Whole-Genome Analysis of Mycobacterium neoaurum DSM 1381 and the Validation of Two Key Enzymes Affecting C22 Steroid Intermediates in Sterol Metabolism
Abstract
1. Introduction
2. Results and Discussion
2.1. Sequencing and Gene Annotation of the Whole Genome of M. neoaurum DSM 1381 and M. neoaurum ATCC 25790
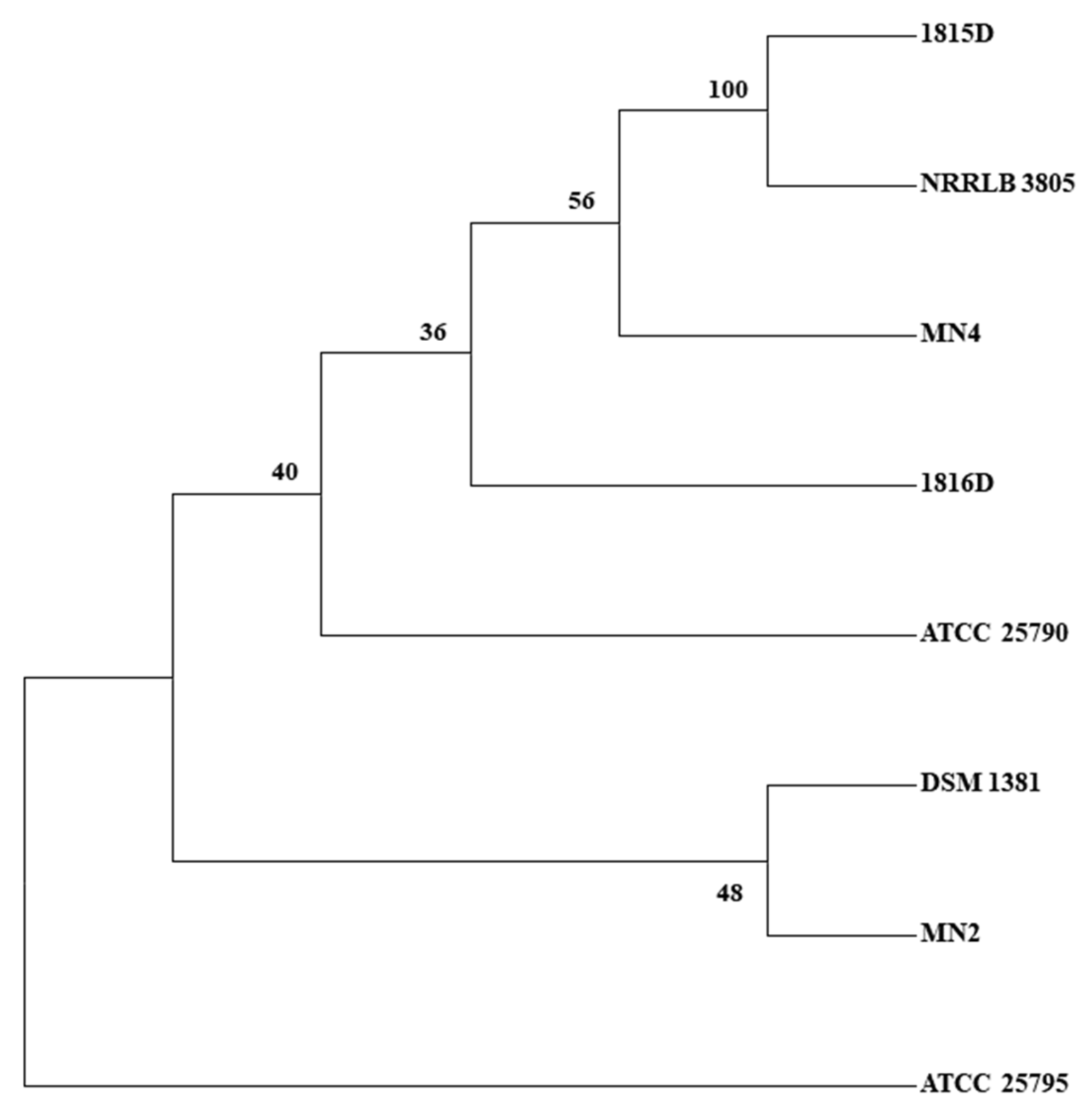
2.2. SNP Discovery Tree Analysis
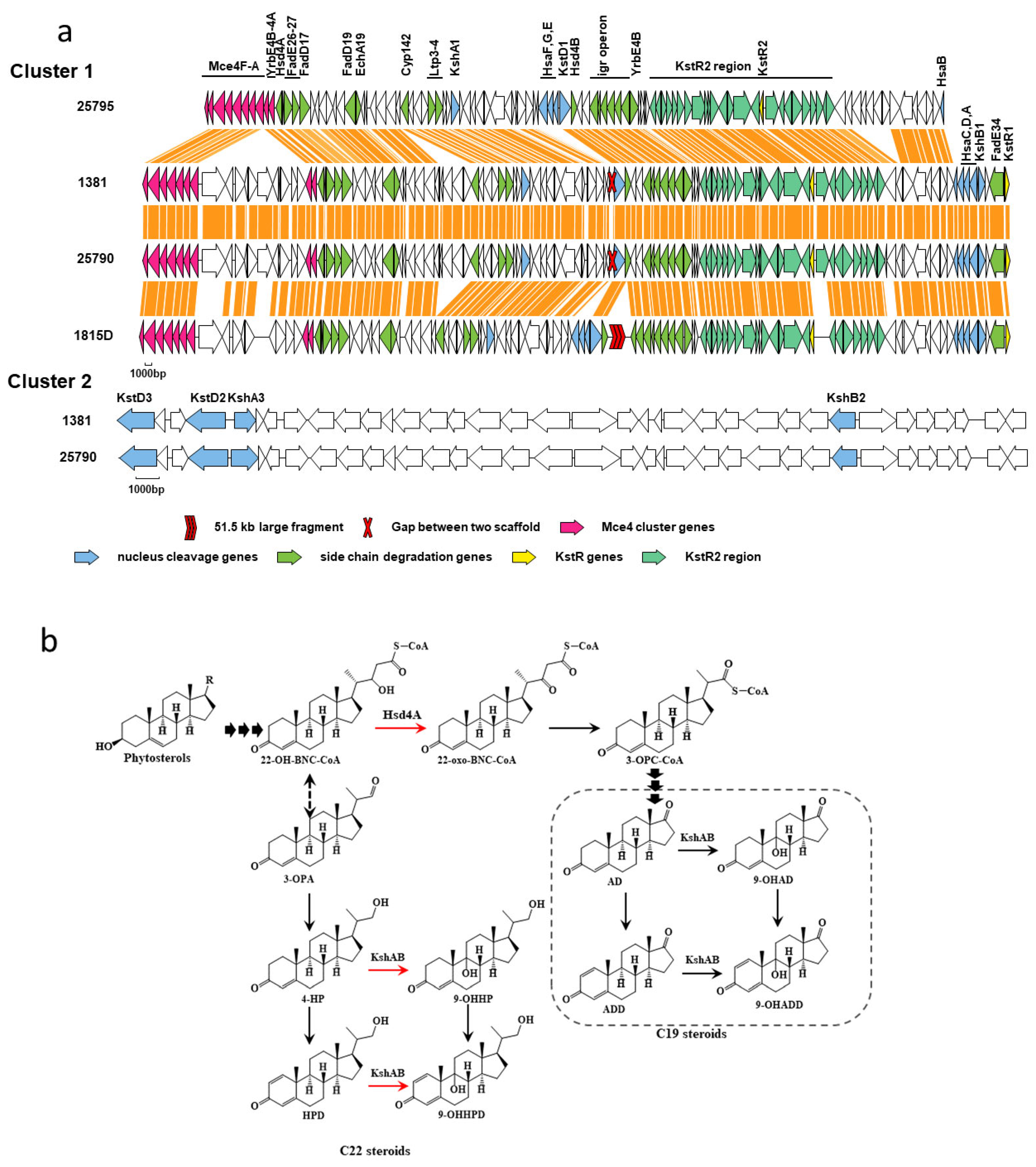
2.3. Prediction of Genes Related to Sterol Metabolism
2.4. Mutation Analysis of Sterol Metabolism-Related Genes
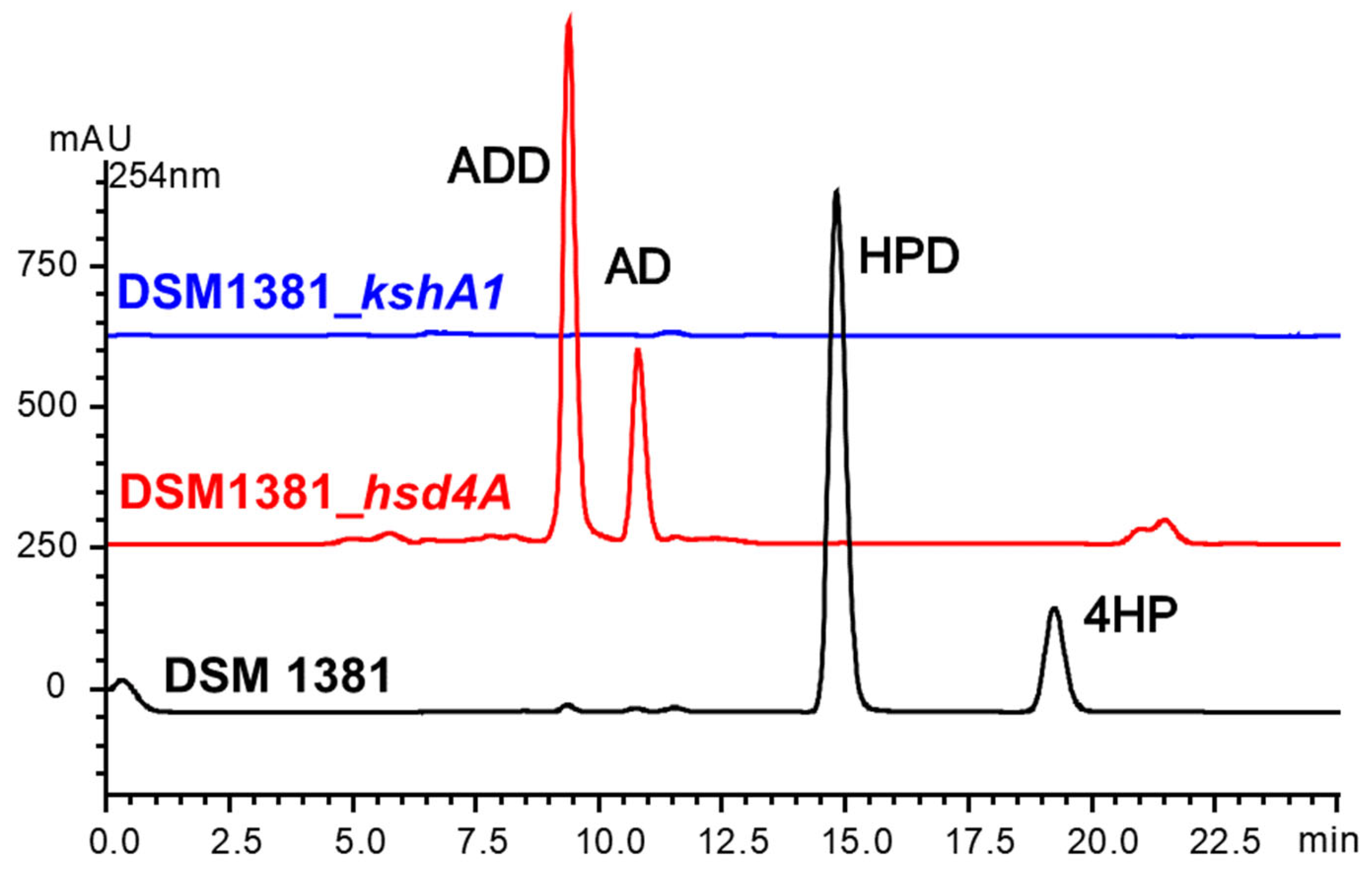
2.5. The Functional Complement of the Selected Mutated Genes
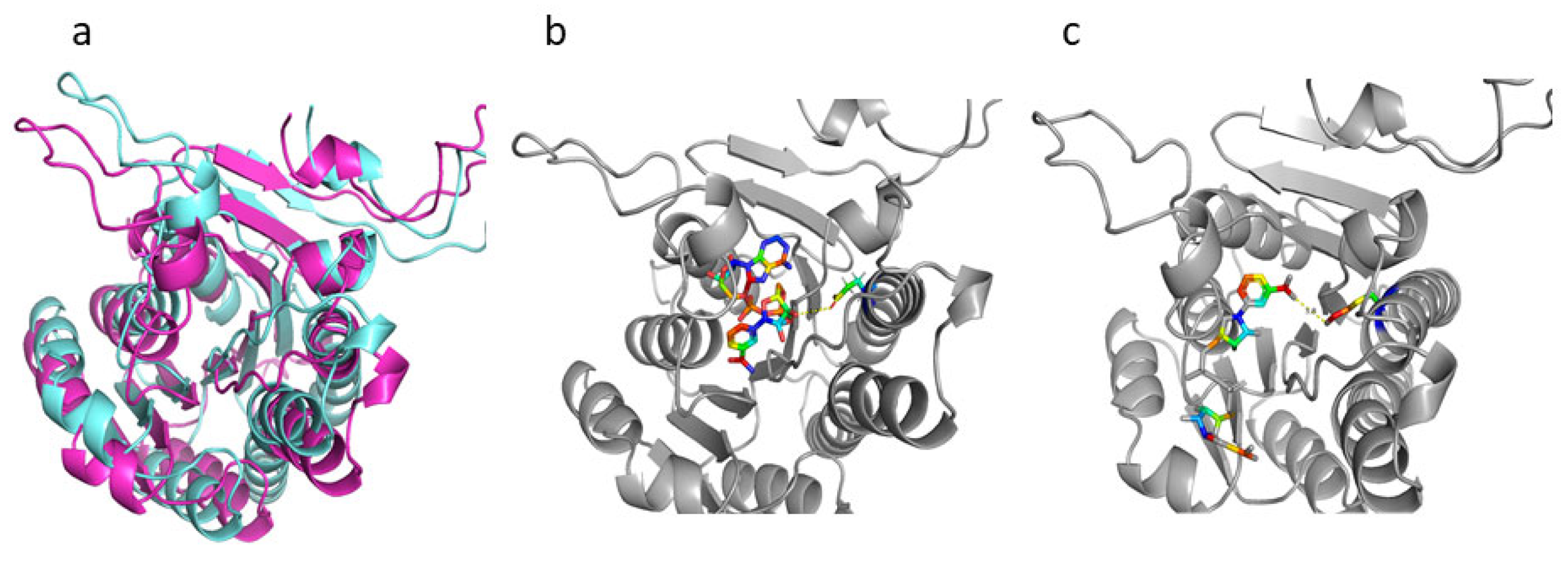
2.6. The Structure and Sequence Analysis of Hsd4A
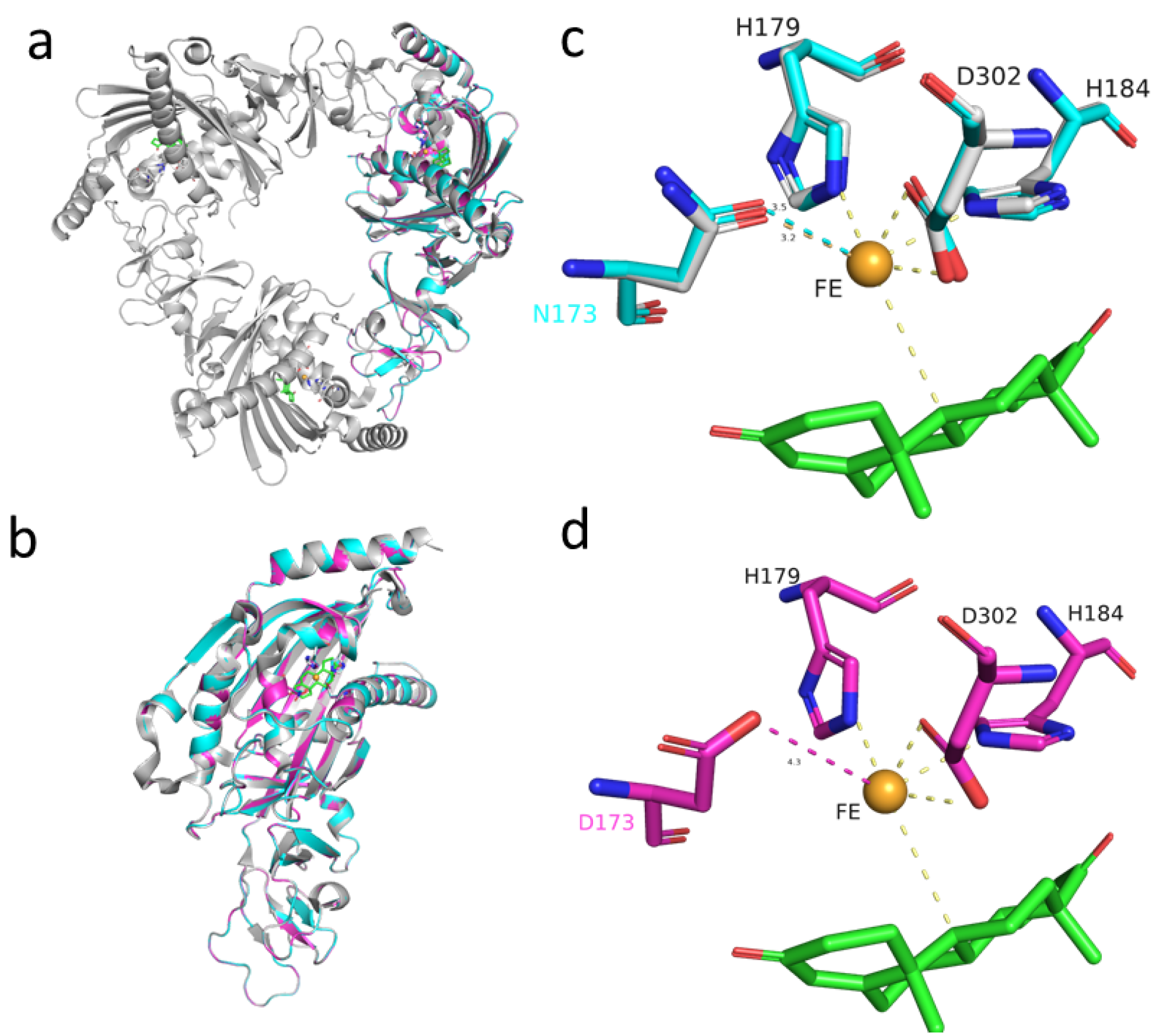
2.7. The Structure and Sequence Analysis of KshA1
3. Materials and Methods
3.1. Chemicals, Strains, and Media
3.2. Characteristics of the Used M. neoaurum Strains
3.3. Genome Sequencing and Annotation
3.4. Whole Gene Alignment Analysis and Visual Display
3.5. SNP and Indel Analysis
3.6. Consensus Parsimonious Tree
3.7. Comparative Analysis of Steroid Degradation-Related Gene Clusters
3.8. Functional Complementation of Differential Genes
3.9. Analysis of Sterol Fermentation Products
3.9.1. Sample Processing
3.9.2. TLC Detection Method for Steroids
3.9.3. The Detection Method of 3-Sterone Compounds
3.9.4. The Quantitative Detection Method of Sterols
3.10. Prediction of the Three-Dimensional Structures of the Hsd4A and KshA1 Enzymes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Thakur, M.K.; Paramanik, V. Role of steroid hormone coregulators in health and disease. Horm. Res. 2009, 71, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Cabezón, L.; Galán, B.; García, J.L. New Insights on Steroid Biotechnology. Front. Microbiol. 2018, 9, 958. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.Q.; Liu, Y.J.; Yao, K.; Liu, H.H.; Tao, X.Y.; Wang, F.Q.; Wei, D.Z. Unraveling and engineering the production of 23,24-bisnorcholenic steroids in sterol metabolism. Sci. Rep. 2016, 6, 21928. [Google Scholar] [CrossRef] [PubMed]
