Navigating through the PD-1/PDL-1 Landscape: A Systematic Review and Meta-Analysis of Clinical Outcomes in Hepatocellular Carcinoma and Their Influence on Immunotherapy and Tumor Microenvironment
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Criteria for Inclusion and Exclusion
2.3. Data Extraction and Assessment of Study Quality
2.4. Statistical Analysis
3. Results
3.1. Eligible Studies
3.2. Characteristics of the Studies Included
3.3. Survival Analysis of PD-1 and PDL-1
3.4. Correlations between PD-1/PDL-1 and Clinicopathological Parameters
3.5. PD-1/PDL-1 and Other Markers
4. Discussion
4.1. PD-1/PDL-1 Survival Assessment
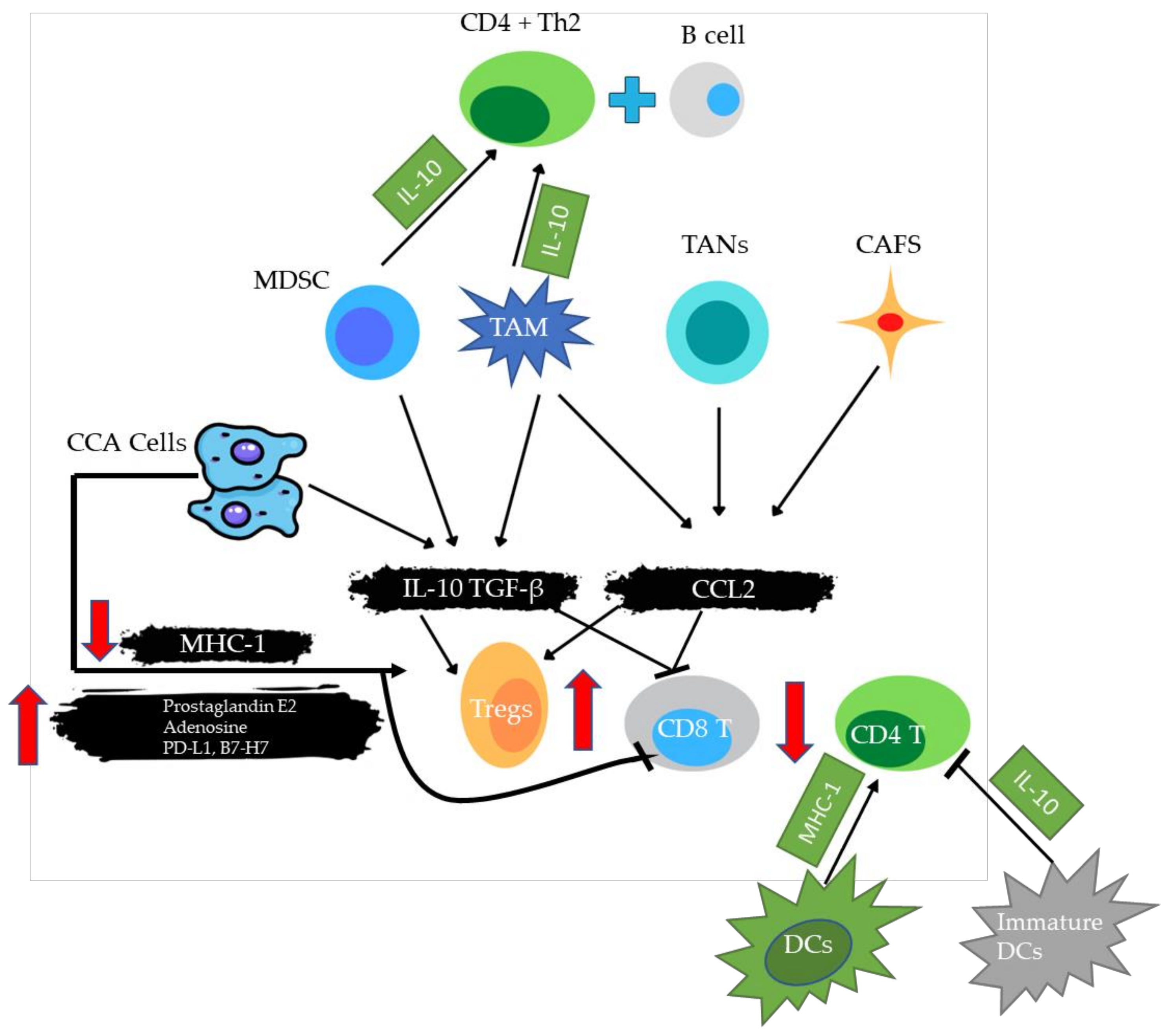
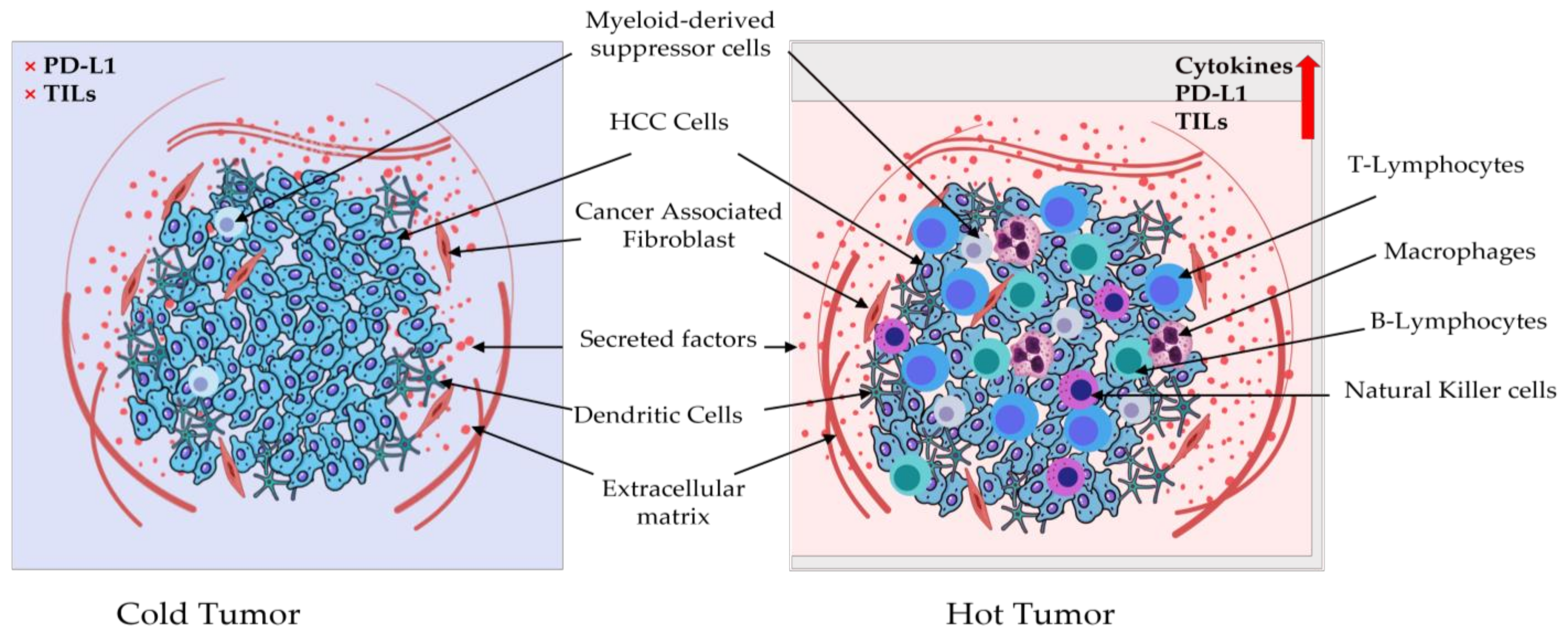
4.2. PD-1/PDL-1 and Tumor Microenvironment
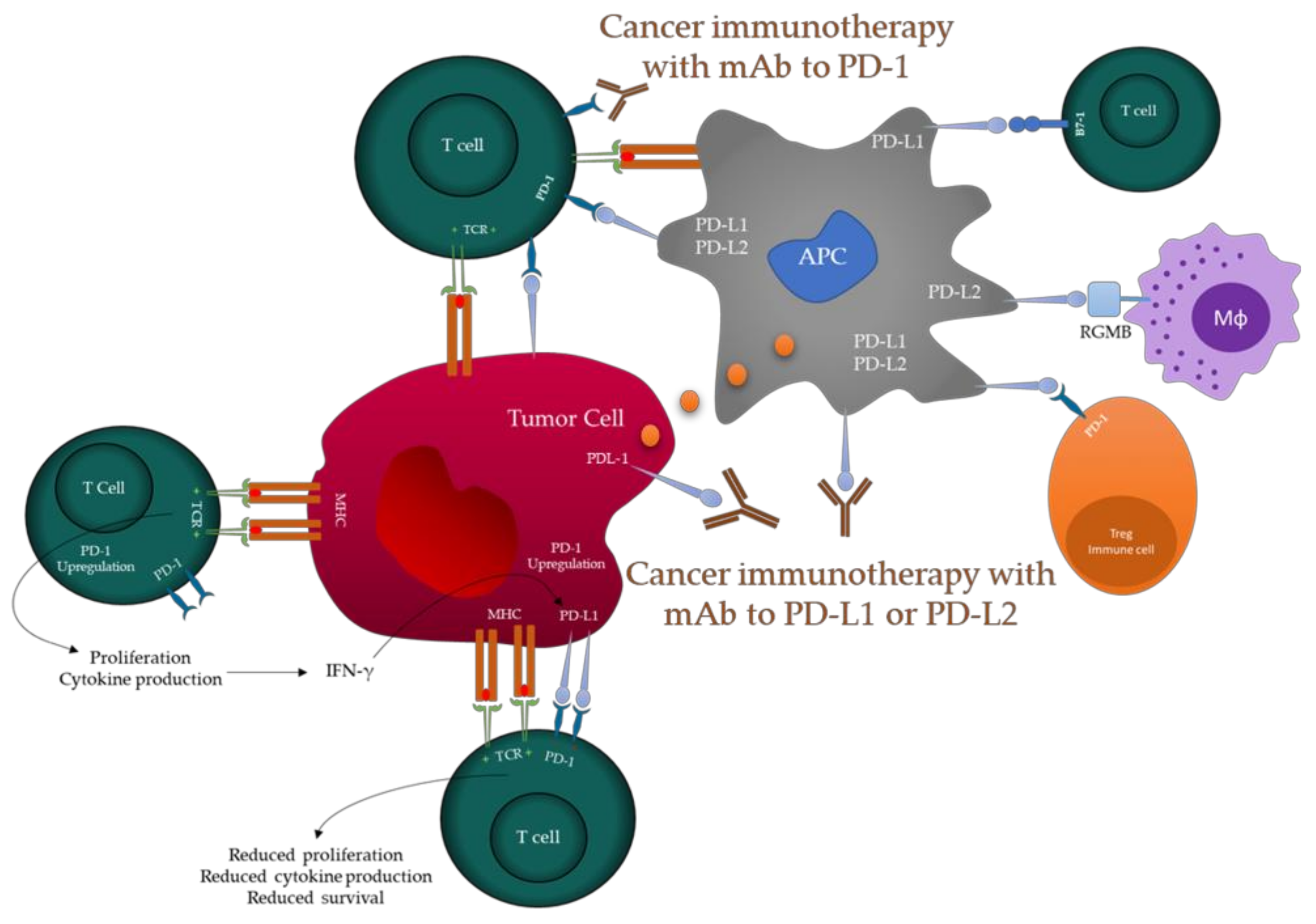
4.3. PD-1/PDL-1 Immune Check Point Inhibitors
4.4. Combination Therapy
4.4.1. Hypoxia-Inducible Factors
4.4.2. Suppressor of Cytokine Signaling 3 (SOCS3)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Papatheodoridis, G.V.; Sypsa, V.; Dalekos, G.N.; Yurdaydin, C.; Van Boemmel, F.; Buti, M.; Calleja, J.L.; Chi, H.; Goulis, J.; Manolakopoulos, S.; et al. Hepatocellular carcinoma prediction beyond year 5 of oral therapy in a large cohort of Caucasian patients with chronic hepatitis B. J. Hepatol. 2020, 72, 1088–1096. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, A.; Llovet, J.M. Targeted Therapies for Hepatocellular Carcinoma. Gastroenterology 2011, 140, 1410–1426. [Google Scholar] [CrossRef] [PubMed]
- Ailia, M.J.; Yoo, S.Y. In Vivo Oncolytic Virotherapy in Murine Models of Hepatocellular Carcinoma: A Systematic Review. Vaccines 2022, 10, 1541. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.Y.; Badrinath, N.; Woo, H.Y.; Heo, J. Oncolytic Virus-Based Immunotherapies for Hepatocellular Carcinoma. Mediat. Inflamm. 2017, 2017, 5198798. [Google Scholar] [CrossRef]
- Heinrich, B.; Czauderna, C.; Marquardt, J.U. Immunotherapy of Hepatocellular Carcinoma. Oncol. Res. Treat. 2018, 41, 292–297. [Google Scholar] [CrossRef]
- Tunger, A.; Kiessler, M.; Wehner, R.; Temme, A.; Meier, F.; Bachmann, M.; Schmitz, M. Immune Monitoring of Cancer Patients Prior to and During CTLA-4 or PD-1/PD-L1 Inhibitor Treatment. Biomedicines 2018, 6, 26. [Google Scholar] [CrossRef]
- Fan, Z.Y.; Liang, Y.; Yang, X.C.; Li, B.; Cui, L.L.; Luo, L.; Jia, Y.F.; Wang, Y.H.; Niu, H.T. A meta-analysis of the efficacy and safety of PD-1/PD-L1 immune checkpoint inhibitors as treatments for metastatic bladder cancer. Onco Targets Ther. 2019, 12, 1791–1801. [Google Scholar] [CrossRef]
- Huang, M.Y.; Jiang, X.M.; Wang, B.L.; Sun, Y.; Lu, J.J. Combination therapy with PD-1/PD-L1 blockade in non-small cell lung cancer: Strategies and mechanisms. Pharmacol. Ther. 2021, 219, 107694. [Google Scholar] [CrossRef]
- Day, C.L.; Kaufmann, D.E.; Kiepiela, P.; Brown, J.A.; Moodley, E.S.; Reddy, S.; Mackey, E.W.; Miller, J.D.; Leslie, A.J.; DePierres, C.; et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 2006, 443, 350–354. [Google Scholar] [CrossRef]
