Current Status of Cell-Based Therapies for Vitiligo
Abstract
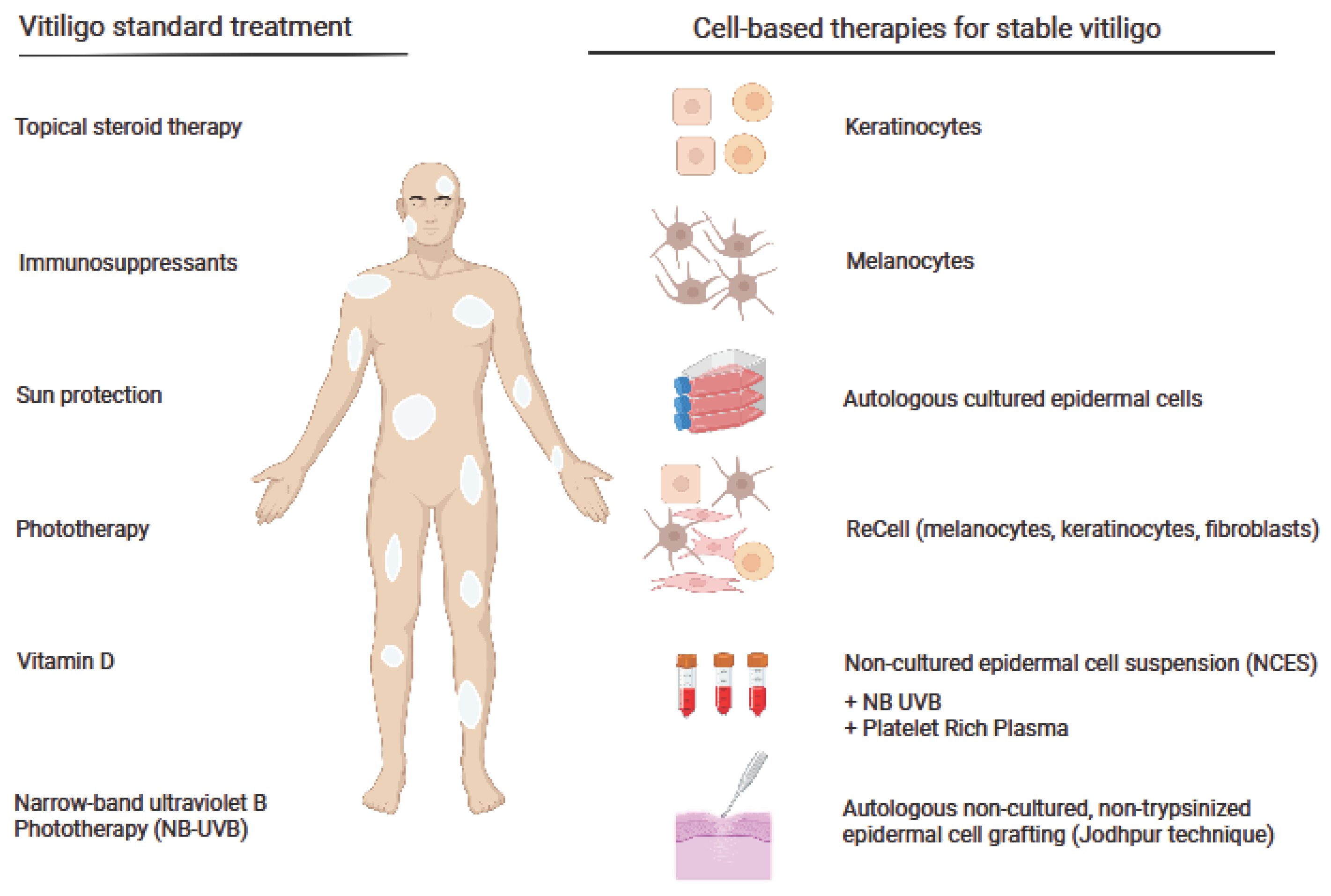
1. Background
2. Melanocytes and Keratinocytes as the Targets for Vitiligo Therapy
3. Melanocyte–Keratinocyte Cell Transplantation (MKCT)
4. ReCell System in Vitiligo Therapy
5. Autologous Non-Cultured, Non-Trypsinized Epidermal Cell Grafting
6. Cell Transplantations in Combination Therapy with a Narrowband Ultraviolet B (NB-UVB) or Autologous Platelet-Rich Plasma
7. Are Cell-Based Therapies Appropriate for All Vitiligo Patients? Limitations and Challenges
8. Conclusions
| Therapy | Vitiligo Characteristics | Patients Number | Age Range (Years) | Clinical Results | Evaluation Time | Reference | Year |
|---|---|---|---|---|---|---|---|
| Melanocyte transplantation | |||||||
| Cultured autologous melanocytes | Stable vitiligo | 25 | 13–72 | Almost complete repigmentation in 6 out of 11 cases, in 4 cases, 40–71% of the grafted achromatic area was repigmented | 1–20 months | Chen et al. [16] | 2000 |
| Autologous transplanted epidermal cell suspensions | Stable vitiligo or vitiligo with doubts about stability | 28 | 5–65 | A total of 77% repigmentation | 12 months | Van Geel et al. [1] | 2004 |
| Autologous cultured pure melanocyte suspension | Stable vitiligo | 120 | 7–72 | Overall, 84% of patients with localized vitiligo experienced 90% to 100% coverage | 6–66 months | Chen et al. [28] | 2004 |
| Autologous melanocyte rich cell suspension (non-cultured) and cultured melanocyte technique | Stable vitiligo | 27 | 21–30 | Over 90% repigmentation in 52.17% of cases with the autologous melanocyte-rich cell suspension technique and in 50% with the melanocyte culture technique | Up to 6 months | Pandya et al. [12] | 2005 |
| Autologous cultured pure melanocytes | Stable vitiligo for at least 6 months | 102 | 8–12, 13–17, Adults: mean 29 | Repigmentation of 50% or more in children, adolescents, and adults were 83.3%, 95.0% and 84.0% cases, respectively | At least 6 months | Hong et al. [17] | 2011 |
| Autologous melanocytes culture on a denuded amniotic membrane | Stable vitiligo | 4 | 13–29 | A total of 90–95% repigmentation in all patients | Up to 6 months | Redondo et al. [18] | 2011 |
| Autologous melanocytes transplantation | Stable vitiligo and developing vitiligo | 16 | 19–40 | In 87.5% of lesions, a repigmentation of >50%; no relapse was observed | 5 years | Zhu et al. [26] | 2017 |
| Autologous epidermal cell suspension | Stable vitiligo | 300 | Range not stated, mean 27.1 | Repigmentation stability remained in most treated patches | Up to 30 months | Orouji et al. [35] | 2017 |
| Autologous non-cultured and trypsinized melanocyte grafting | Stable vitiligo | 28 | Range not stated, mean 25.9 | Over 50% in the face and neck, trunk, upper extremity, and genitals in 57.4%, 20.4%, 16.7%, and 5.5% patients, respectively | 18 months | Ghorbani et al. [14] | 2022 |
| Hair follicle cell transplantation | |||||||
| Non-cultured extracted hair follicular outer root sheath (ORS) cell suspension transplantation | Stable vitiligo for at least 3 months | 14 | 17–32 | Nine out of fourteen patients achieved >75% repigmentation | 1–15 months | Mohanty et al. [19] | 2011 |
| Autologous non-cultured outer root sheath hair follicle cell suspension (NCORSHFS) | Stable vitiligo | 30 | 8–38 | The number of melanocytes and HFSC transplanted were significantly higher among patients achieving optimum (>75%) repigmentation | 24 weeks | Vinay et al. [20] | 2015 |
| Hair follicle outer root sheath cell transplantation (EHF ORS) | Stable vitiligo | 20 | 18–43 | Mean repigmentation of 80.15% with 90–100% in 60% of patients | 6 months | Shah et al. [21] | 2016 |
| Non-cultured, extracted follicular outer root sheath suspension (NC-EHF-ORS-CS) | Stable vitiligo | 2 | 18–36 | The mean repigmentation was 52% and >75% repigmentation in 32% of patients | 6 months | Kumar et al. [22] | 2018 |
| Autologous hair follicle cell derived melanocytes transplantation | Stable vitiligo | 26 | 19–50 | Overall, 34.6% of patients achieved excellent repigmentation, 50% had good, 11.5% had fair, and 3.9% had poor repigmentation | 1 year | Shi et al. [23] | 2020 |
| Keratinocyte transplantation | |||||||
