Adipose Mesenchymal Stromal Cell-Derived Exosomes Carrying MiR-122-5p Antagonize the Inhibitory Effect of Dihydrotestosterone on Hair Follicles by Targeting the TGF-β1/SMAD3 Signaling Pathway
Abstract
1. Introduction
2. Results
2.1. Isolation and Characterization of ADSCs
2.2. ADSC-Exos Promote Hair Growth and Induce HFs to Enter Anagen
2.3. ADSC-Exos Stimulate DPCs Proliferation and Migration
2.4. ADSC-Exos Counteracted the Inhibitory Effect of DHT on DPCs
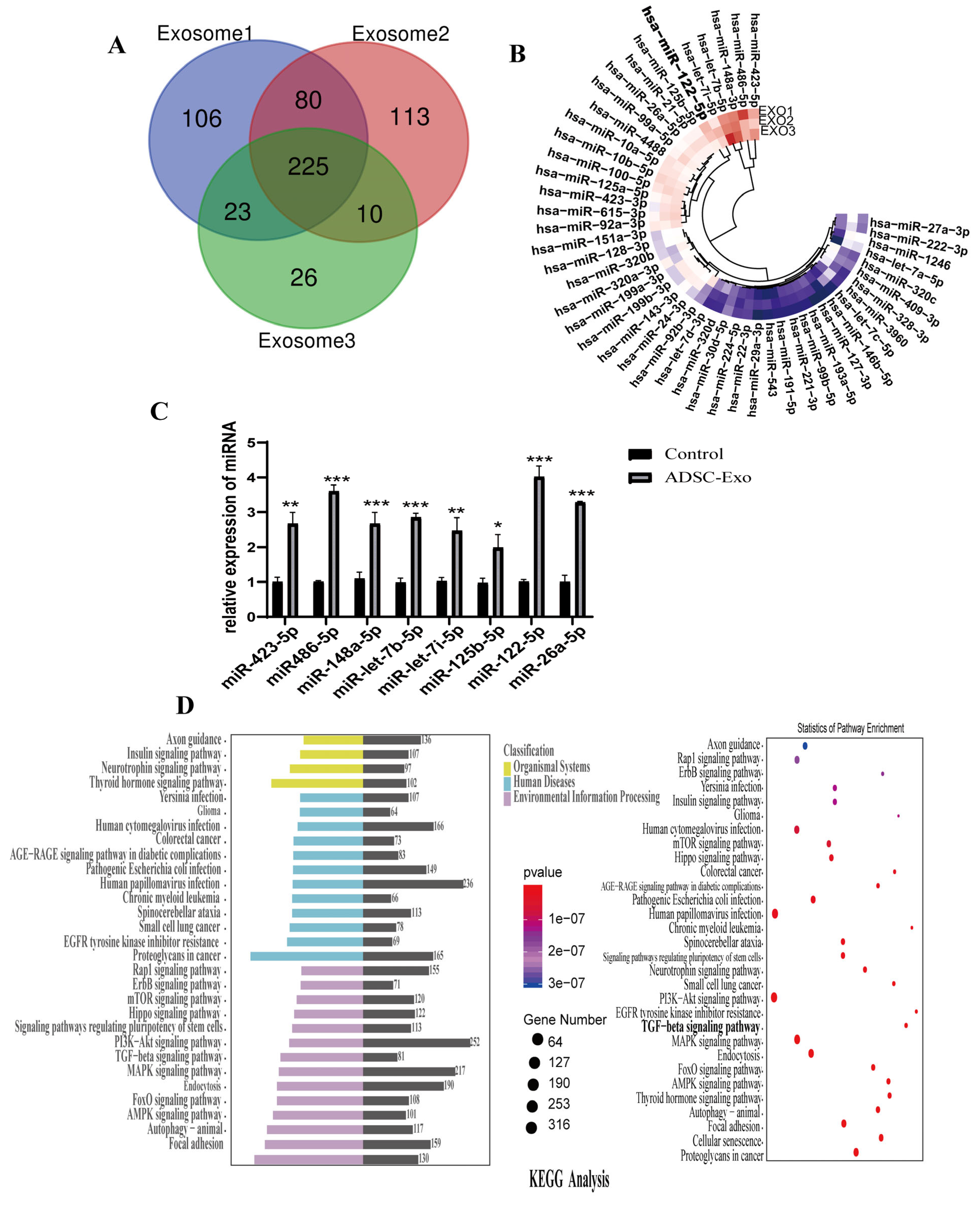
2.5. Screening and Functional Analysis of Target Genes
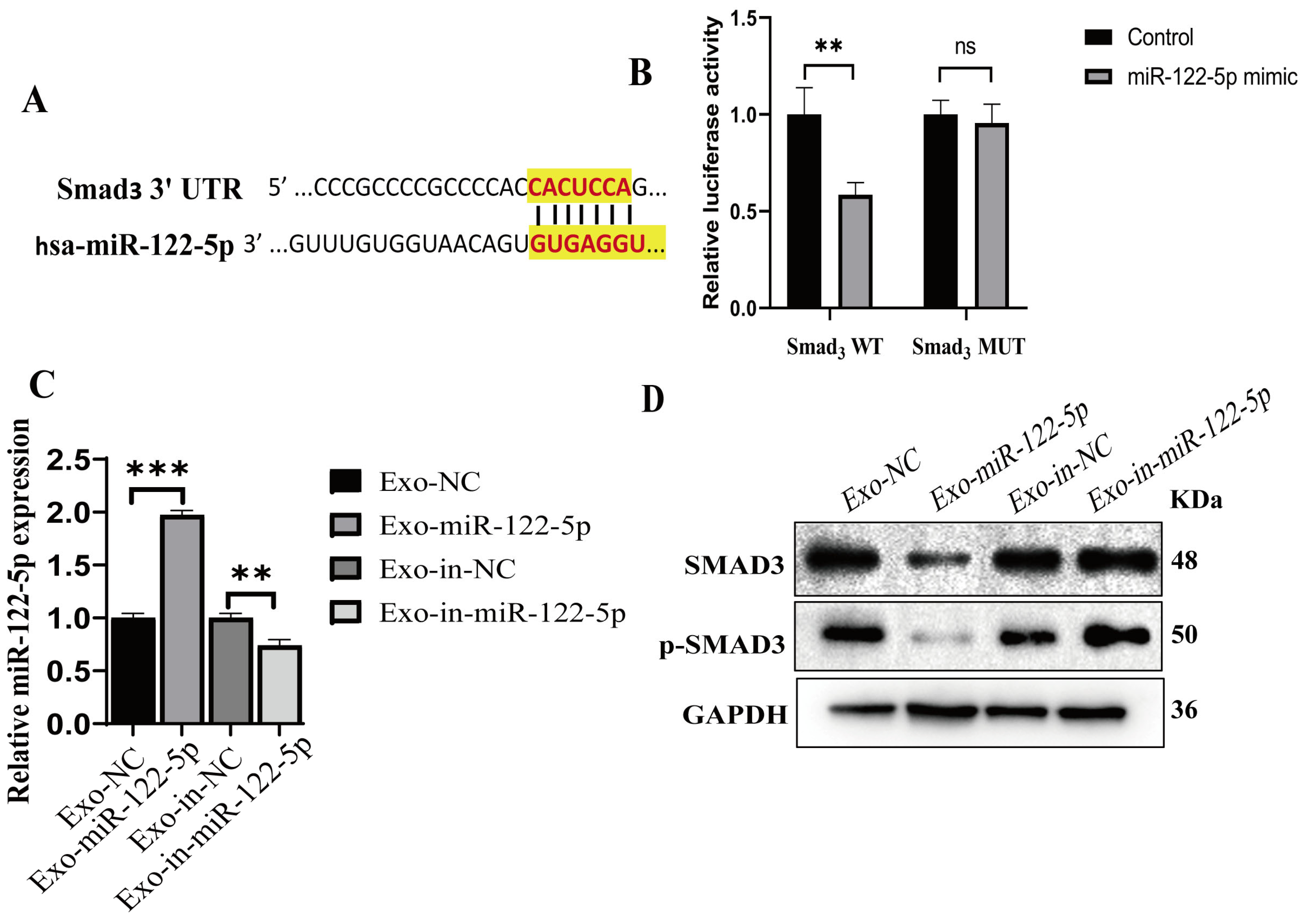
2.6. miR-122-5p Targets and Negatively Regulates SMAD3
2.7. ADSC-Exo-miR-122-5p Counteracted the Inhibitory Effect of DHT on HFs in C57BL/6 Mice
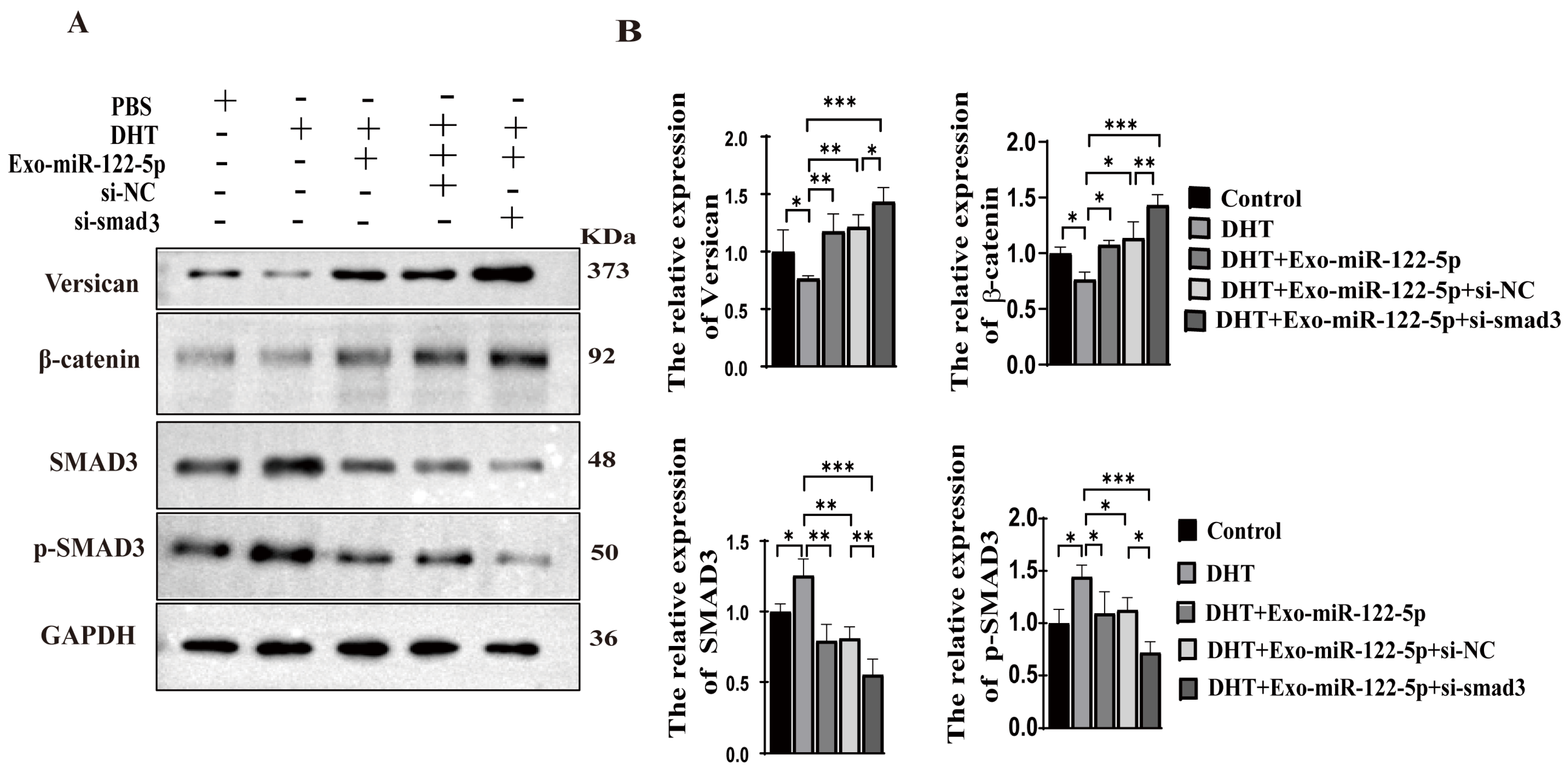
2.8. Inhibition of TGF-β/SMAD Signaling Enhances the Protective Effects of ADSC-Exo-miR-122-5p in DHT-Induced HFs
3. Discussion
4. Methods
4.1. Isolation and Culture of Human HFs
4.2. DPCs Culture
4.3. Isolation and Identification of ADSCs
4.4. Extraction and Identification of ADSC-Deprived Exosomes
4.5. Cellular Uptake and Tracing of ADSC-Exos
4.6. HF Treatment
4.7. Cell Proliferation Assay/Cell Counting Kit-8
4.8. Cell Migration Scratch Test In Vitro
4.9. Cell Transfection and Exosome Editing
4.10. Hematoxylin and Eosin Staining and Immunofluorescence Staining
4.11. RNA Isolation and Quantitative Real Time-PCR
4.12. miRNA Sequencing of ADSC-Exos and Bioinformatics Analysis
4.13. Western Blotting
4.14. Hair Growth In Vivo Study
4.15. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADSCs-Exos | Adipose mesenchymal stromal cell-derived exosomes |
| AGA | Androgenic alopecia |
| DAPI | 4′,6-Diamidino-2-phenylindole |
| DHT | Dihydrotestosterone |
| DMEM | Dulbecco’s modified Eagle’s medium |
| DPCs | Dermal papilla cells |
| FBS | Fetal bovine serum |
| H&E | Hematoxylin and eosin |
| HFs | Hair follicles |
| MSCs | Mesenchymal stromal cells |
| NTA | Nanoparticle tracking analysis |
| SVF | Stromal-vascular fraction |
| SVFs | Stromal-vascular fraction cells |
| TGF-β1 | Transforming growth factor β1 |
| VEGF | Vascular endothelial growth factor |
References
- Varothai, S.; Bergfeld, W. Androgenetic alopecia: An evidence-based treatment update. Am. J. Clin. Dermatol. 2014, 15, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Hordinsky, M.; Donati, A. Alopecia areata: An evidence-based treatment update. Am. J. Clin. Dermatol. 2014, 15, 231–246. [Google Scholar] [CrossRef] [PubMed]
