CrossRef]
- Verges, B. Pathophysiology of diabetic dyslipidaemia: Where are we? Diabetologia 2015, 58, 886–899. [Google Scholar] [CrossRef]
- Kastelein, J.J.; Kereiakes, D.J.; Cannon, C.P.; Bays, H.E.; Minini, P.; Lee, L.V.; Maroni, J.; Farnier, M. Effect of alirocumab dose increase on LDL lowering and lipid goal attainment in patients with dyslipidemia. Coron. Artery Dis. 2017, 28, 190–197. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Banka, P.; Mendez, G.; Garcia, R.; Rosenstock, J.; Rodgers, A.; Mendizabal, G.; Mitchel, Y.; Catapano, A.L. Efficacy and safety of the oral PCSK9 inhibitor MK-0616: A phase 2b randomized controlled trial. J. Am. Coll. Cardiol. 2023, 81, 1553–1564. [Google Scholar] [CrossRef]
- Giugliano, R.P.; Keech, A.; Murphy, S.A.; Huber, K.; Tokgozoglu, S.L.; Lewis, B.S.; Ferreira, J.; Pineda, A.L.; Somaratne, R.; Sever, P.S.; et al. Clinical Efficacy and Safety of Evolocumab in High-Risk Patients Receiving a Statin. JAMA Cardiol. 2017, 2, 1385–1391. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Steg, P.G.; Szarek, M.; Bhatt, D.L.; Bittner, V.A.; Diaz, R.; Edelberg, J.M.; Goodman, S.G.; Hanotin, C.; Harrington, R.A.; et al. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N. Engl. J. Med. 2018, 379, 2097–2107. [Google Scholar] [CrossRef] [PubMed]
- Räber, L.; Ueki, Y.; Otsuka, T.; Losdat, S.; Häner, J.D.; Lonborg, J.; Fahrni, G.; Iglesias, J.F.; van Geuns, R.-J.; Ondracek, A.S.; et al. Effect of Alirocumab Added to High-Intensity Statin Therapy on Coronary Atherosclerosis in Patients With Acute Myocardial Infarction. JAMA 2022, 327, 1771. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wen, D.; Chen, Y.; Ma, L.; You, C. PCSK9 inhibitors for secondary prevention in patients with cardiovascular diseases: A bayesian network meta-analysis. Cardiovasc. Diabetol. 2022, 21, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.U.; Talluri, S.; Riaz, H.; Rahman, H.; Nasir, F.; Bin Riaz, I.; Sattur, S.; Ahmed, H.; Kaluski, E.; Krasuski, R. A Bayesian network meta-analysis of PCSK9 inhibitors, statins and ezetimibe with or without statins for cardiovascular outcomes. Eur. J. Prev. Cardiol. 2018, 25, 844–853. [Google Scholar] [CrossRef]
- E Kosmas, C.; Skavdis, A.; Sourlas, A.; Papakonstantinou, E.J.; Genao, E.P.; Uceta, R.E.; Guzman, E. Safety and Tolerability of PCSK9 Inhibitors: Current Insights. Clin. Pharmacol. Adv. Appl. 2020, 12, 191–202. [Google Scholar] [CrossRef]
- Fitzgerald, K.; Frank-Kamenetsky, M.; Shulga-Morskaya, S.; Liebow, A.; Bettencourt, B.R.; E Sutherland, J.; Hutabarat, R.M.; A Clausen, V.; Karsten, V.; Cehelsky, J.; et al. Effect of an RNA interference drug on the synthesis of proprotein convertase subtilisin/kexin type 9 (PCSK9) and the concentration of serum LDL cholesterol in healthy volunteers: A randomised, single-blind, placebo-controlled, phase 1 trial. Lancet 2013, 383, 60–68. [Google Scholar] [CrossRef]
- Ray, K.K.; Landmesser, U.; Leiter, L.A.; Kallend, D.; Dufour, R.; Karakas, M.; Hall, T.; Troquay, R.P.; Turner, T.; Visseren, F.L.; et al. Inclisiran in Patients at High Cardiovascular Risk with Elevated LDL Cholesterol. N. Engl. J. Med. 2017, 376, 1430–1440. [Google Scholar] [CrossRef]
- Samuel, E.; Watford, M.; Egolum, U.O.; Ombengi, D.N.; Ling, H.; Cates, D.W. Inclisiran: A First-in-Class siRNA Therapy for Lowering Low-Density Lipoprotein Cholesterol. Ann Pharmacother. 2023, 57, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Ray, K.K.; Wright, R.S.; Kallend, D.; Koenig, W.; Leiter, L.A.; Raal, F.J.; Bisch, J.A.; Richardson, T.; Jaros, M.; Wijngaard, P.L.; et al. Two Phase 3 Trials of Inclisiran in Patients with Elevated LDL Cholesterol. N. Engl. J. Med. 2020, 382, 1507–1519. [Google Scholar] [CrossRef]
- Novartis Pharmaceuticals. Efficacy, Safety, Tolerability and Quality of Life of Ongoing Individually Optimized Lipid-Lowering Therapy with or without Inclisiran (KJX839)—A Randomized, Placebo-Controlled, Double-Blind Multicenter Phase IV Study in Participants with Hypercholesterolemia. Available online: https://clinicaltrials.gov/ct2/show/NCT0519294 (accessed on 27 March 2023).
