Master-Key Regulators of Sex Determination in Fish and Other Vertebrates—A Review
Abstract
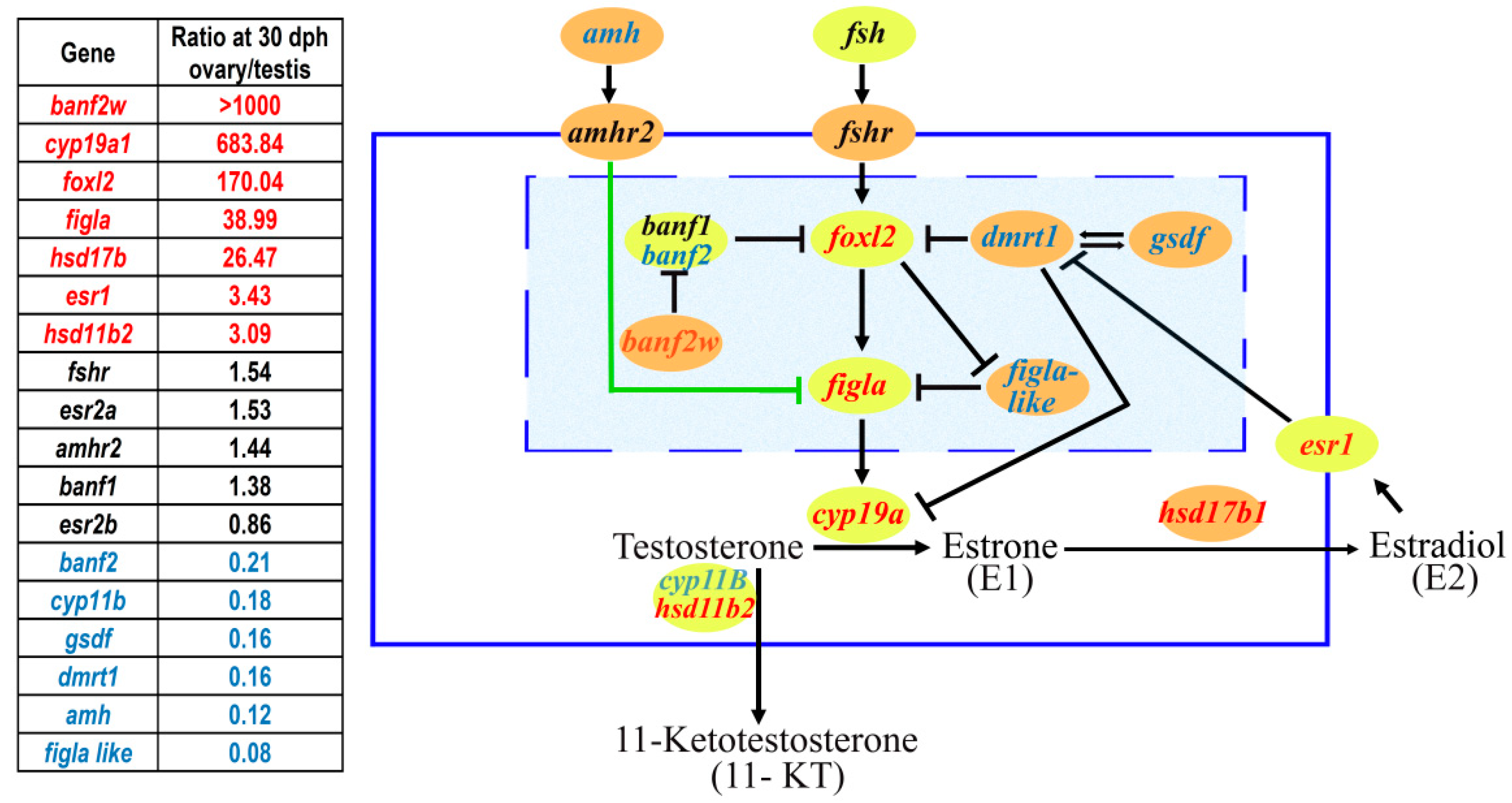
1. Sex Determination
| Human Ortho/Paralog | SD System | MKR | Organism/Species | Type 1 | References/ Chr 2 |
|---|---|---|---|---|---|
| SRY-Box Transcription Factor 3 (SOX3) | XX/XY | sox3Y sry | Oryzias dancena, O. marmoratus, O. profundicola | SV | [26,38]/LG10 |
| mammals (most) | MSD | [39,40]/ChrY | |||
| Anti-Müllerian Hormone Receptor 2 (AMHR2) | XX/XY | amhr2y | Takifugu rubripes, Plecoglossus altivelis | SV | [41,42]/Chr19 |
| Perca flavescens, Phyllopteryx taeniolatus, Syngnathoides biaculeatus, Pangasiidae | MSD | [43]/Chr4 [44]/Chr9 [45,46]/Chr4 | |||
| no ortholog | XX/XY | gsdfY | Oryzias luzonensis, Hippoglossus hippoglossus, Anoplopoma fimbria | SV | [47]/LG12 [48]/Chr13 [49]/LG14 |
| Hydroxysteroid 17-Beta Dehydrogenase 1 (HSD17B1) | WZ/ZZ | hsd17b1w | Seriola genus, Trachinotus anak | SV | [50,51]/Chr24 |
| Bone Morphogenetic Protein Receptor Type 1B (BMPR1B) | XX/XY | bmpr1bbY | Clupea harengus | MSD | [52]/Chr8 |
| Double-sex and Mab-3 Related Transcription Factor 1 (DMRT1) | XX/XY | dmrt1Y dmrt1bY dmW dmrt1z | Oryzias latipes | MSD | [53,54]/LG1 [55,56]/ChrW [57,58,59] |
| XX/XY | Scatophagus argus | ||||
| WZ/ZZ | Xenopus laevis (amphibians) | FSD | |||
| WZ/ZZ | Gallus gallus (birds), Cynoglossus semilaevis | ||||
| Interferon Regulatory Factor 9 (IRF9) | XX/XY | sdY | Salmonids | MSD | [21]/BT18, RT01 |
| Growth Differentiation Factor 6 (GDF6) | XX/XY | gdf6aY gdf6b | Nothobranchius furzeri, Astyanax mexicanus | SV | [60,61]/ChrB |
| no ortholog | XX/XY | zkY | Gadus morhua | MSD | [62]/LG11 |
| Anti-Müllerian Hormone (AMH) | XX/XY | amhΔy | Odontesthes hatcheri, Oreochromis niloticus, Ophiodon elongates, Hypoatherina tsurugae, Esox lucius, Gasterosteus aculeatus, Sebastes schlegelii, Culaea inconstans | MSD | [63] [64]/LG23 [65,66] [67]/LG24 [68,69] [70]/Chr10 |
| Barrier- to-Autointegration- Factor-like protein 2 (BANF2) | WZ/ZZ | banf2w | Oreochromis aureus, Oreochromis urolepis hornorum, Pelmatolapia mariae | FSD | [71]/LG3 |
| Follicle Stimulating Hormone Receptor (FSHR) | XX/XY | fshry | Mugil cephalus | SV | [9]/LG9 |
| Folliculogenesis Specific BHLH Transcription Factor (FIGLA) | XX/XY | figla -like | Oreochromis mossambicus, Coptodon zillii, Sarotherodon galilaeus | MSD | [36]/LG1 |
2. Search for MKRs
3. Identification of SD MKRs
4. From Identification to Validation of MKRs of SD
5. The Significance of Taxonomic Authentication of Species Using DNA Barcoding for MKRs Search
6. Single vs. Polygenic SD Systems
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- West, S.A.; Reece, S.E.; Sheldon, B.C. Sex ratios. Heredity 2002, 88, 117–124. [Google Scholar] [CrossRef]
- West, S.A.; Sheldon, B.C. Constraints in the evolution of sex ratio adjustment. Science 2002, 295, 1685–1688. [Google Scholar] [CrossRef] [PubMed]
- Sarre, S.D.; Georges, A.; Quinn, A. The ends of a continuum: Genetic and temperature-dependent sex determination in reptiles. BioEssays 2004, 26, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, I.; Modi, D. Sex Determination in Teleost Fish. In Recent Updates in Molecular Endocrinology and Reproductive Physiology of Fish; Springer: Singapore, 2021; pp. 121–138. [Google Scholar]
- Harrison, D.A. Sex Determination: Controlling the master. Curr. Biol. 2007, 17, R328–R330. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Sun, Y.; Zhao, J.; Shi, H.; Zeng, S.; Ye, K.; Jiang, D.; Zhou, L.; Sun, L.; Tao, W.; et al. A tandem duplicate of Anti-Müllerian hormone with a missense SNP on the Y chromosome is essential for male sex determination in Nile tilapia, Oreochromis niloticus. PLoS Genet. 2015, 11, e1005678. [Google Scholar] [CrossRef]
