Is Hormone Replacement Therapy a Risk Factor or a Therapeutic Option for Alzheimer’s Disease?
Abstract
1. Introduction
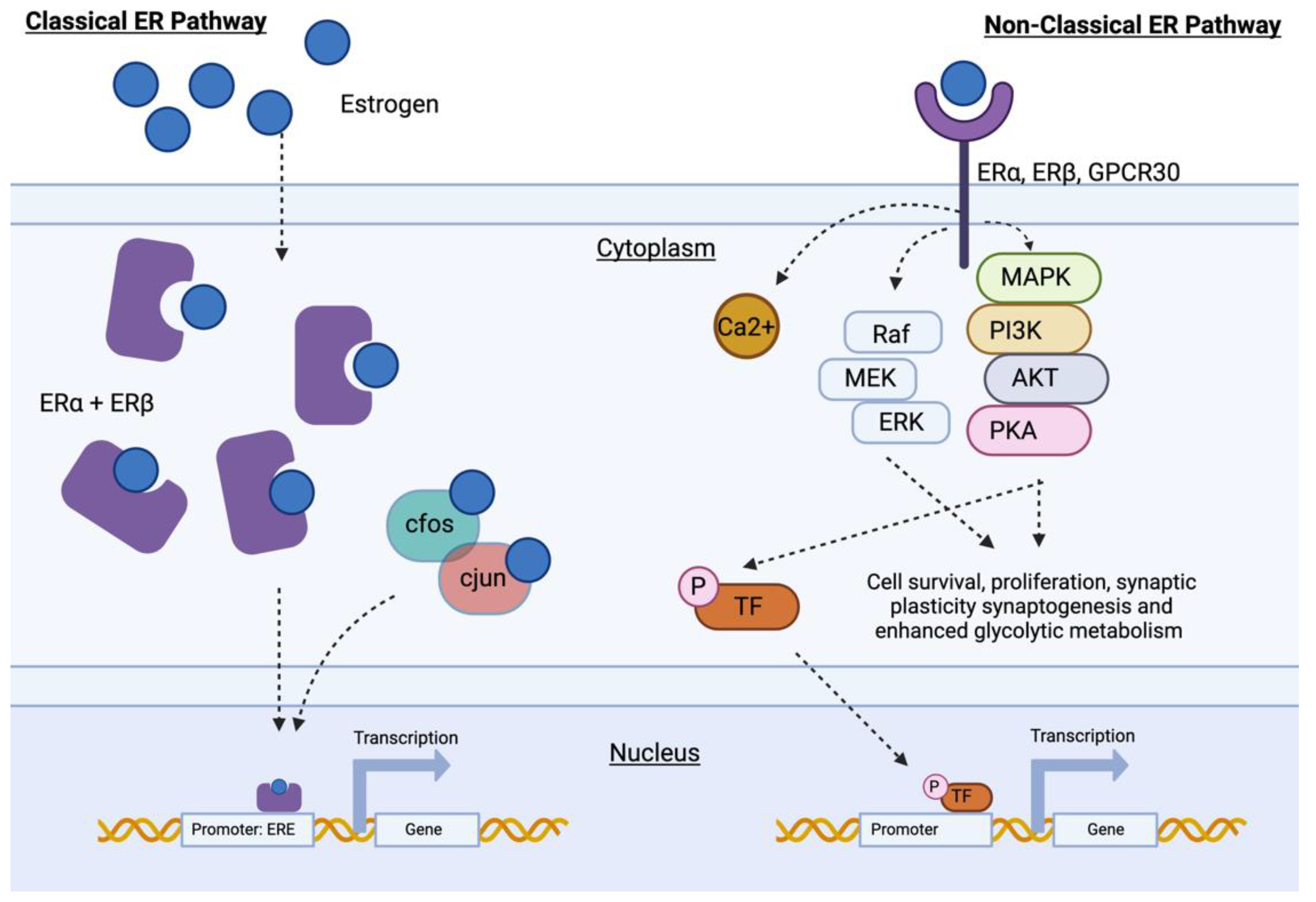
2. Estrogens and Neuroprotection
3. Critical Window Hypothesis
4. Healthy Cell Bias
5. Conflicting Evidence
6. Additional Factors
6.1. Reproductive History
6.2. Route of Administration
6.3. Formulation
6.4. Regime and Dosage
6.5. Progestrone (Combined Therapy)
7. Selective Estrogen Receptor Modulators and Activators of Non-Genomic Estrogen-like Signaling
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Malaiya, A.; Singhai, M.; Singh, M.; Prajapati, S.K.; Choudhury, H.; Fatima, M.; Alexander, A.; Dubey, S.K.; Greish, K.; Kesharwani, P. Recent Update on the Alzheimer’s Disease Progression, Diagnosis and Treatment Approaches. Curr. Drug Targets 2022, 23, 978–1001. [Google Scholar] [PubMed]
- Association, A.S. Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2019, 15, 321–387. [Google Scholar] [CrossRef]
- Li, R.; Singh, M. Sex differences in cognitive impairment and Alzheimer’s disease. Front. Neuroendocr. 2014, 35, 385–403. [Google Scholar] [CrossRef] [PubMed]
- Zagni, E.; Simoni, L.; Colombo, D. Sex and Gender Differences in Central Nervous System-Related Disorders. Neurosci. J. 2016, 2016, 2827090. [Google Scholar] [CrossRef] [PubMed]
- Zucker, I.; Beery, A.K. Males still dominate animal studies. Nature 2010, 465, 690. [Google Scholar] [CrossRef]
- Mogil, J.S.; Chanda, M.L. The case for the inclusion of female subjects in basic science studies of pain. Pain 2005, 117, 1–5. [Google Scholar] [CrossRef]
- Ferretti, M.; Martinkova, J.; Biskup, E.; Benke, T.; Gialdini, G.; Nedelska, Z.; Rauen, K.; Mantua, V.; Religa, D.; Hort, J. Sex and gender differences in Alzheimer’s disease: Current challenges and implications for clinical practice: Position paper of the Dementia and Cognitive Disorders Panel of the European Academy of Neurology. Eur. J. Neurol. 2020, 27, 928–943. [Google Scholar] [CrossRef]
- Pike, C.J. Sex and the development of Alzheimer’s disease. J. Neurosci. Res. 2017, 95, 671–680. [Google Scholar] [CrossRef]
- Rahman, A.; Jackson, H.; Hristov, H.; Isaacson, R.S.; Saif, N.; Shetty, T.; Etingin, O.; Henchcliffe, C.; Brinton, R.D.; Mosconi, L. Sex and Gender Driven Modifiers of Alzheimer’s: The Role for Estrogenic Control Across Age, Race, Medical, and Lifestyle Risks. Front. Aging Neurosci. 2019, 11, 315. [Google Scholar] [CrossRef]
- Guo, L.; Zhong, M.B.; Zhang, L.; Zhang, B.; Cai, D. Sex Differences in Alzheimer’s Disease: Insights from the Multiomics Landscape. Biol. Psychiatry 2022, 91, 61–71. [Google Scholar] [CrossRef]
- Johnson, S.R. Menopause and hormone replacement therapy. Med. Clin. N. Am. 1998, 82, 297–320. [Google Scholar] [CrossRef] [PubMed]
- Henderson, V.W. Alzheimer’s disease: Review of hormone therapy trials and implications for treatment and prevention after menopause. J. Steroid Biochem. Mol. Biol. 2014, 142, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Mortel, K.F.; Meyer, J.S. Lack of postmenopausal estrogen replacement therapy and the risk of dementia. J. Neuropsychiatry Clin. Neurosci. 1995, 7, 334–337. [Google Scholar] [PubMed]
- Tang, M.X.; Jacobs, D.; Stern, Y.; Marder, K.; Schofield, P.; Gurland, B.; Andrews, H.; Mayeux, R. Effect of oestrogen during menopause on risk and age at onset of Alzheimer’s disease. Lancet 1996, 348, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Kawas, C.; Resnick, S.; Morrison, A.; Brookmeyer, R.; Corrada, M.; Zonderman, A.; Bacal, C.; Lingle, D.D.; Metter, E. A prospective study of estrogen replacement therapy and the risk of developing Alzheimer’s disease: The Baltimore Longitudinal Study of Aging. Neurology 1997, 48, 1517–1521. [Google Scholar] [CrossRef] [PubMed]
- Baldereschi, M.; Di Carlo, A.; Lepore, V.; Bracco, L.; Maggi, S.; Grigoletto, F.; Scarlato, G.; Amaducci, L. Estrogen-replacement therapy and Alzheimer’s disease in the Italian Longitudinal Study on Aging. Neurology 1998, 50, 996–1002. [Google Scholar] [CrossRef]
- Slooter, A.J.; Bronzova, J.; Witteman, J.C.; Van Broeckhoven, C.; Hofman, A.; van Duijn, C.M. Estrogen use and early onset Alzheimer’s disease: A population-based study. J. Neurol. Neurosurg. Psychiatry 1999, 67, 779–781. [Google Scholar] [CrossRef]
- Waring, S.C.; Rocca, W.A.; Petersen, R.C.; O’Brien, P.C.; Tangalos, E.G.; Kokmen, E. Postmenopausal estrogen replacement therapy and risk of AD: A population-based study. Neurology 1999, 52, 965–970. [Google Scholar] [CrossRef]
- Lindsay, J.; Laurin, D.; Verreault, R.; Hebert, R.; Helliwell, B.; Hill, G.B.; McDowell, I. Risk factors for Alzheimer’s disease: A prospective analysis from the Canadian Study of Health and Aging. Am. J. Epidemiol. 2002, 156, 445–453. [Google Scholar] [CrossRef]
- Zandi, P.P.; Carlson, M.C.; Plassman, B.L.; Welsh-Bohmer, K.A.; Mayer, L.S.; Steffens, D.C.; Breitner, J.C.; Cache County Memory Study, I. Hormone replacement therapy and incidence of Alzheimer disease in older women: The Cache County Study. JAMA 2002, 288, 2123–2129. [Google Scholar] [CrossRef]
- Yaffe, K.; Sawaya, G.; Lieberburg, I.; Grady, D. Estrogen therapy in postmenopausal women: Effects on cognitive function and dementia. JAMA 1998, 279, 688–695. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, E.S.; Janowsky, J.; Chan, B.K.; Nelson, H.D. Hormone replacement therapy and cognition: Systematic review and meta-analysis. JAMA 2001, 285, 1489–1499. [Google Scholar] [CrossRef] [PubMed]
- Espeland, M.A.; Rapp, S.R.; Shumaker, S.A.; Brunner, R.; Manson, J.E.; Sherwin, B.B.; Hsia, J.; Margolis, K.L.; Hogan, P.E.; Wallace, R.; et al. Women’s Health Initiative Memory, S. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. JAMA 2004, 291, 2959–2968. [Google Scholar] [CrossRef]
- Shumaker, S.A.; Legault, C.; Rapp, S.R.; Thal, L.; Wallace, R.B.; Ockene, J.K.; Hendrix, S.L.; Jones, B.N., 3rd; Assaf, A.R.; Jackson, R.D.; et al. Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: The Women’s Health Initiative Memory Study: A randomized controlled trial. JAMA 2003, 289, 2651–2662. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.E.; Shin, D.W.; Han, K.; Kim, D.; Won, H.S.; Lee, J.; Kim, S.Y.; Nam, G.E.; Park, H.S. Female reproductive factors and the risk of dementia: A nationwide cohort study. Eur. J. Neurol. 2020, 27, 1448–1458. [Google Scholar] [CrossRef]
- Shao, H.; Breitner, J.C.; Whitmer, R.A.; Wang, J.; Hayden, K.; Wengreen, H.; Corcoran, C.; Tschanz, J.; Norton, M.; Munger, R.; et al. Hormone therapy and Alzheimer disease dementia: New findings from the Cache County Study. Neurology 2012, 79, 1846–1852. [Google Scholar] [CrossRef]
- Imtiaz, B.; Tuppurainen, M.; Rikkonen, T.; Kivipelto, M.; Soininen, H.; Kroger, H.; Tolppanen, A.M. Postmenopausal hormone therapy and Alzheimer disease: A prospective cohort study. Neurology 2017, 88, 1062–1068. [Google Scholar] [CrossRef]
- Khoo, S.K.; O’Neill, S.; Byrne, G.; King, R.; Travers, C.; Tripcony, L. Postmenopausal hormone therapy and cognition: Effects of timing and treatment type. Climacteric 2010, 13, 259–264. [Google Scholar] [CrossRef]
- Henderson, V.W.; Paganini-Hill, A.; Miller, B.L.; Elble, R.J.; Reyes, P.F.; Shoupe, D.; McCleary, C.A.; Klein, R.A.; Hake, A.M.; Farlow, M.R. Estrogen for Alzheimer’s disease in women: Randomized, double-blind, placebo-controlled trial. Neurology 2000, 54, 295–301. [Google Scholar] [CrossRef]
- Greendale, G.A.; Huang, M.H.; Wight, R.G.; Seeman, T.; Luetters, C.; Avis, N.E.; Johnston, J.; Karlamangla, A.S. Effects of the menopause transition and hormone use on cognitive performance in midlife women. Neurology 2009, 72, 1850–1857. [Google Scholar] [CrossRef]
- MacLennan, A.H.; Henderson, V.W.; Paine, B.J.; Mathias, J.; Ramsay, E.N.; Ryan, P.; Stocks, N.P.; Taylor, A.W. Hormone therapy, timing of initiation, and cognition in women aged older than 60 years: The REMEMBER pilot study. Menopause 2006, 13, 28–36. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, R.; Schroder, C.M.; Bloss, C.; Bailey, A.M.; Alyeshmerni, A.M.; Mumenthaler, M.S.; Friedman, L.F.; Yesavage, J.A. Hormone replacement therapy and longitudinal cognitive performance in postmenopausal women. Am. J. Geriatr. Psychiatry 2005, 13, 1107–1110. [Google Scholar] [CrossRef]
- Ryan, J.; Carriere, I.; Scali, J.; Dartigues, J.F.; Tzourio, C.; Poncet, M.; Ritchie, K.; Ancelin, M.L. Characteristics of hormone therapy, cognitive function, and dementia: The prospective 3C Study. Neurology 2009, 73, 1729–1737. [Google Scholar] [CrossRef] [PubMed]
- Rippon, G.A.; Tang, M.X.; Lee, J.H.; Lantigua, R.; Medrano, M.; Mayeux, R. Familial Alzheimer disease in Latinos: Interaction between APOE, stroke, and estrogen replacement. Neurology 2006, 66, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Savolainen-Peltonen, H.; Rahkola-Soisalo, P.; Hoti, F.; Vattulainen, P.; Gissler, M.; Ylikorkala, O.; Mikkola, T.S. Use of postmenopausal hormone therapy and risk of Alzheimer’s disease in Finland: Nationwide case-control study. BMJ 2019, 364, l665. [Google Scholar] [CrossRef] [PubMed]
- Matyi, J.M.; Rattinger, G.B.; Schwartz, S.; Buhusi, M.; Tschanz, J.T. Lifetime estrogen exposure and cognition in late life: The Cache County Study. Menopause 2019, 26, 1366. [Google Scholar] [CrossRef] [PubMed]
- Løkkegaard, L.E.; Thinggaard, M.; Nygaard, M.; Hallas, J.; Osler, M.; Christensen, K. Systemic hormone therapy and dementia: A nested case-control and co-twin control study. Maturitas 2022, 165, 113–119. [Google Scholar] [CrossRef]
- Fox, M.; Berzuini, C.; Knapp, L.A. Cumulative estrogen exposure, number of menstrual cycles, and Alzheimer’s risk in a cohort of British women. Psychoneuroendocrinology 2013, 38, 2973–2982. [Google Scholar] [CrossRef]
- Fox, M.; Berzuini, C.; Knapp, L.A. Maternal breastfeeding history and Alzheimer’s disease risk. J. Alzheimer’s Dis. 2013, 37, 809–821. [Google Scholar] [CrossRef]
- Colucci, M.; Cammarata, S.; Assini, A.; Croce, R.; Clerici, F.; Novello, C.; Mazzella, L.; Dagnino, N.; Mariani, C.; Tanganelli, P. The number of pregnancies is a risk factor for Alzheimer’s disease. Eur. J. Neurol. 2006, 13, 1374–1377. [Google Scholar] [CrossRef]
