The Role of α-Linolenic Acid and Its Oxylipins in Human Cardiovascular Diseases
Abstract
1. Introduction
2. ALA Intake, Human Inflammation and CVDs
2.1. Association of ALA with the Lipid Profile and Markers of Inflammation in Human Plasma
2.2. ALA Supplementation and Cardiovascular Diseases
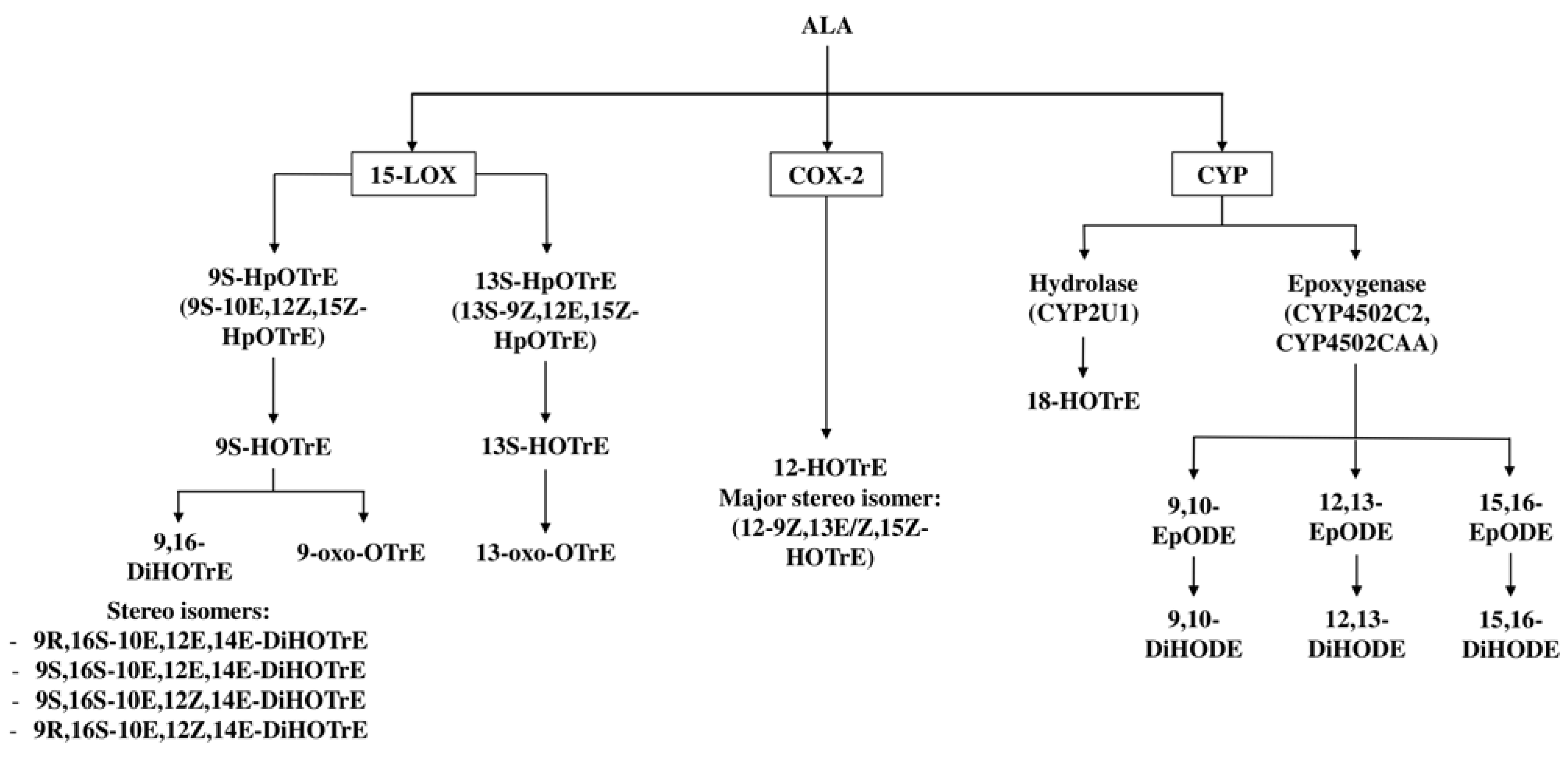
3. Enzymatic Metabolism of ALA to Oxylipins
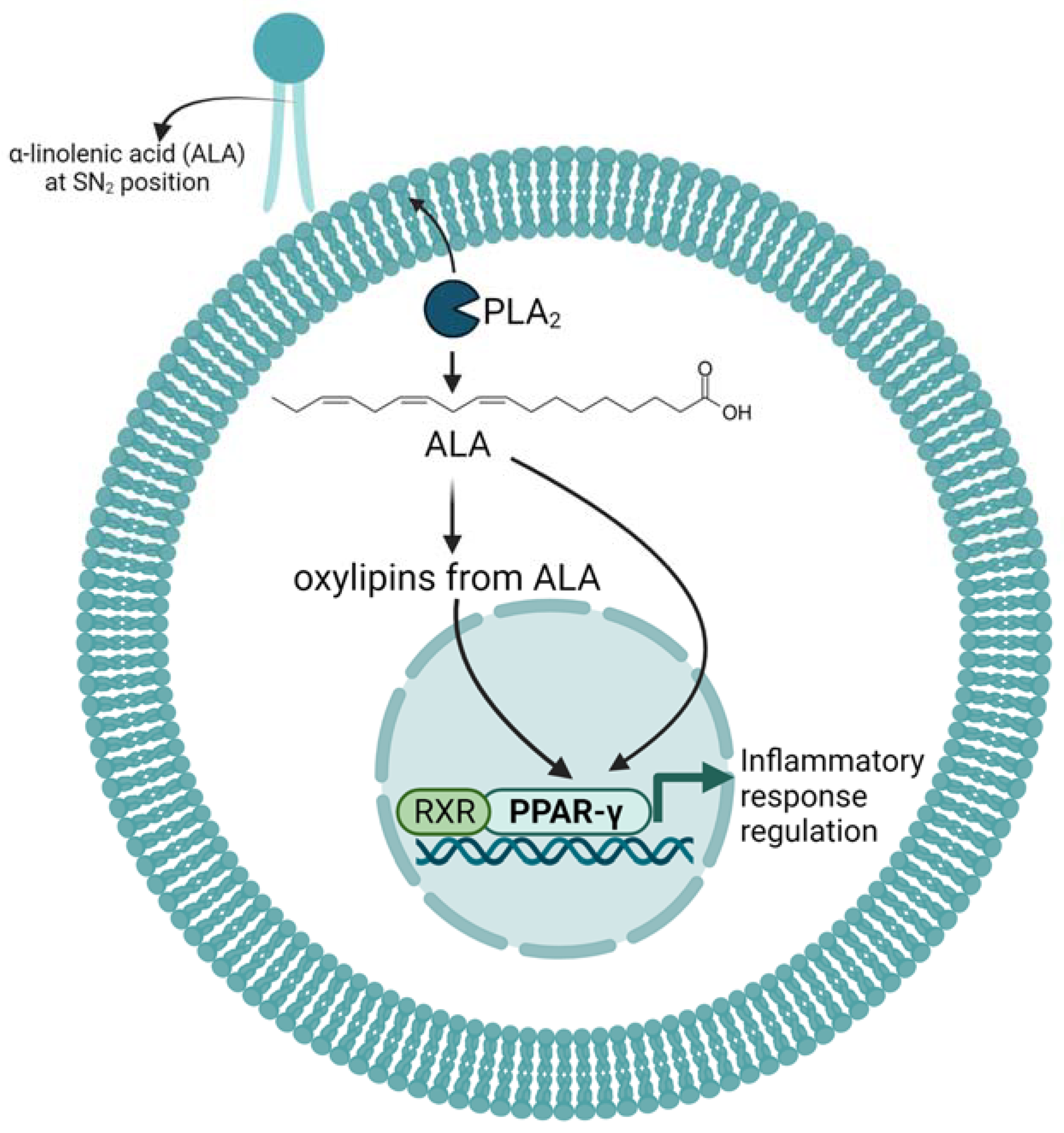
4. Effects and Mechanism of ALA and Its Oxylipins on Inflammation in Mice and Cell Cultures
5. Effect of ALA Supplementation on Oxylipin Profiles
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Burr, G.O.; Burr, M.M. On the nature and rôle of the fatty acids essential in nutrition. J. Biol. Chem. 1930, 86, 587–621. [Google Scholar] [CrossRef]
- Burns-Whitmore, B.; Froyen, E.; Heskey, C.; Parker, T.; San Pablo, G. Alpha-Linolenic and Linoleic Fatty Acids in the Vegan Diet: Do They Require Dietary Reference Intake/Adequate Intake Special Consideration? Nutrients 2019, 11, 2365. [Google Scholar] [CrossRef] [PubMed]
- Cholewski, M.; Tomczykowa, M.; Tomczyk, M. A Comprehensive Review of Chemistry, Sources and Bioavailability of Omega-3 Fatty Acids. Nutrients 2018, 10, 1662. [Google Scholar] [CrossRef]
- Arterburn, L.M.; Hall, E.B.; Oken, H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am. J. Clin. Nutr. 2006, 83, 1467s–1476s. [Google Scholar] [CrossRef] [PubMed]
- Barceló-Coblijn, G.; Murphy, E.J. Alpha-linolenic acid and its conversion to longer chain n-3 fatty acids: Benefits for human health and a role in maintaining tissue n-3 fatty acid levels. Prog. Lipid Res. 2009, 48, 355–374. [Google Scholar] [CrossRef]
- Chen, X.; Wu, Y.; Zhang, Z.; Zheng, X.; Wang, Y.; Yu, M.; Liu, G. Effects of the rs3834458 Single Nucleotide Polymorphism in FADS2 on Levels of n-3 Long-chain Polyunsaturated Fatty Acids: A Meta-analysis. Prostaglandins Leukot. Essent. Fat. Acids 2019, 150, 1–6. [Google Scholar] [CrossRef]
- Wood, K.E.; Mantzioris, E.; Gibson, R.A.; Ramsden, C.E.; Muhlhausler, B.S. The effect of modifying dietary LA and ALA intakes on omega-3 long chain polyunsaturated fatty acid (n-3 LCPUFA) status in human adults: A systematic review and commentary. Prostaglandins Leukot. Essent. Fat. Acids 2015, 95, 47–55. [Google Scholar] [CrossRef]
- Wawrzyniak, P.; Noureddine, N.; Wawrzyniak, M.; Lucchinetti, E.; Krämer, S.D.; Rogler, G.; Zaugg, M.; Hersberger, M. Nutritional Lipids and Mucosal Inflammation. Mol. Nutr. Food Res. 2021, 65, e1901269. [Google Scholar] [CrossRef]
- Dawczynski, C.; Plagge, J.; Jahreis, G.; Liebisch, G.; Höring, M.; Seeliger, C.; Ecker, J. Dietary PUFA Preferably Modify Ethanolamine-Containing Glycerophospholipids of the Human Plasma Lipidome. Nutrients 2022, 14, 3055. [Google Scholar] [CrossRef]
- Turk, H.F.; Chapkin, R.S. Membrane lipid raft organization is uniquely modified by n-3 polyunsaturated fatty acids. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 43–47. [Google Scholar] [CrossRef]
