Fascia Layer—A Novel Target for the Application of Biomaterials in Skin Wound Healing
Abstract
:1. Introduction
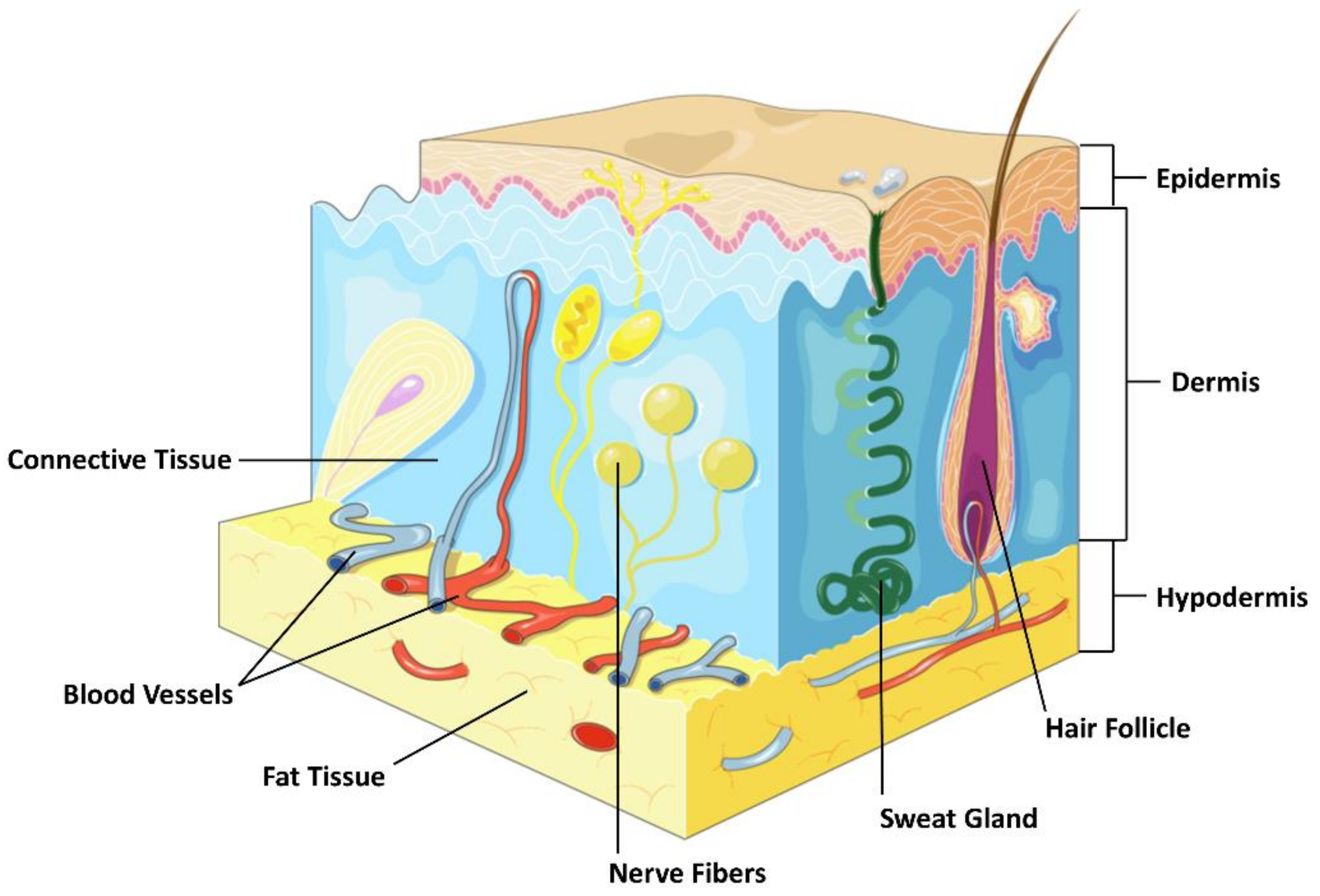
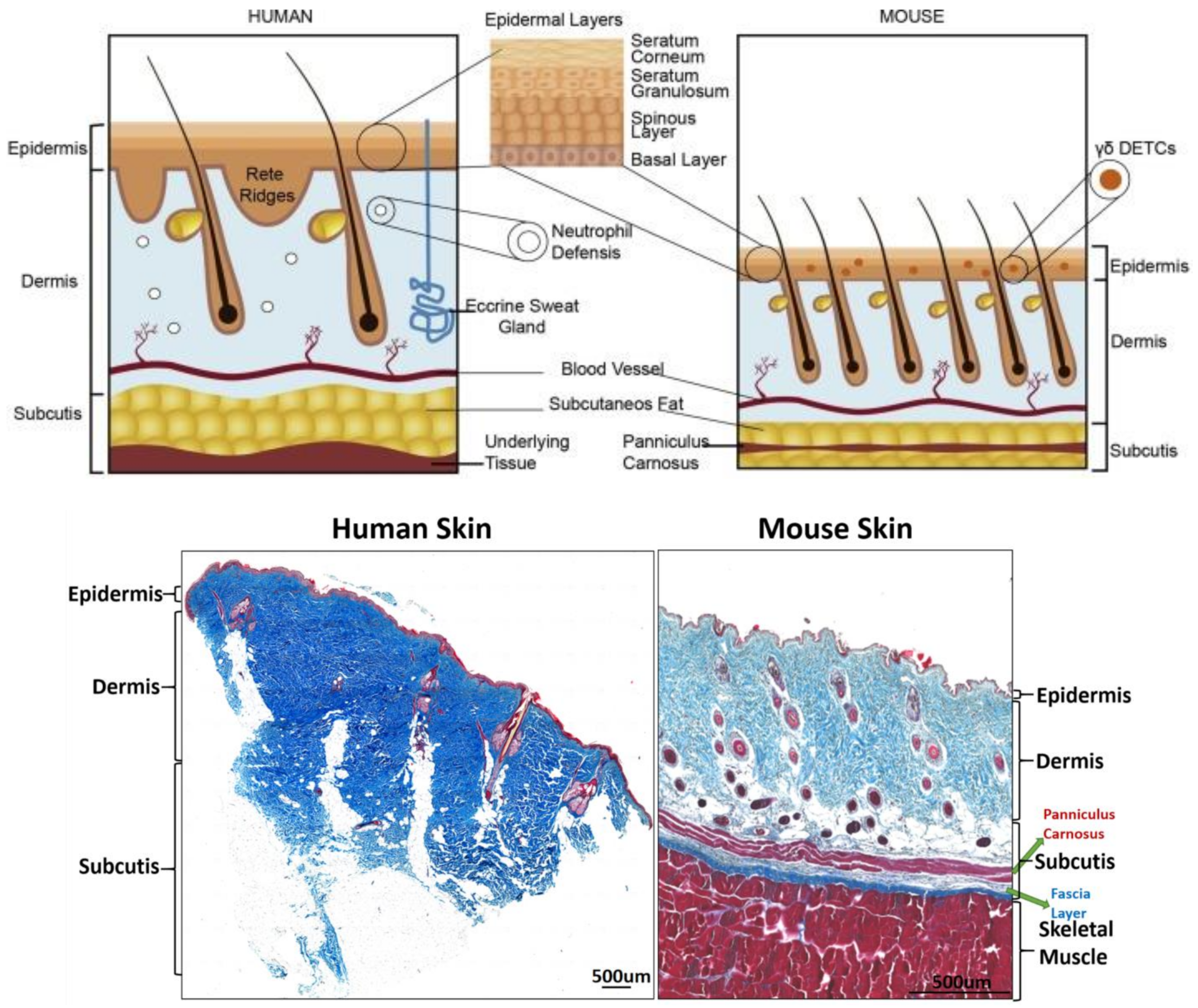
2. The Structure of the Skin
2.1. Epidermis
2.2. Dermis
2.3. Subcutaneous Tissue
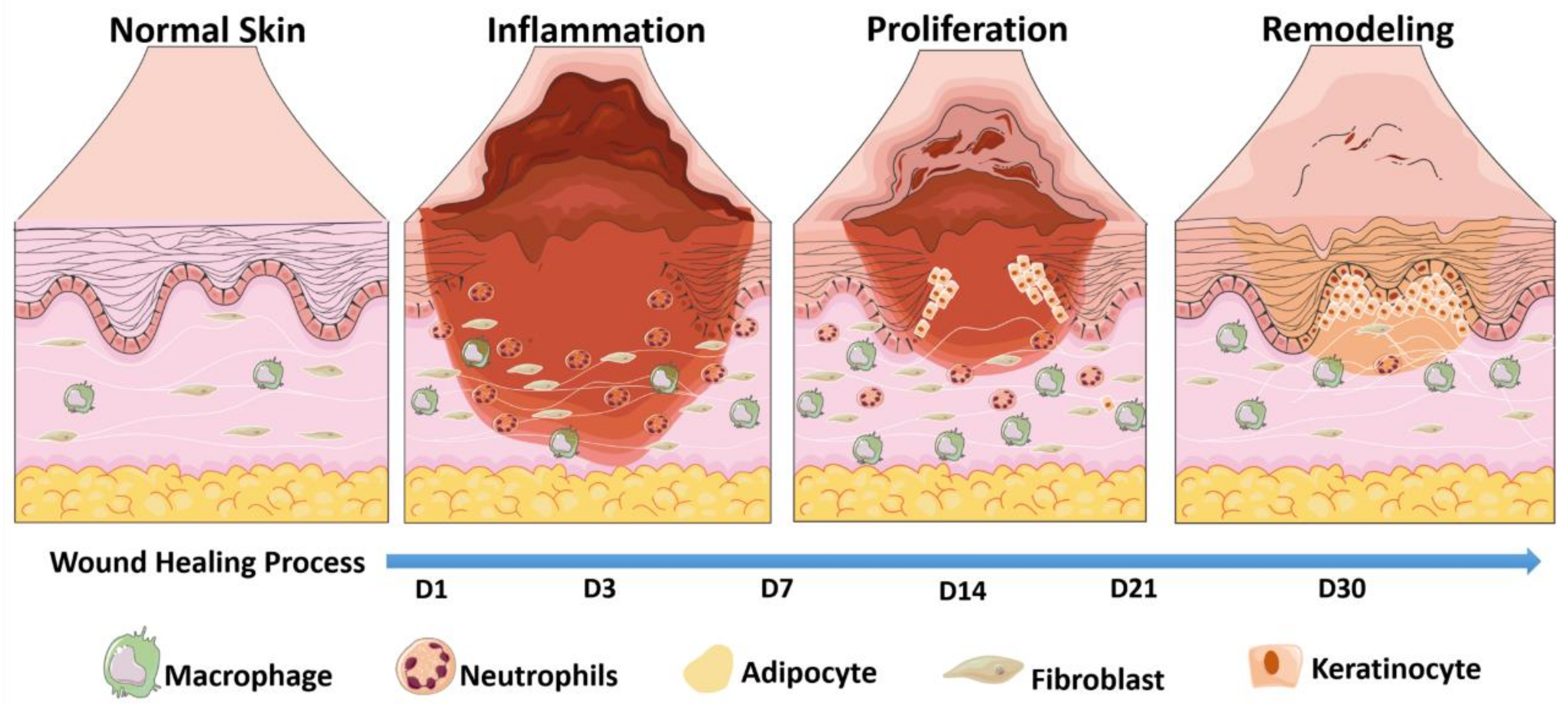
3. The Basic Process of Skin Wound Healing and Its New Finding
3.1. Inflammatory Phase
3.2. Proliferative Phase
3.3. Repair Phase
3.4. New Perspective of Skin Wound Healing
4. The Role of Biomaterials in Skin Wound Healing and Regeneration
4.1. Types of Biomaterials
4.2. Mechanisms by Which Bioactive Materials Promote Wound Repair and Skin Regeneration
4.2.1. Influence on Immune Cell Behavior
4.2.2. Modulation of Non-Immune Cell Behavior
4.2.3. Promotion of Skin Attachment Regeneration
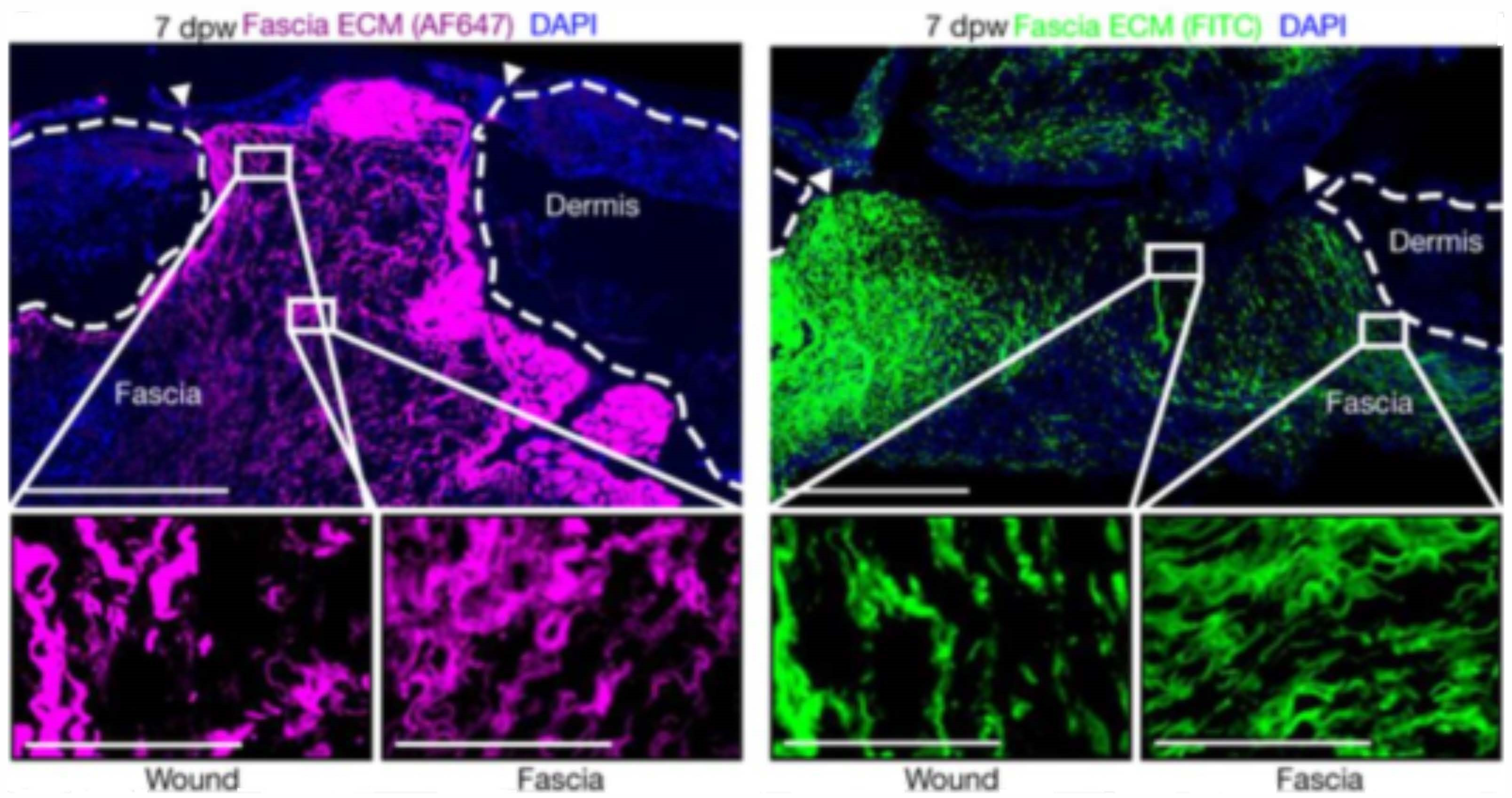
5. Fascia Layer—A New Target for Biomaterials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moreci, R.S.; Lechler, T. Epidermal Structure and Differentiation. Curr. Biol. 2020, 30, R144–R149. [Google Scholar] [CrossRef]
- Baroni, A.; Buommino, E.; De Gregorio, V.; Ruocco, E.; Ruocco, V.; Wolf, R. Structure and Function of the Epidermis Related to Barrier Properties. Clin. Dermatol. 2012, 30, 257–262. [Google Scholar] [CrossRef]
- van Smeden, J.; Janssens, M.; Gooris, G.S.; Bouwstra, J.A. The Important Role of Stratum Corneum Lipids for the Cutaneous Barrier Function. Biochim. Biophys. Acta 2014, 1841, 295–313. [Google Scholar] [CrossRef]
- Rawlings, A.V.; Harding, C.R. Moisturization and Skin Barrier Function. Dermatol. Ther. 2004, 17 (Suppl. S1), 43–48. [Google Scholar] [CrossRef]
