miR-18a and miR-106a Signatures in Plasma Small EVs Are Promising Biomarkers for Early Detection of Pancreatic Ductal Adenocarcinoma
Abstract
1. Introduction
2. Results
2.1. Several miRNAs Are Significantly Enriched in the Small EVs Derived from PDAC Patient Plasma
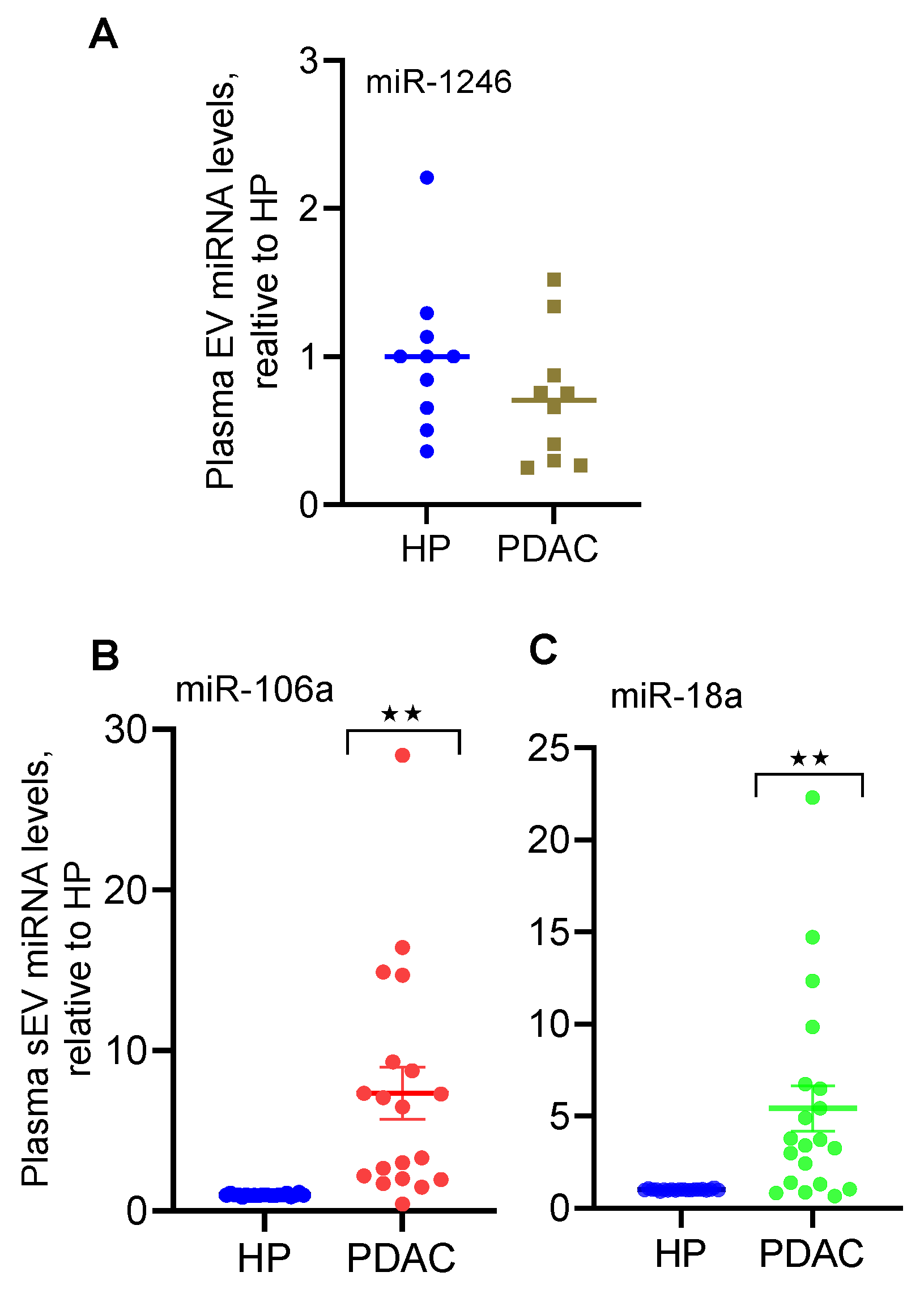
2.2. qRT-PCR Confirms the Elevated Levels of miR-18a and miR-106a in Plasma Small EVs Derived from PDAC Patients
2.3. The Levels of miR-18a and miR-106a Are Significantly Elevated in Immunoprecipitants of Plasma Small EVs from PDAC Versus Those from Colon Cancer and Healthy Controls
3. Discussion
4. Materials and Methods
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, M. Pancreatic cancer. N. Engl. J. Med. 2010, 362, 1605–1617. [Google Scholar] [CrossRef]
- Ryan, D.P.; Hong, T.S.; Bardeesy, N. Pancreatic adenocarcinoma. N. Engl. J. Med. 2014, 371, 1039–1049. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Arora, S.; Singh, S.; Bhardwaj, A.; Averett, C.; Singh, A.P. MicroRNAs in pancreatic malignancy: Progress and promises. Cancer Lett. 2014, 347, 167–174. [Google Scholar] [CrossRef]
- Papaconstantinou, I.G.; Lykoudis, P.M.; Gazouli, M.; Manta, A.; Polymeneas, G.; Voros, D. A review on the role of microRNA in biology, diagnosis, and treatment of pancreatic adenocarcinoma. Pancreas 2012, 41, 671–677. [Google Scholar] [CrossRef]
- Costello, E.; Greenhalf, W.; Neoptolemos, J.P. New biomarkers and targets in pancreatic cancer and their application to treatment. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 435–444. [Google Scholar] [CrossRef]
- Que, R.; Ding, G.; Chen, J.; Cao, L. Analysis of serum exosomal microRNAs and clinicopathologic features of patients with pancreatic adenocarcinoma. World J. Surg. Oncol. 2013, 11, 219. [Google Scholar] [CrossRef]
- Madhavan, B.; Yue, S.; Galli, U.; Rana, S.; Gross, W.; Muller, M.; Giese, N.A.; Kalthoff, H.; Becker, T.; Buchler, M.W.; et al. Combined evaluation of a panel of protein and miRNA serum-exosome biomarkers for pancreatic cancer diagnosis increases sensitivity and specificity. Int. J. Cancer 2015, 136, 2616–2627. [Google Scholar] [CrossRef]
- Takahasi, K.; Iinuma, H.; Wada, K.; Minezaki, S.; Kawamura, S.; Kainuma, M.; Ikeda, Y.; Shibuya, M.; Miura, F.; Sano, K. Usefulness of exosome-encapsulated microRNA-451a as a minimally invasive biomarker for prediction of recurrence and prognosis in pancreatic ductal adenocarcinoma. J. Hepato-Biliary-Pancreat. Sci. 2018, 25, 155–161. [Google Scholar] [CrossRef]
- Lai, X.; Wang, M.; McElyea, S.D.; Sherman, S.; House, M.; Korc, M. A microRNA signature in circulating exosomes is superior to exosomal glypican-1 levels for diagnosing pancreatic cancer. Cancer Lett. 2017, 393, 86–93. [Google Scholar] [CrossRef]