- Matsuda, M.; Sakaizumi, M. Evolution of the sex-determining gene in the teleostean genus Oryzias. Gen. Comp. Endocrinol. 2016, 239, 80–88. [Google Scholar] [CrossRef]
- Gammerdinger, W.J.; Kocher, T.D. Unusual diversity of sex chromosomes in african cichlid fishes. Genes 2018, 9, 480. [Google Scholar] [CrossRef]
- Curzon, A.Y.; Dor, L.; Shirak, A.; Meiri-Ashkenazi, I.; Rosenfeld, H.; Ron, M.; Seroussi, E. A novel c.1759T>G variant in follicle-stimulating hormone-receptor gene is concordant with male determination in the flathead grey mullet (Mugil cephalus). G3 Genes Genomes Genet. 2021, 11, jkaa044. [Google Scholar] [CrossRef]
- Göth, A.; Booth, D.T. Temperature-dependent sex ratio in a bird. Biol. Lett. 2005, 1, 31–33. [Google Scholar] [CrossRef]
- Shirak, A.; Golik, M.; Lee, B.-Y.; Howe, A.E.; Kocher, T.D.; Hulata, G.; Ron, M.; Seroussi, E. Copy number variation of lipocalin family genes for male-specific proteins in tilapia and its association with gender. Heredity 2008, 101, 405–415. [Google Scholar] [CrossRef]
- Shirak, A.; Palti, Y.; Cnaani, A.; Korol, A.; Hulata, G.; Ron, M.; Avtalion, R.R. Association between loci with deleterious alleles and distorted sex ratios in an inbred line of tilapia (Oreochromis aureus). J. Hered. 2002, 93, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Marshall Graves, J.A.; Peichel, C.L. Are homologies in vertebrate sex determination due to shared ancestry or to limited options? Genome Biol. 2010, 11, 205. [Google Scholar] [CrossRef] [PubMed]
- Marshall Graves, J.A. How to evolve new vertebrate sex determining genes. Dev. Dyn. 2013, 242, 354–359. [Google Scholar] [CrossRef]
- Capel, B. Vertebrate sex determination: Evolutionary plasticity of a fundamental switch. Nat. Rev. Genet. 2017, 18, 675–689. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Capel, B. Balancing the bipotential gonad between alternative organ fates: A new perspective on an old problem. Dev. Dyn. 2006, 235, 2292–2300. [Google Scholar] [CrossRef]
- Li, M.; Sun, L.; Wang, D. Roles of estrogens in fish sexual plasticity and sex differentiation. Gen. Comp. Endocrinol. 2019, 277, 9–16. [Google Scholar] [CrossRef]
- Tenugu, S.; Senthilkumaran, B. Sexual plasticity in bony fishes: Analyzing morphological to molecular changes of sex reversal. Aquac. Fish. 2022, 7, 525–539. [Google Scholar] [CrossRef]
- Turner, M.E.; Ely, D.; Prokop, J.; Milsted, A. Sry, more than testis determination? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 301, R561–R571. [Google Scholar] [CrossRef]
- Yano, A.; Guyomard, R.; Nicol, B.; Jouanno, E.; Quillet, E.; Klopp, C.; Cabau, C.; Bouchez, O.; Fostier, A.; Guiguen, Y. An immune-related gene evolved into the master sex-determining gene in rainbow trout, Oncorhynchus mykiss. Curr. Biol. 2012, 22, 1423–1428. [Google Scholar] [CrossRef]
- Yano, A.; Nicol, B.; Jouanno, E.; Quillet, E.; Fostier, A.; Guyomard, R.; Guiguen, Y. The sexually dimorphic on the Y-chromosome gene (sdY) is a conserved male-specific Y-chromosome sequence in many salmonids. Evol. Appl. 2013, 6, 486–496. [Google Scholar] [CrossRef]
- Herpin, A.; Schartl, M. Plasticity of gene-regulatory networks controlling sex determination: Of masters, slaves, usual suspects, newcomers, and usurpators. EMBO Rep. 2015, 16, 1260–1274. [Google Scholar] [CrossRef] [PubMed]
- Bertho, S.; Herpin, A.; Branthonne, A.; Jouanno, E.; Yano, A.; Nicol, B.; Muller, T.; Pannetier, M.; Pailhoux, E.; Miwa, M.; et al. The unusual rainbow trout sex determination gene hijacked the canonical vertebrate gonadal differentiation pathway. Proc. Natl. Acad. Sci. USA 2018, 115, 12781–12786. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, M.; Ma, H.; Liu, X.; Shi, H.; Li, M.; Wang, D. Mutation of foxl2 or cyp19a1a results in female to male sex reversal in XX Nile Tilapia. Endocrinology 2017, 158, 2634–2647. [Google Scholar] [CrossRef] [PubMed]
