Phosphorus-Containing Polybenzoxazine Aerogels with Efficient Flame Retardation and Thermal Insulation
Abstract
1. Introduction
2. Results and Discussion
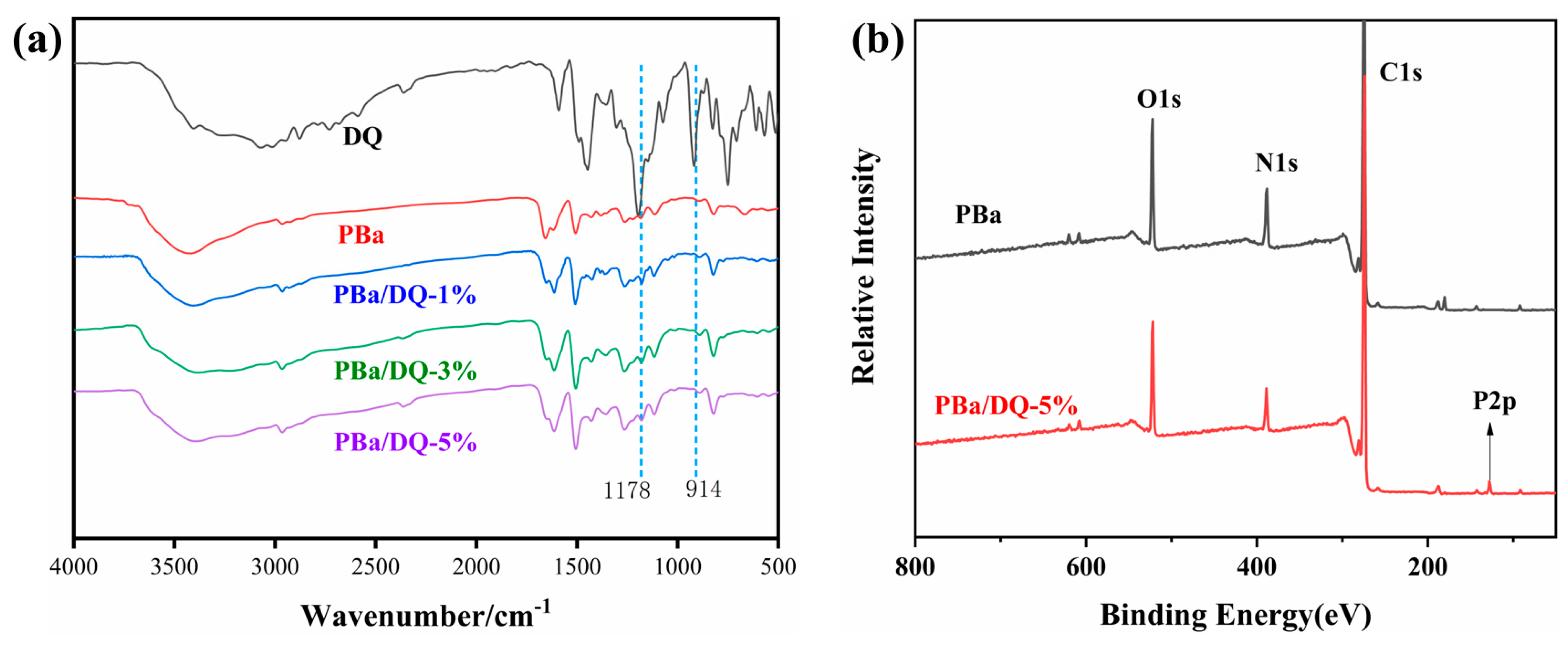
2.1. Chemical Structure of PBa and PBa Composite Aerogels
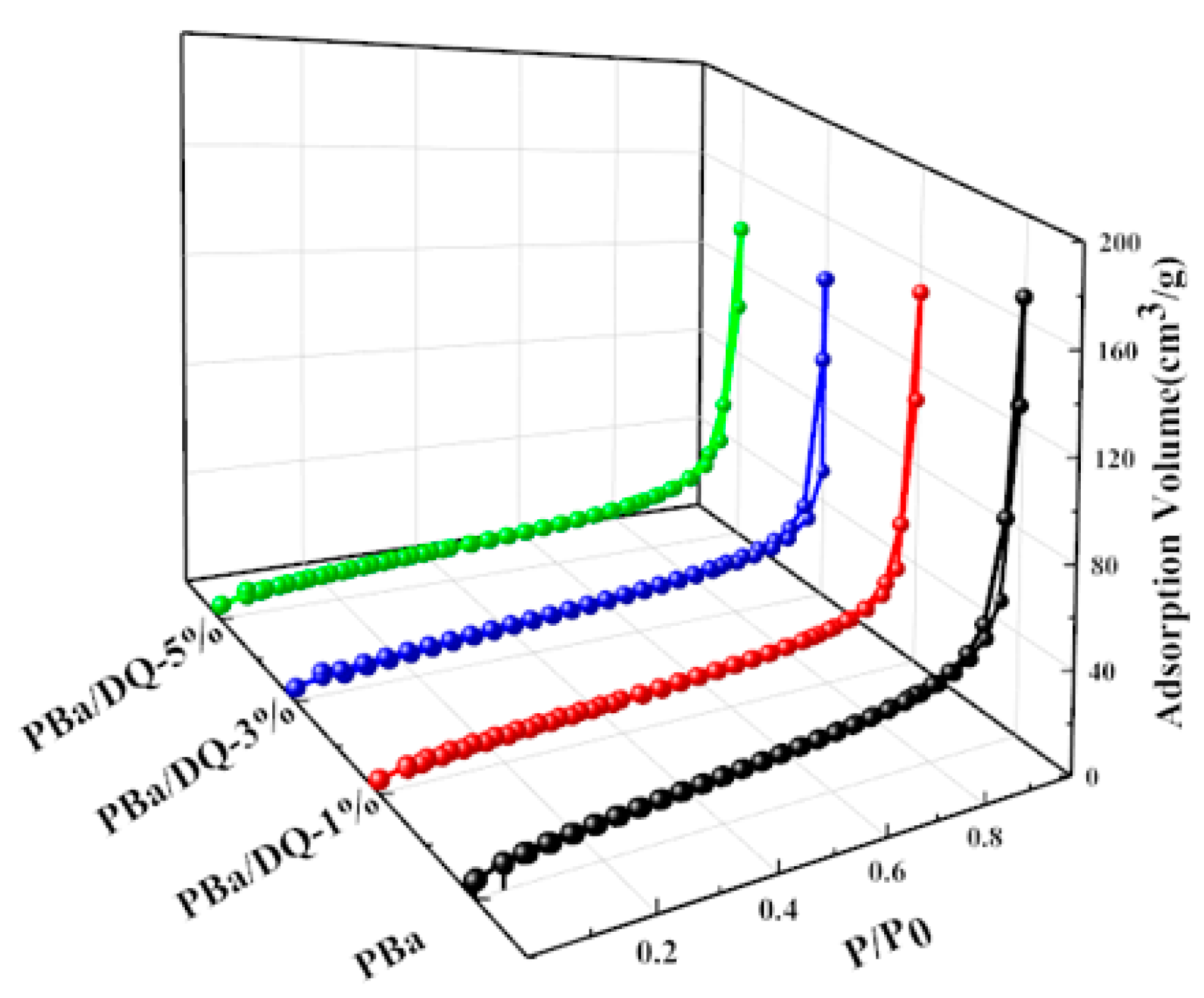
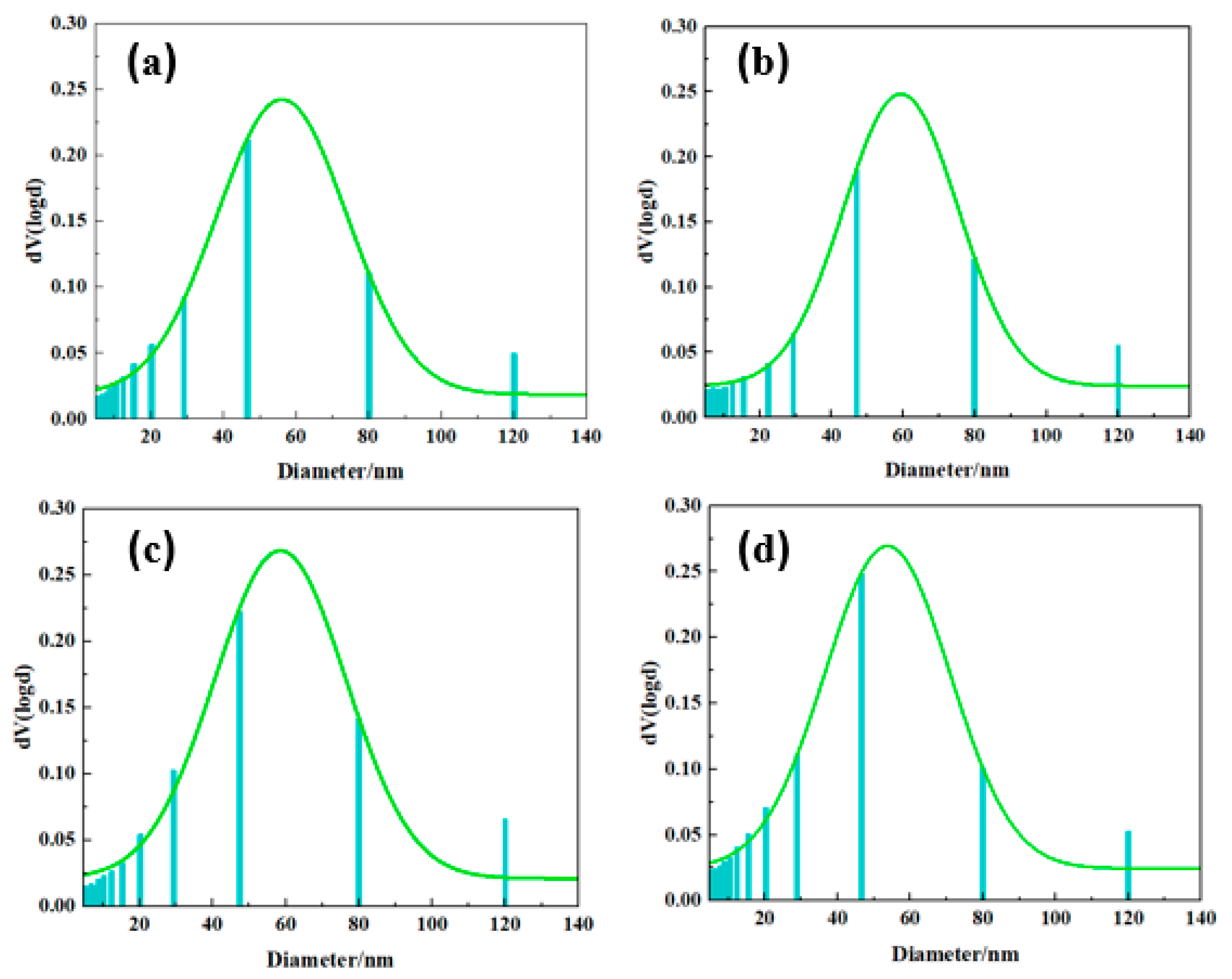
2.2. Surface Morphology of PBa and PBa Composite Aerogels
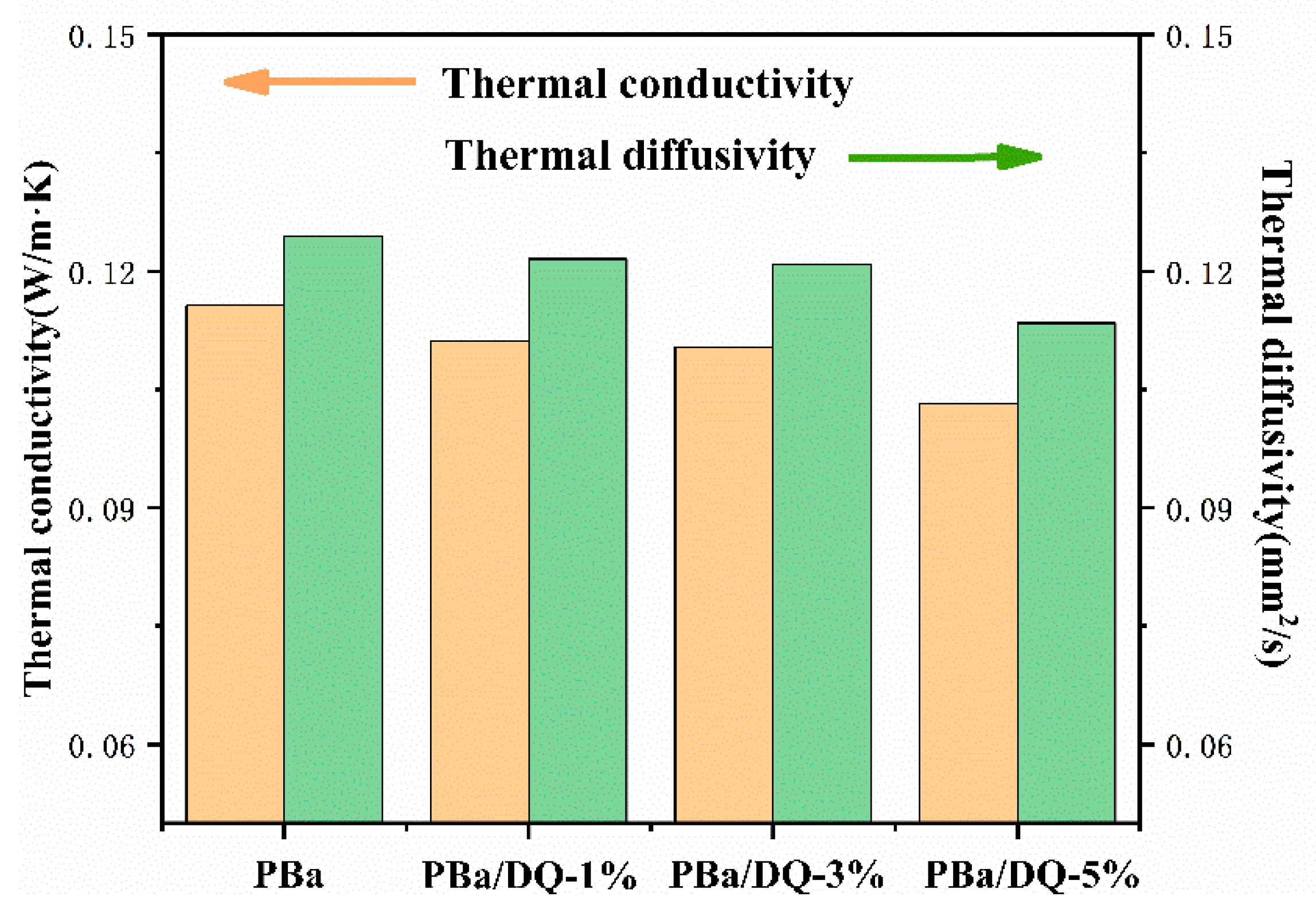
2.3. Thermal Insulation Performance of PBa and PBa Composite Aerogels
2.4. Thermal Degradation Behaviors of PBa and PBa Composite Aerogels
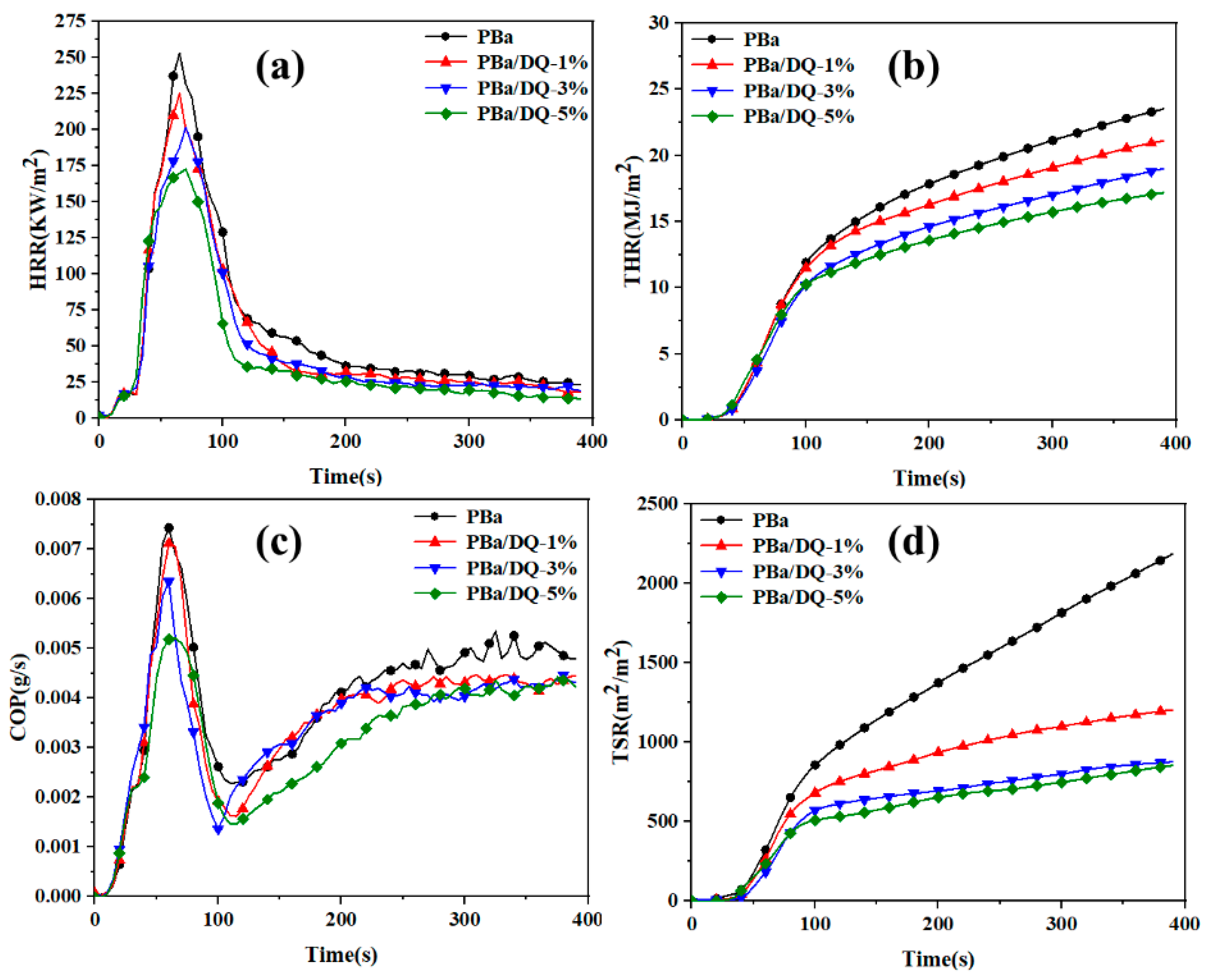
2.5. Flammability Performance of PBa and PBa Composite Aerogels
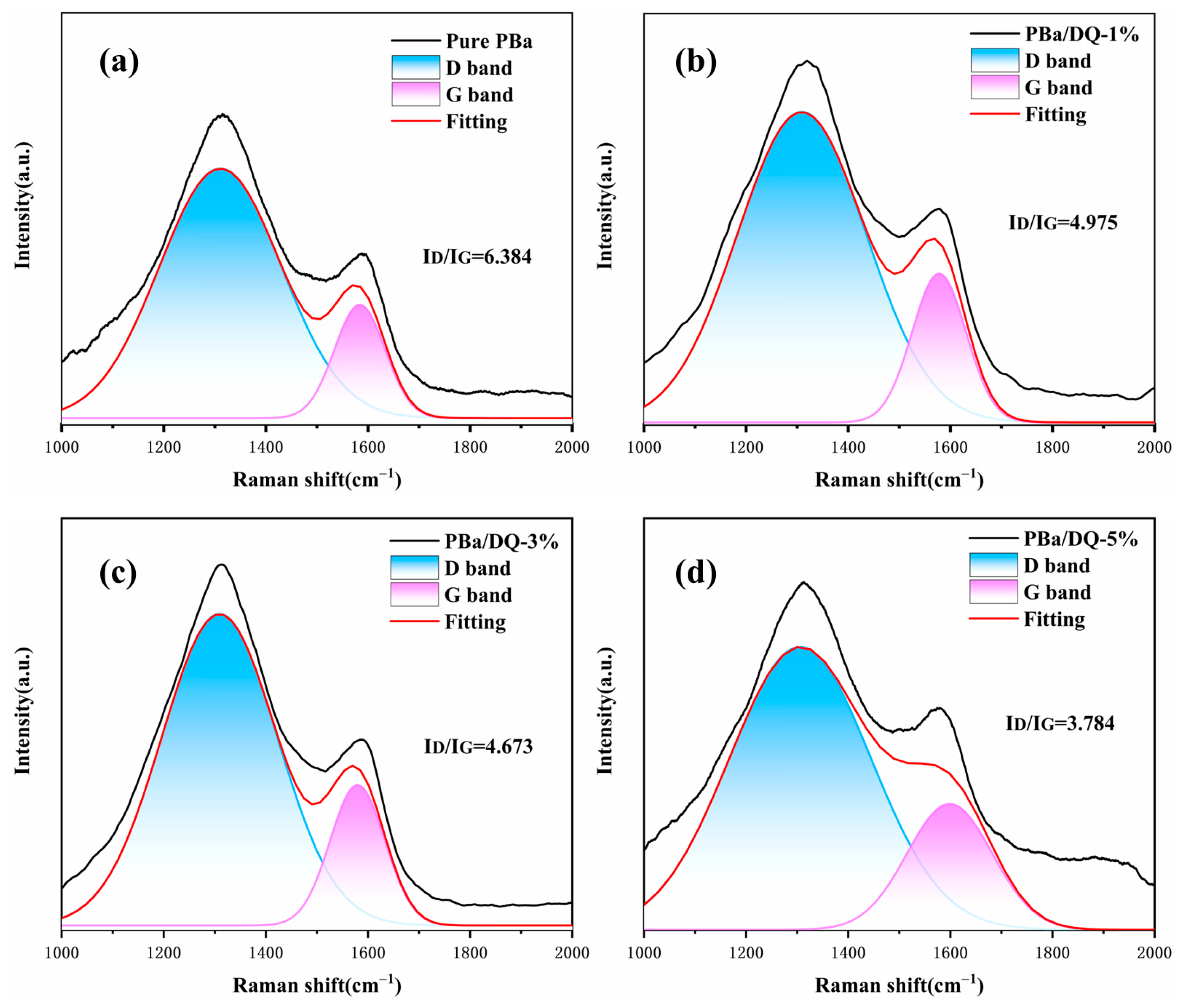
2.6. Analysis of Flame Retardancy Mechanism
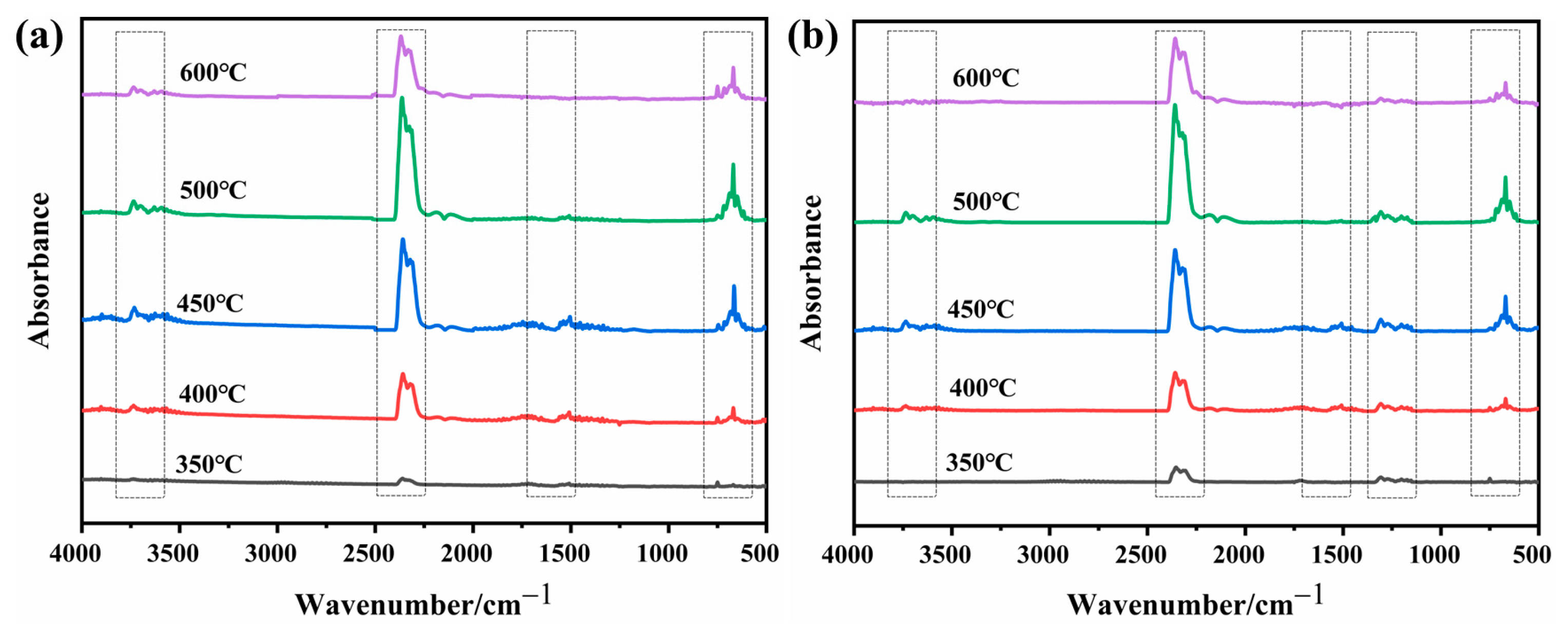
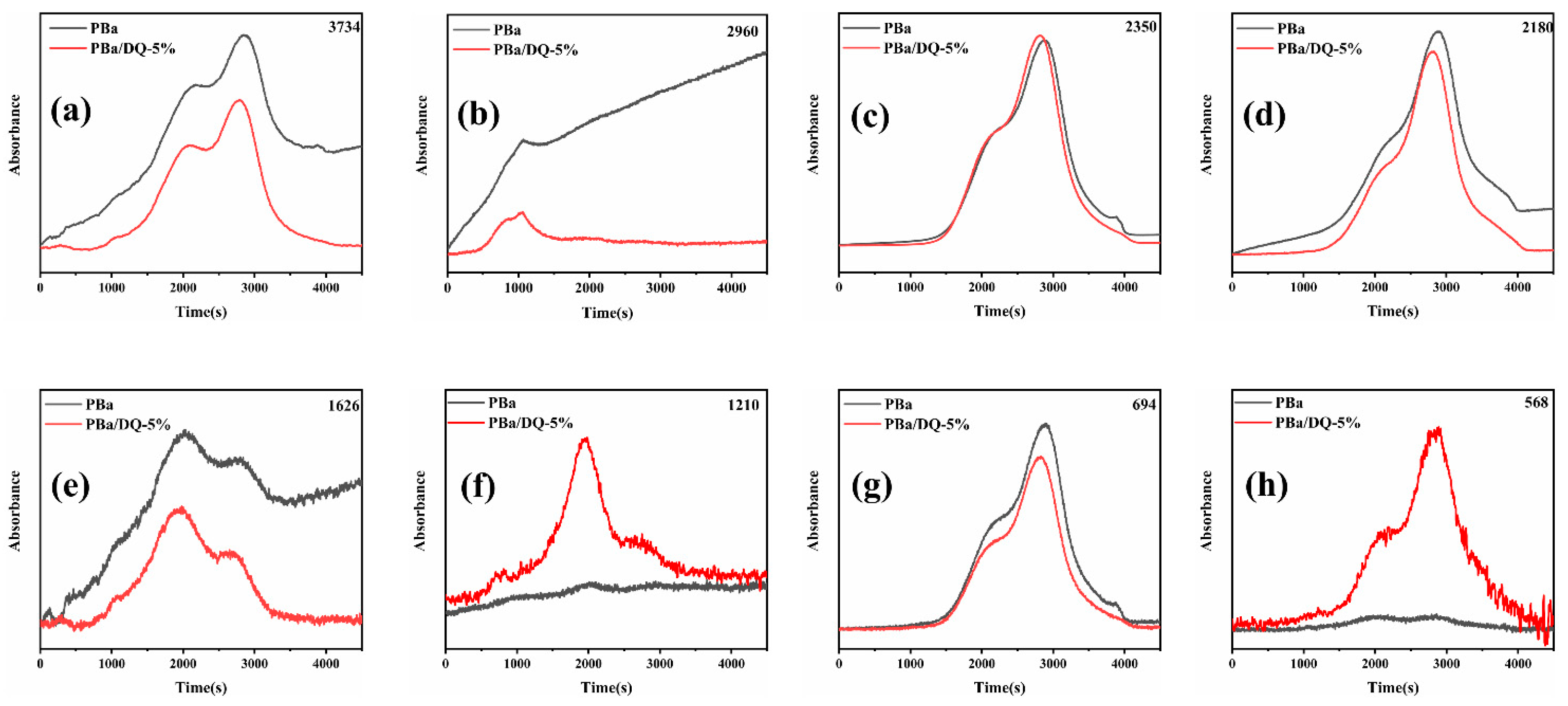
2.7. Gas Phase Analysis
3. Materials and Methods
3.1. Materials
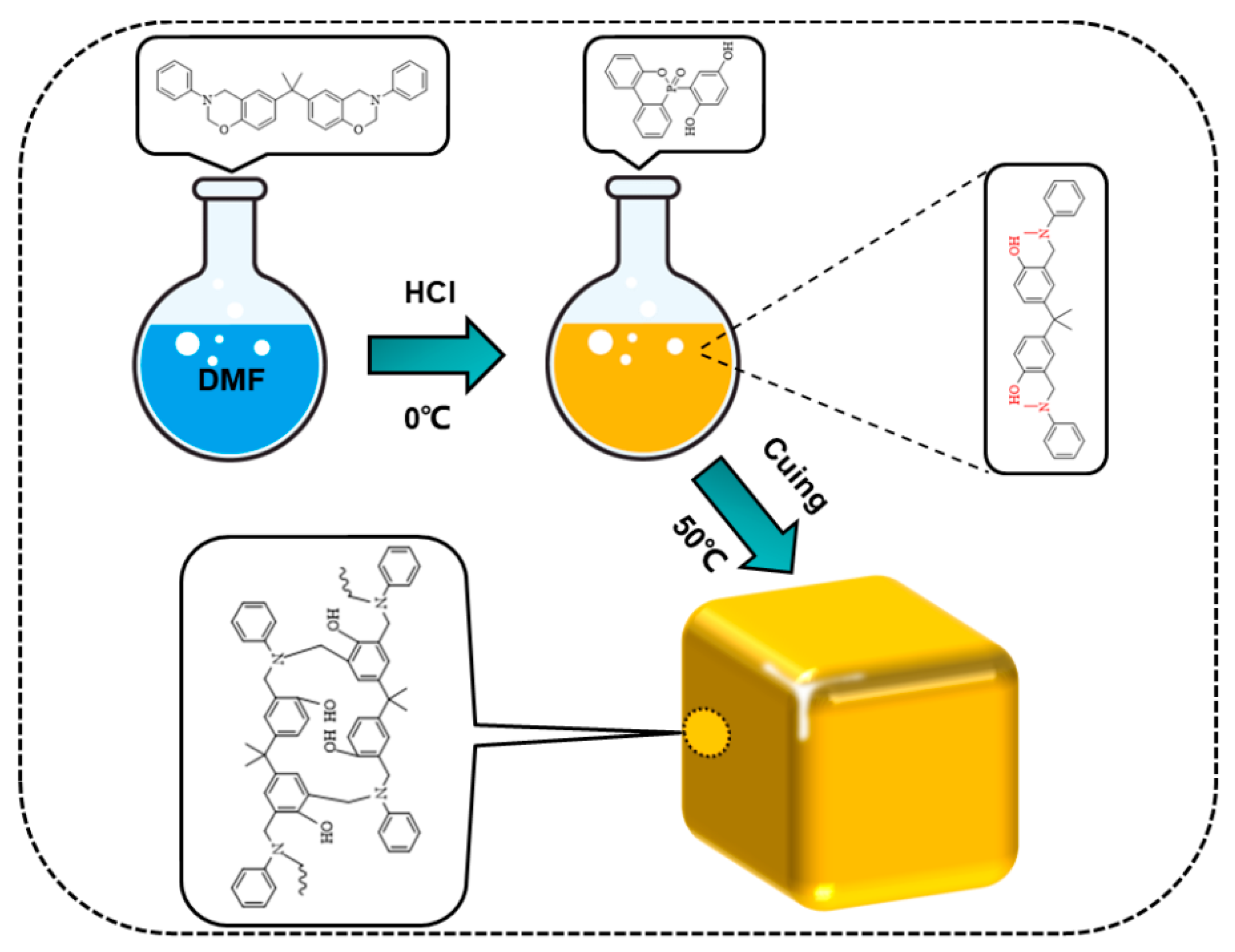
3.2. Preparation of Benzoxazine/DOPO-HQ Mixed Solution
3.3. Preparation of Polybenzoxazine/DOPO-HQ Aerogels
3.4. Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Demir, K.D.; Tasdelen, M.A.; Uyar, T.; Kawaguchi, A.W.; Sudo, A.; Endo, T.; Yagci, Y. Synthesis of polybenzoxazine/clay nanocomposites by in situ thermal ring-opening polymerization using intercalated monomer. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 4213–4220. [Google Scholar] [CrossRef]
- Vengatesan, M.R.; Devaraju, S.; Kannaiyan, D.; Song, J.K.; Alagar, M. Ultrasound-assisted synthesis of benzoxazine monomers: Thermal and mechanical properties of polybenzoxazines. Polym. Int. 2013, 62, 127–133. [Google Scholar] [CrossRef]
- Kimura, H.; Matsumoto, A.; Ohtsuka, K. New type of phenolic resin: Curing reaction of phenol-novolac based benzoxazine with bisoxazoline or epoxy resin using latent curing agent and the properties of the cured resin. J. Appl. Polym. Sci. 2009, 112, 1762–1770. [Google Scholar] [CrossRef]