- Taube, J.M.; Klein, A.; Brahmer, J.R.; Xu, H.Y.; Pan, X.Y.; Kim, J.H.; Chen, L.P.; Pardoll, D.M.; Topalian, S.L.; Anders, R.A. Association of PD-1, PD-1 Ligands, and Other Features of the Tumor Immune Microenvironment with Response to Anti-PD-1 Therapy. Clin. Cancer Res. 2014, 20, 5064–5074. [Google Scholar] [CrossRef]
- Singh, V.; Khurana, A.; Allawadhi, P.; Banothu, A.K.; Bharani, K.K.; Weiskirchen, R. Emerging Role of PD-1/PD-L1 Inhibitors in Chronic Liver Diseases. Front. Pharmacol. 2021, 12, 790963. [Google Scholar] [CrossRef]
- Greenwald, R.J.; Freeman, G.J.; Sharpe, A.H. The B7 family revisited. Annu. Rev. Immunol. 2005, 23, 515–548. [Google Scholar] [CrossRef]
- Riley, J.L. PD-1 signaling in primary T cells. Immunol. Rev. 2009, 229, 114–125. [Google Scholar] [CrossRef]
- Pauken, K.E.; Wherry, E.J. Overcoming T cell exhaustion in infection and cancer. Trends Immunol. 2015, 36, 265–276. [Google Scholar] [CrossRef]
- Iwai, Y.; Terawaki, S.; Honjo, T. PD-1 blockade inhibits hematogenous spread of poorly immunogenic tumor cells by enhanced recruitment of effector T cells. Int. Immunol. 2005, 17, 133–144. [Google Scholar] [CrossRef]
- Yau, T.; Kang, Y.K.; Kim, T.Y.; El-Khoueiry, A.B.; Santoro, A.; Sangro, B.; Melero, I.; Kudo, M.; Hou, M.M.; Matilla, A.; et al. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients with Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020, 6, e204564. [Google Scholar] [CrossRef]
- FDA, U.S. FDA Grants Accelerated Approval to Nivolumab for HCC Previously Treated with Sorafenib. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-nivolumab-hcc-previously-treated-sorafenib (accessed on 9 January 2023).
- Nexavar FDA Approval History. Available online: https://www.drugs.com/history/nexavar.html (accessed on 7 August 2022).
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.N.; Kim, T.Y.; Choo, S.P.; Trojan, J.; Welling, T.H.; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Alsaab, H.O.; Sau, S.; Alzhrani, R.; Tatiparti, K.; Bhise, K.; Kashaw, S.K.; Iyer, A.K. PD-1 and PD-L1 Checkpoint Signaling Inhibition for Cancer Immunotherapy: Mechanism, Combinations, and Clinical Outcome. Front. Pharmacol. 2017, 8, 561. [Google Scholar] [CrossRef]
- Peng, J.H.; Tai, Y.; Zhao, Y.X.; Luo, B.J.; Ou, Q.J.; Pan, Z.Z.; Zhang, L.; Lu, Z.H. Programmed death-ligand 1 expression in the tumour stroma of colorectal liver oligometastases and its association with prognosis after liver resection. Gastroenterol. Rep. 2021, 9, 443–450. [Google Scholar] [CrossRef]
- Dai, X.M.; Pi, G.L.; Yang, S.L.; Chen, G.G.; Liu, L.P.; Dong, H.H. Association of PD-L1 and HIF-1 alpha Coexpression with Poor Prognosis in Hepatocellular Carcinoma. Transl. Oncol. 2018, 11, 559–566. [Google Scholar] [CrossRef]
- Chang, H.; Jung, W.; Kim, A.; Kim, H.K.; Kim, W.B.; Kim, J.H.; Kim, B.H. Expression and prognostic significance of programmed death protein 1 and programmed death ligand-1, and cytotoxic T lymphocyte-associated molecule-4 in hepatocellular carcinoma. Apmis 2017, 125, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.I.; Jeong, D.; Ji, S.; Ahn, T.S.; Bae, S.H.; Chin, S.; Chung, J.C.; Kim, H.C.; Lee, M.S.; Baek, M.J. Overexpression of PD-L1 and PD-L2 Is Associated with Poor Prognosis in Patients with Hepatocellular Carcinoma. Cancer Res. Treat. 