Efficacy of Essential Oils in Relieving Cancer Pain: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Objectives and Protocol
2.2. Information Sources
2.3. Search Strategy
2.4. Study Selection
2.5. Data Synthesis, Assessment of the Risk of Bias and Critical Appraisal
2.6. Statistical Analysis and Effect Measures
3. Results
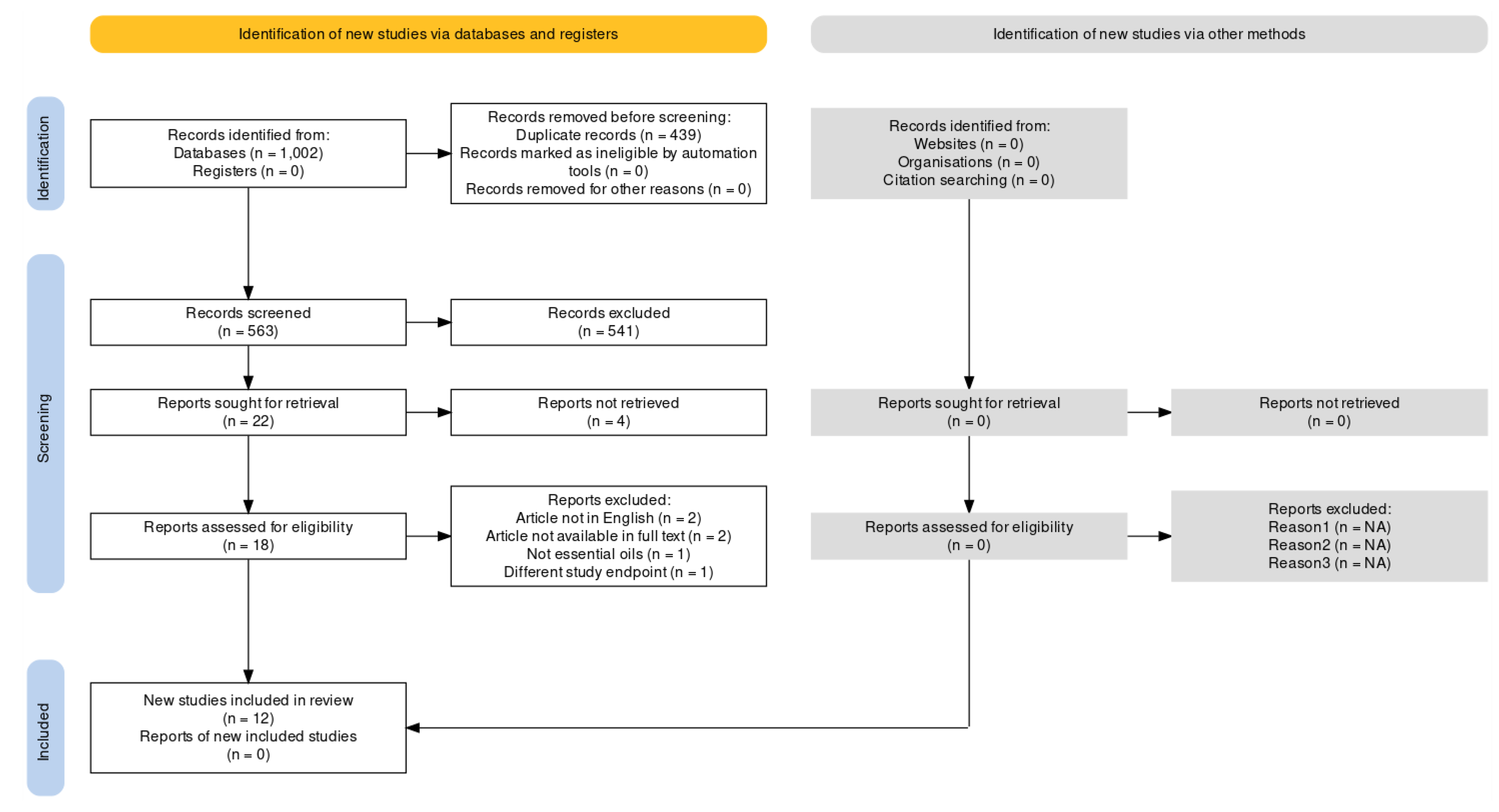
3.1. Studies Selection
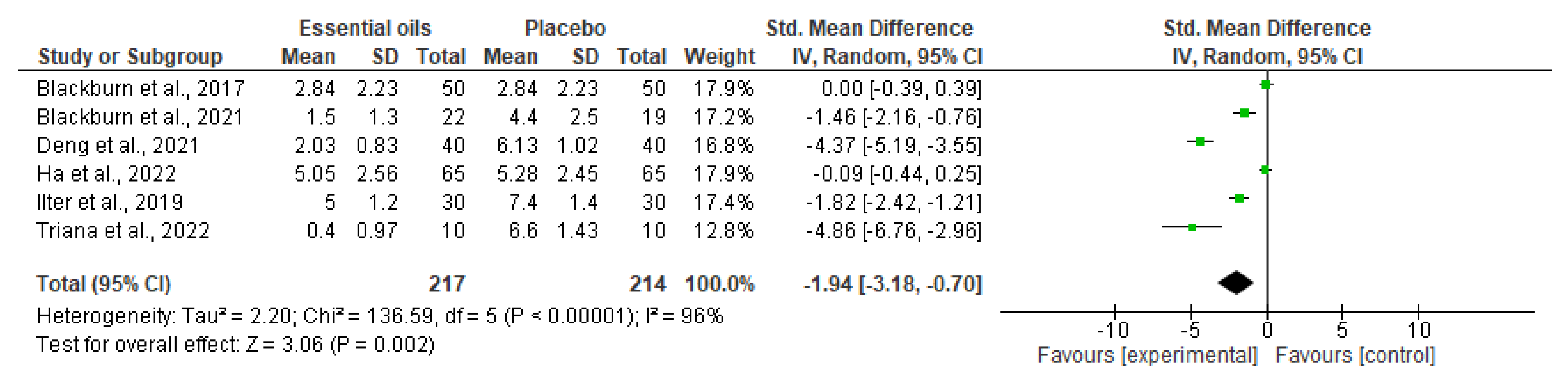
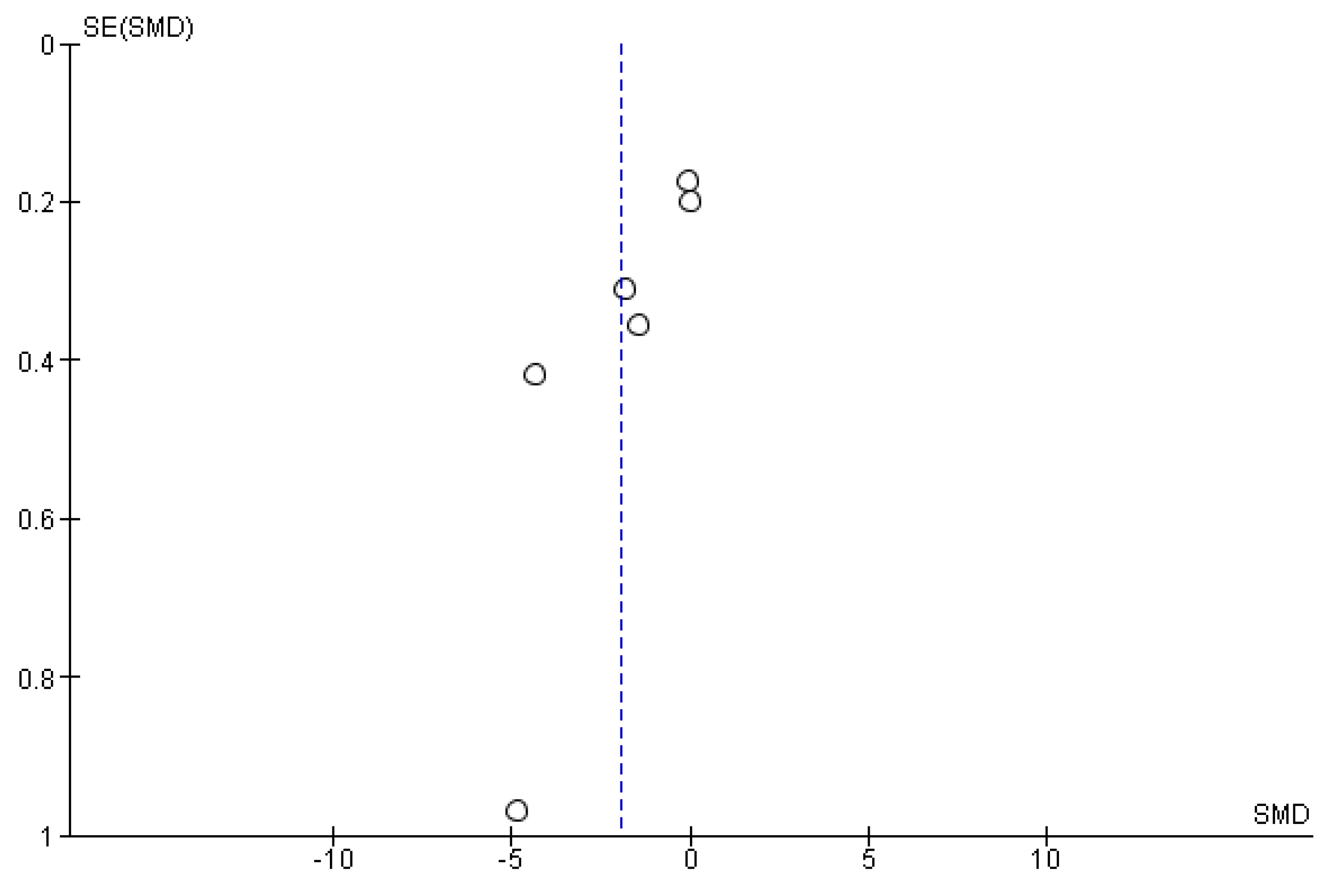
3.2. Data Synthesis, Critical Appraisal, and Meta-Analysis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Portenoy, R.K.; Thaler, H.T.; Kornblith, A.B.; McCarthy Lepore, J.; Friedlander-Klar, H.; Coyle, N.; Smart-Curley, T.; Kemeny, N.; Norton, L.; Hoskins, W.; et al. Symptom prevalence, characteristics and distress in a cancer population. Qual. Life Res. 1994, 3, 183–189. [Google Scholar] [CrossRef]
- Kwekkeboom, K.L. Cancer Symptom Cluster Management. Semin. Oncol. Nurs. 2016, 32, 373–382. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2022–2024; American Cancer Society: Atlanta, GA, USA, 2022. [Google Scholar]
- Mantovani, A.; Allavena, P.; Sica, A.; Balkwill, F. Cancer-related inflammation. Nature 2008, 454, 436–444. [Google Scholar] [CrossRef]
- Van den Beuken-van Everdingen, M.H.; de Rijke, J.M.; Kessels, A.G.; Schouten, H.C.; van Kleef, M.; Patijn, J. Prevalence of pain in patients with cancer: A systematic review of the past 40 years. Ann. Oncol. 2007, 18, 1437–1449. [Google Scholar] [CrossRef]
- Van den Beuken-van Everdingen, M.H.; Hochstenbach, L.M.; Joosten, E.A.; Tjan-Heijnen, V.C.; Janssen, D.J. Update on Prevalence of Pain in Patients with Cancer: Systematic Review and Meta-Analysis. J. Pain Symptom Manag. 2016, 51, 1070–1090.e1079. [Google Scholar] [CrossRef]
- Breivik, H.; Cherny, N.; Collett, B.; de Conno, F.; Filbet, M.; Foubert, A.J.; Cohen, R.; Dow, L. Cancer-related pain: A pan-European survey of prevalence, treatment, and patient attitudes. Ann. Oncol. 2009, 20, 1420–1433. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.M.; Weiss, S.M. Nonpharmacological Interventions with Chronic Cancer Pain in Adults. Cancer Control 2000, 7, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Haenen, V.; Evenepoel, M.; De Baerdemaecker, T.; Meeus, M.; Devoogdt, N.; Morlion, B.; Dams, L.; Van Dijck, S.; Van der Gucht, E.; De Vrieze, T.; et al. Pain prevalence and characteristics in survivors of solid cancers: A systematic review and meta-analysis. Support. Care Cancer 2022, 31, 85. [Google Scholar] [CrossRef]
- Tan, H.S.; Plichta, J.K.; Kong, A.; Tan, C.W.; Hwang, S.; Sultana, R.; Wright, M.C.; Sia, A.T.H.; Sng, B.L.; Habib, A.S. Risk factors for persistent pain after breast cancer surgery: A multicentre prospective cohort study. Anaesthesia 2023. [Google Scholar] [CrossRef]
