Aromatic Bromination Abolishes Deficits in Visuospatial Learning Induced by MDMA (“Ecstasy”) in Rats While Preserving the Ability to Increase LTP in the Prefrontal Cortex
Abstract
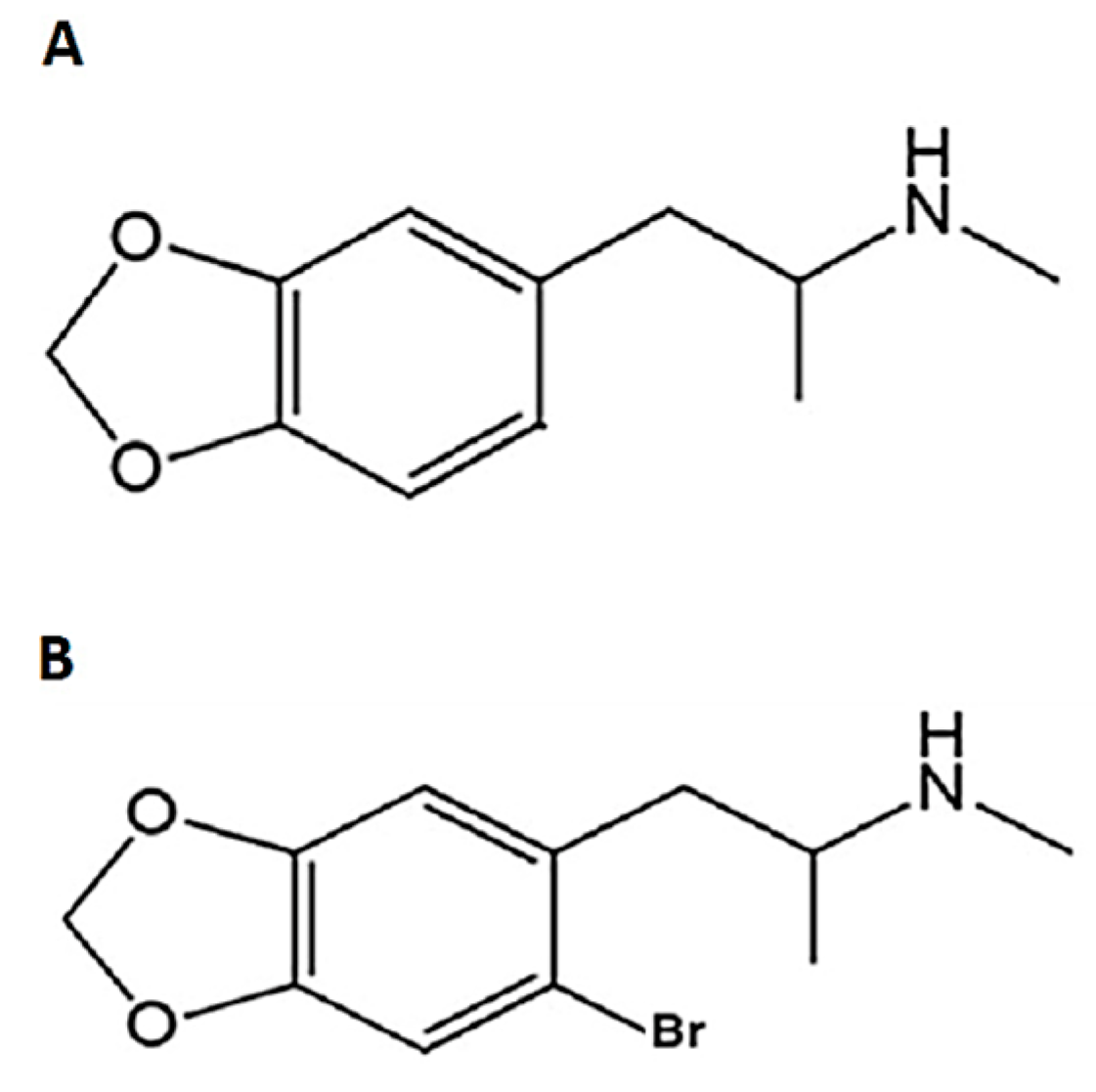
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Visuospatial Memory Performance
4.3. Determination of Prefrontal Cortex LTP
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nichols, D.E. Entactogens: How the Name for a Novel Class of Psychoactive Agents Originated. Front. Psychiatry 2022, 13, 863088. [Google Scholar] [CrossRef] [PubMed]
- Mithoefer, M.C.; Wagner, M.T.; Mithoefer, A.T.; Jerome, L.; Doblin, R. The safety and efficacy of ±3,4-methylenedioxymethamphetamine-assisted psychotherapy in subjects with chronic, treatment-resistant posttraumatic stress disorder: The first randomized controlled pilot study. J. Psychopharmacol. 2011, 25, 439–452. [Google Scholar] [CrossRef] [PubMed]
- Mithoefer, M.C.; Wagner, M.T.; Mithoefer, A.T.; Jerome, L.; Martin, S.F.; Yazar-Klosinski, B.; Michel, Y.; Brewerton, T.D.; Doblin, R. Durability of improvement in post-traumatic stress disorder symptoms and absence of harmful effects or drug dependency after 3,4-methylenedioxymethamphetamine-assisted psychotherapy: A prospective long-term follow-up study. J. Psychopharmacol. 2013, 27, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Feduccia, A.A.; Mithoefer, M.C. MDMA-assisted psychotherapy for PTSD: Are memory reconsolidation and fear extinction underlying mechanisms? Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 84 Pt A, 221–228. [Google Scholar] [CrossRef]
- Szafoni, S.; Więckiewicz, G.; Pudlo, R.; Gorczyca, P.; Piegza, M. Will MDMA-assisted psychotherapy become a breakthrough in treatment-resistant post-traumatic stress disorder? A critical narrative review. Psychiatr. Pol. 2022, 56, 823–836. [Google Scholar] [CrossRef]
- Bouso, J.C.; Doblin, R.; Farré, M.; Alcázar, M.A.; Gómez-Jarabo, G. MDMA-assisted psychotherapy using low doses in a small sample of women with chronic posttraumatic stress disorder. J. Psychoactive Drugs 2008, 40, 225–236. [Google Scholar] [CrossRef]
- Danforth, A.L.; Struble, C.M.; Yazar-Klosinski, B.; Grob, C.S. MDMA-assisted therapy: A new treatment model for social anxiety in autistic adults. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 64, 237–249. [Google Scholar] [CrossRef]
- Sessa, B. Why MDMA therapy for alcohol use disorder? And why now? Neuropharmacology 2018, 142, 83–88. [Google Scholar] [CrossRef]