- Hesson, J. Cumulative estrogen exposure and prospective memory in older women. Brain Cogn. 2012, 80, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Resnick, S.M.; Coker, L.H.; Maki, P.M.; Rapp, S.R.; Espeland, M.A.; Shumaker, S.A. The Women’s Health Initiative Study of Cognitive Aging (WHISCA): A randomized clinical trial of the effects of hormone therapy on age-associated cognitive decline. Clin. Trials 2004, 1, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Schiff, R.; Bulpitt, C.J.; Wesnes, K.A.; Rajkumar, C. Short-term transdermal estradiol therapy, cognition and depressive symptoms in healthy older women. A randomised placebo controlled pilot cross-over study. Psychoneuroendocrinology 2005, 30, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Almeida, O.P.; Lautenschlager, N.T.; Vasikaran, S.; Leedman, P.; Gelavis, A.; Flicker, L. A 20-week randomized controlled trial of estradiol replacement therapy for women aged 70 years and older: Effect on mood, cognition and quality of life. Neurobiol. Aging 2006, 27, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Yaffe, K.; Vittinghoff, E.; Ensrud, K.E.; Johnson, K.C.; Diem, S.; Hanes, V.; Grady, D. Effects of ultra-low-dose transdermal estradiol on cognition and health-related quality of life. Arch. Neurol. 2006, 63, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Shaywitz, S.E.; Naftolin, F.; Zelterman, D.; Marchione, K.E.; Holahan, J.M.; Palter, S.F.; Shaywitz, B.A. Better oral reading and short-term memory in midlife, postmenopausal women taking estrogen. Menopause 2003, 10, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Espeland, M.A.; Shumaker, S.A.; Leng, I.; Manson, J.E.; Brown, C.M.; LeBlanc, E.S.; Vaughan, L.; Robinson, J.; Rapp, S.R.; Goveas, J.S.; et al. Long-term effects on cognitive function of postmenopausal hormone therapy prescribed to women aged 50 to 55 years. JAMA Intern. Med. 2013, 173, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Espeland, M.A.; Rapp, S.R.; Manson, J.E.; Goveas, J.S.; Shumaker, S.A.; Hayden, K.M.; Weitlauf, J.C.; Gaussoin, S.A.; Baker, L.D.; Padula, C.B.; et al. Long-term Effects on Cognitive Trajectories of Postmenopausal Hormone Therapy in Two Age Groups. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Girard, R.; Metereau, E.; Thomas, J.; Pugeat, M.; Qu, C.; Dreher, J.C. Hormone therapy at early post-menopause increases cognitive control-related prefrontal activity. Sci. Rep. 2017, 7, 44917. [Google Scholar] [CrossRef]
- Dunkin, J.; Rasgon, N.; Wagner-Steh, K.; David, S.; Altshuler, L.; Rapkin, A. Reproductive events modify the effects of estrogen replacement therapy on cognition in healthy postmenopausal women. Psychoneuroendocrinology 2005, 30, 284–296. [Google Scholar] [CrossRef]
- Song, X.; Wu, J.; Zhou, Y.; Feng, L.; Yuan, J.M.; Pan, A.; Koh, W.P. Reproductive and hormonal factors and risk of cognitive impairment among Singapore Chinese women. Am. J. Obs. Gynecol. 2020, 223, 410.e1–410.e23. [Google Scholar] [CrossRef] [PubMed]
- Dunkin, J.; Rasgon, N.; Zeller, M.; Wagner-Steh, K.; David, S.; Altshuler, L.; Rapkin, A. Estrogen replacement and cognition in postmenopausal women: Effect of years since menopause on response to treatment. Drug Dev. Res. 2005, 66, 150–159. [Google Scholar] [CrossRef]
- Resnick, S.M.; Espeland, M.A.; Jaramillo, S.A.; Hirsch, C.; Stefanick, M.L.; Murray, A.M.; Ockene, J.; Davatzikos, C. Postmenopausal hormone therapy and regional brain volumes: The WHIMS-MRI Study. Neurology 2009, 72, 135–142. [Google Scholar] [CrossRef]
- Tierney, M.C.; Oh, P.; Moineddin, R.; Greenblatt, E.M.; Snow, W.G.; Fisher, R.H.; Iazzetta, J.; Hyslop, P.S.; MacLusky, N.J. A randomized double-blind trial of the effects of hormone therapy on delayed verbal recall in older women. Psychoneuroendocrinology 2009, 34, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Kantarci, K.; Tosakulwong, N.; Lesnick, T.G.; Zuk, S.M.; Gunter, J.L.; Gleason, C.E.; Wharton, W.; Dowling, N.M.; Vemuri, P.; Senjem, M.L.; et al. Effects of hormone therapy on brain structure: A randomized controlled trial. Neurology 2016, 87, 887–896. [Google Scholar] [CrossRef]
- Shumaker, S.A.; Legault, C.; Kuller, L.; Rapp, S.R.; Thal, L.; Lane, D.S.; Fillit, H.; Stefanick, M.L.; Hendrix, S.L.; Lewis, C.E.; et al. Women’s Health Initiative Memory, S. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. JAMA 2004, 291, 2947–2958. [Google Scholar] [CrossRef]
- Mulnard, R.A.; Cotman, C.W.; Kawas, C.; van Dyck, C.H.; Sano, M.; Doody, R.; Koss, E.; Pfeiffer, E.; Jin, S.; Gamst, A.; et al. Estrogen replacement therapy for treatment of mild to moderate Alzheimer disease: A randomized controlled trial. Alzheimer’s Disease Cooperative Study. JAMA 2000, 283, 1007–1015. [Google Scholar] [CrossRef]
- Wang, P.N.; Liao, S.Q.; Liu, R.S.; Liu, C.Y.; Chao, H.T.; Lu, S.R.; Yu, H.Y.; Wang, S.J.; Liu, H.C. Effects of estrogen on cognition, mood, and cerebral blood flow in AD: A controlled study. Neurology 2000, 54, 2061–2066. [Google Scholar] [CrossRef]
- Valen-Sendstad, A.; Engedal, K.; Stray-Pedersen, B.; Group, A.S.; Strobel, C.; Barnett, L.; Meyer, N.; Nurminemi, M. Effects of hormone therapy on depressive symptoms and cognitive functions in women with Alzheimer disease: A 12 month randomized, double-blind, placebo-controlled study of low-dose estradiol and norethisterone. Am. J. Geriatr. Psychiatry 2010, 18, 11–20. [Google Scholar] [CrossRef]
- Asthana, S.; Craft, S.; Baker, L.D.; Raskind, M.A.; Birnbaum, R.S.; Lofgreen, C.P.; Veith, R.C.; Plymate, S.R. Cognitive and neuroendocrine response to transdermal estrogen in postmenopausal women with Alzheimer’s disease: Results of a placebo-controlled, double-blind, pilot study. Psychoneuroendocrinology 1999, 24, 657–677. [Google Scholar] [CrossRef]
- Asthana, S.; Baker, L.D.; Craft, S.; Stanczyk, F.Z.; Veith, R.C.; Raskind, M.A.; Plymate, S.R. High-dose estradiol improves cognition for women with AD: Results of a randomized study. Neurology 2001, 57, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Wharton, W.; Baker, L.D.; Gleason, C.E.; Dowling, M.; Barnet, J.H.; Johnson, S.; Carlsson, C.; Craft, S.; Asthana, S. Short-term hormone therapy with transdermal estradiol improves cognition for postmenopausal women with Alzheimer’s disease: Results of a randomized controlled trial. J. Alzheimers Dis. 2011, 26, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.J.; Battista, M. Estrogen replacement therapy: Effects on the cognitive functioning and clinical course of women with Alzheimer’s disease. Arch. Clin. Neuropsychol. 2004, 19, 769–778. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wolf, O.T.; Kudielka, B.M.; Hellhammer, D.H.; Torber, S.; McEwen, B.S.; Kirschbaum, C. Two weeks of transdermal estradiol treatment in postmenopausal elderly women and its effect on memory and mood: Verbal memory changes are associated with the treatment induced estradiol levels. Psychoneuroendocrinology 1999, 24, 727–741. [Google Scholar] [CrossRef]
- Maki, P.M. Hormone therapy and cognitive function: Is there a critical period for benefit? Neuroscience 2006, 138, 1027–1030. [Google Scholar] [CrossRef]
- Alhola, P.; Tuomisto, H.; Saarinen, R.; Portin, R.; Kalleinen, N.; Polo-Kantola, P. Estrogen + progestin therapy and cognition: A randomized placebo-controlled double-blind study. J. Obs. Gynaecol. Res. 2010, 36, 796–802. [Google Scholar] [CrossRef]
- Henderson, V.W.; St John, J.A.; Hodis, H.N.; McCleary, C.A.; Stanczyk, F.Z.; Shoupe, D.; Kono, N.; Dustin, L.; Allayee, H.; Mack, W.J. Cognitive effects of estradiol after menopause: A randomized trial of the timing hypothesis. Neurology 2016, 87, 699–708. [Google Scholar] [CrossRef]
- Pefanco, M.A.; Kenny, A.M.; Kaplan, R.F.; Kuchel, G.; Walsh, S.; Kleppinger, A.; Prestwood, K. The effect of 3-year treatment with 0.25 mg/day of micronized 17beta-estradiol on cognitive function in older postmenopausal women. J. Am. Geriatr. Soc. 2007, 55, 426–431. [Google Scholar] [CrossRef]
- Joffe, H.; Hall, J.E.; Gruber, S.; Sarmiento, I.A.; Cohen, L.S.; Yurgelun-Todd, D.; Martin, K.A. Estrogen therapy selectively enhances prefrontal cognitive processes: A randomized, double-blind, placebo-controlled study with functional magnetic resonance imaging in perimenopausal and recently postmenopausal women. Menopause 2006, 13, 411–422. [Google Scholar] [CrossRef]
- Kang, J.H.; Grodstein, F. Postmenopausal hormone therapy, timing of initiation, APOE and cognitive decline. Neurobiol. Aging 2012, 33, 1129–1137. [Google Scholar] [CrossRef]
- Shaywitz, S.E.; Shaywitz, B.A.; Pugh, K.R.; Fulbright, R.K.; Skudlarski, P.; Mencl, W.E.; Constable, R.T.; Naftolin, F.; Palter, S.F.; Marchione, K.E.; et al. Effect of estrogen on brain activation patterns in postmenopausal women during working memory tasks. JAMA 1999, 281, 1197–1202. [Google Scholar] [CrossRef] [PubMed]
- Sherwin, B.B. Estrogen and/or androgen replacement therapy and cognitive functioning in surgically menopausal women. Psychoneuroendocrinology 1988, 13, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Sherwin, B.B. Effects of estrogen on memory function in surgically menopausal women. Psychoneuroendocrinology 1992, 17, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Wroolie, T.E.; Kenna, H.A.; Williams, K.E.; Rasgon, N.L. Cognitive Effects of Hormone Therapy Continuation or Discontinuation in a Sample of Women at Risk for Alzheimer Disease. Am. J. Geriatr. Psychiatry 2015, 23, 1117–1126. [Google Scholar] [CrossRef]
- Silverman, D.H.; Geist, C.L.; Kenna, H.A.; Williams, K.; Wroolie, T.; Powers, B.; Brooks, J.; Rasgon, N.L. Differences in regional brain metabolism associated with specific formulations of hormone therapy in postmenopausal women at risk for AD. Psychoneuroendocrinology 2011, 36, 502–513. [Google Scholar] [CrossRef]
- Grady, D.; Yaffe, K.; Kristof, M.; Lin, F.; Richards, C.; Barrett-Connor, E. Effect of postmenopausal hormone therapy on cognitive function: The Heart and Estrogen/progestin Replacement Study. Am. J. Med. 2002, 113, 543–548. [Google Scholar] [CrossRef]
- Hojo, Y.; Murakami, G.; Mukai, H.; Higo, S.; Hatanaka, Y.; Ogiue-Ikeda, M.; Ishii, H.; Kimoto, T.; Kawato, S. Estrogen synthesis in the brain--role in synaptic plasticity and memory. Mol. Cell. Endocrinol. 2008, 290, 31–43. [Google Scholar] [CrossRef]
- Wright, J.V.; Schliesman, B.; Robinson, L. Comparative measurements of serum estriol, estradiol, and estrone in non-pregnant, premenopausal women; a preliminary investigation. Altern. Med. Rev. 1999, 4, 266–270. [Google Scholar]
- Pfaff, D.; Keiner, M. Atlas of estradiol-concentrating cells in the central nervous system of the female rat. J. Comp. Neurol. 1973, 151, 121–158. [Google Scholar]
- Stumpf, W.E. Autoradiographic techniques for the localization of hormones and drugs at the cellular and subcellular level. Acta Endocrinol. Suppl. 1971, 153, 205–222. [Google Scholar]
- Hara, Y.; Waters, E.M.; McEwen, B.S.; Morrison, J.H. Estrogen Effects on Cognitive and Synaptic Health Over the Lifecourse. Physiol. Rev. 2015, 95, 785–807. [Google Scholar] [CrossRef] [PubMed]
- Cheskis, B.J.; Greger, J.G.; Nagpal, S.; Freedman, L.P. Signaling by estrogens. J. Cell. Physiol. 2007, 213, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Kushner, P.J.; Agard, D.A.; Greene, G.L.; Scanlan, T.S.; Shiau, A.K.; Uht, R.M.; Webb, P. Estrogen receptor pathways to AP-1. J. Steroid Biochem. Mol. Biol. 2000, 74, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Toran-Allerand, C.D. The estrogen/neurotrophin connection during neural development: Is co-localization of estrogen receptors with the neurotrophins and their receptors biologically relevant? Dev. Neurosci. 1996, 18, 36–48. [Google Scholar] [CrossRef]
- Morrison, J.H.; Baxter, M.G. The ageing cortical synapse: Hallmarks and implications for cognitive decline. Nat. Rev. Neurosci. 2012, 13, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Setalo, G., Jr.; Guan, X.; Warren, M.; Toran-Allerand, C.D. Estrogen-induced activation of mitogen-activated protein kinase in cerebral cortical explants: Convergence of estrogen and neurotrophin signaling pathways. J. Neurosci. 1999, 19, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.; Woolley, C.S.; Frankfurt, M.; McEwen, B.S. Gonadal steroids regulate dendritic spine density in hippocampal pyramidal cells in adulthood. J. Neurosci. 1990, 10, 1286–1291. [Google Scholar] [CrossRef] [PubMed]
