Depression and Its Phytopharmacotherapy—A Narrative Review
Abstract
1. Introduction
2. Epidemiology
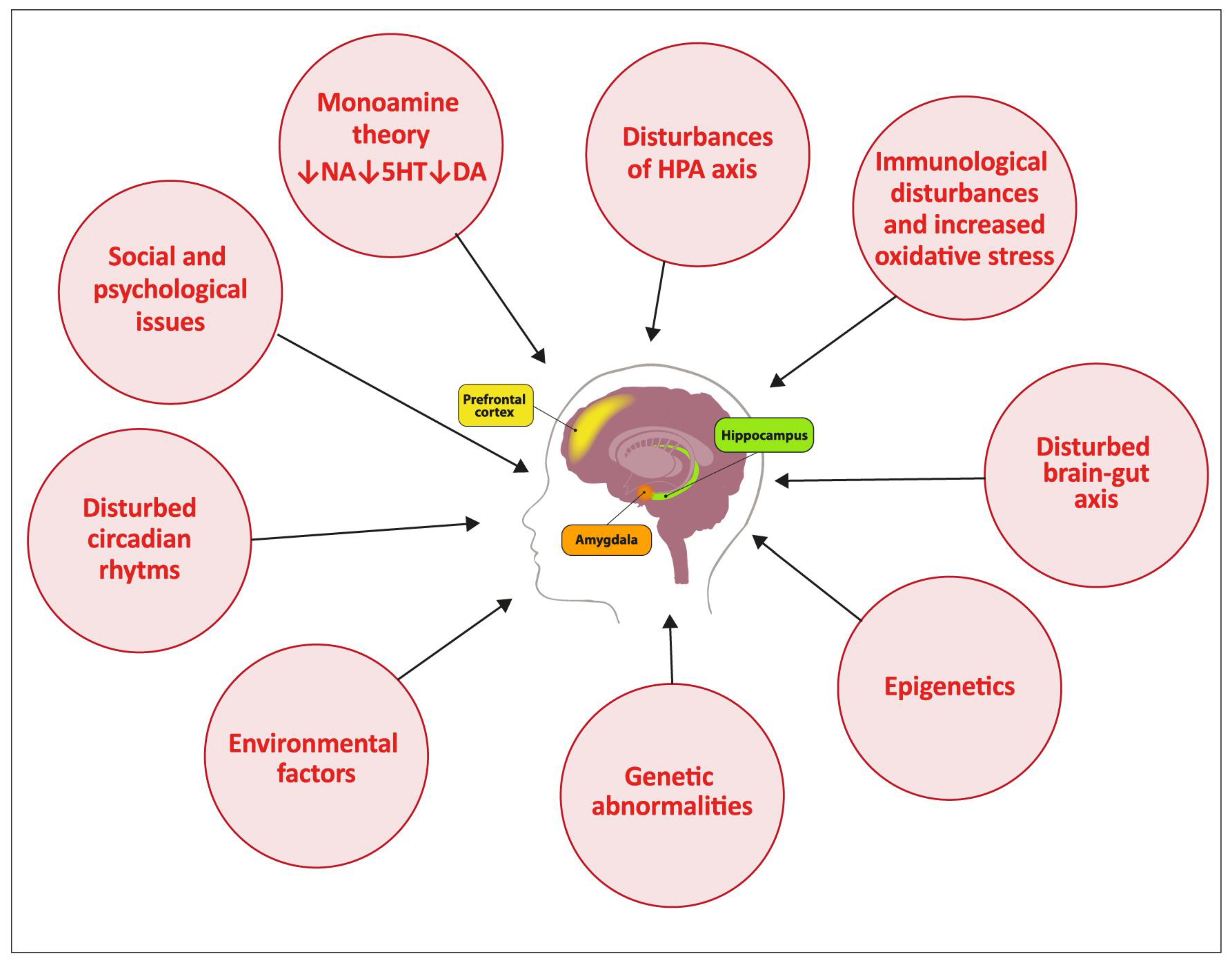
3. Pathophysiology
4. Symptomatology
5. Treatment—Pharmacotherapy and Non-Pharmacological Management of Depression
6. Depression Phytopharmacotherapy as an Alternative to Classical Antidepressant Treatment/Examples of Preparations and General Reasons for Their Antidepressant Effect
7. Side Effects of Herbal Antidepressants Discussed in This Review
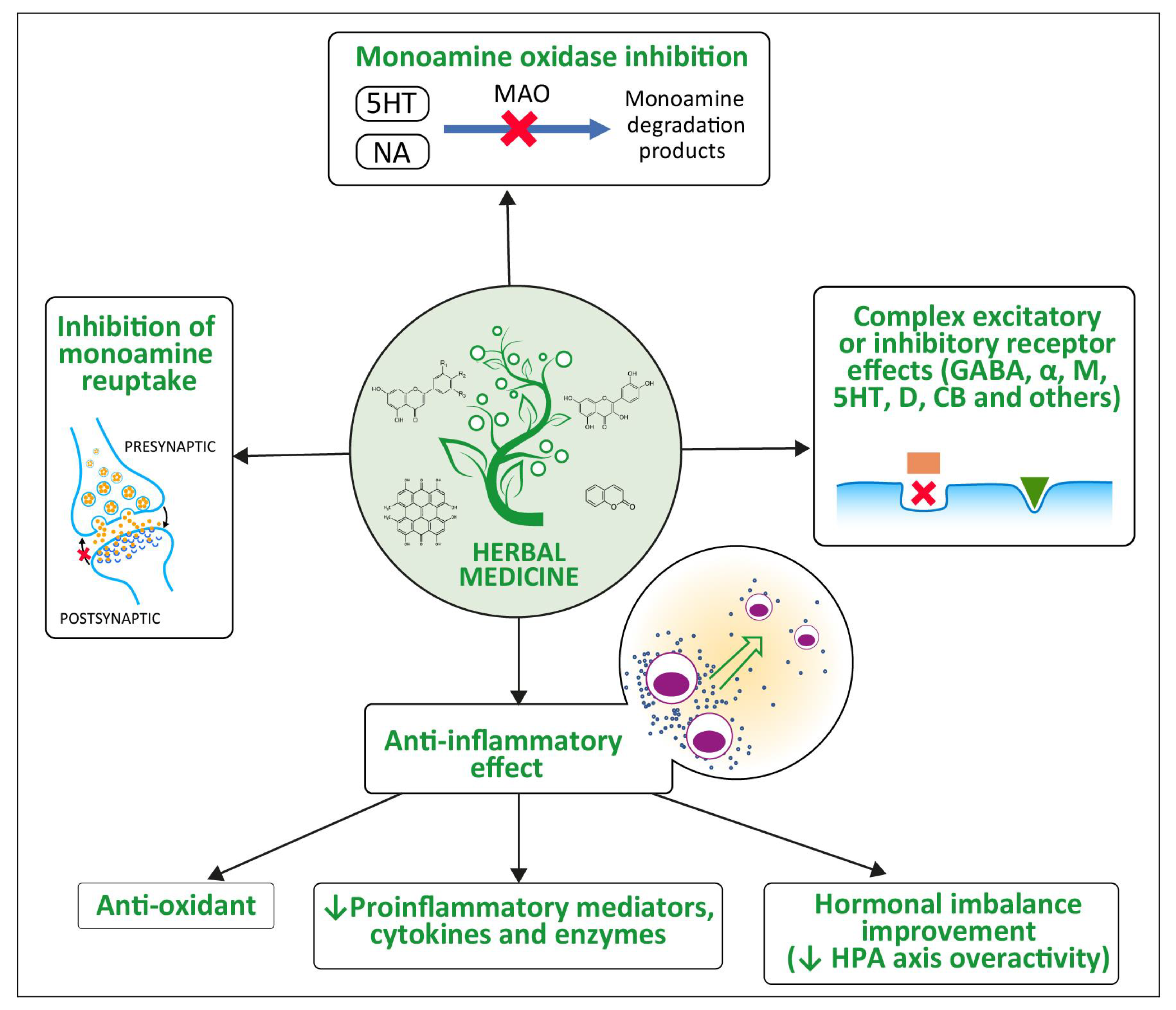
8. A Brief Description of the Phytopharmacodynamics of Plant-Derived Compounds with Antidepressant Activity with Particular Emphasis on Their Anti-Inflammatory Effect
9. Conclusions and Future Research Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5-HT | 5-hydroxytryptamine (serotonin) |
| ADRs | adverse drug reactions |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (glutamate receptors) |
| BDNF | brain derived neurotrophic factor |
| CNS | central nervous system |
| COX-2 | cyclooxygenase 2 |
| GABA | gamma aminobutyric acid |
| HPA | hypothalamic-pituitary-axis |
| SSRIs | selective serotonin reuptake inhibitors |
| Il | interleukin |
| INF-γ | interferon-γ |
| iNOS | inducible nitric oxide synthase |
| MAH | marketing authorization holder |
| MAOIs | monoamine oxidase inhibitors |
| MDD | major depressive disorder |
| NDRIs | noradrenaline and dopamine reuptake inhibitors |
| NE | noradrenaline (norepinephrine) |
| NF-κB | nuclear factor-kappa factor |
| NMDA | N-methyl-D-aspartate (glutamate receptors) |
| NRIs | noradrenaline reuptake inhibitors |
| OTC | over the counter (drugs) |
| OS | oxidative stress |
| SNRIs | selective noradrenaline reuptake inhibitors |
| SJW | St. John’s wort |
| TCAs | tricyclic antidepressants |
| TNF-α | tumour necrosis alpha |
| WHO | World Health Organization |
References
- Clack, S.; Ward, T. The classification and explanation of depression. Behav. Chang. 2019, 36, 41–55. [Google Scholar] [CrossRef]
- World Health Organisation. ICD-11 for Mortality and Morbidity Statistics. Version: 02/2022. Available online: https://icd.who.int/browse11/l-m/en (accessed on 20 November 2022).
- Pardhe, H.A.; Nagalakshmi, N.C.; Hariprasad, M.G.; Chourasia, P.K.; Nandini, S. A review: Medicinal plants with antidepressant properties. Indian J. Neurosci. 2020, 6, 1–5. [Google Scholar] [CrossRef]
- Martins, J.; Brijesh, S. Phytochemistry and pharmacology of anti-depressant medicinal plants: A review. Biomed. Pharmacother. 2018, 104, 343–365. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Depression. Available online: https://www.who.int/news-room/fact-sheets/detail/depression (accessed on 20 November 2022).
- Torre, J.; Vilagut, G.; Ronaldson, A.; Serrano-Blanco, A.; Martín, V.; Peters, M.; Valderas, J.; Dregan, A.; Alonso, J. Prevalence and variability of current depressive disorder in 27 European countries: A population-based study. Lancet Public Health 2021, 6, e729–e738. [Google Scholar] [CrossRef] [PubMed]
- Gałecki, P.; Samochowiec, J.; Mikułowska, M.; Szulc, A. Treatment-Resistant Depression in Poland—Epidemiology and Treatment. J. Clin. Med. 2022, 11, 480. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. National Center for Health Statistics. Anxiety and Depression. Available online: https://www.cdc.gov/nchs/covid19/pulse/mental-health.htm (accessed on 18 January 2023).
- Chiu, E. Epidemiology of depression in the Asia Pacific region. Australas Psychiatry 2004, 12, S4–S10. [Google Scholar] [CrossRef]
- Ogbo, F.A.; Mathsyaraja, S.; Koti, R.K.; Perz, J.; Page, A. The burden of depressive disorders in South Asia, 1990–2016: Findings from the global burden of disease study. BMC Psychiatry 2018, 18, 333. [Google Scholar] [CrossRef]
- Pan American Health Organization and World Health Organization. World Mental Health Day: Depression, the Most Common Mental Disorder. Available online: https://www3.paho.org/hq/index.php?option=com_content&view=article&id=7305:2012-dia-mundial-salud-mental-depresion-trastorno-mental-mas-frecuente&Itemid=0&lang=en#gsc.tab=0 (accessed on 18 January 2023).
- Cuadros, D.F.; Tomita, A.; Vandormael, A.; Slotow, R.; Burns, J.K.; Tanser, F. Spatial structure of depression in South Africa: A longitudinal panel survey of a nationally representative sample of households. Sci. Rep. 2019, 9, 979. [Google Scholar] [CrossRef]
- Bedaso, A.; Mekonnen, N.; Duko, B. Estimate of the prevalence of depression among older people in Africa: A systematic review and meta-analysis. Aging Ment. Health 2022, 26, 1095–1105. [Google Scholar] [CrossRef]
- Australian Institute of Health and Welfare. Prevalence and Impact of Mental Illness. Available online: https://www.aihw.gov.au/mental-health/overview/mental-illness#mental-disorder (accessed on 18 January 2023).