- Im, D.S. Omega-3 fatty acids in anti-inflammation (pro-resolution) and GPCRs. Prog. Lipid Res. 2012, 51, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Moniri, N.H. Free-fatty acid receptor-4 (GPR120): Cellular and molecular function and its role in metabolic disorders. Biochem. Pharmacol. 2016, 110–111, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.Y.; Talukdar, S.; Bae, E.J.; Imamura, T.; Morinaga, H.; Fan, W.; Li, P.; Lu, W.J.; Watkins, S.M.; Olefsky, J.M. GPR120 is an omega-3 fatty acid receptor mediating potent anti-inflammatory and insulin-sensitizing effects. Cell 2010, 142, 687–698. [Google Scholar] [CrossRef]
- Serhan, C.N.; Levy, B.D. Resolvins in inflammation: Emergence of the pro-resolving superfamily of mediators. J. Clin. Investig. 2018, 128, 2657–2669. [Google Scholar] [CrossRef]
- Dyall, S.C.; Balas, L.; Bazan, N.G.; Brenna, J.T.; Chiang, N.; da Costa Souza, F.; Dalli, J.; Durand, T.; Galano, J.M.; Lein, P.J.; et al. Polyunsaturated fatty acids and fatty acid-derived lipid mediators: Recent advances in the understanding of their biosynthesis, structures, and functions. Prog. Lipid Res. 2022, 86, 101165. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 139, e1082–e1143. [Google Scholar] [CrossRef]
- Rimm, E.B.; Appel, L.J.; Chiuve, S.E.; Djoussé, L.; Engler, M.B.; Kris-Etherton, P.M.; Mozaffarian, D.; Siscovick, D.S.; Lichtenstein, A.H. Seafood Long-Chain n-3 Polyunsaturated Fatty Acids and Cardiovascular Disease: A Science Advisory From the American Heart Association. Circulation 2018, 138, e35–e47. [Google Scholar] [CrossRef]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; Chapman, M.J.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U.; Pedersen, T.R.; et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias: The Task Force for the Management of Dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS) Developed with the special contribution of the European Assocciation for Cardiovascular Prevention & Rehabilitation (EACPR). Atherosclerosis 2016, 253, 281–344. [Google Scholar] [CrossRef]
- Skulas-Ray, A.C.; Wilson, P.W.F.; Harris, W.S.; Brinton, E.A.; Kris-Etherton, P.M.; Richter, C.K.; Jacobson, T.A.; Engler, M.B.; Miller, M.; Robinson, J.G.; et al. Omega-3 Fatty Acids for the Management of Hypertriglyceridemia: A Science Advisory From the American Heart Association. Circulation 2019, 140, e673–e691. [Google Scholar] [CrossRef]
- Chen, H.; Deng, G.; Zhou, Q.; Chu, X.; Su, M.; Wei, Y.; Li, L.; Zhang, Z. Effects of eicosapentaenoic acid and docosahexaenoic acid versus α-linolenic acid supplementation on cardiometabolic risk factors: A meta-analysis of randomized controlled trials. Food Funct. 2020, 11, 1919–1932. [Google Scholar] [CrossRef]
- Yue, H.; Qiu, B.; Jia, M.; Liu, W.; Guo, X.F.; Li, N.; Xu, Z.X.; Du, F.L.; Xu, T.; Li, D. Effects of α-linolenic acid intake on blood lipid profiles: A systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2021, 61, 2894–2910. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Liu, R.; Chang, M.; Huang, J.; Jin, Q.; Wang, X. Effect of dietary alpha-linolenic acid on blood inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Nutr. 2018, 57, 877–891. [Google Scholar] [CrossRef] [PubMed]