- Soto, M.; Hiller, M.; Oschkinat, H.; Koschek, K. Multifunctional Benzoxazines Feature Low Polymerization T emperature and Diverse Polymer Structures. Polymers 2016, 8, 278. [Google Scholar] [CrossRef]
- Kudoh, R.; Sudo, A.; Endo, T. A highly reactive benzoxazine monomer, 1-(2-hydroxyethyl)-1,3-benzoxazine: Activation of benzoxazine by neighboring group participation of hydroxyl group. Macromolecules 2010, 43, 1185–1187. [Google Scholar] [CrossRef]
- Kimura, H.; Ohtsuka, K.; Matsumoto, A. New thermosetting resin from benzoxazine and cyanate ester resin. Adv. Polym. Technol. 2013, 32, 651–659. [Google Scholar] [CrossRef]
- Liu, B.-W.; Zhao, H.-B.; Wang, Y.-Z. Advanced Flame-Retardant Methods for Polymeric Materials. Adv. Mater. 2021, 34, 2107905–2107941. [Google Scholar] [CrossRef]
- Zeng, M.; Zhu, W.; Feng, Z.-J.; Chen, J.-B.; Huang, Y.-W.; Xu, Q.-Y.; Wang, J.-X. Two novel halogen-free, phosphorus-free, and intrinsically flame-retardant benzoxazine thermosets containing electron-withdrawing bridge groups. Appl. Polym. 2020, 137, e49300. [Google Scholar] [CrossRef]
- Altikok, C.; Kiskan, B.; Yagci, Y. Synthesis and characterization of sulfone containing main chain oligobenzoxazine precursors. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 2445–2450. [Google Scholar] [CrossRef]
- Lee, H.-W.; Liu, Y.-L. Thermally stable, flame retardant; low-dielectric constants, and flexible thermosetting resins based on a tetrafunctional benzoxazine compound possessing a cyclic siloxane core. Appl. Polym. 2022, 139, e52605. [Google Scholar]
- Naik, D.; Wazarkar, K.; Sabnis, A. UV-curable flame-retardant coatings based on phosphorous and silicon containing oligomers. J. Coat. Technol. Res. 2018, 16, 733–743. [Google Scholar] [CrossRef]
- Sponton, M.; Ronda, J.-C.; Galia, M.; Cadiz, V. Studies on thermal and flame retardant behaviour of mixtures of bis(m-aminophenyl)methylphosphine oxide based benzoxazine and glycidylether or benzoxazine of Bisphenol A. Polym. Degrad. Stab. 2008, 93, 2158–2165. [Google Scholar] [CrossRef]