2017, 49, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.X.; Huang, X.X.; Zhang, W.Z.; Liu, Y.; Chen, B.; Xiang, Y.; Zhang, R.N.; Zhang, M.M.; Feng, J.; Liu, S.P.; et al. Correlation of PD-L1 and SOCS3 Co-expression with the Prognosis of Hepatocellular Carcinoma Patients. J. Cancer 2020, 11, 5440–5448. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Shi, F.; Zhou, L.; Zhang, M.N.; Chen, Y.; Chang, X.J.; Lu, Y.Y.; Bai, W.L.; Qu, J.H.; Wang, C.P.; et al. Upregulation of circulating PD-L1/PD-1 is associated with poor post-cryoablation prognosis in patients with HBV-related hepatocellular carcinoma. PLoS ONE 2011, 6, e23621. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhou, Z.; Li, F.; Sang, J.; Han, Q.; Lv, Y.; Zhao, W.; Li, C.; Liu, Z. Circulating soluble programmed death-1 levels may differentiate immune-tolerant phase from other phases and hepatocellular carcinoma from other clinical diseases in chronic hepatitis B virus infection. Oncotarget 2017, 8, 46020–46033. [Google Scholar] [CrossRef]
- Finkelmeier, F.; Canli, O.; Tal, A.; Pleli, T.; Trojan, J.; Schmidt, M.; Kronenberger, B.; Zeuzem, S.; Piiper, A.; Greten, F.R.; et al. High levels of the soluble programmed death-ligand (sPD-L1) identify hepatocellular carcinoma patients with a poor prognosis. Eur. J. Cancer 2016, 59, 152–159. [Google Scholar] [CrossRef]
- Liu, X.; Li, M.; Wang, X.; Dang, Z.; Jiang, Y.; Wang, X.; Kong, Y.; Yang, Z. PD-1(+) TIGIT(+) CD8(+) T cells are associated with pathogenesis and progression of patients with hepatitis B virus-related hepatocellular carcinoma. Cancer Immunol. Immunother. 2019, 68, 2041–2054. [Google Scholar] [CrossRef]
- Chang, B.Y.; Huang, T.; Wei, H.J.; Shen, L.J.; Zhu, D.; He, W.J.; Chen, Q.F.; Zhang, H.H.; Li, Y.J.; Huang, R.P.; et al. The correlation and prognostic value of serum levels of soluble programmed death protein 1 (sPD-1) and soluble programmed death-ligand 1 (sPD-L1) in patients with hepatocellular carcinoma. Cancer Immunol. Immunother. 2019, 68, 353–363. [Google Scholar] [CrossRef]
- Qin, T.; Zeng, Y.D.; Qin, G.; Xu, F.; Lu, J.B.; Fang, W.F.; Xue, C.; Zhan, J.H.; Zhang, X.K.; Zheng, Q.F.; et al. High PD-L1 expression was associated with poor prognosis in 870 Chinese patients with breast cancer. Oncotarget 2015, 6, 33972–33981. [Google Scholar] [CrossRef]
- Gu, X.B.; Dong, M.L.; Liu, Z.Y.; Mi, Y.; Yang, J.; Zhang, Z.G.; Liu, K.; Jiang, L.; Zhang, Y.; Dong, S.L.; et al. Elevated PD-L1 expression predicts poor survival outcomes in patients with cervical cancer. Cancer Cell. Int. 2019, 19, 146. [Google Scholar] [CrossRef]
- Li, G.P.; Wang, G.L.; Chi, F.Q.; Jia, Y.Q.; Wang, X.; Mu, Q.K.; Qin, K.R.; Zhu, X.X.; Pang, J.; Xu, B.X.; et al. Higher postoperative plasma EV PD-L1 predicts poor survival in patients with gastric cancer. J. Immunother. Cancer 2021, 9, e002218. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Shi, D.B.; Miao, J.J.; Wu, H.J.; Chen, J.W.; Zhou, X.Y.; Hu, D.S.; Zhao, C.; Deng, W.G.; Xie, C.H. PD-L1 predicts poor prognosis for nasopharyngeal carcinoma irrespective of PD-1 and EBV-DNA load. Sci. Rep. 2017, 7, srep43627. [Google Scholar] [CrossRef]