- Krupa Shankar, D.; Chakravarthi, M.; Shilpakar, R. Male androgenetic alopecia: Population-based study in 1005 subjects. Int. J. Trichology 2009, 1, 131–133. [Google Scholar] [CrossRef]
- Cranwell, W.; Sinclair, R. Male Androgenetic Alopecia. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dhatariya, K., Dungan, K., Hershman, J.M., Hofland, J., Kalra, S., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Trüeb, R.M. The impact of oxidative stress on hair. Int. J. Cosmet. Sci. 2015, 37 (Suppl. 2), 25–30. [Google Scholar] [CrossRef] [PubMed]
- Talavera-Adame, D.; Newman, D.; Newman, N. Conventional and novel stem cell based therapies for androgenic alopecia. Stem Cells Cloning Adv. Appl. 2017, 10, 11–19. [Google Scholar] [CrossRef]
- Rose, P. Hair restoration surgery: Challenges and solutions. Clin. Cosmet. Investig. Dermatol. 2015, 8, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P. Autologous Cellular Method Using Micrografts of Human Adipose Tissue Derived Follicle Stem Cells in Androgenic Alopecia. Int. J. Mol. Sci. 2019, 20, 3446. [Google Scholar] [CrossRef]
- Gentile, P.; Scioli, M.; Cervelli, V.; Orlandi, A.; Garcovich, S. Autologous Micrografts from Scalp Tissue: Trichoscopic and Long-Term Clinical Evaluation in Male and Female Androgenetic Alopecia. BioMed Res. Int. 2020, 2020, 7397162. [Google Scholar] [CrossRef]
- Hu, S.; Li, Z.; Lutz, H.; Huang, K.; Su, T.; Cores, J.; Dinh, P.; Cheng, K. Dermal exosomes containing miR-218-5p promote hair regeneration by regulating β-catenin signaling. Sci. Adv. 2020, 6, eaba1685. [Google Scholar] [CrossRef]
- Ogawa, M.; Udono, M.; Teruya, K.; Uehara, N.; Katakura, Y. Exosomes Derived from Fisetin-Treated Keratinocytes Mediate Hair Growth Promotion. Nutrients 2021, 13, 2087. [Google Scholar] [CrossRef]
- Yuan, A.; Bian, Q.; Gao, J. Current advances in stem cell-based therapies for hair regeneration. Eur. J. Pharmacol. 2020, 881, 173197. [Google Scholar] [CrossRef] [PubMed]
- Nestor, M.; Ablon, G.; Gade, A.; Han, H.; Fischer, D. Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics. J. Cosmet. Dermatol. 2021, 20, 3759–3781. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Scioli, M.G.; Bielli, A.; Orlandi, A.; Cervelli, V. Concise Review: The Use of Adipose-Derived Stromal Vascular Fraction Cells and Platelet Rich Plasma in Regenerative Plastic Surgery. Stem. Cells 2017, 35, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Bi, H.; Li, H.; Zhang, C.; Mao, Y.; Nie, F.; Xing, Y.; Sha, W.; Wang, X.; Irwin, D.M.; Tan, H. Stromal vascular fraction promotes migration of fibroblasts and angiogenesis through regulation of extracellular matrix in the skin wound healing process. Stem. Cell Res. 2019, 10, 302. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Scioli, M.; Bielli, A.; Orlandi, A.; Cervelli, V. Comparing different nanofat procedures on scars: Role of the stromal vascular fraction and its clinical implications. Regen. Med. 2017, 12, 939–952. [Google Scholar] [CrossRef]
- Butt, G.; Hussain, I.; Ahmad, F.; Choudhery, M. Stromal vascular fraction-enriched platelet-rich plasma therapy reverses the effects of androgenetic alopecia. J. Cosmet. Dermatol. 2020, 19, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Garcovich, S. Concise Review: Adipose-Derived Stem Cells (ASCs) and Adipocyte-Secreted Exosomal microRNA (A-SE-miR) Modulate Cancer Growth and proMote Wound Repair. J. Clin. Med. 2019, 8, 855. [Google Scholar] [CrossRef]
- Andjelkov, K.; Eremin, I.; Korac, A. Different levels of EGF, VEGF, IL-6, MCP-1, MCP-3, IP-10, Eotaxin and MIP-1α in the adipose-derived stem cell secretome in androgenetic alopecia. Exp. Dermatol. 2022, 31, 946–962. [Google Scholar] [CrossRef]
- Zhao, X.; Ruan, J.; Tang, H.; Li, J.; Shi, Y.; Li, M.; Li, S.; Xu, C.; Lu, Q.; Dai, C. Multi-compositional MRI evaluation of repair cartilage in knee osteoarthritis with treatment of allogeneic human adipose-derived mesenchymal progenitor cells. Stem. Cell Res. Ther. 2019, 10, 308. [Google Scholar] [CrossRef]
- Dong, Y.; Cui, M.; Qu, J.; Wang, X.; Kwon, S.; Barrera, J.; Elvassore, N.; Gurtner, G. Conformable hyaluronic acid hydrogel delivers adipose-derived stem cells and promotes regeneration of burn injury. Acta Biomater. 2020, 108, 56–66. [Google Scholar] [CrossRef]
- Tak, Y.; Lee, S.; Cho, A.; Kim, Y. A randomized, double-blind, vehicle-controlled clinical study of hair regeneration using adipose-derived stem cell constituent extract in androgenetic alopecia. Stem Cells Transl. Med. 2020, 9, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Pegtel, D.; Gould, S. Exosomes. Annu. Rev. Biochem. 2019, 88, 487–514. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, N.; Cheng, Q.; Zhang, H.; Liu, F.; Shang, Y. MiR-21-5p in Macrophage-Derived Exosomes Targets Smad7 to Promote Epithelial Mesenchymal Transition of Airway Epithelial Cells. J. Asthma Allergy 2021, 14, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, H.; Ong, H.; Riau, A.; Stanzel, T.; Mehta, J.; Yam, G. Current Trends and Future Perspective of Mesenchymal Stem Cells and Exosomes in Corneal Diseases. Int. J. Mol. Sci. 2019, 20, 2853. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Zhang, Q.; Hu, W.; Zhao, C.; Lv, W.; Yi, Y.; Wang, Y.; Tang, H.; Wu, M.; Wu, Y. The novel mechanisms and applications of exosomes in dermatology and cutaneous medical aesthetics. Pharmacol. Res. 2021, 166, 105490. [Google Scholar] [CrossRef] [PubMed]
- Feng, N.; Jia, Y.; Huang, X. Exosomes from adipose-derived stem cells alleviate neural injury caused by microglia activation via suppressing NF-kB and MAPK pathway. J. Neuroimmunol. 2019, 334, 576996. [Google Scholar] [CrossRef]
- Hu, L.; Wang, J.; Zhou, X.; Xiong, Z.; Zhao, J.; Yu, R.; Huang, F.; Zhang, H.; Chen, L. Exosomes derived from human adipose mensenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci. Rep. 2016, 6, 32993. [Google Scholar] [CrossRef]
- Xiong, J.; Liu, Z.; Wu, M.; Sun, M.; Xia, Y.; Wang, Y. Comparison of Proangiogenic Effects of Adipose-Derived Stem Cells and Foreskin Fibroblast Exosomes on Artificial Dermis Prefabricated Flaps. Stem. Cells Int. 2020, 2020, 5293850. [Google Scholar] [CrossRef]
- Wu, J.; Yang, Q.; Wu, S.; Yuan, R.; Zhao, X.; Li, Y.; Wu, W.; Zhu, N. Adipose-Derived Stem Cell Exosomes Promoted Hair Regeneration. Tissue Eng. Regen. Med. 2021, 18, 685–691. [Google Scholar] [CrossRef]
- Nilforoushzadeh, M.; Aghdami, N.; Taghiabadi, E. Effects of Adipose-Derived Stem Cells and Platelet-Rich Plasma Exosomes on The Inductivity of Hair Dermal Papilla Cells. Cell J. 2021, 23, 576–583. [Google Scholar]
- Hu, M.; Hong, L.; Liu, C.; Hong, S.; He, S.; Zhou, M.; Huang, G.; Chen, Q. Electrical stimulation enhances neuronal cell activity mediated by Schwann cell derived exosomes. Sci. Rep. 2019, 9, 4206. [Google Scholar] [CrossRef] [PubMed]
- Kazi, T.; Nagata, A.; Nakagawa, T.; Matsuzaki, T.; Inui, S. Dermal Papilla Cell-Derived Extracellular Vesicles Increase Hair Inductive Gene Expression in Adipose Stem Cells via β-Catenin Activation. Cells 2022, 11, 202. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Gao, Y.; Ding, Q.; Liu, J.; Li, Y.; Jin, M.; Xu, H.; Ma, S.; Wang, X.; Zeng, W.; et al. Exosomal Micro RNAs Derived from Dermal Papilla Cells Mediate Hair Follicle Stem Cell Proliferation and Differentiation. Int. J. Biol. Sci. 2019, 15, 1368–1382. [Google Scholar] [CrossRef] [PubMed]
- Driskell, R.R.; Clavel, C.; Rendl, M.; Watt, F.M. Hair follicle dermal papilla cells at a glance. J. Cell Sci. 2011, 124, 1179–1182. [Google Scholar] [CrossRef]