- Naiker, V.E.; Patil, D.A.; More, A.P.; Mhaske, S.T. Synthesis of high-performance bio-based benzoxazine for flame retardant application. Polym. Adv. Technol. 2021, 33, 1481–1495. [Google Scholar] [CrossRef]
- Casarino, A.; Casis, N.; Estenoz, D.; Sponton, M.-E. Synthesis and characterization of polybenzoxazine/silica-based hybrid nanostructures for flame retardancy applications. Polym. Eng. Sci. 2022, 62, 1386–1398. [Google Scholar] [CrossRef]
- Sponton, M.; Ronda, J.; Galia, M.; Cadiz, V. Development of flame retardant phosphorus- and silicon-containing polybenzoxazines. Polym. Degrad. Stab. 2009, 94, 145–150. [Google Scholar] [CrossRef]
- Gu, S.; Miyoshi, T.; Jana, S. Polybenzoxazine aerogels with controllable pore structures. RSC Adv. 2015, 5, 26801. [Google Scholar] [CrossRef]
- Thubsuang, U.; Ishida, H.; Wongkasemjit, S.; Chaisuwan, T. Novel template confinement derived from polybenzoxazine-based carbon xerogels for synthesis of ZSM-5 nanoparticles via microwave irradiation. Microporous Mesoporous Mater. 2012, 156, 7–15. [Google Scholar] [CrossRef]
- Li, H.; Gu, S.; Thomas, S.; Liu, T.; Jana, S.-C. Investigation of polybenzoxazine gelation using laser light scattering. Appl. Polym. 2018, 135, 45709. [Google Scholar] [CrossRef]
- Sahin, I.; Özbakır, Y.; Inönü, Z.; Ulker, Z.; Erkey, C. Kinetics of supercritical drying of gels. Gels 2018, 4, 3. [Google Scholar] [CrossRef]
- González, C.G.; Rey, M.C.; Alnaief, M.; Zetzl, C.; Smirnova, I. Supercritical drying of aerogels using CO2: Effect of extraction time on the end material textural properties. J. Supercrit. Fluids 2012, 66, 297–306. [Google Scholar] [CrossRef]
- Slosarczyk, A.; Barełkowski, M.; Niemier, S. Synthesis and characterisation of silica aerogel/carbon microfibers nanocomposites dried in supercritical and ambient pressure conditions. J. Sol-Gel Sci. Technol. 2015, 76, 227–232. [Google Scholar] [CrossRef]
- Rani, T.S.; Subha, M.C.S.; Reddy, G.V. Synthesis of water-glass-based silica aerogel powder via with and without squeezing of hydrogels. J. Appl. Polym. Sci. 2010, 115, 1675–1679. [Google Scholar] [CrossRef]