- Fernandes-Freitas, I.; Milona, A.; Murphy, K.G.; Dhillo, W.S.; Owen, B.M. Live birth in sex-reversed XY mice lacking the nuclear receptor Dax1. Sci. Rep. 2020, 10, 1703. [Google Scholar] [CrossRef]
- Myosho, T.; Takehana, Y.; Hamaguchi, S.; Sakaizumi, M. Turnover of Sex Chromosomes in Celebensis Group Medaka Fishes. G3 Genes Genomes Genet. 2015, 5, 2685–2691. [Google Scholar] [CrossRef]
- Curzon, A.Y.; Shirak, A.; Zak, T.; Dor, L.; Benet-Perlberg, A.; Naor, A.; Low-Tanne, S.I.; Sharkawi, H.; Ron, M.; Seroussi, E. All-male production by marker-assisted selection for sex determining loci of admixed Oreochromis niloticus and Oreochromis aureus stocks. Anim. Genet. 2021, 52, 361–364. [Google Scholar] [CrossRef]
- Li, M.; Dai, S.; Liu, X.; Xiao, H.; Wang, D. A detailed procedure for CRISPR/Cas9-mediated gene editing in tilapia. Hydrobiologia 2021, 848, 3865–3881. [Google Scholar] [CrossRef]
- Yan, L.; Feng, H.; Wang, F.; Lu, B.; Liu, X.; Sun, L.; Wang, D. Establishment of three estrogen receptors (esr1, esr2a, esr2b) knockout lines for functional study in Nile tilapia. J. Steroid Biochem. Mol. Biol. 2019, 191, 105379. [Google Scholar] [CrossRef]
- Tao, W.; Xu, L.; Zhao, L.; Zhu, Z.; Wu, X.; Min, Q.; Wang, D.; Zhou, Q. High-quality chromosome-level genomes of two tilapia species reveal their evolution of repeat sequences and sex chromosomes. Mol. Ecol. Resour. 2021, 21, 543–560. [Google Scholar] [CrossRef]
- Adolfi, M.C.; Fischer, P.; Herpin, A.; Regensburger, M.; Kikuchi, M.; Tanaka, M.; Schartl, M. Increase of cortisol levels after temperature stress activates dmrt1a causing female-to-male sex reversal and reduced germ cell number in medaka. Mol. Reprod. Dev. 2019, 86, 1405–1417. [Google Scholar] [CrossRef]
- Franch, R.; Louro, B.; Tsalavouta, M.; Chatziplis, D.; Tsigenopoulos, C.S.; Sarropoulou, E.; Antonello, J.; Magoulas, A.; Mylonas, C.C.; Babbucci, M.; et al. A genetic linkage map of the hermaphrodite teleost fish Sparus aurata L. Genetics 2006, 174, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Dor, L.; Shirak, A.; Gorshkov, S.; Ron, M.; Hulata, G. Development of genetic markers for the white grouper (Epinephelus aeneus). Aquaculture 2014, 420–421, S104–S110. [Google Scholar] [CrossRef]
- Baroiller, J.F.; D’Cotta, H.; Bezault, E.; Wessels, S.; Hoerstgen-Schwark, G. Tilapia sex determination: Where temperature and genetics meet. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2009, 153, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Taslima, K.; Khan, M.G.Q.; McAndrew, B.J.; Penman, D.J. Evidence of two XX/XY sex-determining loci in the Stirling stock of Nile tilapia (Oreochromis niloticus). Aquaculture 2021, 532, 735995. [Google Scholar] [CrossRef]
- Curzon, A.Y.; Shirak, A.; Benet-Perlberg, A.; Naor, A.; Low-Tanne, S.I.; Sharkawi, H.; Ron, M.; Seroussi, E. Absence of figla-like gene is concordant with femaleness in cichlids harboring the LG1 sex-determination system. Int. J. Mol. Sci. 2022, 23, 7636. [Google Scholar] [CrossRef]
- Curzon, A.Y.; Shirak, A.; Dor, L.; Zak, T.; Perelberg, A.; Seroussi, E.; Ron, M. A duplication of the Anti-Müllerian hormone gene is associated with genetic sex determination of different Oreochromis niloticus strains. Heredity 2020, 125, 317–327. [Google Scholar] [CrossRef]
- Takehana, Y.; Matsuda, M.; Myosho, T.; Suster, M.L.; Kawakami, K.; Shin-I, T.; Kohara, Y.; Kuroki, Y.; Toyoda, A.; Fujiyama, A.; et al. Co-option of Sox3 as the male-determining factor on the y chromosome in the fish Oryzias dancena. Nat. Commun. 2014, 5, 4157. [Google Scholar] [CrossRef]
- Gubbay, J.; Collignon, J.; Koopman, P.; Capel, B.; Economou, A.; Münsterberg, A.; Vivian, N.; Goodfellow, P.; Lovell-Badge, R. A gene mapping to the sex-determining region of the mouse Y chromosome is a member of a novel family of embryonically expressed genes. Nature 1990, 346, 245–250. [Google Scholar] [CrossRef]
