How Nanoparticles Modify Adsorbed Proteins: Impact of Silica Nanoparticles on the Hemoglobin Active Site
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of the Oxygenation State on Hb Adsorption
2.2. Effect of the Hb Adsorption on Oxygenation Properties
2.3. Effect of Adsorption on the Heme–Iron Electronic and Structural Properties
2.4. Effect of Adsorption on the Heme Environment
2.5. Factors Controlling Hb Affinity
3. Materials and Methods
3.1. Hemoglobin Purification
3.2. Chemicals
3.3. Adsorption Isotherms
3.4. Hemoglobin Oxygenation
3.5. X-ray Absorption Spectroscopy and Data Analysis
3.6. Circular Dichroism in the Soret Region
4. Conclusions
- (i)
- The heme structure affects the adsorption mechanism; depending on the type of Hb, the affinity for the NPs, as well as the maximum number of adsorbed Hb molecules, is modified;
- (ii)
- Adsorption changes the affinity of Hb for O2;
- (iii)
- Soret circular dichroism shows changes in the angles of the vinyl functions upon adsorption;
- (iv)
- These modifications explain the change in the heme pocket environment, as also probed by X-ray absorption spectroscopy.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yamamoto, Y.; Hasegawa, K.; Shibata, T.; Momotake, A.; Ogura, T.; Yanagisawa, S.; Neya, S.; Suzuki, A.; Kobayashi, Y.; Saito, M.; et al. Effect of the Electron Density of the Heme Fe Atom on the Nature of Fe–O2 Bonding in Oxy Myoglobin. Inorg. Chem. 2021, 60, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Benabbas, A.; Sun, Y.; Poulos, T.L.; Champion, P.M. Ultrafast CO Kinetics in Heme Proteins: Adiabatic Ligand Binding and Heavy Atom Tunneling. J. Am. Chem. Soc. 2017, 139, 15738–15747. [Google Scholar] [CrossRef]
- Jahr, J.S.; Guinn, N.R.; Lowery, D.R.; Shore-Lesserson, L.; Shander, A. Blood Substitutes and Oxygen Therapeutics: A Review. Anesth. Analg. 2021, 132, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Devineau, S.; Kiger, L.; Galacteros, F.; Baudin-Creuza, V.; Marden, M.; Renault, J.P.; Pin, S. Manipulating hemoglobin oxygenation using silica nanoparticles: A novel prospect for artificial oxygen carriers. Blood Adv. 2018, 2, 90–94. [Google Scholar] [CrossRef]