- Sáez-Briones, P.; Hernández, A. MDMA (3,4-Methylenedioxymethamphetamine) Analogues as Tools to Characterize MDMA-Like Effects: An Approach to Understand Entactogen Pharmacology. Curr. Neuropharmacol. 2013, 11, 521–534. [Google Scholar] [CrossRef]
- Sitte, H.H.; Freissmuth, M. The reverse operation of Na+/Cl−-coupled neurotransmitter transporters--why amphetamines take two to tango. J. Neurochem. 2010, 112, 340–355. [Google Scholar] [CrossRef]
- Shulgin, A.; Shulgin, A. Pihkal: A Chemical History; Transform Press: Berkeley, CA, USA, 1991; pp. 1–978. ISBN o-9630096-0-5. [Google Scholar]
- Oeri, H.E. Beyond ecstasy: Alternative entactogens to 3,4-methylenedioxymethamphetamine with potential applications in psychotherapy. J. Psychopharmacol. 2021, 35, 512–536. [Google Scholar] [CrossRef]
- Hardegger, L.A.; Kuhn, B.; Spinnler, B.; Anselm, L.; Ecabert, R.; Stihle, M.; Gsell, B.; Thoma, R.; Diez, J.; Benz, J.; et al. Systematic investigation of halogen bonding in protein-ligand interactions. Angew. Chem. Int. Ed. Engl. 2011, 50, 314–318. [Google Scholar] [CrossRef]
- Sáez-Briones, P.; Castro-Castillo, V.; Díaz-Véliz, G.; Valladares, L.; Barra, R.; Hernández, A.; Cassels, B.K. Aromatic Bromination Abolishes the Psychomotor Features and Pro-social Responses of MDMA (“Ecstasy”) in Rats and Preserves Affinity for the Serotonin Transporter (SERT). Front. Pharmacol. 2019, 10, 157. [Google Scholar] [CrossRef]
- Ghaderi, M.; Rezayof, A.; Vousooghi, N.; Zarrindast, M.R. Dorsal hippocampal NMDA receptors mediate the interactive effects of arachidonylcyclopropylamide and MDMA/ecstasy on memory retrieval in rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 66, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Arias-Cavieres, A.; Rozas, C.; Reyes-Parada, M.; Barrera, N.; Pancetti, F.; Loyola, S.; Lorca, R.A.; Zeise, M.L.; Morales, B. MDMA (“ecstasy”) impairs learning in the Morris Water Maze and reduces hippocampal LTP in young rats. Neurosci. Lett. 2010, 469, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Taghizadeh, G.; Mehdizadeh, H.; Lavasani, H.; Hosseinzadeh Ardakani, Y.; Foroumadi, A.; Halvaei Khankahdani, Z.; Moshtagh, A.; Pourahmad, J.; Sharifzadeh, M.; Rouini, M.R. Dose concentration and spatial memory and brain mitochondrial function association after 3,4-methylenedioxymethamphetamine (MDMA) administration in rats. Arch. Toxicol. 2020, 94, 911–925. [Google Scholar] [CrossRef] [PubMed]