- Lefèvre-Utile, A.; Braun, C.; Haftek, M.; Aubin, F. Five Functional Aspects of the Epidermal Barrier. Int. J. Mol. Sci. 2021, 22, 11676. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, R.; He, W.; Yao, Z.; Li, H.; Zhou, J.; Tan, J.; Yang, S.; Zhan, R.; Luo, G.; et al. Three-Dimensional Histological Structures of the Human Dermis. Tissue Eng. Part C Methods 2015, 21, 932–944. [Google Scholar] [CrossRef]
- Driskell, R.R.; Lichtenberger, B.M.; Hoste, E.; Kretzschmar, K.; Simons, B.D.; Charalambous, M.; Ferron, S.R.; Herault, Y.; Pavlovic, G.; Ferguson-Smith, A.C.; et al. Distinct Fibroblast Lineages Determine Dermal Architecture in Skin Development and Repair. Nature 2013, 504, 277–281. [Google Scholar] [CrossRef]
- Rippa, A.L.; Kalabusheva, E.P.; Vorotelyak, E.A. Regeneration of Dermis: Scarring and Cells Involved. Cells 2019, 8, 607. [Google Scholar] [CrossRef]
- Woodley, D.T. Distinct Fibroblasts in the Papillary and Reticular Dermis: Implications for Wound Healing. Dermatol. Clin. 2017, 35, 95–100. [Google Scholar] [CrossRef]
- Krieg, T.; Aumailley, M. The Extracellular Matrix of the Dermis: Flexible Structures with Dynamic Functions. Exp. Dermatol. 2011, 20, 689–695. [Google Scholar] [CrossRef]
- Amano, K.; Naito, M.; Matsuo, M. Morphological Study of Human Facial Fascia and Subcutaneous Tissue Structure by Region through SEM Observation. Tissue Cell 2020, 67, 101437. [Google Scholar] [CrossRef]
- Ishida, T.; Takeuchi, K.; Hayashi, S.; Kawata, S.; Hatayama, N.; Qu, N.; Shibata, M.; Itoh, M. Anatomical Structure of the Subcutaneous Tissue on the Anterior Surface of Human Thigh. Okajimas Folia Anat. Jpn. 2015, 92, 1–6. [Google Scholar] [CrossRef]