- Hanson, J.R. Steroids: Reactions and partial synthesis. Nat. Prod. Rep. 2004, 21, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Donova, M.V. Steroid Bioconversions. Methods Mol. Biol. 2017, 1645, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Donova, M.V.; Egorova, O.V. Microbial steroid transformations: Current state and prospects. Appl. Microbiol. Biotechnol. 2012, 94, 1423–1447. [Google Scholar] [CrossRef]
- Rugutt, J.K.; Rugutt, K.J. Antimycobacterial activity of steroids, long-chain alcohols and lytic peptides. Nat. Prod. Res. 2012, 26, 1004–1011. [Google Scholar] [CrossRef]
- Xie, R.; Shen, Y.; Qin, N.; Wang, Y.; Su, L.; Wang, M. Genetic differences in ksdD influence on the ADD/AD ratio of Mycobacterium neoaurum. J. Ind. Microbiol. Biotechnol. 2015, 42, 507–513. [Google Scholar] [CrossRef]
- Yuan, C.Y.; Ma, Z.G.; Zhang, J.X.; Liu, X.C.; Du, G.L.; Sun, J.S.; Shi, J.P.; Zhang, B.G. Production of 9,21-dihydroxy-20-methyl-pregna-4-en-3-one from phytosterols in Mycobacterium neoaurum by modifying multiple genes and improving the intracellular environment. Microb. Cell Fact. 2021, 20, 229. [Google Scholar] [CrossRef]
- Yao, K.; Xu, L.Q.; Wang, F.Q.; Wei, D.Z. Characterization and engineering of 3-ketosteroid-Δ1-dehydrogenase and 3-ketosteroid-9α-hydroxylase in Mycobacterium neoaurum ATCC 25795 to produce 9α-hydroxy-4-androstene-3,17-dione through the catabolism of sterols. Metab. Eng. 2014, 24, 181–191. [Google Scholar] [CrossRef]
- Egorova, O.V.; Gulevskaya, S.A.; Puntus, I.F.; Filonov, A.E.; Donova, M.V. Production of androstenedione using mutants of Mycobacterium sp. J. Chem. Technol. Biotechnol. Int. Res. Process Environ. Clean Technol. 2002, 77, 141–147. [Google Scholar]
