The IMBG Test for Evaluating the Pharmacodynamic Response to Immunosuppressive Therapy in Kidney Transplant Patients: Current Evidence and Future Applications
Abstract
1. Introduction
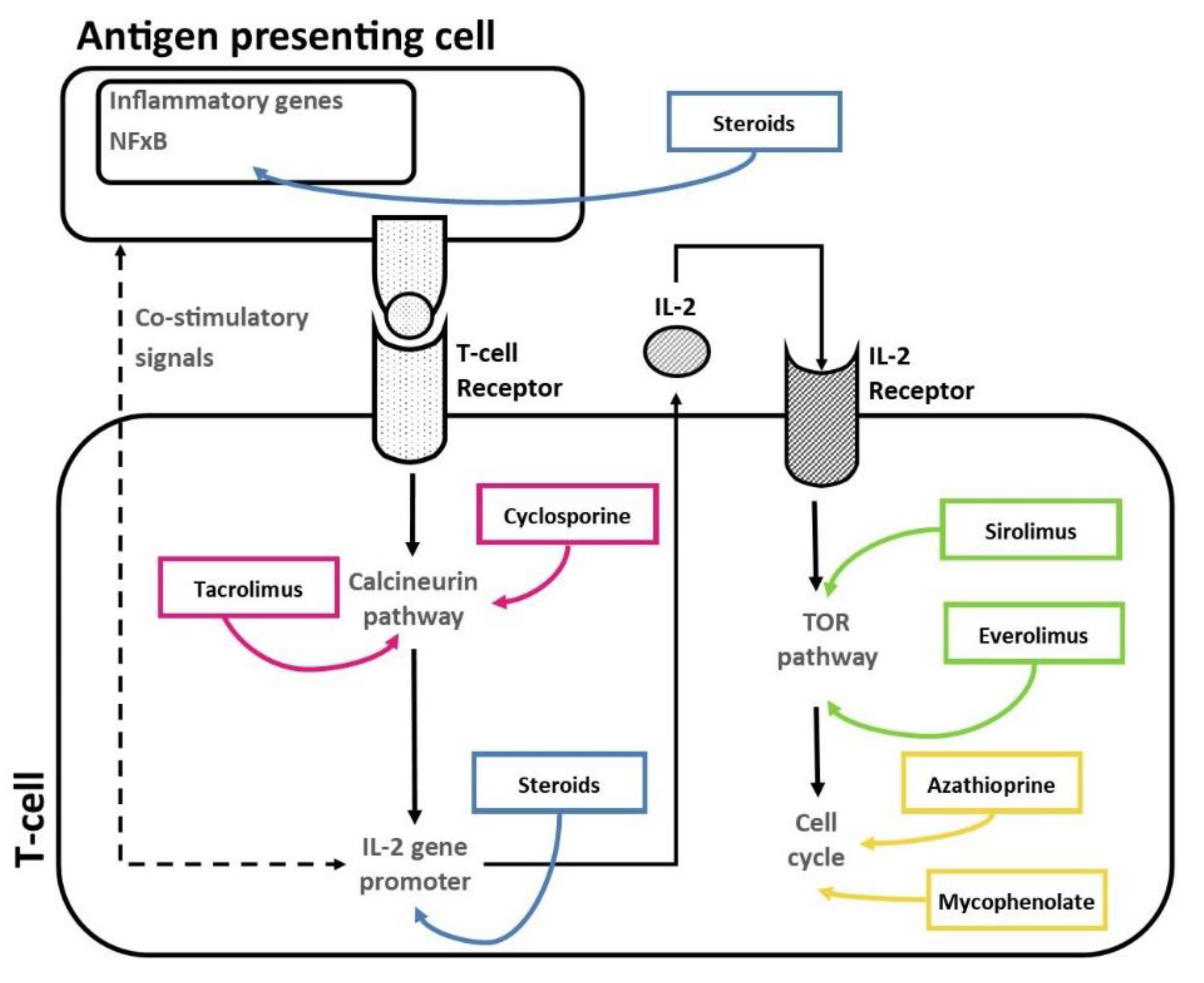
2. Current Approaches for In Vitro Determination of Pharmacological Response to Immunosuppressants
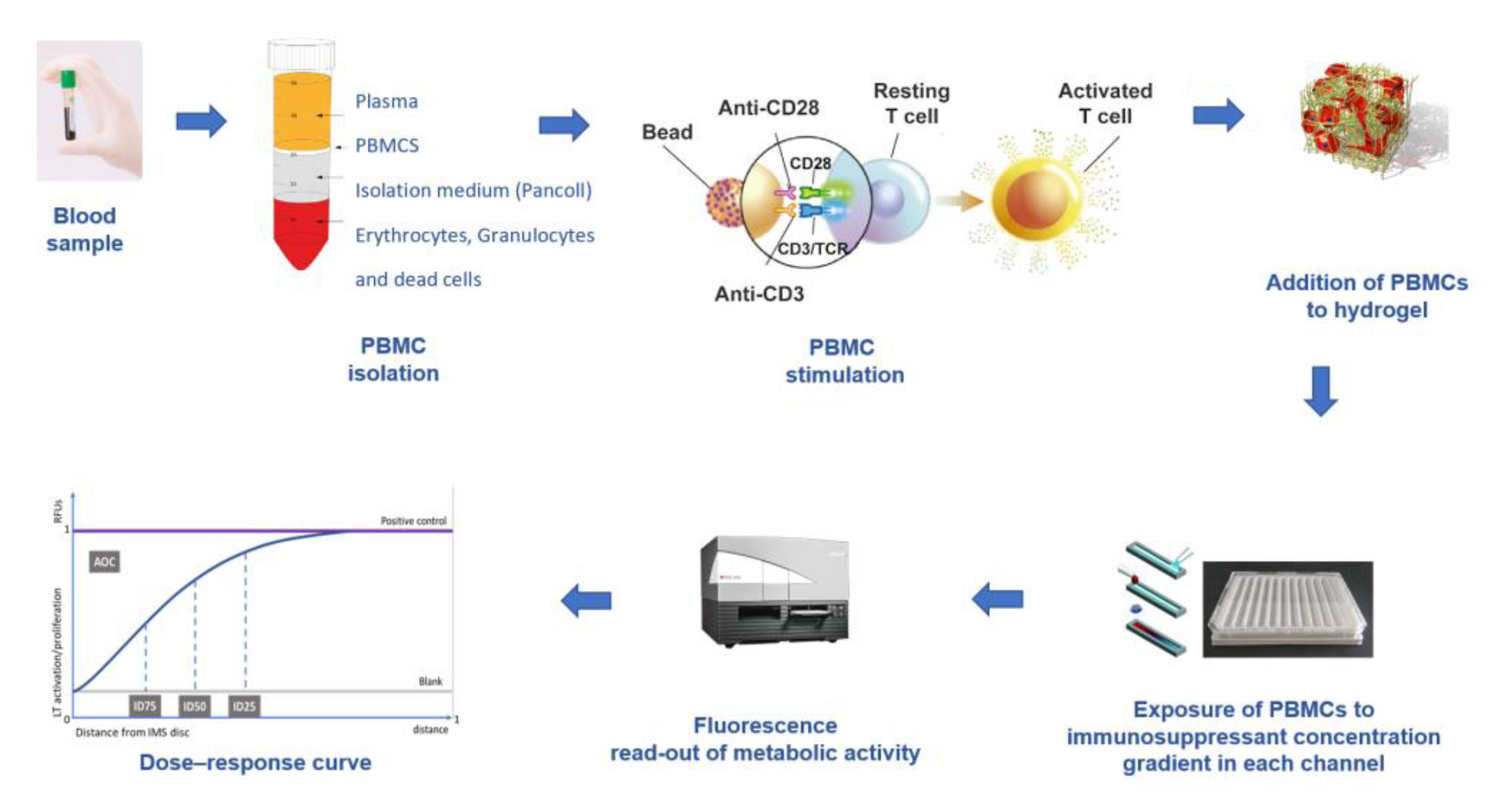
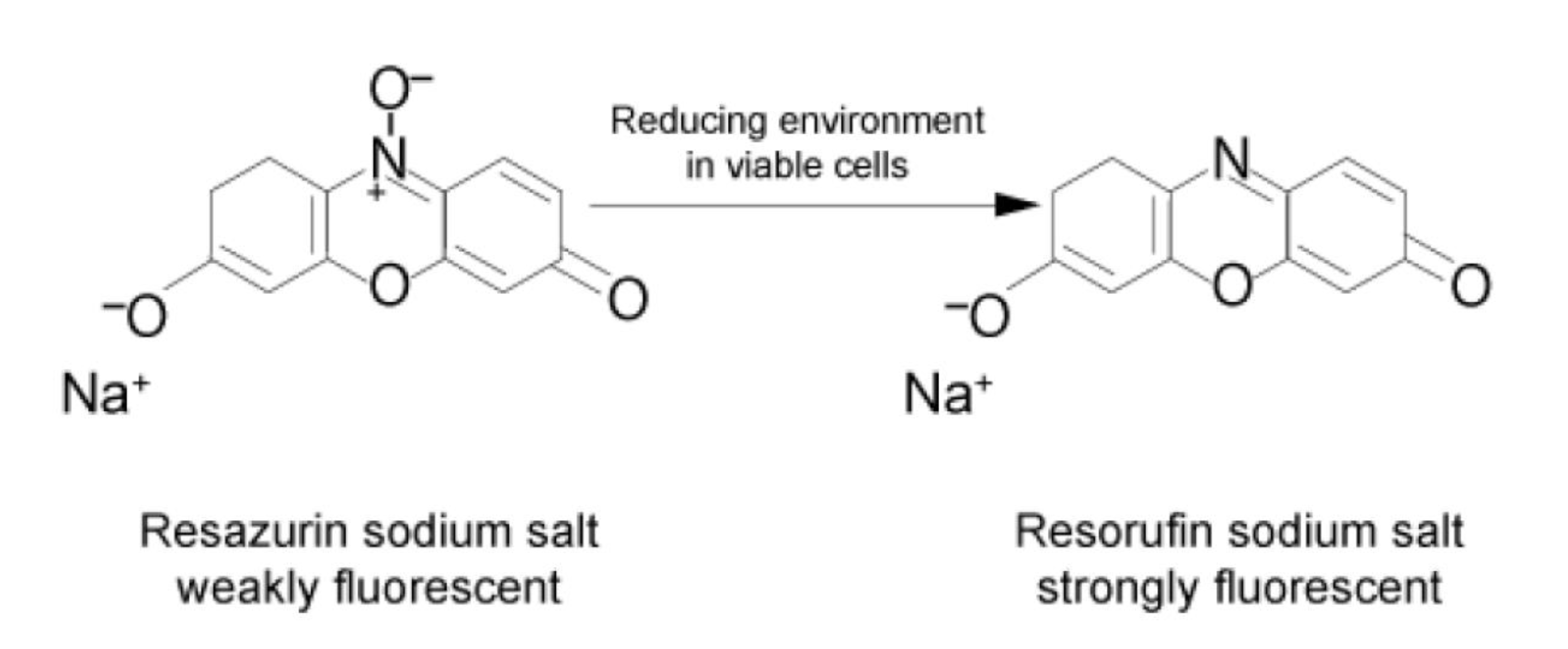
3. The Immunobiogram Test: Methodology and Data Analysis
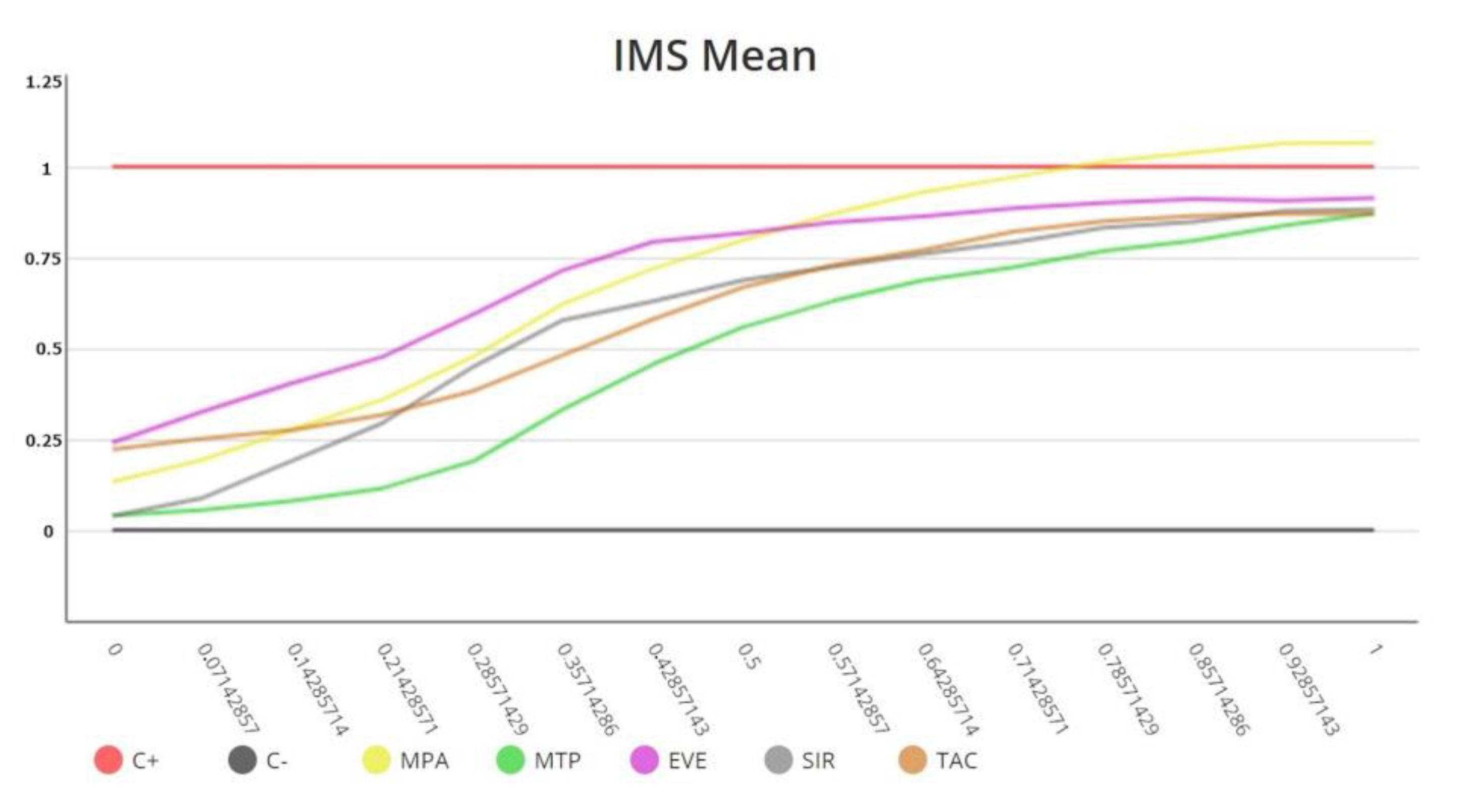
4. The IMBG Measures In Vitro Sensitivity to Immunosuppressive Drugs in KT Patients
5. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hariharan, S.; Israni, A.; Danovitch, G. Long-term survival after kidney transplantation. N. Engl. J. Med. 2021, 385, 729–743. [Google Scholar] [CrossRef]
- Kasiske, L.; Zeier, M.; Craig, J.; Chapman, J.R.; Craig, J.C.; Ekberg, H.; Garvey, C.A.; Green, M.D.; Jha, V.; Josephson, M.A.; et al. Kidney Tx guideline. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am. J. Transplant. 2009, 9, S6–S19. [Google Scholar]
- Baker, R.; Mark, P.; Patel, R.; Stevens, K.; Palmer, N. Renal association clinical practice guideline in post-operative care in the kidney transplant recipient. BMC Nephrol. 2017, 18, 174. [Google Scholar] [CrossRef]
- Neuberger, J.; Bechstein, W.; Kuypers, D.; Burra, P.; Citterio, F.; De Geest, S.; Duvoux, C.; Jardine, A.G.; Kamar, N.; Krämer, B.; et al. Practical recommendations for long-term Management of Modifiable Risks in kidney and liver transplant recipients: A guidance report and clinical checklist by the consensus on managing modifiable risk in transplantation (COMMIT) group. Transplantation 2017, 101, S1–S56. [Google Scholar] [CrossRef] [PubMed]
- Cherukuri, A.; Mehta, R.; Sharma, A.; Sood, P.; Zeevi, A.; Tevar, A.D.; Rothstein, D.M.; Hariharan, S. Post-transplant donor specific antibody is associated with poor kidney transplant outcomes only when combined with both T-cell-mediated rejection and non-adherence. Kidney Int. 2019, 96, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Salinas, F.O.; Santos, J.P.; Mateu, L.P. Inmunosupresión en el trasplante renal. Nefrol. Día. 2020. Available online: https://www.nefrologiaaldia.org/es-articulo-inmunosupresion-el-trasplante-renal-241 (accessed on 10 December 2022).
