Intestinal Carnitine Status and Fatty Acid Oxidation in Response to Clofibrate and Medium-Chain Triglyceride Supplementation in Newborn Pigs
Abstract
1. Introduction
2. Results
2.1. Carnitine Status
2.1.1. Plasma Carnitine
2.1.2. Liver Carnitine
2.1.3. Intestinal Carnitine
2.1.4. Distribution of Carnitine in Plasma and Tissues
2.2. Fatty Acid Oxidation
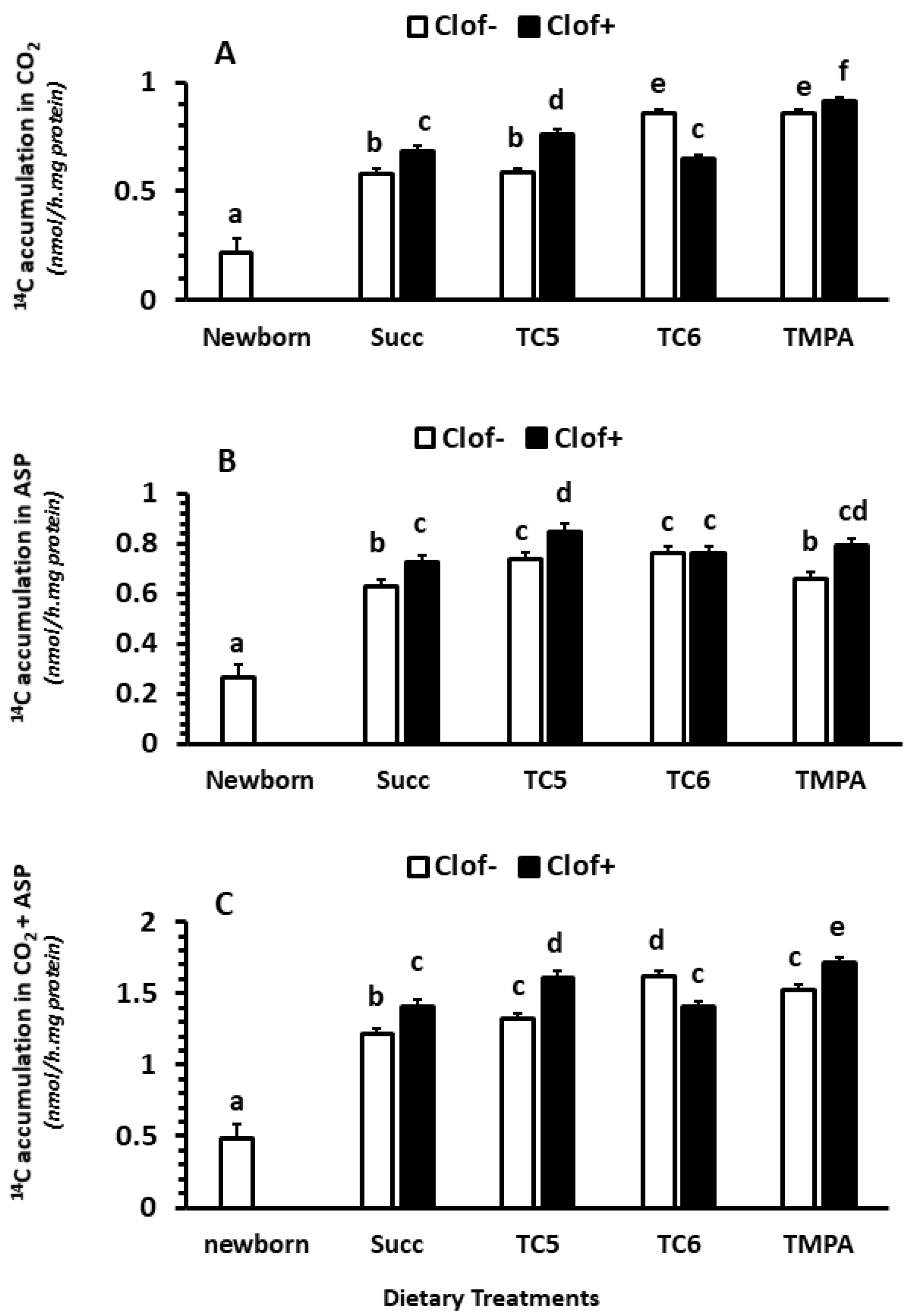
2.2.1. CO2 Production
2.2.2. Acid Soluble Products (ASP)
2.2.3. Total Oxidation (CO2 + ASP)
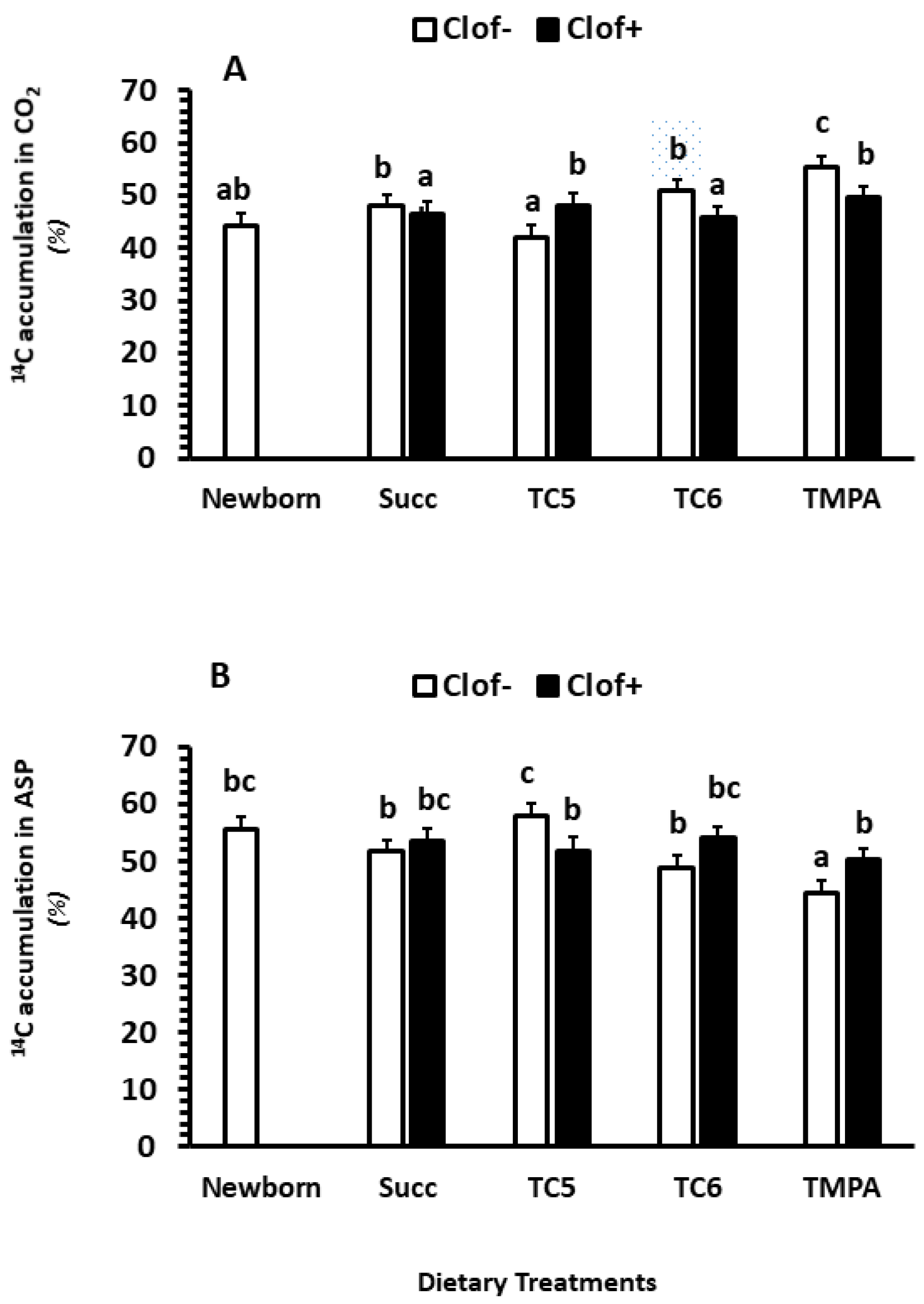
2.2.4. The Distribution between CO2 and ASP (% of Total Fatty Acid Oxidation)
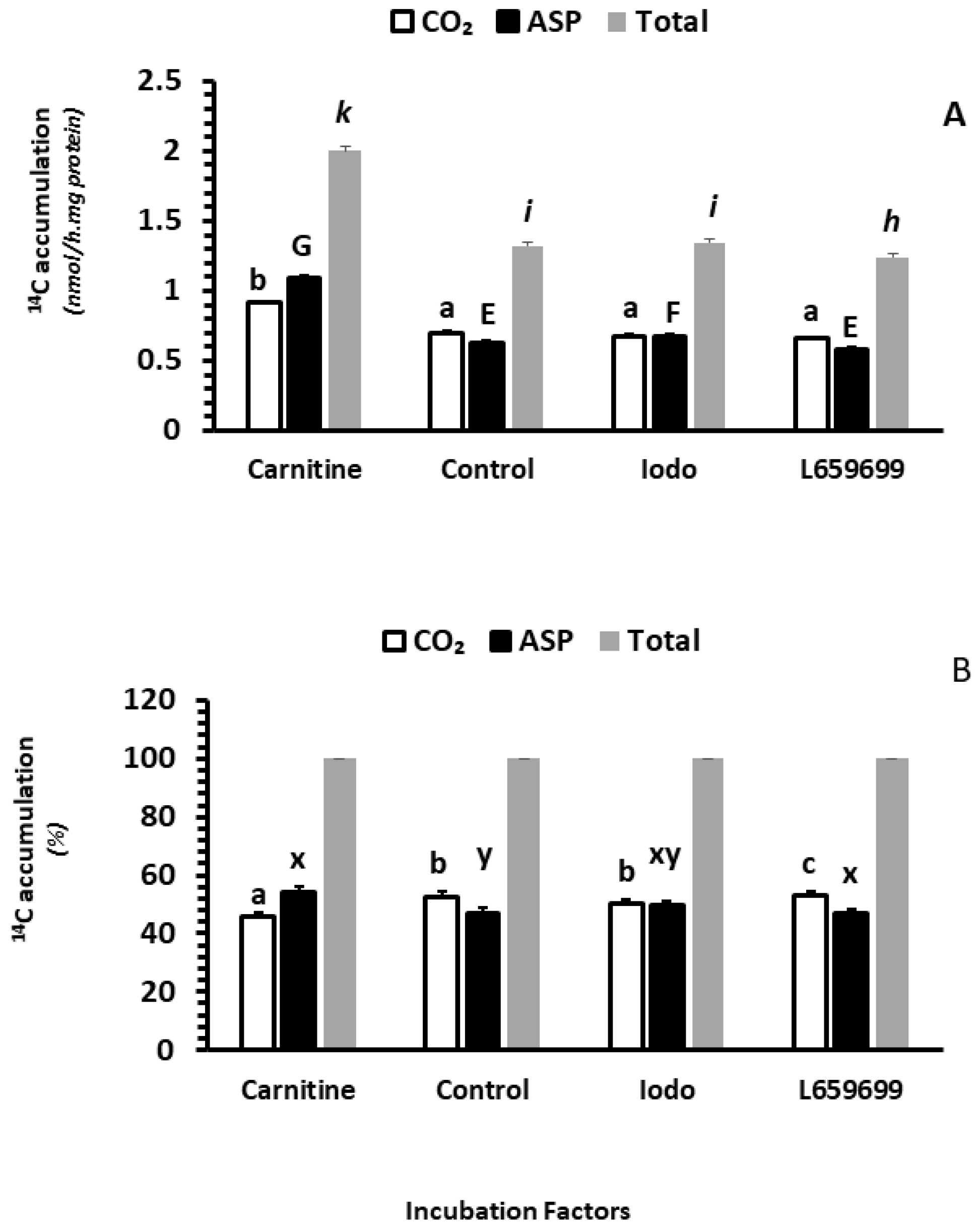
2.2.5. The Effects of Carnitine, Iodoacetamide and L659699 on Fatty Acid Oxidation In Vitro
2.3. Enzyme Activity
2.3.1. Carnitine Palmitoyltransferase
2.3.2. Citrate Synthase (CS)
2.4. Relative mRNA Abundance
2.4.1. Intestinal Mucosa
2.4.2. Liver
3. Discussion
3.1. The Effects of Clofibrate and Dietary Treatments (Succ, TC5, TC6 and TMPA) on Carnitine Status
3.2. The Effects of Clofibrate and Dietary Treatments (Succ, TC5, TC6 and TMPA) on Fatty Acid Oxidation in Intestine
3.3. The Effects of Carnitine and Ketogenic Pathway Inhibitors on Intestinal Fatty Acid Oxidation
4. Materials and Methods
4.1. Animal Care and Animal Protocol (Ethics Statement)
4.2. Triglyceride Synthesis
4.3. Sample Processing
4.4. Carnitine Analysis
4.5. Fatty Acid Oxidation
4.6. Enzyme Activity Assays
4.7. RNA Isolation and qPCR
4.8. Chemicals
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Longo, N.; Frigeni, M.; Pasquali, M. Carnitine transport and fatty acid oxidation. Biochim. Biophys. Acta. 2016, 1863, 2422–2435. [Google Scholar] [CrossRef] [PubMed]
- Baltzell, J.K.; Bazer, F.W.; Miguel, S.G.; Borum, P.R. The neonatal piglet as a model for human neonatal carnitine metabolism. J. Nutr. 1987, 117, 754–757. [Google Scholar] [CrossRef]
- Penn, D.; Bobrowski, P.J.; Zhang, L.; Schmidt-Sommerfeld, E. Neonatal nutritional carnitine deficiency: A piglet model. Pediatr. Res. 1997, 42, 114–121. [Google Scholar] [CrossRef][Green Version]
- Borum, P.R. Carnitine in neonatal nutrition. J. Child Neurol. 1995, 10, S25–S31. [Google Scholar] [CrossRef] [PubMed]
- Rebouche, C.J.; Lehman, L.J.; Olson, L. Epsilon-N-trimethyllysine availability regulates the rate of carnitine biosynthe-sis in the growing rat. J. Nutr. 1986, 116, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Ringseis, R.; Wen, G.; Eder, K. Regulation of Genes Involved in Carnitine Homeostasis by PPARα across Different Species (Rat, Mouse, Pig, Cattle, Chicken, and Human). PPAR Res. 2012, 2012, 868317. [Google Scholar] [CrossRef]
- Shekhawat, P.S.; Sonne, S.; Carter, A.L.; Matern, D.; Ganapathy, V. Enzymes involved in L-carnitine biosynthesis are expressed by small intestinal enterocytes in mice: Implications for gut health. J. Crohn’s Colitis 2013, 7, e197–e205. [Google Scholar] [CrossRef] [PubMed]
- Broderick, T.L.; Cusimano, F.A.; Carlson, C.; Babu, J.R. Biosynthesis of the Essential Fatty Acid Oxidation Cofactor Carnitine Is Stimulated in Heart and Liver after a Single Bout of Exercise in Mice. J. Nutr. Metab. 2018, 2018, 2785090. [Google Scholar] [CrossRef]
- Marciani, P.; Lindi, C.; Marzo, A.; Arrigoni Martelli, E.; Cardace, G.; Esposito, G. L-carnitine and carnitine ester transport in the rat small intestine. Pharmacol. Res. 1991, 23, 157–162. [Google Scholar] [CrossRef]
- Rebouche, C.J. Kinetics, Pharmacokinetics, and Regulation of L-Carnitine and Acetyl-L-carnitine Metabolism. Ann. N.Y. Acad. Sci. 2004, 1033, 30–41. [Google Scholar] [CrossRef]
