Effects of Cannabidiol on Innate Immunity: Experimental Evidence and Clinical Relevance
Abstract
1. Introduction
1.1. CBD Pharmacodynamics
1.2. CBD Pharmacokinetics
1.3. CBD Pharmacogenetics
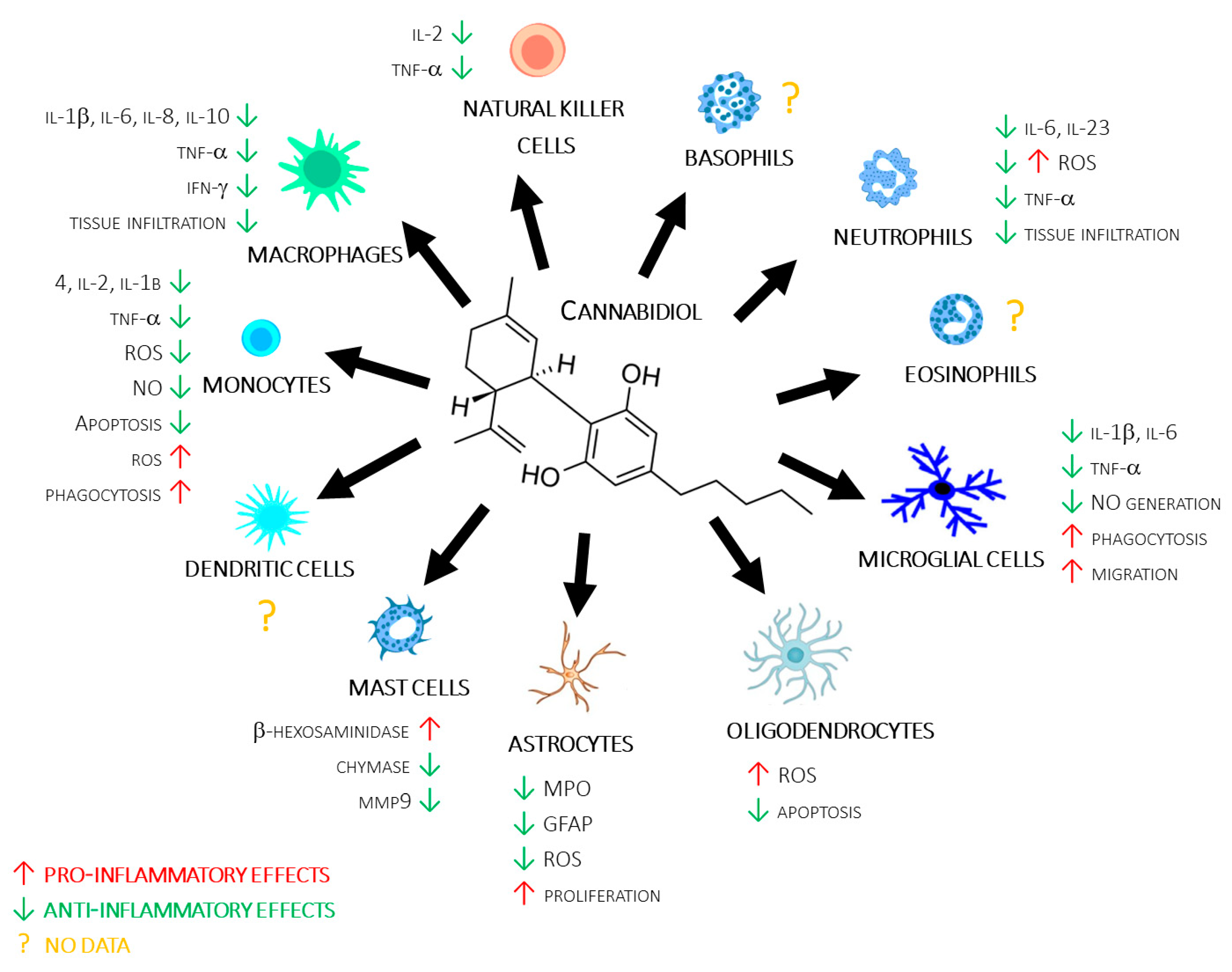
2. Effects of CBD on Immune Systems
2.1. CBD’s Action on the Complement System and Antibacterial Peptides
2.2. Monocytes/Macrophages
2.3. Glial Cells (Astrocytes, Microglia, and Oligodendrocytes)
2.4. Mast Cells
2.5. NK Cells
2.6. Dendritic Cells
2.7. Eosinophils and Basophils
2.8. Polymorphonuclear Neutrophils (PMNs)
3. CDB Modulation of Innate Immunity: Possible Implications for Disease Treatment
3.1. Atherosclerosis
3.2. Neurodegenerative Diseases
3.2.1. Multiple Sclerosis
3.2.2. Parkinson’s Disease
3.2.3. Alzheimer’s Disease
3.3. Neuropsychiatric Diseases
3.4. Autoimmune/Inflammatory Diseases
3.5. Other Therapeutic Possibilities
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Good, P.; Haywood, A.; Gogna, G.; Martin, J.; Yates, P.; Greer, R.; Hardy, J. Oral Medicinal Cannabinoids to Relieve Symptom Burden in the Palliative Care of Patients with Advanced Cancer: A Double-Blind, Placebo Controlled, Randomised Clinical Trial of Efficacy and Safety of Cannabidiol (CBD). BMC Palliat. Care 2019, 18, 110. [Google Scholar] [CrossRef] [PubMed]
- van de Donk, T.; Niesters, M.; Kowal, M.A.; Olofsen, E.; Dahan, A.; van Velzen, M. An Experimental Randomized Study on the Analgesic Effects of Pharmaceutical-Grade Cannabis in Chronic Pain Patients with Fibromyalgia. Pain 2019, 160, 860–869. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Morasco, B.J.; Nugent, S.; Ayers, C.; O’Neil, M.E.; Freeman, M.; Paynter, R.; Kansagara, D. Pharmacotherapy for the Treatment of Cannabis Use Disorder: A Systematic Review; VA Evidence-based Synthesis Program Reports; Department of Veterans Affairs (US): Washington, DC, USA, 2019. [Google Scholar]
- Blessing, E.M.; Steenkamp, M.M.; Manzanares, J.; Marmar, C.R. Cannabidiol as a Potential Treatment for Anxiety Disorders. Neurotherapeutics 2015, 12, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Alves, P.; Amaral, C.; Teixeira, N.; Correia-da-Silva, G. Cannabis Sativa: Much More beyond Δ9-Tetrahydrocannabinol. Pharmacol. Res. 2020, 157, 104822. [Google Scholar] [CrossRef]
- Fraguas-Sánchez, A.I.; Torres-Suárez, A.I. Medical Use of Cannabinoids. Drugs 2018, 78, 1665–1703. [Google Scholar] [CrossRef]
- Babalonis, S.; Haney, M.; Malcolm, R.J.; Lofwall, M.R.; Votaw, V.R.; Sparenborg, S.; Walsh, S.L. Oral Cannabidiol Does Not Produce a Signal for Abuse Liability in Frequent Marijuana Smokers. Drug Alcohol Depend. 2017, 172, 9–13. [Google Scholar] [CrossRef]
- Schoedel, K.A.; Szeto, I.; Setnik, B.; Sellers, E.M.; Levy-Cooperman, N.; Mills, C.; Etges, T.; Sommerville, K. Abuse Potential Assessment of Cannabidiol (CBD) in Recreational Polydrug Users: A Randomized, Double-Blind, Controlled Trial. Epilepsy Behav. 2018, 88, 162–171. [Google Scholar] [CrossRef]
- Lachenmeier, D.W.; Habel, S.; Fischer, B.; Herbi, F.; Zerbe, Y.; Bock, V.; Rajcic de Rezende, T.; Walch, S.G.; Sproll, C. Are Adverse Effects of Cannabidiol (CBD) Products Caused by Tetrahydrocannabinol (THC) Contamination? F1000Research 2021, 8, 1394. [Google Scholar] [CrossRef]
- Bonaccorso, S.; Ricciardi, A.; Zangani, C.; Chiappini, S.; Schifano, F. Cannabidiol (CBD) Use in Psychiatric Disorders: A Systematic Review. NeuroToxicology 2019, 74, 282–298. [Google Scholar] [CrossRef]
- White, C.M. A Review of Human Studies Assessing Cannabidiol’s (CBD) Therapeutic Actions and Potential. J. Clin. Pharmacol. 2019, 59, 923–934. [Google Scholar] [CrossRef]
- Davis, M.P. Oral Nabilone Capsules in the Treatment of Chemotherapy-Induced Nausea and Vomiting and Pain. Expert Opin. Investig. Drugs 2008, 17, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Furgiuele, A.; Cosentino, M.; Ferrari, M.; Marino, F. Immunomodulatory Potential of Cannabidiol in Multiple Sclerosis: A Systematic Review. J. Neuroimmune Pharmacol. 2021, 16, 251–269. [Google Scholar] [CrossRef] [PubMed]
- Zajicek, J.; Fox, P.; Sanders, H.; Wright, D.; Vickery, J.; Nunn, A.; Thompson, A.; UK MS Research Group. Cannabinoids for Treatment of Spasticity and Other Symptoms Related to Multiple Sclerosis (CAMS Study): Multicentre Randomised Placebo-Controlled Trial. Lancet 2003, 362, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Urits, I.; Borchart, M.; Hasegawa, M.; Kochanski, J.; Orhurhu, V.; Viswanath, O. An Update of Current Cannabis-Based Pharmaceuticals in Pain Medicine. Pain Ther. 2019, 8, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Mlost, J.; Bryk, M.; Starowicz, K. Cannabidiol for Pain Treatment: Focus on Pharmacology and Mechanism of Action. Int. J. Mol. Sci. 2020, 21, 8870. [Google Scholar] [CrossRef]
- Chen, J.W.; Borgelt, L.M.; Blackmer, A.B. Cannabidiol: A New Hope for Patients With Dravet or Lennox-Gastaut Syndromes. Ann. Pharmacother. 2019, 53, 603–611. [Google Scholar] [CrossRef]
- Zuardi, A.W.; Crippa, J.a.S.; Hallak, J.E.C.; Pinto, J.P.; Chagas, M.H.N.; Rodrigues, G.G.R.; Dursun, S.M.; Tumas, V. Cannabidiol for the Treatment of Psychosis in Parkinson’s Disease. J. Psychopharmacol. 2009, 23, 979–983. [Google Scholar] [CrossRef]
- Jean-Gilles, L.; Gran, B.; Constantinescu, C.S. Interaction between Cytokines, Cannabinoids and the Nervous System. Immunobiology 2010, 215, 606–610. [Google Scholar] [CrossRef]
- Davis, M.P. Cannabinoids for Symptom Management and Cancer Therapy: The Evidence. J. Natl. Compr. Cancer Netw. 2016, 14, 915–922. [Google Scholar] [CrossRef]
- Pellati, F.; Borgonetti, V.; Brighenti, V.; Biagi, M.; Benvenuti, S.; Corsi, L. Cannabis Sativa L. and Nonpsychoactive Cannabinoids: Their Chemistry and Role against Oxidative Stress, Inflammation, and Cancer. BioMed Res. Int. 2018, 2018, e1691428. [Google Scholar] [CrossRef]
- Castillo, A.; Tolón, M.R.; Fernández-Ruiz, J.; Romero, J.; Martinez-Orgado, J. The Neuroprotective Effect of Cannabidiol in an in Vitro Model of Newborn Hypoxic–Ischemic Brain Damage in Mice Is Mediated by CB2 and Adenosine Receptors. Neurobiol. Dis. 2010, 37, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Jastrząb, A.; Gęgotek, A.; Skrzydlewska, E. Cannabidiol Regulates the Expression of Keratinocyte Proteins Involved in the Inflammation Process through Transcriptional Regulation. Cells 2019, 8, 827. [Google Scholar] [CrossRef] [PubMed]
- Anikwue, R.; Huffman, J.W.; Martin, Z.L.; Welch, S.P. Decrease in Efficacy and Potency of Nonsteroidal Anti-Inflammatory Drugs by Chronic Delta(9)-Tetrahydrocannabinol Administration. J. Pharmacol. Exp. Ther. 2002, 303, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Wheal, A.J.; Cipriano, M.; Fowler, C.J.; Randall, M.D.; O’Sullivan, S.E. Cannabidiol Improves Vasorelaxation in Zucker Diabetic Fatty Rats through Cyclooxygenase Activation. J. Pharmacol. Exp. Ther. 2014, 351, 457–466. [Google Scholar] [CrossRef]
- Cosentino, M.; Legnaro, M.; Luini, A.; Ferrari, M.; Sodergren, M.; Pacchetti, B.; Marino, F. Effect of Cannabidiol on Cyclooxygenase Type 1 and 2 Expression and Function in Human Neutrophils. Cannabis Cannabinoid Res. 2022. [Google Scholar] [CrossRef]
- Pertwee, R.G. Sites and Mechanisms of Action. In Cannabis and Cannabinoids: Pharmacology Toxicology and Therapeutic Potential; Haworth Press: New York, NY, USA, 2002; pp. 73–87. [Google Scholar]
- Mackie, K. Distribution of Cannabinoid Receptors in the Central and Peripheral Nervous System. Handb. Exp. Pharmacol. 2005, 299–325. [Google Scholar] [CrossRef]
- Galiègue, S.; Mary, S.; Marchand, J.; Dussossoy, D.; Carrière, D.; Carayon, P.; Bouaboula, M.; Shire, D.; Le Fur, G.; Casellas, P. Expression of Central and Peripheral Cannabinoid Receptors in Human Immune Tissues and Leukocyte Subpopulations. Eur. J. Biochem. 1995, 232, 54–61. [Google Scholar] [CrossRef]
- Derocq, J.-M.; Jbilo, O.; Bouaboula, M.; Ségui, M.; Clère, C.; Casellas, P. Genomic and Functional Changes Induced by the Activation of the Peripheral Cannabinoid Receptor CB2 in the Promyelocytic Cells HL-60: POSSIBLE INVOLVEMENT OF THE CB2 RECEPTOR IN CELL DIFFERENTIATION*. J. Biol. Chem. 2000, 275, 15621–15628. [Google Scholar] [CrossRef]