- Woolley, C.S.; Gould, E.; Frankfurt, M.; McEwen, B.S. Naturally occurring fluctuation in dendritic spine density on adult hippocampal pyramidal neurons. J. Neurosci. 1990, 10, 4035–4039. [Google Scholar] [CrossRef]
- Pike, C.J. Estrogen modulates neuronal Bcl-xL expression and beta-amyloid-induced apoptosis: Relevance to Alzheimer’s disease. J. Neurochem. 1999, 72, 1552–1563. [Google Scholar] [CrossRef]
- Nilsen, J.; Diaz Brinton, R. Mechanism of estrogen-mediated neuroprotection: Regulation of mitochondrial calcium and Bcl-2 expression. Proc. Natl. Acad. Sci. USA 2003, 100, 2842–2847. [Google Scholar] [CrossRef]
- Wong, M.; Thompson, T.L.; Moss, R.L. Nongenomic actions of estrogen in the brain: Physiological significance and cellular mechanisms. Crit. Rev. Neurobiol. 1996, 10, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Toran-Allerand, C.D. Estrogen and the brain: Beyond ER-alpha, ER-beta, and 17beta-estradiol. Ann. N. Y. Acad. Sci. 2005, 1052, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Filardo, E.J.; Quinn, J.A.; Frackelton, A.R., Jr.; Bland, K.I. Estrogen action via the G protein-coupled receptor, GPR30: Stimulation of adenylyl cyclase and cAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis. Mol. Endocrinol. 2002, 16, 70–84. [Google Scholar] [CrossRef]
- Nilsen, J.; Irwin, R.W.; Gallaher, T.K.; Brinton, R.D. Estradiol in vivo regulation of brain mitochondrial proteome. J. Neurosci. 2007, 27, 14069–14077. [Google Scholar] [CrossRef]
- Bryant, D.N.; Dorsa, D.M. Roles of estrogen receptors alpha and beta in sexually dimorphic neuroprotection against glutamate toxicity. Neuroscience 2010, 170, 1261–1269. [Google Scholar] [CrossRef]
- Lebesgue, D.; Chevaleyre, V.; Zukin, R.S.; Etgen, A.M. Estradiol rescues neurons from global ischemia-induced cell death: Multiple cellular pathways of neuroprotection. Steroids 2009, 74, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Gouras, G.K.; Greenfield, J.P.; Vincent, B.; Naslund, J.; Mazzarelli, L.; Fried, G.; Jovanovic, J.N.; Seeger, M.; Relkin, N.R.; et al. Estrogen reduces neuronal generation of Alzheimer beta-amyloid peptides. Nat. Med. 1998, 4, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Overk, C.R.; Lu, P.Y.; Wang, Y.T.; Choi, J.; Shaw, J.W.; Thatcher, G.R.; Mufson, E.J. Effects of aromatase inhibition versus gonadectomy on hippocampal complex amyloid pathology in triple transgenic mice. Neurobiol. Dis. 2012, 45, 479–487. [Google Scholar] [CrossRef]
- Yue, X.; Lu, M.; Lancaster, T.; Cao, P.; Honda, S.; Staufenbiel, M.; Harada, N.; Zhong, Z.; Shen, Y.; Li, R. Brain estrogen deficiency accelerates Abeta plaque formation in an Alzheimer’s disease animal model. Proc. Natl. Acad. Sci. USA 2005, 102, 19198–19203. [Google Scholar] [CrossRef]
- Duarte, F.H.; Jallad, R.S.; Bronstein, M.D. Estrogens and selective estrogen receptor modulators in acromegaly. Endocrine 2016, 54, 306–314. [Google Scholar] [CrossRef]
- Wang, Y.; Qu, D.; Wang, K. Therapeutic approaches to Alzheimer’s disease through stimulating of non-amyloidogenic processing of amyloid precursor protein. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2389–2403. [Google Scholar]
- Merlo, S.; Sortino, M.A. Estrogen activates matrix metalloproteinases-2 and-9 to increase beta amyloid degradation. Mol. Cell. Neurosci. 2012, 49, 423–429. [Google Scholar] [CrossRef]
- Wang, Z.-F.; Pan, Z.-Y.; Xu, C.-S.; Li, Z.-Q. Activation of G-protein coupled estrogen receptor 1 improves early-onset cognitive impairment via PI3K/Akt pathway in rats with traumatic brain injury. Biochem. Biophys. Res. Commun. 2017, 482, 948–953. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-de-la-Rosa, M.; Silva, I.; Nilsen, J.; Perez, M.M.; Garcia-Segura, L.M.; Avila, J.; Naftolin, F. Estradiol prevents neural tau hyperphosphorylation characteristic of Alzheimer’s disease. Ann. N. Y. Acad. Sci. 2005, 1052, 210–224. [Google Scholar] [CrossRef]
- Lee, E.S.; Sidoryk, M.; Jiang, H.; Yin, Z.; Aschner, M. Estrogen and tamoxifen reverse manganese-induced glutamate transporter impairment in astrocytes. J. Neurochem. 2009, 110, 530–544. [Google Scholar] [CrossRef] [PubMed]
- Simpkins, J.W.; Perez, E.; Wang, X.; Yang, S.; Wen, Y.; Singh, M. The potential for estrogens in preventing Alzheimer’s disease and vascular dementia. Ther. Adv. Neurol. Disord. 2009, 2, 31–49. [Google Scholar] [CrossRef] [PubMed]
- Foy, M.R.; Baudry, M.; Diaz Brinton, R.; Thompson, R.F. Estrogen and hippocampal plasticity in rodent models. J. Alzheimers Dis. 2008, 15, 589–603. [Google Scholar] [CrossRef]
- Rettberg, J.R.; Yao, J.; Brinton, R.D. Estrogen: A master regulator of bioenergetic systems in the brain and body. Front. Neuroendocrinol. 2014, 35, 8–30. [Google Scholar] [CrossRef]
- Lin, J.; Epel, E. Stress and telomere shortening: Insights from cellular mechanisms. Ageing Res. Rev. 2022, 73, 101507. [Google Scholar] [CrossRef]
- Levine, A.J.; Puzio-Kuter, A.M.; Chan, C.S.; Hainaut, P. The role of the p53 protein in stem-cell biology and epigenetic regulation. Cold Spring Harb. Perspect. Med. 2016, 6, a026153. [Google Scholar] [CrossRef]
- Ratnakumar, A.; Zimmerman, S.E.; Jordan, B.A.; Mar, J.C. Estrogen activates Alzheimer’s disease genes. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2019, 5, 906–917. [Google Scholar] [CrossRef] [PubMed]
- Sohrabji, F.; Miranda, R.; Toran-Allerand, C.D. Identification of a putative estrogen response element in the gene encoding brain-derived neurotrophic factor. Proc. Natl. Acad. Sci. USA 1995, 92, 11110–11114. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Ait-Ghezala, G.; Sambamurti, K.; Gao, F.; Shen, Y.; Li, R. Sex-specific regulation of β-Secretase: A novel estrogen response element (ERE)-dependent mechanism in Alzheimer’s disease. J. Neurosci. 2022, 42, 1154–1165. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Jia, J.; Ji, X.; Tian, T. Estrogen associated gene polymorphisms and their interactions in the progress of Alzheimer’s disease. Prog. Neurobiol. 2013, 111, 53–74. [Google Scholar] [CrossRef]
- Mermelstein, P.G.; Micevych, P.E. Nervous system physiology regulated by membrane estrogen receptors. Rev. Neurosci. 2008, 19, 413–424. [Google Scholar] [CrossRef]
- Micevych, P.E.; Mermelstein, P.G. Membrane estrogen receptors acting through metabotropic glutamate receptors: An emerging mechanism of estrogen action in brain. Mol. Neurobiol. 2008, 38, 66–77. [Google Scholar] [CrossRef]
- Micevych, P.; Dominguez, R. Membrane estradiol signaling in the brain. Front. Neuroendocr. 2009, 30, 315–327. [Google Scholar] [CrossRef]
- Boulware, M.I.; Mermelstein, P.G. Membrane estrogen receptors activate metabotropic glutamate receptors to influence nervous system physiology. Steroids 2009, 74, 608–613. [Google Scholar] [CrossRef]
- Rocca, W.A.; Grossardt, B.R.; Shuster, L.T. Oophorectomy, menopause, estrogen treatment, and cognitive aging: Clinical evidence for a window of opportunity. Brain Res. 2011, 1379, 188–198. [Google Scholar] [CrossRef]
- Genazzani, A.R.; Pluchino, N.; Luisi, S.; Luisi, M. Estrogen, cognition and female ageing. Hum. Reprod. Update 2007, 13, 175–187. [Google Scholar] [CrossRef]
- Henderson, V.W.; Sherwin, B.B. Surgical versus natural menopause: Cognitive issues. Menopause 2007, 14, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Sherwin, B.B. The clinical relevance of the relationship between estrogen and cognition in women. J. Steroid Biochem. Mol. Biol. 2007, 106, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Sherwin, B.B. The critical period hypothesis: Can it explain discrepancies in the oestrogen-cognition literature? J. Neuroendocr. 2007, 19, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Sherwin, B.B. Estrogen therapy: Is time of initiation critical for neuroprotection? Nat. Rev. Endocrinol. 2009, 5, 620–627. [Google Scholar] [CrossRef]
- King, R.; Travers, C.; O’Neill, S.; Byrne, G.; Khoo, S.K. The influence of postmenopausal hormone replacement therapy on cognitive functioning: Results from an observational study. J. Br. Menopause Soc. 2004, 10, 103–107. [Google Scholar] [CrossRef]
- Henderson, V.W.; Benke, K.S.; Green, R.C.; Cupples, L.A.; Farrer, L.A.; Group, M.S. Postmenopausal hormone therapy and Alzheimer’s disease risk: Interaction with age. J. Neurol. Neurosurg. Psychiatry 2005, 76, 103–105. [Google Scholar] [CrossRef]
- Whitmer, R.A.; Quesenberry, C.P.; Zhou, J.; Yaffe, K. Timing of hormone therapy and dementia: The critical window theory revisited. Ann. Neurol. 2011, 69, 163–169. [Google Scholar] [CrossRef]
- Hamilton, R.T.; Rettberg, J.R.; Mao, Z.; To, J.; Zhao, L.; Appt, S.E.; Register, T.C.; Kaplan, J.R.; Brinton, R.D. Hippocampal responsiveness to 17beta-estradiol and equol after long-term ovariectomy: Implication for a therapeutic window of opportunity. Brain Res. 2011, 1379, 11–22. [Google Scholar] [CrossRef]
- Daniel, J.M.; Sulzer, J.K.; Hulst, J.L. Estrogen increases the sensitivity of ovariectomized rats to the disruptive effects produced by antagonism of D2 but not D1 dopamine receptors during performance of a response learning task. Horm. Behav. 2006, 49, 38–44. [Google Scholar] [CrossRef]
- Bohacek, J.; Daniel, J.M. The beneficial effects of estradiol on attentional processes are dependent on timing of treatment initiation following ovariectomy in middle-aged rats. Psychoneuroendocrinology 2010, 35, 694–705. [Google Scholar] [CrossRef]
- Smith, C.C.; Vedder, L.C.; Nelson, A.R.; Bredemann, T.M.; McMahon, L.L. Duration of estrogen deprivation, not chronological age, prevents estrogen’s ability to enhance hippocampal synaptic physiology. Proc. Natl. Acad. Sci. USA 2010, 107, 19543–19548. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.W.; Adelman, J.P.; Maylie, J. Ovarian hormone deficiency reduces intrinsic excitability and abolishes acute estrogen sensitivity in hippocampal CA1 pyramidal neurons. J. Neurosci. 2011, 31, 2638–2648. [Google Scholar] [CrossRef] [PubMed]
- Koszegi, Z.; Abraham, I.M. Effect of ageing on post-lesion oestradiol treatment on mouse cholinergic neurones in vivo. J. Neuroendocr. 2012, 24, 1243–1248. [Google Scholar] [CrossRef] [PubMed]
- Rocca, W.A.; Bower, J.H.; Maraganore, D.M.; Ahlskog, J.E.; Grossardt, B.R.; de Andrade, M.; Melton, L.J., 3rd. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology 2007, 69, 1074–1083. [Google Scholar] [CrossRef]
- Adams, M.M.; Fink, S.E.; Shah, R.A.; Janssen, W.G.; Hayashi, S.; Milner, T.A.; McEwen, B.S.; Morrison, J.H. Estrogen and aging affect the subcellular distribution of estrogen receptor-alpha in the hippocampus of female rats. J. Neurosci. 2002, 22, 3608–3614. [Google Scholar] [CrossRef]
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, G.; Guo, Z.; Cui, R.; Wang, L.; Zhou, C. Quantitative evaluation of the interventional effect of estrogen on Alzheimer’s disease. Chin. J. Clin. Rehabil. 2006, 10, 37–91. [Google Scholar]
- Bean, L.A.; Kumar, A.; Rani, A.; Guidi, M.; Rosario, A.M.; Cruz, P.E.; Golde, T.E.; Foster, T.C. Re-Opening the Critical Window for Estrogen Therapy. J. Neurosci. 2015, 35, 16077–16093. [Google Scholar] [CrossRef]