| Autologous cultured keratinocytes | Stable vitiligo | 27 | 9–48 | Twelve patients had 90% or more repigmentation after the first surgery, which increased by two cases when patients with multiple surgeries were included | At least 1 year | Matsuzaki and Kumagai [30] | 2013 |
| Melanocyte–keratinocyte transplantation | |||||||
| Cultured epithelial autographs with keratinocytes seeded at high density | Stable or active vitiligo | 5 | 32–71 | Out of five patients, repigmentation was only achieved in one | N/A | Phillips et al. [33] | 2001 |
| Melanocyte–keratinocyte cell transplantation (MKT) | Stable vitiligo | 184 | 9 to 70 | Overall, 53% in the generalized vitiligo and 84% in the segmental vitiligo group showed 95–100% repigmentation | Up to 1 year | Mulekar et al. [43] | 2003 |
| Autologous non-cultured melanocyte–keratinocyte cell transplantation (MKT) | Stale vitiligo for at least 1 year | 134 | At least 12 | Overall, 84% in the segmental and 73% in the focal vitiligo group showed 95–100% repigmentation | Up to 5 years | Mulekar et al. [47] | 2004 |
| Autologous non-cultured melanocyte–keratinocyte cell transplantation (MKT) | Stable genital vitiligo | 3 | 24–39 | Near-complete repigmentation observed in all patients | Up to 1 year | Mulekar et al. [49] | 2005 |
| Autologus melanocyte–keratinocyte cell transplantation (MKT) | Stable vitiligo (for at least 6 months) | 142 | 18–70 | Overall, 56% patients showed 95–100% repigmentation | Up to 6 years | Mulekar et al. [48] | 2005 |
| Noncultured melanocyte–keratinocyte cell transplantation (MKT) | Stable vitiligo (for at least 6 months) | 49 | 7–65 | For bilateral vitiligo, more than 50% of patients showed >65% repigmentation. For unilateral vitiligo, all but two patients treated for the eyelids vitiligo showed >65% repigmentation | N/A | Mulekar et al. [55] | 2009 |
| Autologous dissociated epidermal cell suspensions | Stable vitiligo | 10 | 17–52 | Overall, 76–100% repigmentation in 40% of patients | 6 months | Khodadadi et al. [34] | 2010 |
| Non-cultured melanocyte–keratinocyte transplantation (MKT) | Stable vitiligo | 25 | 8–45 | Overall, 23% of patients showed 90–100% repigmentation | 6–17 months | El-Zawahry et al. [56] | 2011 |
| Non-cultured melanocyte–keratinocyte transplantation (MKT) | Stable vitiligo | 8 | 13–43 | Of eight lesions treated with non-cultured MKT, four lesions showed 96–100%, one lesion 65–95%, and three lesions 0–25% repigmentation | 4 months | Toossi et al. [50] | 2011 |
| Melanocytes and keratinocytes transplantation using the sandpaper technique combined with dermabrasion or only dermabrasion | Stable vitiligo | 11 | 21–65 | At the end of treatment, both techniques showed similar repigmentation with repigmentation in nine cases (87% to 6%) of pigmenation in the transfer group and nine cases (94% to 5%) in the dermabrasion group | 3 months | Quezada et al. [52] | 2011 |
| Melanocyte–keratinocyte cell suspension with dermabrasion (MKT+ DA) and dermabrasion alone (DA) | Stable vitiligo | 11 | 35–48 | Slightly better pigmentation occurred with DA+MKT in 7 out of 11 patients | 12 months | Vazquez-Martinez et al. [51] | 2011 |
| Autologous melanocyte–keratinocyte transplantation (MKT) | Stable vitiligo | 23 | 18–60 | Overall, 95–100% repigmentation in 17% | 3–6 months | Huggins et al. [53] | 2012 |
| Autologous transplantation of non-cultured melanocyte–keratinocyte cell suspension (MKT) | Stable vitiligo | 20 | 10–50 | Overall, 25% of patients showed ≥90% Repigmentation; the best results were observed in face and neck | Up to 24 months | Ramos et al. [58] | 2017 |
| Autologous melanocyte–keratinocyte transplantation (MKT) | Stable vitiligo without fingertip involvement | 100 | 9–60 | MKT could maintain repigmentation for at least 72 months | 12–72 months | Silpa-Archa et al. [44] | 2017 |
| Autologous non-cultured keratinocyte–melanocyte suspension (MKT) | Stable vitiligo | 5 | Range not stated, mean 20 | Overall, 76–100% repigmentation in 60% of patients | 6 months | Benzekri and Gauthier [25] | 2017 |
| Autologous non-cultured melanocyte–keratinocyte transplantation (MKT) | Stable vitiligo | 602 | 4–67 | Overall, 84.3% of patients achieved ≥50% repigmentation at the 6th month evaluation; at 6 years, 23% showed relapse | 6 years | Altalhab et al. [46] | 2019 |
| Combination therapies | |||||||
| Non-cultured autologous melanocytes and keratinocytes combined with UVA or UVB | Stable vitiligo | 4 | 30–52 | Overall, 85–100% repigmentation achieved at 6 to 20 months | 6–20 months | van Geel et al. [68] | 2001 |