- Zhao, Y.J.; Shi, F.; Zhou, Q.; Li, Y.C.; Wu, J.P.; Wang, R.B.; Song, Q.K. Prognostic significance of PD-L1 in advanced non-small cell lung carcinoma. Medicine 2020, 99, e23172. [Google Scholar] [CrossRef]
- Carlsson, J.; Sundqvist, P.; Kosuta, V.; Falt, A.; Giunchi, F.; Fiorentino, M.; Davidsson, S. PD-L1 Expression is Associated With Poor Prognosis in Renal Cell Carcinoma. Appl. Immunohistochem. Mol. Morphol. 2020, 28, 213–220. [Google Scholar] [CrossRef]
- Giraldo, N.A.; Becht, E.; Remark, R.; Damotte, D.; Sautes-Fridman, C.; Fridman, W.H. The immune contexture of primary and metastatic human tumours. Curr. Opin. Immunol. 2014, 27, 8–15. [Google Scholar] [CrossRef]
- Fridman, W.H.; Pages, F.; Sautes-Fridman, C.; Galon, J. The immune contexture in human tumours: Impact on clinical outcome. Nat. Rev. Cancer 2012, 12, 298–306. [Google Scholar] [CrossRef]
- Bindea, G.; Mlecnik, B.; Tosolini, M.; Kirilovsky, A.; Waldner, M.; Obenauf, A.C.; Angell, H.; Fredriksen, T.; Lafontaine, L.; Berger, A.; et al. Spatiotemporal Dynamics of Intratumoral Immune Cells Reveal the Immune Landscape in Human Cancer. Immunity 2013, 39, 782–795. [Google Scholar] [CrossRef]
- Duan, Q.Q.; Zhang, H.L.; Zheng, J.N.; Zhang, L.J. Turning Cold into Hot: Firing up the Tumor Microenvironment. Trends Cancer 2020, 6, 605–618. [Google Scholar] [CrossRef]
- Galon, J.; Bruni, D. Approaches to treat immune hot, altered and cold tumours with combination immunotherapies. Nat. Rev. Drug. Discov. 2019, 18, 197–218. [Google Scholar] [CrossRef]
- Zemek, R.M.; De Jong, E.; Chin, W.L.; Schuster, I.S.; Fear, V.S.; Casey, T.H.; Forbes, C.; Dart, S.J.; Leslie, C.; Zaitouny, A.; et al. Sensitization to immune checkpoint blockade through activation of a STAT1/NK axis in the tumor microenvironment. Sci. Transl. Med. 2019, 11, eaav7816. [Google Scholar] [CrossRef]
- Makarova-Rusher, O.V.; Medina-Echeverz, J.; Duffy, A.G.; Greten, T.F. The yin and yang of evasion and immune activation in HCC. J. Hepatol. 2015, 62, 1420–1429. [Google Scholar] [CrossRef] [PubMed]
- Sia, D.; Jiao, Y.; Martinez-Quetglas, I.; Kuchuk, O.; Villacorta-Martin, C.; de Moura, M.C.; Putra, J.; Camprecios, G.; Bassaganyas, L.; Akers, N.; et al. Identification of an Immune-specific Class of Hepatocellular Carcinoma, Based on Molecular Features. Gastroenterology 2017, 153, 812–826. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.Y.; Shen, L.J.; Wang, K.F.; Jin, J.T.; Huang, T.; Chen, Q.F.; Li, W.; Wu, P.H. High number of PD-1 positive intratumoural lymphocytes predicts survival benefit of cytokine-induced killer cells for hepatocellular carcinoma patients. Liver Int. 2018, 38, 1449–1458. [Google Scholar] [CrossRef] [PubMed]
- Gabrielson, A.; Wu, Y.; Wang, H.; Jiang, J.; Kallakury, B.; Gatalica, Z.; Reddy, S.; Kleiner, D.; Fishbein, T.; Johnson, L.; et al. Intratumoral CD3 and CD8 T-cell Densities Associated with Relapse-Free Survival in HCC. Cancer Immunol. Res. 2016, 4, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.X.; Zhou, H.Q.; Zhang, L. Which is the optimal immunotherapy for advanced squamous non-small-cell lung cancer in combination with chemotherapy: Anti-PD-1 or anti-PD-L1? J. Immunother. Cancer 2018, 6, 135. [Google Scholar] [CrossRef]