- Brinton, R.D. Investigative models for determining hormone therapy-induced outcomes in brain: Evidence in support of a healthy cell bias of estrogen action. Ann. N. Y. Acad. Sci. 2005, 1052, 57–74. [Google Scholar] [CrossRef]
- Brinton, R.D. The healthy cell bias of estrogen action: Mitochondrial bioenergetics and neurological implications. Trends Neurosci. 2008, 31, 529–537. [Google Scholar] [CrossRef]
- Stone, D.J.; Rozovsky, I.; Morgan, T.E.; Anderson, C.P.; Finch, C.E. Increased synaptic sprouting in response to estrogen via an apolipoprotein E-dependent mechanism: Implications for Alzheimer’s disease. J. Neurosci. 1998, 18, 3180–3185. [Google Scholar] [CrossRef] [PubMed]
- Riedel, B.C.; Thompson, P.M.; Brinton, R.D. Age, APOE and sex: Triad of risk of Alzheimer’s disease. J. Steroid Biochem. Mol. Biol. 2016, 160, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.A.; Krul, E.S.; Lin, R.C.; Schonfeld, G. Regulation of lipoprotein metabolism by estrogen in inbred strains of mice occurs primarily by posttranscriptional mechanisms. Mol. Cell. Biochem. 1997, 173, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.A.K.; Srivastava, N.; Averna, M.; Lin, R.C.; Korach, K.S.; Lubahn, D.B.; Schonfeld, G. Estrogen up-regulates apolipoprotein E (ApoE) gene expression by increasing ApoE mRNA in the translating pool via the estrogen receptor α-mediated pathway. J. Biol. Chem. 1997, 272, 33360–33366. [Google Scholar] [CrossRef]
- Wisniewski, T.; Drummond, E. APOE-amyloid interaction: Therapeutic targets. Neurobiol. Dis. 2020, 138, 104784. [Google Scholar] [CrossRef]
- Lambert, J.; Coyle, N.; Lendon, C. The allelic modulation of apolipoprotein E expression by oestrogen: Potential relevance for Alzheimer’s disease. J. Med. Genet. 2004, 41, 104–112. [Google Scholar] [CrossRef]
- Jacobs, E.G.; Kroenke, C.; Lin, J.; Epel, E.S.; Kenna, H.A.; Blackburn, E.H.; Rasgon, N.L. Accelerated cell aging in female APOE-ε4 carriers: Implications for hormone therapy use. PLoS ONE 2013, 8, e54713. [Google Scholar] [CrossRef]
- Govindpani, K.; McNamara, L.G.; Smith, N.R.; Vinnakota, C.; Waldvogel, H.J.; Faull, R.L.; Kwakowsky, A. Vascular Dysfunction in Alzheimer’s Disease: A Prelude to the Pathological Process or a Consequence of It? J. Clin. Med. 2019, 8, 651. [Google Scholar] [CrossRef]
- Mesulam, M.M. Cholinergic circuitry of the human nucleus basalis and its fate in Alzheimer’s disease. J. Comp. Neurol. 2013, 521, 4124–4144. [Google Scholar] [CrossRef]
- Sims, N.R.; Bowen, D.M.; Allen, S.J.; Smith, C.C.; Neary, D.; Thomas, D.J.; Davison, A.N. Presynaptic cholinergic dysfunction in patients with dementia. J. Neurochem. 1983, 40, 503–509. [Google Scholar] [CrossRef]
- Mufson, E.J.; Counts, S.E.; Perez, S.E.; Ginsberg, S.D. Cholinergic system during the progression of Alzheimer’s disease: Therapeutic implications. Expert Rev. Neurother. 2008, 8, 1703–1718. [Google Scholar] [CrossRef] [PubMed]
- Kwakowsky, A.; Milne, M.R.; Waldvogel, H.J.; Faull, R.L. Effect of Estradiol on Neurotrophin Receptors in Basal Forebrain Cholinergic Neurons: Relevance for Alzheimer’s Disease. Int. J. Mol. Sci. 2016, 17, 2122. [Google Scholar] [CrossRef] [PubMed]
- Milne, M.R.; Haug, C.A.; Abraham, I.M.; Kwakowsky, A. Estradiol modulation of neurotrophin receptor expression in female mouse basal forebrain cholinergic neurons in vivo. Endocrinology 2015, 156, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.; Morici, J.F.; Zanoni, M.B.; Bekinschtein, P. Brain-Derived Neurotrophic Factor: A Key Molecule for Memory in the Healthy and the Pathological Brain. Front. Cell. Neurosci. 2019, 13, 363. [Google Scholar] [CrossRef] [PubMed]
- Su, Q.; Cheng, Y.; Jin, K.; Cheng, J.; Lin, Y.; Lin, Z.; Wang, L.; Shao, B. Estrogen therapy increases BDNF expression and improves post-stroke depression in ovariectomy-treated rats. Exp. Med. 2016, 12, 1843–1848. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tanapat, P.; Hastings, N.B.; Reeves, A.J.; Gould, E. Estrogen stimulates a transient increase in the number of new neurons in the dentate gyrus of the adult female rat. J. Neurosci. 1999, 19, 5792–5801. [Google Scholar] [CrossRef]
- Scharfman, H.E.; MacLusky, N.J. Estrogen and brain-derived neurotrophic factor (BDNF) in hippocampus: Complexity of steroid hormone-growth factor interactions in the adult CNS. Front. Neuroendocr. 2006, 27, 415–435. [Google Scholar] [CrossRef]
- Mangalmurti, A.; Lukens, J.R. How neurons die in Alzheimer’s disease: Implications for neuroinflammation. Curr. Opin. Neurobiol. 2022, 75, 102575. [Google Scholar] [CrossRef]
- Palpagama, T.H.; Waldvogel, H.J.; Faull, R.L.M.; Kwakowsky, A. The Role of Microglia and Astrocytes in Huntington’s Disease. Front. Mol. Neurosci. 2019, 12, 258. [Google Scholar] [CrossRef]
- Dhapola, R.; Hota, S.S.; Sarma, P.; Bhattacharyya, A.; Medhi, B.; Reddy, D.H. Recent advances in molecular pathways and therapeutic implications targeting neuroinflammation for Alzheimer’s disease. Inflammopharmacology 2021, 29, 1669–1681. [Google Scholar] [CrossRef]
- Sarvari, M.; Hrabovszky, E.; Kallo, I.; Solymosi, N.; Toth, K.; Liko, I.; Szeles, J.; Maho, S.; Molnar, B.; Liposits, Z. Estrogens regulate neuroinflammatory genes via estrogen receptors alpha and beta in the frontal cortex of middle-aged female rats. J. Neuroinflammation 2011, 8, 82. [Google Scholar] [CrossRef] [PubMed]
- Vegeto, E.; Belcredito, S.; Ghisletti, S.; Meda, C.; Etteri, S.; Maggi, A. The endogenous estrogen status regulates microglia reactivity in animal models of neuroinflammation. Endocrinology 2006, 147, 2263–2272. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.; Raval, A.P. The peri-menopause in a woman’s life: A systemic inflammatory phase that enables later neurodegenerative disease. J. Neuroinflammation 2020, 17, 317. [Google Scholar] [CrossRef] [PubMed]
- Sortino, M.; Chisari, M.; Merlo, S.; Vancheri, C.; Caruso, M.; Nicoletti, F.; Canonico, P.; Copani, A. Glia mediates the neuroprotective action of estradiol on β-amyloid-induced neuronal death. Endocrinology 2004, 145, 5080–5086. [Google Scholar] [CrossRef] [PubMed]
- Struble, R.G.; Nathan, B.P.; Cady, C.; Cheng, X.; McAsey, M. Estradiol regulation of astroglia and apolipoprotein E: An important role in neuronal regeneration. Exp. Gerontol. 2007, 42, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Nilsen, J.; Brinton, R.D. Dose and temporal pattern of estrogen exposure determines neuroprotective outcome in hippocampal neurons: Therapeutic implications. Endocrinology 2006, 147, 5303–5313. [Google Scholar] [CrossRef]
- Wang, W.; Zhao, F.; Ma, X.; Perry, G.; Zhu, X. Mitochondria dysfunction in the pathogenesis of Alzheimer’s disease: Recent advances. Mol. Neurodegener. 2020, 15, 1–22. [Google Scholar] [CrossRef]
- Hu, Z.; Shi, P. Regularity and Complexity of Human Electroencephalogram Dynamics: Applications to Diagnosis of Alzheimers Disease. In Proceedings of the 18th International Conference on Pattern Recognition (ICPR’06), Hong Kong, China, 20–24 August 2006; pp. 245–248. [Google Scholar]
- Yao, J.; Hamilton, R.T.; Cadenas, E.; Brinton, R.D. Decline in mitochondrial bioenergetics and shift to ketogenic profile in brain during reproductive senescence. Biochim. Biophys. Acta 2010, 1800, 1121–1126. [Google Scholar] [CrossRef]
- Nilsen, J.; Brinton, R.D. Divergent impact of progesterone and medroxyprogesterone acetate (Provera) on nuclear mitogen-activated protein kinase signaling. Proc. Natl. Acad. Sci. USA 2003, 100, 10506–10511. [Google Scholar] [CrossRef]
- Gupta, A.; Caffrey, E.; Callagy, G.; Gupta, S. Oestrogen-dependent regulation of miRNA biogenesis: Many ways to skin the cat. Biochem. Soc. Trans. 2012, 40, 752–758. [Google Scholar] [CrossRef]
- Rao, Y.S.; Mott, N.N.; Wang, Y.; Chung, W.C.; Pak, T.R. MicroRNAs in the aging female brain: A putative mechanism for age-specific estrogen effects. Endocrinology 2013, 154, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Rao, Y.S.; Shults, C.L.; Pinceti, E.; Pak, T.R. Prolonged ovarian hormone deprivation alters the effects of 17β-estradiol on microRNA expression in the aged female rat hypothalamus. Oncotarget 2015, 6, 36965. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, A.S.; Andre, G.; Vellas, B.; Touchon, J.; Pere, J.J.; French Study, G. No additional benefit of HRT on response to rivastigmine in menopausal women with AD. Neurology 2003, 60, 148–149. [Google Scholar] [CrossRef] [PubMed]
- Sundermann, E.; Gilbert, P.E.; Murphy, C. Estrogen and performance in recognition memory for olfactory and visual stimuli in females diagnosed with Alzheimer’s disease. J. Int. Neuropsychol. Soc. 2006, 12, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Ohkura, T.; Isse, K.; Akazawa, K.; Hamamoto, M.; Yaoi, Y.; Hagino, N. Evaluation of estrogen treatment in female patients with dementia of the Alzheimer type. Endocr. J. 1994, 41, 361–371. [Google Scholar] [CrossRef]
- Ohkura, T.; Isse, K.; Akazawa, K.; Hamamoto, M.; Yaoi, Y.; Hagino, N. Long-term estrogen replacement therapy in female patients with dementia of the Alzheimer type: 7 case reports. Dementia 1995, 6, 99–107. [Google Scholar] [CrossRef]
- LeBlanc, E.S.; Neiss, M.B.; Carello, P.E.; Samuels, M.H.; Janowsky, J.S. Hot flashes and estrogen therapy do not influence cognition in early menopausal women. Menopause 2007, 14, 191–202. [Google Scholar] [CrossRef]
- Maki, P.M.; Gast, M.J.; Vieweg, A.J.; Burriss, S.W.; Yaffe, K. Hormone therapy in menopausal women with cognitive complaints: A randomized, double-blind trial. Neurology 2007, 69, 1322–1330. [Google Scholar] [CrossRef]
- Linzmayer, L.; Semlitsch, H.V.; Saletu, B.; Bock, G.; Saletu-Zyhlarz, G.; Zoghlami, A.; Gruber, D.; Metka, M.; Huber, J.; Oettel, M.; et al. Double-blind, placebo-controlled psychometric studies on the effects of a combined estrogen-progestin regimen versus estrogen alone on performance, mood and personality of menopausal syndrome patients. Arzneimittelforschung 2001, 51, 238–245. [Google Scholar] [CrossRef]
- Hendrick, V.; Altshuler, L.L.; Suri, R. Hormonal changes in the postpartum and implications for postpartum depression. Psychosomatics 1998, 39, 93–101. [Google Scholar] [CrossRef]
- Smith, C.; McCleary, C.; Murdock, G.; Wilshire, T.; Buckwalter, D.; Bretsky, P.; Marmol, L.; Gorsuch, R.; Buckwalter, J. Lifelong estrogen exposure and cognitive performance in elderly women. Brain Cogn. 1999, 39, 203–218. [Google Scholar] [CrossRef]
- Rasgon, N.L.; Silverman, D.; Siddarth, P.; Miller, K.; Ercoli, L.M.; Elman, S.; Lavretsky, H.; Huang, S.-C.; Phelps, M.E.; Small, G.W. Estrogen use and brain metabolic change in postmenopausal women. Neurobiol. Aging 2005, 26, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Tulchinsky, D.; Ryan, K.J. Maternal-Fetal Endocrinology; Saunders: Philadelphia, PA, USA, 1980. [Google Scholar]
- Beeri, M.S.; Rapp, M.; Schmeidler, J.; Reichenberg, A.; Purohit, D.P.; Perl, D.P.; Grossman, H.T.; Prohovnik, I.; Haroutunian, V.; Silverman, J.M. Number of children is associated with neuropathology of Alzheimer’s disease in women. Neurobiol. Aging 2009, 30, 1184–1191. [Google Scholar] [CrossRef] [PubMed]
- McLay, R.N.; Maki, P.M.; Lyketsos, C.G. Nulliparity and late menopause are associated with decreased cognitive decline. J. Neuropsychiatry Clin. Neurosci. 2003, 15, 161–167. [Google Scholar] [CrossRef]