- Sandrine, B.; Claudia, H.; Ilbizian, P. Lightweight superinsulating Resorcinol-Formaldehyde-APTES benzoxazine aerogel blankets for space applications. Eur. Polym. J. 2016, 78, 25–37. [Google Scholar]
- Sandrine, B.; Claudia, H.; Ilbizian, P. Evaluation of lightweight and flexible insulating aerogel blankets based on Resorcinol-Formaldehyde-Silica for space applications. Eur. Polym. J. 2017, 93, 403–416. [Google Scholar]
- Alhwaigea, A.; Ishidac, H.; Qutubuddin, S. Chitosan/polybenzoxazine/clay mixed matrix composite aerogels:preparation, physical properties, and water absorbency. Appl. Clay Sci. 2020, 184, 105403. [Google Scholar] [CrossRef]
- Lorjai, P.; Chaisuwan, T.; Wongkasemjit, S. Porous structure of polybenzoxazine-based organic aerogel prepared by sol–gel process and their carbon aerogels. J. Sol-Gel Sci. Technol. 2009, 52, 56–64. [Google Scholar] [CrossRef]
- Li, L.; Xiao, Y.; Zhang, S.; Feng, J.; Jiang, Y.; Feng, J. Lightweight, strong and thermally insulating polymethylsilsesquioxane- polybenzoxazine aerogels by ambient pressure drying. J. Sol-Gel Sci. Technol. 2021, 1–10. [Google Scholar] [CrossRef]
- Zhang, M.; Heb, L.; Shic, T.; Zha, R.-H. Neat 3D C3N4 monolithic aerogels embedded with carbon aerogels via ring-opening polymerization with high photoreactivity. Appl. Catal. B Environ. 2020, 266, 118652. [Google Scholar] [CrossRef]
- Qin, G.-Q.; Jiang, S.-L.; Zhang, H.-T.; Qin, S.-J.; Wu, H.-Y.; Zhang, F.-P.; Zhan, G.-L. Ambient-dried, ultra-high strength, low thermal conductivity, high char residual rate f-type polybenzoxazine aerogel. ACS Omega 2022, 7, 26116–26122. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.-Y.; Li, L.-J.; Zhang, S.-Z.; Feng, J.-Z.; Jiang, Y.-G.; Feng, J. Room temperature oxalic acid-catalyzed, ambient pressure dried, and cost-effective synthesis of polybenzoxazine aerogels for thermal insulation. Adv. Eng. Mater. 2021, 23, 2000856. [Google Scholar] [CrossRef]