- Sarris, J.; Panossian, A.; Schweitzer, I.; Stough, C.; Scholey, A. Herbal medicine for depression, anxiety and insomnia: A review of psychopharmacology and clinical evidence. Eur. Neuropsychopharmacol. 2011, 21, 841–860. [Google Scholar] [CrossRef]
- Bernaras, E.; Jaureguizar, J.; Garaigordobil, M. Child and Adolescent Depression: A Review of theories, evaluation instruments, prevention programs, and treatments. Front. Psychol. 2019, 10, 543. [Google Scholar] [CrossRef]
- Deverteuil, R.L.; Lehmann, H.E. Therapeutic trial of iproniazid (marsilid) in depressed and apathetic patients. Can. Med. Assoc. J. 1958, 78, 131–133. [Google Scholar]
- Kuhn, R. The treatment of depressive states with G 22355 (imipramine hydrochloride). Am. J. Psychiatry 1958, 115, 459–464. [Google Scholar] [CrossRef]
- Schildkraut, J.J.; Kety, S.S. Biogenic amines and emotion. Science 1967, 156, 21–37. [Google Scholar] [CrossRef]
- Qin, D.D.; Rizak, J.; Feng, X.L.; Yang, S.C.; Lü, L.B.; Pan, L.; Yin, Y.; Hu, X.T. Prolonged secretion of cortisol as a possible mechanism underlying stress and depressive behaviour. Sci. Rep. 2016, 6, 30187. [Google Scholar] [CrossRef]
- Herbert, J. Cortisol and depression: Three questions for psychiatry. Psychol. Med. 2013, 43, 449–469. [Google Scholar] [CrossRef]
- Mikulska, J.; Juszczyk, G.; Gawrońska-Grzywacz, M.; Herbet, M. HPA Axis in the pathomechanism of depression and schizophrenia: New therapeutic strategies based on its participation. Brain Sci. 2021, 11, 1298. [Google Scholar] [CrossRef]
- Jesulola, E.; Micalos, P.; Baguley, I.J. Understanding the Pathophysiology of Depression: From Monoamines to the Neurogenesis Hypothesis Model—Are We There Yet? Behav. Brain Res. 2018, 341, 79–90. [Google Scholar]
- Lee, C.H.; Giuliani, F. The Role of Inflammation in Depression and Fatigue. Front. Immunol. 2019, 10, 1696. [Google Scholar] [CrossRef] [PubMed]
- Wohleb, E.S.; Franklin, T.; Iwata, M.; Duman, R.S. Integrating Neuroimmune Systems in the Neurobiology of Depression. Nat. Rev. Neurosci. 2016, 17, 497–511. [Google Scholar] [CrossRef]
- Beurel, E.; Toups, M.; Nemeroff, C.B. The bidirectional relationship of depression and inflammation: Double trouble. Neuron 2020, 107, 234–256. [Google Scholar] [CrossRef] [PubMed]
- Więdłocha, M.; Marcinowicz, P.; Krupa, R.; Janoska-Jaździk, M.; Janus, M.; Dębowska, W.; Mosiołek, A.; Waszkiewicz, N.; Szulc, A. Effect of antidepressant treatment on peripheral inflammation markers—A meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 80, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Savitz, J. Role of kynurenine metabolism pathway activation in major depressive disorders. Curr. Top. Behav. Neurosci. 2017, 31, 249–267. [Google Scholar] [PubMed]
- Berk, M.; Williams, L.J.; Jacka, F.N.; O’Neil, A.; Pasco, J.A.; Moylan, S.; Allen, N.B.; Stuart, A.L.; Hayley, A.C.; Byrne, M.L.; et al. So depression is an inflammatory disease, but where does the inflammation come from? BMC Med. 2013, 11, 200. [Google Scholar] [CrossRef] [PubMed]
- Salim, S. Oxidative stress and the central nervous system. J. Pharmacol. Exp. Ther. 2017, 360, 201–205. [Google Scholar] [CrossRef]
- Jimenez-Fernandez, S.; Gurpegui, M.; Diaz-Atienza, F.; Perez-Costillas, L.; Gerstenberg, M.; Correll, C.U. Oxidative stress and antioxidant parameters in patients with major depressive disorder compared to healthy controls before and after antidepressant treatment: Results from a meta-analysis. J. Clin. Psychiatry 2015, 76, 1658–1667. [Google Scholar] [CrossRef]
- Guilliams, T.G.; Edwards, L. Chronic stress and the HPA axis: Clinical assessment and therapeutic considerations. Standard 2010, 9, 1–12. [Google Scholar]
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The Gut-Brain Axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209. [Google Scholar]
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The Microbiota-Gut-Brain Axis. Physiol. Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef]
- Liang, S.; Wu, X.; Hu, X.; Wang, T.; Jin, F. Recognizing depression from the microbiota–gut–brain axis. Int. J. Mol. Sci. 2018, 19, 1592. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Gómez, A.M.; Orozco, A.; Lahera, G.; Sosa, M.D.; Diaz, D.; Auba, E.; Albillos, A.; Monserrat, J.; Alvarez-Mon, M. Abnormal distribution and function of circulating monocytes and enhanced bacterial translocation in major depressive disorder. Front. Psychiatry 2019, 10, 812. [Google Scholar] [CrossRef]
- Strandwitz, P. Neurotransmitter modulation by the gut microbiota. Brain Res. 2018, 1693, 128. [Google Scholar] [CrossRef]
- Bromberger, J.T.; Schott, L.L.; Kravitz, H.M.; Sowers, M.; Avis, N.E.; Gold, E.B.; Randolph, J.F., Jr.; Matthews, K.A. Longitudinal change in reproductive hormones and depressive symptoms across the menopausal transition: Results from the Study of Women’s Health Across the Nation (SWAN). Arch. Gen. Psychiatry 2010, 67, 598–607. [Google Scholar] [CrossRef]
- Soares, C.N.; Zitek, B. Reproductive hormone sensitivity and risk for depression across the female life cycle: A continuum of vulnerability? J. Psychiatry Neurosci. 2008, 33, 331–343. [Google Scholar]
- Sullivan, P.F.; Neale, M.C.; Kendler, K.S. Genetic epidemiology of major depression: Review and meta-analysis. Am. J. Psychiatry 2000, 157, 1552–1562. [Google Scholar] [CrossRef]
- Menke, A.; Klengel, T.; Binder, E.B. Epigenetics, depression and antidepressant treatment. Curr. Pharm. Des. 2012, 18, 5879–5889. [Google Scholar] [CrossRef]
- Krishnan, V.; Nestler, E.J. The molecular neurobiology of depression. Nature 2008, 455, 894–902. [Google Scholar] [CrossRef]
- Monteggia, L.M.; Luikart, B.; Barrot, M.; Theobold, D.; Malkovska, I.; Nef, S.; Parada, L.F.; Nestler, E.J. Brain-derived neurotrophic factor conditional knockouts show gender differences in depression-related behaviors. Biol. Psychiatry 2007, 15, 187–197. [Google Scholar] [CrossRef]
- Eisch, A.J.; Bolaños, C.A.; de Wit, J.; Simonak, R.D.; Pudiak, C.M.; Barrot, M.; Verhaagen, J.; Nestler, E.J. Brain-derived neurotrophic factor in the ventral midbrain-nucleus accumbens pathway: A role in depression. Biol. Psychiatry. 2003, 54, 994–1005. [Google Scholar] [CrossRef]
- Duman, R.S.; Voleti, B. Signaling pathways underlying the pathophysiology and treatment of depression: Novel mechanisms for rapid-acting agents. Trends Neurosci. 2012, 35, 47–56. [Google Scholar] [CrossRef]
- Shadrina, M.; Bondarenko, E.A.; Slominsky, P.A. Genetics factors in major depression disease. Front. Psychiatry 2018, 9, 334. [Google Scholar] [CrossRef] [PubMed]
- Lohoff, F.W. Overview of the genetics of major depressive disorder. Curr. Psychiatry Rep. 2010, 12, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Mariani, N.; Cattane, N.; Pariante, C.; Cattaneo, A. Gene expression studies in depression development and treatment: An overview of the underlying molecular mechanisms and biological processes to identify biomarkers. Transl. Psychiatry 2021, 11, 354. [Google Scholar] [CrossRef] [PubMed]
- Dupont, C.; Armant, D.R.; Brenner, C.A. Epigenetics: Definition, mechanisms and clinical perspective. Semin. Reprod. Med. 2009, 27, 351–357. [Google Scholar] [CrossRef]
- Al Aboud, N.M.; Tupper, C.; Jialal, I. Genetics, Epigenetic Mechanism. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK532999/ (accessed on 22 January 2023).
- Penner-Goeke, S.; Binder, E.B. Epigenetics and depression. Dialogues Clin. Neurosci. 2019, 21, 397–405. [Google Scholar] [CrossRef]
- Czarny, P.; Białek, K.; Ziółkowska, S.; Strycharz, J.; Barszczewska, G.; Sliwinski, T. The importance of epigenetics in diagnostics and treatment of major depressive disorder. J. Pers. Med. 2021, 11, 167. [Google Scholar] [CrossRef]
- Blatt, S.J. Experiences of Depression: Theoretical, Clinical, and Research Perspectives; American Psychological Association: Worcester, MA, USA, 2004. [Google Scholar] [CrossRef]
- Tonon, A.C.; Pilz, L.K.; Markus, R.P.; Hidalgo, M.P.; Elisabetsky., E. Melatonin and depression: A translational perspective from animal models to clinical studies. Front. Psychiatry 2021, 12, 638981. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Jiang, Y.J.; Zou, M.S.; Liu, J.; Zhao, H.Q.; Wang, Y.H. Antidepressant actions of melatonin and melatonin receptor agonist: Focus on pathophysiology and treatment. Behav. Brain Res. 2022, 420, 113724. [Google Scholar] [CrossRef]
- González-Díaz, S.N.; Arias-Cruz, A.; Elizondo-Villarreal, B.; Monge-Ortega, O.P. Psychoneuroimmunoendocrinology: Clinical implications. World Allergy Organ. J. 2017, 10, 19. [Google Scholar] [CrossRef]
- Singh, T.; Williams, K. Atypical depression. Psychiatry 2006, 3, 33–39. [Google Scholar]
- Munro, M.; Milne, R. Symptoms and causes of depression, and its diagnosis and management. Nurs. Times 2020, 116, 18–22. [Google Scholar]
- Kennedy, S.H. Core symptoms of major depressive disorder: Relevance to diagnosis and treatment. Dialogues Clin. Neurosci. 2008, 10, 271–277. [Google Scholar] [CrossRef]
- Bhowmik, D.; Kumar, K.P.S.; Srivastava, S.; Paswan, S.; Dutta, A.S. Depression-symptoms, causes, medications and therapies. Pharma Innov. 2012, 1, 32–45. [Google Scholar]
- Kuria, M.W.; Ndetei, D.M.; Obot, I.S.; Khasakhala, L.I.; Bagaka, B.M.; Mbugua, M.N.; Kamau, J. The association between alcohol dependence and depression before and after treatment for alcohol dependence. ISRN Psychiatry 2012, 2012, 482802. [Google Scholar] [CrossRef]
- Timonen, M.; Liukkonen, T. Management of depression in adults. BMJ 2008, 336, 435–439. [Google Scholar] [CrossRef]
- Bleakley, S. Review of choice and use of antidepressants. Prog. Neurol. Psychiatry 2009, 13, 14–20. [Google Scholar] [CrossRef]
- Faquih, A.E.; Memon, R.I.; Hafeez, H.; Zeshan, M.; Naveed, S. A review of novel antidepressants: A guide for clinicians. Cureus 2019, 11, e4185. [Google Scholar] [CrossRef]
- Cipriani, A.; Furukawa, T.A.; Salanti, G.; Chaimani, A.; Atkinson, L.Z.; Ogawa, Y.; Leucht, S.; Ruhe, H.G.; Turner, E.H.; Higgins, J.P.T.; et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: A systematic review and network meta-analysis. Lancet 2018, 391, 1357–1366. [Google Scholar] [CrossRef]
- Ramic, E.; Prasko, S.; Gavran, L.; Spahic, E. Assessment of the antidepressant side effects occurrence in patients treated in primary care. Mater. Sociomed. 2020, 32, 131–134. [Google Scholar] [CrossRef]
- Edinoff, A.N.; Akuly, H.A.; Hanna, T.A.; Ochoa, C.O.; Patti, S.J.; Ghaffar, Y.A.; Kaye, A.D.; Viswanath, O.; Urits, I.; Boyer, A.G.; et al. Selective Serotonin Reuptake Inhibitors and Adverse Effects: A Narrative Review. Neurol. Int. 2021, 13, 387–401. [Google Scholar] [CrossRef]
- Moraczewski, J.; Aedma, K.K. Tricyclic Antidepressants. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK557791 (accessed on 25 January 2023).
- Gautam, S.; Jain, A.; Gautam, M.; Vahia, V.N.; Grover, S. Clinical practice guidelines for the management of depression. Indian J. Psychiatry 2017, 59, S34–S50. [Google Scholar]
- Practice Guideline for the Treatment of Patients with Major Depressive Disorder. Available online: https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/mdd.pdf (accessed on 2 December 2022).
- Almeida, S.S.; Zizzi, F.B.; Cattaneo, A.; Comandini, A.; Di Dato, G.; Lubrano, E.; Pellicano, C.; Spallone, V.; Tongiani, S.; Torta, R. Management and treatment of patients with major depressive disorder and chronic diseases: A multidisciplinary approach. Front. Psychol. 2020, 11, 542444. [Google Scholar] [CrossRef] [PubMed]
- American Psychological Association. Clinical Practice Guideline for the Treatment of Depression across Three Age Cohorts. 2019. Available online: https://www.apa.org/depression-guideline (accessed on 2 December 2022).