- Valencia-Sánchez, S.; Nava-Castro, K.E.; Palacios-Arreola, M.I.; Prospéro-García, O.; Morales-Montor, J.; Drucker-Colín, R. Chronic Exercise Modulates the Cellular Immunity and Its Cannabinoid Receptors Expression. PLoS ONE 2019, 14, e0220542. [Google Scholar] [CrossRef]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Front. Mol. Neurosci. 2018, 11, 487. [Google Scholar] [CrossRef] [PubMed]
- Omari, S.A.; Adams, M.J.; Geraghty, D.P. Chapter Six—TRPV1 Channels in Immune Cells and Hematological Malignancies. In Advances in Pharmacology; Geraghty, D.P., Rash, L.D., Eds.; Ion Channels DownUnder; Academic Press: Cambridge, MA, USA, 2017; Volume 79, pp. 173–198. [Google Scholar]
- Russo, E.B.; Burnett, A.; Hall, B.; Parker, K.K. Agonistic Properties of Cannabidiol at 5-HT1a Receptors. Neurochem. Res. 2005, 30, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.; Ferraz-de-Paula, V.; Pinheiro, M.L.; Vitoretti, L.B.; Mariano-Souza, D.P.; Quinteiro-Filho, W.M.; Akamine, A.T.; Almeida, V.I.; Quevedo, J.; Dal-Pizzol, F.; et al. Cannabidiol, a Non-Psychotropic Plant-Derived Cannabinoid, Decreases Inflammation in a Murine Model of Acute Lung Injury: Role for the Adenosine A(2A) Receptor. Eur. J. Pharmacol. 2012, 678, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Kathmann, M.; Flau, K.; Redmer, A.; Tränkle, C.; Schlicker, E. Cannabidiol Is an Allosteric Modulator at Mu- and Delta-Opioid Receptors. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2006, 372, 354–361. [Google Scholar] [CrossRef]
- Pertwee, R.G. The Diverse CB1 and CB2 Receptor Pharmacology of Three Plant Cannabinoids: Delta9-Tetrahydrocannabinol, Cannabidiol and Delta9-Tetrahydrocannabivarin. Br. J. Pharmacol. 2008, 153, 199–215. [Google Scholar] [CrossRef]
- Nelson, K.M.; Bisson, J.; Singh, G.; Graham, J.G.; Chen, S.-N.; Friesen, J.B.; Dahlin, J.L.; Niemitz, M.; Walters, M.A.; Pauli, G.F. The Essential Medicinal Chemistry of Cannabidiol (CBD). J. Med. Chem. 2020, 63, 12137–12155. [Google Scholar] [CrossRef]
- O’Sullivan, S.E.; Sun, Y.; Bennett, A.J.; Randall, M.D.; Kendall, D.A. Time-Dependent Vascular Actions of Cannabidiol in the Rat Aorta. Eur. J. Pharmacol. 2009, 612, 61–68. [Google Scholar] [CrossRef]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2020, 9, 21. [Google Scholar] [CrossRef]
- Ibeas Bih, C.; Chen, T.; Nunn, A.V.W.; Bazelot, M.; Dallas, M.; Whalley, B.J. Molecular Targets of Cannabidiol in Neurological Disorders. Neurotherapeutics 2015, 12, 699–730. [Google Scholar] [CrossRef]
- Ribaudo, G.; Landucci, E.; Giannangeli, M.; Mazzantini, C.; Maccarinelli, G.; Mastinu, A.; Bonini, S.A.; Memo, M.; Pellegrini-Giampietro, D.E.; Gianoncelli, A. Virtual Screening and In Vitro Experiments Highlight Cannabidiol as a Drug-like Phosphodiesterase 9 Inhibitor. Eur. J. Neurosci. 2022. [Google Scholar] [CrossRef]
- Perucca, E.; Bialer, M. Critical Aspects Affecting Cannabidiol Oral Bioavailability and Metabolic Elimination, and Related Clinical Implications. CNS Drugs 2020, 34, 795–800. [Google Scholar] [CrossRef]
- Taylor, L.; Gidal, B.; Blakey, G.; Tayo, B.; Morrison, G. A Phase I, Randomized, Double-Blind, Placebo-Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. CNS Drugs 2018, 32, 1053–1067. [Google Scholar] [CrossRef] [PubMed]
- Hoffmeyer, S.; Burk, O.; von Richter, O.; Arnold, H.P.; Brockmöller, J.; Johne, A.; Cascorbi, I.; Gerloff, T.; Roots, I.; Eichelbaum, M.; et al. Functional Polymorphisms of the Human Multidrug-Resistance Gene: Multiple Sequence Variations and Correlation of One Allele with P-Glycoprotein Expression and Activity in Vivo. Proc. Natl. Acad. Sci. USA 2000, 97, 3473–3478. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.A.; Jirsa, V.K.; Schevon, C.A. Wheels Within Wheels: Theory and Practice of Epileptic Networks. Epilepsy Curr. 2021, 21, 243–247. [Google Scholar] [CrossRef]
- Stout, S.M.; Cimino, N.M. Exogenous Cannabinoids as Substrates, Inhibitors, and Inducers of Human Drug Metabolizing Enzymes: A Systematic Review. Drug Metab. Rev. 2014, 46, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Saviano, A.; Raucci, F.; Tallarico, M.; De Caro, C.; Di Martino, S.; Nesci, V.; Roberti, R.; Iannone, L.F.; Colia, A.L.; Dimonte, S.; et al. Cannabidiol and the Central Nervous System: Translating into Clinics. Pharm. Adv. 2021, 3, 369. [Google Scholar] [CrossRef]
- Ujváry, I.; Hanuš, L. Human Metabolites of Cannabidiol: A Review on Their Formation, Biological Activity, and Relevance in Therapy. Cannabis Cannabinoid Res. 2016, 1, 90–101. [Google Scholar] [CrossRef]
- Ishiguro, H.; Iwasaki, S.; Teasenfitz, L.; Higuchi, S.; Horiuchi, Y.; Saito, T.; Arinami, T.; Onaivi, E.S. Involvement of Cannabinoid CB2 Receptor in Alcohol Preference in Mice and Alcoholism in Humans. Pharm. J. 2007, 7, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Pehlivan, S.; Aydın, P.; Aytaç, H.; Uysal, M.; Sever, U.; Pehlivan, M. Investigation of Catechol-O-Methyltransferase and Cannabinoid Receptor 2 Gene Variants in Tobacco Use Disorder or Tobacco Use Disorder and Schizophrenia Comorbidity. Anadolu Psikiyatri Derg. 2020, 21, 1. [Google Scholar] [CrossRef]
- Kim, H.; Neubert, J.K.; San Miguel, A.; Xu, K.; Krishnaraju, R.K.; Iadarola, M.J.; Goldman, D.; Dionne, R.A. Genetic Influence on Variability in Human Acute Experimental Pain Sensitivity Associated with Gender, Ethnicity and Psychological Temperament. PAIN 2004, 109, 488–496. [Google Scholar] [CrossRef]
- Binder, A.; May, D.; Baron, R.; Maier, C.; Tölle, T.R.; Treede, R.-D.; Berthele, A.; Faltraco, F.; Flor, H.; Gierthmühlen, J.; et al. Transient Receptor Potential Channel Polymorphisms Are Associated with the Somatosensory Function in Neuropathic Pain Patients. PLoS ONE 2011, 6, e17387. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Guo, M.; Huang, D.; Li, J.; Zhou, Y. TRPV1 Genetic Polymorphisms and Risk of COPD or COPD Combined with PH in the Han Chinese Population. Cell Cycle 2020, 19, 3066–3073. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Sadée, W. Searching for Polymorphisms That Affect Gene Expression and MRNA Processing: Example ABCB1 (MDR1). AAPS J. 2006, 8, E515–E520. [Google Scholar] [CrossRef] [PubMed]
- Jarrar, Y.B.; Lee, S.-J. Molecular Functionality of CYP2C9 Polymorphisms and Their Influence on Drug Therapy. Drug Metabol. Drug Interact. 2014, 29, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Chbili, C.; Fathallah, N.; Laouani, A.; Nouira, M.; Hassine, A.; Ben Amor, S.; Ben Ammou, S.; Ben Salem, C.; Saguem, S. Effects of EPHX1 and CYP3A4*22 Genetic Polymorphisms on Carbamazepine Metabolism and Drug Response among Tunisian Epileptic Patients. J. Neurogenet. 2016, 30, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Olson, K.C.; Dellinger, R.W.; Zhong, Q.; Sun, D.; Amin, S.; Spratt, T.E.; Lazarus, P. Functional Characterization of Low-Prevalence Missense Polymorphisms in the UDP-Glucuronosyltransferase 1A9 Gene. Drug Metab. Dispos. 2009, 37, 1999–2007. [Google Scholar] [CrossRef]
- Yamanaka, H.; Nakajima, M.; Katoh, M.; Hara, Y.; Tachibana, O.; Yamashita, J.; McLeod, H.L.; Yokoi, T. A Novel Polymorphism in the Promoter Region of Human UGT1A9 Gene (UGT1A9*22) and Its Effects on the Transcriptional Activity. Pharmacogenetics 2004, 14, 329–332. [Google Scholar] [CrossRef]
- Parkin, J.; Cohen, B. An Overview of the Immune System. Lancet 2001, 357, 1777–1789. [Google Scholar] [CrossRef]
- Roth, M.D.; Baldwin, G.C.; Tashkin, D.P. Effects of Delta-9-Tetrahydrocannabinol on Human Immune Function and Host Defense. Chem. Phys. Lipids 2002, 121, 229–239. [Google Scholar] [CrossRef]
- Specter, S.; Lancz, G. Effects of Marijuana on Human Natural Killer Cell Activity. Adv. Exp. Med. Biol. 1991, 288, 47–56. [Google Scholar] [CrossRef]