- Maghsoudi, K.; Motahari, S. Mechanical, thermal, and hydrophobic properties of silica aerogel–epoxy composites. J. Appl. Polym. Sci. 2018, 7, 45706–45715. [Google Scholar] [CrossRef]
- Volfkovich, Y.; Lobach, A.; Spitsyna, N. Hydrophilic and hydrophobic pores in reduced graphene oxide aerogel. J. Porous Mater. 2019, 26, 1111–1119. [Google Scholar] [CrossRef]
- Xiao, Y.-Y.; Li, L.-J.; Liu, F.-Q.; Zhang, S.-Z.; Feng, J.-Z.; Jiang, Y.-G.; Feng, J. Compressible, flame-resistant and thermally insulating fiber-reinforced polybenzoxazine aerogel composites. Materials 2020, 13, 2809. [Google Scholar] [CrossRef] [PubMed]
- Alhwaige, A.; Alhassan, S.; Katsiotis, M.-S.; Ishida, H. Interactions, morphology and thermal stability of graphene-oxide reinforced polymer aerogel derived from star-like telechelic aldehyde-terminal benzoxazine resin. RSC Adv. 2015, 5, 92719–92731. [Google Scholar] [CrossRef]
- Hoffmann, T.; Pospiech, D.; HÄUßLER, L. Phosphorus-containing polysulfones a comparative study. High Perform. Polym. 2010, 22, 715–741. [Google Scholar] [CrossRef]
- Zhang, S.; Ran, Q.-C.; Gu, Y. Polymerization mechanism of 1,3-benzoxazine catalyzed by PCl5 and rearrangement of chemical structures. Eur. Polym. J. 2021, 142, 110133. [Google Scholar] [CrossRef]
- Semerci, E.; Kiskan, B.; Yagci, Y. Thiol reactive polybenzoxazine precursors: A novel route to functional polymers by thiol-oxazine chemistry. Eur. Polym. J. 2015, 69, 636–641. [Google Scholar] [CrossRef]
- Xiao, Y.-Y.; Li, L.-J.; Zhang, S.-Z.; Feng, J.-Z.; Jiang, Y.-G.; Feng, J. Thermally insulating polybenzoxazine aerogels based on 4,40-diamino-diphenylmethane benzoxazine. J. Mater. Sci. 2019, 54, 12951–12961. [Google Scholar] [CrossRef]
- Feng, J.-Z.; Wang, X.; Jiang, Y.-G.; Du, X.; Feng, J. Study on thermal conductivities of aromatic polyimide aerogels. ACS Appl. Mater. Interfaces 2016, 8, 12992–12996. [Google Scholar] [CrossRef]