- Kang, J.; Kim, S.; Kim, M.; Boo, H.; Kim, E.; Im, G.; Kim, Y.; Hyun, J.; Kang, J.; Koh, Y.; et al. Effects of dihydrotestosterone on rat dermal papilla cells in vitro. Eur. J. Pharmacol. 2015, 757, 74–83. [Google Scholar] [CrossRef]
- Won, C.; Park, G.; Wu, X.; Tran, T.; Park, K.; Park, B.; Kim, D.; Kwon, O.; Kim, K. The Basic Mechanism of Hair Growth Stimulation by Adipose-derived Stem Cells and Their Secretory Factors. Curr. Stem Cell Res. Ther. 2017, 12, 535–543. [Google Scholar] [CrossRef]
- Fu, D.; Huang, J.; Li, K.; Chen, Y.; He, Y.; Sun, Y.; Guo, Y.; Du, L.; Qu, Q.; Miao, Y.; et al. Dihydrotestosterone-induced hair regrowth inhibition by activating androgen receptor in C57BL6 mice simulates androgenetic alopecia. Biomed. Pharmacother. Biomed. Pharmacother. 2021, 137, 111247. [Google Scholar] [CrossRef]
- Shin, H.; Won, C.H.; Chung, W.K.; Park, B.S. Up-to-date Clinical Trials of Hair Regeneration Using Conditioned Media of Adipose-Derived Stem Cells in Male and Female Pattern Hair Loss. Curr. Stem. Cell Res. 2017, 12, 524–530. [Google Scholar] [CrossRef]
- Lou, J.; Wu, J.; Feng, M.; Dang, X.; Wu, G.; Yang, H.; Wang, Y.; Li, J.; Zhao, Y.; Shi, C.; et al. Exercise promotes angiogenesis by enhancing endothelial cell fatty acid utilization via liver-derived extracellular vesicle miR-122-5p. J. Sport Health Sci. 2021, 11, 495–508. [Google Scholar] [CrossRef]
- Wei, Z.; Liu, J.; Xie, H.; Wang, B.; Wu, J.; Zhu, Z. MiR-122-5p Mitigates Inflammation, Reactive Oxygen Species and SH-SY5Y Apoptosis by Targeting CPEB1 After Spinal Cord Injury Via the PI3K/AKT Signaling Pathway. Neurochem. Res. 2021, 46, 992–1005. [Google Scholar] [CrossRef]
- Ding, Z.; Lin, J.; Sun, Y.; Cong, S.; Liu, S.; Zhang, Y.; Chen, Q.; Chen, J. miR-122-5p negatively regulates the transforming growth factor-β/Smad signaling pathway in skeletal muscle myogenesis. Cell Biochem. Funct. 2020, 38, 231–238. [Google Scholar] [CrossRef]
- Domenis, R.; Lazzaro, L.; Calabrese, S.; Mangoni, D.; Gallelli, A.; Bourkoula, E.; Manini, I.; Bergamin, N.; Toffoletto, B.; Beltrami, C.; et al. Adipose tissue derived stem cells: In vitro and in vivo analysis of a standard and three commercially available cell-assisted lipotransfer techniques. Stem Cell Res. Ther. 2015, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Zhu, C.; Jia, J.; Hao, X.Y.; Yu, X.Y.; Liu, X.Y.; Shu, M.G. ADSC-Exos containing MALAT1 promotes wound healing by targeting miR-124 through activating Wnt/β-catenin pathway. Biosci. Rep. 2020, 40, BSR20192549. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Chen, J.; Duscher, D.; Liu, Y.; Guo, G.; Kang, Y.; Xiong, H.; Zhan, P.; Wang, Y.; Wang, C.; et al. Microvesicles from human adipose stem cells promote wound healing by optimizing cellular functions via AKT and ERK signaling pathways. Stem. Cell Res. 2019, 10, 47. [Google Scholar] [CrossRef] [PubMed]
- Zaborowski, M.; Balaj, L.; Breakefield, X.; Lai, C. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience 2015, 65, 783–797. [Google Scholar] [CrossRef]
- Yoo, H.; Won, C.; Lee, S.; Kwon, O.; Kim, K.; Eun, H.; Cho, K. Expression of androgen and estrogen receptors in human scalp mesenchymal cells in vitro. Arch. Dermatol. Res. 2007, 298, 505–509. [Google Scholar] [CrossRef]
- Inui, S.; Fukuzato, Y.; Nakajima, T.; Yoshikawa, K.; Itami, S. Androgen-inducible TGF-beta1 from balding dermal papilla cells inhibits epithelial cell growth: A clue to understand paradoxical effects of androgen on human hair growth. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2002, 16, 1967–1969. [Google Scholar]
- Foitzik, K.; Lindner, G.; Mueller-Roever, S.; Maurer, M.; Botchkareva, N.; Botchkarev, V.; Handjiski, B.; Metz, M.; Hibino, T.; Soma, T.; et al. Control of murine hair follicle regression (catagen) by TGF-beta1 in vivo. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2000, 14, 752–760. [Google Scholar]
- Michel, L.; Reygagne, P.; Benech, P.; Jean-Louis, F.; Scalvino, S.; Ly Ka So, S.; Hamidou, Z.; Bianovici, S.; Pouch, J.; Ducos, B.; et al. Study of gene expression alteration in male androgenetic alopecia: Evidence of predominant molecular signalling pathways. Br. J. Dermatol. 2017, 177, 1322–1336. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, Y.; Liu, Y.; Li, X.; Tang, L.; Duan, M.; Li, J.; Zhang, G. Exosomes derived from human umbilical cord blood mesenchymal stem cells stimulate regenerative wound healing via transforming growth factor-β receptor inhibition. Stem Cell Res. Ther. 2021, 12, 434. [Google Scholar] [CrossRef]
- Yin, K.; Wang, S.; Zhao, R. Exosomes from mesenchymal stem/stromal cells: A new therapeutic paradigm. Biomark. Res. 2019, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wang, H.; Jing, J.; Yu, L.; Wu, X.; Lu, Z. Regulation of hair follicle development by exosomes derived from dermal papilla cells. Biochem. Biophys. Res. Commun. 2018, 500, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yan, J.; Liu, Y.; Chen, Z.; Li, X.; Tang, L.; Li, J.; Duan, M.; Zhang, G. Human Amniotic Fluid Stem Cell-Derived Exosomes as a Novel Cell-Free Therapy for Cutaneous Regeneration. Front. Cell Dev. Biol. 2021, 9, 685873. [Google Scholar] [CrossRef]