- Sinclair, A.H.; Berta, P.; Palmer, M.S.; Hawkins, J.R.; Griffiths, B.L.; Smith, M.J.; Foster, J.W.; Frischauf, A.-M.; Lovell-Badge, R.; Goodfellow, P.N. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature 1990, 346, 240–244. [Google Scholar] [CrossRef]
- Kamiya, T.; Kai, W.; Tasumi, S.; Oka, A.; Matsunaga, T.; Mizuno, N.; Fujita, M.; Suetake, H.; Suzuki, S.; Hosoya, S.; et al. A trans-species missense SNP in amhr2 is associated with sex determination in the tiger Pufferfish, Takifugu rubripes (Fugu). PLoS Genet. 2012, 8, e1002798. [Google Scholar] [CrossRef]
- Nakamoto, M.; Uchino, T.; Koshimizu, E.; Kuchiishi, Y.; Sekiguchi, R.; Wang, L.; Sudo, R.; Endo, M.; Guiguen, Y.; Schartl, M.; et al. A Y-linked anti-Müllerian hormone type-II receptor is the sex-determining gene in ayu, Plecoglossus altivelis. PLoS Genet. 2021, 17, e1009705. [Google Scholar] [CrossRef] [PubMed]
- Qu, M.; Liu, Y.; Zhang, Y.; Wan, S.; Ravi, V.; Qin, G.; Jiang, H.; Wang, X.; Zhang, H.; Zhang, B.; et al. Seadragon genome analysis provides insights into its phenotype and sex determination locus. Sci. Adv. 2021, 7, 5196–5214. [Google Scholar] [CrossRef] [PubMed]
- Feron, R.; Zahm, M.; Cabau, C.; Klopp, C.; Roques, C.; Bouchez, O.; Eché, C.; Valière, S.; Donnadieu, C.; Haffray, P.; et al. Characterization of a Y-specific duplication/insertion of the anti-Mullerian hormone type II receptor gene based on a chromosome-scale genome assembly of yellow perch, Perca flavescens. Mol. Ecol. Resour. 2020, 20, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Wen, M.; Pan, Q.; Jouanno, E.; Montfort, J.; Zahm, M.; Cabau, C.; Klopp, C.; Iampietro, C.; Roques, C.; Bouchez, O.; et al. An ancient truncated duplication of the anti-Müllerian hormone receptor type 2 gene is a potential conserved master sex determinant in the Pangasiidae catfish family. Mol. Ecol. Resour. 2022, 22, 2411–2428. [Google Scholar] [CrossRef]
- Nacif, C.L.; Kratochwil, C.F.; Kautt, A.F.; Nater, A.; Machado-Schiaffino, G.; Meyer, A.; Henning, F. Molecular parallelism in the evolution of a master sex-determining role for the anti-Mullerian hormone receptor 2 gene (amhr2) in Midas cichlids. Mol. Ecol. 2022, 1–13. [Google Scholar] [CrossRef]
- Myosho, T.; Otake, H.; Masuyama, H.; Matsuda, M.; Kuroki, Y.; Fujiyama, A.; Naruse, K.; Hamaguchi, S.; Sakaizumi, M. Tracing the emergence of a novel sex-determining gene in medaka, Oryzias luzonensis. Genetics 2012, 191, 163–170. [Google Scholar] [CrossRef]
- Edvardsen, R.B.; Wallerman, O.; Furmanek, T.; Kleppe, L.; Jern, P.; Wallberg, A.; Kjærner-Semb, E.; Mæhle, S.; Olausson, S.K.; Sundström, E.; et al. Heterochiasmy and the establishment of gsdf as a novel sex determining gene in Atlantic halibut. PLoS Genet. 2022, 18, e1010011. [Google Scholar] [CrossRef]
- Herpin, A.; Schartl, M.; Depincé, A.; Guiguen, Y.; Bobe, J.; Hua-Van, A.; Hayman, E.S.; Octavera, A.; Yoshizaki, G.; Nichols, K.M.; et al. Allelic diversification after transposable element exaptation promoted gsdf as the master sex determining gene of sablefish. Genome Res. 2021, 31, 1366–1380. [Google Scholar] [CrossRef]
- Koyama, T.; Nakamoto, M.; Morishima, K.; Yamashita, R.; Yamashita, T.; Sasaki, K.; Kuruma, Y.; Mizuno, N.; Suzuki, M.; Okada, Y.; et al. A SNP in a steroidogenic enzyme is associated with phenotypic sex in seriola fishes. Curr. Biol. 2019, 29, 1901–1909.e8. [Google Scholar] [CrossRef]
- Fan, B.; Xie, D.; Li, Y.; Wang, X.; Qi, X.; Li, S.; Meng, Z.; Chen, X.; Peng, J.; Yang, Y.; et al. A single intronic single nucleotide polymorphism in splicing site of steroidogenic enzyme hsd17b1 is associated with phenotypic sex in oyster pompano, Trachinotus anak. Proc. R. Soc. B 2021, 288, 20212245. [Google Scholar] [CrossRef]