| Narrow-band ultraviolet B therapy for cultured autologous melanocyte transplantation patients | Stable vitiligo | 437 | 5–55 | A total of 20 sessions of NB-UVB treatment before transplantation and 30 sessions after transplantation gave the best repigmentation | 6 months | Zhang et al. [66] | 2014 |
| Cultured autologous melanocyte transplantation (CMT combined with narrowband ultraviolet B (NB-UVB) | Stable vitiligo | 8 | 7–28 | All patients treated with low-density CMT combined with NB-UVB obtained more than 90% repigmentation | 1 year | Yao et al. [67] | 2017 |
| Platelet-rich plasma (PRP) used to suspend non-cultured epidermal cell suspension (NCES) before transplantation | Stable vitiligo | 21 | Range not stated, mean 23.1 | Suspending NCES in PRP can result in significantly greater mean repigmentation | 6 months | Parambath et al. [70] | 2019 |
| ReCell | |||||||
| ReCell vs. conventional melanocyte–keratinocyte transplantation (MKT) | Stable vitiligo | 5 | 18–40 | Overall, 40% of lesions treated with ReCell showed 100% repigmentation, while 20% of lesions failed to repigment. Overall, 60% of lesions treated with conventional MKT showed 100% repigmentation and 20% failed to repigment | 4 months | Mulekar et al. [62] | 2008 |
| ReCell | Stable vitiligo | 15 | 18–45 | Repigmentation greater than 75% was recorded in 12 (80%) patients | Minimum 6 months | Cervelli et al. [61] | 2009 |
| ReCell | Stable vitiligo | 1 | N/A | The patient had >90% repigmentation | N/A | Cervelli et al. [62] | 2010 |
| Non-cultured cellular grafting | |||||||
| Autologous, non-cultured cellular grafting (MKTP) | Stable vitiligo for at least 6 months | 25 | 4–16 | Overall, 95–100% repigmentation in 62% of patients | 9–54 months | Mulekar et al. [60] | 2010 |
| Autologous non-cultured epidermal suspension (NCES) | Stable vitiligo | 13 | 8–17 | Overall, 79% of lesions had >90% repigmentation | 1 year | Sahni et al. [61] | 2011 |
| Non-cultured cellular grafting | Stable vitiligo | 13 | 15–52 | Overall, 91% of the patients achieved >50% repigmentation | 3–12 months | Gan et al. [59] | 2012 |
| Non-cultured epidermal cell suspension (NCES) | Stable vitiligo | 36 | 16–47 | More than 75% repigmentation in 63.75% of lesions | 6–18 months | Holla et al. [57] | 2013 |
| Non-cultured epidermal cell suspension (NCES) | Stable vitiligo | 37 | Range not stated, mean: 28.3 (group 1) 24.1(group 2) 22.4 (group 3) | Cell count significantly lower in the ORSHFS compared with NCES with no significant difference in the repigmentation outcome | 18 months | El-Zawahry et al. [39] | 2017 |
| Autologous non-cultured epidermal cell suspension (NCES) | Stable vitiligo | 41 | 8–50 | Overall, 80.5% of patients showed 51–75% repigmentation, and 17.1% showed complete or almost complete repigmentation | 6–9 months | Liu et al. [72] | 2019 |
| Non-cultured autologous epidermal cell grafting resuspended in hyaluronic acid (A ready-to-use kit, Viticell®) | Stable vitiligo | 36 | 17–67 | For difficult-to-treat lesions, no repigmentation ≥50% was observed; for other locations, the success rate was significantly higher | 12 months | Bertolotti et al. [76] | 2020 |
| Comparative studies | |||||||
| Three different transplantation methods: autologous cultured melanocytes, ultrathin epidermal sheets, and basal layer cell suspension | Stable or unstable vitiligo | 132 | 8–61 | Stable vitiligo patients responded in most cases with 100% repigmentation in all studied treatments | 1–7 years | Olsson et al. [27] | 2002 |
| Autologous non-cultured epidermal cell suspension (NCES) compared to suction blister epidermal grafting (SBEG) | Stable vitiligo | 41 | 12–40 | Overall, 90–100% repigmentation in 71% of lesions in the NCES group and 27% of lesions in the SBEG group | 4 months | Budania et al. [37] | 2012 |
| Autologous non-cultured epidermal cell suspension (NCES) compared to autologous non-cultured extracted hair follicle outer root sheath cell suspension (NCORSHFS) | Stable vitiligo | 30 | 13–35 | Overall, 90–100% repigmentation in 83% of lesions in the NCES group and 65% of lesions in the NCORSHFS group | 4 months | Singh et al. [38] | 2013 |