- Spahn, D.R. Artificial oxygen carriers: A new future? Crit. Care 2018, 22, 46. [Google Scholar] [CrossRef]
- Marichal, L.; Giraudon-Colas, G.; Cousin, F.; Thill, A.; Labarre, J.; Boulard, Y.; Aude, J.-C.; Pin, S.; Renault, J.P. Protein–Nanoparticle Interactions: What Are the Protein–Corona Thickness and Organization? Langmuir 2019, 35, 10831–10837. [Google Scholar] [CrossRef]
- Perutz, M.F. Stereochemistry of Cooperative Effects in Haemoglobin: Haem–Haem Interaction and the Problem of Allostery. Nature 1970, 228, 726–734. [Google Scholar] [CrossRef]
- Sanchez-Guzman, D.; Giraudon--Colas, G.; Marichal, L.; Boulard, Y.; Wien, F.; Degrouard, J.; Baeza-Squiban, A.; Pin, S.; Renault, J.P.; Devineau, S. In Situ Analysis of Weakly Bound Proteins Reveals Molecular Basis of Soft Corona Formation. ACS Nano 2020, 14, 9073–9088. [Google Scholar] [CrossRef] [PubMed]
- Bikiel, D.E.; Forti, F.; Boechi, L.; Nardini, M.; Luque, F.J.; Martí, M.A.; Estrin, D.A. Role of Heme Distortion on Oxygen Affinity in Heme Proteins: The Protoglobin Case. J. Phys. Chem. B 2010, 114, 8536–8543. [Google Scholar] [CrossRef] [PubMed]
- Kepp, K.P.; Dasmeh, P. Effect of Distal Interactions on O2 Binding to Heme. J. Phys. Chem. B 2013, 117, 3755–3770. [Google Scholar] [CrossRef] [PubMed]
- Kepp, K.P. Heme isomers substantially affect heme’s electronic structure and function. Phys. Chem. Chem. Phys. 2017, 19, 22355–22362. [Google Scholar] [CrossRef] [PubMed]
- Pauling, L. Nature of the Iron–Oxygen Bond in Oxyhæmoglobin. Nature 1964, 203, 182–183. [Google Scholar] [CrossRef]
- Weiss, J.J. Nature of the Iron–Oxygen Bond in Oxyhæmoglobin. Nature 1964, 202, 83–84. [Google Scholar] [CrossRef] [PubMed]