- Rafati, N.; Chen, J.; Herpin, A.; Pettersson, M.E.; Han, F.; Feng, C.; Wallerman, O.; Rubin, C.-J.; Péron, S.; Cocco, A.; et al. Reconstruction of the birth of a male sex chromosome present in Atlantic herring. Proc. Natl. Acad. Sci. USA 2020, 117, 24359–24368. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Nagahama, Y.; Shinomiya, A.; Sato, T.; Matsuda, C.; Kobayashi, T.; Morrey, C.E.; Shibata, N.; Asakawa, S.; Shimizu, N.; et al. DMY is a Y-specific DM-domain gene required for male development in the medaka fish. Nature 2002, 417, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Nanda, I.; Kondo, M.; Hornung, U.; Asakawa, S.; Winkler, C.; Shimizu, A.; Shan, Z.; Haaf, T.; Shimizu, N.; Shima, A.; et al. A duplicated copy of dmrt1 in the sex-determining region of the Y chromosom of the medaka, Oryzias latipes. Proc. Natl. Acad. Sci. USA 2002, 99, 11778–11783. [Google Scholar] [CrossRef]
- Yoshimoto, S.; Okada, E.; Umemoto, H.; Tamura, K.; Uno, Y.; Nishida-Umehara, C.; Matsuda, Y.; Takamatsu, N.; Shiba, T.; Ito, M. A W-linked DM-domain gene, DM-W, participates in primary ovary development in Xenopus laevis. Proc. Natl. Acad. Sci. USA 2008, 105, 2469–2474. [Google Scholar] [CrossRef]
- Smith, C.A.; Roeszler, K.N.; Ohnesorg, T.; Cummins, D.M.; Farlie, P.G.; Doran, T.J.; Sinclair, A.H. The avian Z-linked gene dmrt1 is required for male sex determination in the chicken. Nature 2009, 461, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, G.; Shao, C.; Huang, Q.; Liu, G.; Zhang, P.; Song, W.; An, N.; Chalopin, D.; Volff, J.N.; et al. Whole-genome sequence of a flatfish provides insights into ZW sex chromosome evolution and adaptation to a benthic lifestyle. Nat. Genet. 2014, 46, 253–260. [Google Scholar] [CrossRef]
- Cui, Z.; Liu, Y.; Wang, W.; Wang, Q.; Zhang, N.; Lin, F.; Wang, N.; Shao, C.; Dong, Z.; Li, Y.; et al. Genome editing reveals dmrt1 as an essential male sex-determining gene in Chinese tongue sole (Cynoglossus semilaevis). Sci. Rep. 2017, 7, 42213. [Google Scholar] [CrossRef] [PubMed]
- Mustapha, U.F.; Jiang, D.-N.; Liang, Z.-H.; Gu, H.-T.; Yang, W.; Chen, H.-P.; Deng, S.-P.; Wu, T.-L.; Tian, C.-X.; Zhu, C.-H.; et al. Male-specific dmrt1 is a candidate sex determination gene in spotted scat (Scatophagus argus). Aquaculture 2018, 495, 351–358. [Google Scholar] [CrossRef]
- Reichwald, K.; Petzold, A.; Koch, P.; Downie, B.R.; Hartmann, N.; Pietsch, S.; Baumgart, M.; Chalopin, D.; Felder, M.; Bens, M.; et al. Insights into sex chromosome evolution and aging from the genome of a short-lived fish. Cell 2015, 163, 1527–1538. [Google Scholar] [CrossRef]
- Imarazene, B.; Du, K.; Beille, S.; Jouanno, E.; Feron, R.; Pan, Q.; Torres-Paz, J.; Lopez-Roques, C.; Castinel, A.; Gil, L.; et al. A supernumerary “B-sex” chromosome drives male sex determination in the Pachón cavefish, Astyanax mexicanus. Curr. Biol. 2021, 31, 4800–4809.e9. [Google Scholar] [CrossRef]
- Kirubakaran, T.G.; Andersen, Ø.; de Rosa, M.C.; Andersstuen, T.; Hallan, K.; Kent, M.P.; Lien, S. Characterization of a male specific region containing a candidate sex determining gene in Atlantic cod. Sci. Rep. 2019, 9, 116. [Google Scholar] [CrossRef] [PubMed]
- Hattori, R.S.; Murai, Y.; Oura, M.; Masuda, S.; Majhi, S.K.; Sakamoto, T.; Fernandino, J.I.; Somoza, G.M.; Yokota, M.; Strussmann, C.A. A Y-linked anti-Mullerian hormone duplication takes over a critical role in sex determination. Proc. Natl. Acad. Sci. USA 2012, 109, 2955–2959. [Google Scholar] [CrossRef] [PubMed]
- Eshel, O.; Shirak, A.; Dor, L.; Band, M.; Zak, T.; Markovich-Gordon, M.; Chalifa-Caspi, V.; Feldmesser, E.; Weller, J.I.; Seroussi, E.; et al. Identification of male-specific amh duplication, sexually differentially expressed genes and microRNAs at early embryonic development of Nile tilapia (Oreochromis niloticus). BMC Genom. 2014, 15, 774. [Google Scholar] [CrossRef] [PubMed]