- García-Delgado, M.; Peral, M.J.; Durán, J.M.; García-Miranda, P.; Calonge, M.L.; Ilundáin, A.A. Ontogeny of Na(+)/L-carnitine transporter and of gamma-trimethylaminobutyraldehyde dehydrogenase and gamma-butyrobetaine hydroxylase genes expression in rat kidney. Mech. Ageing Dev. 2009, 130, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Kato, Y.; Sugiura, M.; Sugiura, T.; Wakayama, T.; Kubo, Y.; Kobayashi, D.; Sai, Y.; Tamai, I.; Iseki, S.; Tsuji, A. Organic cation/carnitine transporter OCTN2 (Slc22a5) is responsible for carnitine transport across apical membranes of small intestinal epithelial cells in mouse. Mol. Pharmacol. 2006, 70, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Durán, J.M.; Peral, M.J.; Calonge, M.L.; Ilundáin, A.A. Functional characterization of intestinal L-carnitine transport. J. Membr. Biol. 2002, 185, 65–74. [Google Scholar] [CrossRef]
- Ringseis, R.; Eder, K. Influence of pharmacological PPARalpha activators on carnitine homeostasis in proliferat-ing and non-proliferating species. Pharmacol. Res. 2009, 60, 179–184. [Google Scholar] [CrossRef]
- Wen, G.; Kühne, H.; Rauer, C.; Ringseis, R.; Eder, K. Mouse γ-butyrobetaine dioxygenase is regulated by peroxi-some proliferator-activated receptor α through a PPRE located in the proximal promoter. Biochem. Pharmacol. 2011, 82, 175–183. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wen, G.; Ringseis, R.; Rauer, C.; Eder, K. The mouse gene encoding the carnitine biosynthetic enzyme 4-N-trimethylaminobutyraldehyde dehydrogenase is regulated by peroxisome proliferator-activated receptor α. Biochim. Biophys. Acta Gene Regul. 2012, 1819, 357–365. [Google Scholar] [CrossRef]
- Yu, X.X.; Odle, J.; Drackley, J.K. Differential induction of peroxisomal beta-oxidation enzymes by clofibric acid and as-pirin in piglet tissues. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 281, R1553–R1561. [Google Scholar] [CrossRef]
- Bai, X.; Lin, X.; Drayton, J.; Liu, Y.; Ji, C.; Odle, J. Clofibrate increases long-chain fatty acid oxidation by neona-tal pigs. J. Nutr. 2014, 144, 1688–1693. [Google Scholar] [CrossRef]
- Lin, X.; Jacobi, S.; Odle, J. Transplacental induction of fatty acid oxidation in term fetal pigs by the peroxisome proliferator-activated receptor alpha agonist clofibrate. J. Anim. Sci. Biotechnol. 2015, 6, 11. [Google Scholar] [CrossRef]
- He, Y.; Khan, I.; Bai, X.; Odle, J.; Xi, L. Activation of PPARα by Oral Clofibrate Increases Renal Fatty Acid Oxidation in Developing Pigs. Int. J. Mol. Sci. 2017, 18, 2663. [Google Scholar] [CrossRef]
- Shim, K.; Jacobi, S.; Odle, J.; Lin, X. Pharmacologic activation of peroxisome proliferator-activating receptor-α acceler-ates hepatic fatty acid oxidation in neonatal pigs. Oncotarget 2018, 9, 23900–23914. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lin, X.; Pike, B.; Zhao, J.; Fan, Y.; Zhu, Y.; Zhang, Y.; Wang, F.; Odle, J. Effects of Dietary Anaplerotic and Ketogenic En-ergy Sources on Renal Fatty Acid Oxidation Induced by Clofibrate in Suckling Neonatal Pigs. Int. J. Mol. Sci. 2020, 21, 726. [Google Scholar] [CrossRef] [PubMed]
- Veerkamp, J.H.; van Moerkerk, H.T. Peroxisomal fatty acid oxidation in rat and human tissues. Effect of nutri-tional state, clofibrate treatment and postnatal development in the rat. Biochim. Biophys. Acta 1986, 875, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Grum, D.E.; Hansen, L.R.; Drackley, J.K. Peroxisomal beta-oxidation of fatty acids in bovine and rat liver. Comp. Biochem. Physiol. B. Biochem. Mol. Biol. 1994, 109, 281–292. [Google Scholar] [CrossRef]