- Patrinos, G.P.; Giardine, B.; Riemer, C.; Miller, W.; Chui, D.H.K.; Anagnou, N.P.; Wajcman, H.; Hardison, R.C. Improvements in the HbVar database of human hemoglobin variants and thalassemia mutations for population and sequence variation studies. Nucleic Acids Res. 2004, 32, D537–D541. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.H.; Ghatge, M.S.; Safo, M.K. Hemoglobin: Structure, Function and Allostery. In Vertebrate and Invertebrate Respiratory Proteins, Lipoproteins and other Body Fluid Proteins; Hoeger, U., Harris, J.R., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 345–382. [Google Scholar]
- Orts-Gil, G.; Natte, K.; Drescher, D.; Bresch, H.; Mantion, A.; Kneipp, J.; Österle, W. Characterisation of silica nanoparticles prior to in vitro studies: From primary particles to agglomerates. J. Nanopart. Res. 2011, 13, 1593–1604. [Google Scholar] [CrossRef]
- Latour, R.A. The langmuir isotherm: A commonly applied but misleading approach for the analysis of protein adsorption behavior. J. Biomed. Mater. Res. Part A 2015, 103, 949–958. [Google Scholar] [CrossRef]
- Hill, A.V. The Combinations of Haemoglobin with Oxygen and with Carbon Monoxide. I. Biochem. J. 1913, 7, 471–480. [Google Scholar] [CrossRef]
- Shibayama, N.; Saigo, S. Fixation of the Quaternary Structures of Human Adult Haemoglobin by Encapsulation in Transparent Porous Silica Gels. J. Mol. Biol. 1995, 251, 203–209. [Google Scholar] [CrossRef]
- Adair, G.S.; Bock, A.V.; Field, H. The Hemoglobin System VI: The oxygen dissociation curve of Hemoglobin. J. Biol. Chem. 1925, 63, 529–545. [Google Scholar] [CrossRef]
- Hayashi, T.; Dejima, H.; Matsuo, T.; Sato, H.; Murata, D.; Hisaeda, Y. Blue Myoglobin Reconstituted with an Iron Porphycene Shows Extremely High Oxygen Affinity. J. Am. Chem. Soc. 2002, 124, 11226–11227. [Google Scholar] [CrossRef]
- Matsuo, T.; Dejima, H.; Hirota, S.; Murata, D.; Sato, H.; Ikegami, T.; Hori, H.; Hisaeda, Y.; Hayashi, T. Ligand Binding Properties of Myoglobin Reconstituted with Iron Porphycene: Unusual O2 Binding Selectivity against CO Binding1. J. Am. Chem. Soc. 2004, 126, 16007–16017. [Google Scholar] [CrossRef]