- Wei, W.; Fan, S.Y.; Wang, F.Q.; Wei, D.Z. Accumulation of androstadiene-dione by overexpression of heterologous 3-ketosteroid Δ1-dehydrogenase in Mycobacterium neoaurum NwIB-01. World J. Microbiol. Biotechnol. 2014, 30, 1947–1954. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-García, A.; Fernández-Alegre, E.; Morales, A.; Sola-Landa, A.; Lorraine, J.; Macdonald, S.; Dovbnya, D.; Smith, M.C.; Donova, M.; Barreiro, C. Complete genome sequence of ‘Mycobacterium neoaurum’ NRRL B-3805, an androstenedione (AD) producer for industrial biotransformation of sterols. J. Biotechnol. 2016, 224, 64–65. [Google Scholar] [CrossRef]
- Bragin, E.Y.; Shtratnikova, V.Y.; Dovbnya, D.V.; Schelkunov, M.I.; Pekov, Y.A.; Malakho, S.G.; Egorova, O.V.; Ivashina, T.V.; Sokolov, S.L.; Ashapkin, V.V.; et al. Comparative analysis of genes encoding key steroid core oxidation enzymes in fast-growing Mycobacterium spp. strains. J. Steroid Biochem. Mol. Biol. 2013, 138, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Imada, Y.; Takahashi, K. Process for Producing Steroidal Alcohols. United States patent US 4,223,091, 16 September 1980. [Google Scholar]
- Zhang, R.; Liu, X.; Wang, Y.; Han, Y.; Sun, J.; Shi, J.; Zhang, B. Identification, function, and application of 3-ketosteroid Δ1-dehydrogenase isozymes in Mycobacterium neoaurum DSM 1381 for the production of steroidic synthons. Microb. Cell Fact. 2018, 17, 77. [Google Scholar] [CrossRef]
- Xu, L.X.; Yang, H.L.; Kuang, M.A.; Tu, Z.C.; Wang, X.L. Comparative genomic analysis of Mycobacterium neoaurum MN2 and MN4 substrate and product tolerance. 3 Biotech. 2017, 7, 181. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Song, S.; Peng, F.; Yang, F.; Chen, T.; Li, X.; Cheng, X.; He, Y.; Huang, Y.; Su, Z. Whole-genome and enzymatic analyses of an androstenedione-producing Mycobacterium strain with residual phytosterol-degrading pathways. Microb. Cell Fact. 2020, 19, 187. [Google Scholar] [CrossRef]