- Chaudhary, R.; Garg, J.; Shah, N.; Sumner, A. PCSK9 inhibitors: A new era of lipid lowering therapy. World J. Cardiol. 2017, 9, 76–91. [Google Scholar] [CrossRef]
- Bang, H.O.; Dyerberg, J.; Nielsen, A.B. Plasma lipid and lipoprotein pattern in Greenlandic West-coast Eskimos. Lancet 1971, 1, 1143–1145. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, D.; Lavie, C.J.; Elagizi, A.; Milani, R.V. Update on Omega-3 Polyunsaturated Fatty Acids on Cardiovascular Health. Nutrients 2022, 14, 5146. [Google Scholar] [CrossRef] [PubMed]
- Walti, C.S.; Krantz, E.M.; Maalouf, J.; Boonyaratanakornkit, J.; Keane-Candib, J.; Joncas-Schronce, L.; Stevens-Ayers, T.; Dasgupta, S.; Taylor, J.J.; Hirayama, A.V.; et al. Antibodies to vaccine-preventable infections after CAR-T-cell therapy for B-cell malignancies. J. Clin. Investig. 2021, 6, e146743. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef]
- Tousoulis, D.; Plastiras, A.; Siasos, G.; Oikonomou, E.; Verveniotis, A.; Kokkou, E.; Maniatis, K.; Gouliopoulos, N.; Miliou, A.; Paraskevopoulos, T.; et al. Omega-3 PUFAs improved endothelial function and arterial stiffness with a parallel antiinflammatory effect in adults with metabolic syndrome. Atherosclerosis 2014, 232, 10–16. [Google Scholar] [CrossRef]
- Elagizi, A.; Lavie, C.J.; O’Keefe, E.; Marshall, K.; O’Keefe, J.H.; Milani, R.V. An Update on Omega-3 Polyunsaturated Fatty Acids and Cardiovascular Health. Nutrients 2021, 13, 204. [Google Scholar] [CrossRef]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Japan EPA lipid intervention study (JELIS) Investigators. Effects of eicosapentaenoic acid on major coronary events in hy-percholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098, Erratum in Lancet 2007, 370, 220. [Google Scholar] [CrossRef]
- Hu, Y.; Hu, F.B.; Manson, J.E. Marine Omega-3 Supplementation and Cardiovascular Disease: An Updated Meta-Analysis of 13 Randomized Controlled Trials Involving 127 477 Participants. J. Am. Heart Assoc. 2019, 8, e013543. [Google Scholar] [CrossRef]
- Aung, T.; Halsey, J.; Kromhout, D.; Gerstein, H.; Marchioli, R.; Tavazzi, L.; Geleijnse, J.M.; Rauch, B.; Ness, A.; Galan, P.; et al. Associations of Omega-3 Fatty Acid Supplement Use With Cardiovascular Disease Risks. JAMA Cardiol. 2018, 3, 225–233. [Google Scholar] [CrossRef]
- Ruscica, M.; Sirtori, C.R.; Carugo, S.; Banach, M.; Corsini, A. Bempedoic Acid: For Whom and When. Curr. Atheroscler. Rep. 2022, 24, 791–801. [Google Scholar] [CrossRef]
- Feng, X.; Zhang, L.; Xu, S.; Shen, A.-Z. ATP-citrate lyase (ACLY) in lipid metabolism and atherosclerosis: An updated review. Prog. Lipid Res. 2019, 77, 101006. [Google Scholar] [CrossRef] [PubMed]
- Pinkosky, S.L.; Newton, R.S.; Day, E.A.; Ford, R.J.; Lhotak, S.; Austin, R.C.; Birch, C.M.; Smith, B.K.; Filippov, S.; Groot, P.H.; et al. Liver-specific ATP-citrate lyase inhibition by bempedoic acid decreases LDL-C and attenuates atherosclerosis. Nat. Commun. 2016, 7, 13457. [Google Scholar] [CrossRef]