- Heys, M.; Jiang, C.; Cheng, K.K.; Zhang, W.; Yeung, S.L.A.; Lam, T.H.; Leung, G.M.; Schooling, C.M. Life long endogenous estrogen exposure and later adulthood cognitive function in a population of naturally postmenopausal women from Southern China: The Guangzhou Biobank Cohort Study. Psychoneuroendocrinology 2011, 36, 864–873. [Google Scholar] [CrossRef] [PubMed]
- Egan, K.R.; Gleason, C.E. Longer duration of hormonal contraceptive use predicts better cognitive outcomes later in life. J. Women’s Health 2012, 21, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Harris, K.; Peters, S.A.; Woodward, M. Reproductive factors and the risk of incident dementia: A cohort study of UK Biobank participants. PLoS Med. 2022, 19, e1003955. [Google Scholar] [CrossRef]
- Canonico, M.; Oger, E.; Plu-Bureau, G.; Conard, J.; Meyer, G.; Levesque, H.; Trillot, N.; Barrellier, M.T.; Wahl, D.; Emmerich, J.; et al. Hormone therapy and venous thromboembolism among postmenopausal women: Impact of the route of estrogen administration and progestogens: The ESTHER study. Circulation 2007, 115, 840–845. [Google Scholar] [CrossRef]
- Maki, P.M.; Resnick, S.M. Effects of estrogen on patterns of brain activity at rest and during cognitive activity: A review of neuroimaging studies. Neuroimage 2001, 14, 789–801. [Google Scholar] [CrossRef]
- Ditkoff, E.C.; Cassidenti, D.L.; Paulson, R.J.; Sauer, M.V.; Paul, W.L.; Rivier, J.; Yen, S.S.; Lobo, R.A. The gonadotropin-releasing hormone antagonist (Nal-Glu) acutely blocks the luteinizing hormone surge but allows for resumption of folliculogenesis in normal women. Am. J. Obs. Gynecol. 1991, 165, 1811–1817. [Google Scholar] [CrossRef]
- Sherwin, B.B.; Phillips, S. Estrogen and cognitive functioning in surgically menopausal women. Ann. N. Y. Acad. Sci. 1990, 592, 474–475. [Google Scholar] [CrossRef]
- Chang, W.C.; Wang, J.H.; Ding, D.C. Conjugated equine estrogen used in postmenopausal women associated with a higher risk of stroke than estradiol. Sci. Rep. 2021, 11, 10801. [Google Scholar] [CrossRef] [PubMed]
- Rothman, M.S.; Carlson, N.E.; Xu, M.; Wang, C.; Swerdloff, R.; Lee, P.; Goh, V.H.; Ridgway, E.C.; Wierman, M.E. Reexamination of testosterone, dihydrotestosterone, estradiol and estrone levels across the menstrual cycle and in postmenopausal women measured by liquid chromatography-tandem mass spectrometry. Steroids 2011, 76, 177–182. [Google Scholar] [CrossRef]
- Mashchak, C.A.; Lobo, R.A.; Dozono-Takano, R.; Eggena, P.; Nakamura, R.M.; Brenner, P.F.; Mishell, D.R., Jr. Comparison of pharmacodynamic properties of various estrogen formulations. Am. J. Obs. Gynecol. 1982, 144, 511–518. [Google Scholar] [CrossRef]
- Nelson, H.D. Commonly used types of postmenopausal estrogen for treatment of hot flashes: Scientific review. JAMA 2004, 291, 1610–1620. [Google Scholar] [CrossRef]
- Engler-Chiurazzi, E.; Tsang, C.; Nonnenmacher, S.; Liang, W.S.; Corneveaux, J.J.; Prokai, L.; Huentelman, M.J.; Bimonte-Nelson, H.A. Tonic Premarin dose-dependently enhances memory, affects neurotrophin protein levels and alters gene expression in middle-aged rats. Neurobiol. Aging 2011, 32, 680–697. [Google Scholar] [CrossRef] [PubMed]
- Engler-Chiurazzi, E.B.; Talboom, J.S.; Braden, B.B.; Tsang, C.W.; Mennenga, S.; Andrews, M.; Demers, L.M.; Bimonte-Nelson, H.A. Continuous estrone treatment impairs spatial memory and does not impact number of basal forebrain cholinergic neurons in the surgically menopausal middle-aged rat. Horm. Behav. 2012, 62, 1–9. [Google Scholar] [CrossRef]
- O’Connell, M.B. Pharmacokinetic and pharmacologic variation between different estrogen products. J. Clin. Pharm. 1995, 35, 18S–24S. [Google Scholar] [CrossRef]
- Kristofikova, Z.; Springer, T.; Gedeonova, E.; Hofmannova, A.; Ricny, J.; Hromadkova, L.; Vyhnalek, M.; Laczo, J.; Nikolai, T.; Hort, J.; et al. Interactions of 17beta-Hydroxysteroid Dehydrogenase Type 10 and Cyclophilin D in Alzheimer’s Disease. Neurochem. Res. 2020, 45, 915–927. [Google Scholar] [CrossRef]
- Hiroi, R.; Weyrich, G.; Koebele, S.V.; Mennenga, S.E.; Talboom, J.S.; Hewitt, L.T.; Lavery, C.N.; Mendoza, P.; Jordan, A.; Bimonte-Nelson, H.A. Benefits of Hormone Therapy Estrogens Depend on Estrogen Type: 17beta-Estradiol and Conjugated Equine Estrogens Have Differential Effects on Cognitive, Anxiety-Like, and Depressive-Like Behaviors and Increase Tryptophan Hydroxylase-2 mRNA Levels in Dorsal Raphe Nucleus Subregions. Front. Neurosci. 2016, 10, 517. [Google Scholar]
- Stanczyk, F.Z.; Clarke, N.J. Measurement of estradiol--challenges ahead. J. Clin. Endocrinol. Metab. 2014, 99, 56–58. [Google Scholar] [CrossRef] [PubMed]
- Hodis, H.N.; Mack, W.J.; Henderson, V.W.; Shoupe, D.; Budoff, M.J.; Hwang-Levine, J.; Li, Y.; Feng, M.; Dustin, L.; Kono, N.; et al. Vascular Effects of Early versus Late Postmenopausal Treatment with Estradiol. N. Engl. J. Med. 2016, 374, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Krieger, N.; Lowy, I.; Aronowitz, R.; Bigby, J.; Dickersin, K.; Garner, E.; Gaudilliere, J.P.; Hinestrosa, C.; Hubbard, R.; Johnson, P.A.; et al. Hormone replacement therapy, cancer, controversies, and women’s health: Historical, epidemiological, biological, clinical, and advocacy perspectives. J. Epidemiol. Community Health 2005, 59, 740–748. [Google Scholar] [CrossRef]
- Campagnoli, C.; Clavel-Chapelon, F.; Kaaks, R.; Peris, C.; Berrino, F. Progestins and progesterone in hormone replacement therapy and the risk of breast cancer. J. Steroid Biochem. Mol. Biol. 2005, 96, 95–108. [Google Scholar] [CrossRef]
- Bimonte-Nelson, H.A.; Nelson, M.E.; Granholm, A.C. Progesterone counteracts estrogen-induced increases in neurotrophins in the aged female rat brain. Neuroreport 2004, 15, 2659–2663. [Google Scholar] [CrossRef] [PubMed]
- Bimonte-Nelson, H.A.; Francis, K.R.; Umphlet, C.D.; Granholm, A.C. Progesterone reverses the spatial memory enhancements initiated by tonic and cyclic oestrogen therapy in middle-aged ovariectomized female rats. Eur. J. Neurosci. 2006, 24, 229–242. [Google Scholar] [CrossRef]
- Rosario, E.R.; Ramsden, M.; Pike, C.J. Progestins inhibit the neuroprotective effects of estrogen in rat hippocampus. Brain Res. 2006, 1099, 206–210. [Google Scholar] [CrossRef]
- Carroll, J.C.; Rosario, E.R.; Pike, C.J. Progesterone blocks estrogen neuroprotection from kainate in middle-aged female rats. Neurosci. Lett. 2008, 445, 229–232. [Google Scholar] [CrossRef][Green Version]
- Yao, J.; Chen, S.; Cadenas, E.; Brinton, R.D. Estrogen protection against mitochondrial toxin-induced cell death in hippocampal neurons: Antagonism by progesterone. Brain Res. 2011, 1379, 2–10. [Google Scholar] [CrossRef]
- Pike, C.J.; Carroll, J.C.; Rosario, E.R.; Barron, A.M. Protective actions of sex steroid hormones in Alzheimer’s disease. Front. Neuroendocr. 2009, 30, 239–258. [Google Scholar] [CrossRef]
- Nelson, E.R.; Habibi, H.R. Estrogen receptor function and regulation in fish and other vertebrates. Gen. Comp. Endocrinol. 2013, 192, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Yaffe, K.; Krueger, K.; Cummings, S.R.; Blackwell, T.; Henderson, V.W.; Sarkar, S.; Ensrud, K.; Grady, D. Effect of raloxifene on prevention of dementia and cognitive impairment in older women: The Multiple Outcomes of Raloxifene Evaluation (MORE) randomized trial. Am. J. Psychiatry 2005, 162, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Pickar, J.H.; Boucher, M.; Morgenstern, D. Tissue selective estrogen complex (TSEC): A review. Menopause 2018, 25, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Kloosterboer, H.J. Tissue-selectivity: The mechanism of action of tibolone. Maturitas 2004, 48, S30–S40. [Google Scholar] [CrossRef]
- Sharma, S.; Mahajan, A.; Kumar, S.; Tandon, V.R. Tibolone: A Selective Tissue Estrogenic Activity Regulator. Jk. Sci. 2006, 8, 203–204. [Google Scholar]
- Merlo, S.; Spampinato, S.F.; Sortino, M.A. Estrogen and Alzheimer’s disease: Still an attractive topic despite disappointment from early clinical results. Eur. J. Pharmacol. 2017, 817, 51–58. [Google Scholar] [CrossRef]
- Han, M.; Chang, J.; Choi, S.; Cho, Y.; Lee, G.; Park, S.M. Association of tibolone and dementia risk: A cohort study using Korean claims data. Gynecol. Endocrinol. 2021, 37, 567–571. [Google Scholar] [CrossRef]
- Carlson, R.W. The history and mechanism of action of fulvestrant. Clin. Breast Cancer 2005, 6, S5–S8. [Google Scholar] [CrossRef]
- Kousteni, S.; Chen, J.R.; Bellido, T.; Han, L.; Ali, A.A.; O’Brien, C.A.; Plotkin, L.; Fu, Q.; Mancino, A.T.; Wen, Y.; et al. Reversal of bone loss in mice by nongenotropic signaling of sex steroids. Science 2002, 298, 843–846. [Google Scholar] [CrossRef]
- Kwakowsky, A.; Koszegi, Z.; Cheong, R.Y.; Abraham, I.M. Neuroprotective effects of non-classical estrogen-like signaling activators: From mechanism to potential implications. CNS Neurol. Disord. Drug Targets 2013, 12, 1219–1225. [Google Scholar] [CrossRef]
- Kwakowsky, A.; Potapov, K.; Kim, S.; Peppercorn, K.; Tate, W.P.; Abraham, I.M. Treatment of beta amyloid 1-42 (Abeta(1-42))-induced basal forebrain cholinergic damage by a non-classical estrogen signaling activator in vivo. Sci. Rep. 2016, 6, 21101. [Google Scholar] [CrossRef] [PubMed]
- Cordey, M.; Gundimeda, U.; Gopalakrishna, R.; Pike, C.J. The synthetic estrogen 4-estren-3 alpha,17 beta-diol (estren) induces estrogen-like neuroprotection. Neurobiol. Dis. 2005, 19, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, G.G.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.A. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef]
- Chamniansawat, S.; Chongthammakun, S. A priming role of local estrogen on exogenous estrogen-mediated synaptic plasticity and neuroprotection. Exp. Mol. Med. 2012, 44, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; He, P.; Cui, J.; Staufenbiel, M.; Harada, N.; Shen, Y. Brain endogenous estrogen levels determine responses to estrogen replacement therapy via regulation of BACE1 and NEP in female Alzheimer’s transgenic mice. Mol. Neurobiol. 2013, 47, 857–867. [Google Scholar] [CrossRef] [PubMed]
- Jefferson, W.N.; Padilla-Banks, E.; Newbold, R.R. Disruption of the female reproductive system by the phy-toestrogen genistein. Reprod. Toxicol. 2007, 23, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Cederroth, C.R.; Nef, S. Soy, phytoestrogens and metabolism: A review. Mol. Cell. Endocrinol. 2009, 304, 30–42. [Google Scholar] [CrossRef]
- Penza, M.; Montani, C.; Romani, A.; Vignolini, P.; Pampaloni, B.; Tanini, A.; Di Lorenzo, D. Genistein affects adipose tissue deposition in a dose-dependent and gender-specific manner. Endocrinology 2006, 147, 5740–5751. [Google Scholar] [CrossRef]
- Nikaido, Y.; Yoshizawa, K.; Danbara, N. Effects of maternal xenoestrogen exposure on development of the reproductive tract and mammary gland in female CD-1 mouse offspring. Reprod. Toxicol. 2004, 18, 803–811. [Google Scholar] [CrossRef]
- Duffy, C.; Perez, K.; Partridge, A. Implications of phytoestrogen intake for breast cancer. CA A Cancer J. Clin. 2007, 57, 260–277. [Google Scholar] [CrossRef]