- Samochowiec, J.; Dudek, D.; Mazur, J.K.; Murawiec, S.; Rymaszewska, J.; Cubała, W.J.; Heitzman, J.; Szulc, A.; Bała, M.; Gałecki, P. Pharmacological treatment of a depressive episode and recurrent depressive disorder—Guidelines of the Polish Psychiatric Association and the National Consultant for Adult Psychiatry. Psychiatry Pol. 2021, 55, 235–259. [Google Scholar] [CrossRef] [PubMed]
- Farah, W.H.; Alsawas, M.; Mainou, M.; Alahdab, F.; Farah, M.H.; Ahmed, A.T.; Mohamed, E.A.; Almasri, J.; Gionfriddo, M.R.; Castaneda-Guarderas, A.; et al. Non-pharmacological treatment of depression: A systematic review and evidence map. Evid. Based Med. 2016, 21, 214–221. [Google Scholar] [CrossRef]
- Gautam, M.; Tripathi, A.; Deshmukh, D.; Gaur, M. Cognitive behavioral therapy for depression. Indian J. Psychiatry 2020, 62, S223–S229. [Google Scholar] [CrossRef] [PubMed]
- Thurfah, J.N.; Christine; Bagaskhara, P.P.; Alfian, S.D.; Puspitasari, I.M. Dietary supplementations and depression. J. Multidiscip. Healthc. 2022, 15, 1121–1141. [Google Scholar]
- Ortega, M.A.; Fraile-Martínez, Ó.; García-Montero, C.; Alvarez-Mon, M.A.; Lahera, G.; Monserrat, J.; Llavero-Valero, M.; Gutiérrez-Rojas, L.; Molina, R.; Rodríguez-Jimenez, R.; et al. Biological role of nutrients, food and dietary patterns in the prevention and clinical management of major depressive disorder. Nutrients 2022, 14, 3099. [Google Scholar] [CrossRef]
- Rizvi, S.; Khan, A.M. Use of transcranial magnetic stimulation for depression. Cureus 2019, 11, e4736. [Google Scholar] [CrossRef]
- Porter, R.; Linsley, K.; Ferrier, N. Treatment of severe depression—Non-pharmacological aspects. Adv. Psychiatr. Treat. 2001, 7, 117–124. [Google Scholar] [CrossRef]
- Chen, J.J.; Zhao, L.B.; Liu, Y.Y.; Fan, S.H.; Xie, P. Comparative efficacy and acceptability of electroconvulsive therapy versus repetitive transcranial magnetic stimulation for major depression: A systematic review and multiple-treatments meta-analysis. Behav. Brain Res. 2017, 320, 30–36. [Google Scholar] [CrossRef]
- Micallef-Trigona, B. Comparing the effects of repetitive transcranial magnetic stimulation and electroconvulsive therapy in the treatment of depression: A systematic review and meta-analysis. Depress Res. Treat. 2014, 2014, 135049. [Google Scholar] [CrossRef]
- Iacobucci, G. NHS prescribed record number of antidepressants last year. BMJ 2019, 364, l1508. [Google Scholar] [CrossRef]
- Pratt, L.A.; Brody, D.J.; Gu, Q. Antidepressant use among persons aged 12 and over: United States, 2011–2014. CHS Data Brief 2017, 283, 1–8. [Google Scholar]
- Reinert, M.; Fritze, D.; Nguyen, T. The State of Mental Health in America 2022. Mental Health America: Alexandria, VA, USA. Available online: https://mhanational.org/sites/default/files/2022%20State%20of%20Mental%20Health%20in%20America.pdf (accessed on 2 December 2022).
- Lewer, D.; O’Reilly, C.; Mojtabai, R.; Evans-Lacko, S. Antidepressant use in 27 European countries: Associations with sociodemographic, cultural and economic factors. Br. J. Psychiatry 2015, 207, 221–226. [Google Scholar] [CrossRef]
- Sisay, T.; Wami, R. Adverse drug reactions among major depressive disorders: Patterns by age and gender. Heliyon 2021, 7, e08655. [Google Scholar] [CrossRef]
- Degner, D.; Grohmann, R.; Kropp, S.; Rüther, E.; Bender, S.; Engel, R.R.; Schmidt, L.G. Severe adverse drug reactions of antidepressants: Results of the German multicenter drug surveillance program AMSP. Pharmacopsychiatry 2004, 37, S39–S45. [Google Scholar]
- Uher, R.; Farmer, A.; Henigsberg, N.; Rietschel, M.; Mors, O.; Maier, W.; Kozel, D.; Hauser, J.; Souery, D.; Placentino, A.; et al. Adverse reactions to antidepressants. Br. J. Psychiatry 2009, 195, 202–210. [Google Scholar] [CrossRef]
- Jiao, Y.; Wickett, N.J.; Ayyampalayam, S.; Chanderbali, A.S.; Landherr, L.; Ralph, P.E.; Tomsho, L.P.; Hu, Y.; Liang, H.; Soltis, P.S.; et al. Ancestral polyploidy in seed plants and angiosperms. Nature 2011, 473, 97–100. [Google Scholar] [CrossRef]
- Sahoo, S. A review of some medicinal plants used for nervous system. J. Med. Plants Stud. 2018, 6, 220–224. [Google Scholar]
- Ulrich-Merzenich, G.; Panek, D.; Zeitler, H.; Vetter, H.; Wagner, H. Drug development from natural products: Exploiting synergistic effects. Indian J. Exp. Biol. 2010, 48, 208–219. [Google Scholar]
- Wagner, H.; Ulrich-Merzenich, G. Synergy research: Approaching a new generation of phytopharmaceuticals. Phytomedicine 2009, 16, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Wink, M. Modes of action of herbal medicines and plant secondary metabolites. Medicines 2015, 2, 251–286. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; Soukup, J.; Davis, R.B.; Foster, D.F.; Wilkey, S.A.; Van Rompay, M.I.; Eisenberg, D.M. The use of complementary and alternative therapies to treat anxiety and depression in the United States. Am. J. Psychiatry 2001, 158, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Elkins, G.; Rajab, M.H.; Marcus, J. Complementary and alternative medicine use by psychiatric inpatients. Psychol. Rep. 2005, 96, 163–166. [Google Scholar] [CrossRef]
- Kumar, V. Potential medicinal plants for CNS disorders: An overview. Phytother Res 2006, 20, 1023–1035. [Google Scholar] [CrossRef]
- Peschel, W. The use of community herbal monographs to facilitate registrations and authorisations of herbal medicinal products in the European Union 2004–2012. J. Ethnopharmacol. 2014, 158, 471–486. [Google Scholar] [CrossRef]
- Moreira, D.L.; Teixeira, S.S.; Monteiro, M.H.D.; de-Oliveira, A.C.A.X.; Paumgartten, F.J.R. Traditional use and safety of herbal medicines. Rev. Bras. Farmacogn. 2014, 24, 248–257. [Google Scholar] [CrossRef]
- Papakostas, G.I. The efficacy, tolerability, and safety of contemporary antidepressants. J. Clin. Psychiatry 2010, 71, e03. [Google Scholar] [CrossRef]
- Baldwin, D.S.; Montgomery, S.A.; Nil, R.; Lader, M. Discontinuation symptoms in depression and anxiety disorders. Int. J. Neuropsychopharmacol. 2007, 10, 73–84. [Google Scholar] [CrossRef]
- Wilson, E.; Lader, M. A review of the management of antidepressant discontinuation symptoms. Ther. Adv. Psychopharmacol. 2015, 5, 357–368. [Google Scholar] [CrossRef]
- Alvarez-Mon, M.A.; Ortega, M.A.; García-Montero, C.; Fraile-Martinez, O.; Monserrat, J.; Lahera, G.; Mora, F.; Rodriguez-Quiroga, A.; Fernandez-Rojo, S.; Quintero, J.; et al. Exploring the role of nutraceuticals in major depressive disorder (MDD): Rationale, state of the art and future prospects. Pharmaceuticals 2021, 14, 821. [Google Scholar] [CrossRef]
- Wang, Y.; Su, C.; Zhang, B.; Niu, Y.; Ren, R.; Zhao, X.; Yang, L.; Zhang, W.; Ma, X. Biological activity, hepatotoxicity, and structure-activity relationship of kavalactones and flavokavins, the two main bioactive components in kava (Piper methysticum). Evid. Based Complement. Altern. Med. 2021, 2021, 6851798. [Google Scholar] [CrossRef]
- Staines, S.S. Herbal medicines: Adverse effects and drug-herbs interactions. J. Malta Coll. Pharm. Pract. 2011, 17, 38–42. [Google Scholar]
- Henderson, L.; Yue, Q.Y.; Bergquist, C.; Gerden, B.; Arlett, P. St. John’s wort (Hypericum perforatum): Drug interactions and clinical outcomes. Br. J. Clin. Pharmacol. 2002, 54, 349–356. [Google Scholar] [CrossRef]
- Canenguez Benitez, J.S.; Hernandez, T.E.; Sundararajan, R.; Sarwar, S.; Arriaga, A.J.; Khan, A.T.; Matayoshi, A.; Quintanilla, H.A.; Kochhar, H.; Alam, M.; et al. Advantages and disadvantages of using St. John’s Wort as a treatment for depression. Cureus 2022, 14, e29468. [Google Scholar] [CrossRef]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef]
- Hammerness, P.; Basch, E.; Ulbricht, C.; Barrette, P.; Foppa, I.; Basch, S.; Bent, S.; Boon, H.; Ernst, E. Natural Standard Research Collaboration. St John’s wort: A systematic review of adverse effects and drug interactions for the consultation psychiatrist. Psychosomatics 2003, 44, 271–282. [Google Scholar] [CrossRef]
- Joshi, K.G.; Faubion, M.D. Mania and psychosis associated with St. John’s wort and ginseng. Psychiatry 2005, 2, 56–61. [Google Scholar]
- Nicolussi, S.; Drewe, J.; Butterweck, V.; Zu Schwabedissen, H.E.M. Clinical relevance of St. John’s wort drug interactions revisited. Br. J. Pharmacol. 2020, 177, 1212–1226. [Google Scholar] [CrossRef]
- Grimstein, M.; Huang, S.M. A regulatory science viewpoint on botanical-drug interactions. J. Food Drug Anal. 2018, 26, S12–S25. [Google Scholar] [CrossRef]
- Moore, L.B.; Goodwin, B.; Jones, S.A.; Wisely, G.B.; Serabjit-Singh, C.J.; Willson, T.M.; Collins, J.L.; Kliewer, S.A. St. John’s wort induces hepatic drug metabolism through activation of the pregnane X receptor. Proc. Natl. Acad. Sci. USA 2000, 97, 7500–7502. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, J.S.; Donovan, J.L.; DeVane, C.L.; Taylor, R.M.; Ruan, Y.; Wang, J.S.; Chavin, K.D. Effect of St John’s wort on drug metabolism by induction of cytochrome P450 3A4 enzyme. JAMA 2003, 290, 1500–1504. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, B.; Loos, B.; Scriba, T.J.; Lang, D.; Davids, L.M. St. John’s wort (Hypericum perforatum L.) photomedicine: Hypericin-photodynamic therapy induces metastatic melanoma cell death. PLoS ONE 2014, 9, e103762. [Google Scholar] [CrossRef] [PubMed]
- Vollmer, J.J.; Rosenson, J. Chemistry of St. John’s wort: Hypericin and hyperforin. J. Chem. Educ. 2004, 81, 1450. [Google Scholar] [CrossRef]
- Moshiri, M.; Vahabzadeh, M.; Hosseinzadeh, H. Clinical applications of saffron (Crocus sativus) and its constituents: A review. Drug Res. 2015, 65, 287–295. [Google Scholar] [CrossRef]
- Barnes, J. Saffron. J. Prim. Health Care 2022, 14, 189–190. [Google Scholar] [CrossRef]
- Zam, W.; Quispe, C.; Sharifi-Rad, J.; López, M.D.; Schoebitz, M.; Martorell, M.; Sharopov, F.; Fokou, P.V.T.; Mishra, A.P.; Chandran, D.; et al. An updated review on the properties of Melissa officinalis L.: Not exclusively anti-anxiety. Front. Biosci. 2022, 14, 16. [Google Scholar] [CrossRef]
- Block, K.I.; Gyllenhaal, C.; Mead, M.N. Safety and efficacy of herbal sedatives in cancer care. Integr. Cancer Ther. 2004, 3, 128–148. [Google Scholar] [CrossRef]
- Unger, M. Pharmacokinetic drug interactions involving Ginkgo biloba. Drug Metab. Rev. 2013, 45, 353–385. [Google Scholar] [CrossRef]
- Nguyen, T.; Alzahrani, T. Ginkgo Biloba. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK541024/ (accessed on 23 February 2023).