- Rondeau, E.B.; Laurie, C.V.; Johnson, S.C.; Koop, B.F. A PCR assay detects a male-specific duplicated copy of Anti-Müllerian hormone (amh) in the lingcod (Ophiodon elongatus). BMC Res. Notes 2016, 9, 230. [Google Scholar] [CrossRef]
- Bej, D.K.; Miyoshi, K.; Hattori, R.S.; Strüssmann, C.A.; Yamamoto, Y. A duplicated, truncated amh gene is involved in male sex determination in an old world Silverside. G3 Genes Genomes Genet. 2017, 7, 2489–2495. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Feron, R.; Yano, A.; Guyomard, R.; Jouanno, E.; Vigouroux, E.; Wen, M.; Busne, J.M.; Bobe, J.; Concordet, J.P.; et al. Identification of the master sex determining gene in Northern pike (Esox lucius) reveals restricted sex chromosome differentiation. PLoS Genet. 2019, 15, e1008013. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Xie, Y.; Sun, M.; Li, X.; Fitzpatrick, C.K.; Vaux, F.; O’Malley, K.G.; Zhang, Q.; Qi, J.; He, Y. A duplicated amh is the master sex-determining gene for Sebastes rockfish in the Northwest Pacific. Open Biol. 2021, 11, 210063. [Google Scholar] [CrossRef]
- Peichel, C.L.; McCann, S.R.; Ross, J.A.; Naftaly, A.F.S.; Urton, J.R.; Cech, J.N.; Grimwood, J.; Schmutz, J.; Myers, R.M.; Kingsley, D.M.; et al. Assembly of the threespine stickleback Y chromosome reveals convergent signatures of sex chromosome evolution. Genome Biol. 2020, 21, 177. [Google Scholar] [CrossRef] [PubMed]
- Jeffries, D.L.; Mee, J.A.; Peichel, C.L. Identification of a candidate sex determination gene in Culaea inconstans suggests convergent recruitment of an amh duplicate in two lineages of stickleback. J. Evol. Biol. 2022, 35, 1683–1695. [Google Scholar] [CrossRef]
- Curzon, A.Y.; Shirak, A.; Benet-Perlberg, A.; Naor, A.; Low-Tanne, S.I.; Sharkawi, H.; Ron, M.; Seroussi, E. Gene variant of barrier to autointegration factor 2 (banf2w) is concordant with female determination in Cichlids. Int. J. Mol. Sci. 2021, 22, 7073. [Google Scholar] [CrossRef]
- Liu, F.; Sun, F.; Li, J.; Xia, J.H.; Lin, G.; Tu, R.J.; Yue, G.H. A microsatellite-based linkage map of salt tolerant tilapia (Oreochromis mossambicus × Oreochromis spp.) and mapping of sex-determining loci. BMC Genom. 2013, 14, 58. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Niu, Y.; Ye, X.; Dong, J.; Hu, W.; Zeng, Q.; Chen, Z.; Tian, Y.; Zhang, J.; Lu, M. Construction of a high-density linkage map and mapping of sex determination and growth-related loci in the mandarin fish (Siniperca chuatsi). BMC Genom. 2017, 18, 446. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, L.; Lin, H.; Zhou, Z.; Liu, B.; Zhong, J.; Pu, F.; Shi, Y.; Zhou, T.; Xu, P. Genome-wide association study identifies genomic loci of sex determination, gonadal weight and gonadosomatic index traits in Takifugu bimaculatus. Aquaculture 2022, 546, 737389. [Google Scholar] [CrossRef]
- Martínez, P.; Robledo, D.; Taboada, X.; Blanco, A.; Moser, M.; Maroso, F.; Hermida, M.; Gómez-Tato, A.; Álvarez-Blázquez, B.; Cabaleiro, S.; et al. A genome-wide association study, supported by a new chromosome-level genome assembly, suggests sox2 as a main driver of the undifferentiatiated ZZ/ZW sex determination of turbot (Scophthalmus maximus). Genomics 2021, 113, 1705–1718. [Google Scholar] [CrossRef]
- Lv, J.; Sun, D.; Huan, P.; Song, L.; Liu, P.; Li, J. QTL mapping and marker identification for sex-determining: Indicating XY sex determination system in the swimming crab (Portunus trituberculatus). Front. Genet. 2018, 9, 337. [Google Scholar] [CrossRef]
- Feulner, P.G.D.; Schwarzer, J.; Haesler, M.P.; Meier, J.I.; Seehausen, O. A dense linkage map of Lake Victoria cichlids improved the Pundamilia genome assembly and revealed a major QTL for sex-determination. G3 Genes Genomes Genet. 2018, 8, 2411–2420. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, H.; Wang, X.; Fu, B.; Yu, X.; Tong, J. QTL fine mapping for sex determination region in Bighead Carp (Hypophthalmichthys nobilis) and comparison with Silver Carp (iHypophthalmichthys molitrix). Mar. Biotechnol. 2020, 22, 41–53. [Google Scholar] [CrossRef]