- Liao, H.T.; Chen, W.; Dai, Y.L.; Richardson, J.J.; Guo, J.L.; Yuan, K.F.; Zeng, Y.; Xie, K.L. Expression of Programmed Cell Death-Ligands in Hepatocellular Carcinoma: Correlation with Immune Microenvironment and Survival Outcomes. Front. Oncol. 2019, 9, 883. [Google Scholar] [CrossRef]
- Han, X.; Gu, Y.K.; Li, S.L.; Chen, H.; Chen, M.S.; Cai, Q.Q.; Deng, H.X.; Zuo, M.X.; Huang, J.H. Pre-treatment serum levels of soluble programmed cell death-ligand 1 predict prognosis in patients with hepatitis B-related hepatocellular carcinoma. J. Cancer Res. Clin. 2019, 145, 303–312. [Google Scholar] [CrossRef]
- Sakuishi, K.; Apetoh, L.; Sullivan, J.M.; Blazar, B.R.; Kuchroo, V.K.; Anderson, A.C. Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J. Exp. Med. 2010, 207, 2187–2194. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, S.; Hu, Y.; Yang, Z.; Li, J.; Liu, X.; Deng, L.; Wang, Y.; Zhang, X.; Jiang, T.; et al. Targeting PD-1 and Tim-3 Pathways to Reverse CD8 T-Cell Exhaustion and Enhance Ex Vivo T-Cell Responses to Autologous Dendritic/Tumor Vaccines. J. Immunother. 2016, 39, 171–180. [Google Scholar] [CrossRef]
- Liu, J.H.; Liu, Y.H.; Meng, L.Y.; Liu, K.; Ji, B. Targeting the PD-L1/DNMT1 axis in acquired resistance to sorafenib in human hepatocellular carcinoma. Oncol. Rep. 2017, 38, 899–907. [Google Scholar] [CrossRef]
- Liu, D.; Heij, L.R.; Czigany, Z.; Dahl, E.; Lang, S.A.; Ulmer, T.F.; Luedde, T.; Neumann, U.P.; Bednarsch, J. The role of tumor-infiltrating lymphocytes in cholangiocarcinoma. J. Exp. Clin. Canc Res. 2022, 41, 127. [Google Scholar] [CrossRef]
- Wu, X.M.; Gu, Z.K.; Chen, Y.; Chen, B.R.; Chen, W.; Weng, L.Q.; Liu, X.L. Application of PD-1 Blockade in Cancer Immunotherapy. Comput. Struct. Biotechnol. J. 2019, 17, 661–674. [Google Scholar] [CrossRef]
- Bosco, M.C.; D’Orazi, G.; Del Bufalo, D. Targeting hypoxia in tumor: A new promising therapeutic strategy. J. Exp. Clin. Canc Res. 2020, 39, 8. [Google Scholar] [CrossRef]
- Barsoum, I.B.; Koti, M.; Siemens, D.R.; Graham, C.H. Mechanisms of Hypoxia-Mediated Immune Escape in Cancer. Cancer Res. 2014, 74, 7185–7190. [Google Scholar] [CrossRef]
- Semenza, G.L. Pharmacologic Targeting of Hypoxia-Inducible Factors. Annu. Rev. Pharmacol. 2019, 59, 379–403. [Google Scholar] [CrossRef]
- Semenza, G.L. Targeting HIF-1 for cancer therapy. Nat. Rev. Cancer 2003, 3, 721–732. [Google Scholar] [CrossRef]
- Wu, Q.; You, L.; Nepovimova, E.; Heger, Z.; Wu, W.; Kuca, K.; Adam, V. Hypoxia-inducible factors: Master regulators of hypoxic tumor immune escape. J. Hematol. Oncol. 2022, 15, 77. [Google Scholar] [CrossRef]
- Schito, L.; Semenza, G.L. Hypoxia-Inducible Factors: Master Regulators of Cancer Progression. Trends Cancer 2016, 2, 758–770. [Google Scholar] [CrossRef]
- Triner, D.; Shah, Y.M. Hypoxia-inducible factors: A central link between inflammation and cancer. J. Clin. Investig. 2016, 126, 3689–3698. [Google Scholar] [CrossRef]
- Semenza, G.L. Intratumoral Hypoxia and Mechanisms of Immune Evasion Mediated by Hypoxia-Inducible Factors. Physiology 2021, 36, 73–83. [Google Scholar] [CrossRef]