- Kienzl, M.; Storr, M.; Schicho, R. Cannabinoids and Opioids in the Treatment of Inflammatory Bowel Diseases. Clin. Transl. Gastroenterol. 2020, 11, e00120. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.; Germanos, R.; Weier, M.; Pollard, J.; Degenhardt, L.; Hall, W.; Buckley, N.; Farrell, M. The Use of Cannabis and Cannabinoids in Treating Symptoms of Multiple Sclerosis: A Systematic Review of Reviews. Curr. Neurol. Neurosci. Rep. 2018, 18, 8. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, H.; Qureshi, I.A.; Jahan, N.; Went, T.R.; Sultan, W.; Sapkota, A.; Alfonso, M. A Systematic Review of Fibromyalgia and Recent Advancements in Treatment: Is Medicinal Cannabis a New Hope? Cureus 2021, 13, e17332. [Google Scholar] [CrossRef] [PubMed]
- Mücke, M.; Weier, M.; Carter, C.; Copeland, J.; Degenhardt, L.; Cuhls, H.; Radbruch, L.; Häuser, W.; Conrad, R. Systematic Review and Meta-Analysis of Cannabinoids in Palliative Medicine. J. Cachexia Sarcopenia Muscle 2018, 9, 220–234. [Google Scholar] [CrossRef]
- Costiniuk, C.T.; Jenabian, M.-A. Cannabinoids and Inflammation: Implications for People Living with HIV. AIDS 2019, 33, 2273–2288. [Google Scholar] [CrossRef]
- Mahmud, M.S.; Hossain, M.S.; Ahmed, A.T.M.F.; Islam, M.Z.; Sarker, M.E.; Islam, M.R. Antimicrobial and Antiviral (SARS-CoV-2) Potential of Cannabinoids and Cannabis Sativa: A Comprehensive Review. Molecules 2021, 26, 7216. [Google Scholar] [CrossRef]
- Janecki, M.; Graczyk, M.; Lewandowska, A.A.; Pawlak, Ł. Anti-Inflammatory and Antiviral Effects of Cannabinoids in Inhibiting and Preventing SARS-CoV-2 Infection. Int. J. Mol. Sci. 2022, 23, 4170. [Google Scholar] [CrossRef]
- Arzimanoglou, A.; Brandl, U.; Cross, J.H.; Gil-Nagel, A.; Lagae, L.; Landmark, C.J.; Specchio, N.; Nabbout, R.; Thiele, E.A.; Gubbay, O.; et al. Epilepsy and Cannabidiol: A Guide to Treatment. Epileptic Disord. 2020, 22, 1–14. [Google Scholar] [CrossRef]
- Berger, A.A.; Keefe, J.; Winnick, A.; Gilbert, E.; Eskander, J.P.; Yazdi, C.; Kaye, A.D.; Viswanath, O.; Urits, I. Cannabis and Cannabidiol (CBD) for the Treatment of Fibromyalgia. Best Pract. Res. Clin. Anaesthesiol. 2020, 34, 617–631. [Google Scholar] [CrossRef]
- Baswan, S.M.; Klosner, A.E.; Glynn, K.; Rajgopal, A.; Malik, K.; Yim, S.; Stern, N. Therapeutic Potential of Cannabidiol (CBD) for Skin Health and Disorders. Clin. Cosmet. Investig. Dermatol. 2020, 13, 927–942. [Google Scholar] [CrossRef]
- Noris, M.; Remuzzi, G. Overview of Complement Activation and Regulation. Semin. Nephrol. 2013, 33, 479–492. [Google Scholar] [CrossRef] [PubMed]
- Kozela, E.; Krawczyk, M.; Kos, T.; Juknat, A.; Vogel, Z.; Popik, P. Cannabidiol Improves Cognitive Impairment and Reverses Cortical Transcriptional Changes Induced by Ketamine, in Schizophrenia-Like Model in Rats. Mol. Neurobiol. 2020, 57, 1733–1747. [Google Scholar] [CrossRef] [PubMed]
- Pinoli, M.; Marino, F.; Cosentino, M. Dopaminergic Regulation of Innate Immunity: A Review. J. Neuroimmune Pharmacol. 2017, 12, 602–623. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Chawla, A.; Pollard, J.W. Macrophage Biology in Development, Homeostasis and Disease. Nature 2013, 496, 445–455. [Google Scholar] [CrossRef]
- Epelman, S.; Lavine, K.J.; Randolph, G.J. Origin and Functions of Tissue Macrophages. Immunity 2014, 41, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Nourshargh, S.; Alon, R. Leukocyte Migration into Inflamed Tissues. Immunity 2014, 41, 694–707. [Google Scholar] [CrossRef]
- Sica, A.; Erreni, M.; Allavena, P.; Porta, C. Macrophage Polarization in Pathology. Cell. Mol. Life Sci. 2015, 72, 4111–4126. [Google Scholar] [CrossRef]
- Huang, Y.; Wan, T.; Pang, N.; Zhou, Y.; Jiang, X.; Li, B.; Gu, Y.; Huang, Y.; Ye, X.; Lian, H.; et al. Cannabidiol Protects Livers against Nonalcoholic Steatohepatitis Induced by High-Fat High Cholesterol Diet via Regulating NF-ΚB and NLRP3 Inflammasome Pathway. J. Cell. Physiol. 2019, 234, 21224–21234. [Google Scholar] [CrossRef]
- Kongkadee, K.; Wisuitiprot, W.; Ingkaninan, K.; Waranuch, N. Anti-Inflammation and Gingival Wound Healing Activities of Cannabis Sativa L. Subsp. Sativa (Hemp) Extract and Cannabidiol: An in Vitro Study. Arch. Oral Biol. 2022, 140, 105464. [Google Scholar] [CrossRef]
- Yekhtin, Z.; Khuja, I.; Meiri, D.; Or, R.; Almogi-Hazan, O. Differential Effects of D9 Tetrahydrocannabinol (THC)- and Cannabidiol (CBD)-Based Cannabinoid Treatments on Macrophage Immune Function In Vitro and on Gastrointestinal Inflammation in a Murine Model. Biomedicines 2022, 10, 1793. [Google Scholar] [CrossRef]
- Liu, C.; Ma, H.; Slitt, A.L.; Seeram, N.P. Inhibitory Effect of Cannabidiol on the Activation of NLRP3 Inflammasome Is Associated with Its Modulation of the P2X7 Receptor in Human Monocytes. J. Nat. Prod. 2020, 83, 2025–2029. [Google Scholar] [CrossRef] [PubMed]
- Yeisley, D.J.; Arabiyat, A.S.; Hahn, M.S. Cannabidiol-Driven Alterations to Inflammatory Protein Landscape of Lipopolysaccharide-Activated Macrophages In Vitro May Be Mediated by Autophagy and Oxidative Stress. Cannabis Cannabinoid Res. 2021, 6, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Sermet, S.; Li, J.; Bach, A.; Crawford, R.B.; Kaminski, N.E. Cannabidiol Selectively Modulates Interleukin (IL)-1β and IL-6 Production in Toll-like Receptor Activated Human Peripheral Blood Monocytes. Toxicology 2021, 464, 153016. [Google Scholar] [CrossRef] [PubMed]
- Dopkins, N.; Miranda, K.; Wilson, K.; Holloman, B.L.; Nagarkatti, P.; Nagarkatti, M. Effects of Orally Administered Cannabidiol on Neuroinflammation and Intestinal Inflammation in the Attenuation of Experimental Autoimmune Encephalomyelitis. J. Neuroimmune Pharmacol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Sacerdote, P.; Martucci, C.; Vaccani, A.; Bariselli, F.; Panerai, A.E.; Colombo, A.; Parolaro, D.; Massi, P. The Nonpsychoactive Component of Marijuana Cannabidiol Modulates Chemotaxis and IL-10 and IL-12 Production of Murine Macrophages Both in Vivo and in Vitro. J. Neuroimmunol. 2005, 159, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Muthumalage, T.; Rahman, I. Cannabidiol Differentially Regulates Basal and LPS-Induced Inflammatory Responses in Macrophages, Lung Epithelial Cells, and Fibroblasts. Toxicol. Appl. Pharmacol. 2019, 382, 114713. [Google Scholar] [CrossRef]
- Schultze, N.; Wanka, H.; Zwicker, P.; Lindequist, U.; Haertel, B. Mitochondrial Functions of THP-1 Monocytes Following the Exposure to Selected Natural Compounds. Toxicology 2017, 377, 57–63. [Google Scholar] [CrossRef]
- Wu, H.-Y.; Huang, C.-H.; Lin, Y.-H.; Wang, C.-C.; Jan, T.-R. Cannabidiol Induced Apoptosis in Human Monocytes through Mitochondrial Permeability Transition Pore-Mediated ROS Production. Free Radic. Biol. Med. 2018, 124, 311–318. [Google Scholar] [CrossRef]
- Krzyżewska, A.; Baranowska-Kuczko, M.; Jastrząb, A.; Kasacka, I.; Kozłowska, H. Cannabidiol Improves Antioxidant Capacity and Reduces Inflammation in the Lungs of Rats with Monocrotaline-Induced Pulmonary Hypertension. Molecules 2022, 27, 3327. [Google Scholar] [CrossRef]
- Wu, H.-Y.; Chang, A.-C.; Wang, C.-C.; Kuo, F.-H.; Lee, C.-Y.; Liu, D.-Z.; Jan, T.-R. Cannabidiol Induced a Contrasting Pro-Apoptotic Effect between Freshly Isolated and Precultured Human Monocytes. Toxicol. Appl. Pharmacol. 2010, 246, 141–147. [Google Scholar] [CrossRef]
- Glick, D.; Barth, S.; Macleod, K.F. Autophagy: Cellular and Molecular Mechanisms. J. Pathol. 2010, 221, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Tomer, S.; Mu, W.; Suryawanshi, G.; Ng, H.; Wang, L.; Wennerberg, W.; Rezek, V.; Martin, H.; Chen, I.; Kitchen, S.; et al. Cannabidiol Modulates Expression of Type I IFN Response Genes and HIV Infection in Macrophages. Front. Immunol. 2022, 13, 926696. [Google Scholar] [CrossRef] [PubMed]
- Sofroniew, M.V.; Vinters, H.V. Astrocytes: Biology and Pathology. Acta Neuropathol. 2010, 119, 7–35. [Google Scholar] [CrossRef] [PubMed]