- Eisenberger, P.; Shulman, R.G.; Brown, G.S.; Ogawa, S. Structure-function relations in hemoglobin as determined by x-ray absorption spectroscopy. Proc. Natl. Acad. Sci. USA 1976, 73, 491–495. [Google Scholar] [CrossRef]
- Bianconi, A.; Dell’Ariccia, M.; Durham, P.J.; Pendry, J.B. Multiple-scattering resonances and structural effects in the x-ray-absorption near-edge spectra of Fe II and Fe III hexacyanide complexes. Phys. Rev. B 1982, 26, 6502–6508. [Google Scholar] [CrossRef]
- Kincaid, B.M.; Eisenberger, P.; Hodgson, K.O.; Doniach, S. X-ray absorption spectroscopy using synchrotron radiation for structural investigation of organometallic molecules of biological interest. Proc. Natl. Acad. Sci. USA 1975, 72, 2340–2342. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.A.; Green, E.; Mathews, I.I.; Benfatto, M.; Hodgson, K.O.; Hedman, B.; Sarangi, R. X-ray absorption spectroscopic investigation of the electronic structure differences in solution and crystalline oxyhemoglobin. Proc. Natl. Acad. Sci. USA 2013, 110, 16333–16338. [Google Scholar] [CrossRef] [PubMed]
- Pin, S.; Valat, P.; Cortes, R.; Michalowicz, A.; Alpert, B. Ligand binding processes in hemoglobin. Chemical reactivity of iron studied by XANES spectroscopy. Biophys. J. 1985, 48, 997–1001. [Google Scholar] [CrossRef]
- Longa, S.D.; Arcovito, A.; Benfatto, M.; Congiu-Castellano, A.; Girasole, M.; Hazemann, J.L.; Bosco, A.L. Redox-Induced Structural Dynamics of Fe-Heme Ligand in Myoglobin by X-ray Absorption Spectroscopy. Biophys. J. 2003, 85, 549–558. [Google Scholar] [CrossRef]
- Congiu Castellano, A. Oxygen Binding Site Structure in Hemoproteins by XANES; Springer: Berlin/Heidelberg, Germany, 1987; pp. 89–98. [Google Scholar]