- Venkatachalam, A.B.; Lall, S.P.; Denovan-Wright, E.M.; Wright, J.M. Tissue-specific differential induction of duplicated fatty acid-binding protein genes by the peroxisome proliferator, clofibrate, in zebrafish (Danio rerio). BMC Evol. Biol. 2012, 12, 112. [Google Scholar] [CrossRef]
- Luo, H.; Zhang, Y.; Guo, H.; Zhang, L.; Li, X.; Ringseis, R.; Wen, G.; Hui, D.; Liang, A.; Eder, K.; et al. Transcrip-tional regulation of the human, porcine and bovine OCTN2 gene by PPARα via a conserved PPRE located in intron 1. BMC Genet. 2014, 15, 90. [Google Scholar] [CrossRef]
- Peffer, P.L.; Lin, X.; Jacobi, S.K.; Gatlin, L.A.; Woodworth, J.; Odle, J. Ontogeny of carnitine palmitoyltransferase I activity, carnitine-Km, and mRNA abundance in pigs throughout growth and development. J. Nutr. 2007, 137, 898–903. [Google Scholar] [CrossRef]
- Ringseis, R.; Wege, N.; Wen, G.; Rauer, C.; Hirche, F.; Kluge, H.; Eder, K. Carnitine synthesis and uptake into cells are stimulated by fasting in pigs as a model of nonproliferating species. J. Nutr. Biochem. 2009, 20, 840–847. [Google Scholar] [CrossRef]
- Koch, A.; König, B.; Luci, S.; Stangl, G.I.; Eder, K. Dietary oxidised fat up regulates the expression of organic cation transporters in liver and small intestine and alters carnitine concentrations in liver, muscle and plasma of rats. Br. J. Nutr. 2007, 98, 882–889. [Google Scholar] [CrossRef]
- Bhuiyan, A.K.; Jackson, S.; Turnbull, D.M.; Aynsley-Green, A.; Leonard, J.V.; Bartlett, K. The measurement of carnitine and acyl-carnitines: Application to the investigation of patients with suspected inherited disorders of mitochondrial fatty acid oxidation. Clin. Chim. Acta 1992, 207, 185–204. [Google Scholar] [CrossRef]
- Lin, X.; House, R.; Odle, J. Ontogeny and kinetics of carnitine palmitoyltransferase in liver and skeletal muscle of the domestic felid (Felis domestica). J. Nutr. Biochem. 2005, 16, 331–338. [Google Scholar] [CrossRef]
- Lin, X.; Odle, J. Changes in Kinetics of Carnitine Palmitoyltransferase in Liver and Skeletal Muscle of Dogs (Canis familiaris) throughout Growth and Development. J. Nutr. 2003, 133, 1113–1119. [Google Scholar] [CrossRef]
- Xi, L.; Brown, K.; Woodworth, J.; Shim, K.; Johnson, B.; Odle, J. Maternal dietary L-carnitine supplementation influences fetal carnitine status and stimulates carnitine palmitoyltransferase and pyruvate dehydrogenase complex activities in swine. J. Nutr. 2008, 138, 2356–2362. [Google Scholar] [CrossRef]
- Fischer, M.; Keller, J.; Hirche, F.; Kluge, H.; Ringseis, R.; Eder, K. Activities of gamma-butyrobetaine dioxygenase and concentrations of carnitine in tissues of pigs. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 2009, 153, 324–331. [Google Scholar] [CrossRef]
- Fischer, M.; Varady, J.; Hirche, F.; Kluge, H.; Eder, K. Supplementation of L-carnitine in pigs: Absorption of carnitine and effect on plasma and tissue carnitine concentrations. Arch. Anim. Nutr. 2009, 63, 1–15. [Google Scholar] [CrossRef]
- Borum, P.R.; York, C.M.; Bennett, S.G. Carnitine concentration of red blood cells. Am. J. Clin. Nutr. 1982, 41, 653–656. [Google Scholar] [CrossRef] [PubMed]
- Birkenfeld, C.; Doberenz, J.; Kluge, H.; Eder, K. Effect of l-carnitine supplementation of sows on l-carnitine status, body composition and concentrations of lipids in liver and plasma of their piglets at birth and during the suckling period. Anim. Feed Sci. Technol. 2006, 129, 23–38. [Google Scholar] [CrossRef]