- Zhu, Z.X.; Lin, Y.L.; Ai, C.H.; Xiong, Y.Y.; Huang, D.D.; Yao, Y.Y.; Liu, T.D.; Chen, C.H.; Lin, H.R.; Xia, J.H. First identification of two co-existing genome-wide significant sex quantitative trait loci (QTL) in red tilapia using integrative QTL mapping. Zool Res. 2022, 43, 205. [Google Scholar] [CrossRef]
- Bachtrog, D. A dynamic view of sex chromosome evolution. Curr. Opin. Genet. Dev. 2006, 16, 578–585. [Google Scholar] [CrossRef]
- Dor, L.; Shirak, A.; Kohn, Y.Y.; Gur, T.; Weller, J.I.; Zilberg, D.; Seroussi, E.; Ron, M. Mapping of the sex determining region on linkage group 12 of guppy (Poecilia reticulata). G3 Genes Genomes Genet. 2019, 9, 3867–3875. [Google Scholar] [CrossRef]
- Charlesworth, D.; Bergero, R.; Graham, C.; Gardner, J.; Yong, L. Locating the sex determining region of linkage group 12 of guppy (Poecilia reticulata). G3 Genes Genomes Genet. 2020, 10, 3639–3649. [Google Scholar] [CrossRef] [PubMed]
- Kocher, T.D.; Behrens, K.A.; Conte, M.A.; Aibara, M.; Mrosso, H.D.J.; Green, E.C.J.; Kidd, M.R.; Nikaido, M.; Koblmüller, S. New sex chromosomes in Lake Victoria cichlid fishes (Cichlidae: Haplochromini). Genes 2022, 13, 804. [Google Scholar] [CrossRef] [PubMed]
- Duncan, L.E.; Ostacher, M.; Ballon, J. How genome-wide association studies (GWAS) made traditional candidate gene studies obsolete. Neuropsychopharmacology 2019, 44, 1518–1523. [Google Scholar] [CrossRef] [PubMed]
- Altshuler, D.; Daly, M.J.; Lander, E.S. Genetic mapping in human disease. Science 2008, 322, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.W.; Graves, J.A. An SRY-related sequence on the marsupial X chromosome: Implications for the evolution of the mammalian testis-determining gene. Proc. Natl. Acad. Sci. USA 1994, 91, 1927–1931. [Google Scholar] [CrossRef] [PubMed]
- Sreenivasan, R.; Gonen, N.; Sinclair, A. SOX genes and their role in disorders of sex development. Sex Dev. 2022, 16, 80–91. [Google Scholar] [CrossRef]
- Haines, B.; Hughes, J.; Corbett, M.; Shaw, M.; Innes, J.; Patel, L.; Gecz, J.; Clayton-Smith, J.; Thomas, P. Interchromosomal insertional translocation at Xq26.3 alters SOX3 expression in an individual with XX male sex reversal. J. Clin. Endocrinol. Metab. 2015, 100, E815–E820. [Google Scholar] [CrossRef]
- Sutton, E.; Hughes, J.; White, S.; Sekido, R.; Tan, J.; Arboleda, V.; Rogers, N.; Knower, K.; Rowley, L.; Eyre, H.; et al. Identification of SOX3 as an XX male sex reversal gene in mice and humans. J. Clin. Investig. 2011, 121, 328–341. [Google Scholar] [CrossRef]
- Shirak, A.; Seroussi, E.; Cnaani, A.; Howe, A.E.; Domokhovsky, R.; Zilberman, N.; Kocher, T.D.; Hulata, G.; Ron, M. Amh and dmrta2 genes map to tilapia (Oreochromis spp.) linkage group 23 within quantitative trait locus regions for sex determination. Genetics 2006, 174, 1573–1581. [Google Scholar] [CrossRef]
- Guiguen, Y.; Fostier, A.; Piferrer, F.; Chang, C.F. Ovarian aromatase and estrogens: A pivotal role for gonadal sex differentiation and sex change in fish. Gen. Comp. Endocrinol. 2010, 165, 352–366. [Google Scholar] [CrossRef]
- Simpson, E.R.; Mahendroo, M.S.; Means, G.D.; Kilgore, M.W.; Hinshelwood, M.M.; Graham-Lorence, S.; Amarneh, B.; Ito, Y.; Fisher, C.R.; Michael, M.D.; et al. Aromatase cytochrome P450, the enzyme responsible for estrogen biosynthesis. Endocr. Rev. 1994, 15, 342–355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, S.; Lu, H.; Zhang, L.; Zhang, W. Genes encoding aromatases in teleosts: Evolution and expression regulation. Gen. Comp. Endocrinol. 2014, 205, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tang, Y.; Wang, L.; Li, X.; Deng, L.; Deng, W.; Zheng, Y.; Wang, D.; Wei, L. Transcription factor sox3 is required for oogenesis in the teleost fish Nile tilapia. Int. J. Biol. Macromol. 2022, 222, 2639–2647. [Google Scholar] [CrossRef]