- Zentz, C.; El Antri, S.; Pin, S.; Cortes, R.; Massat, A.; Simon, M.; Alpert, B. Alteration of heme axial ligands in hemoglobin by organic solvents analyzed by CD, FTIR and XANES techniques. Biochemistry 1991, 30, 2804–2810. [Google Scholar] [CrossRef]
- D’Angelo, P.; Lapi, A.; Migliorati, V.; Arcovito, A.; Benfatto, M.; Roscioni, O.M.; Meyer-Klaucke, W.; Della-Longa, S. X-ray Absorption Spectroscopy of Hemes and Hemeproteins in Solution: Multiple Scattering Analysis. Inorg. Chem. 2008, 47, 9905–9918. [Google Scholar] [CrossRef]
- Nagai, M.; Nagai, Y.; Imai, K.; Neya, S. Circular Dichroism of Hemoglobin and Myoglobin. Chirality 2014, 26, 438–442. [Google Scholar] [CrossRef]
- Woody, R.W.; Pescitelli, G. The Role of Heme Chirality in the Circular Dichroism of Heme Proteins. Z. Fur Nat. Sect. A-A J. Phys. Sci. 2014, 69, 313–325. [Google Scholar] [CrossRef]
- Geraci, G.; Parkhurst, L.J. Circular dichroism spectra of hemoglobins. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1981; Volume 76, pp. 262–275. [Google Scholar]
- Sugita, Y.; Nagai, M.; Yoneyama, Y. Circular Dichroism of Hemoglobin in Relation to the Structure Surrounding the Heme. J. Biol. Chem. 1971, 246, 383–388. [Google Scholar] [CrossRef]
- Hu, D.; Kluger, R. Functional Cross-Linked Hemoglobin Bis-tetramers: Geometry and Cooperativity. Biochemistry 2008, 47, 12551–12561. [Google Scholar] [CrossRef]
- Woody, R.W.; Kiefl, C.; Sreerama, N.; Lu, Y.; Qiu, Y.; Shelnutt, J.A. Molecular Dynamics Simulations of Carbonmonoxy Myoglobin and Calculations of Heme Circular Dichroism. In Insulin & Related Proteins—Structure to Function and Pharmacology; Dieken, M.L., Federwisch, M., De Meyts, P., Eds.; Springer: Dordrecht, The Netherlands, 2002; pp. 233–248. [Google Scholar]
- Perutz, M.F. Mechanisms Regulating the Reactions of Human Hemoglobin with Oxygen and Carbon Monoxide. Annu. Rev. Physiol. 1990, 52, 1–26. [Google Scholar] [CrossRef]