- Chen, C.C.; Koenig, A.; Saison, C.; Dahdal, S.; Rigault, G.; Barba, T.; Taillardet, M.; Chartoire, D.; Ovize, M.; Morelon, E.; et al. CD4+ T Cell Help Is Mandatory for Naive and Memory Donor-Specific Antibody Responses: Impact of Therapeutic Immunosuppression. Front. Immunol. 2018, 19, 275. [Google Scholar] [CrossRef]
- Gorbacheva, V.; Fan, R.; Fairchild, R.L.; Baldwin, W.M.; Valujskikh, A. Memory CD4 T Cells Induce Antibody-Mediated Rejection of Renal Allografts. J Am. Soc. Nephrol. 2016, 27, 3299–3307. [Google Scholar] [CrossRef]
- Christians, U.; Schmitz, V.; Schoning, W.; Bendrick-Peart, J.; Klawitter, J.; Haschke, M.; Klawitter, J. Toxicodynamic therapeutic drug monitoring of immunosuppressants: Promises, reality, and challenges. Ther. Drug Monit. 2008, 30, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Kuypers, D. Immunosuppressive drug monitoring—What to use in clinical practice today to improve renal graft outcome. Transp. Int. 2005, 18, 140–150. [Google Scholar] [CrossRef]
- Andrews, M.L.Y.; Brenda, C.; Shi, Y.Y.; Baan, C.C.; Hesselink, D.A. Pharmacokinetic considerations related to therapeutic drug monitoring of tacrolimus in kidney transplant patients. Expert Opin. Drug Metab. Toxicol. 2017, 13, 1225–1236. [Google Scholar] [CrossRef]
- Bestard, O.; Nickel; Cruzado, J.; Schoenemann, C.; Boenisch, O.; Sefrin, A.; Grinyó, J.M.; Volk, H.D.; Reinke, P. Circulating alloreactive T cells correlate with graft function in longstanding renal transplant recipients. J. Am. Soc. Nephrol. 2008, 19, 1419–1429. [Google Scholar] [CrossRef]
- Sottong, P.; Rosebrock, J.; Britz, J.; Kramer, T. Measurement of T-lymphocyte responses in whole-blood cultures using newly synthesized DNA and AT. Clin. Diagn. Lab. Immunol. 2000, 7, 307–311. [Google Scholar] [CrossRef]
- Bestard, O.; Cravedi, P. Monitoring alloimmune response in kidney transplantation. J. Nephrol. 2017, 30, 187–200. [Google Scholar] [CrossRef]
- Portoles, J.; Jimenez, C.; Janeiro, D.; Lopez-Oliva, M.; Ortega-Carrion, A.; Blanquez, D.; Arribas, L.; Gomez, C.; Diez, T.; Pascual, P.; et al. The Immunobiogram, a novel in vitro assay to evaluate treatment resistance in patients receiving immunosuppressive therapy. Front. Immunol. 2021, 11, 3483. [Google Scholar] [CrossRef] [PubMed]
- Pascual, J.; Jiménez, C.; Krajewskad, M.; Seron, D.; Kotton, C.N.; Portolés, J.; Witzke, O.; Sorensen, S.S.; Andrés, A.; Crespo, M.; et al. The Immunobiogram, a novel in vitro diagnostic test to measure the pharmacodynamic response to immunosuppressive therapy in kidney transplant patients. Transpl. Immunol. 2022, 75, 101711. [Google Scholar] [CrossRef] [PubMed]
- Heemann, U.; Abramowicz, D.; Spasovski, G.; Vanholder, R. Aprobación de las directrices Kidney Disease Improving Global Outcomes (KDIGO) sobre el trasplante renal: Una declaración de posición de la European Renal Best Practice (ERBP). Nephrol. Dial. Transplant. 2011, 26, 2099–2106. [Google Scholar] [CrossRef]
- Schinstock, C.; Dadhania, D.; Everly, M.; Smith, B.; Gandhi, M.; Farkash, E.; Sharma, V.K.; Samaniego-Picota, M.; Stegall, M.D. Factors at de novo donor-specific antibody initial detection associated with allograft loss: A multicenter study. Transp. Int. 2019, 32, 502–515. [Google Scholar] [CrossRef] [PubMed]