- Li, W.; Ahn, A.C. Subcutaneous Fascial Bands—A Qualitative and Morphometric Analysis. PLoS ONE 2011, 6, e23987. [Google Scholar] [CrossRef]
- Ahn, A.C.; Kaptchuk, T.J. Spatial Anisotropy Analyses of Subcutaneous Tissue Layer: Potential Insights into Its Biomechanical Characteristics. J. Anat. 2011, 219, 515–524. [Google Scholar] [CrossRef]
- Stecco, C.; Macchi, V.; Porzionato, A.; Duparc, F.; De Caro, R. The Fascia: The Forgotten Structure. Ital. J. Anat. Embryol. 2011, 116, 127–138. [Google Scholar]
- Zomer, H.D.; Trentin, A.G. Skin Wound Healing in Humans and Mice: Challenges in Translational Research. J. Dermatol. Sci. 2018, 90, 3–12. [Google Scholar] [CrossRef]
- Wang, P.-H.; Huang, B.-S.; Horng, H.-C.; Yeh, C.-C.; Chen, Y.-J. Wound Healing. J. Chin. Med. Assoc. 2018, 81, 94–101. [Google Scholar] [CrossRef]
- Eming, S.A.; Krieg, T.; Davidson, J.M. Inflammation in Wound Repair: Molecular and Cellular Mechanisms. J. Invest. Dermatol. 2007, 127, 514–525. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Kanno, E.; Tanno, H.; Sasaki, A.; Kitai, Y.; Miura, T.; Takagi, N.; Shoji, M.; Kasamatsu, J.; Sato, K.; et al. Distinct Roles for Dectin-1 and Dectin-2 in Skin Wound Healing and Neutrophilic Inflammatory Responses. J. Invest. Dermatol. 2021, 141, 164–176.e8. [Google Scholar] [CrossRef]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin Wound Healing: An Update on the Current Knowledge and Concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef]
- Rodero, M.P.; Khosrotehrani, K. Skin Wound Healing Modulation by Macrophages. Int. J. Clin. Exp. Pathol. 2010, 3, 643–653. [Google Scholar]
- Skin Acute Wound Healing: A Comprehensive Review—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/31275545/ (accessed on 25 August 2022).