- Schappert, S.M.; Burt, C.W. Ambulatory care visits to physician offices, hospital outpatient departments, and emergency departments: United States, 2001–2002. Vital Health Stat. 2006, 159, 1–66. [Google Scholar]
- Russo, M.M.; Sundaramurthi, T. An Overview of Cancer Pain: Epidemiology and Pathophysiology. Semin. Oncol. Nurs. 2019, 35, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Treede, R.D.; Jensen, T.S.; Campbell, J.N.; Cruccu, G.; Dostrovsky, J.O.; Griffin, J.W.; Hansson, P.; Hughes, R.; Nurmikko, T.; Serra, J. Neuropathic pain: Redefinition and a grading system for clinical and research purposes. Neurology 2008, 70, 1630–1635. [Google Scholar] [CrossRef] [PubMed]
- Bajwa, S.J.; Haldar, R. Pain management following spinal surgeries: An appraisal of the available options. J. Craniovertebral Junction Spine 2015, 6, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Masri, R.; Keller, A. Chronic pain following spinal cord injury. Adv. Exp. Med. Biol. 2012, 760, 74–88. [Google Scholar] [CrossRef]
- Scuteri, D.; Mantovani, E.; Tamburin, S.; Sandrini, G.; Corasaniti, M.T.; Bagetta, G.; Tonin, P. Opioids in Post-stroke Pain: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2020, 11, 587050. [Google Scholar] [CrossRef]
- Portenoy, R.K.; Ahmed, E. Cancer Pain Syndromes. Hematol. Oncol. Clin. N. Am. 2018, 32, 371–386. [Google Scholar] [CrossRef]
- Farquhar-Smith, P. Chemotherapy-induced neuropathic pain. Curr. Opin. Support. Palliat. Care 2011, 5, 1–7. [Google Scholar] [CrossRef]
- Quintão, N.L.M.; Santin, J.R.; Stoeberl, L.C.; Corrêa, T.P.; Melato, J.; Costa, R. Pharmacological Treatment of Chemotherapy-Induced Neuropathic Pain: PPARγ Agonists as a Promising Tool. Front. Neurosci. 2019, 13, 907. [Google Scholar] [CrossRef]
- Bayer, A.; Tadd, W. Unjustified exclusion of elderly people from studies submitted to research ethics committee for approval: Descriptive study. BMJ 2000, 321, 992–993. [Google Scholar] [CrossRef]
- Scuteri, D.; Adornetto, A.; Rombolà, L.; Naturale, M.D.; De Francesco, A.E.; Esposito, S.; Zito, M.; Morrone, L.A.; Bagetta, G.; Tonin, P.; et al. Pattern of triptans use: A retrospective prescription study in Calabria, Italy. Neural Regen. Res. 2020, 15, 1340–1343. [Google Scholar] [CrossRef]
- Scuteri, D.; Corasaniti, M.T.; Tonin, P.; Bagetta, G. Eptinezumab for the treatment of migraine. Drugs Today 2019, 55, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Corasaniti, M.T.; Tonin, P.; Nicotera, P.; Bagetta, G. Role of CGRP pathway polymorphisms in migraine: A systematic review and impact on CGRP mAbs migraine therapy. J Headache Pain 2021, 22, 87. [Google Scholar] [CrossRef] [PubMed]