- Chang, Y.L.; Yang, C.Y.; Lin, M.W.; Wu, C.T.; Yang, P.C. High co-expression of PD-L1 and HIF-1alpha correlates with tumour necrosis in pulmonary pleomorphic carcinoma. Eur. J. Cancer 2016, 60, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Noman, M.Z.; Desantis, G.; Janji, B.; Hasmim, M.; Karray, S.; Dessen, P.; Bronte, V.; Chouaib, S. PD-L1 is a novel direct target of HIF-1α, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J. Exp. Med. 2014, 211, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.R.; Tao, Y.R.; Shi, Z.M.; Liang, W.L.; Hu, W.H.; Xing, Z.; Zhou, S.L.; Guo, X.Y.; Fu, X.D.; Wang, X.J. SOCS3 Acts as an Onco-immunological Biomarker with Value in Assessing the Tumor Microenvironment, Pathological Staging, Histological Subtypes, Therapeutic Effect, and Prognoses of Several Types of Cancer. Front. Oncol. 2022, 12, 881801. [Google Scholar] [CrossRef] [PubMed]
- Muthukutty, P.; Woo, H.Y.; Ragothaman, M.; Yoo, S.Y. Recent Advances in Cancer Immunotherapy Delivery Modalities. Pharmaceutics 2023, 15, 504. [Google Scholar] [CrossRef]
- Rizzo, A.; Ricci, A.D.; Di Federico, A.; Frega, G.; Palloni, A.; Tavolari, S.; Brandi, G. Predictive Biomarkers for Checkpoint Inhibitor-Based Immunotherapy in Hepatocellular Carcinoma: Where Do We Stand? Front. Oncol. 2021, 11, 803133. [Google Scholar] [CrossRef]
- Viscardi, G.; Tralongo, A.C.; Massari, F.; Lambertini, M.; Mollica, V.; Rizzo, A.; Comito, F.; Di Liello, R.; Alfieri, S.; Imbimbo, M.; et al. Comparative assessment of early mortality risk upon immune checkpoint inhibitors alone or in combination with other agents across solid malignancies: A systematic review and meta-analysis. Eur. J. Cancer 2022, 177, 175–185. [Google Scholar] [CrossRef]
- Rizzo, A.; Cusmai, A.; Gadaleta-Caldarola, G.; Palmiotti, G. Which role for predictors of response to immune checkpoint inhibitors in hepatocellular carcinoma? Expert Rev. Gastroenterol. Hepatol. 2022, 16, 333–339. [Google Scholar] [CrossRef]

 The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;
The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;  the black line depicts the study’s confidence interval at 95%;
the black line depicts the study’s confidence interval at 95%;  the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.
the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.
 The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;
The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;  the black line depicts the study’s confidence interval at 95%;
the black line depicts the study’s confidence interval at 95%;  the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.
the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.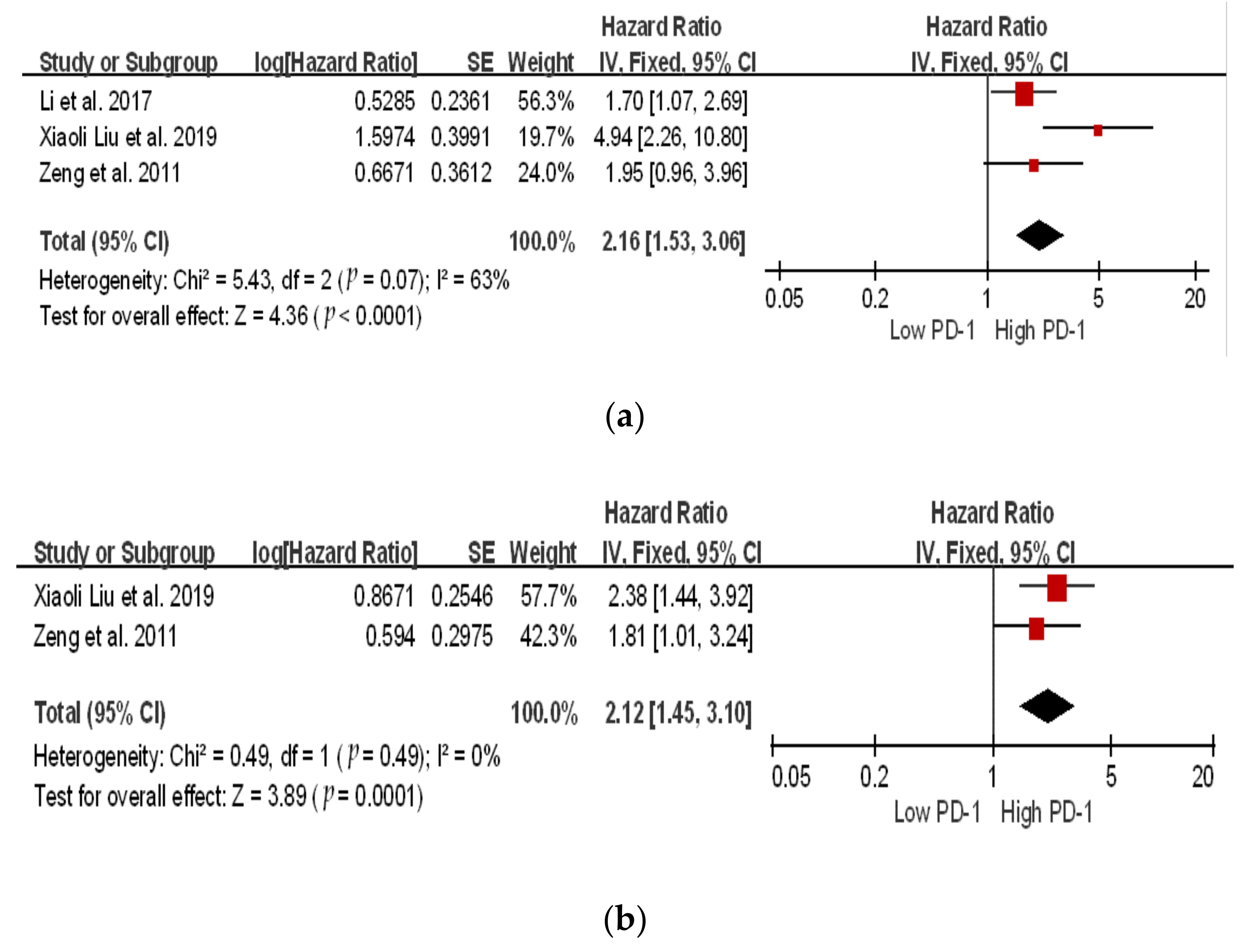
 The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;
The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;  the black line depicts the study’s confidence interval at 95%;
the black line depicts the study’s confidence interval at 95%;  the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.
the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.