- Sadler, C.; Charrois, T.L.; Vohra, S. Gingko biloba: Practical management of adverse effects and drug interactions. Canadian Pharm. J. Rev. Pharm. Can. 2006, 139, 32–35. [Google Scholar] [CrossRef]
- Kiefer, D.; Pantuso, T. Panax ginseng. Am. Fam. Phys. 2003, 68, 1539–1542. [Google Scholar]
- Tao, H.; Wu, X.; Cao, J.; Peng, Y.; Wang, A.; Pei, J.; Xiao, J.; Wang, S.; Wang, Y. Rhodiola species: A comprehensive review of traditional use, phytochemistry, pharmacology, toxicity, and clinical study. Med. Res. Rev. 2019, 39, 1779–1850. [Google Scholar] [CrossRef]
- Sarrica, A.; Kirika, N.; Romeo, M.; Salmona, M.; Diomede, L. Safety and toxicology of magnolol and honokiol. Planta Med. 2018, 84, 1151–1164. [Google Scholar] [CrossRef]
- Sayyah, M.; Siahpoosh, A.; Khalili, H.; Malayeri, A.; Samaee, H. A double-blind, placebo-controlled study of the aqueous extract of Echium amoenum for patients with general anxiety disorder. Iran J. Pharm. Res. 2012, 11, 697–701. [Google Scholar]
- Peth-Nui, T.; Wattanathorn, J.; Muchimapura, S.; Tong-Un, T.; Piyavhatkul, N.; Rangseekajee, P.; Ingkaninan, K.; Vittaya-Areekul, S. Effects of 12-week Bacopa monnieri consumption on attention, cognitive processing, working memory, and functions of both cholinergic and monoaminergic systems in healthy elderly volunteers. Evid. Based Complement. Alternat. Med. 2012, 2012, 606424. [Google Scholar] [CrossRef]
- Sanaye, M.M.; Joglekar, C.S.; Pagare, N.P. Mimosa—A brief overview. J. Pharmacogn. Phytochem. 2015, 4, 182–187. [Google Scholar]
- Wang, Y.S.; Shen, C.Y.; Jiang, J.G. Antidepressant active ingredients from herbs and nutraceuticals used in TCM: Pharmacological mechanisms and prospects for drug discovery. Pharmacol. Res. 2019, 150, 104520. [Google Scholar] [CrossRef]
- Farahani, M.S.; Bahramsoltani, R.; Farzaei, M.H.; Abdollahi, M.; Rahimi, R. Plant-derived natural medicines for the management of depression: An overview of mechanisms of action. Rev. Neurosci. 2015, 26, 305–321. [Google Scholar] [CrossRef]
- Zhang, Z.; Deng, T.; Wu, M.; Zhu, A.; Zhu, G. Botanicals as modulators of depression and mechanisms involved. Chin. Med. 2019, 14, 24. [Google Scholar] [CrossRef]
- Lundstrom, K.; Pham, H.T.; Dinh, L.D. Interaction of plant extracts with central nervous system receptors. Medicines 2017, 4, 12. [Google Scholar] [CrossRef]
- Parveen, A.; Parveen, B.; Parveen, R.; Ahmad, S. Challenges and guidelines for clinical trial of herbal drugs. J. Pharm. Bioallied Sci. 2015, 7, 329–333. [Google Scholar] [PubMed]
- Jachak, S.M.; Saklani, A. Challenges and opportunities in drug discovery from plants. Curr. Sci. 2007, 92, 1251–1257. [Google Scholar]
- Williams, R.; Münch, G.; Gyengesi, E.; Bennett, L. Bacopa monnieri (L.) exerts anti-inflammatory effects on cells of the innate immune system in vitro. Food Funct. 2014, 5, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Nemetchek, M.D.; Stierle, A.A.; Stierle, D.B.; Lurie, D.I. The Ayurvedic plant Bacopa monnieri inhibits inflammatory pathways in the brain. J. Ethnopharmacol. 2017, 197, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Mathur, A.S.; Verma, S.K.; Purohit, R.; Singh, S.K.; Mathur, D.; Prasad, G.K.; Dua, V.K. Pharmacological investigation of Bacopa monnieri on the basis of antioxidant, antimicrobial and anti-inflammatory properties. J. Chem. Pharm. Res. 2010, 2, 191–198. [Google Scholar]
- Rai, K.; Gupta, N.S.; Dharamdasani, L.; Nair, P.; Bodhankar, P. Bacopa monnieri: A wonder drug changing fortune of people. IJASBT 2017, 5, 127–132. [Google Scholar] [CrossRef]
- Bhattacharya, S.K.; Bhattacharya, A.; Kumar, A.; Ghosal, S. Antioxidant activity of Bacopa monniera in rat frontal cortex, striatum and hippocampus. Phytother. Res. 2000, 14, 174–179. [Google Scholar] [CrossRef]
- Jin, J.; Boersch, M.; Nagarajan, A.; Davey, A.K.; Zunk, M. Antioxidant properties and reported ethnomedicinal use of the genus Echium (Boraginaceae). Antioxidants 2020, 9, 722. [Google Scholar] [CrossRef]
- Naseri, N.; Kalantar, K.; Amirghofran, Z. Anti-inflammatory activity of Echium amoenum extract on macrophages mediated by inhibition of inflammatory mediators and cytokines expression. Res. Pharm. Sci. 2018, 13, 73–81. [Google Scholar]
- Das Noor-E-Tabassum, R.; Lami, M.S.; Chakraborty, A.J.; Mitra, S.; Tallei, T.E.; Idroes, R.; Mohamed, A.A.; Hossain, M.J.; Dhama, K.; Mostafa-Hedeab, G.; et al. Ginkgo biloba: A treasure of functional phytochemicals with multimedicinal applications. Evid. Based Complement. Alternat. Med. 2022, 2022, 8288818. [Google Scholar] [CrossRef]
- Kaur, S.; Sharma, N.; Nehru, B. Anti-inflammatory effects of Ginkgo biloba extract against trimethyltin-induced hippocampal neuronal injury. Inflammopharmacology 2018, 26, 87–104. [Google Scholar] [CrossRef]
- Gargouri, B.; Carstensen, J.; Bhatia, H.S.; Huell, M.; Dietz, G.P.H.; Fiebich, B.L. Anti-neuroinflammatory effects of Ginkgo biloba extract EGb761 in LPS-activated primary microglial cells. Phytomedicine 2018, 44, 45–55. [Google Scholar] [CrossRef]
- Li, M.; Li, B.; Hou, Y.; Tian, Y.; Chen, L.; Liu, S.; Zhang, N.; Dong, J. Anti-inflammatory effects of chemical components from Ginkgo biloba L. male flowers on lipopolysaccharide-stimulated RAW264.7 macrophages. Phytother. Res. 2019, 33, 989–997. [Google Scholar] [CrossRef]
- Piazza, S.; Pacchetti, B.; Fumagalli, M.; Bonacina, F.; Dell’Agli, M.; Sangiovanni, E. Comparison of two Ginkgo biloba L. extracts on oxidative stress and inflammation markers in human endothelial cells. Mediat. Inflamm. 2019, 2019, 6173893. [Google Scholar] [CrossRef]
- Barbalho, S.M.; Direito, R.; Laurindo, L.F.; Marton, L.T.; Guiguer, E.L.; Goulart, R.A.; Tofano, R.J.; Carvalho, A.C.A.; Flato, U.A.P.; Tofano, V.A.C.; et al. Ginkgo biloba in the aging process: A narrative review. Antioxidants 2022, 11, 525. [Google Scholar] [CrossRef]
- An, M.Y.; Lee, S.R.; Hwang, H.J.; Yoon, J.G.; Lee, H.J.; Cho, J.A. Antioxidant and anti-inflammatory effects of korean black ginseng extract through ER stress pathway. Antioxidants 2021, 10, 62. [Google Scholar] [CrossRef]
- Park, J.; Cho, J.Y. Anti-inflammatory effects of ginsenosides from Panax ginseng and their structural analogs. Afr. J. Biotechnol. 2009, 8, 3682–3690. [Google Scholar]
- Pandur, E.; Balatinácz, A.; Micalizzi, G.; Mondello, L.; Horváth, A.; Sipos, K.; Horváth, G. Anti-inflammatory effect of lavender (Lavandula angustifolia Mill.) essential oil prepared during different plant phenophases on THP-1 macrophages. BMC Complement. Med. Ther. 2021, 21, 287. [Google Scholar] [CrossRef]
- Silva, G.L.; Luft, C.; Lunardelli, A.; Amaral, R.H.; Melo, D.A.; Donadio, M.V.; Nunes, F.B.; de Azambuja, M.S.; Santana, J.C.; Moraes, C.M.; et al. Antioxidant, analgesic and anti-inflammatory effects of lavender essential oil. An. Acad. Bras. Cienc. 2015, 87, 1397–1408. [Google Scholar] [CrossRef]
- Cardia, G.F.; Silva-Filho, S.E.; Silva, E.L.; Uchida, N.S.; Cavalcante, H.A.; Cassarotti, L.L.; Salvadego, V.E.; Spironello, R.A.; Bersani-Amado, C.A.; Cuman, R.K. Effect of lavender (Lavandula angustifolia) essential oil on acute inflammatory response. Evid. Based Complement. Alternat. Med. 2018, 2018, 1413940. [Google Scholar] [CrossRef]
- Świąder, K.; Startek, K.; Wijaya, C.H. The therapeutic properties of lemon balm (Melissa officinalis L.): Reviewing novel findings and medical indications. J. Appl. Bot. Food Qual. 2019, 92, 327–335. [Google Scholar]
- Draginic, N.; Andjic, M.; Jeremic, J.; Zivkovic, V.; Kocovic, A.; Tomovic, M.; Bozin, B.; Kladar, N.; Bolevich, S.; Jakovljevic, V.; et al. Anti-inflammatory and antioxidant effects of Melissa officinalis extracts: A comparative study. Iran J. Pharm. Res. 2022, 21, e126561. [Google Scholar] [CrossRef] [PubMed]
- Miraj, S.; Rafieian-Kopaei; Kiani, S. Melissa officinalis L: A review study with an antioxidant prospective. J. Evid. Based Complement. Altern. Med. 2017, 22, 385–394. [Google Scholar]
- Eliaz, I. Honokiol research review. A promising extract with multiple applications. Nat. Med. J. 2014. Available online: https://www.naturalmedicinejournal.com/journal/honokiol-research-review (accessed on 27 January 2023).
- Walker, J.M.; Maitra, A.; Walker, J.; Ehrnhoefer-Ressler, M.M.; Inui, T.; Somoza, V. Identification of Magnolia officinalis L. bark extract as the most potent anti-inflammatory of four plant extracts. Am. J. Chin. Med. 2013, 41, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jung, E.; Park, J.; Jung, K.; Lee, S.; Hong, S.; Park, J.; Park, E.; Kim, J.; Park, S.; et al. Anti-inflammatory effects of magnolol and honokiol are mediated through inhibition of the downstream pathway of MEKK-1 in NF-kappaB activation signaling. Planta Med. 2005, 71, 338–343. [Google Scholar] [CrossRef]
- Ramachandran, C.; Wilk, B.; Melnick, S.J.; Eliaz, I. Synergistic antioxidant and anti-inflammatory effects between modified citrus pectin and honokiol. Evid. Based Complement. Alternat. Med. 2017, 2017, 8379843. [Google Scholar] [CrossRef]
- Kokila, K.; Priyadharshini, S.D.; Sujatha, V. Phytopharmacological properties of Albizia species: A review. Int. J. Pharm. Pharm. Sci. 2013, 5, 70–73. [Google Scholar]
- Balkrishna, A.; Sakshi; Chauhan, M.; Dabas, A.; Arya, V. A comprehensive insight into the phytochemical, pharmacological potential, and traditional medicinal uses of Albizia lebbeck (L.) Benth. Evid. Based Complement. Alternat. Med. 2022, 2022, 5359669. [Google Scholar] [CrossRef]
- Babu, N.P.; Pandikumar, P.; Ignacimuthu, S. Anti-inflammatory activity of Albizia lebbeck Benth., an ethnomedicinal plant, in acute and chronic animal models of inflammation. J. Ethnopharmacol. 2009, 125, 356–360. [Google Scholar] [CrossRef]
- Kuum, M.G.M.; Guemmogne, R.J.T.; Ndzana, M.T.B.; Tchadji, J.C.; Lissom, A.; Dimo, T. Anti-inflammatory effects of the stem barks from Albizia ferruginea (Mimosaceae) on chronic inflammation induced in rats. Int. J. Innov. Res. Med. Sci. 2018, 3, 2183–2195. [Google Scholar]
- Meshram, G.G.; Kumar, A.; Rizvi, W.; Tripathi, C.D.; Khan, R.A. Evaluation of the anti-inflammatory activity of the aqueous and ethanolic extracts of the leaves of Albizzia lebbeck in rats. J. Tradit. Complement. Med. 2015, 6, 172–175. [Google Scholar] [CrossRef]
- Bakasatae, N.; Kunworarath, N.; Takahashi Yupanqui, C.; Voravuthikunchai, S.P.; Joycharat, N. Bioactive components, antioxidant, and anti-inflammatory activities of the wood of Albizia myriophylla. Rev. Bras. Farmacogn. 2018, 28, 444–450. [Google Scholar] [CrossRef]
- Pu, W.L.; Zhang, M.Y.; Bai, R.Y.; Sun, L.K.; Li, W.H.; Yu, Y.L.; Zhang, Y.; Song, L.; Wang, Z.X.; Peng, Y.F.; et al. Anti-inflammatory effects of Rhodiola rosea L.: A review. Biomed. Pharmacother. 2020, 121, 109552. [Google Scholar] [CrossRef]
- Kosakowska, O.; Bączek, K.; Przybył, J.L.; Pióro-Jabrucka, E.; Czupa, W.; Synowiec, A.; Gniewosz, M.; Costa, R.; Mondello, L.; Węglarz, Z. Antioxidant and antibacterial activity of roseroot (Rhodiola rosea L.) dry extracts. Molecules 2018, 23, 1767. [Google Scholar] [CrossRef]
- Lee, Y.; Jung, J.C.; Jang, S.; Kim, J.; Ali, Z.; Khan, I.A.; Oh, S. Anti-inflammatory and neuroprotective effects of constituents isolated from Rhodiola rosea. Evid. Based Complement Alternat. Med. 2013, 2013, 514049. [Google Scholar]
- Zeinali, M.; Zirak, M.R.; Rezaee, S.A.; Karimi, G.; Hosseinzadeh, H. Immunoregulatory and anti-inflammatory properties of Crocus sativus (Saffron) and its main active constituents: A review. Iran J. Basic Med. Sci. 2019, 22, 334–344. [Google Scholar]
- Ebrahimi, F.; Sahebkar, A.; Aryaeian, N.; Pahlavani, N.; Fallah, S.; Moradi, N.; Abbasi, D.; Hosseini, A.F. Effects of saffron supplementation on inflammation and metabolic responses in type 2 diabetic patients: A randomized, double-blind, placebo-controlled trial. Diabetes Metab. Syndr. Obes. 2019, 12, 2107–2115. [Google Scholar] [CrossRef]
- Ghobadi, H.; Abdollahi, N.; Madani, H.; Aslani, M.R. Effect of crocin from saffron (Crocus sativus L.) supplementation on oxidant/antioxidant markers, exercise capacity, and pulmonary function tests in COPD patients: A randomized, double-blind, placebo-controlled trial. Front. Pharmacol. 2022, 13, 884710. [Google Scholar] [CrossRef]