- Nørgaard, A.; Jensen-Dahm, C.; Gasse, C.; Hansen, E.S.; Waldemar, G. Psychotropic Polypharmacy in Patients with Dementia: Prevalence and Predictors. J. Alzheimer’s Dis. 2017, 56, 707–716. [Google Scholar] [CrossRef]
- Scuteri, D.; Berliocchi, L.; Rombolà, L.; Morrone, L.A.; Tonin, P.; Bagetta, G.; Corasaniti, M.T. Effects of Aging on Formalin-Induced Pain Behavior and Analgesic Activity of Gabapentin in C57BL/6 Mice. Front. Pharmacol. 2020, 11, 663. [Google Scholar] [CrossRef]
- McLachlan, A.J.; Hilmer, S.N.; Le Couteur, D.G. Variability in response to medicines in older people: Phenotypic and genotypic factors. Clin. Pharmacol. Ther. 2009, 85, 431–433. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.J.; Ismaila, N.; Bao, T.; Barton, D.; Ben-Arye, E.; Garland, E.L.; Greenlee, H.; Leblanc, T.; Lee, R.T.; Lopez, A.M.; et al. Integrative Medicine for Pain Management in Oncology: Society for Integrative Oncology–ASCO Guideline. J. Clin. Oncol. 2022, 40, 3998–4024. [Google Scholar] [CrossRef]
- Bardia, A.; Barton, D.L.; Prokop, L.J.; Bauer, B.A.; Moynihan, T.J. Efficacy of Complementary and Alternative Medicine Therapies in Relieving Cancer Pain: A Systematic Review. J. Clin. Oncol. 2006, 24, 5457–5464. [Google Scholar] [CrossRef]
- Rombolà, L.; Scuteri, D.; Watanabe, C.; Sakurada, S.; Hamamura, K.; Sakurada, T.; Tonin, P.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Role of 5-HT1A Receptor in the Anxiolytic-Relaxant Effects of Bergamot Essential Oil in Rodent. Int. J. Mol. Sci. 2020, 21, 2597. [Google Scholar] [CrossRef]
- Scuteri, D.; Cassano, R.; Trombino, S.; Russo, R.; Mizoguchi, H.; Watanabe, C.; Hamamura, K.; Katsuyama, S.; Komatsu, T.; Morrone, L.A.; et al. Development and Translation of NanoBEO, a Nanotechnology-Based Delivery System of Bergamot Essential Oil Deprived of Furocumarins, in the Control of Agitation in Severe Dementia. Pharmaceutics 2021, 13, 379. [Google Scholar] [CrossRef]
- Scuteri, D.; Hamamura, K.; Sakurada, T.; Watanabe, C.; Sakurada, S.; Morrone, L.A.; Rombolà, L.; Tonin, P.; Bagetta, G.; Corasaniti, M.T. Efficacy of Essential Oils in Pain: A Systematic Review and Meta-Analysis of Preclinical Evidence. Front. Pharmacol. 2021, 12, 640128. [Google Scholar] [CrossRef]