- Hind, W.H.; England, T.J.; O’Sullivan, S.E. Cannabidiol Protects an in Vitro Model of the Blood-Brain Barrier from Oxygen-Glucose Deprivation via PPARγ and 5-HT1A Receptors. Br. J. Pharmacol. 2016, 173, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Di Giacomo, V.; Chiavaroli, A.; Recinella, L.; Orlando, G.; Cataldi, A.; Rapino, M.; Di Valerio, V.; Ronci, M.; Leone, S.; Brunetti, L.; et al. Antioxidant and Neuroprotective Effects Induced by Cannabidiol and Cannabigerol in Rat CTX-TNA2 Astrocytes and Isolated Cortexes. Int. J. Mol. Sci. 2020, 21, 3575. [Google Scholar] [CrossRef]
- Hayakawa, K.; Mishima, K.; Irie, K.; Hazekawa, M.; Mishima, S.; Fujioka, M.; Orito, K.; Egashira, N.; Katsurabayashi, S.; Takasaki, K.; et al. Cannabidiol Prevents a Post-Ischemic Injury Progressively Induced by Cerebral Ischemia via a High-Mobility Group Box1-Inhibiting Mechanism. Neuropharmacology 2008, 55, 1280–1286. [Google Scholar] [CrossRef]
- Schiavon, A.P.; Soares, L.M.; Bonato, J.M.; Milani, H.; Guimarães, F.S.; Weffort de Oliveira, R.M. Protective Effects of Cannabidiol against Hippocampal Cell Death and Cognitive Impairment Induced by Bilateral Common Carotid Artery Occlusion in Mice. Neurotox. Res. 2014, 26, 307–316. [Google Scholar] [CrossRef]
- Nayak, D.; Roth, T.L.; McGavern, D.B. Microglia Development and Function. Annu. Rev. Immunol. 2014, 32, 367–402. [Google Scholar] [CrossRef]
- Walter, L.; Franklin, A.; Witting, A.; Wade, C.; Xie, Y.; Kunos, G.; Mackie, K.; Stella, N. Nonpsychotropic Cannabinoid Receptors Regulate Microglial Cell Migration. J. Neurosci. 2003, 23, 1398–1405. [Google Scholar] [CrossRef]
- Amenta, P.S.; Jallo, J.I.; Tuma, R.F.; Elliott, M.B. A Cannabinoid Type 2 Receptor Agonist Attenuates Blood-Brain Barrier Damage and Neurodegeneration in a Murine Model of Traumatic Brain Injury. J. Neurosci. Res. 2012, 90, 2293–2305. [Google Scholar] [CrossRef]
- Landucci, E.; Mazzantini, C.; Lana, D.; Calvani, M.; Magni, G.; Giovannini, M.G.; Pellegrini-Giampietro, D.E. Cannabidiol Inhibits Microglia Activation and Mitigates Neuronal Damage Induced by Kainate in an In-Vitro Seizure Model. Neurobiol. Dis. 2022, 174, 105895. [Google Scholar] [CrossRef] [PubMed]
- Malek, N.; Popiolek-Barczyk, K.; Mika, J.; Przewlocka, B.; Starowicz, K. Anandamide, Acting via CB2 Receptors, Alleviates LPS-Induced Neuroinflammation in Rat Primary Microglial Cultures. Neural Plast. 2015, 2015, 130639. [Google Scholar] [CrossRef] [PubMed]
- Kozela, E.; Pietr, M.; Juknat, A.; Rimmerman, N.; Levy, R.; Vogel, Z. Cannabinoids Delta(9)-Tetrahydrocannabinol and Cannabidiol Differentially Inhibit the Lipopolysaccharide-Activated NF-KappaB and Interferon-Beta/STAT Proinflammatory Pathways in BV-2 Microglial Cells. J. Biol. Chem. 2010, 285, 1616–1626. [Google Scholar] [CrossRef] [PubMed]
- Juknat, A.; Kozela, E.; Kaushansky, N.; Mechoulam, R.; Vogel, Z. Anti-Inflammatory Effects of the Cannabidiol Derivative Dimethylheptyl-Cannabidiol—Studies in BV-2 Microglia and Encephalitogenic T Cells. J. Basic Clin. Physiol. Pharmacol. 2016, 27, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Martín-Moreno, A.M.; Reigada, D.; Ramírez, B.G.; Mechoulam, R.; Innamorato, N.; Cuadrado, A.; de Ceballos, M.L. Cannabidiol and Other Cannabinoids Reduce Microglial Activation in Vitro and in Vivo: Relevance to Alzheimer’s Disease. Mol. Pharmacol. 2011, 79, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Chen, N.; Liu, Y.; Godlewski, G.; Kaplan, H.J.; Shrader, S.H.; Song, Z.-H.; Shao, H. Studies of Involvement of G-Protein Coupled Receptor-3 in Cannabidiol Effects on Inflammatory Responses of Mouse Primary Astrocytes and Microglia. PLoS ONE 2021, 16, e0251677. [Google Scholar] [CrossRef]
- Wu, H.-Y.; Goble, K.; Mecha, M.; Wang, C.-C.; Huang, C.-H.; Guaza, C.; Jan, T.-R. Cannabidiol-Induced Apoptosis in Murine Microglial Cells through Lipid Raft. Glia 2012, 60, 1182–1190. [Google Scholar] [CrossRef]
- Hassan, S.; Eldeeb, K.; Millns, P.J.; Bennett, A.J.; Alexander, S.P.H.; Kendall, D.A. Cannabidiol Enhances Microglial Phagocytosis via Transient Receptor Potential (TRP) Channel Activation. Br. J. Pharmacol. 2014, 171, 2426–2439. [Google Scholar] [CrossRef]
- Yang, S.; Du, Y.; Zhao, X.; Tang, Q.; Su, W.; Hu, Y.; Yu, P. Cannabidiol Enhances Microglial Beta-Amyloid Peptide Phagocytosis and Clearance via Vanilloid Family Type 2 Channel Activation. Int. J. Mol. Sci. 2022, 23, 5367. [Google Scholar] [CrossRef]
- Snaidero, N.; Simons, M. The Logistics of Myelin Biogenesis in the Central Nervous System. Glia 2017, 65, 1021–1031. [Google Scholar] [CrossRef]
- Mato, S.; Victoria Sánchez-Gómez, M.; Matute, C. Cannabidiol Induces Intracellular Calcium Elevation and Cytotoxicity in Oligodendrocytes. Glia 2010, 58, 1739–1747. [Google Scholar] [CrossRef] [PubMed]
- Mecha, M.; Torrao, A.S.; Mestre, L.; Carrillo-Salinas, F.J.; Mechoulam, R.; Guaza, C. Cannabidiol Protects Oligodendrocyte Progenitor Cells from Inflammation-Induced Apoptosis by Attenuating Endoplasmic Reticulum Stress. Cell Death Dis. 2012, 3, e331. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Gaudenzio, N.; Tsai, M. Mast Cells in Inflammation and Disease: Recent Progress and Ongoing Concerns. Annu. Rev. Immunol. 2020, 38, 49–77. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lu, M.; Ren, J.; Wu, X.; Long, M.; Chen, L.; Chen, Z. Electroacupuncture Inhibits Mast Cell Degranulation via Cannabinoid CB2 Receptors in a Rat Model of Allergic Contact Dermatitis. Acupunct. Med. 2019, 37, 348–355. [Google Scholar] [CrossRef]
- Giudice, E.D.; Rinaldi, L.; Passarotto, M.; Facchinetti, F.; D’Arrigo, A.; Guiotto, A.; Carbonare, M.D.; Battistin, L.; Leon, A. Cannabidiol, Unlike Synthetic Cannabinoids, Triggers Activation of RBL-2H3 Mast Cells. J. Leukoc. Biol. 2007, 81, 1512–1522. [Google Scholar] [CrossRef]
- De Filippis, D.; Esposito, G.; Cirillo, C.; Cipriano, M.; Winter, B.Y.D.; Scuderi, C.; Sarnelli, G.; Cuomo, R.; Steardo, L.; Man, J.G.D.; et al. Cannabidiol Reduces Intestinal Inflammation through the Control of Neuroimmune Axis. PLoS ONE 2011, 6, e28159. [Google Scholar] [CrossRef]
- Chiossone, L.; Dumas, P.-Y.; Vienne, M.; Vivier, E. Natural Killer Cells and Other Innate Lymphoid Cells in Cancer. Nat. Rev. Immunol. 2018, 18, 671–688. [Google Scholar] [CrossRef]
- Vivier, E.; Raulet, D.H.; Moretta, A.; Caligiuri, M.A.; Zitvogel, L.; Lanier, L.L.; Yokoyama, W.M.; Ugolini, S. Innate or Adaptive Immunity? The Example of Natural Killer Cells. Science 2011, 331, 44–49. [Google Scholar] [CrossRef]
- Luetke-Eversloh, M.; Cicek, B.B.; Siracusa, F.; Thom, J.T.; Hamann, A.; Frischbutter, S.; Baumgrass, R.; Chang, H.-D.; Thiel, A.; Dong, J.; et al. NK Cells Gain Higher IFN-γ Competence during Terminal Differentiation. Eur. J. Immunol. 2014, 44, 2074–2084. [Google Scholar] [CrossRef]
- Zitti, B.; Bryceson, Y.T. Natural Killer Cells in Inflammation and Autoimmunity. Cytokine Growth Factor Rev. 2018, 42, 37–46. [Google Scholar] [CrossRef]
- Kishimoto, S.; Muramatsu, M.; Gokoh, M.; Oka, S.; Waku, K.; Sugiura, T. Endogenous Cannabinoid Receptor Ligand Induces the Migration of Human Natural Killer Cells. J. Biochem. 2005, 137, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, M.E.; Hong, S.; Stierle, A.; Stierle, D.; Stella, N.; Roberts, K.; Jaffar, Z. CB2 Receptors Regulate Natural Killer Cells That Limit Allergic Airway Inflammation in a Murine Model of Asthma. Allergy 2017, 72, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Matias, I.; Pochard, P.; Orlando, P.; Salzet, M.; Pestel, J.; Di Marzo, V. Presence and Regulation of the Endocannabinoid System in Human Dendritic Cells. Eur. J. Biochem. 2002, 269, 3771–3778. [Google Scholar] [CrossRef] [PubMed]
- Do, Y.; McKallip, R.J.; Nagarkatti, M.; Nagarkatti, P.S. Activation through Cannabinoid Receptors 1 and 2 on Dendritic Cells Triggers NF-KappaB-Dependent Apoptosis: Novel Role for Endogenous and Exogenous Cannabinoids in Immunoregulation. J. Immunol. 2004, 173, 2373–2382. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, H.F.; Dyer, K.D.; Foster, P.S. Eosinophils: Changing Perspectives in Health and Disease. Nat. Rev. Immunol. 2013, 13, 9–22. [Google Scholar] [CrossRef]