- Liu, J.; Zhao, B.; Zhu, H.; Pan, Q.; Cai, M.; Bai, X.; Li, X.; Hu, X.; Zhang, M.; Shi, J.; et al. Wnt4 Negatively Regulates the TGF-Β1-Induced Human Dermal Fibroblast-to-Myofibroblast Transition via Targeting Smad3 and ERK. Cell Tissue Res. 2020, 379, 537–548. [Google Scholar] [CrossRef]
- Monika, P.; Waiker, P.V.; Chandraprabha, M.N.; Rangarajan, A.; Murthy, K.N.C. Myofibroblast Progeny in Wound Biology and Wound Healing Studies. Wound Repair Regen. 2021, 29, 531–547. [Google Scholar] [CrossRef]
- Yao, L.; Rathnakar, B.H.; Kwon, H.R.; Sakashita, H.; Kim, J.H.; Rackley, A.; Tomasek, J.J.; Berry, W.L.; Olson, L.E. Temporal Control of PDGFRα Regulates the Fibroblast-to-Myofibroblast Transition in Wound Healing. Cell Rep. 2022, 40, 111192. [Google Scholar] [CrossRef]
- Zhao, B.; Guan, H.; Liu, J.-Q.; Zheng, Z.; Zhou, Q.; Zhang, J.; Su, L.-L.; Hu, D.-H. Hypoxia Drives the Transition of Human Dermal Fibroblasts to a Myofibroblast-like Phenotype via the TGF-Β1/Smad3 Pathway. Int. J. Mol. Med. 2017, 39, 153–159. [Google Scholar] [CrossRef]
- Correa-Gallegos, D.; Jiang, D.; Christ, S.; Ramesh, P.; Ye, H.; Wannemacher, J.; Kalgudde Gopal, S.; Yu, Q.; Aichler, M.; Walch, A.; et al. Patch Repair of Deep Wounds by Mobilized Fascia. Nature 2019, 576, 287–292. [Google Scholar] [CrossRef]
- Wan, L.; Jiang, D.; Correa-Gallegos, D.; Ramesh, P.; Zhao, J.; Ye, H.; Zhu, S.; Wannemacher, J.; Volz, T.; Rinkevich, Y. Connexin43 Gap Junction Drives Fascia Mobilization and Repair of Deep Skin Wounds. Matrix Biol. 2021, 97, 58–71. [Google Scholar] [CrossRef]
- Jiang, D.; Christ, S.; Correa-Gallegos, D.; Ramesh, P.; Kalgudde Gopal, S.; Wannemacher, J.; Mayr, C.H.; Lupperger, V.; Yu, Q.; Ye, H.; et al. Injury Triggers Fascia Fibroblast Collective Cell Migration to Drive Scar Formation through N-Cadherin. Nat. Commun. 2020, 11, 5653. [Google Scholar] [CrossRef]
- Rajendran, V.; Ramesh, P.; Dai, R.; Kalgudde Gopal, S.; Ye, H.; Machens, H.-G.; Adler, H.; Jiang, D.; Rinkevich, Y. Therapeutic Silencing of P120 in Fascia Fibroblasts Ameliorates Tissue Repair. J. Invest. Dermatol. 2022. [Google Scholar] [CrossRef]
- Rinkevich, Y.; Correa-Gallegos, D.; Ye, H.; Dasgupta, B.; Sardogan, A.; Ichijo, R.; Strunz, M.; Ansari, M.; Angelidis, I.; Schiller, H.; et al. CD201+ Fascia Progenitors Choreograph Injury Repair. Res. Square, 2022; preprint. [Google Scholar] [CrossRef]
- Julier, Z.; Park, A.J.; Briquez, P.S.; Martino, M.M. Promoting Tissue Regeneration by Modulating the Immune System. Acta Biomater. 2017, 53, 13–28. [Google Scholar] [CrossRef]
- Huang, C.; Dong, L.; Zhao, B.; Lu, Y.; Huang, S.; Yuan, Z.; Luo, G.; Xu, Y.; Qian, W. Anti-inflammatory Hydrogel Dressings and Skin Wound Healing. Clin. Transl. Med. 2022, 12, e1094. [Google Scholar] [CrossRef]
- Konop, M.; Czuwara, J.; Kłodzińska, E.; Laskowska, A.K.; Zielenkiewicz, U.; Brzozowska, I.; Nabavi, S.M.; Rudnicka, L. Development of a Novel Keratin Dressing Which Accelerates Full-Thickness Skin Wound Healing in Diabetic Mice: In Vitro and in Vivo Studies. J. Biomater. Appl. 2018, 33, 527–540. [Google Scholar] [CrossRef]
- Ashouri, F.; Beyranvand, F.; Beigi Boroujeni, N.; Tavafi, M.; Sheikhian, A.; Varzi, A.M.; Shahrokhi, S. Macrophage Polarization in Wound Healing: Role of Aloe Vera/Chitosan Nanohydrogel. Drug Deliv. Transl. Res. 2019, 9, 1027–1042. [Google Scholar] [CrossRef]
- You, C.; Li, Q.; Wang, X.; Wu, P.; Ho, J.K.; Jin, R.; Zhang, L.; Shao, H.; Han, C. Silver Nanoparticle Loaded Collagen/Chitosan Scaffolds Promote Wound Healing via Regulating Fibroblast Migration and Macrophage Activation. Sci. Rep. 2017, 7, 10489. [Google Scholar] [CrossRef]
- Zhao, H.; Huang, J.; Li, Y.; Lv, X.; Zhou, H.; Wang, H.; Xu, Y.; Wang, C.; Wang, J.; Liu, Z. ROS-Scavenging Hydrogel to Promote Healing of Bacteria Infected Diabetic Wounds. Biomaterials 2020, 258, 120286. [Google Scholar] [CrossRef]
- Shen, T.; Dai, K.; Yu, Y.; Wang, J.; Liu, C. Sulfated Chitosan Rescues Dysfunctional Macrophages and Accelerates Wound Healing in Diabetic Mice. Acta Biomater. 2020, 117, 192–203. [Google Scholar] [CrossRef]
- Lohmann, N.; Schirmer, L.; Atallah, P.; Wandel, E.; Ferrer, R.A.; Werner, C.; Simon, J.C.; Franz, S.; Freudenberg, U. Glycosaminoglycan-Based Hydrogels Capture Inflammatory Chemokines and Rescue Defective Wound Healing in Mice. Sci. Transl. Med. 2017, 9, eaai9044. [Google Scholar] [CrossRef]
- Park, C.J.; Clark, S.G.; Lichtensteiger, C.A.; Jamison, R.D.; Johnson, A.J.W. Accelerated Wound Closure of Pressure Ulcers in Aged Mice by Chitosan Scaffolds with and without BFGF. Acta Biomater. 2009, 5, 1926–1936. [Google Scholar] [CrossRef]
- Burmeister, D.M.; Roy, D.C.; Becerra, S.C.; Natesan, S.; Christy, R.J. In Situ Delivery of Fibrin-Based Hydrogels Prevents Contraction and Reduces Inflammation. J. Burn. Care Res. 2017, 1, 40–53. [Google Scholar] [CrossRef]
- Oryan, A.; Alemzadeh, E.; Tashkhourian, J.; Nami Ana, S.F. Topical Delivery of Chitosan-Capped Silver Nanoparticles Speeds up Healing in Burn Wounds: A Preclinical Study. Carbohydr. Polym. 2018, 200, 82–92. [Google Scholar] [CrossRef]
- Artuc, M.; Hermes, B.; Steckelings, U.M.; Grützkau, A.; Henz, B.M. Mast Cells and Their Mediators in Cutaneous Wound Healing--Active Participants or Innocent Bystanders? Exp. Dermatol. 1999, 8, 1–16. [Google Scholar] [CrossRef]