- de Abreu, A.M.; Copetti, C.L.K.; Hauschild, D.B.; Di Pietro, P.F.; Wazlawik, E. Effects of supplementation with vegetable sources of alpha-linolenic acid (ALA) on inflammatory markers and lipid profile in individuals with chronic kidney disease: A systematic review and meta-analysis. Clin. Nutr. 2022, 41, 1434–1444. [Google Scholar] [CrossRef]
- Wei, J.; Hou, R.; Xi, Y.; Kowalski, A.; Wang, T.; Yu, Z.; Hu, Y.; Chandrasekar, E.K.; Sun, H.; Ali, M.K. The association and dose-response relationship between dietary intake of α-linolenic acid and risk of CHD: A systematic review and meta-analysis of cohort studies. Br. J. Nutr. 2018, 119, 83–89. [Google Scholar] [CrossRef]
- Pan, A.; Chen, M.; Chowdhury, R.; Wu, J.H.; Sun, Q.; Campos, H.; Mozaffarian, D.; Hu, F.B. α-Linolenic acid and risk of cardiovascular disease: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2012, 96, 1262–1273. [Google Scholar] [CrossRef] [PubMed]
- Naghshi, S.; Aune, D.; Beyene, J.; Mobarak, S.; Asadi, M.; Sadeghi, O. Dietary intake and biomarkers of alpha linolenic acid and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of cohort studies. BMJ 2021, 375, n2213. [Google Scholar] [CrossRef]
- Abdelhamid, A.S.; Brown, T.J.; Brainard, J.S.; Biswas, P.; Thorpe, G.C.; Moore, H.J.; Deane, K.H.; Summerbell, C.D.; Worthington, H.V.; Song, F.; et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2020, 3, Cd003177. [Google Scholar] [CrossRef]
- Lawler, P.R.; Bhatt, D.L.; Godoy, L.C.; Lüscher, T.F.; Bonow, R.O.; Verma, S.; Ridker, P.M. Targeting cardiovascular inflammation: Next steps in clinical translation. Eur. Heart J. 2021, 42, 113–131. [Google Scholar] [CrossRef]
- Chareonrungrueangchai, K.; Wongkawinwoot, K.; Anothaisintawee, T.; Reutrakul, S. Dietary Factors and Risks of Cardiovascular Diseases: An Umbrella Review. Nutrients 2020, 12, 1088. [Google Scholar] [CrossRef]
- Bernasconi, A.A.; Wiest, M.M.; Lavie, C.J.; Milani, R.V.; Laukkanen, J.A. Effect of Omega-3 Dosage on Cardiovascular Outcomes: An Updated Meta-Analysis and Meta-Regression of Interventional Trials. Mayo Clin. Proc. 2021, 96, 304–313. [Google Scholar] [CrossRef]
- Elagizi, A.; Lavie, C.J.; O’Keefe, E.; Marshall, K.; O’Keefe, J.H.; Milani, R.V. An Update on Omega-3 Polyunsaturated Fatty Acids and Cardiovascular Health. Nutrients 2021, 13, 204. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Xu, Z.; Xu, D.; Tan, X. Effect of n-3 polyunsaturated fatty acids on ischemic heart disease and cardiometabolic risk factors: A two-sample Mendelian randomization study. BMC Cardiovasc. Disord. 2021, 21, 532. [Google Scholar] [CrossRef]
- Sala-Vila, A.; Guasch-Ferré, M.; Hu, F.B.; Sánchez-Tainta, A.; Bulló, M.; Serra-Mir, M.; López-Sabater, C.; Sorlí, J.V.; Arós, F.; Fiol, M.; et al. Dietary α-Linolenic Acid, Marine ω-3 Fatty Acids, and Mortality in a Population With High Fish Consumption: Findings From the PREvención con DIeta MEDiterránea (PREDIMED) Study. J. Am. Heart Assoc. 2016, 5, e002543. [Google Scholar] [CrossRef] [PubMed]
- Gabbs, M.; Leng, S.; Devassy, J.G.; Monirujjaman, M.; Aukema, H.M. Advances in Our Understanding of Oxylipins Derived from Dietary PUFAs. Adv. Nutr. 2015, 6, 513–540. [Google Scholar] [CrossRef] [PubMed]