 The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;
The location of the square shows the risk ratio, while the size of the square represents the individual effect of research;  the black line depicts the study’s confidence interval at 95%;
the black line depicts the study’s confidence interval at 95%;  the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.
the edge of the diamond denotes the 95% confidence interval for the pooled hazard ratio.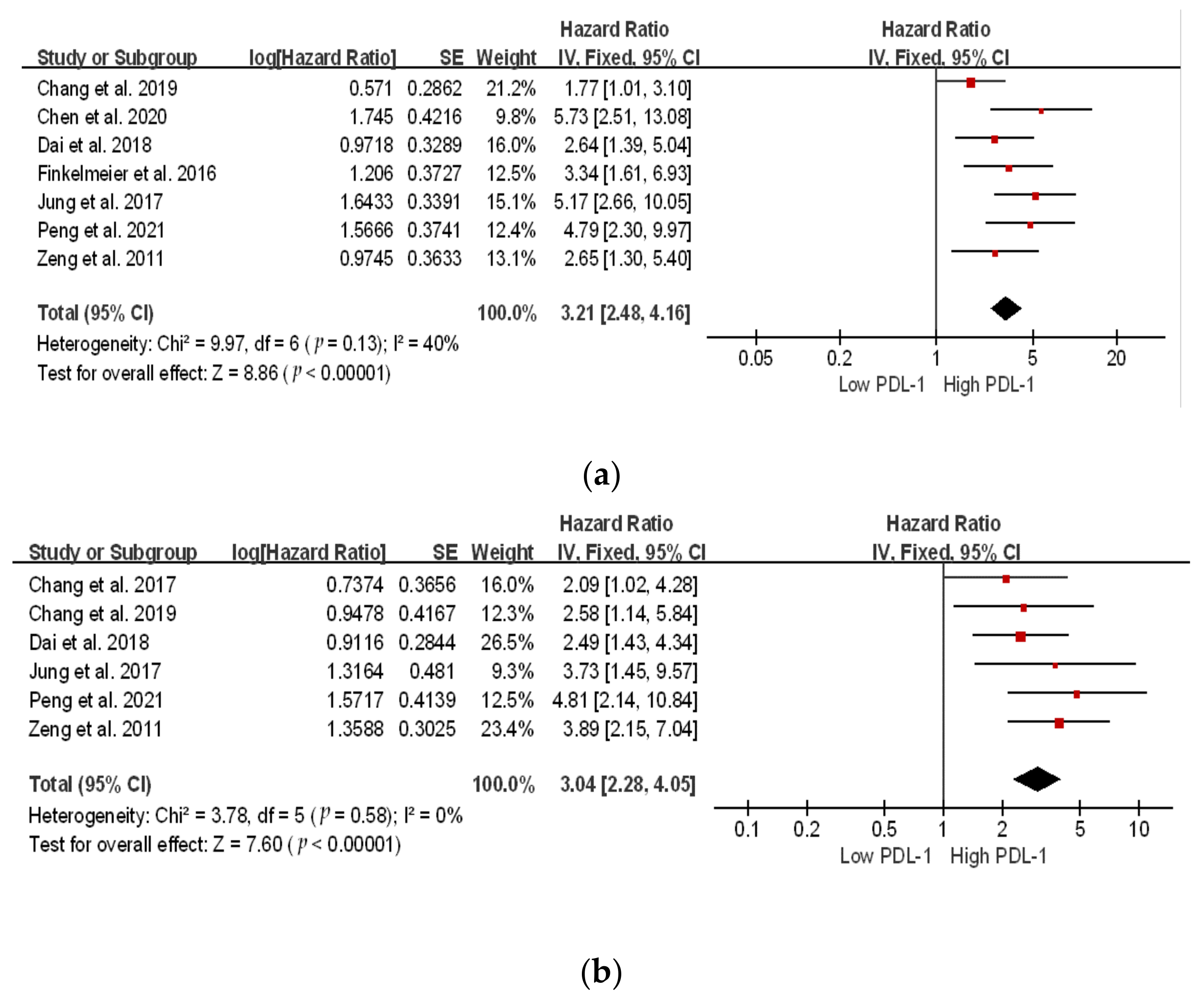

| Reference | Year | Country | Patients (n) | Sample Source | Assessment Method | Cut-Off | Outcome | Follow-Up (Months) |
|---|---|---|---|---|---|---|---|---|
| Peng et al. [21] | 2021 | China | 126 | Tumor tissue | IHC | ≥3 | OS, DFS * | 84 |
| Dai et al. [22] | 2018 | China | 90 | OS, DFS * | 228 | |||
| Chang et al. [23] | 2017 | Korea | 146 | DFS * | 33 | |||
| Jung et al. [24] | 2017 | Korea | 85 | OS, DFS * | - | |||
| Chen et al. [25] | 2020 | China | 74 | Tumor tissue | IHC | ≥4 | OS, DFS * | - |
| Zeng et al. [26] | 2011 | China | 141 | Tumor tissue | IHC | ≥2 | OS, DFS † | |
| Li et al. [27] | 2017 | China | 285 | Serum | ELISA | >10 ng/mL | OS ** | 36 |
| Finkelmeier et al. [28] | 2016 | Germany | 215 | >0.8 ng/mL | OS * | 10 | ||
| Xiaoli Liu et al. [29] | 2019 | China | 122 | PMBC | Flow cytometry | 12.8% CD8+ Cells | OS, PFS ** | 14.75 |
| Characteristics | Studies (n) | Participants (n) | Pooled OR (95% CI) | p-Value | Heterogeneity | ||
|---|---|---|---|---|---|---|---|
| I2 (%) | p-Value | Model | |||||
| Sex (Male vs. Female) | 3 | 407 | 1.39 [0.81, 2.37] | 0.23 | 0 | 0.60 | Fixed |
| Age (<50 vs. >50) | 3 | 417 | 1.12 [0.73, 1.71] | 0.61 | 26 | 0.26 | Fixed |
| BCLC Stage (A vs. B) | 2 | 229 | 0.52 [0.04, 7.27] | 0.62 | 92 | 0.0005 | Random |
| HBV History (Present vs. Absent) | 2 | 261 | 1.25 [0.26, 5.94] | 0.68 | 77 | 0.001 | Random |
| AFP (ng/mL) (≤25 vs. ≥25) | 6 | 646 | 4.63 [1.13, 19.09] | 0.03 * | 86 | 0.001 | Random |
| Tumor Size (≤5 vs. ≥5) | 6 | 646 | 0.58 [0.29, 1.16] | 0.12 | 0 | 0.57 | Fixed |
| Tumor Number (Single vs. Multiple) | 4 | 451 | 1.89 [1.08, 3.33] | 0.003 * | 10 | 0.33 | Fixed |
| Characteristics | Studies (n) | Participants (n) | Pooled OR (95% CI) | p-Value | Heterogeneity | ||
|---|---|---|---|---|---|---|---|
| I2 (%) | p-Value | Model | |||||
| Sex (Male vs. Female) | 6 | 656 | 1.95 [1.24, 3.06] | 0.004 * | 50 | 0.08 | Fixed |
| Age (<50 vs. >50) | 6 | 656 | 1.21 [0.88, 1.67] | 0.25 | 30 | 0.21 | Fixed |
| BCLC Stage (A vs. B) | 3 | 283 | 1.04 [0.11, 9.85] | 0.97 | 90 | <0.0001 | Random |
| HBV History (Present vs. Absent) | 5 | 498 | 1.04 [0.63, 1.70] | 0.88 | 0 | 0.49 | Fixed |
| Child Pugh Score (5 vs. 6) | 2 | 261 | 0.84 [0.42, 1.67] | 0.62 | 0 | 0.80 | Fixed |
| AFP (ng/mL) (≤25 vs. ≥25) | 6 | 646 | 0.85 [0.61, 1.18] | 0.33 | 38 | 0.16 | Fixed |
| Tumor Size (≤5 vs. ≥5) | 6 | 646 | 2.08 [1.46, 2.98] | <0.0001 * | 75 | 0.001 | Fixed |
| Tumor Number (Single vs. Multiple) | 4 | 451 | 1.16 [0.73, 1.86] | 0.52 | 50 | <0.11 | Fixed |