- Jiang, T.; Wang, Z.; Sun, J. Human bone marrow mesenchymal stem cell-derived exosomes stimulate cutaneous wound healing mediates through TGF-β/Smad signaling pathway. Stem Cell Res. Ther. 2020, 11, 198. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wu, H.; Peng, Y.; Zhao, Y.; Qin, Y.; Zhang, Y.; Xiao, Z. Hypoxia adipose stem cell-derived exosomes promote high-quality healing of diabetic wound involves activation of PI3K/Akt pathways. J. Nanobiotechnol. 2021, 19, 202. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, D.; Ma, T.; Liu, Q. Vascular Endothelial Growth Factor Protects CD200-Rich and CD34-Positive Hair Follicle Stem Cells Against Androgen-Induced Apoptosis Through the Phosphoinositide 3-Kinase/Akt Pathway in Patients with Androgenic Alopecia. Dermatol. Surg. Off. Publ. Am. Soc. Dermatol. Surg. 2020, 46, 358–368. [Google Scholar] [CrossRef]
- Lachgar, S.; Charveron, M.; Gall, Y.; Bonafe, J. Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. Br. J. Dermatol. 1998, 138, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Ying, Z.; Ke, F.; Mei-Lin, X. Apigenin suppresses TGF-β1-induced cardiac fibroblast differentiation and collagen synthesis through the downregulation of HIF-1α expression by miR-122-5p. Phytomed. Int. J. Phytother. Phytopharm. 2021, 83, 153481. [Google Scholar] [CrossRef]
- Jiang, M.; Ma, W.; Gao, Y.; Jia, K.; Zhang, Y.; Liu, H.; Sun, Q. IL-22-induced miR-122-5p promotes keratinocyte proliferation by targeting Sprouty2. Exp. Dermatol. 2017, 26, 368–374. [Google Scholar] [CrossRef]
- Li, S.; Chen, J.; Chen, F.; Wang, C.; Guo, X.; Wang, C.; Fan, Y.; Wang, Y.; Peng, Y.; Li, W. Liposomal honokiol promotes hair growth via activating Wnt3a/β-catenin signaling pathway and down regulating TGF-β1 in C57BL/6N mice. Biomed. Pharmacother. Biomed. Pharmacother. 2021, 141, 111793. [Google Scholar] [CrossRef]