- Dennis, E.A.; Cao, J.; Hsu, Y.H.; Magrioti, V.; Kokotos, G. Phospholipase A2 enzymes: Physical structure, biological function, disease implication, chemical inhibition, and therapeutic intervention. Chem. Rev. 2011, 111, 6130–6185. [Google Scholar] [CrossRef] [PubMed]
- Hersberger, M. Potential role of the lipoxygenase derived lipid mediators in atherosclerosis: Leukotrienes, lipoxins and resolvins. Clin. Chem. Lab. Med. 2010, 48, 1063–1073. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N.; Dalli, J.; Levy, B.D. Lipid mediators in the resolution of inflammation. Cold Spring Harb. Perspect. Biol. 2014, 7, a016311. [Google Scholar] [CrossRef]
- Jimbo, H.; Yuasa, K.; Takagi, K.; Hirashima, T.; Keta, S.; Aichi, M.; Wada, H. Specific Incorporation of Polyunsaturated Fatty Acids into the sn-2 Position of Phosphatidylglycerol Accelerates Photodamage to Photosystem II under Strong Light. Int. J. Mol. Sci. 2021, 22, 10432. [Google Scholar] [CrossRef]
- Defries, D.; Curtis, K.; Petkau, J.C.; Shariati-Ievari, S.; Blewett, H.; Aliani, M. Patterns of alpha-linolenic acid incorporation into phospholipids in H4IIE cells. J. Nutr. Biochem. 2022, 106, 109014. [Google Scholar] [CrossRef]
- Shindou, H.; Hishikawa, D.; Harayama, T.; Eto, M.; Shimizu, T. Generation of membrane diversity by lysophospholipid acyltransferases. J. Biochem. 2013, 154, 21–28. [Google Scholar] [CrossRef]
- Mouchlis, V.D.; Chen, Y.; McCammon, J.A.; Dennis, E.A. Membrane Allostery and Unique Hydrophobic Sites Promote Enzyme Substrate Specificity. J. Am. Chem. Soc. 2018, 140, 3285–3291. [Google Scholar] [CrossRef] [PubMed]
- Pauls, S.D.; Rodway, L.A.; Winter, T.; Taylor, C.G.; Zahradka, P.; Aukema, H.M. Alpha-linolenic acid enhances the phagocytic and secretory functions of alternatively activated macrophages in part via changes to the oxylipin profile. Int. J. Biochem. Cell Biol. 2020, 119, 105662. [Google Scholar] [CrossRef] [PubMed]
- Grechkin, A.N.; Kuramshin, R.A.; Safonova, E.Y.; Yefremov, Y.J.; Latypov, S.K.; Ilyasov, A.V.; Tarchevsky, I.A. Double hydroperoxidation of alpha-linolenic acid by potato tuber lipoxygenase. Biochim. Biophys. Acta 1991, 1081, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Sok, D.E.; Kim, M.R. Enzymatic formation of 9,16-dihydro(pero)xyoctadecatrienoic acid isomers from alpha-linolenic acid. Arch. Biochem. Biophys. 1990, 277, 86–93. [Google Scholar] [CrossRef]
- Liu, M.; Chen, P.; Véricel, E.; Lelli, M.; Béguin, L.; Lagarde, M.; Guichardant, M. Characterization and biological effects of di-hydroxylated compounds deriving from the lipoxygenation of ALA. J. Lipid Res. 2013, 54, 2083–2094. [Google Scholar] [CrossRef]
- Vliegenthart, J.F.; Feiters, M.C.; Veldink, G.A. Conjugated dihydroperoxyoctadecatrienoic fatty acids formed upon double dioxygenation of α-linolenic acid by lipoxygenase-2 from soybeans. In Oxidative Damage and Related Enzymes; Rotilio, G., Bannister, J.V., Eds.; Harwood Academic Publishers: Reading, UK, 1984; pp. 132–138. [Google Scholar]