- Eslami, S.M.; Khorshidi, L.; Ghasemi, M.; Rashidian, A.; Mirghazanfari, M.; Nezhadi, A.; Chamanara, M.; Mirjani, R. Protective effects of atorvastatin and rosuvastatin on 3,4-methylenedioxymethamphetamine (MDMA)-induced spatial learning and memory impairment. Inflammopharmacology 2021, 29, 1807–1818. [Google Scholar] [CrossRef]
- Pantoni, M.M.; Anagnostaras, S.G. Cognitive Effects of MDMA in Laboratory Animals: A Systematic Review Focusing on Dose. Pharmacol. Rev. 2019, 71, 413–449. [Google Scholar] [CrossRef]
- Blumenfeld, R.S.; Ranganath, C. The lateral prefrontal cortex and human long-term memory. Handb. Clin. Neurol. 2019, 163, 221–235. [Google Scholar] [CrossRef]
- Chapman, C.A.; Trepel, C.; Ivanco, T.L.; Froc, D.J.; Wilson, K.; Racine, R.J. Changes in field potentials and membrane currents in rat sensorimotor cortex following repeated tetanization of the corpus callosum in vivo. Cereb. Cortex 1998, 8, 730–742. [Google Scholar] [CrossRef]
- Trepel, C.; Racine, R.J. Long-term potentiation in the neocortex of the adult, freely moving rat. Cereb. Cortex 1998, 8, 719–729. [Google Scholar] [CrossRef]
- Barra, R.; Soto-Moyano, R.; Valladares, L.; Morgan, C.; Pérez, H.; Burgos, H.; Olivares, R.; Sáez-Briones, P.; Laurido, C.; Hernández, A. Knockdown of α2C-adrenoceptors in the occipital cortex rescued long-term potentiation in hidden prenatally malnourished rats. Neurobiol. Learn. Mem. 2012, 98, 228–234. [Google Scholar] [CrossRef]
- Sáez-Briones, P.; Soto-Moyano, R.; Burgos, H.; Castillo, A.; Valladares, L.; Morgan, C.; Pérez, H.; Barra, R.; Constandil, L.; Laurido, C.; et al. β2-adrenoceptor stimulation restores frontal cortex plasticity and improves visuospatial performance in hidden-prenatally-malnourished young-adult rats. Neurobiol. Learn. Mem. 2015, 119, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kay, C.; Harper, D.N.; Hunt, M. Differential effects of MDMA and scopolamine on working versus reference memory in the radial arm maze task. Neurobiol. Learn Mem. 2010, 93, 151–156. [Google Scholar] [CrossRef] [PubMed]
- McNamara, M.G.; Kelly, J.P.; Leonard, B.E. Some behavioural and neurochemical aspects of subacute ±3,4-methylenedioxymethamphetamine administration in rats. Pharmacol. Biochem. Behav. 1995, 52, 479–484. [Google Scholar] [CrossRef]
- Rodsiri, R.; Spicer, C.; Green, A.R.; Marsden, C.A.; Fone, K.C. Acute concomitant effects of MDMA binge dosing on extracellular 5-HT, locomotion and body temperature and the long-term effect on novel object discrimination in rats. Psychopharmacology 2011, 213, 365–376. [Google Scholar] [CrossRef]