- Gross, C.J.; Savaiano, D.A. Effect of development and nutritional state on the uptake, metabolism and release of free and acetyl-l-carnitine by the rodent small intestine. Biochim. Biophys. Acta (BBA)/Lipids Lipid Metab. 1993, 1170, 265–274. [Google Scholar] [CrossRef]
- Brown, K.R.; Goodband, R.D.; Tokach, M.D.; Dritz, S.S.; Nelssen, J.L.; Minton, J.E.; Higgins, J.J.; Lin, X.; Odle, J.; Wood-worth, J.C.; et al. Effects of feeding L-carnitine to gilts through day 70 of gestation on litter traits and the ex-pression of insulin-like growth factor system components and L-carnitine concentration in foetal tissues. J. Anim. Physiol. Anim. Nutr. 2008, 92, 660–667. [Google Scholar] [CrossRef]
- Paul, H.S.; Adibi, S.A. Paradoxical effects of clofibrate on liver and muscle metabolism in rats. Induction of myotonia and alteration of fatty acid and glucose oxidation. J. Clin. Investig. 1979, 64, 405–412. [Google Scholar] [CrossRef]
- Shibani, M.; Keller, J.; König, B.; Kluge, H.; Hirche, F.; Stangl, G.I.; Ringseis, R.; Eder, K. Effects of fish oil and conjugated linoleic acids on carnitine homeostasis in laying hens. Br. Poult. Sci. 2012, 53, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Paul, H.S.; Gleditsch, C.E.; Adibi, S.A. Mechanism of increased hepatic concentration of carnitine by clofibrate. Am. J. Physiol. 1986, 251, E311–E315. [Google Scholar] [CrossRef] [PubMed]
- Pande, S.V.; Parvin, R. Clofibrate enhancement of mitochondrial carnitine transport system of rat liver and augmen-tation of liver carnitine and gamma-butyrobetaine hydroxylase activity by thyroxine. Biochim. Biophys. Acta 1980, 21, 363–370. [Google Scholar] [CrossRef]
- Lin, X.; Lyvers Peffer, P.A.; Woodworth, J.; Odle, J. Ontogeny of carnitine biosynthesis in Sus scrofa domesticus, in-ferred from γ-butyrobetaine hydroxylase (dioxygenase) activity and substrate inhibition. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020, 319, R43–R49. [Google Scholar] [CrossRef] [PubMed]
- Van Vlies, N.; Ferdinandusse, S.; Turkenburg, M.; Wanders, R.J.A.; Vaz, F.M. PPARα-activation results in enhanced car-nitine biosynthesis and OCTN2-mediated hepatic carnitine accumulation. Biochim. Biophys. Acta Bioenerg. 2007, 1767, 1134–1142. [Google Scholar] [CrossRef]
- Ringseis, R.; Pösel, S.; Hirche, F.; Eder, K. Treatment with pharmacological peroxisome proliferator-activated receptor alpha agonist clofibrate causes upregulation of organic cation transporter 2 in liver and small intestine of rats. Pharmacol Res. 2007, 56, 175–183. [Google Scholar] [CrossRef]
- Luci, S.; Hirche, F.; Eder, K. Fasting and caloric restriction increases mRNA concentrations of novel organic cation transporter-2 and carnitine concentrations in rat tissues. Ann. Nutr. Metab. 2008, 52, 58–67. [Google Scholar] [CrossRef]
- Eder, K.; Ringseis, R. The role of peroxisome proliferator-activated receptor alpha in transcriptional regulation of nov-el organic cation transporters. Eur. J. Pharmacol. 2010, 628, 1–5. [Google Scholar] [CrossRef]
- Wen, G.; Ringseis, R.; Eder, K. Mouse OCTN2 is directly regulated by peroxisome proliferator-activated receptor alpha (PPARalpha) via a PPRE located in the first intron. Biochem. Pharmacol. 2010, 79, 768–776. [Google Scholar] [CrossRef]
- Luci, S.; Geissler, S.; König, B.; Koch, A.; Stangl, G.I.; Hirche, F.; Eder, K. PPARα agonists up-regulate organic cation transporters in rat liver cells. Biochem. Biophys. Res. Commun. 2006, 350, 704–708. [Google Scholar] [CrossRef]