- Pinkosky, S.L.; Filippov, S.; Srivastava, R.; Hanselman, J.C.; Bradshaw, C.D.; Hurley, T.R.; Cramer, C.T.; Spahr, M.A.; Brant, A.F.; Houghton, J.L.; et al. AMP-activated protein kinase and ATP-citrate lyase are two distinct molecular targets for ETC-1002, a novel small molecule regulator of lipid and carbohydrate metabolism. J. Lipid Res. 2013, 54, 134–151. [Google Scholar] [CrossRef] [PubMed]
- Pinkosky, S.L.; Groot, P.H.E.; Lalwani, N.D.; Steinberg, G.R. Targeting ATP-Citrate Lyase in Hyperlipidemia and Metabolic Disorders. Trends Mol. Med. 2017, 23, 1047–1063. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Zhang, M.; Song, Q.; Cheng, L.; Zhang, X.; Song, G.; Sun, X.; Gu, M.; Zhou, C.; Zhang, Y.; et al. Development of the novel ACLY inhibitor 326E as a promising treatment for hypercholesterolemia. Acta Pharm. Sin. B 2023, 13, 739–753. [Google Scholar] [CrossRef]
- Samsoondar, J.P.; Burke, A.C.; Sutherland, B.G.; Telford, D.E.; Sawyez, C.G.; Edwards, J.Y.; Pinkosky, S.L.; Newton, R.S.; Huff, M.W. Prevention of Diet-Induced Metabolic Dysregulation, Inflammation, and Atherosclerosis in Ldlr−/− Mice by Treatment With the ATP-Citrate Lyase Inhibitor Bempedoic Acid. Arter. Thromb. Vasc. Biol. 2017, 37, 647–656. [Google Scholar] [CrossRef]
- Ruscica, M.; Tokgözoğlu, L.; Corsini, A.; Sirtori, C.R. PCSK9 inhibition and inflammation: A narrative review. Atherosclerosis 2019, 288, 146–155. [Google Scholar] [CrossRef]
- Morrow, M.R.; Batchuluun, B.; Wu, J.; Ahmadi, E.; Leroux, J.M.; Mohammadi-Shemirani, P.; Desjardins, E.M.; Wang, Z.; Tsakiridis, E.E.; Lavoie, D.C.; et al. Inhibition of ATP-citrate lyase improves NASH, liver fibrosis, and dyslipidemia. Cell Metab. 2022, 34, 919–936. [Google Scholar] [CrossRef]
- Pirillo, A.; Catapano, A.L. New insights into the role of bempedoic acid and ezetimibe in the treatment of hypercholesterolemia. Curr. Opin. Endocrinol. Diabetes 2022, 29, 161–166. [Google Scholar] [CrossRef]
- Alam, U.; Al-Bazz, D.Y.; Soran, H. Bempedoic Acid: The New Kid on the Block for the Treatment of Dyslipidemia and LDL Cholesterol: A Narrative Review. Diabetes Ther. 2021, 12, 1779–1789. [Google Scholar] [CrossRef]
- Laufs, U.; Ballantyne, C.M.; Banach, M.; Bays, H.; Catapano, A.L.; Duell, P.B.; Goldberg, A.C.; Gotto, A.M.; Leiter, L.A.; Ray, K.K.; et al. Efficacy and safety of bempedoic acid in patients not receiving statins in phase 3 clinical trials. J. Clin. Lipidol. 2022, 16, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, S.B.; Crass, R.L.; Chapel, S.; Kerschnitzki, M.; Sasiela, W.J.; Emery, M.G.; Amore, B.M.; Barrett, P.H.R.; Watts, G.F.; Catapano, A.L. Pharmacodynamic effect of bempedoic acid and statin combinations: Predictions from a dose–response model. Eur. Heart J. Cardiovasc. Pharmacother. 2021, 8, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Agha, A.M.; Jones, P.H.; Ballantyne, C.M.; Virani, S.S.; Nambi, V. Greater than expected reduction in low-density lipoprotein-cholesterol (LDL-C) with bempedoic acid in a patient with heterozygous familial hypercholesterolemia (HeFH). J. Clin. Lipidol. 2021, 15, 649–652. [Google Scholar] [CrossRef] [PubMed]