- Scuteri, D.; Sandrini, G.; Tamburin, S.; Corasaniti, M.T.; Nicotera, P.; Tonin, P.; Bagetta, G. Bergamot rehabilitation AgaINst agitation in dementia (BRAINAID): Study protocol for a randomized, double-blind, placebo-controlled trial to assess the efficacy of furocoumarin-free bergamot loaded in a nanotechnology-based delivery system of the essential oil in the treatment of agitation in elderly affected by severe dementia. Phytother. Res. 2021, 35, 5333–5338. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Rombolà, L.; Crudo, M.; Watanabe, C.; Mizoguchi, H.; Sakurada, S.; Hamamura, K.; Sakurada, T.; Morrone, L.A.; Tonin, P.; et al. Translational Value of the Transdermal Administration of Bergamot Essential Oil and of Its Fractions. Pharmaceutics 2022, 14, 1006. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Rombolà, L.; Crudo, M.; Watanabe, C.; Mizoguchi, H.; Sakurada, S.; Hamamura, K.; Sakurada, T.; Tonin, P.; Corasaniti, M.T.; et al. Preclinical Characterization of Antinociceptive Effect of Bergamot Essential Oil and of Its Fractions for Rational Translation in Complementary Therapy. Pharmaceutics 2022, 14, 312. [Google Scholar] [CrossRef]
- Hamamura, K.; Katsuyama, S.; Komatsu, T.; Scuteri, D.; Bagetta, G.; Aritake, K.; Sakurada, T. Behavioral Effects of Continuously Administered Bergamot Essential Oil on Mice With Partial Sciatic Nerve Ligation. Front. Pharmacol. 2020, 11, 1310. [Google Scholar] [CrossRef]
- Scuteri, D.; Crudo, M.; Rombolà, L.; Watanabe, C.; Mizoguchi, H.; Sakurada, S.; Sakurada, T.; Greco, R.; Corasaniti, M.T.; Morrone, L.A.; et al. Antinociceptive effect of inhalation of the essential oil of bergamot in mice. Fitoterapia 2018, 129, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Rombolà, L.; Hayashi, T.; Watanabe, C.; Sakurada, S.; Hamamura, K.; Sakurada, T.; Tonin, P.; Bagetta, G.; Morrone, L.A.; et al. Analgesic Characteristics of NanoBEO Released by an Airless Dispenser for the Control of Agitation in Severe Dementia. Molecules 2022, 27, 4987. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ Clin. Res. Ed. 2021, 372, n71. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Jones, J.; Hunter, D. Qualitative Research: Consensus methods for medical and health services research. BMJ 1995, 311, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, C.; Glanville, J.; Briscoe, S.; Littlewood, A.; Marshall, C.; Metzendorf, M.-I.; Noel-Storr, A.; Rader, T.; Shokraneh, F.; Thomas, J.; et al. Searching for and selecting studies. In Cochrane Handbook for Systematic Reviews of Interventions; Wiley: Hoboken, NJ, USA, 2019. [Google Scholar]
- McGowan, J.; Sampson, M.; Salzwedel, D.M.; Cogo, E.; Foerster, V.; Lefebvre, C. PRESS Peer Review of Electronic Search Strategies: 2015 Guideline Statement. J. Clin. Epidemiol. 2016, 75, 40–46. [Google Scholar] [CrossRef]
- Ryan, R. Cochrane Consumers and Communication Review Group: Data Synthesis and Analysis. Available online: http://cccrg.cochrane.org (accessed on 13 March 2019).