| Authors (Year) | Origin | Study Type | Study Groups | Outcome | Type | Dose (mg/day) | ROA | Menopause Stage | Age (Years) | Duration of HRT Use | Main Finding or Conclusion (HRT Related) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mortel and Meyer (1995) [13] | USA | Observational | Control HRT− (n = 75) Control HRT+ (n = 17) AD HRT− (n = 158) AD HRT+ (n = 10) | HRT use and probable AD diagnosis | NR | NR | NR | Postmenopausal | >60 | NR | Non-patients were more likely to have taken HRT than patients diagnosed with AD. This evidence suggests that HRT may be a prophylactic agent in reducing dementia of the AD type in postmenopausal women. |
| Tang et al. (1996) [14] | USA | Observational | Control HRT− (119) Control HRT+ (29) AD HRT− (86) AD HRT+ (7) | HRT use and AD diagnosis | CEE Other | NR | NR | Postmenopausal | Mean 74.2 ± 7 | ≤1 >1 | The annual incidence of AD was lower among users of HRT. Long-term HRT use over 1 year provided the lowest risk for AD. |
| Kawas et al. (1997) [15] | USA | Observational | Control HRT− (810) Control HRT+ (147) AD HRT− (158) AD HRT+ (9) | HRT use and AD diagnosis | NR | NR | Oral TD | Postmenopausal | 28–94 Mean 61.5 | <0–5 years 5–10 years >10 years | Fewer diagnoses of AD were made among women taking HRT. HRT showed a protective influence against AD, with no effect associated with the duration of HRT use. |
| Baldereschi et al. (1998) [16] | ITA | Observational | Control HRT− (217) Control HRT+ (221) AD HRT− (25) AD HRT+ (9) | HRT use AD diagnosis Cognitive tests: MMSE, CAMDEX, Pfeffer Functional Activities Questionnaire, HDRS. | NR | NR | NR | Postmenopausal | 65–84 | NR | There was a decreased risk of AD among women taking HRT. The age of disease onset did not differ between groups. Women taking HRT had better scores on the MMSE test. |
| Slooter et al. (1999) [17] | NLD | Observational | Control HRT− (1293) Control HRT+ (183) AD HRT− (89) AD HRT+ (3) | HRT use and AD diagnosis | NR | NR | NR | Postmenopausal | <65 | NR | HRT use was negatively associated with the development of early onset AD. |
| Waring et al. (1999) [18] | USA | Observational | Control HRT− (95) Control HRT+ (24) AD HRT− (98) AD HRT+ (11) | HRT use and AD diagnosis | NR | NR | Oral Topical | Postmenopausal | 57–96 | <6 months >6 months | HRT use was more common among non-patients than AD patients. Increased duration of HRT use positively correlated with decreased AD risk. |
| Lindsay et al. (2002) [19] | CAN | Observational | Control HRT− (175) Control HRT+ (47) AD HRT− (189) AD HRT+ (33) | HRT use and AD diagnosis | NR | NR | NR | Postmenopausal | 65+ | NR | HRT was not a protective or adverse factor for the risk of AD. |
| Zandi et al. (2002) [20] | USA | Observational | Control HRT− (1837) Control HRT+ (125) AD HRT− (106) AD HRT+ (4) | HRT use and AD diagnosis | NR | NR | NR | Postmenopausal | Mean 74.5 | <3 years 3–10 years >10 years | Previous HRT use was associated with a reduced risk of AD. This benefit was not observed if HRT was used for a duration of more than 10 years. |
| Yoo et al. (2020) [25] | KOR | Observational | HRT− (3,641,065) HRT+ (679,456) AD HRT− (189,459) AD HRT+ (14,151) | Dementia diagnosis AD diagnosis Cognitive tests: MMSE | NR | NR | NR | Postmenopausal | Mean 61.2 ± 8.6 | Mean 5.74 years | The risk of dementia increased in women who reached menarcheal age at an older age, and decreased for women who reached menopausal age later. The use of HRT reduced the risk of dementia. The findings from this study demonstrate that female reproductive factors are risk factors for dementia incidence with higher risk associated with shorter lifetime endogenous estrogen exposure. |
| Shao et al. (2012) [26] | USA | Observational | Control HRT− (574) Control HRT+ (1081) AD HRT− (89) AD HRT+ (87) | HRT use AD diagnosis | Unopposed Opposed Unknown | NR | Oral TD | Postmenopausal | 65+ | <3 years 3–10 years ≥10 years | Women who began HRT within 5 years of menopause showed a decreased risk of AD. This risk was shown to decrease further if HRT was used for 10 years or more. This effect was not seen in women who initiated HRT 5 years or more after menopause. Additionally, unopposed HRT use, but not opposed HRT use, was associated with a decreased AD risk. |
| Imtiaz et al. (2017) [27] | FIN | Observational | Whole cohort (8938) | AD diagnosis | Unopposed Opposed | NR | Oral TD | Postmenopausal | 47–56 | <1 year 1–3 years 3–5 years 5–10 years >10 years Follow up: 20 years | There was no association found between HRT use and AD risk. In long-term users of unopposed HRT, self-reported data showed a decreased risk of AD although there was no evidence to suggest a protective association. |
| Imtiaz et al. (2017) Tuppurainen [27] | FIN | Observational | Whole cohort (8195) | AD diagnosis | Unopposed Opposed | NR | Oral TD | Premenopausal Menopausal Postmenopausal | 47–56 | <1 year 1–3 years 3–5 years 5–10 years >10 years Follow up: 20 years | A protective association was found between postmenopausal HRT use and AD. |
| Imtiaz et al. (2017) [27] | FIN | Observational | HRT− (488) HRT+ (243) | MCI diagnosis Dementia diagnosis Cognitive tests: MMSE. Immediate word recall, Stroop Test, CF Test, Bimanual Purdue Pegboard Test, LD substitution test. | Unopposed Opposed | NR | NR | Postmenopausal | 65–79 | <5 years <5 years Follow up: 8 years | Overall, no strong protective effect of HRT on cognition was found. Long-term HRT users had improved global cognition in some domains. |
| Khoo et al. (2010) [28] | AUS | Observational | HRT− (213) Early HRT+ (158) Late HRT+ (39) | Cognitive test: MMSE, NART, WMS-3, General Memory Index, Working Memory Index. | Unopposed Opposed | NR | NR | Postmenopausal | 40–80 | >12 months Follow up: 5 years | Early start use of unopposed HRT was associated with a reduced risk of global cognitive decline, while opposed HRT showed increased risk of memory decline. There were no major effects on subgroup regarding type or timing of HRT on cognitive function. This study suggest that cognitive effects of hormone therapies may be mixed depending on cognitive domain, timing of use and preparation of HRT. |
| Henderson et al. (2005) [29] | USA | Observational | Control HRT− (353) Control HRT+ (192) AD HRT− (339) AD HRT+ (87) | HRT use AD diagnosis | NR | NR | NR | Postmenopausal | 65+ | >6 months | The findings of this study support evidence of a critical window of initiation for HRT use and AD risk. Association of HT use and AD risk was found to be dependent on timing of use with a significant protective association found only in the youngest age tertile. |
| Greendale et al. (2009) [30] | USA | Observational | Control HRT− (1887) Pre/Peri HRT+ (380) Post HRT+ (95) | Cognitive tests: SDMT, EBMT, DSB. | NR | NR | NR | Peri-menopausal Post-menopausal | 42–59 Mean 45.9 | 2- 3.2 years prior to MP Follow up: 4 years | HRT initiated pre-menopause was shown to have a beneficial effect on cognitive performance, while initiation post-menopause was detrimental. There was no difference in performance scores between postmenopausal non-HRT users and that of premenopausal women. |
| MacLennan et al. (2006) [31] | AUD | Observational | Never Users (194) Current HRT Users (70) Past HRT Users (140) | Cognitive Tests: MMSE, CERAD, VFT (FAS), BNT, TMTA and TMTB. | Unopposed Opposed | NR | NR | Postmenopausal | 60+ | <1 year 1–5 years <6–11 years >11 years | Early initiators of HRT performed better on cognitive tests than never users. Late initiators performed worse than never users on the MMSE but not the FAS test. Early initiation of HRT showed beneficial effects, while later initiation may have a detrimental effect on cognitive performance. |
| Slooter et al. (1999) [17] | NLD | Observational | Control HRT+ (24) Control HRT− (95) AD HRT+ (11) AD HRT− (98) | AD diagnosis | NR | NR | NR | Postmenopausal | AD onset <65 | NR | An inverse association was found between early HRT use and early onset AD, where HRT was beneficial in risk reduction. |
| O’Hara et al. (2005) [32] | USA | Observational | HRT+ (37) HRT− (32) | Cognitive tests: MMSE, Revised Benton Visual Retention test, WMT, symbol digit modalities test, list learning measure of delayed recall, GDS | Estradiol Valerate ± Norethisterone | 0.625 ± 2.5 | Oral | Postmenopausal | 60–93 Mean 72.74 ± 7.58 | 5 years | Findings from this study showed no effect on HRT on any measures of cognition. |
| Ryan et al. (2009) [33] | FRA | Observational | Never Users (2169) Past HRT+ (487) Current HRT+ (474) | Dementia diagnosis Cognitive tests: MMSE, Issacs Set Test, Benton Visual Retention Test, immediate and delayed recall (word recall) Trail Making Test A and B. | 17-β Estradiol | NR | NR | Postmenopausal | Never 74.9 ± 5.3 Past 72.4 ± 4.7 Current 70.2 ± 3.3 | 4 years | Current HRT users had greater performance on verbal fluency, working memory and psychomotor speed compared to never users. The strength of association varied depending on the type of treatment with longer duration appearing to be more beneficial. However, initiation of HRT close to menopause was not associated with improved cognition. HRT did not significantly reduce the risk of dementia. |
| Rippon et al. (2006) [34] | USA | Observational | HRT+ (77) HRT− (289) AD HRT+ (19) AD HRT− (61) | AD diagnosis | NR | NR | NR | Postmenopausal | HRT+ 63.97 ± 9.98) HRT- 74.4 ± 13.4 | NR | In women with history of stroke, ERT was a deleterious effect monitor. |
| Savolainen-Peltonen et al. (2019) [35] | FIN | Observational | Control HRT− (59,175) Control HRT+ (25,564) AD HRT− (58,186) AD HRT+ (26,553) | HRT use AD diagnosis | Unopposed Opposed Tibolone | NR | Oral TD Vaginal | Postmenopausal | 50+ | <3 years 3–4 years 5–9 years ≥10 years | Long-term use (10+ years) of HRT was associated with an increased risk of AD, regardless of use of opposed or unopposed HRT. This increased risk remained apparent after 3–5 years of HRT use in older women. Users of vaginal estradiol did not show the same increased risk. |
| Matyi et al. (2019) [36] | USA | Observational | Whole cohort (2114) | Dementia screening Cognitive tests: 3 MS | Unopposed Opposed | NR | NR | Postmenopausal | 74.94 (mean age) | HRT duration was used as a variable | Longer duration of HRT exposure was associated with better cognitive health. Women who initiated HRT earlier showed higher cognitive scores than those who initiated later. |
| Kim et al. (2022) [25] | KOR | Observational | Dementa—(183,510) Dementia + (26078) | Dementia diagnosis (AD and vascular) | NR | NR | NR | Postmenopausal | Dementia—(61.54) Demenia + (70.46) | <2 years 2–5 years >5 years Unknown Follow up: 7.72 years | HRT use after menopause transition was associated with decreased risk of AD and VD in depressed female patients. Lifetime oral contraceptive use was also associated with lower AD risk. |
| Lokkegaard et al. (2022) [37] | NLD | Observational | Dementia + (2235) Dementia—(11028) Dementia twin + (204) Dementia twin—(204) | HRT initiation (age) Dementia diagnosis (age) | Unopposed Opposed Continuous Cyclic | NR | Oral TD | NR | 45+ (for first HRT use) | NR | HRT use was associated with increased dementia risk if used before 2003. |