- Atar, D.; Jukema, J.W.; Molemans, B.; Taub, P.R.; Goto, S.; Mach, F.; CerezoOlmos, C.; Underberg, J.; Keech, A.; Tokgözoğlu, L.; et al. New cardiovascular prevention guidelines: How to optimally manage dyslipidaemia and cardiovascular risk in 2021 in patients needing secondary prevention? Atherosclerosis 2020, 319, 51–61. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Banach, M.; Mancini, G.J.; Lepor, N.E.; Hanselman, J.C.; Zhao, X.; Leiter, L.A. Efficacy and safety of bempedoic acid added to ezetimibe in statin-intolerant patients with hypercholesterolemia: A randomized, placebo-controlled study. Atherosclerosis 2018, 277, 195–203. [Google Scholar] [CrossRef]
- Ray, K.K.; Bays, H.E.; Catapano, A.L.; Lalwani, N.D.; Bloedon, L.T.; Sterling, L.R.; Robinson, P.L.; Ballantyne, C.M. Safety and Efficacy of Bempedoic Acid to Reduce LDL Cholesterol. N. Engl. J. Med. 2019, 380, 1022–1032. [Google Scholar] [CrossRef]
- Goldberg, A.C.; Leiter, L.A.; Stroes, E.S.G.; Baum, S.J.; Hanselman, J.C.; Bloedon, L.T.; Lalwani, N.D.; Patel, P.M.; Zhao, X.; Duell, P.B. Effect of Bempedoic Acid vs Placebo Added to Maximally Tolerated Statins on Low-Density Lipoprotein Cholesterol in Patients at High Risk for Cardiovascular Disease. JAMA 2019, 322, 1780–1788. [Google Scholar] [CrossRef]
- Laufs, U.; Banach, M.; Mancini, G.B.J.; Gaudet, D.; Bloedon, L.T.; Sterling, L.R.; Kelly, S.; Stroes, E.S.G. Efficacy and Safety of Bempedoic Acid in Patients With Hypercholesterolemia and Statin Intolerance. J. Am. Heart Assoc. 2019, 8, e011662. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Banach, M.; Bays, H.E.; Catapano, A.L.; Laufs, U.; Stroes, E.S.; Robinson, P.; Lei, L.; Ray, K.K. Long-Term Safety and Efficacy of Bempedoic Acid in Patients With Atherosclerotic Cardiovascular Disease and/or Heterozygous Familial Hypercholesterolemia (from the CLEAR Harmony Open-Label Extension Study). Am. J. Cardiol. 2022, 174, 1–11. [Google Scholar] [CrossRef]
- Banach, M.; Duell, P.B.; Gotto, A.M.; Laufs, U.; Leiter, L.A.; Mancini, G.B.J.; Ray, K.K.; Flaim, J.; Ye, Z.; Catapano, A.L. Association of Bempedoic Acid Administration With Atherogenic Lipid Levels in Phase 3 Randomized Clinical Trials of Patients With Hypercholesterolemia. JAMA Cardiol. 2020, 5, 1124–1135. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Fogacci, F.; Hernandez, A.V.; Banach, M.; on behalf of the Lipid and Blood Pressure Meta-Analysis Collaboration (LBPMC) Group and the International Lipid Expert Panel (ILEP). Efficacy and safety of bempedoic acid for the treatment of hypercholesterolemia: A systematic review and meta-analysis. PLoS Med. 2020, 17, e1003121. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.E.; Lincoff, A.M.; Brennan, D.; Ray, K.K.; Mason, D.; Kastelein, J.J.; Thompson, P.D.; Libby, P.; Cho, L.; Plutzky, J.; et al. Bempedoic Acid and Cardiovascular Outcomes in Statin-Intolerant Patients. N. Engl. J. Med. 2023, 388, 1353–1364. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Masuda, D.; Matsuzawa, Y. Pemafibrate, a New Selective PPARα Modulator: Drug Concept and Its Clinical Applications for Dyslipidemia and Metabolic Diseases. Curr. Atheroscler. Rep. 2020, 22, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, M.; Kambe, A.; Yamamoto, Y.; Arulmozhiraja, S.; Ito, S.; Nakagawa, Y.; Tokiwa, H.; Nakano, S.; Shimano, H. Elucidation of Molecular Mechanism of a Selective PPARα Modulator, Pemafibrate, through Combinational Approaches of X-ray Crystallography, Thermodynamic Analysis, and First-Principle Calculations. Int. J. Mol. Sci. 2020, 21, 361. [Google Scholar] [CrossRef]
- Arai, H.; Yamashita, S.; Yokote, K.; Araki, E.; Suganami, H.; Ishibashi, S. Efficacy and Safety of Pemafibrate Versus Fenofibrate in Patients with High Triglyceride and Low HDL Cholesterol Levels: A Multicenter, Pla-cebo-Controlled, Double-Blind, Randomized Trial. J. Atheroscler. Thromb. 2018, 25, 521–538. [Google Scholar] [CrossRef] [PubMed]
- Fruchart, J.C.; Santos, R.D.; Aguilar-Salinas, C.; Aikawa, M.; Al Rasadi, K.; Amarenco, P. The selective peroxisome prolifera-tor-activated receptor alpha modulator (SPPARMα) paradigm: Conceptual framework and therapeutic potential: A consensus statement from the International Atherosclerosis Society (IAS) and the Residual Risk Reduction Initiative (R3i) Foundation. Cardiovasc. Diabetol. 2019, 18, 71. [Google Scholar]
- Pradhan, A.D.; Paynter, N.P.; Everett, B.M.; Glynn, R.J.; Amarenco, P.; Elam, M. Rationale and design of the Pemafibrate to Reduce Cardiovascular Outcomes by Reducing Triglycerides in Patients with Diabetes (PROMINENT) study. Am Heart J. 2018, 206, 80–93. [Google Scholar] [CrossRef]
- Ida, S.; Kaneko, R.; Murata, K. Efficacy and safety of pemafibrate administration in patients with dyslipidemia: A systematic review and meta-analysis. Cardiovasc. Diabetol. 2019, 18, 1–14. [Google Scholar] [CrossRef]
- Viney, N.J.; van Capelleveen, J.C.; Geary, R.S.; Xia, S.; A Tami, J.; Yu, R.Z.; Marcovina, S.M.; Hughes, S.G.; Graham, M.J.; Crooke, R.M.; et al. Antisense oligonucleotides targeting apolipoprotein(a) in people with raised lipoprotein(a): Two randomised, double-blind, placebo-controlled, dose-ranging trials. Lancet 2016, 388, 2239–2253. [Google Scholar] [CrossRef]
- Viney, N.J.; Yeang, C.; Yang, X.; Xia, S.; Witztum, J.L.; Tsimikas, S. Relationship between “LDL-C”, estimated true LDL-C, apolipo-protein B-100, and PCSK9 levels following lipoprotein(a) lowering with an antisense oligonucleotide. J. Clin. Lipidol. 2018, 12, 702–710. [Google Scholar] [CrossRef]
- Graham, M.J.; Viney, N.; Crooke, R.M.; Tsimikas, S. Antisense inhibition of apolipoprotein (a) to lower plasma lipoprotein (a) levels in humans. J. Lipid Res. 2016, 57, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Viney, N.J.; Hughes, S.G.; Singleton, W.; Graham, M.J.; Baker, B.F.; Burkey, J.L.; Yang, Q.; Marcovina, S.M.; Geary, R.S.; et al. Antisense therapy targeting apolipoprotein(a): A randomised, double-blind, placebo-controlled phase 1 study. Lancet 2015, 386, 1472–1483. [Google Scholar] [CrossRef] [PubMed]