- Rothenberg, M.E.; Hogan, S.P. The Eosinophil. Annu. Rev. Immunol. 2006, 24, 147–174. [Google Scholar] [CrossRef]
- Cadman, E.T.; Thysse, K.A.; Bearder, S.; Cheung, A.Y.N.; Johnston, A.C.; Lee, J.J.; Lawrence, R.A. Eosinophils Are Important for Protection, Immunoregulation and Pathology during Infection with Nematode Microfilariae. PLoS Pathog. 2014, 10, e1003988. [Google Scholar] [CrossRef]
- Gleich, G.J. The Pathology of Asthma: With Emphasis on the Role of the Eosinophil. New Engl. Reg. Allergy Proc. 1986, 7, 421–424. [Google Scholar] [CrossRef]
- Chusid, M.J. Eosinophils: Friends or Foes? J. Allergy Clin. Immunol. Pract. 2018, 6, 1439–1444. [Google Scholar] [CrossRef]
- Karasuyama, H.; Obata, K.; Wada, T.; Tsujimura, Y.; Mukai, K. Newly Appreciated Roles for Basophils in Allergy and Protective Immunity. Allergy 2011, 66, 1133–1141. [Google Scholar] [CrossRef]
- Cabral, G.A.; Rogers, T.J.; Lichtman, A.H. Turning Over a New Leaf: Cannabinoid and Endocannabinoid Modulation of Immune Function. J. Neuroimmune Pharmacol. 2015, 10, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Frei, R.B.; Luschnig, P.; Parzmair, G.P.; Peinhaupt, M.; Schranz, S.; Fauland, A.; Wheelock, C.E.; Heinemann, A.; Sturm, E.M. Cannabinoid Receptor 2 Augments Eosinophil Responsiveness and Aggravates Allergen-induced Pulmonary Inflammation in Mice. Allergy 2016, 71, 944–956. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C. Neutrophils and Immunity: Challenges and Opportunities. Nat. Rev. Immunol. 2006, 6, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Condliffe, A.M.; Chilvers, E.R.; Haslett, C.; Dransfield, I. Priming Differentially Regulates Neutrophil Adhesion Molecule Expression/Function. Immunology 1996, 89, 105–111. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Levy, D.; Vasan, R.S.; Leip, E.P.; Wolf, P.A.; D’Agostino, R.B.; Murabito, J.M.; Kannel, W.B.; Benjamin, E.J. Temporal Relations of Atrial Fibrillation and Congestive Heart Failure and Their Joint Influence on Mortality: The Framingham Heart Study. Circulation 2003, 107, 2920–2925. [Google Scholar] [CrossRef]
- Wójcik, P.; Garley, M.; Wroński, A.; Jabłońska, E.; Skrzydlewska, E. Cannabidiol Modifies the Formation of NETs in Neutrophils of Psoriatic Patients. Int. J. Mol. Sci. 2020, 21, 6795. [Google Scholar] [CrossRef]
- Kolaczkowska, E.; Kubes, P. Neutrophil Recruitment and Function in Health and Inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef]
- Marino, F.; Maresca, A.M.; Castiglioni, L.; Cosentino, M.; Maio, R.C.; Schembri, L.; Klersy, C.; Mongiardi, C.; Robustelli Test, L.; Grandi, A.M.; et al. Simvastatin Down-Regulates the Production of Interleukin-8 by Neutrophil Leukocytes from Dyslipidemic Patients. BMC Cardiovasc. Disord. 2014, 14, 37. [Google Scholar] [CrossRef]
- Haschka, D.; Tymoszuk, P.; Bsteh, G.; Petzer, V.; Berek, K.; Theurl, I.; Berger, T.; Weiss, G. Expansion of Neutrophils and Classical and Nonclassical Monocytes as a Hallmark in Relapsing-Remitting Multiple Sclerosis. Front. Immunol. 2020, 11, 594. [Google Scholar] [CrossRef]
- Rumble, J.M.; Huber, A.K.; Krishnamoorthy, G.; Srinivasan, A.; Giles, D.A.; Zhang, X.; Wang, L.; Segal, B.M. Neutrophil-Related Factors as Biomarkers in EAE and MS. J. Exp. Med. 2015, 212, 23–35. [Google Scholar] [CrossRef]
- Lucchinetti, C.F.; Mandler, R.N.; McGavern, D.; Bruck, W.; Gleich, G.; Ransohoff, R.M.; Trebst, C.; Weinshenker, B.; Wingerchuk, D.; Parisi, J.E.; et al. A Role for Humoral Mechanisms in the Pathogenesis of Devic’s Neuromyelitis Optica. Brain 2002, 125, 1450–1461. [Google Scholar] [CrossRef] [PubMed]
- Brazil, J.C.; Louis, N.A.; Parkos, C.A. The Role of Polymorphonuclear Leukocyte Trafficking in the Perpetuation of Inflammation During Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2013, 19, 1556–1565. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Peng, W.; Zhang, Y.; Guo, J.; Fu, J.; Wang, W. Correlation of PMN Elastase and PMN Elastase-to-Neutrophil Ratio with Disease Activity in Patients with Myositis. J. Transl. Med. 2019, 17, 420. [Google Scholar] [CrossRef] [PubMed]
- Galkina, E.; Ley, K. Immune and Inflammatory Mechanisms of Atherosclerosis. Annu. Rev. Immunol. 2009, 27, 165–197. [Google Scholar] [CrossRef] [PubMed]
- Baetta, R.; Corsini, A. Role of Polymorphonuclear Neutrophils in Atherosclerosis: Current State and Future Perspectives. Atherosclerosis 2010, 210, 1–13. [Google Scholar] [CrossRef]
- Fialkow, L.; Wang, Y.; Downey, G.P. Reactive Oxygen and Nitrogen Species as Signaling Molecules Regulating Neutrophil Function. Free Radic. Biol. Med. 2007, 42, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Marino, F.; Guasti, L.; Cosentino, M.; Ferrari, M.; Rasini, E.; Maio, R.C.; Cimpanelli, M.G.; Cereda, E.; Crespi, C.; Simoni, C.; et al. Angiotensin II Type 1 Receptor Expression in Polymorphonuclear Leukocytes from High-Risk Subjects: Changes after Treatment with Simvastatin. J. Cardiovasc. Pharmacol. 2007, 49, 299–305. [Google Scholar] [CrossRef]
- Guasti, L.; Marino, F.; Cosentino, M.; Cimpanelli, M.; Maio, R.C.; Klersy, C.; Crespi, C.; Restelli, D.; Simoni, C.; Franzetti, I.; et al. Simvastatin Treatment Modifies Polymorphonuclear Leukocyte Function in High-Risk Individuals: A Longitudinal Study. J. Hypertens. 2006, 24, 2423–2430. [Google Scholar] [CrossRef] [PubMed]
- Guasti, L.; Marino, F.; Cosentino, M.; Maio, R.C.; Rasini, E.; Ferrari, M.; Castiglioni, L.; Klersy, C.; Gaudio, G.; Grandi, A.M.; et al. Prolonged Statin-Associated Reduction in Neutrophil Reactive Oxygen Species and Angiotensin II Type 1 Receptor Expression: 1-Year Follow-Up. Eur. Heart J. 2008, 29, 1118–1126. [Google Scholar] [CrossRef][Green Version]
- McHugh, D.; Tanner, C.; Mechoulam, R.; Pertwee, R.G.; Ross, R.A. Inhibition of Human Neutrophil Chemotaxis by Endogenous Cannabinoids and Phytocannabinoids: Evidence for a Site Distinct from CB1 and CB2. Mol. Pharmacol. 2008, 73, 441–450. [Google Scholar] [CrossRef]
- Gómez, C.T.; Lairion, F.; Repetto, M.; Ettcheto, M.; Merelli, A.; Lazarowski, A.; Auzmendi, J. Cannabidiol (CBD) Alters the Functionality of Neutrophils (PMN). Implications in the Refractory Epilepsy Treatment. Pharmaceuticals 2021, 14, 220. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, P.; Rajesh, M.; Horváth, B.; Bátkai, S.; Park, O.; Tanchian, G.; Gao, R.Y.; Patel, V.; Wink, D.A.; Liaudet, L.; et al. Cannabidiol Protects against Hepatic Ischemia/Reperfusion Injury by Attenuating Inflammatory Signaling and Response, Oxidative/Nitrative Stress, and Cell Death. Free Radic. Biol. Med. 2011, 50, 1368–1381. [Google Scholar] [CrossRef]
- Mabou Tagne, A.; Marino, F.; Legnaro, M.; Luini, A.; Pacchetti, B.; Cosentino, M. A Novel Standardized Cannabis Sativa L. Extract and Its Constituent Cannabidiol Inhibit Human Polymorphonuclear Leukocyte Functions. Int. J. Mol. Sci. 2019, 20, 1833. [Google Scholar] [CrossRef] [PubMed]
- Napimoga, M.H.; Benatti, B.B.; Lima, F.O.; Alves, P.M.; Campos, A.C.; Pena-Dos-Santos, D.R.; Severino, F.P.; Cunha, F.Q.; Guimarães, F.S. Cannabidiol Decreases Bone Resorption by Inhibiting RANK/RANKL Expression and pro-Inflammatory Cytokines during Experimental Periodontitis in Rats. Int. Immunopharmacol. 2009, 9, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Thapa, D.; Cairns, E.A.; Szczesniak, A.-M.; Toguri, J.T.; Caldwell, M.D.; Kelly, M.E.M. The Cannabinoids Δ8THC, CBD, and HU-308 Act via Distinct Receptors to Reduce Corneal Pain and Inflammation. Cannabis Cannabinoid Res. 2018, 3, 11–20. [Google Scholar] [CrossRef]
- Wang, Y.; Mukhopadhyay, P.; Cao, Z.; Wang, H.; Feng, D.; Haskó, G.; Mechoulam, R.; Gao, B.; Pacher, P. Cannabidiol Attenuates Alcohol-Induced Liver Steatosis, Metabolic Dysregulation, Inflammation and Neutrophil-Mediated Injury. Sci. Rep. 2017, 7, 12064. [Google Scholar] [CrossRef] [PubMed]
- Robaina Cabrera, C.L.; Keir-Rudman, S.; Horniman, N.; Clarkson, N.; Page, C. The Anti-Inflammatory Effects of Cannabidiol and Cannabigerol Alone, and in Combination. Pulm. Pharmacol. Ther. 2021, 69, 102047. [Google Scholar] [CrossRef]