- Tait, B.; Süsal, C.; Gebel, H.; Nickerson, P.W.; Zachary, A.A.; Claas, F.H.; Reed, E.F.; Bray, R.A.; Campbell, P.; Chapman, J.R.; et al. Consensus guidelines on the testing and clinical management issues associated with HLA and non-HLA antibodies in transplantation. Transplantation 2013, 15, 19–47. [Google Scholar] [CrossRef] [PubMed]
- Crespo, M.; Llinàs-Mallol, L.; Redondo-Pachón, D.; Butler, C.; Gimeno, J.; Pérez-Sáez, M.; Burballa, C.; Buxeda, A.; Arias-Cabrales, C.; Folgueiras, M.; et al. Non-HLA Antibodies and Epitope Mismatches in Kidney Transplant Recipients With Histological Antibody-Mediated Rejection. Front. Immunol. 2021, 12, 703457. [Google Scholar] [CrossRef]
- Bloom, R.; Bromberg, J.; Poggio, E.; Bunnapradist, S.; Langone, A.J.; Sood, P.; Matas, A.J.; Mehta, S.; Mannon, R.B.; Sharfuddin, A.; et al. Cell-free DNA and active rejection in kidney allografts. J. Am. Soc. Nephrol. 2017, 28, 2221–2232. [Google Scholar] [CrossRef]
- Sigdel, T.; Archila, F.; Constantin, T.; Prins, S.A.; Liberto, J.; Damm, I.; Towfighi, P.; Navarro, S.; Kirkizlar, E.; Demko, Z.P.; et al. Optimizing detection of kidney transplant injury by assessment of donor derived cell-free DNA via massively multiplex PCR. J. Clin. Med. 2018, 8, 8. [Google Scholar] [CrossRef]
- Sigdel, T.; Yang, J.; Bestard, O.; Schroeder, A.; Hsieh, S.C.; Liberto, J.M.; Damm, I.; Geraedts, A.C.M.; Sarwal, M.M. A urinary Common Rejection Module (uCRM) score for non-invasive kidney transplant monitoring. PLoS ONE 2019, 31, e0220052. [Google Scholar] [CrossRef]
- Suthanthiran, M.; Schwartz, J.; Ding, R.; Abecassis, M.; Dadhania, D.; Samstein, B.; Knechtle, S.J.; Friedewald, J.; Becker, Y.T.; Sharma, V.K.; et al. Urinary-cell mRNA profile and acute cellular rejection in kidney allografts. N. Engl. J. Med. 2013, 369, 20–31. [Google Scholar] [CrossRef]
- Roedder, S.; Sigdel, T.; Salomonis, N.; Hsieh, S.; Dai, H.; Bestard, O.; Metes, D.; Zeevi, A.; Gritsch, A.; Cheeseman, J.; et al. The kSORT assay to detect renal transplant patients at high risk for acute rejection: Results of the multicenter AART study. PLoS Med. 2014, 11, e1001759. [Google Scholar] [CrossRef]
- Zhang, W.; Yi, Z.; Keung, K.; Wei, C.; Cravedi, P.; Sun, Z.; Xi, C.; Woytovich, C.; Farouk, S.; Huang, W.; et al. Peripheral Blood Gene Expression Signature to Diagnose Subclinical Acute Rejection. J. Am. Soc. Nephrol. 2019, 30, 1481–1494. [Google Scholar] [CrossRef] [PubMed]
- Friedewald, J.; Kurian, S.; Heilman, R.; Whisenant, T.C.; Poggio, E.D.; Marsh, C.; Baliga, P.; Odim, J.; Brown, M.M.; Ikle, D.N.; et al. Development and clinical validity of a novel blood-based molecular biomarker for subclinical acute rejection following kidney transplant. Am. J. Transplant. 2019, 19, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Loupy, A.; Aubert, O.; Orandi, B.; Naesens, M.; Bouatou, Y.; Raynaud, M.; Divard, G.; Jackson, A.M.; Viglietti, D.; Giral, M.; et al. Prediction system for risk of allograft loss in patients receiving kidney transplants: International derivation and validation study. BMJ 2019, 17, l4923. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Sarwal, R.; Sigdel, T.; Damm, I.; Rosenbaum, B.; Liberto, J.M.; Chan-On, C.; Arreola-Guerra, J.M.; Alberu, J.; Vincenti, F.; et al. A urine score for noninvasive accurate diagnosis and prediction of kidney transplant rejection. Sci. Transl. Med. 2020, 12, eaba250. [Google Scholar] [CrossRef]