- Pin, S.; Cortes, R.; Alpert, B. XANES spectroscopy of carp hemoglobin-iron in correlation with the affinity changes of the protein for ligand. FEBS Lett. 1986, 208, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Devineau, S.; Zanotti, J.-M.; Loupiac, C.; Zargarian, L.; Neiers, F.; Pin, S.; Renault, J.P. Myoglobin on Silica: A Case Study of the Impact of Adsorption on Protein Structure and Dynamics. Langmuir 2013, 29, 13465–13472. [Google Scholar] [CrossRef]
- Shibayama, N.; Sato-Tomita, A.; Ohki, M.; Ichiyanagi, K.; Park, S.Y. Direct observation of ligand migration within human hemoglobin at work. Proc. Natl. Acad. Sci. USA 2020, 117, 4741–4748. [Google Scholar] [CrossRef]
- Pandey, P.; Singh, S.P.; Arya, S.K.; Gupta, V.; Datta, M.; Singh, S.; Malhotra, B.D. Application of Thiolated Gold Nanoparticles for the Enhancement of Glucose Oxidase Activity. Langmuir 2007, 23, 3333–3337. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Narsimhan, G. Effect of surface concentration on secondary and tertiary conformational changes of lysozyme adsorbed on silica nanoparticles. Biochim. Biophys. Acta BBA-Proteins Proteom. 2008, 1784, 1694–1701. [Google Scholar] [CrossRef]
- Zoungrana, T.; Findenegg, G.H.; Norde, W. Structure, Stability, and Activity of Adsorbed Enzymes. J. Colloid Interface Sci. 1997, 190, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Perutz, M.F. Preparation of Haemoglobin crystals. J. Cryst. Growth 1968, 2, 54–56. [Google Scholar] [CrossRef]
- Jelkmann, W.; Bauer, C. What is the best method to remove 2,3-diphosphoglycerate from hemoglobin? Anal. Biochem. 1976, 75, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, R.; Alpert, Y.; Leterrier, F.; Williams, R.J.P. Visible absorption and electron spin resonance spectra of the isolated chains of human hemoglobin. Discussion of chain-mediated heme-heme interaction. Biochemistry 1969, 8, 2862–2867. [Google Scholar] [CrossRef]