| Comparison between autologous melanocyte rich cell suspension (NCMT technique) and cultured melanocyte technique (CMT) | Stable vitiligo | 25 | N/A | More than 70% repigmentation in 62.17% cases treated with NCMT and in 52% with the CMT | 6 months | Verma et al. [13] | 2014 |
| Blister roof grafting (BG), cultured melanocytes transplantation (CMT) and non-cultured epidermal cell suspension transplantation (NCES) | Stable vitiligo | 83 | Range not stated, mean 25 | More than 50% repigmentation in 92%, 82%, and 81% of the 83 patients treated with the BG, CMT, and NCES methods, respectively | 12 months | Bao et al. [36] | 2015 |
| Autologous cultured melanocytes transplantation (CMT) and non-cultured epidermal cell suspension transplantation (NCES) | Stable vitiligo | 30 | Range not stated, mean 26.1 | Overall, 66.7% of cases showed more than 70% repigmentation with CMT; NCES resulted in less than 40% repigmentation in most of the cases | 3–6 months | Verma et al. [40] | 2015 |
| Excimer laser alone compared to non-cultured melanocyte–keratinocyte transplantation (MKCT) alone and combination therapy | Stable vitiligo | 10 | 21–48 | Excimer laser combined with non-cultured MKCT improves the repigmentation rate, with an average of 41.9% reduction of depigmented area surface | 2 weeks | Ebadi et al. [54] | 2015 |
| Epidermal melanocyte transfer (EMT) compared to hair follicular melanocyte transfer (HFMT) | Stable vitiligo | 11 | 12–42 | More than 90% repigmentation observed in 83.33% patches of the EMT group and 43.33% in the HFMT group | 6 months | Donaparthi et al. [77] | 2016 |
| Autologous non-cultured epidermal cell suspension (ECS) and follicular cell suspension (FCS) | Stable vitiligo | 5 | 21–33 | Superior repigmentation obtained in combined ECS and FCS treatment | 4 months | Razmi et al. [41] | 2017 |
| Hair follicle transplantation (follicular unit transplantation, FUT) and autologous non-cultured, non-trypsinized epidermal cells grafting (Jodhpur Technique-JT) | Stable vitiligo | 30 | 21–25 | More than 75% repigmentation was observed in 70% lesions in the FUT group and 72% of lesions in the JT group | 20 weeks | Lamoria et al. [24] | 2020 |
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- van Geel, N.; Ongenae, K.; De Mil, M.; Vander Haeghen, Y.; Vervaet, C.; Naeyaert, J.M. Double-Blind Placebo-Controlled Study of Autologous Transplanted Epidermal Cell Suspensions for Repigmenting Vitiligo. Arch. Dermatol. 2004, 140, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Alkhateeb, A.; Fain, P.R.; Thody, A.; Bennett, D.C.; Spritz, R.A. Epidemiology of Vitiligo and Associated Autoimmune Diseases in Caucasian Probands and Their Families. Pigment. Cell Res. 2003, 16, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Alikhan, A.; Felsten, L.M.; Daly, M.; Petronic-Rosic, V. Vitiligo: A Comprehensive Overview. J. Am. Acad. Dermatol. 2011, 65, 473–491. [Google Scholar] [CrossRef] [PubMed]
- Felsten, L.M.; Alikhan, A.; Petronic-Rosic, V. Vitiligo: A Comprehensive Overview. J. Am. Acad. Dermatol. 2011, 65, 493–514. [Google Scholar] [CrossRef]
- Sarkar, S.; Sarkar, T.; Sarkar, A.; Das, S. Vitiligo and Psychiatric Morbidity: A Profile from a Vitiligo Clinic of a Rural-Based Tertiary Care Center of Eastern India. Indian J. Dermatol. 2018, 63, 281. [Google Scholar] [CrossRef]
- Bonotis, K.; Pantelis, K.; Karaoulanis, S.; Katsimaglis, C.; Papaliaga, M.; Zafiriou, E.; Tsogas, P. Investigation of Factors Associated with Health-Related Quality of Life and Psychological Distress in Vitiligo: Quality of Life and Psychological Distress in Vitiligo. JDDG J. Dtsch. Dermatol. Ges. 2016, 14, 45–48. [Google Scholar] [CrossRef]
- Khaitan, B.; Kathuria, S.; Ramam, M. A Descriptive Study to Characterize Segmental Vitiligo. Indian J. Dermatol. Venereol. Leprol. 2012, 78, 715. [Google Scholar] [CrossRef]
- Toriyama, K.; Kato, H.; Sato, H.; Tanaka, T.; Inoie, M.; Morita, A. Cultured Epidermal Autografts for Treatment of Stable Vitiligo: Quantitative Analysis of Color Matching with Surrounding Normally Pigmented Skin. J. Dermatol. 2021, 48, 1405–1408. [Google Scholar] [CrossRef]
- Picardo, M.; Bastonini, E. A New View of Vitiligo: Looking at Normal-Appearing Skin. J. Investig. Dermatol. 2015, 135, 1713–1714. [Google Scholar] [CrossRef]
- Available online: https://pubmed.ncbi.nlm.nih.gov/ (accessed on 20 November 2022).
- Available online: https://clinicaltrials.gov/ (accessed on 20 November 2022).