- Guo, H.; Meador, M.A.; McCorkle, L.; Quade, D.J.; Guo, J.; Hamilton, B.; Cakmak, M.; Sprowl, G. Polyimide aerogels cross-linked through amine functionalized poly-oligomeric silsesquioxane. ACS Appl. Mater. Interfaces 2011, 3, 546–552. [Google Scholar] [CrossRef]
- Cuce, E.; Cuce, P.M.; Wood, C.J.; Riffat, S.B. Toward aerogel based thermal superinsulation in buildings: A comprehensive review. Renew. Sustain. Energy Rev. 2014, 34, 273–299. [Google Scholar] [CrossRef]
- Liu, B.-W.; Cao, M.; Zhang, Y.-Y.; Wang, Y.-Z.; Zhao, H.-B. Multifunctional protective aerogel with superelasticity over -196 to 500 °C. Nano Res. 2022, 15, 7797–7805. [Google Scholar] [CrossRef]
- Chen, M.-J.; Lin, Y.-C.; Wang, X.-N.; Zhong, L.; Li, Q.-L.; Liu, Z.-G. Influence of Cuprous Oxide on Enhancing the Flame Retardancy and Smoke Suppression of Epoxy Resins Containing Microencapsulated Ammonium Polyphosphate. Am. Chem. Soc. 2015, 54, 12705–12713. [Google Scholar] [CrossRef]
- Chen, M.-J.; Xu, Y.-J.; Rao, W.-H.; Huang, J.-Q.; Wang, X.-L.; Chen, L. Influence of Valence and Structure of Phosphorus-Containing Melamine Salts on the Decomposition and Fire Behaviors of Flexible Polyurethane Foams. Am. Chem. Soc. 2014, 53, 8773–8783. [Google Scholar] [CrossRef]
- Battig, A.; Müller, P.; Bertin, A. Hyperbranched rigid aromatic phosphorus-containing flame retardants for epoxy resins. Macromol. Mater. Eng. 2021, 306, 2000731. [Google Scholar] [CrossRef]
- Kolibaba, T.; Lazar, S.; Grunlan, J. Facile two-step phosphazine-based network coating for flame retardant cotton. Cellulose 2020, 27, 4123–4132. [Google Scholar]
- Haubold, T.; Hartwig, A.; Koschek, K. Synthesis and application studies of DOPO-Based organophosphorous derivatives to modify the thermal behavior of polybenzoxazine. Polymers 2022, 14, 606. [Google Scholar] [CrossRef]
- He, J.; Hou, D.-F.; Ma, H.-B.; Li, X.-Y. Preparation of phosphorus-containing cyanate resin with low curing temperature while excellent flame resistance and dielectric properties. J. Macromol. Sci. Part A 2019, 56, 629–639. [Google Scholar] [CrossRef]