- Vollenweider, S.; Weber, H.; Stolz, S.; Chételat, A.; Farmer, E.E. Fatty acid ketodienes and fatty acid ketotrienes: Michael addition acceptors that accumulate in wounded and diseased Arabidopsis leaves. Plant J. 2000, 24, 467–476. [Google Scholar] [CrossRef]
- Koch, T.; Hoskovec, M.; Boland, W. Efficient syntheses of (10E,12Z,15Z)-9-oxo- and (9Z,11E,15E)-13-oxo-octadecatrienoic acids; two stress metabolites of wounded plants. Tetrahedron 2002, 58, 3271–3274. [Google Scholar] [CrossRef]
- Laneuville, O.; Breuer, D.K.; Xu, N.; Huang, Z.H.; Gage, D.A.; Watson, J.T.; Lagarde, M.; DeWitt, D.L.; Smith, W.L. Fatty acid substrate specificities of human prostaglandin-endoperoxide H synthase-1 and -2. Formation of 12-hydroxy-(9Z, 13E/Z, 15Z)- octadecatrienoic acids from alpha-linolenic acid. J. Biol. Chem. 1995, 270, 19330–19336. [Google Scholar] [CrossRef]
- Laethem, R.M.; Balazy, M.; Koop, D.R. Epoxidation of C18 unsaturated fatty acids by cytochromes P4502C2 and P4502CAA. Drug Metab. Dispos. 1996, 24, 664–668. [Google Scholar]
- Schuchardt, J.P.; Schmidt, S.; Kressel, G.; Dong, H.; Willenberg, I.; Hammock, B.D.; Hahn, A.; Schebb, N.H. Comparison of free serum oxylipin concentrations in hyper- vs. normolipidemic men. Prostaglandins Leukot. Essent. Fat. Acids 2013, 89, 19–29. [Google Scholar] [CrossRef]
- Morisseau, C.; Hammock, B.D. Epoxide hydrolases: Mechanisms, inhibitor designs, and biological roles. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 311–333. [Google Scholar] [CrossRef] [PubMed]
- Morisseau, C.; Inceoglu, B.; Schmelzer, K.; Tsai, H.J.; Jinks, S.L.; Hegedus, C.M.; Hammock, B.D. Naturally occurring monoepoxides of eicosapentaenoic acid and docosahexaenoic acid are bioactive antihyperalgesic lipids. J. Lipid Res. 2010, 51, 3481–3490. [Google Scholar] [CrossRef] [PubMed]
- Konkel, A.; Schunck, W.H. Role of cytochrome P450 enzymes in the bioactivation of polyunsaturated fatty acids. Biochim. Biophys. Acta 2011, 1814, 210–222. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.S.; Helvig, C.; Taimi, M.; Ramshaw, H.A.; Collop, A.H.; Amad, M.; White, J.A.; Petkovich, M.; Jones, G.; Korczak, B. CYP2U1, a novel human thymus- and brain-specific cytochrome P450, catalyzes omega- and (omega-1)-hydroxylation of fatty acids. J. Biol. Chem. 2004, 279, 6305–6314. [Google Scholar] [CrossRef]
- Burdge, G.C.; Finnegan, Y.E.; Minihane, A.M.; Williams, C.M.; Wootton, S.A. Effect of altered dietary n-3 fatty acid intake upon plasma lipid fatty acid composition, conversion of [13C]alpha-linolenic acid to longer-chain fatty acids and partitioning towards beta-oxidation in older men. Br. J. Nutr. 2003, 90, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Pawlosky, R.J.; Hibbeln, J.R.; Lin, Y.; Goodson, S.; Riggs, P.; Sebring, N.; Brown, G.L.; Salem, N., Jr. Effects of beef- and fish-based diets on the kinetics of n-3 fatty acid metabolism in human subjects. Am. J. Clin. Nutr. 2003, 77, 565–572. [Google Scholar] [CrossRef]
- Schiessel, D.L.; Yamazaki, R.K.; Kryczyk, M.; Coelho de Castro, I.; Yamaguchi, A.A.; Pequito, D.C.; Brito, G.A.; Borghetti, G.; Aikawa, J.; Nunes, E.A.; et al. Does Oil Rich in Alpha-Linolenic Fatty Acid Cause the Same Immune Modulation as Fish Oil in Walker 256 Tumor-Bearing Rats? Nutr. Cancer 2016, 68, 1369–1380. [Google Scholar] [CrossRef]