- Meng, F.; Alayash, A.I. Determination of extinction coefficients of human hemoglobin in various redox states. Anal. Biochem. 2017, 521, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Marichal, L.; Degrouard, J.; Gatin, A.; Raffray, N.; Aude, J.-C.; Boulard, Y.; Combet, S.; Cousin, F.; Hourdez, S.; Mary, J.; et al. From Protein Corona to Colloidal Self-Assembly: The Importance of Protein Size in Protein–Nanoparticle Interactions. Langmuir 2020, 36, 8218–8230. [Google Scholar] [CrossRef]
- Barahona, F.; Geiss, O.; Urbán, P.; Ojea-Jimenez, I.; Gilliland, D.; Barrero-Moreno, J. Simultaneous Determination of Size and Quantification of Silica Nanoparticles by Asymmetric Flow Field-Flow Fractionation Coupled to ICPMS Using Silica Nanoparticles Standards. Anal. Chem. 2015, 87, 3039–3047. [Google Scholar] [CrossRef] [PubMed]
- Devineau, S.; Zargarian, L.; Renault, J.P.; Pin, S. Structure and Function of Adsorbed Hemoglobin on Silica Nanoparticles: Relationship between the Adsorption Process and the Oxygen Binding Properties. Langmuir 2017, 33, 3241–3252. [Google Scholar] [CrossRef] [PubMed]
- Ravel, B.; Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: Data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 2005, 12, 537–541. [Google Scholar] [CrossRef]
- Filipponi, A.; DiCicco, A.; Natoli, C.R. X-ray-absorption spectroscopy and n-body distribution functions in condensed matter. I. Theory. Phys. Rev. B 1995, 52, 15122–15134. [Google Scholar] [CrossRef] [PubMed]
- Filipponi, A.; DiCicco, A. X-ray-absorption spectroscopy and n-body distribution functions in condensed matter. II. Data analysis and applications. Phys. Rev. B 1995, 52, 15135–15149. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Yokoyama, T.; Shibayama, N.; Shiro, Y.; Tame, J.R.H. 1.25 angstrom resolution crystal structures of human haemoglobin in the oxy, deoxy and carbonmonoxy forms. J. Mol. Biol. 2006, 360, 690–701. [Google Scholar] [CrossRef]
- Rangus, M.; Mazaj, M.; Dražić, G.; Popova, M.; Tušar, N.N. Active Iron Sites of Disordered Mesoporous Silica Catalyst FeKIL-2 in the Oxidation of Volatile Organic Compounds (VOC). Materials 2014, 7, 4243–4257. [Google Scholar] [CrossRef]