- Liu, Y.-F.; Liao, C.-Y.; Hao, Z.-Z.; Luo, X.-X.; Jing, S.-S.; Run, M.-T. The polymerization behavior and thermal properties of benzoxazine based on o-allylphenol and 4,40-diaminodiphenyl methane. React. Funct. Polym. 2014, 75, 9–15. [Google Scholar] [CrossRef]
- Lorjai, P.; Wongkasemjit, S.; Chaisuwan, T.; Jamieson, A.-M. Significant enhancement of thermal stability in the non-oxidative thermal degradation of bisphenol-A/aniline based polybenzoxazine aerogel. Polym. Degrad. Stab. 2011, 96, 708–718. [Google Scholar] [CrossRef]
- Jian, R.-K.; Lin, X.-B.; Liu, Z.-Q.; Zhang, W.; Zhang, J.; Zhang, L.; Li, Z.; Wang, D.-Y. Rationally designed zinc borate@ZIF-8 core-shell nanorods for curing epoxy resins along with low flammability and high mechanical property. Composites 2020, 200, 108349. [Google Scholar] [CrossRef]
- Peng, C.-H.; Chen, T.; Zeng, B.-R.; Chen, G.-R.; Yuan, C.-H.; Xu, Y.-T.; Dai, L.-Z. Anderson-type polyoxometalate-based hybrid with high flame retardant efficiency for the preparation of multifunctional epoxy resin nanocomposites. Composites 2020, 186, 107780. [Google Scholar] [CrossRef]
- Ferrari, A.; Meyer, J.; Casiraghi, C. Raman Spectrum of Graphene and Graphene Layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.-Y.; Lin, Y.-C.; Tawiah, B.; Sun, J.; Yuen, R.K.; Fei, B. DOPO-decorated two-dimensional MXene nanosheets for flame-retardant, ultraviolet-protective, and reinforced polylactide composites. ACS Appl. Mater. Interfaces 2021, 13, 21876–21887. [Google Scholar] [CrossRef] [PubMed]
- Kundu, C.; Gangireddy, S.; Song, L.; Hu, Y. Flame retardant treatments for polyamide 66 textiles: Analysis the role of phosphorus compounds. Polym. Degrad. Stab. 2020, 182, 109376. [Google Scholar] [CrossRef]
- Zeng, F.-R.; Men, X.-F.; Chen, M.-J.; Liu, B.-W.; Han, Q.-W.; Huang, S.-C.; Zhao, H.-B.; Wang, Y.-Z. Molecular-micron multiscale toughening and flame retarding for polyurethane foams. Chem. Eng. J. 2023, 454, 140023. [Google Scholar] [CrossRef]