- Hultcrantz, M.; Rind, D.; Akl, E.A.; Treweek, S.; Mustafa, R.A.; Iorio, A.; Alper, B.S.; Meerpohl, J.J.; Murad, M.H.; Ansari, M.T.; et al. The GRADE Working Group clarifies the construct of certainty of evidence. J. Clin. Epidemiol. 2017, 87, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ Clin. Res. Ed. 2021, 372, n160. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res. Synth. Methods 2020, 12, 55–61. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Kacker, R. Random-effects model for meta-analysis of clinical trials: An update. Contemp. Clin. Trials 2007, 28, 105–114. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629. [Google Scholar] [CrossRef]
- Sterne, J.A.; Egger, M. Funnel plots for detecting bias in meta-analysis: Guidelines on choice of axis. J. Clin. Epidemiol. 2001, 54, 1046–1055. [Google Scholar] [CrossRef]
- Duval, S.; Tweedie, R. Trim and Fill: A Simple Funnel-Plot–Based Method of Testing and Adjusting for Publication Bias in Meta-Analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef]
- Hasheminasab, F.S.; Hashemi, S.M.; Dehghan, A.; Sharififar, F.; Setayesh, M.; Sasanpour, P.; Tasbandi, M.; Raeiszadeh, M. Effects of a Plantago ovata-based herbal compound in prevention and treatment of oral mucositis in patients with breast cancer receiving chemotherapy: A double-blind, randomized, controlled crossover trial. J. Integr. Med. 2020, 18, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Aghamohammadi, A.; Moslemi, D.; Akbari, J.; Ghasemi, A.; Azadbakht, M.; Asgharpour, A.; Hosseinimehr, S.J. The effectiveness of Zataria extract mouthwash for the management of radiation-induced oral mucositis in patients: A randomized placebo-controlled double-blind study. Clin. Oral Investig. 2018, 22, 2263–2272. [Google Scholar] [CrossRef] [PubMed]
- Arantes, D.A.C.; da Silva, A.C.G.; Freitas, N.M.A.; Lima, E.M.; de Oliveira, A.C.; Marreto, R.N.; Mendonça, E.F.; Valadares, M.C. Safety and efficacy of a mucoadhesive phytomedication containing curcuminoids and Bidens pilosa L. extract in the prevention and treatment of radiochemotherapy-induced oral mucositis: Triple-blind, randomized, placebo-controlled, clinical trial. Head Neck 2021, 43, 3922–3934. [Google Scholar] [CrossRef]
- Czakert, J.; Stritter, W.; Blakeslee, S.B.; Grabowski, J.P.; Sehouli, J.; Seifert, G. “Like one part of a puzzle”—Individualized aromatherapy for women with gynecological cancers in aftercare: Results from a qualitative-focused mixed-methods study. Support. Care Cancer 2022, 31, 80. [Google Scholar] [CrossRef]
- Ho, S.S.M.; Kwong, A.N.L.; Wan, K.W.S.; Ho, R.M.L.; Chow, K.M. Experiences of aromatherapy massage among adult female cancer patients: A qualitative study. J. Clin. Nurs. 2017, 26, 4519–4526. [Google Scholar] [CrossRef]
- Oyston, E.; McGee, M. Oncology Massage Research and Training Update. J. Aust. Tradit. Med. Soc. 2012, 18, 11–14. [Google Scholar]
- Lee, W.J.; Park, H. Effects of aroma inhalation on blood pressure, heart rate, pain and quality of sleep among lung cancer patients who underwent pulmonary resection. Asia Life Sci. 2018, 1 (Suppl. 15), 901–912. [Google Scholar]
- Cheong, M.J.; Ha, W.B.; Cho, H.B.; Choi, U.J.; Woo, H.J.; Han, Y.H.; Kang, H.W. Improving the quality of life in a breast cancer patient and caregiver: Protocol for the application of the integrative medical service model. Medicine 2022, 101, e32244. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Li, L.; Xie, Y.; Xu, J.; Liu, Y. [Effects of aroma therapy and music intervention on pain and anxious for breast cancer patients in the perioperative period]. Zhong Nan Da Xue Xue Bao Yi Xue Ban J. Cent. South Univ. Med. Sci. 2018, 43, 656–661. [Google Scholar] [CrossRef]
- Chang, S.Y. Effects of aroma hand massage on pain, state anxiety and depression in hospice patients with terminal cancer. J. Korean Acad. Nurs. 2008, 38, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Nekuzad, N.; Torab, T.A.; Mojab, F.; Alavi-Majd, H.; Azadeh, P.; Ehtejab, G. Effect of external use of Sesame Oil in the prevention of chemotherapy-induced phlebitis. Iran. J. Pharm. Res. 2012, 11, 1065–1071. [Google Scholar] [PubMed]
- Yayla, E.M.; Ozdemir, L. Effect of Inhalation Aromatherapy on Procedural Pain and Anxiety after Needle Insertion into an Implantable Central Venous Port Catheter: A Quasi-Randomized Controlled Pilot Study. Cancer Nurs. 2019, 42, 35–41. [Google Scholar] [CrossRef]
- Corbin, L.W.; Mellis, B.K.; Beaty, B.L.; Kutner, J.S. The Use of Complementary and Alternative Medicine Therapies by Patients with Advanced Cancer and Pain in a Hospice Setting: A Multicentered, Descriptive Study. J. Palliat. Med. 2009, 12, 7–8. [Google Scholar] [CrossRef]
- Ovayolu, Ö.; Seviğ, Ü.; Ovayolu, N.; Sevinç, A. The effect of aromatherapy and massage administered in different ways to women with breast cancer on their symptoms and quality of life. Int. J. Nurs. Pract. 2014, 20, 408–417. [Google Scholar] [CrossRef]
- Elhadad, M.A.; El-Negoumy, E.; Taalab, M.R.; Ibrahim, R.S.; Elsaka, R.O. The effect of topical chamomile in the prevention of chemotherapy-induced oral mucositis: A randomized clinical trial. Oral Dis. 2022, 28, 164–172. [Google Scholar] [CrossRef]