- Gill, S.J.; Di Cera, E.; Doyle, M.L.; Bishop, G.A.; Robert, C.H. Oxygen binding constants for human hemoglobin tetramers. Biochemistry 1987, 26, 3995–4002. [Google Scholar] [CrossRef]
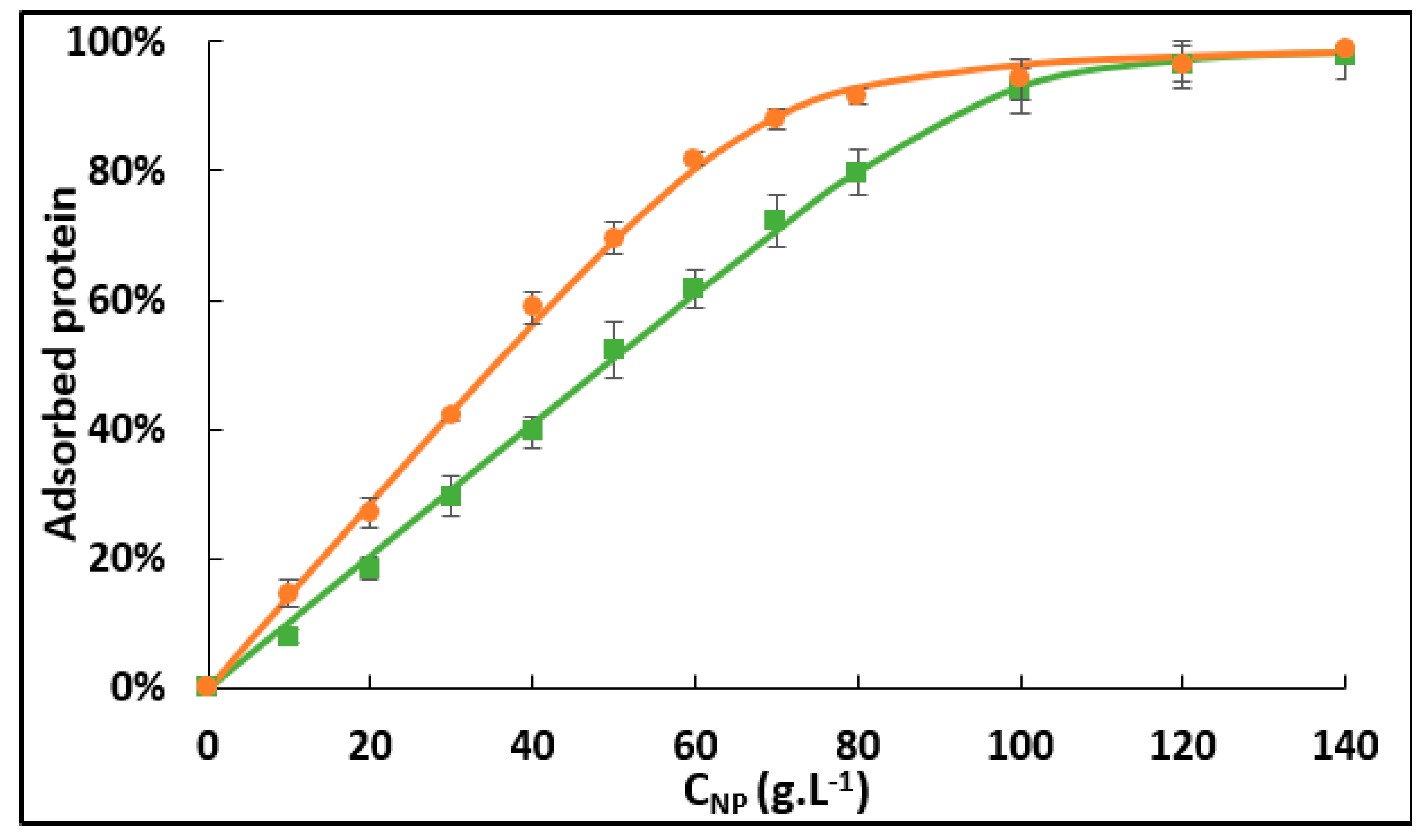

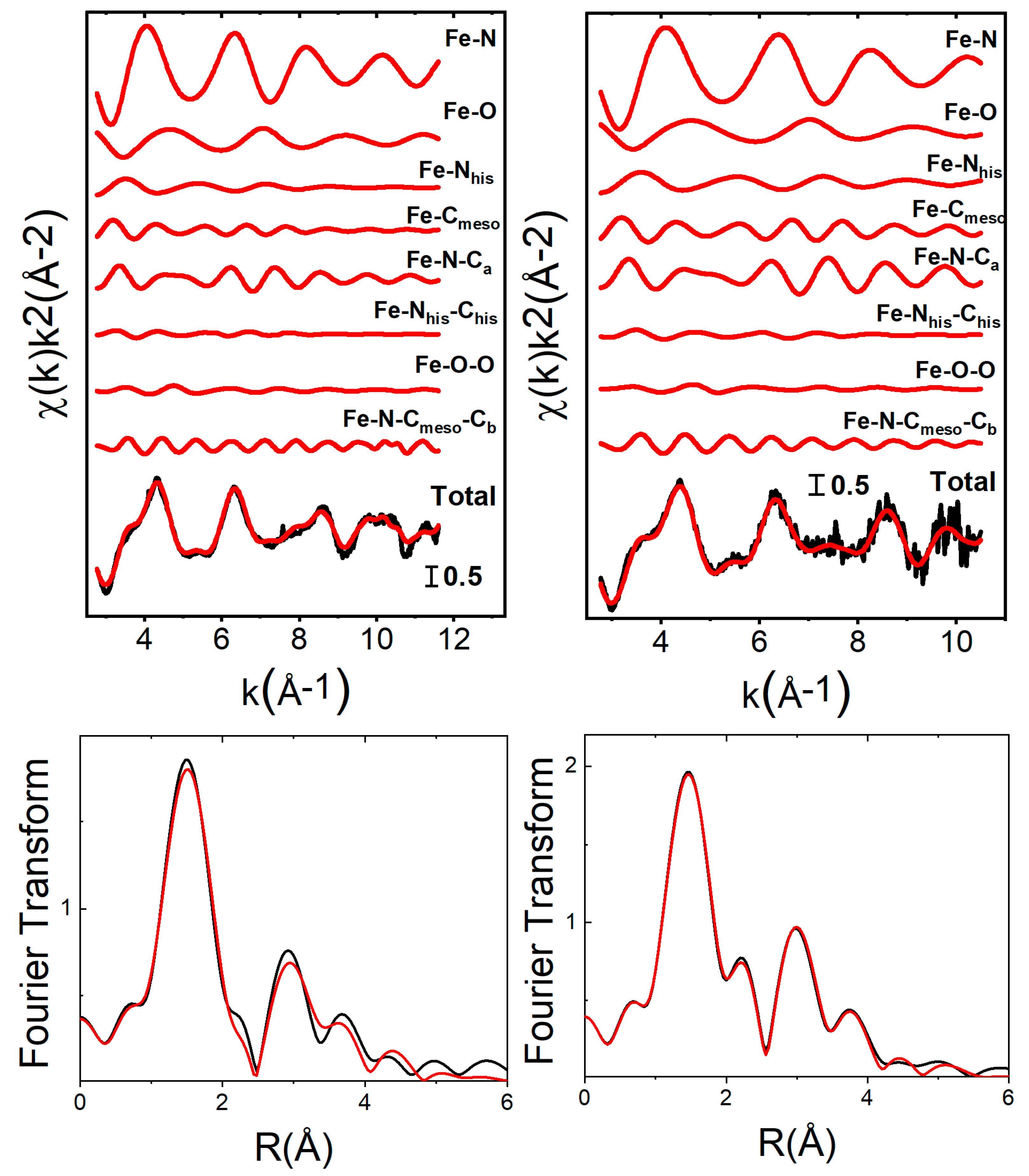
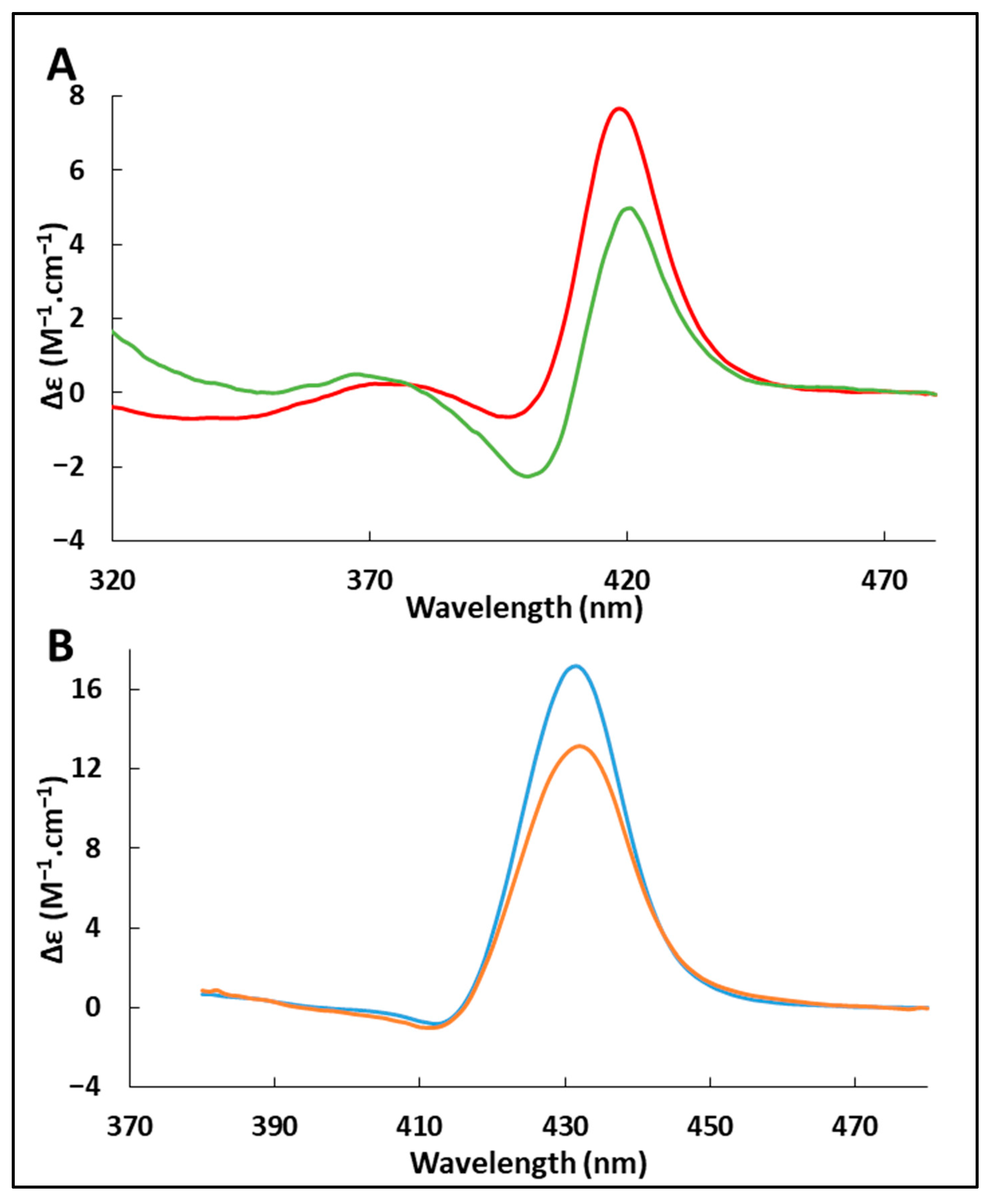
| nsites | m∞ (mg.m−2) | Kads (L.mol−1) | ΔrG0 (kJ.mol−1) | |
|---|---|---|---|---|
| deoxyHb | 170 ± 7 | 2.3 ± 0.1 | 5.1 104 | −27 |
| oxyHb | 120 ± 8 | 1.6 ± 0.1 | 1.2 105 | −29 |
| Free Hb | Adsorbed Hb | |
|---|---|---|
| P1/2 (mmHg) | 9.4 | 6.0 |
| nHill | 2.9 | 2.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giraudon--Colas, G.; Devineau, S.; Marichal, L.; Barruet, E.; Zitolo, A.; Renault, J.-P.; Pin, S. How Nanoparticles Modify Adsorbed Proteins: Impact of Silica Nanoparticles on the Hemoglobin Active Site. Int. J. Mol. Sci. 2023, 24, 3659. https://doi.org/10.3390/ijms24043659
Giraudon--Colas G, Devineau S, Marichal L, Barruet E, Zitolo A, Renault J-P, Pin S. How Nanoparticles Modify Adsorbed Proteins: Impact of Silica Nanoparticles on the Hemoglobin Active Site. International Journal of Molecular Sciences. 2023; 24(4):3659. https://doi.org/10.3390/ijms24043659
Chicago/Turabian StyleGiraudon--Colas, Gaël, Stéphanie Devineau, Laurent Marichal, Elodie Barruet, Andrea Zitolo, Jean-Philippe Renault, and Serge Pin. 2023. "How Nanoparticles Modify Adsorbed Proteins: Impact of Silica Nanoparticles on the Hemoglobin Active Site" International Journal of Molecular Sciences 24, no. 4: 3659. https://doi.org/10.3390/ijms24043659
APA StyleGiraudon--Colas, G., Devineau, S., Marichal, L., Barruet, E., Zitolo, A., Renault, J.-P., & Pin, S. (2023). How Nanoparticles Modify Adsorbed Proteins: Impact of Silica Nanoparticles on the Hemoglobin Active Site. International Journal of Molecular Sciences, 24(4), 3659. https://doi.org/10.3390/ijms24043659