- Van der Geize, R.; Yam, K.; Heuser, T.; Wilbrink, M.H.; Hara, H.; Anderton, M.C.; Sim, E.; Dijkhuizen, L.; Davies, J.E.; Mohn, W.W.; et al. A gene cluster encoding cholesterol catabolism in a soil actinomycete provides insight into Mycobacterium tuberculosis survival in macrophages. Proc. Natl. Acad. Sci. USA 2007, 104, 1947–1952. [Google Scholar] [CrossRef]
- Yao, K.; Wang, F.Q.; Zhang, H.C.; Wei, D.Z. Identification and engineering of cholesterol oxidases involved in the initial step of sterols catabolism in Mycobacterium neoaurum. Metab. Eng. 2013, 15, 75–87. [Google Scholar] [CrossRef]
- Mindnich, R.; Möller, G.; Adamski, J. The role of 17 beta-hydroxysteroid dehydrogenases. Mol. Cell. Endocrinol. 2004, 218, 7–20. [Google Scholar] [CrossRef]
- Uhía, I.; Galán, B.; Kendall, S.L.; Stoker, N.G.; García, J.L. Cholesterol metabolism in Mycobacterium smegmatis. Environ. Microbiol. Rep. 2012, 4, 168–182. [Google Scholar] [CrossRef]
- Kreit, J. Aerobic catabolism of sterols by microorganisms: Key enzymes that open the 3-ketosteroid nucleus. FEMS Microbiol. Lett. 2019, 366, fnz173. [Google Scholar] [CrossRef] [PubMed]
- Capyk, J.K.; Kalscheuer, R.; Stewart, G.R.; Liu, J.; Kwon, H.; Zhao, R.; Okamoto, S.; Jacobs, W.R., Jr.; Eltis, L.D.; Mohn, W.W. Mycobacterial cytochrome p450 125 (cyp125) catalyzes the terminal hydroxylation of c27 steroids. J. Biol. Chem. 2009, 284, 35534–35542. [Google Scholar] [CrossRef]
- Fernandes, P.; Cruz, A.; Angelova, B.; Pinheiro, H.M.; Cabral, J.M.S. Microbial conversion of steroid compounds: Recent developments. Enzym. Microb. Technol. 2003, 32, 688–705. [Google Scholar] [CrossRef]
- García, J.L.; Uhía, I.; Galán, B. Catabolism and biotechnological applications of cholesterol degrading bacteria. Microb. Biotechnol. 2012, 5, 679–699. [Google Scholar] [CrossRef]
- Xiong, L.B.; Liu, H.H.; Xu, L.Q.; Sun, W.J.; Wang, F.Q.; Wei, D.Z. Improving the production of 22-hydroxy-23,24-bisnorchol-4-ene-3-one from sterols in Mycobacterium neoaurum by increasing cell permeability and modifying multiple genes. Microb. Cell Fact. 2017, 16, 89. [Google Scholar] [CrossRef] [PubMed]