- Ringseis, R.; Luci, S.; Spielmann, J.; Kluge, H.; Fischer, M.; Geissler, S.; Wen, G.; Hirche, F.; Eder, K. Clofibrate treatment up-regulates novel organic cation transporter (OCTN)-2 in tissues of pigs as a model of non-proliferating species. Eur. J. Pharmacol. 2008, 583, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Ringseis, R.; Keller, J.; Eder, K. Basic mechanisms of the regulation of L-carnitine status in monogastrics and efficacy of L-carnitine as a feed additive in pigs and poultry. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1686–1719. [Google Scholar] [CrossRef] [PubMed]
- Gudjonsson, H.; Li, B.U.; Shug, A.l.; Olsen, W.A. In vivo studies of intestinal carnitine absorption in rats. Gastroenterology 1985, 88, 1880–1887. [Google Scholar] [CrossRef] [PubMed]
- Cederblad, G. Effect of diet on plasma carnitine levels and urinary carnitine excretion in humans. Am. J. Clin. Nutr. 1987, 45, 725–729. [Google Scholar] [CrossRef]
- Stadler, D.D.; Chenard, C.A.; Rebouche, C.J. Effect of dietary macronutrient content on carnitine excretion and efficien-cy of carnitine reabsorption. Am. J. Clin. Nutr. 1993, 58, 868–872. [Google Scholar] [CrossRef]
- Peffer, P.L.; Lin, X.; Odle, J. Hepatic beta-oxidation and carnitine palmitoyltransferase I in neonatal pigs after dietary treatments of clofibric acid, isoproterenol, and medium-chain triglycerides. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R1518–R1524. [Google Scholar] [CrossRef]
- Zhao, J.; Pike, B.; Huang, J.; Feng, Z.; Odle, J.; Lin, X. Effects of medium chain triglycerides on hepatic fatty acid oxida-tion in clofibrate-fed newborn piglets. Anim. Nutr. 2022, 12, 334–344. [Google Scholar] [CrossRef]
- Asins, G.; Serra, D.; Arias, G.; Hegardt, F.G. Developmental changes in carnitine palmitoyltransferases I and II gene expression in intestine and liver of suckling rats. Biochem. J. 1995, 306, 379–384. [Google Scholar] [CrossRef]
- Azari, E.K.; Leitner, C.; Jaggi, T.; Langhans, W.; Mansouri, A. Possible role of intestinal fatty acid oxidation in the eating-inhibitory effect of the PPAR-α agonist Wy-14643 in high-fat diet fed rats. PLoS ONE 2013, 8, e74869. [Google Scholar]
- Poirier, H.; Degrace, P.; Niot, I.; Bernard, A.; Besnard, P. Localization and regulation of the putative membrane fatty-acid transporter (FAT) in the small intestine Comparison with fatty acid-binding proteins (FABP). Eur. J. Biochem. 1996, 238, 368–373. [Google Scholar] [CrossRef]
- Storch, J.; Thumser, A.E. Tissue-specific functions in the fatty acid-binding protein family. J. Biol. Chem. 2010, 285, 32679–32683. [Google Scholar] [CrossRef] [PubMed]
- Wolfrum, C. Cytoplasmic fatty acid binding protein sensing fatty acids for peroxisome proliferator activated receptor activation. Cell Mol. Life Sci. 2007, 64, 2465–2476. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, A.L.; Atshaves, B.P.; Hostetler, H.A.; Huang, H.; Davis, J.; Lyuksyutova, O.I.; Landrock, D.; Kier, A.B.; Schroeder, F. Liver type fatty acid binding protein (L-FABP) gene ablation reduces nuclear ligand distribution and pe-roxisome proliferator-activated receptor-alpha activity in cultured primary hepatocytes. Arch. Biochem. Biophys. 2009, 485, 160–173. [Google Scholar] [CrossRef]
- MacDonald, M.J.; Fahien, F.A.; Mertz, R.J.; Rana, R.S. Effect of esters of succinic acid and other citric acid cycle interme-diates on insulin release and inositol phosphate formation by pancreatic islets. Arch. Biochem. Biophys. 1989, 269, 400–406. [Google Scholar] [CrossRef]