- Karmaus, P.W.F.; Wagner, J.G.; Harkema, J.R.; Kaminski, N.E.; Kaplan, B.L.F. Cannabidiol (CBD) Enhances Lipopolysaccharide (LPS)-Induced Pulmonary Inflammation in C57BL/6 Mice. J. Immunotoxicol. 2013, 10, 321–328. [Google Scholar] [CrossRef]
- Makwana, R.; Venkatasamy, R.; Spina, D.; Page, C. The Effect of Phytocannabinoids on Airway Hyper-Responsiveness, Airway Inflammation, and Cough. J. Pharmacol. Exp. Ther. 2015, 353, 169–180. [Google Scholar] [CrossRef]
- Malfait, A.M.; Gallily, R.; Sumariwalla, P.F.; Malik, A.S.; Andreakos, E.; Mechoulam, R.; Feldmann, M. The Nonpsychoactive Cannabis Constituent Cannabidiol Is an Oral Anti-Arthritic Therapeutic in Murine Collagen-Induced Arthritis. Proc. Natl. Acad. Sci. USA 2000, 97, 9561–9566. [Google Scholar] [CrossRef]
- Biernacki, M.; Brzóska, M.M.; Markowska, A.; Gałażyn-Sidorczuk, M.; Cylwik, B.; Gęgotek, A.; Skrzydlewska, E. Oxidative Stress and Its Consequences in the Blood of Rats Irradiated with UV: Protective Effect of Cannabidiol. Antioxidants 2021, 10, 821. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 5th ed.; Oxford University Press: Oxford, UK, 2015; ISBN 978-0-19-871747-8. [Google Scholar]
- Ohms, M.; Möller, S.; Laskay, T. An Attempt to Polarize Human Neutrophils Toward N1 and N2 Phenotypes in Vitro. Front. Immunol. 2020, 11, 532. [Google Scholar] [CrossRef] [PubMed]
- Baban, B.; Hoda, N.; Malik, A.; Khodadadi, H.; Simmerman, E.; Vaibhav, K.; Mozaffari, M.S. Impact of Cannabidiol Treatment on Regulatory T-17 Cells and Neutrophil Polarization in Acute Kidney Injury. Am. J. Physiol.-Ren. Physiol. 2018, 315, F1149–F1158. [Google Scholar] [CrossRef] [PubMed]
- Lecluse, L.L.A.; Dowlatshahi, E.A.; Limpens, C.E.J.M.; de Rie, M.A.; Bos, J.D.; Spuls, P.I. Etanercept: An Overview of Dermatologic Adverse Events. Arch. Dermatol. 2011, 147, 79–94. [Google Scholar] [CrossRef] [PubMed]
- Lis, K.; Kuzawińska, O.; Bałkowiec-Iskra, E. Tumor Necrosis Factor Inhibitors—State of Knowledge. Arch. Med. Sci. 2014, 10, 1175–1185. [Google Scholar] [CrossRef]
- Patel, D.; Madani, S.; Patel, S.; Guglani, L. Review of Pulmonary Adverse Effects of Infliximab Therapy in Crohn’s Disease. Expert Opin. Drug Saf. 2016, 15, 769–775. [Google Scholar] [CrossRef]
- Subedi, S.; Gong, Y.; Chen, Y.; Shi, Y. Infliximab and Biosimilar Infliximab in Psoriasis: Efficacy, Loss of Efficacy, and Adverse Events. DDDT 2019, 13, 2491–2502. [Google Scholar] [CrossRef]
- Frostegård, J. Immunity, Atherosclerosis and Cardiovascular Disease. BMC Med. 2013, 11, 117. [Google Scholar] [CrossRef]
- Frostegård, J.; Ulfgren, A.K.; Nyberg, P.; Hedin, U.; Swedenborg, J.; Andersson, U.; Hansson, G.K. Cytokine Expression in Advanced Human Atherosclerotic Plaques: Dominance of pro-Inflammatory (Th1) and Macrophage-Stimulating Cytokines. Atherosclerosis 1999, 145, 33–43. [Google Scholar] [CrossRef]
- Marino, F.; Tozzi, M.; Schembri, L.; Ferraro, S.; Tarallo, A.; Scanzano, A.; Legnaro, M.; Castelli, P.; Cosentino, M. Correction: Production of IL-8, VEGF and Elastase by Circulating and Intraplaque Neutrophils in Patients with Carotid Atherosclerosis. PLoS ONE 2017, 12, e0181389. [Google Scholar] [CrossRef]
- Mach, F.; Montecucco, F.; Steffens, S. Cannabinoid Receptors in Acute and Chronic Complications of Atherosclerosis. Br. J. Pharmacol. 2008, 153, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Sahu, M.R.; Mondal, A.C. The Emerging Role of Hippo Signaling in Neurodegeneration. J. Neurosci. Res. 2020, 98, 796–814. [Google Scholar] [CrossRef] [PubMed]
- Teeling, J.L.; Perry, V.H. Systemic Infection and Inflammation in Acute CNS Injury and Chronic Neurodegeneration: Underlying Mechanisms. Neuroscience 2009, 158, 1062–1073. [Google Scholar] [CrossRef] [PubMed]
- Salmina, A.B.; Kharitonova, E.V.; Gorina, Y.V.; Teplyashina, E.A.; Malinovskaya, N.A.; Khilazheva, E.D.; Mosyagina, A.I.; Morgun, A.V.; Shuvaev, A.N.; Salmin, V.V.; et al. Blood-Brain Barrier and Neurovascular Unit In Vitro Models for Studying Mitochondria-Driven Molecular Mechanisms of Neurodegeneration. Int. J. Mol. Sci. 2021, 22, 4661. [Google Scholar] [CrossRef] [PubMed]
- Sulzer, D.; Alcalay, R.N.; Garretti, F.; Cote, L.; Kanter, E.; Agin-Liebes, J.; Liong, C.; McMurtrey, C.; Hildebrand, W.H.; Mao, X.; et al. T Cells from Patients with Parkinson’s Disease Recognize α-Synuclein Peptides. Nature 2017, 546, 656–661. [Google Scholar] [CrossRef]
- Marogianni, C.; Sokratous, M.; Dardiotis, E.; Hadjigeorgiou, G.M.; Bogdanos, D.; Xiromerisiou, G. Neurodegeneration and Inflammation-An Interesting Interplay in Parkinson’s Disease. Int. J. Mol. Sci. 2020, 21, E8421. [Google Scholar] [CrossRef]
- Yi, W.; Schlüter, D.; Wang, X. Astrocytes in Multiple Sclerosis and Experimental Autoimmune Encephalomyelitis: Star-Shaped Cells Illuminating the Darkness of CNS Autoimmunity. Brain Behav. Immun. 2019, 80, 10–24. [Google Scholar] [CrossRef]
- van Langelaar, J.; Rijvers, L.; Smolders, J.; van Luijn, M.M. B and T Cells Driving Multiple Sclerosis: Identity, Mechanisms and Potential Triggers. Front. Immunol. 2020, 11, 760. [Google Scholar] [CrossRef]
- Naegele, M.; Tillack, K.; Reinhardt, S.; Schippling, S.; Martin, R.; Sospedra, M. Neutrophils in Multiple Sclerosis Are Characterized by a Primed Phenotype. J. Neuroimmunol. 2012, 242, 60–71. [Google Scholar] [CrossRef]
- Milo, R.; Korczyn, A.D.; Manouchehri, N.; Stüve, O. The Temporal and Causal Relationship between Inflammation and Neurodegeneration in Multiple Sclerosis. Mult. Scler. 2020, 26, 876–886. [Google Scholar] [CrossRef]
- Healy, L.M.; Stratton, J.A.; Kuhlmann, T.; Antel, J. The Role of Glial Cells in Multiple Sclerosis Disease Progression. Nat. Rev. Neurol. 2022, 18, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, J. Parkinson’s Disease: Clinical Features and Diagnosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Kustrimovic, N.; Marino, F.; Cosentino, M. Peripheral Immunity, Immunoaging and Neuroinflammation in Parkinson’s Disease. CMC 2019, 26, 3719–3753. [Google Scholar] [CrossRef] [PubMed]
- Blandini, F.; Cosentino, M.; Mangiagalli, A.; Marino, F.; Samuele, A.; Rasini, E.; Fancellu, R.; Tassorelli, C.; Pacchetti, C.; Martignoni, E.; et al. Modifications of Apoptosis-Related Protein Levels in Lymphocytes of Patients with Parkinson’s Disease. The Effect of Dopaminergic Treatment. J. Neural Transm. 2004, 111, 1017–1030. [Google Scholar] [CrossRef] [PubMed]
- Kustrimovic, N.; Rasini, E.; Legnaro, M.; Bombelli, R.; Aleksic, I.; Blandini, F.; Comi, C.; Mauri, M.; Minafra, B.; Riboldazzi, G.; et al. Dopaminergic Receptors on CD4+ T Naive and Memory Lymphocytes Correlate with Motor Impairment in Patients with Parkinson’s Disease. Sci. Rep. 2016, 6, 33738. [Google Scholar] [CrossRef]
- Kustrimovic, N.; Comi, C.; Magistrelli, L.; Rasini, E.; Legnaro, M.; Bombelli, R.; Aleksic, I.; Blandini, F.; Minafra, B.; Riboldazzi, G.; et al. Parkinson’s Disease Patients Have a Complex Phenotypic and Functional Th1 Bias: Cross-Sectional Studies of CD4+ Th1/Th2/T17 and Treg in Drug-Naïve and Drug-Treated Patients. J. Neuroinflamm. 2018, 15, 205. [Google Scholar] [CrossRef]
- Storelli, E.; Cassina, N.; Rasini, E.; Marino, F.; Cosentino, M. Do Th17 Lymphocytes and IL-17 Contribute to Parkinson’s Disease? A Systematic Review of Available Evidence. Front. Neurol. 2019, 10, 13. [Google Scholar] [CrossRef]
- Magistrelli, L.; Storelli, E.; Rasini, E.; Contaldi, E.; Comi, C.; Cosentino, M.; Marino, F. Relationship between Circulating CD4+ T Lymphocytes and Cognitive Impairment in Patients with Parkinson’s Disease. Brain Behav. Immun. 2020, 89, 668–674. [Google Scholar] [CrossRef]
- Contaldi, E.; Magistrelli, L.; Milner, A.V.; Cosentino, M.; Marino, F.; Comi, C. Expression of Transcription Factors in CD4+ T Cells as Potential Biomarkers of Motor Complications in Parkinson’s Disease. J. Parkinsons Dis. 2021, 11, 507–514. [Google Scholar] [CrossRef]