- van der Geize, R.; Hessels, G.I.; van Gerwen, R.; van der Meijden, P.; Dijkhuizen, L. Molecular and functional characterization of kshA and kshB, encoding two components of 3-ketosteroid 9alpha-hydroxylase, a class IA monooxygenase, in Rhodococcus erythropolis strain SQ1. Mol. Microbiol. 2002, 45, 1007–1018. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology modelling of protein structures and complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef]
- Eddy, S.R. Accelerated Profile HMM Searches. PLoS Comput. Biol. 2011, 7, e1002195. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Dranow, D.M.; Fairman, J.W.; Edwards, T.E.; Lorimer, D. Crystal Structure of a Putative Short Chain Dehydrogenase from Mycobacterium smegmatis. Acta Crystallogr. Sect. F Struct. Biol. Commun. 2022, 78, 25–30. [Google Scholar]
- Petrusma, M.; Dijkhuizen, L.; van der Geize, R. Rhodococcus rhodochrous DSM 43269 3-ketosteroid 9alpha-hydroxylase, a two-component iron-sulfur-containing monooxygenase with subtle steroid substrate specificity. Appl. Environ. Microbiol. 2009, 75, 5300–5307. [Google Scholar] [CrossRef] [PubMed]
- Capyk, J.K.; D’Angelo, I.; Strynadka, N.C.; Eltis, L.D. Characterization of 3-ketosteroid 9{alpha}-hydroxylase, a Rieske oxygenase in the cholesterol degradation pathway of Mycobacterium tuberculosis. J. Biol. Chem. 2009, 284, 9937–9946. [Google Scholar] [CrossRef] [PubMed]
- Capyk, J.K.; Casabon, I.; Gruninger, R.; Strynadka, N.C.; Eltis, L.D. Activity of 3-ketosteroid 9α-hydroxylase (KshAB) indicates cholesterol side chain and ring degradation occur simultaneously in Mycobacterium tuberculosis. J. Biol. Chem. 2011, 286, 40717–40724. [Google Scholar] [CrossRef]
- Penfield, J.S.; Worrall, L.J.; Strynadka, N.C.; Eltis, L.D. Substrate specificities and conformational flexibility of 3-ketosteroid 9α-hydroxylases. J. Biol. Chem. 2014, 289, 25523–25536. [Google Scholar] [CrossRef]