- Ishihara, K.; Komatsu, W.; Saito, H.; Shinohara, K. Comparison of the effects of dietary alpha-linolenic, stearidonic, and eicosapentaenoic acids on production of inflammatory mediators in mice. Lipids 2002, 37, 481–486. [Google Scholar] [CrossRef]
- Ren, J.; Chung, S.H. Anti-inflammatory effect of alpha-linolenic acid and its mode of action through the inhibition of nitric oxide production and inducible nitric oxide synthase gene expression via NF-kappaB and mitogen-activated protein kinase pathways. J. Agric. Food Chem. 2007, 55, 5073–5080. [Google Scholar] [CrossRef]
- Zhao, G.; Etherton, T.D.; Martin, K.R.; Vanden Heuvel, J.P.; Gillies, P.J.; West, S.G.; Kris-Etherton, P.M. Anti-inflammatory effects of polyunsaturated fatty acids in THP-1 cells. Biochem. Biophys. Res. Commun. 2005, 336, 909–917. [Google Scholar] [CrossRef]
- Cao, J.; Schwichtenberg, K.A.; Hanson, N.Q.; Tsai, M.Y. Incorporation and clearance of omega-3 fatty acids in erythrocyte membranes and plasma phospholipids. Clin. Chem. 2006, 52, 2265–2272. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C. Metabolism of alpha-linolenic acid in humans. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Leyton, J.; Drury, P.J.; Crawford, M.A. Differential oxidation of saturated and unsaturated fatty acids in vivo in the rat. Br. J. Nutr. 1987, 57, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Koch, E.; Wiebel, M.; Löwen, A.; Willenberg, I.; Schebb, N.H. Characterization of the Oxylipin Pattern and Other Fatty Acid Oxidation Products in Freshly Pressed and Stored Plant Oils. J. Agric. Food Chem. 2022, 70, 12935–12945. [Google Scholar] [CrossRef] [PubMed]
- Caligiuri, S.P.; Love, K.; Winter, T.; Gauthier, J.; Taylor, C.G.; Blydt-Hansen, T.; Zahradka, P.; Aukema, H.M. Dietary linoleic acid and α-linolenic acid differentially affect renal oxylipins and phospholipid fatty acids in diet-induced obese rats. J. Nutr. 2013, 143, 1421–1431. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.; Winter, T.; Aukema, H.M. Dietary ALA, EPA and DHA have distinct effects on oxylipin profiles in female and male rat kidney, liver and serum. J. Nutr. Biochem. 2018, 57, 228–237. [Google Scholar] [CrossRef]
- Pauls, S.D.; Rodway, L.A.; Winter, T.; Taylor, C.G.; Zahradka, P.; Aukema, H.M. Anti-inflammatory effects of α-linolenic acid in M1-like macrophages are associated with enhanced production of oxylipins from α-linolenic and linoleic acid. J. Nutr. Biochem. 2018, 57, 121–129. [Google Scholar] [CrossRef]
- Kumar, N.; Gupta, G.; Anilkumar, K.; Fatima, N.; Karnati, R.; Reddy, G.V.; Giri, P.V.; Reddanna, P. 15-Lipoxygenase metabolites of α-linolenic acid, [13-(S)-HPOTrE and 13-(S)-HOTrE], mediate anti-inflammatory effects by inactivating NLRP3 inflammasome. Sci. Rep. 2016, 6, 31649. [Google Scholar] [CrossRef]
- Lucchinetti, E.; Lou, P.H.; Holtzhauer, G.; Noureddine, N.; Wawrzyniak, P.; Hartling, I.; Lee, M.; Strachan, E.; Clemente-Casares, X.; Tsai, S.; et al. Novel lipid emulsion for total parenteral nutrition based on 18-carbon n-3 fatty acids elicits a superior immunometabolic phenotype in a murine model compared with standard lipid emulsions. Am. J. Clin. Nutr. 2022, 116, 1805–1819. [Google Scholar] [CrossRef]