- Sajadi, A.; Amiri, I.; Gharebaghi, A.; Komaki, A.; Asadbegi, M.; Shahidi, S.; Mehdizadeh, M.; Soleimani Asl, S. Treadmill exercise alters ecstasy- induced long- term potentiation disruption in the hippocampus of male rats. Metab. Brain Dis. 2017, 32, 1603–1607. [Google Scholar] [CrossRef] [PubMed]
- Morini, R.; Mlinar, B.; Baccini, G.; Corradetti, R. Enhanced hippocampal long-term potentiation following repeated MDMA treatment in Dark-Agouti rats. Eur. Neuropsychopharmacol. 2011, 21, 80–91. [Google Scholar] [CrossRef]
- Mlinar, B.; Stocca, G.; Corradetti, R. Endogenous serotonin facilitates hippocampal long-term potentiation at CA3/CA1 synapses. J. Neural. Transm. 2015, 122, 177–185. [Google Scholar] [CrossRef]
- Rozas, C.; Loyola, S.; Ugarte, G.; Zeise, M.L.; Reyes-Parada, M.; Pancetti, F.; Rojas, P.; Morales, B. Acutely applied MDMA enhances long-term potentiation in rat hippocampus involving D1/D5 and 5-HT2 receptors through a polysynaptic mechanism. Eur. Neuropsychopharmacol. 2012, 22, 584–595. [Google Scholar] [CrossRef]
- Wilson, D.A.; Racine, R.J. The postnatal development of post-activation potentiation in the rat neocortex. Brain Res. 1983, 283, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Conti, F.; Manzoni, T. The neurotransmitters and postsynaptic actions of callosally projecting neurons. Behav. Brain Res. 1994, 64, 37–53. [Google Scholar] [CrossRef]
- Eckert, M.J.; Racine, R.J. Metabotropic glutamate receptors contribute to neocortical synaptic plasticity in vivo. Neuroreport 2004, 15, 2685–2689. [Google Scholar] [CrossRef]
- Andrade, R. Serotonergic regulation of neuronal excitability in the prefrontal cortex. Neuropharmacology 2011, 61, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Joushi, S.; Esmaeilpour, K.; Masoumi-Ardakani, Y.; Esmaeili-Mahani, S.; Sheibani, V. Intranasal oxytocin administration facilitates the induction of long-term potentiation and promotes cognitive performance of maternally separated rats. Psychoneuroendocrinology 2021, 123, 105044. [Google Scholar] [CrossRef]
- Chen, G.; Greengard, P.; Yan, Z. Potentiation of NMDA receptor currents by dopamine D1 receptors in prefrontal cortex. Proc. Natl. Acad. Sci. USA 2004, 101, 2596–2600. [Google Scholar] [CrossRef]
- Zhou, H.C.; Sun, Y.Y.; Cai, W.; He, X.T.; Yi, F.; Li, B.M.; Zhang, X.H. Activation of β2-adrenoceptor enhances synaptic potentiation and behavioral memory via cAMP-PKA signaling in the medial prefrontal cortex of rats. Learn. Mem. 2013, 20, 274–284. [Google Scholar] [CrossRef]
- National Research Council. Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011; pp. 1–120. ISBN 978-0-309-38629-6. [Google Scholar]