- Donova, M.V.; Gulevskaya, S.A.; Dovbnya, D.V.; Puntus, I.F. Mycobacterium sp. mutant strain producing 9alpha-hydroxyandrostenedione from sitosterol. Appl. Microbiol. Biotechnol. 2005, 67, 671–678. [Google Scholar] [CrossRef]
- Marsheck, W.J.; Kraychy, S.; Muir, R.D. Microbial degradation of sterols. Appl. Microbiol. 1972, 23, 72–77. [Google Scholar] [CrossRef]
- Delcher, A.L.; Bratke, K.A.; Powers, E.C.; Salzberg, S.L. Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics 2007, 23, 673–679. [Google Scholar] [CrossRef]
- Delcher, A.L.; Harmon, D.; Kasif, S.; White, O.; Salzberg, S.L. Improved microbial gene identification with GLIMMER. Nucleic Acids Res. 1999, 27, 4636–4641. [Google Scholar] [CrossRef]
- Wang, K.; Li, M.; Hakonarson, H. ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010, 38, e164. [Google Scholar] [CrossRef]
- Stover, C.K.; de la Cruz, V.F.; Fuerst, T.R.; Burlein, J.E.; Benson, L.A.; Bennett, L.T.; Bansal, G.P.; Young, J.F.; Lee, M.H.; Hatfull, G.F.; et al. New use of BCG for recombinant vaccines. Nature 1991, 351, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Mirdita, M.; Schütze, K.; Moriwaki, Y.; Heo, L.; Ovchinnikov, S.; Steinegger, M. ColabFold: Making protein folding accessible to all. Nat. Methods 2022, 19, 679–682. [Google Scholar] [CrossRef] [PubMed]
- AlQuraishi, M. End-to-End Differentiable Learning of Protein Structure. Cell Syst. 2019, 8, 292–301.e3. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, F.B.; Wagstaff, E.; Dauparas, J.; Posner, I. Iterative SE(3)-Transformers. In Proceedings of the 5th International Conference on Geometric Science of Information (GSI), Sorbonne University, Paris, France, 21–23 July 2021; pp. 585–595. [Google Scholar]
- Vaswani, A.; Shazeer, N.; Parmar, N.; Uszkoreit, J.; Jones, L.; Gomez, A.N.; Kaiser, L.; Polosukhin, I. Attention Is All You Need. In Proceedings of the 31st Annual Conference on Neural Information Processing Systems (NIPS), Long Beach, CA, USA, 4–9 December 2017. [Google Scholar]