- Kofanova, O.; Henry, E.; Aguilar Quesada, R.; Bulla, A.; Navarro Linares, H.; Lescuyer, P.; Shea, K.; Stone, M.; Tybring, G.; Bellora, C.; et al. IL8 and IL16 levels indicate serum and plasma quality. Clin. Chem. Lab. Med. 2018, 56, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Hannafon, B.N.; Trigoso, Y.D.; Calloway, C.L.; Zhao, Y.D.; Lum, D.H.; Welm, A.L.; Zhao, Z.J.; Blick, K.E.; Dooley, W.C.; Ding, W.Q. Plasma exosome microRNAs are indicative of breast cancer. Breast Cancer Res. BCR 2016, 18, 90. [Google Scholar] [CrossRef]
- Xu, Y.F.; Hannafon, B.N.; Zhao, Y.D.; Postier, R.G.; Ding, W.Q. Plasma exosome miR-196a and miR-1246 are potential indicators of localized pancreatic cancer. Oncotarget 2017, 8, 77028–77040. [Google Scholar] [CrossRef]
- Manri, C.; Yokoi, T.; Nishida, H. Size-Selective Harvesting of Extracellular Vesicles for Strategic Analyses Towards Tumor Diagnoses. Appl. Biochem. Biotechnol. 2017, 182, 609–623. [Google Scholar] [CrossRef]
- Thind, A.; Wilson, C. Exosomal miRNAs as cancer biomarkers and therapeutic targets. J. Extracell. Vesicles 2016, 5, 31292. [Google Scholar] [CrossRef]
- Hannafon, B.N.; Ding, W.Q. Intercellular Communication by Exosome-Derived microRNAs in Cancer. Int. J. Mol. Sci. 2013, 14, 14240–14269. [Google Scholar] [CrossRef]
- Salehi, M.; Sharifi, M. Exosomal miRNAs as novel cancer biomarkers: Challenges and opportunities. J. Cell. Physiol. 2018, 233, 6370–6380. [Google Scholar] [CrossRef]
- Xu, Y.F.; Xu, X.; Bhandari, K.; Gin, A.; Rao, C.V.; Morris, K.T.; Hannafon, B.N.; Ding, W.Q. Isolation of extra-cellular vesicles in the context of pancreatic adenocarcinomas: Addition of one stringent filtration step improves recovery of specific microRNAs. PLoS ONE 2021, 16, e0259563. [Google Scholar] [CrossRef]
- Yang, Z.; LaRiviere, M.J.; Ko, J.; Till, J.E.; Christensen, T.; Yee, S.S.; Black, T.A.; Tien, K.; Lin, A.; Shen, H.; et al. A Multianalyte Panel Consisting of Extracellular Vesicle miRNAs and mRNAs, cfDNA, and CA19-9 Shows Utility for Diagnosis and Staging of Pancreatic Ductal Adenocarcinoma. Clin. Cancer Res. 2020, 26, 3248–3258. [Google Scholar] [CrossRef]
- Xu, Y.F.; Hannafon, B.N.; Khatri, U.; Gin, A.; Ding, W.Q. The origin of exosomal miR-1246 in human cancer cells. RNA Biol. 2019, 16, 770–784. [Google Scholar] [CrossRef]