- Haddaway, N.R.; Page, M.J.; Pritchard, C.C.; McGuinness, L.A. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst. Rev. 2022, 18, e1230. [Google Scholar] [CrossRef]
- Blackburn, L.; Achor, S.; Allen, B.; Bauchmire, N.; Dunnington, D.; Klisovic, R.B.; Naber, S.J.; Roblee, K.; Samczak, A.; Tomlinson-Pinkham, K.; et al. The Effect of Aromatherapy on Insomnia and Other Common Symptoms Among Patients With Acute Leukemia. Oncol. Nurs. Forum 2017, 44, E185–E193. [Google Scholar] [CrossRef]
- Blackburn, L.; Hill, C.; Lindsey, A.L.; Sinnott, L.T.; Thompson, K.; Quick, A. Effect of Foot Reflexology and Aromatherapy on Anxiety and Pain During Brachytherapy for Cervical Cancer. Oncol. Nurs. Forum 2021, 48, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Xie, Y.; Liu, Y.; Li, Y.; Xiao, Y. Aromatherapy Plus Music Therapy Improve Pain Intensity and Anxiety Scores in Patients With Breast Cancer During Perioperative Periods: A Randomized Controlled Trial: Aromatherapy plus Music Therapy on Pain and Anxiety. Clin. Breast Cancer 2022, 22, 115–120. [Google Scholar] [CrossRef]
- Ha, Y.S.; Kim, J.W.; Lee, J.E.; Yang, J.H.; Lee, M.J.; Lee, E.Y.; You, J.Y.; Jung, S.P.; Choi, H.J.; Park, K.H. Aroma lymphatic massage (Aroma lymphatic tressage) for symptom relief in breast cancer patients with taxane acute pain syndrome: A randomized controlled crossover trial. Eur. J. Integr. Med. 2022, 54, 102167. [Google Scholar] [CrossRef]
- Ilter, S.M.; Ovayolu, O.; Ovayolu, N. The Effect of Inhaler Aromatherapy on Invasive Pain, Procedure Adherence, Vital Signs, and Saturation During Port Catheterization in Oncology Patients. Holist. Nurs. Pract. 2019, 33, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Izgu, N.; Ozdemir, L.; Basal, F.B. Effect of Aromatherapy Massage on Chemotherapy-Induced Peripheral Neuropathic Pain and Fatigue in Patients Receiving Oxaliplatin An Open Label Quasi-Randomized Controlled Pilot Study. Cancer Nurs. 2019, 42, 139–147. [Google Scholar] [CrossRef]
- Louis, M.; Kowalski, S.D. Use of aromatherapy with hospice patients to decrease pain, anxiety, and depression and to promote an increased sense of well-being. Am. J. Hosp. Palliat. Med. 2022, 19, 381–386. [Google Scholar] [CrossRef]
- Maddocks-Jennings, W.; Wilkinson, J.M.; Cavanagh, H.M.; Shillington, D. Evaluating the effects of the essential oils Leptospermum scoparium (manuka) and Kunzea ericoides (kanuka) on radiotherapy induced mucositis: A randomized, placebo controlled feasibility study. Eur. J. Oncol. Nurs. 2009, 13, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Ndao, D.H.; Ladas, E.J.; Cheng, B.; Sands, S.A.; Snyder, K.T.; Garvin, J.H., Jr.; Kelly, K.M. Inhalation aromatherapy in children and adolescents undergoing stem cell infusion: Results of a placebo-controlled double-blind trial. Psycho-Oncol. 2012, 21, 247–254. [Google Scholar] [CrossRef]
- Shammas, R.L.; Marks, C.E.; Broadwater, G.; Le, E.; Glener, A.D.; Sergesketter, A.R.; Cason, R.W.; Rezak, K.M.; Phillips, B.T.; Hollenbeck, S.T. The Effect of Lavender Oil on Perioperative Pain, Anxiety, Depression, and Sleep after Microvascular Breast Reconstruction: A Prospective, Single-Blinded, Randomized, Controlled Trial. J. Reconstr. Microsurg. 2021, 37, 530–540. [Google Scholar] [CrossRef]
- Soden, K.; Vincent, K.; Craske, S.; Lucas, C.; Asley, S. A randomized controlled trial of aromatherapy massage in a hospice setting. Palliat. Med. 2004, 18, 87–92. [Google Scholar] [CrossRef]
- Triana, K.Y.; Allenidekania, A.; Hayati, H. The Effect of Aromatherapy Inhalation on Reducing Chronic Pain for Children with Cancer: A Pilot Study. Trends Sci. 2022, 19, 2669. [Google Scholar] [CrossRef]
- Bruera, E.; Kim, H.N. Cancer Pain. JAMA 2003, 290, 2476–2479. [Google Scholar] [CrossRef] [PubMed]
- Ahles, T.A.; Blanchard, E.B.; Ruckdeschel, J.C. The multidimensional nature of cancer-related pain. Pain 1983, 17, 277–288. [Google Scholar] [CrossRef]
- Cleeland, C.S.; Ryan, K.M. Pain assessment: Global use of the Brief Pain Inventory. Ann. Acad. Med. Singap. 1994, 23, 129–138. [Google Scholar]
- Scuteri, D.; Watanabe, C.; Sakurada, S.; Hamamura, K.; Sakurada, T.; Tonin, P.; Bagetta, G.; Corasaniti, M.T. Pharmacotechnological Advances for Clinical Translation of Essential Oils for the Treatment of Pain and Agitation in Severe Dementia. Processes 2022, 10, 1340. [Google Scholar] [CrossRef]
- Scuteri, D.; Corasaniti, M.T.; Tonin, P.; Nicotera, P.; Bagetta, G. New trends in pharmacological control of neuropsychiatric symptoms of dementia. Curr. Opin. Pharmacol. 2021, 61, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Guida, F.; Boccella, S.; Palazzo, E.; Maione, S.; Rodríguez-Landa, J.F.; Martínez-Mota, L.; Tonin, P.; Bagetta, G.; Corasaniti, M.T. Effects of Palmitoylethanolamide (PEA) on Nociceptive, Musculoskeletal and Neuropathic Pain: Systematic Review and Meta-Analysis of Clinical Evidence. Pharmaceutics 2022, 14, 1672. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Rombolà, L.; Watanabe, C.; Sakurada, S.; Corasaniti, M.T.; Bagetta, G.; Tonin, P.; Russo, R.; Nucci, C.; Morrone, L.A. Impact of nutraceuticals on glaucoma: A systematic review. Prog. Brain Res. 2020, 257, 141–154. [Google Scholar] [PubMed]