- Weisel, J.W. Fibrinogen and Fibrin. In Advances in Protein Chemistry; Fibrous Proteins: Coiled-Coils, Collagen and Elastomers; Academic Press: Cambridge, MA, USA, 2005; Volume 70, pp. 247–299. [Google Scholar]
- Hartmann, K.; Henz, B.M.; Krüger-Krasagakes, S.; Köhl, J.; Burger, R.; Guhl, S.; Haase, I.; Lippert, U.; Zuberbier, T. C3a and C5a Stimulate Chemotaxis of Human Mast Cells. Blood 1997, 89, 2863–2870. [Google Scholar] [CrossRef]
- Trautmann, A.; Toksoy, A.; Engelhardt, E.; Bröcker, E.-B.; Gillitzer, R. Mast Cell Involvement in Normal Human Skin Wound Healing: Expression of Monocyte Chemoattractant Protein-1 Is Correlated with Recruitment of Mast Cells Which Synthesize Interleukin-4 in Vivo. J. Pathol. 2000, 190, 100–106. [Google Scholar] [CrossRef]
- Gillitzer, R.; Goebeler, M. Chemokines in Cutaneous Wound Healing. J. Leukoc. Biol. 2001, 69, 513–521. [Google Scholar]
- Mast Cells in Tissue Healing: From Skin to the Gastrointestinal Tract—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/22103847/ (accessed on 26 January 2023).
- Monocyte Chemoattractant Peptide-1 Expression during Cutaneous Allergic Reactions in Mice Is Mast Cell Dependent and Largely Mediates the Monocyte Recruitment Response—ScienceDirect. Available online: https://www.sciencedirect.com/science/article/pii/S009167490038006X?casa_token=UGe26hM9GeMAAAAA:cp04PcW9yGbC9VKhHzt6-xPEg6O0Yjm9SjDLpcN3MRhfNgYlzNPwThR1DEEyrenShIgpUGgJ (accessed on 26 January 2023).
- Egozi, E.I.; Ferreira, A.M.; Burns, A.L.; Gamelli, R.L.; Dipietro, L.A. Mast Cells Modulate the Inflammatory but Not the Proliferative Response in Healing Wounds. Wound Repair Regen. 2003, 11, 46–54. [Google Scholar] [CrossRef]
- Li, J.; Chen, J.; Kirsner, R. Pathophysiology of Acute Wound Healing. Clin. Dermatol. 2007, 25, 9–18. [Google Scholar] [CrossRef]
- Huttunen, M.; Aalto, M.L.; Harvima, R.J.; Horsmanheimo, M.; Harvima, I.T. Alterations in Mast Cells Showing Tryptase and Chymase Activity in Epithelializating and Chronic Wounds. Exp. Dermatol. 2000, 9, 258–265. [Google Scholar] [CrossRef]
- Huttunen, M.; Hyttinen, M.; Nilsson, G.; Butterfield, J.H.; Horsmanheimo, M.; Harvima, I.T. Inhibition of Keratinocyte Growth in Cell Culture and Whole Skin Culture by Mast Cell Mediators. Exp. Dermatol. 2001, 10, 184–192. [Google Scholar] [CrossRef]
- Tellechea, A.; Leal, E.C.; Kafanas, A.; Auster, M.E.; Kuchibhotla, S.; Ostrovsky, Y.; Tecilazich, F.; Baltzis, D.; Zheng, Y.; Carvalho, E.; et al. Mast Cells Regulate Wound Healing in Diabetes. Diabetes 2016, 65, 2006–2019. [Google Scholar] [CrossRef]
- Yamamoto, T.; Hartmann, K.; Eckes, B.; Krieg, T. Mast Cells Enhance Contraction of Three-Dimensional Collagen Lattices by Fibroblasts by Cell–Cell Interaction: Role of Stem Cell Factor/c-Kit. Immunology 2000, 99, 435–439. [Google Scholar] [CrossRef]
- Hiromatsu, Y.; Toda, S. Mast Cells and Angiogenesis. Microsc. Res. Tech. 2003, 60, 64–69. [Google Scholar] [CrossRef]
- Rao, K.N.; Brown, M.A. Mast Cells. Ann. N. Y. Acad. Sci. 2008, 1143, 83–104. [Google Scholar] [CrossRef]
- Maltby, S.; Khazaie, K.; McNagny, K.M. Mast Cells in Tumor Growth: Angiogenesis, Tissue Remodelling and Immune-Modulation. Biochim. Biophys. Acta (BBA)—Rev. Cancer 2009, 1796, 19–26. [Google Scholar] [CrossRef]
- Ozpinar, E.W.; Frey, A.L.; Cruse, G.; Freytes, D.O. Mast Cell—Biomaterial Interactions and Tissue Repair. Tissue Eng. Part B Rev. 2021, 27, 590–603. [Google Scholar] [CrossRef]
- Rinkevich, Y.; Walmsley, G.G.; Hu, M.S.; Maan, Z.N.; Newman, A.M.; Drukker, M.; Januszyk, M.; Krampitz, G.W.; Gurtner, G.C.; Lorenz, H.P.; et al. Identification and Isolation of a Dermal Lineage with Intrinsic Fibrogenic Potential. Science 2015, 348, aaa2151. [Google Scholar] [CrossRef]
- Yannas, I.V.; Tzeranis, D.S.; So, P.T.C. Regeneration of Injured Skin and Peripheral Nerves Requires Control of Wound Contraction, Not Scar Formation: Regeneration Requires Contraction Blocking. Wound Rep. Reg. 2017, 25, 177–191. [Google Scholar] [CrossRef]
- Wang, X.; Chung, L.; Hooks, J.; Maestas, D.R.; Lebid, A.; Andorko, J.I.; Huleihel, L.; Chin, A.F.; Wolf, M.; Remlinger, N.T.; et al. Type 2 Immunity Induced by Bladder Extracellular Matrix Enhances Corneal Wound Healing. Sci. Adv. 2021, 7, eabe2635. [Google Scholar] [CrossRef]
- Gholipourmalekabadi, M.; Khosravimelal, S.; Nokhbedehghan, Z.; Sameni, M.; Jajarmi, V.; Urbanska, A.M.; Mirzaei, H.; Salimi, M.; Chauhan, N.P.S.; Mobaraki, M.; et al. Modulation of Hypertrophic Scar Formation Using Amniotic Membrane/Electrospun Silk Fibroin Bilayer Membrane in a Rabbit Ear Model. ACS Biomater. Sci. Eng. 2019, 5, 1487–1496. [Google Scholar] [CrossRef]
- Izadyari Aghmiuni, A.; Heidari Keshel, S.; Sefat, F.; Akbarzadeh Khiyavi, A. Quince Seed Mucilage-Based Scaffold as a Smart Biological Substrate to Mimic Mechanobiological Behavior of Skin and Promote Fibroblasts Proliferation and h-ASCs Differentiation into Keratinocytes. Int. J. Biol. Macromol. 2020, 142, 668–679. [Google Scholar] [CrossRef]
- Ekambaram, R.; Dharmalingam, S. Fabrication and Evaluation of Electrospun Biomimetic Sulphonated PEEK Nanofibrous Scaffold for Human Skin Cell Proliferation and Wound Regeneration Potential. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 115, 111150. [Google Scholar] [CrossRef]