- Pandya, V.; Parmar, K.; Shah, B.; Bilimoria, F. A Study of Autologous Melanocyte Transfer in Treatment of Stable Vitiligo. Indian J. Dermatol. Venereol. Leprol. 2005, 71, 393. [Google Scholar] [CrossRef]
- Verma, R.; Grewal, R.S.; Chatterjee, M.; Pragasam, V.; Vasudevan, B.; Mitra, D. A Comparative Study of Efficacy of Cultured versus Non Cultured Melanocyte Transfer in the Management of Stable Vitiligo. Med. J. Armed Forces India 2014, 70, 26–31. [Google Scholar] [CrossRef]
- Ghorbani, I.; Khazaei, M.; Kavoussi, H.; Ebrahimi, A.; Rezaei, M.; Kavoussi, R.; Mansouri, K. Treatment of Recalcitrant Vitiligo by Autologous Non-Cultured and Trypsinized Melanocyte Grafting in the West of Iran. An. Bras. Dermatol. 2022, 97, 315–320. [Google Scholar] [CrossRef]
- Thingnes, J.; Lavelle, T.J.; Hovig, E.; Omholt, S.W. Understanding the Melanocyte Distribution in Human Epidermis: An Agent-Based Computational Model Approach. PLoS ONE 2012, 7, e40377. [Google Scholar] [CrossRef]
- Chen, Y.-F.; Chang, J.S.; Yang, P.-Y.; Hung, C.-M.; Huang, M.-H.; Hu, D.-N. Transplant of Cultured Autologous Pure Melanocytes after Laser-Abrasion for the Treatment of Segmental Vitiligo. J. Dermatol. 2000, 27, 434–439. [Google Scholar] [CrossRef]
- Hong, W.; Hu, D.; Qian, G.; McCormick, S.; Xu, A. Treatment of Vitiligo in Children and Adolescents by Autologous Cultured Pure Melanocytes Transplantation with Comparison of Efficacy to Results in Adults: Melanocyte Transplantation in Children with Vitiligo. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 538–543. [Google Scholar] [CrossRef]
- Redondo, P.; Giménez de Azcarate, A.; Marqués, L.; García-Guzman, M.; Andreu, E.; Prósper, F. Amniotic Membrane as a Scaffold for Melanocyte Transplantation in Patients with Stable Vitiligo. Dermatol. Res. Pract. 2011, 2011, 1–6. [Google Scholar] [CrossRef]
- Mohanty, S.; Kumar, A.; Dhawan, J.; Sreenivas, V.; Gupta, S. Noncultured Extracted Hair Follicle Outer Root Sheath Cell Suspension for Transplantation in Vitiligo: ORS Cell Transplantation in Vitiligo. Br. J. Dermatol. 2011, 164, 1241–1246. [Google Scholar] [CrossRef]
- Vinay, K.; Dogra, S.; Parsad, D.; Kanwar, A.J.; Kumar, R.; Minz, R.W.; Saikia, U.N. Clinical and Treatment Characteristics Determining Therapeutic Outcome in Patients Undergoing Autologous Non-Cultured Outer Root Sheath Hair Follicle Cell Suspension for Treatment of Stable Vitiligo. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 31–37. [Google Scholar] [CrossRef]
- Shah, A.; Marfatia, R.; Saikia, S. A Study of Noncultured Extracted Hair Follicle Outer Root Sheath Cell Suspension for Transplantation in Vitiligo. Int. J. Trichol. 2016, 8, 67. [Google Scholar] [CrossRef]
- Kumar, P.; Bhari, N.; Tembhre, M.K.; Mohanty, S.; Arava, S.; Sharma, V.K.; Gupta, S. Study of Efficacy and Safety of Noncultured, Extracted Follicular Outer Root Sheath Cell Suspension Transplantation in the Management of Stable Vitiligo. Int. J. Dermatol. 2018, 57, 245–249. [Google Scholar] [CrossRef]
- Shi, H.-X.; Zhang, R.-Z.; Xu, B.; Xu, C.-X.; Li, D.; Wang, L.; Xiao, L. Experimental Study and Clinical Observations of Autologous Hair Follicle Cell Transplants to Treat Stable Vitiligo. Indian J. Dermatol. Venereol. Leprol. 2020, 86, 124. [Google Scholar] [CrossRef] [PubMed]
- Lamoria, A.; Agrawal, A.; Rao, P.; Kachhawa, D. A Comparative Study between Follicular Unit Transplantation and Autologous Non-Cultured Non-Trypsinized Epidermal Cells Grafting (Jodhpur Technique) in Stable Vitiligo. J. Cutan. Aesthet. Surg. 2020, 13, 204. [Google Scholar] [CrossRef] [PubMed]
- Benzekri, L.; Gauthier, Y. The First Transepidermal Transplantation of Non-Cultured Epidermal Suspension Using a Dermarolling System in Vitiligo: A Sequential Histological and Clinical Study. Pigment Cell Melanoma Res. 2017, 30, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.-C.; Ma, H.-Y.; Zhan, Z.; Liu, C.-G.; Luo, W.; Zhao, G. Detection of Auto Antibodies and Transplantation of Cultured Autologous Melanocytes for the Treatment of Vitiligo. Exp. Ther. Med. 2017, 13, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Olsson, M.J.; Juhlin, L. Long-Term Follow-up of Leucoderma Patients Treated with Transplants of Autologous Cultured Melanocytes, Ultrathin Epidermal Sheets and Basal Cell Layer Suspension. Br. J. Dermatol. 2002, 147, 893–904. [Google Scholar] [CrossRef]
- Chen, Y.-F.; Yang, P.-Y.; Hu, D.-N.; Kuo, F.-S.; Hung, C.-S.; Hung, C.-M. Treatment of Vitiligo by Transplantation of Cultured Pure Melanocyte Suspension: Analysis of 120 Cases. J. Am. Acad. Dermatol. 2004, 51, 68–74. [Google Scholar] [CrossRef]