- Tardif, J.C.; Karwatowska-Prokopczuk, E.; Amour, E.S.; Ballantyne, C.M.; Shapiro, M.D.; Moriarty, P.M.; Baum, S.J.; Hurh, E.; Bartlett, V.J.; Kingsbury, J.; et al. lipoprotein C-III reduction in subjects with moderate hypertriglyceridaemia and at high cardiovascular risk. Eur Heart J. 2022, 43, 1401–1412. [Google Scholar] [CrossRef]
- Ionis Pharmaceuticals, Inc. Akcea Therapeutics. A Randomized, Double-Blind, Placebo-Controlled, Phase 3 Study of AKCEA-APOCIII-LRx Administered Subcutaneously to Patients with Familial Chylomicronemia Syndrome (FCS). clinicaltri-als.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04568434 (accessed on 31 March 2023).
- Akcea Therapeutics, Ionis Pharmaceuticals, Inc. A Randomized, Double-Blind, Placebo-Controlled, with an Open Label Ex-tension, Phase 2/3 Study of ISIS 304801 Administered Subcutaneously to Patients with Familial Partial Lipodystrophy. clini-caltrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02527343 (accessed on 31 March 2023).
- Novartis Pharmaceuticals. A Randomized Double-blind, Placebo-controlled, Multicenter Trial Assessing the Impact of Lipo-protein (a) Lowering with Pelacarsen (TQJ230) on Major Cardiovascular Events in Patients with Established Cardiovascular Disease. clinicaltrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04023552 (accessed on 31 March 2023).
- Michael Gibson, C.; Korjian, S.; Tricoci, P.; Daaboul, Y.; Yee, M.; Jain, P. Safety and Tolerability of CSL112, a Reconstituted, Infusible, Plasma-Derived Apolipoprotein A-I, after Acute Myocardial Infarction: The AEGIS-I Trial (ApoA-I Event Reducing in Ischemic Syndromes I). Circulation 2016, 134, 1918–1930. [Google Scholar] [CrossRef] [PubMed]
- Rader, D.J. Apolipoprotein A-I Infusion Therapies for Coronary Disease: Two outs in the ninth inning and swinging for the fences. JAMA Cardiol. 2018, 3, 799–801. [Google Scholar] [CrossRef] [PubMed]
- Kingwell, B.A.; Nicholls, S.J.; Velkoska, E.; Didichenko, S.A.; Duffy, D.; Korjian, S.; Gibson, C.M. Antiatherosclerotic Effects of CSL112 Mediated by Enhanced Cholesterol Efflux Capacity. J. Am. Heart Assoc. 2022, 11, e024754. [Google Scholar] [CrossRef]
- Gibson, C.M.; Kastelein, J.J.; Phillips, A.T.; Aylward, P.E.; Yee, M.K.; Tendera, M.; Nicholls, S.J.; Pocock, S.; Goodman, S.G.; Alexander, J.H.; et al. Rationale and design of ApoA-I Event Reducing in Ischemic Syndromes II (AEGIS-II): A phase 3, multicenter, double-blind, randomized, placebo-controlled, parallel-group study to investigate the efficacy and safety of CSL112 in subjects after acute myocardial infarction. Am. Heart J. 2020, 231, 121–127. [Google Scholar] [CrossRef]
- Nicholls, S.; Puri, R.; Ballantyne, C.M.; Jukema, J.W.; Kastelein, J.J.P.; Koenig, W.; Wright, R.S.; Kallend, D.; Wijngaard, P.; Borgman, M.; et al. Effect of Infusion of High-Density Lipoprotein Mimetic Containing Recombinant Apolipoprotein A-I Milano on Coronary Disease in Patients With an Acute Coronary Syndrome in the MILANO-PILOT Trial. JAMA Cardiol. 2018, 3, 806–814. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Andrews, J.; Kastelein, J.J.P.; Merkely, B.; Nissen, S.E.; Ray, K.K.; Schwartz, G.G.; Worthley, S.G.; Keyserling, C.; Dasseux, J.L.; et al. Effect of serial infusions of CER-001, a pre-beta high-density lipoprotein mimetic, on coronary atherosclerosis in patients following acute coronary syndromes in the CER-001 atherosclerosis regression acute coronary syndrome trial: A randomized clinical trial. JAMA Cardiol. 2018, 3, 815–822. [Google Scholar]