- Lucchinetti, E.; Lou, P.H.; Lemal, P.; Bestmann, L.; Hersberger, M.; Rogler, G.; Krämer, S.D.; Zaugg, M. Gut microbiome and circulating bacterial DNA (“blood microbiome”) in a mouse model of total parenteral nutrition: Evidence of two distinct separate microbiotic compartments. Clin. Nutr. ESPEN 2022, 49, 278–288. [Google Scholar] [CrossRef]
- Zahradka, P.; Neumann, S.; Aukema, H.M.; Taylor, C.G. Adipocyte lipid storage and adipokine production are modulated by lipoxygenase-derived oxylipins generated from 18-carbon fatty acids. Int. J. Biochem. Cell Biol. 2017, 88, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Itoh, T.; Fairall, L.; Amin, K.; Inaba, Y.; Szanto, A.; Balint, B.L.; Nagy, L.; Yamamoto, K.; Schwabe, J.W. Structural basis for the activation of PPARgamma by oxidized fatty acids. Nat. Struct. Mol. Biol. 2008, 15, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Umeno, A.; Sakashita, M.; Sugino, S.; Murotomi, K.; Okuzawa, T.; Morita, N.; Tomii, K.; Tsuchiya, Y.; Yamasaki, K.; Horie, M.; et al. Comprehensive analysis of PPARγ agonist activities of stereo-, regio-, and enantio-isomers of hydroxyoctadecadienoic acids. Biosci. Rep. 2020, 40, BSR20193767. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xu, Y.W.; Han, J.; Liang, H.; Wang, N.; Cheng, Y. 12/15-Lipoxygenase metabolites of arachidonic acid activate PPARγ: A possible neuroprotective effect in ischemic brain. J. Lipid Res. 2015, 56, 502–514. [Google Scholar] [CrossRef]
- Takahashi, H.; Hara, H.; Goto, T.; Kamakari, K.; Wataru, N.; Mohri, S.; Takahashi, N.; Suzuki, H.; Shibata, D.; Kawada, T. 13-Oxo-9(Z),11(E),15(Z)-octadecatrienoic acid activates peroxisome proliferator-activated receptor γ in adipocytes. Lipids 2015, 50, 3–12. [Google Scholar] [CrossRef]
- Balas, L.; Dey, S.K.; Béraud-Dufour, S.; Riechers, D.E.; Landau, O.A.; Bertrand-Michel, J.; Durand, T.; Blondeau, N. Linotrins: Omega-3 oxylipins featuring an E,Z,E conjugated triene motif are present in the plant kingdom and alleviate inflammation in LPS-challenged microglial cells. Eur. J. Med. Chem. 2022, 231, 114157. [Google Scholar] [CrossRef]
- Gabbs, M.; Zahradka, P.; Taylor, C.G.; Aukema, H.M. Time Course and Sex Effects of α-Linolenic Acid-Rich and DHA-Rich Supplements on Human Plasma Oxylipins: A Randomized Double-Blind Crossover Trial. J. Nutr. 2021, 151, 513–522. [Google Scholar] [CrossRef]
- Caligiuri, S.P.; Aukema, H.M.; Ravandi, A.; Guzman, R.; Dibrov, E.; Pierce, G.N. Flaxseed consumption reduces blood pressure in patients with hypertension by altering circulating oxylipins via an α-linolenic acid-induced inhibition of soluble epoxide hydrolase. Hypertension 2014, 64, 53–59. [Google Scholar] [CrossRef]
- Pauls, S.D.; Rodway, L.R.; Sidhu, K.K.; Winter, T.; Sidhu, N.; Aukema, H.M.; Zahradka, P.; Taylor, C.G. Oils Rich in α-Linolenic Acid or Docosahexaenoic Acid Have Distinct Effects on Plasma Oxylipin and Adiponectin Concentrations and on Monocyte Bioenergetics in Women with Obesity. J. Nutr. 2021, 151, 3053–3066. [Google Scholar] [CrossRef]