- Ehinger, J.K.; Piel, S.; Ford, R.; Karlsson, M.; Sjövall, F.; Frostner, E.A.; Morota, S.; Taylor, R.W.; Turnbull, D.M.; Cornell, C.; et al. Cell-permeable succinate prodrugs bypass mitochondrial complex I deficiency. Nat. Commun. 2016, 7, 12317. [Google Scholar] [CrossRef]
- Tretter, L.; Patocs, A.; Chinopoulos, C. Succinate, an intermediate in metabolism, signal transduction, ROS, hy-poxia, and tumorigenesis. Biochim. Biophys. Acta Bioenerg. 2016, 1857, 1086–1101. [Google Scholar] [CrossRef]
- Koldovský, O.; Dobiásová, M.; Drahota, Z.; Hahn, P. Developmental aspects of lipid metabolism. Physiol. Res. 1995, 44, 353–356. [Google Scholar] [PubMed]
- Wolfe, R.G.; Maxwell, C.V.; Nelson, E.C. Effect of Age and Dietary Fat Level on Fatty Acid Oxidation in the Neo-natal Pig. J. Nutr. 1978, 108, 1621–1634. [Google Scholar] [CrossRef]
- Coffey, M.T.; Shireman, R.B.; Herman, D.L.; Jones, E.E. Carnitine status and lipid utilization in neonatal piglets fed diets low in carnitine. J. Nutr. 1991, 121, 1047–1053. [Google Scholar] [CrossRef]
- van Kempen, T.A.T.G.; Odle, J. Medium-Chain Fatty Acid Oxidation in Colostrum-Deprived Newborn Piglets: Stimulative Effect of L-Carnitine Supplementation. J. Nutr. 1993, 123, 1531–1537. [Google Scholar] [CrossRef] [PubMed]
- Odle, J.; Lin, X.; van Kempen, T.A.; Drackley, J.K.; Adams, S.H. Carnitine low ketogenesis in neonatal pigs. J. Nutr. 1995, 125, 2541–2549. [Google Scholar] [PubMed]
- Heo, K.N.; Lin, X.; Han, I.K.; Odle, J. Medium-chain fatty acids but not L-carnitine accelerate the kinetics of [14C]triacylglycerol utilization by colostrum-deprived newborn pigs. J. Nutr. 2002, 132, 1989–1994. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Shim, K.; Odle, J. Carnitine palmitoyltransferase I control of acetogenesis, the major pathway of fatty acid {beta}-oxidation in liver of neonatal swine. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1435–R1443. [Google Scholar] [CrossRef]
- Williamson, D.H.; Bates, M.W.; Krebs, H.A. Activity and intracellular distribution of enzymes of ketone-body metabolism in rat liver. Biochem. J. 1968, 108, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Greenspan, M.D.; Yudkovitz, J.B.; Lo, C.L.; Chen, J.S.; Alberts, A.W.; Hunt, V.W.; Chang, M.N.; Yang, S.S.; Thompson, K.L.; Chiang, Y.P.; et al. Inhibition of hydroxymethylglutaryl-coenzyme A synthase by L-659,699. Proc. Natl. Acad. Sci. USA 1987, 84, 7488–7492. [Google Scholar] [CrossRef]
- Hahn, P.; Taller, M. Ketone formation in the intestinal mucosa of infant rats. Life Sci. 1987, 41, 1525–1528. [Google Scholar] [CrossRef]
- Békési, A.; Williamson, D.H. An explanation for ketogenesis by the intestine of the suckling rat: The presence of an active hydroxymethylglutaryl-coenzyme A pathway. Biol. Neonat. 1990, 58, 160–165. [Google Scholar] [CrossRef]
- Vilà-Brau, A.; De Sousa-Coelho, A.L.; Mayordomo, C.; Haro, D.; Marrero, P.F. Human HMGCS2 regulates mito-chondrial fatty acid oxidation and FGF21 expression in HepG2 cell line. J. Biol. Chem. 2011, 286, 20423–20430. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhou, Y.; Rychahou, P.; Fan, T.W.; Lane, A.N.; Weiss, H.L.; Evers, B.M. Ketogenesis contributes to intes-tinal cell differentiation. Cell Death Differ. 2017, 24, 458–468. [Google Scholar] [CrossRef]
- Adams, S.H.; Alho, C.S.; Asins, G.; Hegardt, F.G.; Marrero, P.F. Gene expression of mitochondrial 3-hydroxy-3-methylglutaryl-CoA synthase in a poorly ketogenic mammal: Effect of starvation during the neonatal pe-riod of the piglet. Biochem. J. 1997, 324, 65–73. [Google Scholar] [CrossRef]