- De Francesco, E.; Terzaghi, M.; Storelli, E.; Magistrelli, L.; Comi, C.; Legnaro, M.; Mauri, M.; Marino, F.; Versino, M.; Cosentino, M. CD4+ T-Cell Transcription Factors in Idiopathic REM Sleep Behavior Disorder and Parkinson’s Disease. Mov. Disord. 2021, 36, 225–229. [Google Scholar] [CrossRef]
- Contaldi, E.; Magistrelli, L.; Cosentino, M.; Marino, F.; Comi, C. Lymphocyte Count and Neutrophil-to-Lymphocyte Ratio Are Associated with Mild Cognitive Impairment in Parkinson’s Disease: A Single-Center Longitudinal Study. J. Clin. Med. 2022, 11, 5543. [Google Scholar] [CrossRef] [PubMed]
- Chagas, M.H.N.; Eckeli, A.L.; Zuardi, A.W.; Pena-Pereira, M.A.; Sobreira-Neto, M.A.; Sobreira, E.T.; Camilo, M.R.; Bergamaschi, M.M.; Schenck, C.H.; Hallak, J.E.C.; et al. Cannabidiol Can Improve Complex Sleep-Related Behaviours Associated with Rapid Eye Movement Sleep Behaviour Disorder in Parkinson’s Disease Patients: A Case Series. J. Clin. Pharm. Ther. 2014, 39, 564–566. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, C.; Francavilla, M.; Ongari, G.; Petese, A.; Ghezzi, C.; Rossini, N.; Blandini, F.; Cerri, S. Neuroprotective and Symptomatic Effects of Cannabidiol in an Animal Model of Parkinson’s Disease. Int. J. Mol. Sci. 2021, 22, 8920. [Google Scholar] [CrossRef] [PubMed]
- Clayton, K.A.; Van Enoo, A.A.; Ikezu, T. Alzheimer’s Disease: The Role of Microglia in Brain Homeostasis and Proteopathy. Front. Neurosci. 2017, 11, 680. [Google Scholar] [CrossRef] [PubMed]
- Princiotta Cariddi, L.; Mauri, M.; Cosentino, M.; Versino, M.; Marino, F. Alzheimer’s Disease: From Immune Homeostasis to Neuroinflammatory Condition. Int. J. Mol. Sci. 2022, 23, 13008. [Google Scholar] [CrossRef]
- Heneka, M.T.; Kummer, M.P.; Latz, E. Innate Immune Activation in Neurodegenerative Disease. Nat. Rev. Immunol. 2014, 14, 463–477. [Google Scholar] [CrossRef]
- McDonough, A.; Lee, R.V.; Weinstein, J.R. Microglial Interferon Signaling and White Matter. Neurochem. Res. 2017, 42, 2625–2638. [Google Scholar] [CrossRef]
- Aso, E.; Ferrer, I. CB2 Cannabinoid Receptor As Potential Target against Alzheimer’s Disease. Front. Neurosci. 2016, 10, 243. [Google Scholar] [CrossRef]
- Ribaudo, G.; Memo, M.; Gianoncelli, A. Multi-Target Natural and Nature-Inspired Compounds against Neurodegeneration: A Focus on Dual Cholinesterase and Phosphodiesterase Inhibitors. Appl. Sci. 2021, 11, 5044. [Google Scholar] [CrossRef]
- Xi, M.; Sun, T.; Chai, S.; Xie, M.; Chen, S.; Deng, L.; Du, K.; Shen, R.; Sun, H. Therapeutic Potential of Phosphodiesterase Inhibitors for Cognitive Amelioration in Alzheimer’s Disease. Eur. J. Med. Chem. 2022, 232, 114170. [Google Scholar] [CrossRef]
- Bernstein, H.-G.; Steiner, J.; Guest, P.C.; Dobrowolny, H.; Bogerts, B. Glial Cells as Key Players in Schizophrenia Pathology: Recent Insights and Concepts of Therapy. Schizophr. Res. 2015, 161, 4–18. [Google Scholar] [CrossRef] [PubMed]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms Underlying Inflammation in Neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar] [CrossRef] [PubMed]
- van Kesteren, C.F.M.G.; Gremmels, H.; de Witte, L.D.; Hol, E.M.; Van Gool, A.R.; Falkai, P.G.; Kahn, R.S.; Sommer, I.E.C. Immune Involvement in the Pathogenesis of Schizophrenia: A Meta-Analysis on Postmortem Brain Studies. Transl. Psychiatry 2017, 7, e1075. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-C.; Yang, Y.-Y.; Chou, Y.-M.; Chen, K.-P.; Shen, W.W.; Leu, S.-J. Immunologic Variables in Acute Mania of Bipolar Disorder. J. Neuroimmunol. 2004, 150, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, B.I.; Kemp, D.E.; Soczynska, J.K.; McIntyre, R.S. Inflammation and the Phenomenology, Pathophysiology, Comorbidity, and Treatment of Bipolar Disorder: A Systematic Review of the Literature. J. Clin. Psychiatry 2009, 70, 1078–1090. [Google Scholar] [CrossRef]
- Altamura, A.C.; Buoli, M.; Pozzoli, S. Role of Immunological Factors in the Pathophysiology and Diagnosis of Bipolar Disorder: Comparison with Schizophrenia. Psychiatry Clin. Neurosci. 2014, 68, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, M.; Godio, M.; Martini, S.; Callegari, C.; Cosentino, M.; Marino, F. Inflammatory Markers at Baseline Correlate with Subsequent Clinical Response to Quetiapine in Patients with Bipolar Disorder. Hum. Psychopharmacol. 2022, 37, e2854. [Google Scholar] [CrossRef]
- Khandaker, G.M.; Cousins, L.; Deakin, J.; Lennox, B.R.; Yolken, R.; Jones, P.B. Inflammation and Immunity in Schizophrenia: Implications for Pathophysiology and Treatment. Lancet Psychiatry 2015, 2, 258–270. [Google Scholar] [CrossRef]
- Pearlstein, J.G.; Staudenmaier, P.J.; West, A.E.; Geraghty, S.; Cosgrove, V.E. Immune Response to Stress Induction as a Predictor of Cognitive-Behavioral Therapy Outcomes in Adolescent Mood Disorders: A Pilot Study. J. Psychiatr. Res. 2020, 120, 56–63. [Google Scholar] [CrossRef]
- Saccaro, L.F.; Schilliger, Z.; Dayer, A.; Perroud, N.; Piguet, C. Inflammation, Anxiety, and Stress in Bipolar Disorder and Borderline Personality Disorder: A Narrative Review. Neurosci. Biobehav. Rev. 2021, 127, 184–192. [Google Scholar] [CrossRef]
- Hiles, S.A.; Baker, A.L.; de Malmanche, T.; Attia, J. Interleukin-6, C-Reactive Protein and Interleukin-10 after Antidepressant Treatment in People with Depression: A Meta-Analysis. Psychol. Med. 2012, 42, 2015–2026. [Google Scholar] [CrossRef] [PubMed]
- Haring, L.; Koido, K.; Vasar, V.; Leping, V.; Zilmer, K.; Zilmer, M.; Vasar, E. Antipsychotic Treatment Reduces Psychotic Symptoms and Markers of Low-Grade Inflammation in First Episode Psychosis Patients, but Increases Their Body Mass Index. Schizophr. Res. 2015, 169, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, M.; Godio, M.; Callegari, C.; Cosentino, M.; Marino, F. 6 Weeks Monotherapy with Antipsychotic Drug Reduced Inflammatory Markers in Bipolar Disorder Patients. Psychopharmacol. Bull. 2022, 52, 34–44. [Google Scholar] [PubMed]
- Rolland, B.; Jardri, R.; Amad, A.; Thomas, P.; Cottencin, O.; Bordet, R. Pharmacology of Hallucinations: Several Mechanisms for One Single Symptom? BioMed Res. Int. 2014, 2014, 307106. [Google Scholar] [CrossRef]
- De Almeida, V.; Martins-de-Souza, D. Cannabinoids and Glial Cells: Possible Mechanism to Understand Schizophrenia. Eur. Arch. Psychiatry Clin. Neurosci. 2018, 268, 727–737. [Google Scholar] [CrossRef]
- Esposito, G.; Scuderi, C.; Valenza, M.; Togna, G.I.; Latina, V.; De Filippis, D.; Cipriano, M.; Carratù, M.R.; Iuvone, T.; Steardo, L. Cannabidiol Reduces Aβ-Induced Neuroinflammation and Promotes Hippocampal Neurogenesis through PPARγ Involvement. PLoS ONE 2011, 6, e28668. [Google Scholar] [CrossRef]
- Farina, C.; Aloisi, F.; Meinl, E. Astrocytes Are Active Players in Cerebral Innate Immunity. Trends Immunol. 2007, 28, 138–145. [Google Scholar] [CrossRef]
- Tufano, M.; Pinna, G. Is There a Future for PPARs in the Treatment of Neuropsychiatric Disorders? Molecules 2020, 25, 1062. [Google Scholar] [CrossRef]
- Emrich, H.M.; Leweke, F.M.; Schneider, U. Towards a Cannabinoid Hypothesis of Schizophrenia: Cognitive Impairments Due to Dysregulation of the Endogenous Cannabinoid System. Pharmacol. Biochem. Behav. 1997, 56, 803–807. [Google Scholar] [CrossRef]
- Leweke, F.M.; Mueller, J.K.; Lange, B.; Rohleder, C. Therapeutic Potential of Cannabinoids in Psychosis. Biol. Psychiatry 2016, 79, 604–612. [Google Scholar] [CrossRef]
- Zuccoli, G.S.; Brandão-Teles, C.; Vieira, G.M.; Gomes, F.V.; Crunfli, F. Molecular Findings Guiding the Modulation of the Endocannabinoid System as a Potential Target to Treat Schizophrenia. Adv. Exp. Med. Biol. 2022, 1400, 89–103. [Google Scholar] [CrossRef]
- Husain, M.I.; Strawbridge, R.; Stokes, P.R.; Young, A.H. Anti-Inflammatory Treatments for Mood Disorders: Systematic Review and Meta-Analysis. J. Psychopharmacol. 2017, 31, 1137–1148. [Google Scholar] [CrossRef]
- Altinoz, M.A.; Ozpinar, A. Acetylsalicylic Acid and Its Metabolite Gentisic Acid May Act as Adjunctive Agents in the Treatment of Psychiatric Disorders. Behav. Pharmacol. 2019, 30, 627–641. [Google Scholar] [CrossRef]