- Holt, R.R.; Yim, S.J.; Shearer, G.C.; Hackman, R.M.; Djurica, D.; Newman, J.W.; Shindel, A.W.; Keen, C.L. Effects of short-term walnut consumption on human microvascular function and its relationship to plasma epoxide content. J. Nutr. Biochem. 2015, 26, 1458–1466. [Google Scholar] [CrossRef]
| Study Population | Trials Included | Number of Participants | ALA Source | Dose | Duration | Effects | Reference |
|---|---|---|---|---|---|---|---|
| At least one of hyperlipidemia, type 2 diabetes, impaired glucose metabolism, hypertriglyceridemia, hypertension, metabolic syndrome (n = 10), healthy subjects (n = 3), unknown (n = 1) | 14 | 1107 | Flaxseed oil (n = 6) rapeseed oil (n = 1) linseed oil (n = 2) camelina sativa oil (n = 1) botanical oil (n = 1) | 1.9–10 g/d | 2–12+ Weeks | ↓ LDL | Chen, H. et al. [20] |
| Healthy (34%), type 2 diabetes (23.4%), dyslipidemia (23.4%), other (19.2%) | 47 | 2630 | Flaxseed (46%), walnut (17%), other (37%) | 0.4–16 g | 3–104 Weeks (mean 15 weeks) | ↓ TG, LDL-C, VLDL-C, TC/HDL-C ratio, LDL-C/HDL-C ratio | Yue, H. et al. [21] |
| Healthy (30%), dyslipidemia (19%), obese (15%), other (36%) | 25 | 2579 | Flaxseed oil/linseed (60%) other (40%) | 1–14 g/d | 4 Weeks-2 years | No CRP reduction | Su, H. et al. [22] |
| CKD | 19 | 1145 | Olive oil (47%), corn oil (21%), flaxseed oil (21%), other (11%) | 2–30 g | 4–48 Weeks | ↓ CRP | de Abreu, A.M. et al. [23] |
| Unspecified | 14 | 345,202 | Unspecified | 0.36–2.8 g/d * | 4–22 Years follow-up | ↓ Risk of composite CHD ↓ risk of fatal CHD | Wei, J. et al. [24] |
| CVD events | 27 | 251,049 | Unspecified | Unspecified | 5–30.7 Years | ↓ Fatal CHD | Pan, A. et al. [25] |
| Unspecified | 41 | 1,197,564 | Unspecified | 0.38–2.69 g/d | 2–32 Years follow-up | ↓ All-cause mortality, CVD mortality, CHD mortality ↑ cancer mortality | Naghshi, S. et al. [26] |
| Unspecified | 86 | 162,796 | Unspecified | Unspecified | 12–88 Months | ↓ CVD events ↓ arrhythmia | Abdelhamid, A.S. et al. [27] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cambiaggi, L.; Chakravarty, A.; Noureddine, N.; Hersberger, M. The Role of α-Linolenic Acid and Its Oxylipins in Human Cardiovascular Diseases. Int. J. Mol. Sci. 2023, 24, 6110. https://doi.org/10.3390/ijms24076110
Cambiaggi L, Chakravarty A, Noureddine N, Hersberger M. The Role of α-Linolenic Acid and Its Oxylipins in Human Cardiovascular Diseases. International Journal of Molecular Sciences. 2023; 24(7):6110. https://doi.org/10.3390/ijms24076110
Chicago/Turabian StyleCambiaggi, Lucia, Akash Chakravarty, Nazek Noureddine, and Martin Hersberger. 2023. "The Role of α-Linolenic Acid and Its Oxylipins in Human Cardiovascular Diseases" International Journal of Molecular Sciences 24, no. 7: 6110. https://doi.org/10.3390/ijms24076110
APA StyleCambiaggi, L., Chakravarty, A., Noureddine, N., & Hersberger, M. (2023). The Role of α-Linolenic Acid and Its Oxylipins in Human Cardiovascular Diseases. International Journal of Molecular Sciences, 24(7), 6110. https://doi.org/10.3390/ijms24076110