- Toro, A.; Ambrus, G. Oxidative decarboxylation of 17(20)-dehydro-23,24-dinorcholanoic acids. Tetrahedron Lett. 1990, 31, 3475–3476. [Google Scholar] [CrossRef]
- Xiong, L.B.; Liu, H.H.; Zhao, M.; Liu, Y.J.; Song, L.; Xie, Z.Y.; Xu, Y.X.; Wang, F.Q.; Wei, D.Z. Enhancing the bioconversion of phytosterols to steroidal intermediates by the deficiency of kasB in the cell wall synthesis of Mycobacterium neoaurum. Microb. Cell Fact. 2020, 19, 80. [Google Scholar] [CrossRef]
- Sun, W.J.; Wang, L.; Liu, H.H.; Liu, Y.J.; Ren, Y.H.; Wang, F.Q.; Wei, D.Z. Characterization and engineering control of the effects of reactive oxygen species on the conversion of sterols to steroid synthons in Mycobacterium neoaurum. Metab. Eng. 2019, 56, 97–110. [Google Scholar] [CrossRef]

| Strains | DSM 1381 | ATCC 25790 |
|---|---|---|
| Gene num | 5232 | 5348 |
| Gene total length | 5,577,916 bp | 5,577,210 bp |
| Gene average length | 971 bp | 951 bp |
| Gene density | 0.937 genes per kb | 0.958 genes per kb |
| GC content in gene region (%) | 67.2 | 67.2 |
| Gene/Genome (%) | 91.1 | 91.3 |
| Intergenetic region length | 494,836 bp | 487,875 bp |
| GC content in the intergenetic region (%) | 62.2 | 62.2 |
| Intergenetic length/Genome (%) | 8.97 | 8.74 |
| Description of the Gene Product | Gene ID | SNV | aa Mutation | |||
|---|---|---|---|---|---|---|
| Nonsynonymous SNV | ATCC 25790 | DSM 1381 | ATCC 25790 | DSM 1381 | ATCC 25790 | DSM 1381 |
| KshA1 | 807 | 14 | 517A | 517G | 173Asn | 173Asp |
| Hsd4A | 775 | 46 | 511A | 511G | 171Lys | 171Glu |
| NAD(P)/FAD-dependent oxidoreductase | 1328 | 1296 | 1025C | 1025G | 342Ala | 342Gly |
| FadR family transcriptional regulator | 2235 | 2188 | 446T | 446A | 149Leu | 149Gln |
| SDR family NAD(P)-dependent oxidoreductase | 1151 | 1123 | upstream 34G | upstream 34T | ||
| Indel | ATCC 25790 | DSM 1381 | Indel in DSM 1381 | Mutation start position | ||
| 2,4-dienoyl-CoA reductase | 2435 | 2389 | insert, 4 bp | 660 | ||
| acyl-CoA synthetase | 2617–2616 | 2573 | gap, 34 bp | 807 | ||