- Tchemtchoua, V.T.; Atanasova, G.; Aqil, A.; Filée, P.; Garbacki, N.; Vanhooteghem, O.; Deroanne, C.; Noël, A.; Jérome, C.; Nusgens, B.; et al. Development of a Chitosan Nanofibrillar Scaffold for Skin Repair and Regeneration. Biomacromolecules 2011, 12, 3194–3204. [Google Scholar] [CrossRef]
- Tutuianu, R.; Rosca, A.-M.; Albu Kaya, M.G.; Pruna, V.; Neagu, T.P.; Lascar, I.; Simionescu, M.; Titorencu, I. Mesenchymal Stromal Cell-Derived Factors Promote the Colonization of Collagen 3D Scaffolds with Human Skin Cells. J. Cell. Mol. Med. 2020, 24, 9692–9704. [Google Scholar] [CrossRef]
- Kalyanaraman, B.; Boyce, S. Assessment of an Automated Bioreactor to Propagate and Harvest Keratinocytes for Fabrication of Engineered Skin Substitutes. Tissue Eng. 2007, 13, 983–993. [Google Scholar] [CrossRef]
- Kim, S.-S.; Gwak, S.-J.; Choi, C.Y.; Kim, B.-S. Skin Regeneration Using Keratinocytes and Dermal Fibroblasts Cultured on Biodegradable Microspherical Polymer Scaffolds. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 75, 369–377. [Google Scholar] [CrossRef]
- Revi, D.; Paul, W.; Anilkumar, T.V.; Sharma, C.P. Chitosan Scaffold Co-Cultured with Keratinocyte and Fibroblast Heals Full Thickness Skin Wounds in Rabbit. J. Biomed. Mater. Res. A 2014, 102, 3273–3281. [Google Scholar] [CrossRef]
- Bannasch, H.; Stark, G.B.; Knam, F.; Horch, R.E.; Föhn, M. Decellularized Dermis in Combination with Cultivated Keratinocytes in a Short- and Long-Term Animal Experimental Investigation. J. Eur. Acad. Derm. Venereol. 2008, 22, 41–49. [Google Scholar] [CrossRef]
- Pelipenko, J.; Kocbek, P.; Govedarica, B.; Rošic, R.; Baumgartner, S.; Kristl, J. The Topography of Electrospun Nanofibers and Its Impact on the Growth and Mobility of Keratinocytes. Eur. J. Pharm. Biopharm. 2013, 84, 401–411. [Google Scholar] [CrossRef]
- Liu, X.; Lee, P.-Y.; Ho, C.-M.; Lui, V.C.H.; Chen, Y.; Che, C.-M.; Tam, P.K.H.; Wong, K.K.Y. Silver Nanoparticles Mediate Differential Responses in Keratinocytes and Fibroblasts during Skin Wound Healing. ChemMedChem 2010, 5, 468–475. [Google Scholar] [CrossRef]
- Kalirajan, C.; Palanisamy, T. A ZnO–Curcumin Nanocomposite Embedded Hybrid Collagen Scaffold for Effective Scarless Skin Regeneration in Acute Burn Injury. J. Mater. Chem. B 2019, 7, 5873–5886. [Google Scholar] [CrossRef]
- Yang, C.-C.; Cotsarelis, G. Review of Hair Follicle Dermal Cells. J. Dermatol. Sci. 2010, 57, 2–11. [Google Scholar] [CrossRef]
- Qi, C.; Xu, L.; Deng, Y.; Wang, G.; Wang, Z.; Wang, L. Sericin Hydrogels Promote Skin Wound Healing with Effective Regeneration of Hair Follicles and Sebaceous Glands after Complete Loss of Epidermis and Dermis. Biomater. Sci. 2018, 6, 2859–2870. [Google Scholar] [CrossRef]
- Liu, W.; Wang, M.; Cheng, W.; Niu, W.; Chen, M.; Luo, M.; Xie, C.; Leng, T.; Zhang, L.; Lei, B. Bioactive Antiinflammatory Antibacterial Hemostatic Citrate-Based Dressing with Macrophage Polarization Regulation for Accelerating Wound Healing and Hair Follicle Neogenesis. Bioact. Mater. 2021, 6, 721–728. [Google Scholar] [CrossRef]
- Tsai, H.-C.; Sheng, C.; Chang, L.-S.; Wen, Z.-H.; Ho, C.-Y.; Chen, C.-M. Chitosan-Microencapsulated RhEGF in Promoting Wound Healing. J. Wound Care 2021, 30, IXi–IXxi. [Google Scholar] [CrossRef]
- Leng, Q.; Li, Y.; Pang, X.; Wang, B.; Wu, Z.; Lu, Y.; Xiong, K.; Zhao, L.; Zhou, P.; Fu, S. Curcumin Nanoparticles Incorporated in PVA/Collagen Composite Films Promote Wound Healing. Drug Deliv. 2020, 27, 1676–1685. [Google Scholar] [CrossRef]
- Zhang, Z.; Dai, Q.; Zhang, Y.; Zhuang, H.; Wang, E.; Xu, Q.; Ma, L.; Wu, C.; Huan, Z.; Guo, F.; et al. Design of a Multifunctional Biomaterial Inspired by Ancient Chinese Medicine for Hair Regeneration in Burned Skin. ACS Appl. Mater. Interfaces 2020, 12, 12489–12499. [Google Scholar] [CrossRef]
- Weng, T.; Wu, P.; Zhang, W.; Zheng, Y.; Li, Q.; Jin, R.; Chen, H.; You, C.; Guo, S.; Han, C.; et al. Regeneration of Skin Appendages and Nerves: Current Status and Further Challenges. J. Transl. Med. 2020, 18, 53. [Google Scholar] [CrossRef]
- Abaci, H.E.; Coffman, A.; Doucet, Y.; Chen, J.; Jacków, J.; Wang, E.; Guo, Z.; Shin, J.U.; Jahoda, C.A.; Christiano, A.M. Tissue Engineering of Human Hair Follicles Using a Biomimetic Developmental Approach. Nat. Commun. 2018, 9, 5301. [Google Scholar] [CrossRef]
- Li, H.; Chen, L.; Zhang, M.; Tang, S.; Fu, X. Three-Dimensional Culture and Identification of Human Eccrine Sweat Glands in Matrigel Basement Membrane Matrix. Cell Tissue Res. 2013, 354, 897–902. [Google Scholar] [CrossRef]
- Chen, L.; Du, L.; Zhang, L.; Xie, S.; Zhang, X.; Li, H. EGFR Inhibitor AG1478 Blocks the Formation of 3D Structures Mainly through ERK Signaling Pathway in Matrigel-Induced 3D Reconstruction of Eccrine Sweat Gland-like Structures. J. Mol. Histol. 2020, 51, 191–197. [Google Scholar] [CrossRef]
- Li, H.; Chen, L.; Zeng, S.; Li, X.; Zhang, X.; Lin, C.; Zhang, M.; Xie, S.; He, Y.; Shu, S.; et al. Matrigel Basement Membrane Matrix Induces Eccrine Sweat Gland Cells to Reconstitute Sweat Gland-like Structures in Nude Mice. Exp. Cell Res. 2015, 332, 67–77. [Google Scholar] [CrossRef]