- Bahraini, M.; Hosseini, S.A.; Cheraghian, B.; Shoushtari, M.H. Effect of saffron on anti-inflammatory and oxidative stress in asthma. Int. J. Pharm. Phytopharm. Res. 2020, 10, 179–184. [Google Scholar]
- Poursamimi, J.; Shariati-Sarabi, Z.; Tavakkol-Afshari, J.; Mohajeri, S.A.; Mohammadi, M. Crocus Sativus (Saffron): An immunoregulatory factor in the autoimmune and non-autoimmune diseases. Iran J. Allergy Asthma Immunol. 2020, 19, 27–42. [Google Scholar] [CrossRef]
- Olajide, O.A. Inhibitory effects of St. John’s Wort on inflammation: Ignored potential of a popular herb. J. Diet Suppl. 2009, 6, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Tedeschi, E.; Menegazzi, M.; Margotto, D.; Suzuki, H.; Förstermann, U.; Kleinert, H. Anti-inflammatory actions of St. John’s wort: Inhibition of human inducible nitric-oxide synthase expression by down-regulating signal transducer and activator of transcription-1alpha (STAT-1alpha) activation. J. Pharmacol. Exp. Ther. 2003, 307, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Koeberle, A.; Rossi, A.; Bauer, J.; Dehm, F.; Verotta, L.; Northoff, H.; Sautebin, L.; Werz, O. Hyperforin, an anti-inflammatory constituent from St. John’s Wort, inhibits microsomal prostaglandin E(2) synthase-1 and suppresses prostaglandin E(2) formation in vivo. Front. Pharmacol. 2011, 2, 7. [Google Scholar] [CrossRef] [PubMed]
- Bonaterra, G.A.; Schwendler, A.; Hüther, J.; Schwarzbach, H.; Schwarz, A.; Kolb, C.; Abdel-Aziz, H.; Kinscherf, R. Neurotrophic, cytoprotective, and anti-inflammatory effects of St. John’s Wort extract on differentiated mouse hippocampal HT-22 neurons. Front. Pharmacol. 2018, 8, 955. [Google Scholar] [CrossRef] [PubMed]
- Brady, K.T.; Verduin, M.L. Pharmacotherapy of comorbid mood, anxiety, and substance use disorders. Subst. Use Misuse 2005, 40, 2043–2048. [Google Scholar] [CrossRef]
- Butterweck, V.; Schmidt, M. St. John’s wort: Role of active compounds for its mechanism of action and efficacy. Wien. Med. Wochenschr. 2007, 157, 356–361. [Google Scholar] [CrossRef]
- Paulke, A.; Nöldner, M.; Schubert-Zsilavecz, M.; Wurglics, M. St. John’s wort flavonoids and their metabolites show antidepressant activity and accumulate in brain after multiple oral doses. Pharmazie 2008, 63, 296–302. [Google Scholar]
- Butterweck, V. Mechanism of action of St. John’s wort in depression: What is known? CNS Drugs 2003, 17, 539–562. [Google Scholar] [CrossRef]
- Di Carlo, G.; Borrelli, F.; Ernst, E.; Izzo, A.A. St. John’s wort: Prozac from the plant kingdom. Trends Pharmacol. Sci. 2001, 22, 292–297. [Google Scholar] [CrossRef]
- Kholghi, G.; Arjmandi-Rad, S.; Zarrindast, M.R.; Vaseghi, S. St. John’s wort (Hypericum perforatum) and depression: What happens to the neurotransmitter systems? Naunyn. Schmiedebergs. Arch. Pharmacol. 2022, 395, 629–642. [Google Scholar] [CrossRef]
- Avila-Sosa, R.; Nevárez-Moorillón, G.V.; Ochoa-Velasco, C.E.; Navarro-Cruz, A.R.; Hernández-Carranza, P.; Cid-Pérez, T.S. Detection of saffron’s main bioactive compounds and their relationship with commercial quality. Foods 2022, 11, 3245. [Google Scholar] [CrossRef]
- Gohari, A.R.; Saeidnia, S.; Mahmoodabadi, M.K. An overview on saffron, phytochemicals, and medicinal properties. Pharmacogn. Rev. 2013, 7, 61–66. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Ali Redha, A.; Snoeck, E.R.; Singh, S.; Simal-Gandara, J.; Ibrahim, S.A.; Jafari, S.M. Anti-depressant properties of crocin molecules in saffron. Molecules 2022, 27, 2076. [Google Scholar] [CrossRef]
- Shafiee, M.; Arekhi, S.; Omranzadeh, A.; Sahebkar, A. Saffron in the treatment of depression, anxiety and other mental disorders: Current evidence and potential mechanisms of action. J. Affect. Disord. 2018, 227, 330–337. [Google Scholar] [CrossRef]
- Maqbool, Z.; Arshad, M.S.; Ali, A.; Aziz, A.; Khalid, W.; Afzal, M.F.; Bangar, S.P.; Addi, M.; Hano, C.; Lorenzo, J.M. Potential role of phytochemical extract from saffron in development of functional foods and protection of brain-related disorders. Oxid. Med. Cell. Longev. 2022, 2022, 6480590. [Google Scholar] [CrossRef]
- Petrisor, G.; Motelica, L.; Craciun, L.N.; Oprea, O.C.; Ficai, D.; Ficai, A. Melissa officinalis: Composition, pharmacological effects and derived release systems-a review. Int. J. Mol. Sci. 2022, 23, 3591. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Quispe, C.; Herrera-Bravo, J.; Akram, M.; Abbaass, W.; Semwal, P.; Painuli, S.; Konovalov, D.A.; Alfred, M.A.; Kumar, N.V.A.; et al. Phytochemical constituents, biological activities, and health-promoting effects of the Melissa officinalis. Oxid. Med. Cell. Longev. 2021, 2021, 6584693. [Google Scholar] [CrossRef]
- Wake, G.; Court, J.; Pickering, A.; Lewis, R.; Wilkins, R.; Perry, E. CNS acetylcholine receptor activity in European medicinal plants traditionally used to improve failing memory. J. Ethnopharmacol. 2000, 69, 105–114. [Google Scholar] [CrossRef]
- Kenda, M.; Glavač, N.K.; Nagy, M.; Dolenc, M.S. Medicinal plants used for anxiety, depression, or stress treatment: An update. Molecules 2022, 27, 6021. [Google Scholar] [CrossRef]
- Lin, S.H.; Chou, M.L.; Chen, W.C.; Lai, Y.S.; Lu, K.H.; Hao, C.W.; Sheen, L.Y. A medicinal herb, Melissa officinalis L. ameliorates depressive-like behavior of rats in the forced swimming test via regulating the serotonergic neurotransmitter. J. Ethnopharmacol. 2015, 175, 266–272. [Google Scholar] [CrossRef]
- Białoń, M.; Krzyśko-Łupicka, T.; Nowakowska-Bogdan, E.; Wieczorek, P.P. Chemical composition of two different lavender essential oils and their effect on facial skin microbiota. Molecules 2019, 24, 3270. [Google Scholar] [CrossRef] [PubMed]
- Prusinowska, R.; Śmigielski, K.B. Composition, biological properties and therapeutic effects of lavender. A review. Herba Pol. 2014, 60, 56–66. [Google Scholar] [CrossRef]
- López, V.; Nielsen, B.; Solas, M.; Ramírez, M.J.; Jäger, A.K. Exploring pharmacological mechanisms of lavender (Lavandula angustifolia) essential oil on central nervous system targets. Front. Pharmacol. 2017, 8, 280. [Google Scholar] [CrossRef] [PubMed]
- Shamabadi, A.; Akhondzadeh, S. Bioactive components for depression: Naringin, caffeine, probiotics, saffron and lavender may exert antidepressant effects through inflammation modulation. J. Iran Med. Counc. 2022, 5, 1–3. [Google Scholar] [CrossRef]
- Schuwald, A.M.; Nöldner, M.; Wilmes, T.; Klugbauer, N.; Leuner, K.; Müller, W.E. Lavender oil-potent anxiolytic properties via modulating voltage dependent calcium channels. PLoS ONE 2013, 8, e59998. [Google Scholar] [CrossRef] [PubMed]
- Adamczak, A.; Buchwald, W.; Gryszczyńska, A. Biometric features and content of phenolic compounds of roseroot (Rhodiola rosea L.). Acta Soc. Bot. Pol. 2016, 85, 3500. [Google Scholar] [CrossRef]
- Chiang, H.M.; Chen, H.C.; Wu, C.S.; Wu, P.Y.; Wen, K.C. Rhodiola plants: Chemistry and biological activity. J. Food Drug Anal. 2015, 23, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Amsterdam, J.D.; Panossian, A.G. Rhodiola rosea L. as a putative botanical antidepressant. Phytomedicine 2016, 23, 770–783. [Google Scholar] [CrossRef] [PubMed]
- Konstantinos, F.; Heun, R. The effects of Rhodiola rosea supplementation on depression, anxiety and mood—A systematic review. Glob. Psychiatry 2020, 3, 72–82. [Google Scholar] [CrossRef]
- Stojcheva, E.I.; Quintela, J.C. The effectiveness of Rhodiola rosea L. preparations in alleviating various aspects of life-stress symptoms and stress-induced conditions-encouraging clinical evidence. Molecules 2022, 27, 3902. [Google Scholar] [CrossRef] [PubMed]
- Fermino, B.L.; Milanez, M.C.; de Freitas, G.B.L.; da Silva, W.C.F.N.; Pereira, R.P.; da Rocha, J.B.T.; Bonini, J.S. Ginkgo biloba L.: Phytochemical components and antioxidant activity. AJPP 2015, 9, 950–955. [Google Scholar]
- Bai, S.; Zhang, X.; Chen, Z.; Wang, W.; Hu, Q.; Liang, Z.; Shen, P.; Gui, S.; Zeng, L.; Liu, Z.; et al. Insight into the metabolic mechanism of diterpene ginkgolides on antidepressant effects for attenuating behavioural deficits compared with venlafaxine. Sci. Rep. 2017, 7, 9591. [Google Scholar] [CrossRef]
- Fehske, C.J.; Leuner, K.; Müller, W.E. Ginkgo biloba extract (EGb761) influences monoaminergic neurotransmission via inhibition of NE uptake, but not MAO activity after chronic treatment. Pharmacol. Res. 2009, 60, 68–73. [Google Scholar] [CrossRef]
- Sloley, B.D.; Urichuk, L.J.; Morley, P.; Durkin, J.; Shan, J.J.; Pang, P.K.; Coutts, R.T. Identification of kaempferol as a monoamine oxidase inhibitor and potential neuroprotectant in extracts of Ginkgo biloba leaves. J. Pharm. Pharmacol. 2000, 52, 451–459. [Google Scholar] [CrossRef]
- Lü, J.M.; Yao, Q.; Chen, C. Ginseng compounds: An update on their molecular mechanisms and medical applications. Curr. Vasc. Pharmacol. 2009, 7, 293–302. [Google Scholar] [CrossRef]
- Ratan, Z.A.; Haidere, M.F.; Hong, Y.H.; Park, S.H.; Lee, J.O.; Lee, J.; Cho, J.Y. Pharmacological potential of ginseng and its major component ginsenosides. J. Ginseng Res. 2021, 45, 199–210. [Google Scholar] [CrossRef]
- Jin, Y.; Cui, R.; Zhao, L.; Fan, J.; Li, B. Mechanisms of Panax ginseng action as an antidepressant. Cell Prolif. 2019, 52, e12696. [Google Scholar] [CrossRef]
- Mu, D.; Ma, Q. A review of antidepressant effects and mechanisms of three common herbal medicines: Panax ginseng, Bupleurum chinense, and Gastrodia elata. CNS Neurol. Disord. Drug Targets 2022. Online ahead of print. [Google Scholar] [CrossRef]
- Hou, W.; Wang, Y.; Zheng, P.; Cui, R. Effects of Ginseng on neurological disorders. Front. Cell. Neurosci. 2020, 14, 55. [Google Scholar] [CrossRef]
- Abolhassani, M. Antiviral activity of borage (Echium amoenum). Arch. Med. Sci. 2010, 6, 366–369. [Google Scholar] [CrossRef]
- Zannou, O.; Pashazadeh, H.; Ghellam, M.; Ibrahim, S.A.; Koca, I. Extraction of anthocyanins from Borage (Echium amoenum) flowers using choline chloride and a glycerol-based, deep eutectic solvent: Optimization, antioxidant activity, and in vitro bioavailability. Molecules 2021, 27, 134. [Google Scholar] [CrossRef] [PubMed]
- Nouri, M.; Farajdokht, F.; Torbati, M.; Ranjbar, F.; Hamedyazdan, S.; Araj-Khodaei, M.; Sadigh-Eteghad, S. A close look at Echium amoenum processing, neuroactive components, and effects on neuropsychiatric disorders. Galen Med. J. 2019, 8, e1559. [Google Scholar] [CrossRef] [PubMed]
- Abdol, N.; Setorki, M. Evaluation of antidepressant, antianxiolytic, and antioxidant effects of Echium amoenum L. extract on social isolation stress of male mice. Iran Red Crescent Med. J. 2020, 22, e97593. [Google Scholar] [CrossRef]
- Jeyasri, R.; Muthuramalingam, P.; Suba, V.; Ramesh, M.; Chen, J.T. Bacopa monnieri and their bioactive compounds inferred multi-target treatment strategy for neurological diseases: A cheminformatics and system pharmacology approach. Biomolecules 2020, 10, 536. [Google Scholar] [CrossRef]
- Mathur, D.; Goyal, K.; Koul, V.; Anand, A. The molecular links of re-emerging therapy: A review of evidence of Brahmi (Bacopa monniera). Front. Pharmacol. 2016, 7, 44. [Google Scholar] [CrossRef]
- Liu, X.; Liu, F.; Yue, R.; Li, Y.; Zhang, J.; Wang, S.; Zhang, S.; Wang, R.; Shan, L.; Zhang, W. The antidepressant-like effect of bacopaside I: Possible involvement of the oxidative stress system and the noradrenergic system. Pharmacol. Biochem. Behav. 2013, 110, 224–230. [Google Scholar] [CrossRef]
- Sekhar, V.C.; Viswanathan, G.; Baby, S. Insights into the molecular aspects of neuroprotective Bacoside A and Bacopaside I. Curr. Neuropharmacol. 2019, 17, 438–446. [Google Scholar] [CrossRef]
- Fatima, U.; Roy, S.; Ahmad, S.; Ali, S.; Elkady, W.M.; Khan, I.; Alsaffar, R.M.; Adnan, M.; Islam, A.; Hassan, M.I. Pharmacological attributes of Bacopa monnieri extract: Current updates and clinical manifestation. Front. Nutr. 2022, 9, 972379. [Google Scholar] [CrossRef]
- Asgarirad, H.; Chabra, A.; Rahimnejad, M.; Zaghi Hosseinzadeh, A.; Davoodi, A.; Azadbakht, M. Comparison of Albizia Julibressin and silver sulfadiazine in healing of second and third degree burns. World J. Plast. Surg. 2018, 7, 34–44. [Google Scholar]