- Petagna, L.; Antonelli, A.; Ganini, C.; Bellato, V.; Campanelli, M.; Divizia, A.; Efrati, C.; Franceschilli, M.; Guida, A.M.; Ingallinella, S.; et al. Pathophysiology of Crohn’s Disease Inflammation and Recurrence. Biol. Direct 2020, 15, 23. [Google Scholar] [CrossRef]
- Mandl, L.A. Osteoarthritis Year in Review 2018: Clinical. Osteoarthr. Cartil. 2019, 27, 359–364. [Google Scholar] [CrossRef]
- Orlowsky, E.W.; Kraus, V.B. The Role of Innate Immunity in Osteoarthritis: When Our First Line of Defense Goes on the Offensive. J. Rheumatol. 2015, 42, 363–371. [Google Scholar] [CrossRef]
- Dijkstra, D.J.; Joeloemsingh, J.V.; Bajema, I.M.; Trouw, L.A. Complement Activation and Regulation in Rheumatic Disease. Semin. Immunol. 2019, 45, 101339. [Google Scholar] [CrossRef]
- Dinarello, C.A. The IL-1 Family of Cytokines and Receptors in Rheumatic Diseases. Nat. Rev. Rheumatol. 2019, 15, 612–632. [Google Scholar] [CrossRef]
- Kafil, T.S.; Nguyen, T.M.; MacDonald, J.K.; Chande, N. Cannabis for the Treatment of Crohn’s Disease. Cochrane Database Syst. Rev. 2018, 2018, CD012853. [Google Scholar] [CrossRef]
- Philpott, H.T.; O’Brien, M.; McDougall, J.J. Attenuation of Early Phase Inflammation by Cannabidiol Prevents Pain and Nerve Damage in Rat Osteoarthritis. Pain 2017, 158, 2442–2451. [Google Scholar] [CrossRef]
- Lowin, T.; Schneider, M.; Pongratz, G. Joints for Joints: Cannabinoids in the Treatment of Rheumatoid Arthritis. Curr. Opin. Rheumatol. 2019, 31, 271–278. [Google Scholar] [CrossRef]
- Fitzcharles, M.-A.; Clauw, D.J.; Hauser, W. A Cautious Hope for Cannabidiol (CBD) in Rheumatology Care. Arthritis Care Res. 2020. [Google Scholar] [CrossRef]
- Pike, C.K.; Kim, M.; Schnitzer, K.; Mercaldo, N.; Edwards, R.; Napadow, V.; Zhang, Y.; Morrissey, E.J.; Alshelh, Z.; Evins, A.E.; et al. Study Protocol for a Phase II, Double-Blind, Randomised Controlled Trial of Cannabidiol (CBD) Compared with Placebo for Reduction of Brain Neuroinflammation in Adults with Chronic Low Back Pain. BMJ Open 2022, 12, e063613. [Google Scholar] [CrossRef]
- Mabou Tagne, A.; Pacchetti, B.; Sodergren, M.; Cosentino, M.; Marino, F. Cannabidiol for Viral Diseases: Hype or Hope? Cannabis Cannabinoid Res. 2020, 5, 121–131. [Google Scholar] [CrossRef]
- Amulic, B.; Cazalet, C.; Hayes, G.L.; Metzler, K.D.; Zychlinsky, A. Neutrophil Function: From Mechanisms to Disease. Annu. Rev. Immunol. 2012, 30, 459–489. [Google Scholar] [CrossRef]
- De Volder, J.; Vereecke, L.; Joos, G.; Maes, T. Targeting Neutrophils in Asthma: A Therapeutic Opportunity? Biochem. Pharmacol. 2020, 182, 114292. [Google Scholar] [CrossRef]
- Silvestre-Roig, C.; Braster, Q.; Ortega-Gomez, A.; Soehnlein, O. Neutrophils as Regulators of Cardiovascular Inflammation. Nat. Rev. Cardiol. 2020, 17, 327–340. [Google Scholar] [CrossRef]
| Cannabinoid Components | Brand Name | Origin | Indication(s) | Authorisation |
|---|---|---|---|---|
| Nabiximols (Δ9-THC and CBD 1:1) | Sativex | Natural | Multiple sclerosis-related spasticity and neuropathic pain, and cancer-related pain | FDA/EMA |
| Nabilone (Δ9-THC analogue) | Cesamet/Canemes | Synthetic | Chemotherapy-induced nausea and vomiting | FDA only |
| Dronabinol ((-)-trans-Δ9-THC) | Marinol/Syndros | Synthetic | Anorexia related to AIDS, and nausea and vomiting induced by chemotherapy | FDA only |
| CBD | Epidyolex | Natural | Epilepsy | FDA/EMA |
| CBDV | GWP42006 | Natural | Rett syndrome, fragile X syndrome *, and autism ** | / |
| (Whole plant) | Cannabis FM2, Cannabis Flos, Pedanios | Natural | Neuropathic and spasticity-associated pain, nausea and vomiting due to chemo/radiotherapy and HIV therapy, anorexia and cachexia, glaucoma, and Tourette syndrome | AIFA |
| Target | Action | Affinity (pKi) | Reference(s) |
|---|---|---|---|
| CB1 | Weak agonist, negative allosteric modulator | 3.64 | [38,39] |
| CB2 | Weak agonist, inverse agonist | 3.46 | [38,39] |
| TRPV1, TRPV2, TRPV3, TRPA1 | Agonist | N/A | [33] |
| TRPM8 | Antagonist | N/A | [33] |
| PPARγ | Agonist | N/A | [40] |
| GPR55 | Antagonist | N/A | [41] |
| GPR3, GPR6, GPR12 | Inverse agonist | N/A | [41] |
| 5-HT1A, 5-HT2A, 5-HT2C | Agonist | 1.1 (5-HT2C) | [35,39,42] |
| A2A | Agonist | N/A | [36] |
| μ, κ, and δ opioid receptors | Allosteric modulator | 1.3 (µ), 2.3 (κ), 6.4 (δ) | [37,39] |
| GLRA1, GLRB | Allosteric modulator | N/A | [42] |
| GLRA3 | Potentiator | N/A | [42] |
| GPR18 | Unknown | N/A | [42] |
| ADRA2B, ADRA2C | Unknown | 3.2 (B), 3.7 (C) | [39] |
| PDE9 | Inhibitor | N/A | [43] |
| Characteristic | Value | Note | Reference(s) |
|---|---|---|---|
| Oral bioavailability (fasting) | ~6% | Greatly increases with food intake (about 4-fold increase for both CMAX and AUC) | [44] |
| TMAX | 2.5–5 h | / | [45] |
| T1/2 | 10–17 h | / | [45] |
| Binding to plasma proteins (%) | ≥88% | / | [45] |
| Vd | 20,963–42,849 L | / | [45] |
| CL | 1111–1909 L/h | Mainly hepatic clearance | [45] |
| Protein | Gene | Variant | Nucleotide Change | AF (%) | Biological Effect |
|---|---|---|---|---|---|
| Cannabinoid receptor 1 (CB1R) | CNR1 | rs806368 | T>A | 21 | Associated with alcohol dependence [51] |
| rs806380 | A>G | 34 | Associated with alcohol dependence [51] | ||
| rs1049353 | C>A | 27 | Associated with alcohol dependence [51] | ||
| rs2023239 | T>C | 17 | Increased cannabinoids-induced side-effects [51] | ||
| Cannabinoid receptor 2 (CB2R) | CNR2 | rs2229579 | G>A | 10 | Increased mRNA expression [52] |
| Transient receptor potential cation channel subfamily V member 1 (TRPV1) | TRPV1 | rs8065080 | T>C | 38 | Associated with hypoalgesia [53,54] |
| rs222747 | C>A | 75 | Associated with hypoalgesia [53,54] | ||
| rs4790521 | T>C | 32 | Association with the COPD risk [55] | ||
| ATP-binding cassette subfamily C member 5 (ABCC5) | ABCC5 | rs3749442 | G>A | 17 | Increase in CBD response [47] |
| P-glycoprotein (P-gp) | ABCB1 | rs2032582 | A>C | 55 | Reduced P-gp expression and activity [56] |
| rs1045642 | A>C | 48 | Reduced P-gp expression and activity [56] | ||
| rs1128503 | A>G | 57 | Reduced P-gp expression and activity [56] | ||
| Solute carrier family 15 member 1 (SLC15A1) | SLC15A1 | rs1339067 | A>C | 66 | Decreased transporter expression in CNS and lower response to CBD [47] |
| Cytochrome P450 2C9 (CYP2C9) | CYP2C9 | rs1799853 (*2) | C>T | 12 | Decreased enzyme activity [57] |
| rs1057910 (*3) | A>C | 7 | Decreased enzyme activity [57] | ||
| Cytochrome P450 3A4 (CYP3A4) | CYP3A4 | rs35599367 | G>A | 5 | Decreased enzyme activity [58] |
| Aldehyde oxidase 1 (AOX1) | AOX1 | rs6729738 | C>A | 53 | CBD low seizure control in epilepsy [47] |
| Amine oxidase copper 1 (AOC1) | AOC1 | rs12539 | C>T | 22 | Increased diarrhoea and low seizure control in epileptic patients [47] |
| UGT1A9 (UDP-glucuronosyltransferase 1A9) | UGT1A9 | rs72551330 (*3) | T>A | 2-3 | Decreased enzymatic activity [59] |
| rs3832043 (*22) | (DT) 9>10 | 40 | Increased enzymatic activity [60] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martini, S.; Gemma, A.; Ferrari, M.; Cosentino, M.; Marino, F. Effects of Cannabidiol on Innate Immunity: Experimental Evidence and Clinical Relevance. Int. J. Mol. Sci. 2023, 24, 3125. https://doi.org/10.3390/ijms24043125
Martini S, Gemma A, Ferrari M, Cosentino M, Marino F. Effects of Cannabidiol on Innate Immunity: Experimental Evidence and Clinical Relevance. International Journal of Molecular Sciences. 2023; 24(4):3125. https://doi.org/10.3390/ijms24043125
Chicago/Turabian StyleMartini, Stefano, Alessandra Gemma, Marco Ferrari, Marco Cosentino, and Franca Marino. 2023. "Effects of Cannabidiol on Innate Immunity: Experimental Evidence and Clinical Relevance" International Journal of Molecular Sciences 24, no. 4: 3125. https://doi.org/10.3390/ijms24043125
APA StyleMartini, S., Gemma, A., Ferrari, M., Cosentino, M., & Marino, F. (2023). Effects of Cannabidiol on Innate Immunity: Experimental Evidence and Clinical Relevance. International Journal of Molecular Sciences, 24(4), 3125. https://doi.org/10.3390/ijms24043125