- Xu, Y.F.; Xu, X.; Gin, A.; Nshimiyimana, J.D.; Mooers, B.H.M.; Caputi, M.; Hannafon, B.N.; Ding, W.Q. SRSF1 regulates exosome microRNA enrichment in human cancer cells. Cell Commun. Signal. 2020, 18, 130. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Jiang, T.; Liu, Y.; Huai, G.; Lan, C.; Li, G.; Jia, G.; Wang, K.; Yang, M. Droplet digital PCR-based circulating microRNA detection serve as a promising diagnostic method for gastric cancer. BMC Cancer 2018, 18, 676. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.M.S.; Barros-Filho, M.C.; Wong, D.V.T.; Mello, J.B.H.; Nobre, L.M.S.; Wanderley, C.W.S.; Lucetti, L.T.; Muniz, H.A.; Paiva, I.K.D.; Kuasne, H.; et al. Circulating let-7e-5p, miR-106a-5p, miR-28-3p, and miR-542-5p as a Promising microRNA Signature for the Detection of Colorectal Cancer. Cancers 2021, 13, 1493. [Google Scholar] [CrossRef] [PubMed]
- Sharova, E.; Grassi, A.; Marcer, A.; Ruggero, K.; Pinto, F.; Bassi, P.; Zanovello, P.; Zattoni, F.; D’Agostino, D.M.; Iafrate, M.; et al. A circulating miRNA assay as a first-line test for prostate cancer screening. Br. J. Cancer 2016, 114, 1362–1366. [Google Scholar] [CrossRef]
- Morimura, R.; Komatsu, S.; Ichikawa, D.; Takeshita, H.; Tsujiura, M.; Nagata, H.; Konishi, H.; Shiozaki, A.; Ikoma, H.; Okamoto, K.; et al. Novel diagnostic value of circulating miR-18a in plasma of patients with pancreatic cancer. Br. J. Cancer 2011, 105, 1733–1740. [Google Scholar] [CrossRef]
- Komatsu, S.; Ichikawa, D.; Takeshita, H.; Morimura, R.; Hirajima, S.; Tsujiura, M.; Kawaguchi, T.; Miyamae, M.; Nagata, H.; Konishi, H.; et al. Circulating miR-18a: A sensitive cancer screening biomarker in human cancer. In Vivo 2014, 28, 293–297. [Google Scholar]
- Prinz, C.; Fehring, L.; Frese, R. MicroRNAs as Indicators of Malignancy in Pancreatic Ductal Adenocarcinoma (PDAC) and Cystic Pancreatic Lesions. Cells 2022, 11, 2374. [Google Scholar] [CrossRef]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef]
| Plasma Sample | Age (years) | Sex a | Race b | Diagnosis | Tumor Size Largest Dimension | Stage c |
|---|---|---|---|---|---|---|
| PL1 | 72 | F | W | PDAC | 2.5 cm | IIA |
| PL2 | 43 | F | W | PDAC | 1.8 cm | I |
| PL3 | 50 | F | W | PDAC | 1.8 cm | I |
| PL4 | 52 | M | W | PDAC | 1.8 cm | I |
| PL5 | 55 | M | W | PDAC | 1.6 cm | I |
| PL6 | 62 | M | W | PDAC | 2.1 cm | IIA |
| PL7 | 60 | M | W | PDAC | NA | IIA |
| PL8 | 47 | F | W | PDAC | NA | IIA |
| PL9 | 55 | F | W | PDAC | NA | IIA |
| PL10 | 67 | F | W | PDAC | 0.8 cm | I |
| PL11 | 50 | M | W | PDAC | NA | I |
| PL12 | 59 | M | W | PDAC | 1.8 cm | I |
| PL13 | 55 | M | B | PDAC | 1.8 cm | I |
| PL14 | 67 | M | W | PDAC | NA | IIA |
| PL15 | 63 | F | W | PDAC | 3.2 cm | IIA |
| PL16 | 69 | M | W | PDAC | 3.1 cm | IIA |
| PL17 | 60 | F | W | PDAC | 2.8 cm | IIA |
| PL18 | 67 | F | W | PDAC | 3.3 cm | IIA |