| acyltransferase | 187 | 624 | gap, 1 bp | 461 | ||
| Strain | Abbreviation | Main Product | Side Products | Reference |
|---|---|---|---|---|
| M. neoaurum DSM 1381 | DSM 1381 | HPD | 4HP | [15] |
| M. neoaurum ATCC 25790 (M. neoaurum ATCC 25790) | ATCC 25790 | NA | NA | [15] |
| M. neoaurum ATCC 25795 | ATCC 25795 | NA | NA | |
| Mycobacterium sp. VKM Ac-1815D | Ac 1815D | AD | ADD, HP, HPD | [37] |
| Mycobacterium sp. VKM Ac-1816D | Ac 1816D | ADD | AD, HP, HPD | [14] |
| M. neoaurum MN2 | MN2 | AD | ADD | [17] |
| M. neoaurum MN4 | MN4 | AD | ADD | [17] |
| M. neoaurum NRRLB 3805 | NRRLB 3805 | NA | NA | [38] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Zhang, R.; Song, S.; Su, Z.; Shi, J.; Cao, H.; Zhang, B. Whole-Genome Analysis of Mycobacterium neoaurum DSM 1381 and the Validation of Two Key Enzymes Affecting C22 Steroid Intermediates in Sterol Metabolism. Int. J. Mol. Sci. 2023, 24, 6148. https://doi.org/10.3390/ijms24076148
Zhang J, Zhang R, Song S, Su Z, Shi J, Cao H, Zhang B. Whole-Genome Analysis of Mycobacterium neoaurum DSM 1381 and the Validation of Two Key Enzymes Affecting C22 Steroid Intermediates in Sterol Metabolism. International Journal of Molecular Sciences. 2023; 24(7):6148. https://doi.org/10.3390/ijms24076148
Chicago/Turabian StyleZhang, Jingxian, Ruijie Zhang, Shikui Song, Zhengding Su, Jiping Shi, Huijin Cao, and Baoguo Zhang. 2023. "Whole-Genome Analysis of Mycobacterium neoaurum DSM 1381 and the Validation of Two Key Enzymes Affecting C22 Steroid Intermediates in Sterol Metabolism" International Journal of Molecular Sciences 24, no. 7: 6148. https://doi.org/10.3390/ijms24076148
APA StyleZhang, J., Zhang, R., Song, S., Su, Z., Shi, J., Cao, H., & Zhang, B. (2023). Whole-Genome Analysis of Mycobacterium neoaurum DSM 1381 and the Validation of Two Key Enzymes Affecting C22 Steroid Intermediates in Sterol Metabolism. International Journal of Molecular Sciences, 24(7), 6148. https://doi.org/10.3390/ijms24076148