- Langhoff, E.; Ladefoged, J.; Jakobsen, B.; Platz, P.; Ryder, L.P.; Svejgaard, A.; Thaysen, J.H. Recipient lymphocyte sensitivity to methylprednisolone affects cadaver kidney graft survival. Lancet 1986, 327, 1296–1297. [Google Scholar] [CrossRef]
- Langhoff, E.; Ladefoged, J. The impact of high lymphocyte sensitivity to glucocorticoids on kidney graft survival in patients treated with azathioprine and cyclosporine. Transplantation 1987, 43, 380–384. [Google Scholar] [CrossRef] [PubMed]
- Francis, D.; Dumble, L.; Bowes, L.; Clunie, G.J.; Macdonald, I.M. Adverse influence of recipient lymphoid resistance to in vitro immunosuppression on the outcome of kidney transplants. Transplantation 1988, 46, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Oka, K.; Takeuchi, H.; Sakurai, E.; Matsuno, N.; Tamaki, T.; Kozaki, M. Clinical significance of glucocorticoid pharmacodynamics assessed by anti-lymphocite action in kidney transplantation: Marked difference between prednisolone and methylprednisolone. Transplantation 1994, 57, 1341–1348. [Google Scholar] [CrossRef]
- Hirano, T.; Oka, K.; Sakurai, E.; Iwamoto, H.; Shimazu, M.; Unezaki, S.; Hirano, T. Impaired prednisolone sensitivity of endogenous system as well as that of peripheral-blood lymphocytes is closely related to clinical incidence in renal transplantation. J. Pharm. Pharmacol. 1991, 43, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Hirano, T.; Oka, K.; Mizumoto, K.; Akashi, T.; Sakurai, E.; Degawa, T.; Uchiyama, M.; Kozaki, K.; Matsuno, N.; et al. Lymphocyte-sensitivities to cyclosporine and tacrolimus in chronic renal failure patients and their significance on renal transplantation. Transplant. Proc. 1998, 30, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Muhetaer, G.; Takeuchi, H.; Unezaki, S.; Kawachi, S.; Iwamoto, H.; Nakamura, Y.; Shimazu, M.; Sugiyama, K.; Hirano, T. Clinical Significance of Peripheral Blood Lymphocyte Sensitivity toGlucocorticoids for the Differentiation of High-risk Patients With Decreased Allograft Function After Glucocorticoid Withdrawal in Renal Transplantation. Clin. Ther. 2014, 36, 1264–1272. [Google Scholar] [CrossRef]
- Sugiyama, K.; Isogai, K.; Toyama, A.; Satoh, H.; Saito, K.; Nakagawa, Y.; Tasaki, M.; Takahashi, K.; Saito, N.; Hirano, T. Cyclosporine pharmacological efficacy estimated by lymphocyte immunosuppressant sensitivity test before and after renal transplantation. J. Clin. Pharm. Ther. 2009, 34, 539–545. [Google Scholar] [CrossRef]
- Sugiyama, K.; Isogai, K.; Horisawa, S.; Toyama, A.; Satoh, H.; Saito, K.; Nakagawa, Y.; Tasaki, M.; Takahashi, K.; Hirano, T.; et al. The pharmacological efficacy of mycophenolic acid before and after renal transplantation as estimated by the lymphocyte immunosuppressant sensitivity test (LIST). Immunopharmacol. Immunotoxicol. 2010, 32, 430–436. [Google Scholar] [CrossRef]
- Sugiyama, K.; Sasahara, H.; Tsukaguchi, M.; Isogai, K.; Toyama, A.; Satoh, H.; Saitoh, K.; Nakagawa, Y.; Takahashi, K.; Tanaka, S.; et al. Peripheral Lymphocyte Response to Mycophenolic Acid In Vitro and Incidence of Cytomegalovirus Infection in Renal Transplantation. Cell Med. 2013, 6, 47–55. [Google Scholar] [CrossRef][Green Version]