- Mahasweta, R.; Yadav, D.K.; Kumar, B.; Kaur, J.; Patel, A.K.; Kumar, N. A review on phytochemical and pharmacological studies of Albizia julibrissin: An ornamental plant. World J. Pharm. Res. 2016, 5, 598–604. [Google Scholar]
- Patro, G.; Kumar Bhattamisra, S.; Kumar Mohanty, B. Effects of Mimosa pudica L. leaves extract on anxiety, depression and memory. Avicenna J. Phytomed. 2016, 6, 696–710. [Google Scholar]
- Udyavar, S.; Kumar, K.S.; Rai, M.; Gopalakrishna, H.N.; Sowmya, C.R. Evaluation of antidepressant activity of ethanolic extract of Mimosa pudica in swiss albino mice. Indian J. Pharm. Pharmacol. 2020, 7, 240–244. [Google Scholar] [CrossRef]
- Duarte-Filho, L.A.M.S.; Amariz, I.A.; Nishimura, R.H.V.; Massaranduba, A.B.R.; Menezes, P.M.N.; Damasceno, T.A.; Brys, I.; Rolim, L.A.; Silva, F.S.; Ribeiro, L.A.A. β-carboline-independent antidepressant-like effect of the standardized extract of the barks of Mimosa tenuiflora (Willd) Poir. occurs via 5-HT2A/2C receptors in mice. J. Psychopharmacol. 2022, 36, 836–848. [Google Scholar] [CrossRef]
- Patočka, J.; Jakl, J.; Strunecká, A. Expectations of biologically active compounds of the genus Magnolia in biomedicine. J. Appl. Biomed. 2006, 4, 171–178. [Google Scholar] [CrossRef]
- Lee, Y.J.; Lee, Y.M.; Lee, C.K.; Jung, J.K.; Han, S.B.; Hong, J.T. Therapeutic applications of compounds in the Magnolia family. Pharmacol. Ther. 2011, 130, 157–176. [Google Scholar] [CrossRef]
- Yi, L.T.; Xu, Q.; Li, Y.C.; Yang, L.; Kong, L.D. Antidepressant-like synergism of extracts from magnolia bark and ginger rhizome alone and in combination in mice. Prog. Neuropsychopharmacol. Biol. Psychiatry 2009, 33, 616–624. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, P.P.; Hu, K.L.; Li, L.N.; Yu, X.; Lu, Y.; Chang, H.S. Antidepressant-like effect and mechanism of action of honokiol on the mouse lipopolysaccharide (LPS) depression model. Molecules 2019, 24, 2035. [Google Scholar] [CrossRef]
- Fan, X.X.; Sun, W.Y.; Li, Y.; Tang, Q.; Li, L.N.; Yu, X.; Wang, S.Y.; Fan, A.R.; Xu, X.Q.; Chang, H.S. Honokiol improves depression-like behaviors in rats by HIF-1α-VEGF signaling pathway activation. Front. Pharmacol. 2022, 13, 968124. [Google Scholar] [CrossRef]
- Tenny, S.; Varacallo, M. Evidence Based Medicine. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK470182/ (accessed on 9 December 2022).
- Szajewska, H. Evidence-Based Medicine and clinical research: Both are needed, neither is perfect. Ann. Nutr. Metab. 2018, 72, 13–23. [Google Scholar] [CrossRef]
- Murad, M.H.; Asi, N.; Alsawas, M.; Alahdab, F. New evidence pyramid. Evid. Based Med. 2016, 21, 125–127. [Google Scholar] [CrossRef]
- Cui, Y.H.; Zheng, Y. A meta-analysis on the efficacy and safety of St. John’s wort extract in depression therapy in comparison with selective serotonin reuptake inhibitors in adults. Neuropsychiatr. Dis. Treat. 2016, 12, 1715–1723. [Google Scholar] [PubMed]
- Apaydin, E.A.; Maher, A.R.; Shanman, R.; Booth, M.S.; Miles, J.N.; Sorbero, M.E.; Hempel, S. A systematic review of St. John’s wort for major depressive disorder. Syst. Rev. 2016, 5, 148. [Google Scholar] [CrossRef] [PubMed]
- Ng, Q.X.; Venkatanarayanan, N.; Ho, C.Y. Clinical use of Hypericum perforatum (St. John’s wort) in depression: A meta-analysis. J. Affect Disord. 2017, 210, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Hausenblas, H.A.; Heekin, K.; Mutchie, H.L.; Anton, S. A systematic review of randomized controlled trials examining the effectiveness of saffron (Crocus sativus L.) on psychological and behavioral outcomes. J. Integr. Med. 2015, 13, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Hausenblas, H.A.; Saha, D.; Dubyak, P.J.; Anton, S.D. Saffron (Crocus sativus L.) and major depressive disorder: A meta-analysis of randomized clinical trials. J. Integr. Med. 2013, 11, 377–383. [Google Scholar] [CrossRef]
- Jackson, P.A.; Forster, J.; Khan, J.; Pouchieu, C.; Dubreuil, S.; Gaudout, D.; Moras, B.; Pourtau, L.; Joffre, F.; Vaysse, C.; et al. Effects of saffron extract supplementation on mood, well-being, and response to a psychosocial stressor in healthy adults: A randomized, double-blind, parallel group, clinical trial. Front. Nutr. 2021, 7, 606124. [Google Scholar] [CrossRef]
- Akhondzadeh, S.; Mostafavi, S.A.; Keshavarz, S.A.; Mohammadi, M.R.; Hosseini, S.; Eshraghian, M.R. A placebo controlled randomized clinical trial of Crocus sativus L. (saffron) on depression and food craving among overweight women with mild to moderate depression. J. Clin. Pharm. Ther. 2020, 45, 134–143. [Google Scholar] [CrossRef]
- Ghazizadeh, J.; Sadigh-Eteghad, S.; Marx, W.; Fakhari, A.; Hamedeyazdan, S.; Torbati, M.; Taheri-Tarighi, S.; Araj-Khodaei, M.; Mirghafourvand, M. The effects of lemon balm (Melissa officinalis L.) on depression and anxiety in clinical trials: A systematic review and meta-analysis. Phytother. Res. 2021, 35, 6690–6705. [Google Scholar] [CrossRef]
- Haybar, H.; Javid, A.Z.; Haghighizadeh, M.H.; Valizadeh, E.; Mohaghegh, S.M.; Mohammadzadeh, A. The effects of Melissa officinalis supplementation on depression, anxiety, stress, and sleep disorder in patients with chronic stable angina. Clin. Nutr. ESPEN 2018, 26, 47–52. [Google Scholar] [CrossRef]
- Heidari, M.; Soltanpour, A.; Naseri, M.; Kazemnezhad, A. The effect of Lemon Balm (Melissa officinalis) on depression in patients after coronary artery bypass graft. Iran J. Cardiovasc. Nurs. 2015, 4, 43–46. [Google Scholar]
- Firoozeei, T.S.; Feizi, A.; Rezaeizadeh, H.; Zargaran, A.; Roohafza, H.R.; Karimi, M. The antidepressant effects of lavender (Lavandula angustifolia Mill.): A systematic review and meta-analysis of randomized controlled clinical trials. Complement. Ther. Med. 2021, 59, 102679. [Google Scholar] [CrossRef]
- Nategh, M.; Reza, H.M.; Abbas, E.; Reza, N.; Zahra, M.; Bahman, A. Lavender aromatherapy on anxiety and depression in patients with Acute Coronary Syndrome: A single-blind randomized clinical trial. Front. Nurs. 2022, 9, 233–239. [Google Scholar] [CrossRef]
- Tayebi, A.; Kasra Dehkordi, A.; Ebadi, A.; Sahraei, H.; Einollahi, B. The Effect of aromatherapy with Lavender essential oil on depression, anxiety and stress in hemodialysis patients: A clinical trial. Evid. Based Care J. 2015, 5, 65–74. [Google Scholar]
- Jokar, M.; Delam, H.; Bakhtiari, S.; Paki, S.; Askari, A.; Bazrafshan, M.R.; Shokrpour, N. The effects of inhalation Lavender aromatherapy on postmenopausal women’s depression and anxiety: A randomized clinical trial. JNP 2020, 16, 617–622. [Google Scholar] [CrossRef]
- Mao, J.J.; Xie, S.X.; Zee, J.; Soeller, I.; Li, Q.S.; Rockwell, K.; Amsterdam, J.D. Rhodiola rosea versus sertraline for major depressive disorder: A randomized placebo-controlled trial. Phytomedicine 2015, 22, 394–399. [Google Scholar] [CrossRef]
- Liang, Z.H.; Jia, Y.B.; Wang, M.L.; Li, Z.R.; Li, M.; Yun, Y.L.; Zhu, R.X. Efficacy of Ginkgo Biloba extract as augmentation of venlafaxine in treating post-stroke depression. Neuropsychiatr. Dis. Treat. 2019, 15, 2551–2557. [Google Scholar] [CrossRef]
- Dai, C.X.; Hu, C.C.; Shang, Y.S.; Xie, J. Role of Ginkgo biloba extract as an adjunctive treatment of elderly patients with depression and on the expression of serum S100B. Medicine 2018, 97, e12421. [Google Scholar] [CrossRef]
- Jeong, H.G.; Ko, Y.H.; Oh, S.Y.; Han, C.; Kim, T.; Joe, S.H. Effect of Korean Red Ginseng as an adjuvant treatment for women with residual symptoms of major depression. Asia Pac. Psychiatry 2015, 7, 330–366. [Google Scholar] [CrossRef]
- Lee, K.H.; Bahk, W.M.; Lee, S.J.; Pae, C.U. Effectiveness and tolerability of Korean Red Ginseng augmentation in major depressive disorder patients with difficult-to-treat in routine practice. Clin. Psychopharmacol. Neurosci. 2020, 18, 621–626. [Google Scholar] [CrossRef]
- Sayyah, M.; Sayyah, M.; Kamalinejad, M. A preliminary randomized double blind clinical trial on the efficacy of aqueous extract of Echium amoenum in the treatment of mild to moderate major depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2006, 30, 166–169. [Google Scholar] [CrossRef]
- Najafabady, M.T.; Baghbadoranee, P.Y.; Moghimipour, E.; Haghighizadeh, M.H.; Boostani, H. Comparison the effect of Echium amoenum extract with fluoxetine on depression in menopausal women. A double-blind randomized controlled trial. Asian J. Pharm. 2019, 13, 271–275. [Google Scholar]
- Bargard, M.S.; Assadi, S.; Amini, H.; Saiiah, M.; Akhondzadeh, S.; Kamalinejad, M. Efficacy of aqueous extract of Echium amoenum L. in the treatment of mild to moderate major depressive disorder: A randomized double blind clinical trial. J. Med. Plants 2004, 3, 61–68. [Google Scholar]
- Shetty, S.K.; Rao, P.N.; U, S.; Raj, A.; Ks, S.; Sv, S. The effect of Brahmi (Bacopa monnieri (L.) Pennell) on depression, anxiety and stress during Covid-19. Eur. J. Integr. Med. 2021, 48, 101898. [Google Scholar] [CrossRef]
- Calabrese, C.; Gregory, W.L.; Leo, M.; Kraemer, D.; Bone, K.; Oken, B. Effects of a standardized Bacopa monnieri extract on cognitive performance, anxiety, and depression in the elderly: A randomized, double-blind, placebo-controlled trial. J. Altern. Complement. Med. 2008, 14, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Jiang, C.; Zhao, X.; Zhang, Y.Q. Study on the Effects of Albizia julibrissin flower on cognitive function and plasma 5-HT, NE and DA in patients with depression: A randomized parallel controlled multicenter clinical trial. J. Pract. Tradit. Chin. Intern. Med. 2013, 27, 18–20. [Google Scholar]
- Xue, L.; Zhang, J.; Shen, H.; Ai, L.; Wu, R. A randomized controlled pilot study of the effectiveness of magnolia tea on alleviating depression in postnatal women. Food Sci. Nutr. 2020, 8, 1554–1561. [Google Scholar] [CrossRef]
- Panossian, A.G.; Efferth, T.; Shikov, A.N.; Pozharitskaya, O.N.; Kuchta, K.; Mukherjee, P.K.; Banerjee, S.; Heinrich, M.; Wu, W.; Guo, D.A.; et al. Evolution of the adaptogenic concept from traditional use to medical systems: Pharmacology of stress- and aging-related diseases. Med. Res. Rev. 2021, 41, 630–703. [Google Scholar] [CrossRef]
- Panossian, A.; Efferth, T. Network pharmacology of adaptogens in the assessment of their pleiotropic therapeutic activity. Pharmaceuticals 2022, 15, 1051. [Google Scholar] [CrossRef]
- Jawaid, T.; Gupta, R.; Siddiqui, Z.A. A review on herbal plants showing antidepressant activity. IJPSR 2011, 2, 3051–3060. [Google Scholar]
- Rabiei, Z.; Rabiei, S. A review on antidepressant effect of medicinal plants. Bangladesh J. Pharmacol. 2017, 12, 1–11. [Google Scholar] [CrossRef]
- Taboada, T.; Alvarenga, N.L.; Galeano, A.K.; Arrúa, W.J.; Campuzano-Bublitz, M.A.; Kennedy, M.L. In vivo antidepressant-like effect assessment of two Aloysia species in mice and LCMS chemical characterization of ethanol extract. Molecules 2022, 27, 7828. [Google Scholar] [CrossRef]
- Wang, J.; Hu, D.; Hou, J.; Li, S.; Wang, W.; Li, J.; Bai, J. Ethyl acetate fraction of Hemerocallis citrina Baroni decreases tert-butyl hydroperoxide-induced oxidative stress damage in BRL-3A cells. Oxid. Med. Cell. Longev. 2018, 2018, 1526125. [Google Scholar] [CrossRef]
- Matraszek-Gawron, R.; Chwil, M.; Terlecka, P.; Skoczylas, M.M. Recent studies on anti-depressant bioactive substances in selected species from the genera Hemerocallis and Gladiolus: A systematic review. Pharmaceuticals 2019, 12, 172. [Google Scholar] [CrossRef]
- Odhiambo, J.A.; Siboe, G.M.; Lukhoba, C.W.; Dossaji, S.F. Antifungal activity of crude extracts of Gladiolus dalenii van geel (iridaceae). Afr. J. Tradit. Complement. Altern. Med. 2009, 7, 53–58. [Google Scholar] [CrossRef]
- Agarwa, P.; Sharma, B.; Fatima, A.; Jain, S.K. An update on Ayurvedic herb Convolvulus pluricaulis Choisy. Asian Pac. J. Trop. Biomed. 2014, 4, 245–252. [Google Scholar] [CrossRef]
- Sharma, R.; Singla, R.K.; Banerjee, S.; Sinha, B.; Shen, B.; Sharma, R. Role of Shankhpushpi (Convolvulus pluricaulis) in neurological disorders: An umbrella review covering evidence from ethnopharmacology to clinical studies. Neurosci. Biobehav. Rev. 2022, 140, 104795. [Google Scholar] [CrossRef]