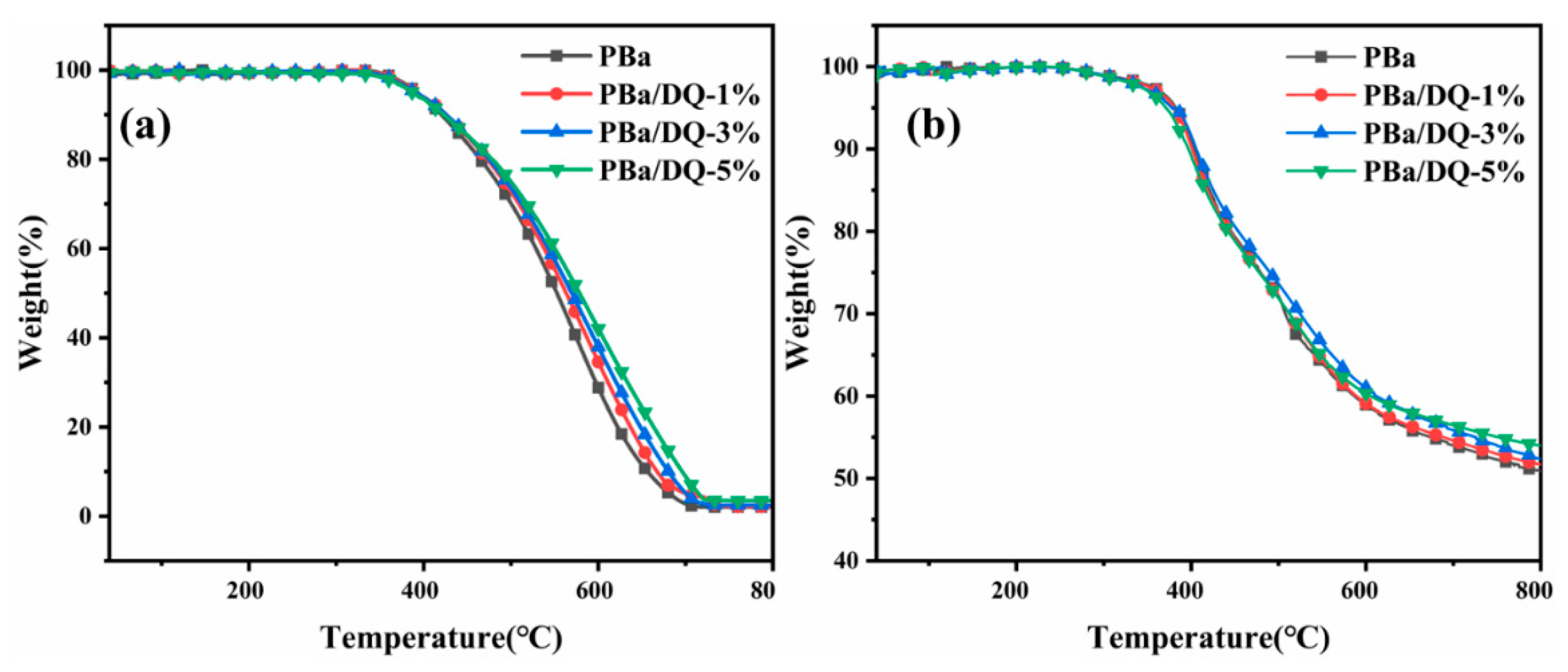



| Sample | Tonset (°C) | Tmax (°C) | Char Residue(%) | |||||
|---|---|---|---|---|---|---|---|---|
| 700 °C | 800 °C | |||||||
| Air | N2 | Air | N2 | Air | N2 | Air | N2 | |
| PBa | 392 | 382 | 521 | 405 | 2.7 | 54.0 | 2.0 | 50.9 |
| PBa/DQ-1% | 390 | 380 | 520 | 403 | 5.1 | 54.6 | 2.1 | 51.7 |
| PBa/DQ-3% | 389 | 377 | 517 | 403 | 5.2 | 55.8 | 2.4 | 52.4 |
| PBa/DQ-5% | 386 | 371 | 511 | 401 | 8.8 | 56.5 | 3.4 | 53.9 |
| Sample | TTI (s) | PHRR (kW/m2) | AvHRR (kW/m2) | THR (MJ/m2) | TSP (m2) |
|---|---|---|---|---|---|
| PBr | 86 | 312 | 98 | 79.0 | 109 |
| PBa | 35 | 253 | 64 | 23.3 | 19 |
| PBa/DQ-1% | 32 | 225 | 57 | 20.9 | 15 |
| PBa/DQ-3% | 29 | 201 | 51 | 18.8 | 10 |
| PBa/DQ-5% | 27 | 172 | 46 | 17.1 | 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Que, Y.; Zhao, C.; Wei, J.; Yang, F.; Li, H.; Cheng, J.; Xiang, D.; Wu, Y.; Wang, B. Phosphorus-Containing Polybenzoxazine Aerogels with Efficient Flame Retardation and Thermal Insulation. Int. J. Mol. Sci. 2023, 24, 4314. https://doi.org/10.3390/ijms24054314
Que Y, Zhao C, Wei J, Yang F, Li H, Cheng J, Xiang D, Wu Y, Wang B. Phosphorus-Containing Polybenzoxazine Aerogels with Efficient Flame Retardation and Thermal Insulation. International Journal of Molecular Sciences. 2023; 24(5):4314. https://doi.org/10.3390/ijms24054314
Chicago/Turabian StyleQue, Yusheng, Chunxia Zhao, Jixuan Wei, Fahong Yang, Hui Li, Jinbo Cheng, Dong Xiang, Yuanpeng Wu, and Bin Wang. 2023. "Phosphorus-Containing Polybenzoxazine Aerogels with Efficient Flame Retardation and Thermal Insulation" International Journal of Molecular Sciences 24, no. 5: 4314. https://doi.org/10.3390/ijms24054314
APA StyleQue, Y., Zhao, C., Wei, J., Yang, F., Li, H., Cheng, J., Xiang, D., Wu, Y., & Wang, B. (2023). Phosphorus-Containing Polybenzoxazine Aerogels with Efficient Flame Retardation and Thermal Insulation. International Journal of Molecular Sciences, 24(5), 4314. https://doi.org/10.3390/ijms24054314