| Fox et al. (2013) [38,39] | ENG | Observational | AD (38) Controls (51) | Endogenous estrogen exposure span Reproductive factors HRT use | NR | NR | NR | NR | 70 -100 | NR | HRT had no effect on AD risk. Increased total months of endogenous estrogen exposure did result in a negative associated risk of AD. |
| Colucci et al. (2006) [40] | ITA | Observational | AD HRT− (188) AD HRT+ (16) Control HRT− (167) Control HRT+ (34) | HRT use Reproductive factors | NR | NR | NR | NR | AD (75.3) Controls (74.3) | NR | The AD cohort had a higher number of pregnancies than the control group. Findings suggest that is it an increase in progesterone or estrogen levels and not a decrease that is associated with AD pathology. |
| Hesson et al. (2012) [41] | CAN | Observational | Whole cohort (50) | Cognitive tests: STW Test, GDS, WMS-III LM I and II, time-based memory tests, RAND health survey. Estrogen exposure HRT use | NR | NR | NR | NR | 69.3 (mean) | Initiated HRT within 5 years of menopause Current Former | Cumulative lifetime exposure to estrogen was associated with memory performance. |
| Espeland et al. (2004) [42] | USA | Clinical | Placebo− (1421) Placebo MPA− (2213) CEE+ (1387) CEE+ MPA+ (2131) | Cognitive test: 3 MSE | CEE ± MPA | 0.625 mg | Oral TD | Postmenopausal | 65–79 | Mean 5.4 years | HRT was shown to have adverse effects on cognition. In women with lower baseline cognitive scores this effect was stronger. |
| Schiff et al. (2005) [43] | UK | Clinical | 12 women (crossover design) | Cognitive test: simple and choice reaction time, immediate word recall, delayed word recall, digital vigilance, visual tracking, spatial working memory, word recognition, picture and face recognition, NART. | 17-β Estradiol | 50 ug | TD | Surgically menopausal | 62–89 | 8 weeks follow up: for 26 weeks | HRT therapy, when administered short term, did consistently improve the speed and accuracy of older women in cognitive tests, including reaction time and depression scores. These findings support an effect of estradiol on mood and cognitive function and not simply as a result of relief of climacteric symptoms. |
| Almeida et al. (2006) [44] | AUS | Clinical | HRT+ (58) Placebo (57) | Cognitive tests: CAMCOG, VFT, BD, CVLT-II | 17-β Estradiol | 2 | Oral | Postmenopausal | 70+ | 20 weeks | High-dose HRT when administered for a course of 20 weeks was not associated with significant changes in cognitive function, mood or quality of life. |
| Yaffe et al. (2006) [45] | USA | Clinical | HRT+ (208) Placebo (209) | Cognitive tests: MMSE, Episodic Memory, M-BNT, Verbal Fluency, Medical Outcomes Study Health survey (SF-36) | 17-β Estradiol | 0.014 | TD | Postmenopausal | 60–80 | 2 years | Ultra-low dose HRT treatment for a course of 2 years had no effect on change in cognitive function of health-related life quality. |
| Shaywhitz et al. (2003) [46] | USA | Clinical | 60 (Cross-over design) | Cognitive tests: Grey Oral reading test (3rd edition), WMS, sentence span) | CEE | 1.25 | Oral | Postmenopausal | 32.8–64.9 Mean 51.2 ± 5 | 3 weeks | Participants in the HRT-active treatment arm showed better performance on oral reading and verbal memory than the placebo group. HRT had greater beneficial effects on midlife postmenopausal women. |
| Espeland et al. (2013) [47] | USA | Clinical | HRT+ (609) Placebo (559) | Cognitive tests: TISC-m, E-BMT, OTMT, VF-A, WAIS-R | CEE ± MPA | 0.625 ± 2.5 | Oral | Postmenopausal | 50–55 | 7.2 years | HRT produced no overall sustained benefit or risk to cognitive function in any domain. HRT therapy may adversely affect verbal fluency among women with prior use of HRT. |
| Espeland et al. (2017) [48] | USA | Clinical | Younger placebo (635) Younger HRT+ (701) Older placebo (1478) Older HRT+ (1402) | Cognitive tests: TICS-m, E-BMT, OTMT, VF-A, DST | CEE ± MPA | 0.625 ± 2.5 | Oral | Postmenopausal | 50–54 65–79 | CEE + MPA mean 5.4 years CEE mean 7.1 years | HRT in younger women had no significant long-term effects on cognitive function or changes in cognitive function. In older women, HRT use was associated with long-term detrimental effects in global cognitive function, working memory and executive function. When HRT was initiated near the time of menopause, there was neither a benefit nor a detrimental effect on cognition. |
| Girard et al. (2017) [49] | FRA | Clinical | HRT+ (6) Placebo (6) | fMRI Cognitive tests: Task switching | 17-β Estradiol ± Progesterone | 2 100 | Postmenopausal | 48–55 | 56 days | HRT enhanced brain activation and recruitment of dorsolateral prefrontal cortex neurons during task switching, and this change was associated with the performance on the task. The results suggest that HRT, when taken early, may have a beneficial effect on cognitive control of prefrontal mechanisms. | |
| Dunkin et al. (2005a) [50] | USA | Clinical | HRT+ (8) Placebo (9) | Cognitive tests: Stroop Task, WAIS-R, DST, Grooved Pegboard Test, CVLT, WMS-R, WCST, Controlled Oral Word Association Test, Trail Making Test—B | 17-β Estradiol | 0.1 | TD | Postmenopausal | Control Mean: 57 ± 6.89 Depressed Mean: 54.10 ± 6.51 | 10 weeks | Reproductive events and levels of endogenous estrogens were found to be related to the clinical response to HRT. Overall, little beneficial effect of HRT was found although years after menopause were significantly related to change in executive functioning in the treatment group but not in the placebo. Women who were more recently menopausal demonstrated a greater positive change than older women. |
| Song et al. (2020) [51] | SGP | Clinical | MCI HRT− (6216) MCI HRT–(1273) HRT+ (674) HRT+ (59) | MCI diagnosis Cognitive tests: SM-MMSE | Unopposed Opposed | NR | NR | Postmenopausal | 45–74 Mean 53.4 ± 6.4 | NR | Users of HRT showed a decreased risk of mild cognitive impairment diagnosis compared to non-users of HRT. |
| Dunkin et al. (2005b) [52] | USA | Clinical | Control HRT+ (8) Control HRT− (9) Depressed HRT+ (10) Depressed HRT− (10) | Cognitive tests: Stroop Task A and B, WAIS-R, Grooved Pegboard Test, CVLT, WMS-R, WCST, Controlled Oral Word Association Test (FAS), Trail Making Test Part B, WAIS-R, AMNART, HAM-D | 17-β Estradiol | 0.1 | TD | Postmenopausal | Control: 57 ± 6.89 Depressed: 54.1 ± 6.51 | 10 weeks | Recently menopausal women demonstrated greater positive change than older women, especially in domains of executive functioning. This finding occurred in both depressed and control subject. |
| Resnick et al. (2009) [53] | USA | Clinical | HRT+ (257) HRT+ MPA+ (435) Placebo (263) Placebo MPA− (447) | MRI Cognitive tests: 3MSE | CEE ± MPA | 0.625 ± 2.5 | Oral | Postmenopausal | 71–89 | Mean 4 years | Opposed or unopposed HRT was associated with greater brain atrophy in women ages 65+. Greater adverse effects were observed in women who had lower cognitive baseline scores. |
| Tierney et al. (2009) [54] | CAN | Clinical | HRT+ (70) Placebo (72) | Cognitive tests: CVLT, Rey–Osterrieth Complex Figure Test, BNT, Trail Making Test Parts A and B | 17-β Estradiol + progesterone | 1 1 + 0.35 | Oral | Postmenopausal | 61–87 HRT+ Mean 75 ± 6.4 Placebo Mean 74.5 ± 7.4 | 2 years | Women in the treatment group who scored above average at baseline showed significantly less decline than the placebo group in short delay verbal recall. No treatment effect was found in women who scored below average on these measures. These findings support the hypothesis that HRT will have a greater beneficial effect in women with healthy neurons. |
| Kantarci et al. (2016) [55] | USA | Clinical | CEE+ (29) 17 B Estradiol+ (30) Placebo (36) | MRI Cognitive tests | 17-β Estradiol CEE Progesterone | 50 ug 0.45 200 | TD Oral | Postmenopausal | 52–54 | 4 years | Ventricular volumes increased without changes in cognitive performance in women who received CEE compared to placebo. Rates of ventricular expansion were not different between groups; however, only differences in the CEE group reached significance. |
| Shumaker et al.(2004) [56] | USA | Clinical | HRT+ (1464) HRT+ MPA+ (3693) Placebo (1483) Placebo MPA− (3786) | AD diagnosis MCI diagnosis Cognitive tests: 3MSE, CERAD | CEE ± MPA | 0.635 ± 2.5 | Oral | Postmenopausal | 65–79 | Opposed: 7 years Unopposed: 9 years | Unopposed HRT did not reduce the incidence of dementia or MCI. Pooling data from unopposed and opposed groups resulted in increased risk for both dementia and MCI. |
| Henderson et al. (2000) [29] | USA | Clinical | AD HRT+ (21) AD Placebo (21) | AD diagnosis Cognitive tests: ADAS-cog, CGIC, ADL/IADL, MADRS, WMS, BNT, TMT, logical and visual reproduction subsets, Token Test, GDS | CEE | 1.25 | Oral | Postmenopausal | HRT+ 77 ± 1.4 Placebo 78 ± 1 | 16 weeks | Short-term HRT use did not improve global, cognitive, or functional performance among women with mild to moderate AD. |
| Mulnard et al. (2000) [57] | USA | Clinical | Low Dose HRT+ (42) High Dose HRT+ (39) Placebo (39) | HRT use and AD diagnosis Cognitive tests: CGIC, MMSE, CDRS, HDRS, MAACL, ADAS-cog, TMT, EFRT, BDRS, New Dot, Letter Cancellation, Digit Symbol, Category and Letter Fluency, Grooved Pegboard, Finger Tapping, Dependency Scale. | CEE | Low (0.625) High (1.25) | Oral | Surgically Menopausal | >60 | 48 weeks | HRT did not slow disease progression or improve global, cognitive, or functional outcomes regardless of dose. HRT group showed some benefit at 8 weeks compared to placebo, but this effect changed to become detrimental following 12 months. The CEE treatment group showed benefit at 8 weeks, became worse than placebo following 12 months of CEE. |
| Wang et al. (2000) [58] | TWN | Clinical | HRT+ (24) Placebo (39) | AD diagnosis Cognitive tests: CASI, MMSE, CIBIC-plus, BEHAVE-AD, HDRS, HARS, rCBF | CEE | 1.25 | Oral | Postmenopausal | >/=60 | 12 weeks | HRT did not cause any effect on tested domains in postmenopausal women with AD. |
| Valen-Sendstad et al. (2010) [59] | NOR | Clinical | HRT+ (33) Placebo (32) | AD Diagnosis Cognitive tests: WLM, CERAD-modified MMSE, HDRS, TMT, CERAD-modified BNT, CERAD depression screen, WAIS, Constructional Praxis, Global Deterioration Scale, Barthel Index | 17-β Estradiol Norethisterone | 1 0.5 | Oral | Postmenopausal | 65–89 | 48 weeks | HRT improved mood, depressive symptom scores, and appeared to slow the progression of cognitive decline. This effect was stronger in those without the APOE allele. |
| Asthana et al. (1999) [60] | USA | Clinical | AD HRT+ (6) AD Placebo (6) | HRT use and AD diagnosis Cognitive tests: BSRT, SCWIT, TMT, MMSE, BMICT, BPRS, paragraph recall, verbal reproduction and fluency, Token Test | 17-β Estradiol | 0.05 | TD | Postmenopausal | HRT+ 79.5 ± 7.6 Placebo 77.6 ± 6.6 | 8 weeks | The HRT group showed improvement in attentional and verbal memory domains in women with AD. These improvements were not stable and did not remain after HRT discontinuation. |
| Asthana et al. (2001) [61] | USA | Clinical | HRT+ (10) Placebo (10) | AD diagnosis Cognitive tests: MMSE, BMICT, BPRS, CIBIC, IADL, PSMS, SCWIT, TMT, BSRT, OMDR, BNT, RCFT, Story recall, Visual Paired- Associates, Treisman VS | 17-β Estradiol | 0.10 | TD | Postmenopausal | HRT+ 79.0 ± 9.7 Placebo 80.2 ± 6.7 | 8 weeks | HRT improved attention, verbal memory and visual memory in postmenopausal women with AD. |