- Scuteri, D.; Contrada, M.; Loria, T.; Sturino, D.; Cerasa, A.; Tonin, P.; Sandrini, G.; Tamburin, S.; Bruni, A.C.; Nicotera, P.; et al. Pain and agitation treatment in severe dementia patients: The need for Italian Mobilization–Observation–Behavior–Intensity–Dementia (I-MOBID2) pain scale translation, adaptation and validation with psychometric testing. Biomed. Pharmacother. 2022, 150, 113013. [Google Scholar] [CrossRef]
- Scuteri, D.; Contrada, M.; Loria, T.; Tonin, P.; Sandrini, G.; Tamburin, S.; Nicotera, P.; Bagetta, G.; Corasaniti, M.T. Pharmacological Treatment of Pain and Agitation in Severe Dementia and Responsiveness to Change of the Italian Mobilization–Observation–Behavior–Intensity–Dementia (I-MOBID2) Pain Scale: Study Protocol. Brain Sci. 2022, 12, 573. [Google Scholar] [CrossRef]
- Scuteri, D.; Matamala-Gomez, M.; Bottiroli, S.; Corasaniti, M.T.; De Icco, R.; Bagetta, G.; Tonin, P. Pain assessment and treatment in dementia at the time of coronavirus disease COVID-19. Front. Neurol. 2020, 11, 890. [Google Scholar] [CrossRef]
- Pignolo, L.; Tonin, P.; Nicotera, P.; Bagetta, G.; Scuteri, D. ROBOCOP (ROBOtic Care of Poststroke Pain): Study Protocol for a Randomized Trial to Assess Robot-Assisted Functional and Motor Recovery and Impact on Poststroke Pain Development. Front. Neurol. 2022, 13, 813282. [Google Scholar] [CrossRef]
- Scuteri, D.; Contrada, M.; Tonin, P.; Corasaniti, M.T.; Nicotera, P.; Bagetta, G. Dementia and COVID-19: A Case Report and Literature Review on Pain Management. Pharmaceuticals 2022, 15, 199. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Tonin, P.; Nicotera, P.; Bagetta, G.; Corasaniti, M.T. Real world considerations for newly approved CGRP receptor antagonists in migraine care. Expert Rev. Neurother. 2022, 22, 221–230. [Google Scholar] [CrossRef]
- Scuteri, D.; Tonin, P.; Nicotera, P.; Vulnera, M.; Altieri, G.C.; Tarsitano, A.; Bagetta, G.; Corasaniti, M.T. Pooled Analysis of Real-World Evidence Supports Anti-CGRP mAbs and OnabotulinumtoxinA Combined Trial in Chronic Migraine. Toxins 2022, 14, 529. [Google Scholar] [CrossRef]
- Contrada, M.; Cerasa, A.; Tonin, P.; Bagetta, G.; Scuteri, D. Aromatherapy in Stroke Patients: Is it Time to Begin? Front. Behav. Neurosci. 2021, 15, 749353. [Google Scholar] [CrossRef] [PubMed]
- Silva, G.L.; Luft, C.; Lunardelli, A.; Amaral, R.H.; Melo, D.A.; Donadio, M.V.; Nunes, F.B.; de Azambuja, M.S.; Santana, J.C.; Moraes, C.M.; et al. Antioxidant, analgesic and anti-inflammatory effects of lavender essential oil. An. Acad. Bras. Cienc. 2015, 87, 1397–1408. [Google Scholar] [CrossRef] [PubMed]
- Scuteri, D.; Rombolà, L.; Tridico, L.; Mizoguchi, H.; Watanabe, C.; Sakurada, T.; Sakurada, S.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Neuropharmacological properties of the essential oil of bergamot for the clinical management of pain-related BPSDs. Curr. Med. Chem. 2019, 26, 3764–3774. [Google Scholar] [CrossRef]
- Van de Ven, T.J.; John Hsia, H.L. Causes and prevention of chronic postsurgical pain. Curr. Opin. Crit. Care 2012, 18, 366–371. [Google Scholar] [CrossRef]
- Kitade, M.; Nakajima, H.; Tsujikawa, T.; Noriki, S.; Mori, T.; Kiyono, Y.; Okazawa, H.; Matsumine, A. Evaluation of (R)-[11C]PK11195 PET/MRI for Spinal Cord-Related Neuropathic Pain in Patients with Cervical Spinal Disorders. J. Clin. Med. 2023, 12, 116. [Google Scholar] [CrossRef]

| Study | Population | Intervention | Comparator | Outcome Measures | Study Design | Power Analysis | Ethical Committee/ Institutional Review Board Approval and Request for Informed Consent | Results |
|---|---|---|---|---|---|---|---|---|
| Blackburn et al., 2017 [71] | Patients aged 18 years or older, having a new diagnosis of acute, myelogenous leukemia, and initiating four weeks of intensive induction chemotherapy | Lavender, peppermint, or chamomile | Placebo | Edmonton Symptom Assessment Scale–Revised with Visual Analog Scale (VAS) | Randomized, cross-over, wash-out trial, N = 50; First Aromatherapy, N = 25; First placebo, N = 28 | + | + | Aromatherapy = 2.84 ± 2.23; Placebo = 2.84 ± 2.23 |
| Blackburn et al., 2021 [72] | Women affected by locally advanced cervical cancer | Lavender, lemon, peppermint, and foot reflexology | Standard of- care management medications | Numeric rating scale (NRS) | Randomized, controlled trial, N = 41; Intervention, N = 22; Control, N = 19 | + | + | Intervention = 1.5 ± 1.3; Control = 4.4 ± 2.5 |
| Deng et al., 2021 [73] | Women > 18 years old, subjected to breast cancer surgery | Aromatherapy alone or in combination with music therapy | Usual care or music therapy | VAS | Randomized, open-label, controlled trial, N = 160; Aromatherapy, N = 40; Combination therapy, N = 40; Music therapy, N = 40; Usual care, N = 40 | Not reported | + | Combination therapy = 2.03 ± 0.83; aromatherapy alone = 3.38 ± 0.90; Usual care = 6.13 ± 1.02 |
| Ha et al., 2022 [74] | Breast cancer patients with taxane acute pain syndrome 20 years of age or older | Aroma lymphatic tressage (Frankincense is used as essential oil and sweet almond oil as a carrier oil) in addition to standard care | Standard care (acetaminophen/tramadol) | VAS | Phase II, randomized, cross-over trial, N = 65. First standard care, N = 22; First aroma lymphatic tressage, N = 33 | + | +; Trial registration = KCT0005758 | No significant difference in pain score (p = 0.368) or toxicity are reported |