- Sugiyama, K.; Isogai, K.; Toyama, A.; Satoh, H.; Saito, K.; Nakagawa, Y.; Tasaki, M.; Takahashi, K.; Hirano, T. Clinical Significance of the Pharmacological Efficacy of Tacrolimus Estimated by the Lymphocyte Immunosuppressant Sensitivity Test (LIST) Before and After Renal Transplantation. Cell Med. 2012, 3, 81–88. [Google Scholar] [CrossRef]
- Kurata, Y.; Kato, M.; Kuzuya, T.; Miwa, Y.; Iwasaki, K.; Haneda, M.; Amioka, K.; Watarai, Y.; Uchida, K.; Nakao, A.; et al. Pretransplant pharmacodynamic analysis of immunosuppressive agents using CFSE-based T-cell proliferation assay. Clin. Pharmacol. Ther. 2009, 86, 285–289. [Google Scholar] [CrossRef]
- Kurata, Y.; Kuzuya, T.; Miwa, Y.; Iwasaki, K.; Haneda, M.; Amioka, K.; Yamada, K.; Watarai, Y.; Katayama, A.; Uchida, K.; et al. Clinical relevance of post-transplant pharmacodynamic analysis of cyclosporine in renal transplantation. Int. Immunopharmacol. 2014, 22, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Mijiti, A.; Matsuno, N.; Takeuchi, H.; Unezaki, S.; Nagao, T.; Hirano, T. Clinical Significance of the Cellular Pharmacodynamics of Tacrolimus in Living-Donor Liver Transplantation. Cell Transplant. 2009, 18, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Kleiveland, C.R. Peripheral Blood Mononuclear Cells. In The Impact of Food Bioactives on Health: In Vitro and Ex Vivo Models; Verhoeckx, K., Cotter, P., Eds.; Springer: New York, NY, USA, 2015. [Google Scholar]
- Jimenez, C.; Portolés, J.; Crespo, M.; Rubio-Terrés, C.; Rubio-Rodríguez, D.; Diez, T.; Portero, I. Economic evaluation of the personalization of immunosuppressive therapy in kidney transplantation by means of an in vitro diagnostic test (Immunobiogram) in Spain. Rev. EsEco Salud. 2022, 17, 23–34. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pascual, J.; Crespo, M.; Portoles, J.; Jimenez, C.; Ortega-Carrion, A.; Diez, T.; Portero, I. The IMBG Test for Evaluating the Pharmacodynamic Response to Immunosuppressive Therapy in Kidney Transplant Patients: Current Evidence and Future Applications. Int. J. Mol. Sci. 2023, 24, 5201. https://doi.org/10.3390/ijms24065201
Pascual J, Crespo M, Portoles J, Jimenez C, Ortega-Carrion A, Diez T, Portero I. The IMBG Test for Evaluating the Pharmacodynamic Response to Immunosuppressive Therapy in Kidney Transplant Patients: Current Evidence and Future Applications. International Journal of Molecular Sciences. 2023; 24(6):5201. https://doi.org/10.3390/ijms24065201
Chicago/Turabian StylePascual, Julio, Marta Crespo, Jose Portoles, Carlos Jimenez, Alvaro Ortega-Carrion, Teresa Diez, and Isabel Portero. 2023. "The IMBG Test for Evaluating the Pharmacodynamic Response to Immunosuppressive Therapy in Kidney Transplant Patients: Current Evidence and Future Applications" International Journal of Molecular Sciences 24, no. 6: 5201. https://doi.org/10.3390/ijms24065201
APA StylePascual, J., Crespo, M., Portoles, J., Jimenez, C., Ortega-Carrion, A., Diez, T., & Portero, I. (2023). The IMBG Test for Evaluating the Pharmacodynamic Response to Immunosuppressive Therapy in Kidney Transplant Patients: Current Evidence and Future Applications. International Journal of Molecular Sciences, 24(6), 5201. https://doi.org/10.3390/ijms24065201