| Wharton et al. (2011) [62] | USA | Clinical | Low Dose HRT+(10) Low Dose HRT+(8) High Dose HRT+ (8) High Dose HRT (9) Placebo (8) | AD Diagnosis Cognitive tests: BNT, POMS, VPA, TMT, SCWIT, List Learning, paragraph recall, Complex Figure and Figural Memory Tests. | 17-β Estradiol ± MPA | 0.05 ± 2.5 | TD | Postmenopausal | 55–85 | 48 Weeks | HRT administration improved semantic and episodic visual memory domains in postmenopausal women with AD. Changes to episodic visual memory were more pronounced in women receiving opposed HRT. |
| Levine and Battista (2004) [63] | USA | Clinical | HRT− (99) HRT+ (20) | AD diagnosis Cognitive tests: BDRS, MMSE, NCSE, CSDD, NRS, GDS | NR | NR | NR | Postmenopausal | HRT- 78.78 ± 6.38 HRT+ 77.48 ± 5.08 | NR | No differences in cognition, mood and age of onset AD between groups. Women in the HRT group had greater behavioral deficits in self-care. |
| Wolf et al. (1999) [64] | DEU | Clinical | HRT+ (21) Placebo (17) | Cognitive tests: VFT, city maps task, Paired Associated Test, Stoop Test, mental rotation, mood assessment | 17-β Estradiol | 0.1 | Percutaneously | Perimenopausal and Postmenopausal | HRT+ 69.5 ± 1.4 Placebo 67.8 ± 1.2 | 2 weeks | The treatment group analysis showed that women who received HRT and reached higher estradiol levels performed significantly better after treatment on delayed recall of paired associate tests. As these effects were seen after 2 weeks in women who were ~17 years postmenopausal, the findings of this study suggest that the brain remains sensitive to HRT where mediated changes occur rapidly. |
| LeBlanc et al. (2007) [22] | USA | Clinical | HRT+ (14) Placebo (18) | Cognitive tests: Oregon Sleep Health Diary, profile of mood states, paragraph recall and Verbal Paired Associates, facial emotion, abstract painting recall, letter category’s | 17-β Estradiol | 2 | Oral | Postmenopausal | HRT+ 53.26 ± 0.64 Placebo 52.08 ± 0.64 | 8 weeks | Women receiving HRT did not have any improvements in cognitive performance or mood compared with those receiving placebo. |
| Maki et al. (2007) [65] | USA | Clinical | HRT+ (78) Placebo (80) | Cognitive tests: CVLT, (MFQ), BTA, Benton Visual Retention Test, Educational Testing Service Card rotation Test, Letter Fluency Test, WMS-R/LM-R, PANAS, The Greene Climacteric Scale, The Utian Quality of Life Scale, McCoy Female Sexuality Scale Questionnaire, PSQI. | CEE + MPA | 0.625 + 2.5 | Oral | Postmenopausal | 45–55 | 4 months | There were no differences between the HRT group and placebo on any measures of cognition or quality of life. Modest negative effects on short-term and long-term verbal memory were observed but did not reach significance. |
| Alhola et al. (2010) [66] | FIN | Clinical | PreMP HRT+ (8) PreMP Placebo (8) Post PM HRT+ (7) Placebo (9) | Cognitive tests: Simple Reaction Time, Two Choice Reaction Time, 10 Choice Reaction Time, Subtraction task, Verification task, vigilance task, WAIS-R, PASAT, Bourdon–Wiersma Test, modified Rey Auditory Verbal Learning Test, Benton Visual Retention test. | Estradiol Valerate + Norethisterone | 2 mg + 1 (0.7) | Oral | Premenopausal and Postmenopausal | PreMP 47.9 ± 1.7 PostMP 62.9 ± 2.9 | 6 months | In comparison to baseline, premenopausal women improved in tests of reaction time with the use of HRT, while the placebo group performed better in tests of attention. In postmenopausal women, HRT use was associated with improved performance in verbal episodic memory and minor declines in auditory attention. |
| Henderson et al. (2016) [67] | USA | Clinical | Early HRT+ (271) Late HRT+ (372) | Cognitive tests: SDMT, Complex Scanning and Visual Tracking, Trail Making Test (B), Shipley Institute of Living Scale, Abstraction Scale, Concept Formation, Letter Number Sequencing, Block Design, Animal Naming, WTAR, BNT, CVLT-s, EBMT | CEE ± MPA | 1 ± 45 | Oral Vaginal(Gel) | Postmenopausal | Early 55.4 ± 4.1 Late 65.4 ± 6.0 | 57 months | Results from the early and late groups did not differ significantly. Progesterone levels were significantly and positively associated with verbal memory and global cognition in early HRT initiation group. No evidence was found to support temporal proximity to menopause as a modifier of cognitive effect. |
| Perfanco et al. (2007) [68] | USA | Clinical | HRT+ (32) Placebo (25) | Cognitive tests: COWAT, Trail Making Test, WCST, BNT, the Digital Symbol Modalities Test, the Complex Figure Test, the Fold Object Memory Test, WMSIII, GDS, Beck Anxiety Inventory. | 17-β Estradiol ± cyclic micronized progesterone | 0.25 ± 100 | NR | Postmenopausal | HRT+ 76 ± 6 Placebo 75 ± 4 | 3 years | No differences were found between groups on any neurocognitive or depression scores. Additionally, no differences were found when stratifying according to age. |
| Joffe et al. (2006) [69] | USA | Clinical | HRT+ (26) Placebo (26) | fMRI Cognitive tests: CVLT, WMS-R, Rey–Osterreith Complex Figure Test, PSQI, Beck Depression Inventory. | Micronized 17-β Estradiol ± micronized progesterone | 0.05 | TD | Perimenopausal and Postmenopausal | 40–60 HRT+ 50.8 ± 3.4 Placebo 51.3 ± 4.2 | 12 weeks | HRT selectively reduced errors of preservation during verbal recall compared to placebo without influencing any other cognitive processes. Women with higher baseline scores of hot-flash severity demonstrated a greater cognitive benefit of HRT. Measures of fMRI BOLD activation during test of verbal and spatial working memory showed significant increases in frontal activity with HRT. |
| Kang and Grodstein (2012) [70] | USA | Clinical | Non HRT Users (5075) Past HRT users (5765) Current HRT unopposed Users (4188) Current HRT opposed users (1486) | Cognitive tests: TICS, EBMT-a, MMSE, Category Fluency, delayed recall of word list, digit span backwards | NR | NR | NR | Postmenopausal | 70–81 | 26 years | In comparison to never users, past or current users of HRT showed worse rates of decline in the TICS. No protective association was found with early timing of initiation. |
| Shaywitz et al. (1999) [71] | USA | Clinical | 46 (Crossover design) | fMRI Cognitive tests: verbal and non-verbal memory tasks | CEE | 1.25 | Oral | Postmenopausal | 33–61 Mean 50.8 ± 4.7 | 21 days | HRT in a therapeutic dosage alters brain activation patterns in specific brain regions. However, HRT did not affect the performance score of verbal or non-verbal memory tasks. |
| Sherwin (1988) [72] | USA | Clinical | Climacteron (10) Estradiol Valerate (10) Testosterone enanthate (10) Placebo (10) Hysterectomy control (10) | Cognitive tests: Digit Span, WMS, Clerical Speed and Accuracy (Differential Aptitude Test), paragraph recall, Abstract Reasoning subset of DAT. | Climacteron (Testosterone, Estradiol dienanthate, Estradiol benzoate) Estradiol Valerate Testosterone enanthate | (150,7.5,1.0) 10 200 | Intramuscular | Surgically menopausal | 36.6–45.4 years | 6 months | Test scores on short-term, long-term memory, or logical reasoning were not different during post-op treatment phase of HRT to their preoperative performance. Oophorectomized women who received placebo had lower scores of cognitive functioning postoperatively with lower concentrations of plasma estradiol and testosterone. Women who underwent hysterectomy but maintained their ovaries showed stable cognitive performance and in circulatory sex steroid concentrations. These results suggest that the drastic change in endocrine milieu following surgical menopause may have a direct, albeit modest, effect on aspects of cognitive function. |
| Phillips and Sherwin (1992) [73] | USA | Clinical | HRT+ (10) Placebo (9) | Cognitive tests: WMS, Menopausal Index, Multiple Affect Check List | Estradiol Valerate | 10 mg/month | Intramuscular Injection | Surgically menopausal | Mean 48.2 ± 4.7 | 3 months | Scores for immediate and delayed recall paired associates stayed at same level in HRT-treated women, whereas they decreased significantly in pre- to post-op in the placebo group. |
| Wroolie et al. (2015) [74] | USA | Clinical | Continued HRT (30) Discontinued HRT (24) | Cognitive tests: WAIS, Auditory Consonant Trigrams Total, BSRT first trial learning; WMS-III, Color Trails 1; DKEFS, BSRT, Benton Visual Retention Test, Rey–Osterrieth Complex Figure Test, DKEFS, Memory Functioning Questionnaire | 17-β Estradiol CEE | NR | NR | Postmenopausal | 49–69 Discontinued 57.9 ± 5.01 Continued 58.2 ± 4.37 | >3 years | Among women who were selected on a basis for a higher risk of AD and current HRT use, the continuation of HRT was associated with increased performance on cognitive domains, including verbal memory, combined attention, working memory and processing speed measures at the end of the follow-up period. All female patients diagnosed with AD declined in verbal memory; however, women who continued HRT declined less than women who discontinued HRT. |
| Silverman et al. (2011) [75] | USA | Clinical | 17B Estradiol (35) CEE (18) | FDG Pet scan Cognitive tests: Auditory Consonant Trigrams, Benton Visual Retention Test, BNT, Color Trail Making Test, DKEFS, Rey–Osterrieth Complex Figure Test, WAISIII, WMS 3, Memory Function Questionnaire. | CEE 17-β Estradiol (Unopposed and Opposed) | NR | NR | Postmenopausal | 50–65 | 2 years | Women taking 17-β estradiol performed better in verbal memory tasks than those taking CEE, where verbal memory performance positively correlated with metabolism in Wernicke’s and auditory association areas. Women taking opposed HRT had lower metabolism than women taking unopposed HRT in some brain regions. |
| Shumaker et al. (2003) [24] | USA | Clinical | HRT+ (2229) Placebo (2303) | MCI and AD diagnosis Cognitive tests: 3MSE, CERAD, BNT, word list memory task, contractional praxis, executive function. | CEE + MPA | 0.625 + 2.5 | Oral | Postmenopausal | 65+ | Mean 4.05 years | Opposed HRT increased the risk for probable dementia. |
| Grady et al. (2002) [76] | USA | Clinical | HRT+ (517) Placebo (546) | Cognitive tests: 3MSE, VFT, BNT, Word List Memory, Word List Recall, + Trials B) | CEE + MPA | 0.625 + 2.5 | Oral | Postmenopausal | Mean 71 ± 6 | 4 years | There were no differences in cognitive function performance between groups when adjusted for age in women with a history of coronary disease. However, women assigned to HRT scored worse on the VFT than women assigned to placebo. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mills, Z.B.; Faull, R.L.M.; Kwakowsky, A. Is Hormone Replacement Therapy a Risk Factor or a Therapeutic Option for Alzheimer’s Disease? Int. J. Mol. Sci. 2023, 24, 3205. https://doi.org/10.3390/ijms24043205
Mills ZB, Faull RLM, Kwakowsky A. Is Hormone Replacement Therapy a Risk Factor or a Therapeutic Option for Alzheimer’s Disease? International Journal of Molecular Sciences. 2023; 24(4):3205. https://doi.org/10.3390/ijms24043205
Chicago/Turabian StyleMills, Zoe B., Richard L. M. Faull, and Andrea Kwakowsky. 2023. "Is Hormone Replacement Therapy a Risk Factor or a Therapeutic Option for Alzheimer’s Disease?" International Journal of Molecular Sciences 24, no. 4: 3205. https://doi.org/10.3390/ijms24043205
APA StyleMills, Z. B., Faull, R. L. M., & Kwakowsky, A. (2023). Is Hormone Replacement Therapy a Risk Factor or a Therapeutic Option for Alzheimer’s Disease? International Journal of Molecular Sciences, 24(4), 3205. https://doi.org/10.3390/ijms24043205