- Moragrega, I.; Ríos, J.L. Medicinal plants in the treatment of depression: Evidence from preclinical studies. Planta Med. 2021, 87, 656–685. [Google Scholar] [CrossRef]
- Moragrega, I.; Ríos, J.L. Medicinal plants in the treatment of depression. II: Evidence from clinical trials. Planta Med. 2022, 88, 1092–1110. [Google Scholar] [CrossRef]
| Brand Name | Marketing Authorization Holder | Pharmaceutical Form | Active Ingredients (per Dose) | Dosage | Declared Effects by MAH |
|---|---|---|---|---|---|
| MIRALO | USP Zdrowie, Warszawa | Capsules | 100 mg Withania somnifera (ashwagandha) extract (7 mg withanolides), 28 mg saffron stigma extract (0.84 mg crocin and 0.56 mg safranal), 100 mg lemon balm extract (2 mg rosemary acid) | 1 capsule a day | Improves mood, helps maintain a state of relaxation, contributes to emotional balance. |
| JASNUM MOOD | USP Zdrowie, Warszawa | Capsules | 30 mg saffron stigma extract, vitamin B6 (1.4 mg), vitamin B12 (100 μg) | 1 capsule a day | Alleviates hormonal disorders during menopause that cause mood swings. |
| DEPRIBON | Bonimed, Żywiec | Capsules | 150 mg of St. John’s wort extract, 50 mg of Ginseng root extract | 1 capsule a day | Improves mood. |
| INTRACTUM HYPERICI | Phyto Pharm, Klęka | Liquid | Ethanol extract of Hypericum perforatumL., herba (fresh St. John’s wort) Hyperici herbae recentis intractum (1:1). Extraction solvent: ethanol 96% (v/v) | 4 times a day, 5 mL of the preparation in a small amount of liquid. The recommended treatment time is 4 weeks. | Traditional herbal medicine used to relieve temporary states of nervous exhaustion. It also relieves depression. |
| DEPRESANUM | Novascon, Warszawa | Coated tablets | Inositol 100 mg, L-tryptophan 100 mg, saffron flower extract 15 mg, vitamin B6 0.210 mg, folic acid 100 μg | 1 tablet 2 times a day during a meal | Helps maintain a positive mood, reduces fatigue and tiredness, improves nervous system function. |
| SZAFRACEUM | Lekam, Warszawa | Tablets | Saffron extract 30 mg, inositol 100 mg, L-tryptophan 50 mg, ginseng root extract 50 mg, vitamin B6 1.3 mg, vitamin B2 1.3 mg, vitamin B12 2.5 µg, folic acid 200 µg | 1 tablet a day | Helps maintain a positive mood, supports the maintenance of emotional balance. |
| DEPRELLA | Organic Pharma, Warszawa | Capsules | L-tryptophan 150 mg, L-phenylalanine 150 mg, turmeric extract (Curcuma longa) 10:1–100 mg, saffron crocus extract 30 mg, vitamin D (cholecalciferol) 5 µg, vitamin B6 (pyridoxine hydrochloride) 1.4 mg, vitamin B9 (pteroylmonoglutamic acid) 200 µg, vitamin B12 (cyanocobalamin) 2.5 µg | 1 capsule a day | Improves mood and has an antidepressant effect by using the bio-potential of a harmonious combination of ingredients that support the production of neurotransmitters and happiness hormones and the transmission of nerve impulses. The composition is enriched with the addition of B vitamins, essential for the proper functioning of the nervous system and the maintenance of mental balance. |
| ALINESS | MedicaLine, Karczew | Tablets | Cultivated saffron crocus extract (Crocus sativus) SafraSOL 30 mg, including crocin 3 mg, safranal 600 µg | 1–3 tablets a day during or after a meal with water | Improves mood, helps maintain emotional balance, improves libido, helps maintain comfort before and during the menstrual cycle. |
| HYPERHERBA | Labofarm, Starogard Gdański | Tablets | 30 mg Hypericum perforatum L., herba (St. John’s wort) | 1 tablet 3 times a day with water | Traditionally used to relieve temporary symptoms of mental exhaustion. |
| DEPREMIN | Colfarm, Mielec | Coated tablets | 612 mg of extract (in the form of a dry, quantified extract) from Hypericum perforatum L., herba (St. John’s wort), which corresponds to: 0.6 mg–1.8 mg of total hypericin expressed as hypericin, 36.72 mg–91.80 mg of total flavonoids per rutin, not more than 36.72 mg of hyperforin Extraction solvent: ethanol 60% (v/v) | 1 tablet a day | Used in the short-term treatment of mild depressive disorders. |
| MAGNOLIAMAX | K2Pharm, s.r.o. Opava, Czech Republic | Coated tablets | 350 mg of magnolia bark extract 6% (21 mg honokiol), black pepper extract—5 mg | 1 tablet a day | Supports restful sleep and helps maintain a good mood. Relieves fatigue and stress. |
| Drugs | Possible Mechanism of Interaction | Effect of Interaction on Drug |
|---|---|---|
| Pharmacokinetic interactions | ||
| HIV protease inhibitors (indinavir, nelfinavir, ritonavir and saquinavir) | Induction of CYP3A4 | Reduced blood concentrations with possible loss of HIV suppression |
| HIV non-nucleoside reverse transcriptase inhibitors (efavirenz, nevirapine) | Induction of CYP3A4 | Reduced blood concentrations with possible loss of HIV suppression |
| Anticonvulsants (carbamazepine, phenobarbitone and phenytoin) | Induction of CYP3A4 | Reduced blood concentrations with risk of seizures |
| Oral contraceptives | Induction of CYP3A4 and CYP1A2 | Reduced blood concentrations with risk of unintended pregnancy and breakthrough bleeding |
| Oral anticoagulant-vitamin K antagonists (warfarin and phenprocoumon) | Induction of CYP2C9 | Reduced anticoagulant effect and need for increased dose |
| Cyclosporin | Induction of CYP3A4 and the transport protein P-glycoprotein | Reduced blood concentrations with risk of transplant rejection |
| Theophylline | Induction of CYP1A2 | Reduced blood concentrations and possible loss of control of asthma or chronic obstructive pulmonary disease |
| Digoxin | Induction of transport protein P-glycoprotein | Reduced blood concentrations and possible loss of control of heart rhythm or heart failure |
| Pharmacodynamic interactions | ||
| SSRI (citalopram, fluoxetine, fluvoxamine, paroxetine and sertraline) | Increasing of serotonin concentrations | Increased serotonergic effects with risk of increased incidence of serotonin syndrome |
| Serotonin 5HT 1B/1D agonists (“Triptans”; (sumatriptan, naratriptan, Rizatriptan and zolmitriptan) | Increasing of serotonin concentrations | Increased serotonergic effects with risk of increased incidence of serotonin syndrome |
| Scientific (Latin) Name | Common Name |
|---|---|
| Albizia julibrissin | Mimosa tree |
| Bacopa monnieri | Brahmi water hyssop |
| Crocus sativus | Saffron crocus |
| Echium amoenum | Borage |
| Ginkgo biloba | Gingko maidenhair tree |
| Hypericum perforatum | St. John’s wort (SJW) |
| Lavandula officinalis | Lavender |
| Magnolia officinalis | Magnolia bark houpu magnolia |
| Melissa officinalis | Lemon balm common balm balm mint |
| Panax ginseng | Korean ginseng |
| Rhodiola rosea | Roseroot golden root |
| Medicinal Plant and Its Commonly Used Parts | Major Active Constituents | Mechanism of Action | Additional Effects on the Nervous System | References |
|---|---|---|---|---|
| St. John’s wort flowers leaves |
|
| Neuroprotective; Improves cognitive functions | Composition: [179,180] Mechanisms: [181,182,183] |
| Saffron crocus dried thread-like parts of the flower (stigmas) |
|
| Anxiolytic Anti-inflammatory | Composition: [184,185] Mechanisms: [186,187,188] |
| Lemon balm leaves |
|
| Anxiolytic Sedative Neuroprotective | Composition: [189,190] Mechanisms: [191,192,193] |
| Lavender flowers |
|
| Anxiolytic Sedative | Composition: [194,195] Mechanisms: [196,197,198] |
| Roseroot root rhizomes |
|
| Adaptogenic; Increases resistance to stress, reduces fatigue | Composition: [199,200] Mechanisms: [201,202,203] |
| Ginkgo leaves |
|
| Neuroprotective; Improves cognitive functions | Composition: [142,204] Mechanisms: [205,206,207] |
| Korean ginseng root |
|
| Adaptogenic; Increases resistance to stress, reduces fatigue, improves cognitive functions | Composition: [208,209] Mechanisms: [210,211,212] |
| Borage leaves stems flowers |
|
| Anxiolytic | Composition: [213,214] Mechanisms: [140,215,216] |
| Brahmi leaves stems |
|
| Adaptogenic-increases resistance to stress, improves cognitive functions | Composition: [217,218] Mechanisms: [219,220,221] |
| Mimosa tree stem bark flowers |
|
| Anxiolytic Improves sleep | Composition: [222,223] Mechanisms: [224,225,226] |
| Magnolia bark tree bark |
|
| Anxiolytic | Composition: [227,228] Mechanisms: [229,230,231] |
| Author, Year, References | Type of Publication | Characteristic | Conclusions |
|---|---|---|---|
| St. John’s wort | |||
| Cui Y.H. et al. 2016 [235] | A meta-analysis | 27 clinical trials (3126 patients). Evaluation of the efficacy and safety of SJW extract and selective serotonin reuptake inhibitors (SSRIs) in the treatment of depression. | SJW extract did not differ from SSRIs in clinical response, remission and the mean reduction in Hamilton Rating Scale for Depression score. St. John’s wort extract had a significantly lower rate of adverse events than SSRIs. SJW extract and SSRIs effectively treat mild-to-moderate depression. |
| Apaydin E.A. et al. 2016 [236] | A systematic review | 35 clinical trials (6993 patients). Evaluation of the efficacy and safety of SJW in adults with major depressive disorder (MDD) compared to placebo and active comparator and evaluation of whether the effects vary by severity of MDD. | Compared to antidepressants, participants taking SJW were less likely to experience adverse events, with no difference in treatment effectiveness in mild and moderate depression. Monotherapy with SJW for mild and moderate depression was superior to placebo in reducing depression symptoms and not significantly different from antidepressant medication. |
| Ng X.N. et al. 2017 [237] | A meta-analysis | 27 clinical trials (3808 patients). Evaluation of SJW efficacy and comparison to SSRIs. | SJW showed comparable response and remission in patients with depression and a significantly lower withdrawal/depression rate compared to standard SSRIs. |
| Saffron crocus | |||
| Hausenblas H.A. et al. 2015 [238] | A systematic review | 12 clinical trials (928 patients). The evaluation of scientific evidence from randomized controlled trials (RCTs) regarding the efficacy of saffron crocus on psychological and behavioural outcomes. | Saffron crocus can reduce the symptoms and effects of depression, premenstrual syndrome, sexual dysfunction and infertility. |
| Hausenblas H.A. et al. 2013 [239] | A meta-analysis | 5 clinical trials (177 patients). A review of the clinical trials examining the effects of saffron crocus on the treatment of major depressive disorders (MDDs). | Saffron supplementation can reduce symptoms of depression in adults with MDD. |
| Jackson P.A. et al. 2021 [240] | A randomized, double-blind, parallel group clinical trial Clinical trial number: NCT03639831 | The study assessed the efficacy of 8 weeks’ supplementation with 30 mg standardized saffron extract or placebo for 8 weeks on emotional well-being in 56 healthy males and females aged 18–54 years with subclinical feelings of low mood and anxiety and/or stress. | Participants who received the saffron crocus extract reported reduced depression scores and improved social relationships at the end of the study. This extract appears to improve subclinical depressive symptoms in healthy individuals and may contribute to increased resilience against the development of stress-related psychiatric disorders. |
| Akhondzadeh S. et al. 2020 [241] | A placebo-controlled, double-blind, randomized clinical trial | The study evaluated the effects of 30 mg of Crocus sativus capsules administered for 12 weeks on food craving, body weight and depression among 73 overweight (BMI ≥ 25) women with mild and moderate depression compared to placebo. | Saffron crocus capsules were not effective in reducing food cravings, but as a safe over-the-counter supplement, they were effective in reducing the symptoms of depression in patients who experience mild or moderate depression and are overweight. |
| Lemon balm | |||
| Ghazizadeh J. et al. 2021 [242] | A systemic review and meta-analysis | Investigation of the effects and side effects of lemon balm as a medicinal herb on anxiety and depression in clinical trials published up to 30 October 2020. | Lemon balm significantly improved mean anxiety and depression scores compared with the placebo without serious side effects. Current evidence suggests that lemon balm may be effective in improving anxiety and depressive symptoms, particularly in the acute setting. |