| Tumor Stage (I + II vs. III + IV) | 3 | 264 | 1.39 [0.70, 2.75] | 0.35 | 0 | 0.54 | Fixed |
| Cirrhosis (Positive vs. Negative) | 3 | 305 | 0.34 [0.12, 0.95] | 0.84 | 23 | 0.27 | Fixed |
| Clinical Setting | Regimen | Starting Date | Estimated Study Completion Date | Patients | Trial Name/Number |
|---|---|---|---|---|---|
| First-line systemic | Atezolizumab–cabozantinib | 10 June 2018 | 1 December 2023 | 740 | COSMIC-312/NCT03755791 |
| SHR-1210-apatinib | 10 June 2019 | June 2022 | 543 | NA/NCT03764293 | |
| Durvalumab–tremelimumab | 11 October 2017 | 27 August 2024 | 1504 | HIMALAYA/NCT03298451 | |
| Pembrolizumab–envatinib | 31 December 2018 | 31 December 2023 | 794 | LEAP-002/NCT03713593 | |
| Tislelizumab | 28 December 2017 | May 2022 | 674 | RATIONLALE-301/ NCT03412773 | |
| Sintilimab-IBI305 (anti-VEGF) | 11 February 2019 | December 2022 | 595 | ORIENT-32/ NCT03794440 | |
| Adjuvant resection/ablation | Nivolumab | 18 April 2018 | 16 December 2025 | 545 | CheckMate-9DX/ NCT03383458 |
| Durvalumab–bevacizumab | 29 April 2019 | 31 May 2024 | 908 | EMERALD-2/ NCT03847428 | |
| Pembrolizumab | 28 May 2019 | 30 June 2025 | 950 | KEYNOTE-937/ NCT03867084 | |
| Atezolizumab–bevacizumab | 31 December 2019 | 16 July 2027 | 668 | IMbrave-050/ NCT04102098 | |
| Adjuvant TACE | Durvalumab or Durvalumab–bevacizumab | 30 November 2018 | 19 August 2024 | 724 | EMERALD-1/ NCT03778957 |
| Pembrolizumab–lenvatinib | 22 May 2020 | 31 December 2029 | 950 | LEAP-012/ NCT04246177 | |
| Nivolumab or Nivolumab–ipilimumab | 15 September 2020 | 29 January 2024 | 26 | CheckMate-74W/ NCT04340193 | |
| Adjuvant TACE beads | Nivolumab | 8 May 2019 | June 2026 | 522 | TACE-3/NCT04268888 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ailia, M.J.; Heo, J.; Yoo, S.Y. Navigating through the PD-1/PDL-1 Landscape: A Systematic Review and Meta-Analysis of Clinical Outcomes in Hepatocellular Carcinoma and Their Influence on Immunotherapy and Tumor Microenvironment. Int. J. Mol. Sci. 2023, 24, 6495. https://doi.org/10.3390/ijms24076495
Ailia MJ, Heo J, Yoo SY. Navigating through the PD-1/PDL-1 Landscape: A Systematic Review and Meta-Analysis of Clinical Outcomes in Hepatocellular Carcinoma and Their Influence on Immunotherapy and Tumor Microenvironment. International Journal of Molecular Sciences. 2023; 24(7):6495. https://doi.org/10.3390/ijms24076495
Chicago/Turabian StyleAilia, Muhammad Joan, Jeong Heo, and So Young Yoo. 2023. "Navigating through the PD-1/PDL-1 Landscape: A Systematic Review and Meta-Analysis of Clinical Outcomes in Hepatocellular Carcinoma and Their Influence on Immunotherapy and Tumor Microenvironment" International Journal of Molecular Sciences 24, no. 7: 6495. https://doi.org/10.3390/ijms24076495
APA StyleAilia, M. J., Heo, J., & Yoo, S. Y. (2023). Navigating through the PD-1/PDL-1 Landscape: A Systematic Review and Meta-Analysis of Clinical Outcomes in Hepatocellular Carcinoma and Their Influence on Immunotherapy and Tumor Microenvironment. International Journal of Molecular Sciences, 24(7), 6495. https://doi.org/10.3390/ijms24076495