| PL19 | 66 | M | W | PDAC | 2.6 cm | IIA |
| PL20 | 63 | M | B | PDAC | 1.5 cm | IIA |
| Mean Age | 59.1 |
| microRNA | PDAC Mean Reads | Healthy Mean Reads | Fold Change | Adjusted p-Value |
|---|---|---|---|---|
| miR-502 | 144 ± 116 | 0 ± 0 | 371 | 0.023 |
| miR-18a | 186 ± 110 | 1 ± 1 | 97 | 0.011 |
| miR-106a | 153 ± 66 | 1 ± 1 | 147 | 0.047 |
| miR-660 | 445 ± 50 | 144 ± 3 | 33 | 0.011 |
| miR-21 | 207.461 ± 52,898 | 125.842 ± 3966 | 1 | 0.733 |
| microRNA | Primer |
|---|---|
| miR-502 | 5′-CATCCTTGCTATCTGGGTGCTA-3′ |
| miR-18a | 5′-GCTAAGGTGCATCTAGTGCAGATAG-3′ |
| miR-106a | 5′-CAAAAGTGCTTACAGTGCAGGTAG-3′ |
| miR-1246 | 5′-GCGCGATGGATTTTTGGAGCAG-3′ |
| microRNA | Healthy Control Mean ± SEM | PDAC Mean ± SEM | Colon Cancer Mean ± SEM |
|---|---|---|---|
| miR-18a | 1.0 ± 0.09 | 3.05 ± 0.81 * | 0.44 ± 0.06 |
| miR-106a | 1.0 ± 0.12 | 4.60 ± 0.79 ** | 0.74 ± 0.05 |
| miR-196-a | 1.0 ± 0.25 | 1.59 ± 0.45 | 1.27 ± 0.14 |
| Plasma Sample | Age (years) | Sex a | Race b | Diagnosis c | Stage d |
|---|---|---|---|---|---|
| CoP1 | 65 | F | W | CRC | IIA |
| CoP2 | 68 | F | W | CC | II |
| CoP3 | 67 | F | W | CC | IIIA |
| CoP4 | 41 | F | W | CC | IIIC |
| CoP5 | 53 | F | W | RC | I |
| CoP6 | 56 | F | W | CC | I |
| CoP7 | 43 | M | W | CRC | IIA |
| CoP8 | 67 | M | W | CRC | IIA |
| CoP9 | 49 | M | W | RC | NA |
| CoP10 | 41 | M | W | CC | IIIB |
| Mean Age | 55 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.; Bhandari, K.; Xu, C.; Morris, K.; Ding, W.-Q. miR-18a and miR-106a Signatures in Plasma Small EVs Are Promising Biomarkers for Early Detection of Pancreatic Ductal Adenocarcinoma. Int. J. Mol. Sci. 2023, 24, 7215. https://doi.org/10.3390/ijms24087215
Xu X, Bhandari K, Xu C, Morris K, Ding W-Q. miR-18a and miR-106a Signatures in Plasma Small EVs Are Promising Biomarkers for Early Detection of Pancreatic Ductal Adenocarcinoma. International Journal of Molecular Sciences. 2023; 24(8):7215. https://doi.org/10.3390/ijms24087215
Chicago/Turabian StyleXu, Xiaohui, Kritisha Bhandari, Chao Xu, Katherine Morris, and Wei-Qun Ding. 2023. "miR-18a and miR-106a Signatures in Plasma Small EVs Are Promising Biomarkers for Early Detection of Pancreatic Ductal Adenocarcinoma" International Journal of Molecular Sciences 24, no. 8: 7215. https://doi.org/10.3390/ijms24087215
APA StyleXu, X., Bhandari, K., Xu, C., Morris, K., & Ding, W.-Q. (2023). miR-18a and miR-106a Signatures in Plasma Small EVs Are Promising Biomarkers for Early Detection of Pancreatic Ductal Adenocarcinoma. International Journal of Molecular Sciences, 24(8), 7215. https://doi.org/10.3390/ijms24087215