- Lee, A.-Y. Role of Keratinocytes in the Development of Vitiligo. Ann. Dermatol. 2012, 24, 115. [Google Scholar] [CrossRef]
- Matsuzaki, K.; Kumagai, N. Treatment of Vitiligo with Autologous Cultured Keratinocytes in 27 Cases. Eur. J. Plast. Surg. 2013, 36, 651–656. [Google Scholar] [CrossRef]
- Gauthier, Y.; Surleve-Bazeille, J.-E. Autologous Grafting with Non-cultured Melanocytes: A Simplified Method for Treatment of Depigmented Lesions. J. Am. Acad. Dermatol. 1992, 26, 191–194. [Google Scholar] [CrossRef]
- Billingham, R.E.; Medawar, P.B. Pigment Spread and Cell Heredity in Guinea-Pigs’ Skin. Heredity 1948, 2, 29–47. [Google Scholar] [CrossRef]
- Phillips, J.; Gawkrodger, D.J.; Caddy, C.M.; Hedley, S.; Dawson, R.A.; Smith-Thomas, L.; Freedlander, E.; Mac Neil, S. Keratinocytes Suppress TRP-1 Expression and Reduce Cell Number of Co-Cultured Melanocytes—Implications For Grafting of Patients with Vitiligo: Keratinocytes Down-Regulate Melanocyte Number and Pigmentation. Pigment. Cell Res. 2001, 14, 116–125. [Google Scholar] [CrossRef]
- Khodadadi, L.; Shafieyan, S.; Sotoudeh, M.; Dizaj, A.V.; Shahverdi, A.; Aghdami, N.; Baharvand, H. Intraepidermal Injection of Dissociated Epidermal Cell Suspension Improves Vitiligo. Arch. Dermatol. Res. 2010, 302, 593–599. [Google Scholar] [CrossRef]
- Orouji, Z.; Bajouri, A.; Ghasemi, M.; Mohammadi, P.; Fallah, N.; Shahbazi, A.; Rezvani, M.; Vaezirad, F.; Khalajasadi, Z.; Alizadeh, A.; et al. A Single-Arm Open-Label Clinical Trial of Autologous Epidermal Cell Transplantation for Stable Vitiligo: A 30-Month Follow-Up. J. Dermatol. Sci. 2018, 89, 52–59. [Google Scholar] [CrossRef]
- Bao, H.; Hong, W.; Fu, L.; Wei, X.; Qian, G.; Xu, A. Blister Roof Grafting, Cultured Melanocytes Transplantation and Non-Cultured Epidermal Cell Suspension Transplantation in Treating Stable Vitiligo: A Mutual Self-Control Study. J. Dermatol. Treat. 2015, 26, 571–574. [Google Scholar] [CrossRef]
- Budania, A.; Parsad, D.; Kanwar, A.J.; Dogra, S. Comparison between Autologous Noncultured Epidermal Cell Suspension and Suction Blister Epidermal Grafting in Stable Vitiligo: A Randomized Study. Br. J. Dermatol. 2012, 167, 1295–1301. [Google Scholar] [CrossRef]
- Singh, C.; Parsad, D.; Kanwar, A.J.; Dogra, S.; Kumar, R. Comparison between Autologous Noncultured Extracted Hair Follicle Outer Root Sheath Cell Suspension and Autologous Noncultured Epidermal Cell Suspension in the Treatment of Stable Vitiligo: A Randomized Study. Br. J. Dermatol. 2013, 169, 287–293. [Google Scholar] [CrossRef]
- El-Zawahry, B.M.; Esmat, S.; Bassiouny, D.; Zaki, N.S.; Sobhi, R.; Saleh, M.A.; Abdel-Halim, D.; Hegazy, R.; Gawdat, H.; Samir, N.; et al. Effect of Procedural-Related Variables on Melanocyte–Keratinocyte Suspension Transplantation in Nonsegmental Stable Vitiligo: A Clinical and Immunocytochemical Study. Dermatol. Surg. 2017, 43, 226–235. [Google Scholar] [CrossRef]
- Verma, G.; Varkhande, S.R.; Kar, H.K.; Rani, R. Evaluation of Repigmentation with Cultured Melanocyte Transplantation (CMT) Compared with Non-Cultured Epidermal Cell Transplantation in Vitiligo at 12th Week Reveals Better Repigmentation with CMT. J. Investig. Dermatol. 2015, 135, 2533–2535. [Google Scholar] [CrossRef]
- Razmi, T.M.; Parsad, D.; Kumaran, S.M. Combined Epidermal and Follicular Cell Suspension as a Novel Surgical Approach for Acral Vitiligo. J. Am. Acad. Dermatol. 2017, 76, 564–567. [Google Scholar] [CrossRef]
- Olsson, M.J.; Juhlin, L. Juhlin Leucoderma Treated by Transplantation of a Basal Cell Layer Enriched Suspension: BASAL CELL TRANSPLANT FOR LEUCODERMA. Br. J. Dermatol. 1998, 138, 644–648. [Google Scholar] [CrossRef]
- Mulekar, S.V. Melanocyte-Keratinocyte Cell Transplantation for Stable Vitiligo. Int. J. Dermatol. 2003, 42, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Silpa-Archa, N.; Griffith, J.L.; Huggins, R.H.; Henderson, M.D.; Kerr, H.A.; Jacobsen, G.; Mulekar, S.V.; Lim, H.W.; Hamzavi, I.H. Long-Term Follow-up of Patients Undergoing Autologous Noncultured Melanocyte-Keratinocyte Transplantation for Vitiligo and Other Leukodermas. J. Am. Acad. Dermatol. 2017, 77, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Nahhas, A.F.; Mohammad, T.F.; Hamzavi, I.H. Vitiligo surgery: Shuffling melanocytes. J. Investig. Dermatol. Symp. Proc. 2017, 18, S34–S37. [Google Scholar] [CrossRef] [PubMed]
- Altalhab, S.; AlJasser, M.I.; Mulekar, S.V.; Al Issa, A.; Mulekar, S.; Diaz, J.; Diallo, A.; Ezzedine, K. Six-year Follow-up of Vitiligo Patients Successfully Treated with Autologous Non-cultured Melanocyte–Keratinocyte Transplantation. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1172–1176. [Google Scholar] [CrossRef] [PubMed]