- Lei, X.; Liu, B.; Wu, J.; Lu, Y.; Yang, Y. Matrigel-Induced Tubular Morphogenesis of Human Eccrine Sweat Gland Epithelial Cells. Anat. Rec. 2011, 294, 1525–1531. [Google Scholar] [CrossRef]
- Huang, S.; Xu, Y.; Wu, C.; Sha, D.; Fu, X. In Vitro Constitution and in Vivo Implantation of Engineered Skin Constructs with Sweat Glands. Biomaterials 2010, 31, 5520–5525. [Google Scholar] [CrossRef]
- Chao, F.-C.; Wu, M.-H.; Chen, L.-C.; Lin, H.-L.; Liu, D.-Z.; Ho, H.-O.; Sheu, M.-T. Preparation and Characterization of Chemically TEMPO-Oxidized and Mechanically Disintegrated Sacchachitin Nanofibers (SCNF) for Enhanced Diabetic Wound Healing. Carbohydr. Polym. 2020, 229, 115507. [Google Scholar] [CrossRef]
- Diao, J.; Liu, J.; Wang, S.; Chang, M.; Wang, X.; Guo, B.; Yu, Q.; Yan, F.; Su, Y.; Wang, Y. Sweat Gland Organoids Contribute to Cutaneous Wound Healing and Sweat Gland Regeneration. Cell Death Dis. 2019, 10, 238. [Google Scholar] [CrossRef]
- Eweida, A.; Saad, M.; Gabr, E.; Marei, M.; Khalil, M.R. Cultured Keratinocytes on Urinary Bladder Matrix Scaffolds Increase Angiogenesis and Help in Rapid Healing of Wounds. Adv. Ski. Wound Care 2011, 24, 268–273. [Google Scholar] [CrossRef]
- do Amaral, R.J.F.C.; Zayed, N.M.A.; Pascu, E.I.; Cavanagh, B.; Hobbs, C.; Santarella, F.; Simpson, C.R.; Murphy, C.M.; Sridharan, R.; González-Vázquez, A.; et al. Functionalising Collagen-Based Scaffolds With Platelet-Rich Plasma for Enhanced Skin Wound Healing Potential. Front. Bioeng. Biotechnol. 2019, 7, 371. [Google Scholar] [CrossRef]
- Li, Y.; Xu, T.; Tu, Z.; Dai, W.; Xue, Y.; Tang, C.; Gao, W.; Mao, C.; Lei, B.; Lin, C. Bioactive Antibacterial Silica-Based Nanocomposites Hydrogel Scaffolds with High Angiogenesis for Promoting Diabetic Wound Healing and Skin Repair. Theranostics 2020, 10, 4929–4943. [Google Scholar] [CrossRef]
- Wang, M.; Wang, C.; Chen, M.; Xi, Y.; Cheng, W.; Mao, C.; Xu, T.; Zhang, X.; Lin, C.; Gao, W.; et al. Efficient Angiogenesis-Based Diabetic Wound Healing/Skin Reconstruction through Bioactive Antibacterial Adhesive Ultraviolet Shielding Nanodressing with Exosome Release. ACS Nano 2019, 13, 10279–10293. [Google Scholar] [CrossRef]
- Li, J.; Zhai, D.; Lv, F.; Yu, Q.; Ma, H.; Yin, J.; Yi, Z.; Liu, M.; Chang, J.; Wu, C. Preparation of Copper-Containing Bioactive Glass/Eggshell Membrane Nanocomposites for Improving Angiogenesis, Antibacterial Activity and Wound Healing. Acta Biomater. 2016, 36, 254–266. [Google Scholar] [CrossRef]
- Wang, X.; Cheng, F.; Liu, J.; Smått, J.-H.; Gepperth, D.; Lastusaari, M.; Xu, C.; Hupa, L. Biocomposites of Copper-Containing Mesoporous Bioactive Glass and Nanofibrillated Cellulose: Biocompatibility and Angiogenic Promotion in Chronic Wound Healing Application. Acta Biomater. 2016, 46, 286–298. [Google Scholar] [CrossRef]
- Romero-Sánchez, L.B.; Marí-Beffa, M.; Carrillo, P.; Medina, M.Á.; Díaz-Cuenca, A. Copper-Containing Mesoporous Bioactive Glass Promotes Angiogenesis in an in Vivo Zebrafish Model. Acta Biomater. 2018, 68, 272–285. [Google Scholar] [CrossRef]
- de Laia, A.G.S.; Valverde, T.M.; Barrioni, B.R.; Cunha, P.d.S.; de Goes, A.M.; de Miranda, M.C.; Gomes, D.A.; Queiroz-Junior, C.M.; de Sá, M.A.; de Magalhães Pereira, M. Cobalt-Containing Bioactive Glass Mimics Vascular Endothelial Growth Factor A and Hypoxia Inducible Factor 1 Function. J. Biomed. Mater. Res. A 2021, 109, 1051–1064. [Google Scholar] [CrossRef]
- Solanki, A.K.; Lali, F.V.; Autefage, H.; Agarwal, S.; Nommeots-Nomm, A.; Metcalfe, A.D.; Stevens, M.M.; Jones, J.R. Bioactive Glasses and Electrospun Composites That Release Cobalt to Stimulate the HIF Pathway for Wound Healing Applications. Biomater. Res. 2021, 25, 1. [Google Scholar] [CrossRef]
- Cattalini, J.P.; Hoppe, A.; Pishbin, F.; Roether, J.; Boccaccini, A.R.; Lucangioli, S.; Mouriño, V. Novel Nanocomposite Biomaterials with Controlled Copper/Calcium Release Capability for Bone Tissue Engineering Multifunctional Scaffolds. J. R Soc. Interface 2015, 12, 0509. [Google Scholar] [CrossRef]
- Chen, S.; Michálek, M.; Galusková, D.; Michálková, M.; Švančárek, P.; Talimian, A.; Kaňková, H.; Kraxner, J.; Zheng, K.; Liverani, L.; et al. Multi-Targeted B and Co Co-Doped 45S5 Bioactive Glasses with Angiogenic Potential for Bone Regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 112, 110909. [Google Scholar] [CrossRef]
- Bordoni, B.; Mahabadi, N.; Varacallo, M. Anatomy, Fascia. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Pirri, C.; Fede, C.; Petrelli, L.; Guidolin, D.; Fan, C.; De Caro, R.; Stecco, C. Elastic Fibres in the Subcutaneous Tissue: Is There a Difference between Superficial and Muscular Fascia? A Cadaver Study. Ski. Res. Technol. 2022, 28, 21–27. [Google Scholar] [CrossRef]
- Blasi, M.; Blasi, J.; Domingo, T.; Pérez-Bellmunt, A.; Miguel-Pérez, M. Anatomical and Histological Study of Human Deep Fasciae Development. Surg. Radiol. Anat. 2015, 37, 571–578. [Google Scholar] [CrossRef]
- Stecco, C.; Sfriso, M.M.; Porzionato, A.; Rambaldo, A.; Albertin, G.; Macchi, V.; De Caro, R. Microscopic Anatomy of the Visceral Fasciae. J. Anat. 2017, 231, 121–128. [Google Scholar] [CrossRef]
- Ibrahim, M.M. Subcutaneous and Visceral Adipose Tissue: Structural and Functional Differences. Obes. Rev. 2010, 11, 11–18. [Google Scholar] [CrossRef]