| Haybar H. et al. 2018 [243] | A double-blind, randomized, placebo-controlled clinical trial | The study aimed to determine the effects of lemon balm supplementation (3 g during 8 weeks) on depression, anxiety, stress, and sleep disturbances in patients with chronic stable angina (CSA). | The intervention group receiving 8-week supplementation with 3 g lemon balm capsules had a significant reduction in scores of depression, anxiety, stress and total sleep disturbance in patients with CSA, compared to the placebo group. |
| Heidari M. et al. 2016 [244] | A double-blind, randomized, placebo-controlled clinical trial | The study evaluated the effect of lemon balm (Melissa Officinalis; 500 mg three times daily over 7 days) on depression in patients after coronary artery bypass graft. | After the intervention, herbal balm decreased depression more in the intervention group compared to the placebo group. Lemon balm may reduce depression after a coronary artery bypass graft. |
| Lavender | |||
| Firoozeei T.S. et al. 2021 [245] | A systematic review and meta-analysis | 17 clinical trials (1859 patients). Determining the efficacy of lavender on depression severity by performing a systematic review and meta-analysis. | The results showed a significant efficacy of lavender in reducing depression scores compared to the control group. Subgroup analysis showed that the effect of lavender was slightly more pronounced in participants with a diagnosis of depression, while its effect was statistically significant in patients with other illnesses with associated depressive symptoms, and the oral route was the most effective route of administration. |
| Nategh M. et al. 2022 [246] | A single-blind, randomized clinical trial | The study aimed to investigate the effects of lavender essential oil (two drops of lavender essence dropped on non-absorbent three-ply tissue paper attached to the patient’s shirt collar) on anxiety and depression in 110 patients with the acute coronary syndrome (ACS). | The study confirmed the effectiveness of lavender aromatherapy in reducing anxiety and depression in ACS patients and supports the use of lavender oil aromatherapy by intensive care nurses as a non-pharmacological and cost-effective intervention to reduce psychological tension and increase patient satisfaction during hospitalization in cardiac care units. |
| Tayebi A. et al. 2015 [247] | A randomized controlled clinical trial | The study evaluated the effect of aromatherapy with lavender essential oil on depression, anxiety and stress rates in 60 haemodialysis patients who inhaled the lavender essential oil smeared on a piece of cloth (three drops of oil) for one hour during the haemodialysis procedure. | The administration of lavender oil decreased the scores of depression and stress in the intervention group in comparison with the control group. Still, the reduction in the anxiety score was not statistically significant. Aromatherapy with lavender essential oil might reduce depression and stress among haemodialysis patients. Therefore, this method can be used as a complementary method with less complication to improve the quality of life of these patients. |
| Jokar M. et al. 2020 [248] | A single-blind, placebo-controlled, randomized clinical trial | Evaluation of the effect of inhaled lavender aromatherapy on depression and anxiety levels among 46 postmenopausal women who were administered 2% inhaled lavender essence or distilled water 20 min in the evening before bedtime for 4 consecutive weeks. | Depression and anxiety mean scores decreased in the lavender group compared with controls, providing evidence that lavender aromatherapy may be an effective non-invasive treatment during the postmenopausal stage. |
| Roseroot | |||
| Mao J.J. et al. 2014 [249] | A randomized, double-blind, placebo-controlled, parallel-group study clinical trial | Evaluation of the safety and efficacy of R. rosea (extract 340–1360 mg daily) versus sertraline or placebo therapy in 57 subjects with major depressive disorder (MDD) in a 12-week treatment. | Although R. rosea produced less antidepressant effect versus sertraline, it also resulted in significantly fewer adverse events and was better tolerated. These findings suggest that R. rosea, although less effective than sertraline, may have a more favourable risk–benefit ratio for people with mild to moderate depression. |
| Ginkgo | |||
| Liang Z.H. et al. 2019 [250] | A randomized, double-blind clinical trial | Evaluation of the efficacy of ginkgo extract (40 mg three times a day) as augmentation of treatment in 80 venlafaxine-treated (75–225 mg/day) post-stroke depression (PSD) patients. Moreover, serum 5-hydroxytryptamine (5-HT) and BDNF levels were measured before and after treatment. | Patients receiving ginkgo extract plus venlafaxine had significantly lower mean depression scale scores. Meanwhile, compared to the control group, patients in the experimental group additionally receiving ginkgo extract had significantly higher 5-HT levels and BDNF levels and required a lower dose of venlafaxine and experienced fewer adverse events. The results showed that ginkgo extract was a good augmentation of venlafaxine in the treatment of PSD. |
| Dai C.X. et al. 2018 [251] | A randomized clinical trial | Evaluation of the efficacy of ginkgo extract (EGb; 19.2 mg three times daily) as adjunctive treatment in 136 elderly depressed patients treated with citalopram (Cit; 20 mg daily) and the effect on serum glioma-derived protein (S100B) expression, which is reduced in depressive disorders. | The time to onset of efficacy was significantly shorter in the EGb + Cit group. After treatment, S100B levels decreased, and the change in S100B levels in the EGb + Cit group was greater than in the Cit group. EGb, as an adjunctive treatment, can effectively improve depressive symptoms and reduce serum S100B expression, which is a marker of brain damage, suggesting that EGb restores neurological function when treating depression in elderly patients. |
| Korean ginseng | |||
| Jeong H.G. et al. 2015 [252] | An open-label, prospective clinical study | Evaluation of the efficacy and tolerability of adjuvant treatment with Korean red ginseng (3 g/day for 8 weeks) in 25 female patients with residual symptoms of major depression. | Subjects reported a significant reduction in depressive symptoms over 8 weeks. The results suggest that Korean red ginseng is effective as an adjunctive treatment for patients with residual symptoms of major depression. |
| Lee K.H. et al. 2020 [253] | An open-label, prospective clinical study | Evaluation of the efficacy and tolerability of Korean red ginseng supplementation (RGA; 2 g/day for 6 weeks) in major depressive disorder (MDD) of 36 difficult-to-treat patients who were currently on antidepressant monotherapy (duloxetine, venlafaxine, paroxetine, sertraline, fluoxetine, escitalopram, mirtazapine). | The study demonstrated the presumed efficacy and tolerability of RGA in the treatment of MDD, which is difficult to treat in clinical practice. Remission rates were significantly better in patients treated with RGA. |
| Borage | |||
| Sayyah M. et al. 2006 [254] | A randomized, double-blind, placebo-controlled clinical trial | The evaluation of the efficacy of an aqueous extract of E. amoenum (375 mg daily for 6 weeks) in 35 patients with mild to moderate major depressive disorder. | At week 4, the extract showed a significant advantage over the placebo in reducing depressive symptoms. The effect on anxiety was not significant. |
| Najafabady M.T. et al. 2019 [255] | A double-blind, randomized clinical trial | The aim of the study was to compare the effects of Echium amoenum extract (500 mg daily for 8 weeks) with fluoxetine on depression in 72 menopausal women. | At the start of the study, a comparison of the mean depression scores in the two intervention and control groups showed no significant difference. However, a statistically significant difference was observed in the mean depression score in both groups at week 4 of the intervention. No statistically significant difference was observed between the mean depression scores of the two groups at the end of the study. |
| Saiiah Bargard M. et al. 2004 [256] | A double-blind, randomized clinical trial | Evaluation of the efficacy and safety of an aqueous extract of Echium amoenum (375 mg daily for 6 weeks) in 35 patients with mild to moderate major depressive disorder (MDD). | At week 4 of the study, the extract was superior to the placebo, and at week 6, the difference was marginally insignificant. Echium amoenum extract did not cause more side effects than placebo during the study. It was concluded that the aqueous extract of Echium amoenum can be considered an effective and safe treatment for MDD. |
| Brahmi | |||
| Shetty S.K. et al. 2021 [257] | An open-label, prospective clinical study | Evaluation of the efficacy of Brahmi (Bacopa monnieri; 500 mg twice daily for 30 days) in reducing depression, anxiety and stress among 198 in COVID-19 negative Indian patients aged 12 to 60 years. | The study showed a significant decrease in the severity of depression, anxiety and stress after Brahmi supplementation for 1 month. |
| Calabrese C. et al. 2008 [258] | A randomized, double-blind, placebo-controlled trial | Evaluation of the effects of Bacopa monnieri standardized dry extract (300 mg daily for 12 weeks) on cognitive function and affect, and its safety and tolerability in 54 healthy elderly participants (65 years or older). | Epidemiologic Studies Depression scale (CESD-10) depression scores, joint state plus trait anxiety scores, and decreased heart rate over time for the Bacopa group. |
| Mimosa tree | |||
| Xueli S. 2013 [259] | A randomized, parallel controlled clinical trial | Evaluation of the effects of Albizia julibrissin flower decoctum (AJF; 30 g, 1 dose/day, decocted in water up to 200 mL, in the afternoon; treatment for 6 weeks) in patients with depression compared to those treated with venlafaxine. | In the treatment group, 8 cases were cured, 20 cases were markedly effective, 10 cases were effective, 12 cases were invalid, the total efficacy was 76.00%. In the control group, 10 cases were cured, 20 cases were markedly effective, 10 cases were effective, 10 cases were invalid, the total efficacy was 80.00%. |
| Magnolia bark | |||
| Xue L. et al. 2020 [260] | A randomized controlled clinical trial | The aim of the study was to investigate whether magnolia tea drunk for 3 weeks (one cup per day, prepared by infusing the tea in 300 mL of hot water for 12 min) has a palliative effect on postnatal depression in 143 postpartum women. | Compared to the control group, the intervention group showed a significant difference in sleep deprivation related to physical symptoms at week 3 post-test. The comparison results also showed alleviation of depressive symptoms at week 3 post-test and week 6 post-test. The results suggested that drinking a single-ingredient magnolia tea for a 3-week duration had positive effects on postpartum women. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobrek, L.; Głowacka, K. Depression and Its Phytopharmacotherapy—A Narrative Review. Int. J. Mol. Sci. 2023, 24, 4772. https://doi.org/10.3390/ijms24054772
Dobrek L, Głowacka K. Depression and Its Phytopharmacotherapy—A Narrative Review. International Journal of Molecular Sciences. 2023; 24(5):4772. https://doi.org/10.3390/ijms24054772
Chicago/Turabian StyleDobrek, Lukasz, and Krystyna Głowacka. 2023. "Depression and Its Phytopharmacotherapy—A Narrative Review" International Journal of Molecular Sciences 24, no. 5: 4772. https://doi.org/10.3390/ijms24054772
APA StyleDobrek, L., & Głowacka, K. (2023). Depression and Its Phytopharmacotherapy—A Narrative Review. International Journal of Molecular Sciences, 24(5), 4772. https://doi.org/10.3390/ijms24054772