- Tall, A.R.; Rader, D.J. Trials and Tribulations of CETP Inhibitors. Circ. Res. 2018, 122, 106–112. [Google Scholar] [CrossRef]
- Zhu, Y.; Xian, X.; Wang, Z.; Bi, Y.; Chen, Q.; Han, X.; Tang, D.; Chen, R. Research Progress on the Relationship between Atherosclerosis and Inflammation. Biomolecules 2018, 8, 80. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; MacFadyen, J.G.; Everett, B.M.; Libby, P.; Thuren, T.; Glynn, R.J.; Kastelein, J.; Koenig, W.; Genest, J.; Lorenzatti, A.; et al. Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: A secondary analysis from the CANTOS randomised controlled trial. Lancet 2017, 391, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Tardif, J.-C.; Kouz, S.; Waters, D.D.; Bertrand, O.F.; Diaz, R.; Maggioni, A.P.; Pinto, F.J.; Ibrahim, R.; Gamra, H.; Kiwan, G.S.; et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N. Engl. J. Med. 2019, 381, 2497–2505. [Google Scholar] [CrossRef] [PubMed]
- Nidorf, S.M.; Fiolet, A.T.L.; Mosterd, A.; Eikelboom, J.W.; Schut, A.; Opstal, T.S.J.; The, S.H.K.; Xu, X.-F.; Ireland, M.A.; Lenderink, T.; et al. Colchicine in Patients with Chronic Coronary Disease. N. Engl. J. Med. 2020, 383, 1838–1847. [Google Scholar] [CrossRef]
- Vaidya, K.; Arnott, C.; Martínez, G.J.; Ng, B.; McCormack, S.; Sullivan, D.R.; Celermajer, D.S.; Patel, S. Colchicine Therapy and Plaque Stabilization in Patients With Acute Coronary Syndrome. JACC Cardiovasc. Imaging 2017, 11, 305–316. [Google Scholar] [CrossRef]
- Andreis, A.; Imazio, M.; Avondo, S.; Casula, M.; Paneva, E.; Piroli, F.; De Ferrari, G.M. Adverse events of colchicine for cardiovascular diseases: A comprehensive meta-analysis of 14 188 patients from 21 randomized controlled trials. J. Cardiovasc. Med. 2021, 22, 637–644. [Google Scholar] [CrossRef]
- Leung, Y.Y.; Yao Hui, L.L.; Kraus, V.B. Colchicine—Update on mechanisms of action and therapeutic uses. Semin. Arthritis Rheum. 2015, 45, 341–350. [Google Scholar] [CrossRef]
- Martínez, G.J.; Robertson, S.; Barraclough, J.; Xia, Q.; Mallat, Z.; Bursill, C.; Celermajer, D.S.; Patel, S. Colchicine Acutely Suppresses Local Cardiac Production of Inflammatory Cytokines in Patients With an Acute Coronary Syndrome. J. Am. Heart Assoc. 2015, 4, e002128. [Google Scholar] [CrossRef]
- Casey, A.; Quinn, S.; McAdam, B.; Kennedy, M.; Sheahan, R. Colchicine-regeneration of an old drug. Ir. J. Med. Sci. 2023, 192, 115–123. [Google Scholar] [CrossRef]
- Addamo, A.M. CIRT Investigators. Low-dose methotrexate for the prevention of atherosclerotic events. N. Engl. J. Med. 2019, 380, 752–762. [Google Scholar] [CrossRef]
- Tsiantoulas, D.; Diehl, C.; Witztum, J.L.; Binder, C.J. B Cells and Humoral Immunity in Atherosclerosis. Circ. Res. 2014, 114, 1743–1756. [Google Scholar] [CrossRef] [PubMed]