- Burgos, H.; Castillo, A.; Flores, O.; Puentes, G.; Morgan, C.; Gatica, A.; Cofré, C.; Hernández, A.; Laurido, C.; Constandil, L. Effect of modafinil on learning performance and neocortical long-term potentiation in rats. Brain Res. Bull. 2010, 83, 238–244. [Google Scholar] [CrossRef]
- Flores, O.; Pérez, H.; Valladares, L.; Morgan, C.; Gatica, A.; Burgos, H.; Olivares, R.; Hernández, A. Hidden prenatal malnutrition in the rat: Role of β₁-adrenoceptors on synaptic plasticity in the frontal cortex. J. Neurochem. 2011, 119, 314–323. [Google Scholar] [CrossRef]
- Pellegrino, L.J.; Cushman, A.J. A Stereotaxic Atlas of the Rat Brain; Appleton-Century Crofts: New York, NY, USA, 1967; ISBN 0306500639. [Google Scholar]
- Soto-Moyano, R.; Valladares, L.; Sierralta, W.; Pérez, H.; Mondaca, M.; Fernández, V.; Burgos, H.; Hernández, A. Mild prenatal protein malnutrition increases alpha2C-adrenoceptor density in the cerebral cortex during postnatal life and impairs neocortical long-term potentiation and visuo-spatial performance in rats. J. Neurochem. 2005, 93, 1099–1109. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sáez-Briones, P.; Palma, B.; Burgos, H.; Barra, R.; Hernández, A. Aromatic Bromination Abolishes Deficits in Visuospatial Learning Induced by MDMA (“Ecstasy”) in Rats While Preserving the Ability to Increase LTP in the Prefrontal Cortex. Int. J. Mol. Sci. 2023, 24, 3724. https://doi.org/10.3390/ijms24043724
Sáez-Briones P, Palma B, Burgos H, Barra R, Hernández A. Aromatic Bromination Abolishes Deficits in Visuospatial Learning Induced by MDMA (“Ecstasy”) in Rats While Preserving the Ability to Increase LTP in the Prefrontal Cortex. International Journal of Molecular Sciences. 2023; 24(4):3724. https://doi.org/10.3390/ijms24043724
Chicago/Turabian StyleSáez-Briones, Patricio, Boris Palma, Héctor Burgos, Rafael Barra, and Alejandro Hernández. 2023. "Aromatic Bromination Abolishes Deficits in Visuospatial Learning Induced by MDMA (“Ecstasy”) in Rats While Preserving the Ability to Increase LTP in the Prefrontal Cortex" International Journal of Molecular Sciences 24, no. 4: 3724. https://doi.org/10.3390/ijms24043724
APA StyleSáez-Briones, P., Palma, B., Burgos, H., Barra, R., & Hernández, A. (2023). Aromatic Bromination Abolishes Deficits in Visuospatial Learning Induced by MDMA (“Ecstasy”) in Rats While Preserving the Ability to Increase LTP in the Prefrontal Cortex. International Journal of Molecular Sciences, 24(4), 3724. https://doi.org/10.3390/ijms24043724