- Mulekar, S.V. Long-Term Follow-up Study of Segmental and Focal Vitiligo Treated by Autologous, Noncultured Melanocyte-Keratinocyte Cell Transplantation. Arch. Dermatol. 2004, 140, 1211–1215. [Google Scholar] [CrossRef]
- Mulekar, S.V. Long-Term Follow-up Study of 142 Patients with Vitiligo Vulgaris Treated by Autologous, Non-Cultured Melanocyte-Keratinocyte Cell Transplantation: Vitiligo Vulgaris Patients Treated by Epidermal Cell Transplantation. Int. J. Dermatol. 2005, 44, 841–845. [Google Scholar] [CrossRef]
- Mulekar, S.V.; Issa, A.; Eisa, A.; Asaad, M. Genital Vitiligo Treated by Autologous, Noncultured Melanocyte-Keratinocyte Cell Transplantation. Dermatol. Surg. 2005, 31, 1737–1740. [Google Scholar] [CrossRef]
- Toossi, P.; Shahidi-Dadras, M.; Mahmoudi Rad, M.; Fesharaki, R.J. Non-Cultured Melanocyte-Keratinocyte Transplantation for the Treatment of Vitiligo: A Clinical Trial in an Iranian Population: Cell Grafting for Vitiligo. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 1182–1186. [Google Scholar] [CrossRef]
- Vazquez-Martínez, O.T.; Martínez-Rodríguez, H.G.; Velásquez-Arenas, L.; Baños-González, D.; Ortíz-López, R.; Padilla-Rivas, G.; Welsh, O.; Ocampo-Candiani, J. Treatment of Vitiligo with a Melanocyte-Keratinocyte Cell Suspension versus Dermabrasion Only: A Pilot Study with a 12-Month Follow Up. J. Drugs Dermatol. 2011, 10, 1032–1036. [Google Scholar]
- Quezada, N.; Filho, C. App.M.; De La Sotta, P.; Uribe, P. Melanocytes and Keratinocytes Transfer Using Sandpaper Technique Combined with Dermabrasion for Stable Vitiligo. Dermatol. Surg. 2011, 37, 192–198. [Google Scholar] [CrossRef]
- Huggins, R.H.; Henderson, M.D.; Mulekar, S.V.; Ozog, D.M.; Kerr, H.A.; Jabobsen, G.; Lim, H.W.; Hamzavi, I.H. Melanocyte-Keratinocyte Transplantation Procedure in the Treatment of Vitiligo: The Experience of an Academic Medical Center in the United States. J. Am. Acad. Dermatol. 2012, 66, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Ebadi, A.; Rad, M.M.; Nazari, S.; Fesharaki, R.J.; Ghalamkarpour, F.; Younespour, S. The Additive Effect of Excimer Laser on Non-Cultured Melanocyte-Keratinocyte Transplantation for the Treatment of Vitiligo: A Clinical Trial in an Iranian Population. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Mulekar, S.V.; Al Issa, A.; Al Eisa, A. Treatment of Vitiligo on Difficult-to-Treat Sites Using Autologous Noncultured Cellular Grafting. Dermatol. Surg. 2009, 35, 66–71. [Google Scholar] [CrossRef] [PubMed]
- El-Zawahry, B.; Zaki, N.; Bassiouny, D.; Sobhi, R.; Zaghloul, A.; Khorshied, M.; Gouda, H. Autologous Melanocyte-Keratinocyte Suspension in the Treatment of Vitiligo: MC-KC Suspension in Treatment of Vitiligo. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Holla, A.P.; Sahni, K.; Kumar, R.; Parsad, D.; Kanwar, A.; Mehta, S.D. Acral Vitiligo and Lesions over Joints Treated with Non-Cultured Epidermal Cell Suspension Transplantation. Clin. Exp. Dermatol. 2013, 38, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Ramos, M.G.; Ramos, D.G.; Ramos, C.G. Evaluation of Treatment Response to Autologous Transplantation of Noncultured Melanocyte/Keratinocyte Cell Suspension in Patients with Stable Vitiligo. An. Bras. Dermatol. 2017, 92, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Gan, E.Y.; van Geel, N.; Goh, B.K. Repigmentation of Leucotrichia in Vitiligo with Noncultured Cellular Grafting: Leucotrichia Repigmentation with Noncultured Cellular Grafting. Br. J. Dermatol. 2012, 166, 196–199. [Google Scholar] [CrossRef]
- Mulekar, S.V.; Al Eisa, A.; Delvi, M.B.; Al Issa, A.; Al Saeed, A.H. Childhood Vitiligo: A Long-Term Study of Localized Vitiligo Treated by Non-cultured Cellular Grafting. Pediatr. Dermatol. 2010, 27, 132–136. [Google Scholar] [CrossRef]
- Sahni, K.; Parsad, D.; Kanwar, A.J. Noncultured Epidermal Suspension Transplantation for the Treatment of Stable Vitiligo in Children and Adolescents: Noncultured Melanocyte Transplantation in Children and Adolescents. Clin. Exp. Dermatol. 2011, 36, 607–612. [Google Scholar] [CrossRef]
- Cervelli, V.; De Angelis, B.; Balzani, A.; Colicchia, G.; Spallone, D.; Grimaldi, M. Treatment of Stable Vitiligo by ReCell System. Acta Dermatovenerol. Croat. 2009, 17, 273–278. [Google Scholar]
- Mulekar, S.V.; Ghwish, B.; Al Issa, A.; Al Eisa, A. Treatment of Vitiligo Lesions by ReCell ® vs. Conventional Melanocyte–Keratinocyte Transplantation: A Pilot Study. Br. J. Dermatol. 2007, 19, 507–527. [Google Scholar] [CrossRef]
- Cervelli, V.; Spallone, D.; Lucarini, L.; Palla, L.; Brinci, L.; De Angelis, B. Treatment of Stable Vitiligo Hands by ReCell System: A Preliminary Report. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 691–694. [Google Scholar]