| Ilter et al., 2019 [75] | Patients diagnosed with cancer undergoing port catheter insertion aged 18 years or older | Aromatic mixture prepared by diluting orange, chamomile, and lavender oil at the ratio of 1:1:1 in 70 mL distilled water for inhalation | Routine practices | VAS | Non-randomized, controlled trial, N = 60; Intervention, N = 30; Control, N = 30 | + | + | Aromatherapy (5 ± 1.2) reduces pain during and after the procedure in a statistically significant manner in comparison with control (i.e., no treatment; 7.4 ± 1.4) |
| Izgu et al., 2019 [76] | Patients with Chemotherapy-Induced Peripheral Neuropathic (CIPN) Pain due to oxaliplatin of age of 18 years or older | Aromatherapy massage (peppermint, chamomile, and rosemary blended in 1:1:1 proportion at 1.5% in 50 mL of coconut oil) | Routine care | Douleur Neuropathique 4 Questions | Open-label, quasi-randomized, controlled, pilot study, N = 46; Intervention, N = 22; Control, N = 24 | + | + | At week 6, the rate of neuropathic pain is significantly lower in the aromatherapy massage group |
| Louis and Kowalski, 2022 [77] | Homecare hospice patients with terminal cancer | Aromatherapy with humidified lavender essential oil (3%) | Control (no treatment/water humidification) | VAS | Quasi-experimental, repeated measures, one-group design N = 17 | - | + | Not significant difference |
| Maddocks-Jennings et al., 2009 [78] | Patients aged over 18 affected by radiation-induced mucositis of the oropharyngeal area during treatment for head and neck cancers | Gargle containing 2 drops of a 1:1 mix of the essential oils of manuka and kanuka in water + usual oral care as prescribed | Bottle of sterile water for gargling + usual oral care as prescribed. Instead of placebo, receives usual care | VAS | Randomized, placebo-controlled, feasibility study, N = 19; Active group, N = 9; Placebo group, N = 6; Control group, N = 8 | - | + | Within the active group, n = 2 patients experience pain scores ≥ 3, n = 5 from the control group, and n = 4 from the placebo group |
| Ndao et al., 2012 [79] | Children and adolescents undergoing stem cell infusion | Respiratory administration of bergamot essential oil (BEO) through stream aromatherapy diffuser | Placebo consisting in non-essential-oil-based scented shampoo | VAS | Randomized, placebo-controlled, double-blind trial, N = 37; Intervention, N = 17; Placebo N = 20 | + | + | No significant effect on pain |
| Shammas et al., 2021 [80] | Women (ages 18 to 85 years) subjected to microvascular breast reconstruction due to breast cancer | Lavender oil | Coconut oil | VAS | Prospective, single-blinded, randomized, controlled trial, Intervention, N = 27; Control, N = 22 | - | + | No significant differences in the perioperative setting for pain scores (p = 0.30) |
| Soden et al., 2004 [81] | Patients with advanced cancer | Aromatherapy massage with lavender essential oil and an inert carrier oil | Massage with an inert carrier oil alone or no intervention | VAS and Verbal Rating Scale (VRS) | Randomized, controlled trial, N = 42; Aromatherapy massage, N = 16; Massage, N = 13; Control, N = 13 | + | + | No statistically significant difference in pain intensity from final to baseline assessment |
| Triana et al., 2022 [82] | School-age children and adolescents (7–17 years), with a diagnosis of cancer, experiencing chronic pain (for longer than 1 month), and receiving moderate analgesics/opioids | Aromatherapy inhalation of a scent that the participant likes (favorite, aloe vera) | Standard care (painkillers and a relaxing technique) | VAS | Quasi-experimental with a consecutive sampling, N = 20; Intervention group, N = 10; Control group, N = 10 | - | + | Inhaled aromatherapy significantly reduces chronic pain (p = 0.001) compared with standard care. At minute 30, Intervention = 0.4 ± 0.97 and Control = 6.6 ± 1.43 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corasaniti, M.T.; Bagetta, G.; Morrone, L.A.; Tonin, P.; Hamamura, K.; Hayashi, T.; Guida, F.; Maione, S.; Scuteri, D. Efficacy of Essential Oils in Relieving Cancer Pain: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2023, 24, 7085. https://doi.org/10.3390/ijms24087085
Corasaniti MT, Bagetta G, Morrone LA, Tonin P, Hamamura K, Hayashi T, Guida F, Maione S, Scuteri D. Efficacy of Essential Oils in Relieving Cancer Pain: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2023; 24(8):7085. https://doi.org/10.3390/ijms24087085
Chicago/Turabian StyleCorasaniti, Maria Tiziana, Giacinto Bagetta, Luigi Antonio Morrone, Paolo Tonin, Kengo Hamamura, Takafumi Hayashi, Francesca Guida, Sabatino Maione, and Damiana Scuteri. 2023. "Efficacy of Essential Oils in Relieving Cancer Pain: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 24, no. 8: 7085. https://doi.org/10.3390/ijms24087085
APA StyleCorasaniti, M. T., Bagetta, G., Morrone, L. A., Tonin, P., Hamamura, K., Hayashi, T., Guida, F., Maione, S., & Scuteri, D. (2023). Efficacy of Essential Oils in Relieving Cancer Pain: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 24(8), 7085. https://doi.org/10.3390/ijms24087085