- Chen, X.; Liu, B.; Li, Y.; Han, L.; Tang, X.; Deng, W.; Lai, W.; Wan, M. Dihydrotestosterone Regulates Hair Growth Through the Wnt/β-Catenin Pathway in C57BL/6 Mice and In Vitro Organ Culture. Front. Pharmacol. 2019, 10, 1528. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Huang, J.; Liu, Z.; Chen, R.; Fu, D.; Yang, L.; Wang, J.; Du, L.; Wen, L.; Miao, Y.; et al. miR-140-5p in Small Extracellular Vesicles From Human Papilla Cells Stimulates Hair Growth by Promoting Proliferation of Outer Root Sheath and Hair Matrix Cells. Front. Cell Dev. Biol. 2020, 8, 593638. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Chen, J.; Guo, J.; Liu, Y.; Xiong, H.; Jing, B.; Yang, X.; Li, G.; Kang, Y.; Wang, C.; et al. Exosomes from Adipose Stem Cells Promote Diabetic Wound Healing through the eHSP90/LRP1/AKT Axis. Cells 2022, 11, 3229. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Yang, Y.; Xu, Y.; Jiang, W.; Shao, Y.; Xing, J.; Chen, Y.; Han, Y. Biomimetic nerve guidance conduit containing engineered exosomes of adipose-derived stem cells promotes peripheral nerve regeneration. Stem Cell Res. Ther. 2021, 12, 442. [Google Scholar] [CrossRef] [PubMed]
- Qu, H.; Wu, S.; Li, J.; Ma, T.; Li, J.; Xiang, B.; Jiang, H.; Zhang, Q. MiR-125b regulates the differentiation of hair follicles in Fine-wool Sheep and Cashmere goats by targeting MXD4 and FGFR2. Anim. Biotechnol. 2021, 1–8. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Y.; Tang, X.; Zhang, X.; Cao, C.; Yu, M.; Wan, M. Adipose Mesenchymal Stromal Cell-Derived Exosomes Carrying MiR-122-5p Antagonize the Inhibitory Effect of Dihydrotestosterone on Hair Follicles by Targeting the TGF-β1/SMAD3 Signaling Pathway. Int. J. Mol. Sci. 2023, 24, 5703. https://doi.org/10.3390/ijms24065703
Liang Y, Tang X, Zhang X, Cao C, Yu M, Wan M. Adipose Mesenchymal Stromal Cell-Derived Exosomes Carrying MiR-122-5p Antagonize the Inhibitory Effect of Dihydrotestosterone on Hair Follicles by Targeting the TGF-β1/SMAD3 Signaling Pathway. International Journal of Molecular Sciences. 2023; 24(6):5703. https://doi.org/10.3390/ijms24065703
Chicago/Turabian StyleLiang, Yunxiao, Xin Tang, Xue Zhang, Cuixiang Cao, Miao Yu, and Miaojian Wan. 2023. "Adipose Mesenchymal Stromal Cell-Derived Exosomes Carrying MiR-122-5p Antagonize the Inhibitory Effect of Dihydrotestosterone on Hair Follicles by Targeting the TGF-β1/SMAD3 Signaling Pathway" International Journal of Molecular Sciences 24, no. 6: 5703. https://doi.org/10.3390/ijms24065703
APA StyleLiang, Y., Tang, X., Zhang, X., Cao, C., Yu, M., & Wan, M. (2023). Adipose Mesenchymal Stromal Cell-Derived Exosomes Carrying MiR-122-5p Antagonize the Inhibitory Effect of Dihydrotestosterone on Hair Follicles by Targeting the TGF-β1/SMAD3 Signaling Pathway. International Journal of Molecular Sciences, 24(6), 5703. https://doi.org/10.3390/ijms24065703