| Scaffold Types | Advantages | Disadvantages | Future Prospects |
|---|---|---|---|
| Porous scaffolds | High porosity provides a suitable environment for extracellular matrix (ECM) secretion and nutrient supplies to the cells. Pore sizes specific to the cell types prevent clustering of the cells, thus avoiding necrotic center formation. | Time consuming post-manufacturing cell inoculation, low cell viability, and high cost. | Improvement in the connectivity of pores and thereby the structure of the scaffolds is required. |
| Fibrous scaffolds | Highly microporous structure is best suitable for cell adhesion, proliferation, and differentiation. Low inflammatory response upon implantation. | Surface functionalization is required to create the nanofibers of these scaffolds. | Drugs and biological molecules such as proteins, genes, growth factors, etc., can be incorporated in fibrous scaffolds for release applications. |
| Hydrogel scaffolds | Highly biocompatible and controlled biodegradation rate. | Limited mechanical strength due to soft structures. | Degradation behavior of the hydrogels and tenability should be well-defined. Hydrogels incorporating growth factors to facilitate cell differentiation. |
| Microsphere scaffolds | Easily fabricated with controlled physical characteristics suitable for slow or fast drug delivery. Provides enhanced cell attachment and migration properties. | Microsphere sintering methods are sometimes not compatible to the cells and reduces the cell viability. | These scaffolds can be used as a target specific delivery vehicle for the drugs such as antibiotics, anti-cancer, etc. |
| Composite scaffolds | Highly biodegradable and offer mechanical strength. Greater absorbability. | Acidic byproducts are generated upon degradation. Poor cell affinity. Require tedious efforts to develop composite scaffolds. | Nano-bioceramic and polymer composites with faster degradation are currently being developed. |
| Acellular scaffolds | Native ECM is retained, and thus normal anatomical features are maintained. Less inflammatory and immune response with higher mechanical strength. | Incomplete decellularization is required to avoid immune responses. | Such scaffolds hold promise towards developing artificial organs. |
| Extracellular Matrix-Based Scaffolds | Retains native ECM, less inflammation and immune response. | Rapid degradation and poor mechanical properties. | Scaffold cross-linking, blending with other biomaterials, and the addition of bioactive substances to create multifunctional scaffolds for ECM. |
| Porous Scaffolds | Porous scaffolds have excellent load-carrying capacity, many of which can payload cargos with relatively large sizes. | Time consuming post-manufacturing cell inoculation, low cell viability, and high cost. | There is a need to improve the connectivity of the pores and the structure of the scaffolds. |
| Fibrous Scaffolds | Suitable for cell adhesion, proliferation, and differentiation with low inflammatory response, giving the cells their typical in vivo morphology. | Surface functionalization required. | Hybrid fiber scaffolds with enhanced properties (biomechanical, physico-chemical and biological) need to be developed. |
| Microsphere Scaffolds | With spatial expansion, temporal duration control and site targeting. | Reduces cell viability and has certain incompatibility with cells. | Development of an efficient drug delivery system. |
| Hydrogel Scaffolds | Hydrogel scaffolds are highly hydrophilic, flexible, biocompatible, and biodegradable. | Limited mechanical properties, difficult purification and sometimes pathogenic transmission and immunogenicity. | Development of a “smart” 4D hydrogel. |
| Nanoparticle scaffolds | Nanoparticle scaffolds have high mechanical properties, antibacterial ability. | Nanoparticle scaffolds may be toxic, cancerous, and teratogenic. | More efficient and safe nanoparticle scaffolds to be developed. |
| Polymer Scaffolds | Better biocompatibility, reproducible mechanical properties, processability, and low price. | Sometimes triggers an immune response and toxicity and poor cell affinity. Require tedious efforts to develop composite scaffolds. | Faster and safer polymer scaffolds are currently being developed. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, H.; Rinkevich, Y. Fascia Layer—A Novel Target for the Application of Biomaterials in Skin Wound Healing. Int. J. Mol. Sci. 2023, 24, 2936. https://doi.org/10.3390/ijms24032936
Ye H, Rinkevich Y. Fascia Layer—A Novel Target for the Application of Biomaterials in Skin Wound Healing. International Journal of Molecular Sciences. 2023; 24(3):2936. https://doi.org/10.3390/ijms24032936
Chicago/Turabian StyleYe, Haifeng, and Yuval Rinkevich. 2023. "Fascia Layer—A Novel Target for the Application of Biomaterials in Skin Wound Healing" International Journal of Molecular Sciences 24, no. 3: 2936. https://doi.org/10.3390/ijms24032936
APA StyleYe, H., & Rinkevich, Y. (2023). Fascia Layer—A Novel Target for the Application of Biomaterials in Skin Wound Healing. International Journal of Molecular Sciences, 24(3), 2936. https://doi.org/10.3390/ijms24032936