- McGarry, J.D.; Foster, D.W. An improved and simplified radioisotopic assay for the determination of free and esterified carnitine. J. Lipid Res. 1976, 17, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Gornall, A.G.; Bardawill, C.J.; David, M.M. Determination of serum proteins by means of the biuret reaction. J. Biol. Chem. 1949, 177, 751–766. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
| PPAR Activator | MCT | PPAR × CT | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| −Clof | +Clof | SEM | p-Value | Succ | TC5 | TC6 | TMPA | SEM | p-Value | p-Value | |
| nmol/µL | nmol/µL | ||||||||||
| Free Carnitine | 0.078 | 0.090 | 0.009 | 0.362 | 0.075 | 0.091 | 0.096 | 0.075 | 0.012 | 0.497 | 0.847 |
| Acyl-Carnitine | 0.130 a | 0.153 b | 0.008 | 0.049 | 0.136 | 0.142 | 0.163 | 0.125 | 0.011 | 0.111 | 0.696 |
| Total Carnitine | 0.208 | 0.243 | 0.016 | 0.132 | 0.210 | 0.232 | 0.259 | 0.201 | 0.022 | 0.243 | 0.934 |
| % | % | ||||||||||
| Free Carnitine | 38.0 | 34.9 | 1.29 | 0.064 | 36.8 | 38.7 | 34.9 | 35.6 | 1.57 | 0.387 | 0.259 |
| Acyl-Carnitine | 62.0 | 65.1 | 1.29 | 0.064 | 63.2 | 61.3 | 65.1 | 64.4 | 1.57 | 0.387 | 0.259 |
| PPAR Activator | MCT | PPAR × MCT | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| −Clof | +Clof | SEM | p-Value | Succ | TC5 | TC6 | TMPA | SEM | p-Value | p-Value | |
| nmol/mg fresh tissue | nmol/mg fresh tissue | ||||||||||
| Free Carnitine | 0.211 a | 0.360 b | 0.015 | 0.001 | 0.269 | 0.275 | 0.309 | 0.287 | 0.020 | 0.552 | 0.841 |
| Acyl-Carnitine | 0.181 a | 0.323 b | 0.016 | 0.001 | 0.231 | 0.269 | 0.247 | 0.261 | 0.022 | 0.659 | 0.284 |
| Total Carnitine | 0.392 a | 0.683 b | 0.026 | 0.001 | 0.501 | 0.545 | 0.557 | 0.548 | 0.037 | 0.737 | 0.467 |
| % | % | ||||||||||
| Free Carnitine | 51.6 | 51.9 | 1.30 | 0.877 | 52.8 | 49.1 | 52.5 | 52.6 | 1.86 | 0.483 | 0.619 |
| Acyl-Carnitine | 48.4 | 48.1 | 1.30 | 0.877 | 47.2 | 50.9 | 47.5 | 47.3 | 1.86 | 0.483 | 0.619 |
| PPAR Activator | MCT | PPAR × MCT | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| −Clof | +Clof | SEM | p-Value | Succ | TC5 | TC6 | TMPA | SEM | p-Value | p-Value | |
| nmol/mg fresh tissue | nmol/mg fresh tissue | ||||||||||
| Free Carnitine | 0.291 | 0.323 | 0.015 | 0.134 | 0.296 | 0.317 | 0.301 | 0.313 | 0.020 | 0.878 | 0.351 |
| Acyl-Carnitine | 0.566 | 0.613 | 0.045 | 0.473 | 0.552 | 0.572 | 0.720 | 0.515 | 0.064 | 0.659 | 0.284 |
| Total Carnitine | 0.857 | 0.936 | 0.051 | 0.287 | 0.848 | 0.889 | 1.021 | 0.828 | 0.072 | 0.107 | 0.593 |
| % | % | ||||||||||
| Free Carnitine | 36.6 | 36.9 | 1.97 | 0.921 | 36.7 | 39.0 | 32.1 | 39.2 | 2.85 | 0.227 | 0.756 |
| Acyl-Carnitine | 63.4 | 63.1 | 1.97 | 0.901 | 63.3 | 61.0 | 67.9 | 60.8 | 2.85 | 0.227 | 0.756 |
| Clof− | Clof+ | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Plasma | Liver | Intestine | Plasma | Liver | Intestine | SEM | p-Value | ||
| nmol/mg fresh tissue | |||||||||
| Free carnitine | 0.078 a | 0.210 b | 0.292 c | 0.090 a | 0.358 d | 0.328 cd | 0.0196 | 0.001 | |
| Acyl-carnitine | 0.128 a | 0.179 a | 0.572 c | 0.151 a | 0.318 b | 0.614 c | 0.0381 | 0.258 | |
| Total carnitine | 0.207 a | 0.389 b | 0.865 d | 0.241 a | 0.676 c | 0.942 d | 0.0488 | 0.022 | |
| % | |||||||||
| Free carnitine | 38.21 a | 51.67 b | 36.50 a | 35.54 a | 52.12 b | 37.31 a | 2.617 | 0.783 | |
| Acyl-carnitine | 61.79 b | 48.33 a | 63.50 b | 64.46 b | 47.88 a | 62.69 b | 2.617 | 0.783 | |
| PPAR Activator | MCT | PPAR × MCT | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| −Clof | +Clof | SEM | p-Value | Succ | TC5 | TC6 | TMPA | SEM | p-Value | p-Value | |
| nmol/mg protein | nmol/mg protein | ||||||||||