- Chyu, K.-Y.; Dimayuga, P.C.; Shah, P.K. Vaccine against arteriosclerosis: An update. Ther. Adv. Vaccines 2017, 5, 39–47. [Google Scholar] [CrossRef]
- Gisterå, A.; Hermansson, A.; Strodthoff, D.; Klement, M.L.; Hedin, U.; Fredrikson, G.N.; Nilsson, J.; Hansson, G.K.; Ketelhuth, D.F.J. Vaccination against T-cell epitopes of native ApoB100 reduces vascular inflammation and disease in a humanized mouse model of atherosclerosis. J. Intern. Med. 2017, 281, 383–397. [Google Scholar] [CrossRef]
- Wolf, D.; Gerhardt, T.; Ley, K. Vaccination to Prevent Cardiovascular Disease. In Platelets, Haemostasis and Inflammation Cham; Zirlik, A., Bode, C., Gawaz, M., Eds.; Springer International Publishing: New York, NY, USA, 2017; pp. 29–52. [Google Scholar]
- MD JC, Cambridge University Hospitals NHS Foundation Trust, University of Cambridge. Low Dose Interleukin-2 in Patients With Stable Ischaemic Heart Disease and Acute Coronary Syndromes. clinicaltrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03113773 (accessed on 31 March 2023).
- Moore, J.X.; Chaudhary, N.; Akinyemiju, T. Metabolic Syndrome Prevalence by Race/Ethnicity and Sex in the United States, National Health and Nutrition Examination Survey, 1988–2012. Prev. Chronic Dis. 2017, 14, E24. [Google Scholar] [CrossRef] [PubMed]
- Blaum, C.; Brunner, F.J.; Goßling, A.; Kröger, F.; Bay, B.; Lorenz, T.; Graef, A.; Zeller, T.; Schnabel, R.; Clemmensen, P.; et al. Target Populations and Treatment Cost for Bempedoic Acid and PCSK9 Inhibitors: A Simulation Study in a Contemporary CAD Cohort. Clin. Ther. 2021, 43, 1583–1600. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumric, M.; Urlic, H.; Bozic, J.; Vilovic, M.; Ticinovic Kurir, T.; Glavas, D.; Miric, D.; Zanchi, J.; Bradaric-Slujo, A.; Lozo, M.; et al. Emerging Therapies for the Treatment of Atherosclerotic Cardiovascular Disease: From Bench to Bedside. Int. J. Mol. Sci. 2023, 24, 8062. https://doi.org/10.3390/ijms24098062
Kumric M, Urlic H, Bozic J, Vilovic M, Ticinovic Kurir T, Glavas D, Miric D, Zanchi J, Bradaric-Slujo A, Lozo M, et al. Emerging Therapies for the Treatment of Atherosclerotic Cardiovascular Disease: From Bench to Bedside. International Journal of Molecular Sciences. 2023; 24(9):8062. https://doi.org/10.3390/ijms24098062
Chicago/Turabian StyleKumric, Marko, Hrvoje Urlic, Josko Bozic, Marino Vilovic, Tina Ticinovic Kurir, Duska Glavas, Dino Miric, Jaksa Zanchi, Anteo Bradaric-Slujo, Mislav Lozo, and et al. 2023. "Emerging Therapies for the Treatment of Atherosclerotic Cardiovascular Disease: From Bench to Bedside" International Journal of Molecular Sciences 24, no. 9: 8062. https://doi.org/10.3390/ijms24098062
APA StyleKumric, M., Urlic, H., Bozic, J., Vilovic, M., Ticinovic Kurir, T., Glavas, D., Miric, D., Zanchi, J., Bradaric-Slujo, A., Lozo, M., & Borovac, J. A. (2023). Emerging Therapies for the Treatment of Atherosclerotic Cardiovascular Disease: From Bench to Bedside. International Journal of Molecular Sciences, 24(9), 8062. https://doi.org/10.3390/ijms24098062