- Tyagi, S.; Malhotra, S.K.; Kaur, T. Comparative Evaluation of Efficacy of Non-Cultured Epidermal Cell Suspension and Epidermal Curettage in Stable Vitiligo. J. Cutan. Aesthet. Surg. 2021, 14, 32–40. [Google Scholar] [CrossRef]
- Samson Yashar, S.; Gielczyk, R.; Scherschun, L.; Lim, H.W. Narrow-Band Ultraviolet B Treatment for Vitiligo, Pruritus, and Inflammatory Dermatoses: Narrow-Band UVB Treatment. Photodermatol. Photoimmunol. Photomed. 2003, 19, 164–168. [Google Scholar] [CrossRef]
- Zhang, D.; Hong, W.; Fu, L.; Wei, X.; Xu, A. A Randomized Controlled Study of the Effects of Different Modalities of Narrow-Band Ultraviolet B Therapy on the Outcome of Cultured Autologous Melanocytes Transplantation in Treating Vitiligo. Dermatol. Surg. 2014, 40, 420–426. [Google Scholar] [CrossRef]
- Yao, L.; Liu, Y.; Song, Y.; Zhong, S.; Li, S. Successful Treatment of Stable Vitiligo by Low-Density Cultured Autologous Melanocyte Transplantation Combined With Narrowband Ultraviolet B Therapy. Dermatol. Surg. 2017, 43, 1281–1287. [Google Scholar] [CrossRef]
- van Geel, N.; Ongenae, K.; De Mil, M.; Naeyaert, J.M. Modified Technique of Autologous Noncultured Epidermal Cell Transplantation for Repigmenting Vitiligo: A Pilot Study. Dermatol. Surg. 2001, 27, 873–876. [Google Scholar] [CrossRef]
- Ibrahim, Z.A.; El-Ashmawy, A.A.; El-Tatawy, R.A.; Sallam, F.A. The Effect of Platelet-Rich Plasma on the Outcome of Short-Term Narrowband-Ultraviolet B Phototherapy in the Treatment of Vitiligo: A Pilot Study. J. Cosmet. Dermatol. 2016, 15, 108–116. [Google Scholar] [CrossRef]
- Parambath, N.; Sharma, V.K.; Parihar, A.S.; Sahni, K.; Gupta, S. Use of Platelet-Rich Plasma to Suspend Non-cultured Epidermal Cell Suspension Improves Repigmentation after Autologous Transplantation in Stable Vitiligo: A Double-Blind Randomized Controlled Trial. Int. J. Dermatol. 2019, 58, 472–476. [Google Scholar] [CrossRef]
- Liu, B.; Chen, H.-H.; Liu, Z.-H.; Liang, J.-F.; Xue, R.-J.; Chen, P.-J.; Li, C.-X.; Liang, X.-D.; Deng, J.; Ye, R.-X.; et al. The Clinical Efficacy of Treatment Using the Autologous Non-Cultured Epidermal Cell Suspension Technique for Stable Vitiligo in 41 Patients. J. Dermatol. Treat. 2021, 32, 90–94. [Google Scholar] [CrossRef]
- Available online: https://www.aafp.org/pubs/afp/issues/2017/1215/p797.html (accessed on 20 November 2022).
- Abdolahzadeh, H.; Mohammadi, P.; Ghasemi, M.; Mousavi, S.A.; Bajouri, A.; Ataei-Fashtami, L.; Totonchi, M.; Rezvani, M.; Aghdami, N.; Shafieyan, S. Comparison of Skin Transcriptome between Responder and Non-Responder Vitiligo Lesions to Cell Transplantation: A Clinical Trial Study. Cell J. 2022, 24, 316. [Google Scholar] [CrossRef] [PubMed]
- Ju, H.J.; Bae, J.M.; Lee, R.W.; Kim, S.H.; Parsad, D.; Pourang, A.; Hamzavi, I.; Shourick, J.; Ezzedine, K. Surgical Interventions for Patients With Vitiligo: A Systematic Review and Meta-analysis. JAMA Dermatol. 2021, 157, 307–331. [Google Scholar] [CrossRef] [PubMed]
- Bertolotti, A.; Leone, G.; Taïeb, A.; Soriano, E.; Pascal, M.; Maillard, H.; Geel, N. Assessment of Non-Cultured Autologous Epidermal Cell Grafting Resuspended in Hyaluronic Acid for Repigmenting Vitiligo and Piebaldism Lesions: A Randomized Clinical Trial. Acta Derm. Venereol. 2021, 2021, 101. [Google Scholar] [CrossRef] [PubMed]
- Donaparthi, N.; Chopra, A. Comparative Study of Efficacy of Epidermal Melanocyte Transfer Versus Hair Follicular Melanocyte Transfer in Stable Vitiligo. Indian J. Dermatol. 2016, 61, 640–644. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domaszewska-Szostek, A.; Polak, A.; Słupecka-Ziemilska, M.; Krzyżanowska, M.; Puzianowska-Kuźnicka, M. Current Status of Cell-Based Therapies for Vitiligo. Int. J. Mol. Sci. 2023, 24, 3357. https://doi.org/10.3390/ijms24043357
Domaszewska-Szostek A, Polak A, Słupecka-Ziemilska M, Krzyżanowska M, Puzianowska-Kuźnicka M. Current Status of Cell-Based Therapies for Vitiligo. International Journal of Molecular Sciences. 2023; 24(4):3357. https://doi.org/10.3390/ijms24043357
Chicago/Turabian StyleDomaszewska-Szostek, Anna, Agnieszka Polak, Monika Słupecka-Ziemilska, Marta Krzyżanowska, and Monika Puzianowska-Kuźnicka. 2023. "Current Status of Cell-Based Therapies for Vitiligo" International Journal of Molecular Sciences 24, no. 4: 3357. https://doi.org/10.3390/ijms24043357
APA StyleDomaszewska-Szostek, A., Polak, A., Słupecka-Ziemilska, M., Krzyżanowska, M., & Puzianowska-Kuźnicka, M. (2023). Current Status of Cell-Based Therapies for Vitiligo. International Journal of Molecular Sciences, 24(4), 3357. https://doi.org/10.3390/ijms24043357