| CPT I | 2.064 a | 2.660 b | 0.094 | 0.0001 | 2.418 | 2.368 | 2.199 | 2.462 | 0.135 | 0.503 | 0.626 |
| CPT II | 1.140 a | 1.335 b | 0.052 | 0.0120 | 1.168 | 1.310 | 1.234 | 1.234 | 0.074 | 0.629 | 0.718 |
| Total CPT | 3.204 a | 3.994 b | 0.105 | 0.0001 | 3.585 | 3.678 | 3.433 | 3.700 | 0.141 | 0.550 | 0.397 |
| CS | 1.705 | 1.568 | 0.617 | 0.128 | 1.751 | 1.621 | 1.648 | 1.525 | 0.088 | 0.340 | 0.490 |
| Total protein | 6.403 | 6.412 | 0.112 | 0.953 | 6.417 | 6.166 | 6.454 | 6.594 | 0.160 | 0.319 | 0.296 |
| NB | PPAR Activator | MCT | PPAR × MCT | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NB | −Clof | +Clof | SEM | p-Value | Succ | TC5 | TC6 | TMPA | SEM | p-Value | p-Value | |
| Intestine | Fold | Fold | ||||||||||
| ACOX | 1 | 1.52 | 1.52 | 0.086 | 0.318 | 1.50 | 1.62 | 1.64 | 1.59 | 0.121 | 0.861 | 0.259 |
| CPT Ia | 1 | 0.804 | 0.804 | 0.094 | 0.978 | 0.854 | 0.846 | 0.801 | 0.723 | 0.125 | 0.896 | 0.782 |
| CPT Ib | 1 | 0.229 | 0.229 | 0.020 | 0.161 | 0.200 | 0.256 | 0.198 | 0.184 | 0.028 | 0.316 | 0.821 |
| BBOX | 1 | 1.51 | 1.51 | 0.144 | 0.178 | 1.11 | 1.31 | 1. 44 | 1.62 | 0.205 | 0.354 | 0.845 |
| FABP | 1 | 0.623 | 0.623 | 0.056 | 0.944 | 0.438 a | 0.805 b | 0.623 ab | 0.629 ab | 0.080 | 0.029 | 0.425 |
| OCTN1 | 1 | 1.363 b | 1.363 b | 0.133 | 0.045 | 1.331 | 1.099 | 1.030 | 1.211 | 0.187 | 0.689 | 0.730 |
| OCTN2 | 1 | 0.686 | 0.686 | 0.095 | 0.308 | 0.538 | 0.657 | 0.616 | 0.657 | 0.135 | 0.918 | 0.222 |
| PPARα | 1 | 1.074 | 1.074 | 0.159 | 0.058 | 0.804 | 0.894 | 1.010 | 0.714 | 0.224 | 0.795 | 0.846 |
| TMLHE | 1 | 0.761 | 0.761 | 0.135 | 0.225 | 0.637 | 0.659 | 0.700 | 0.579 | 0.191 | 0.973 | 0.933 |
| ALDH9A1 | 1 | 1.142 | 1.142 | 0.119 | 0.339 | 1.133 | 0.935 | 1.237 | 0.938 | 0.170 | 0.489 | 0.609 |
| Liver | ||||||||||||
| ALDH9A1 | 1 | 1.68 a | 3.54 b | 0.39 | 0.003 | 2.27 | 2.03 | 3.04 | 3.11 | 0.550 | 0.430 | 0.606 |
| BBOX | 1 | 2.11 | 2.02 | 0.19 | 0.758 | 2.07 | 2.10 | 2.06 | 2.03 | 0.267 | 0.998 | 0.738 |
| TMLHE | 1 | 2.04 a | 3.70 b | 0.47 | 0.022 | 3.30 | 2.54 | 2.59 | 3.04 | 0.671 | 0.838 | 0.670 |
| OCTN2 | 1 | 0.92 a | 1.90 b | 0.20 | 0.002 | 1.02 | 1.21 | 1.48 | 1.94 | 0.279 | 0.132 | 0.385 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pike, B.; Zhao, J.; Hicks, J.A.; Wang, F.; Hagen, R.; Liu, H.-C.; Odle, J.; Lin, X. Intestinal Carnitine Status and Fatty Acid Oxidation in Response to Clofibrate and Medium-Chain Triglyceride Supplementation in Newborn Pigs. Int. J. Mol. Sci. 2023, 24, 6066. https://doi.org/10.3390/ijms24076066
Pike B, Zhao J, Hicks JA, Wang F, Hagen R, Liu H-C, Odle J, Lin X. Intestinal Carnitine Status and Fatty Acid Oxidation in Response to Clofibrate and Medium-Chain Triglyceride Supplementation in Newborn Pigs. International Journal of Molecular Sciences. 2023; 24(7):6066. https://doi.org/10.3390/ijms24076066
Chicago/Turabian StylePike, Brandon, Jinan Zhao, Julie A. Hicks, Feng Wang, Rachel Hagen, Hsiao-Ching Liu, Jack Odle, and Xi Lin. 2023. "Intestinal Carnitine Status and Fatty Acid Oxidation in Response to Clofibrate and Medium-Chain Triglyceride Supplementation in Newborn Pigs" International Journal of Molecular Sciences 24, no. 7: 6066. https://doi.org/10.3390/ijms24076066
APA StylePike, B., Zhao, J., Hicks, J. A., Wang, F., Hagen, R., Liu, H.-C., Odle, J., & Lin, X. (2023). Intestinal Carnitine Status and Fatty Acid Oxidation in Response to Clofibrate and Medium-Chain Triglyceride Supplementation in Newborn Pigs. International Journal of Molecular Sciences, 24(7), 6066. https://doi.org/10.3390/ijms24076066