- Vining, B.; Ming, Z.; Bagheri-Fam, S.; Harley, V.; Barrionuevo Jimenez, J.; Burgos, M.; Jiménez, R. Diverse regulation but conserved function: SOX9 in vertebrate sex determination. Genes 2021, 12, 486. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Li, X.; Li, M.; Tang, Y.; Wei, J.; Wang, D. Dmrt1 directly regulates the transcription of the testis-biased Sox9b gene in Nile tilapia (Oreochromis niloticus). Gene 2019, 687, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Yang, C.; Tao, W.; Wang, D. Genome-wide identification and transcriptome-based expression profiling of the sox gene family in the Nile tilapia (Oreochromis niloticus). Int. J. Mol. Sci. 2016, 17, 270. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, M.; Suzuki, A.; Matsuda, M.; Nagahama, Y.; Shibata, N. Testicular type sox9 is not involved in sex determination but might be in the development of testicular structures in the medaka, Oryzias latipes. Biochem. Biophys. Res. Commun. 2005, 333, 729–736. [Google Scholar] [CrossRef]
- Heinosalo, T.; Saarinen, N.; Poutanen, M. Role of hydroxysteroid (17beta) dehydrogenase type 1 in reproductive tissues and hormone-dependent diseases. Mol. Cell. Endocrinol. 2019, 489, 9–31. [Google Scholar] [CrossRef]
- Ijiri, S.; Kaneko, H.; Kobayashi, T.; Wang, D.-S.; Sakai, F.; Paul-Prasanth, B.; Nakamura, M.; Nagahama, Y. Sexual dimorphic expression of genes in gonads during early differentiation of a Teleost fish, the Nile tilapia Oreochromis niloticus. Biol. Reprod. 2008, 78, 333–341. [Google Scholar] [CrossRef]
- Hsu, C.W.; Chung, B.C. Evolution, expression, and function of gonadal somatic cell-derived factor. Front. Cell Dev. Biol. 2021, 9, 1600. [Google Scholar] [CrossRef]
- Kaneko, H.; Ijiri, S.; Kobayashi, T.; Izumi, H.; Kuramochi, Y.; Wang, D.S.; Mizuno, S.; Nagahama, Y. Gonadal soma-derived factor (gsdf), a TGF-beta superfamily gene, induces testis differentiation in the teleost fish Oreochromis niloticus. Mol. Cell. Endocrinol. 2015, 415, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.N.; Yang, H.H.; Li, M.H.; Shi, H.J.; Zhang, X.B.; Wang, D.S. Gsdf is a downstream gene of dmrt1 that functions in the male sex determination pathway of the Nile tilapia. Mol. Reprod. Dev. 2016, 83, 497–508. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chang, Y.; Zhai, W.; Qian, F.; Zhang, Y.; Xu, S.; Guo, H.; Wang, S.; Hu, R.; Zhong, X.; et al. A potential role for the Gsdf–eEF1α complex in inhibiting germ cell proliferation: A protein-interaction analysis in Medaka (Oryzias latipes) from a proteomics perspective. Mol. Cell. Proteom. 2021, 20, 100023. [Google Scholar] [CrossRef]
- Jiang, D.N.; Peng, Y.X.; Liu, X.Y.; Mustapha, U.F.; Huang, Y.Q.; Shi, H.J.; Li, M.H.; Li, G.L.; Wang, D.S. Homozygous mutation of gsdf causes infertility in female Nile tilapia (Oreochromis niloticus). Front. Endocrinol. 2022, 13, 135. [Google Scholar] [CrossRef]
- Lu, J.; Li, W.; Hu, R.; Zhou, Y.; Fei, Y.; Zhang, Y.; Zhai, W.; Chen, L. Molecular and morphological changes in Nile tilapia (Oreochromis niloticus) gonads during high-temperature-induced masculinization. Aquac. Res. 2022, 53, 921–931. [Google Scholar] [CrossRef]
- Romano, S.; Kaufman, O.H.; Marlow, F.L. Loss of dmrt1 restores zebrafish female fates in the absence of cyp19a1a but not rbpms2a/b. Development 2020, 147, dev190942. [Google Scholar] [CrossRef]
- Wu, K.; Song, W.; Zhang, Z.; Ge, W. Disruption of dmrt1 rescues the all-male phenotype of the cyp19a1a mutant in zebrafish—A novel insight into the roles of aromatase/estrogens in gonadal differentiation and early folliculogenesis. Development 2020, 147, dev182758. [Google Scholar] [CrossRef]
- Webster, K.A.; Schach, U.; Ordaz, A.; Steinfeld, J.S.; Draper, B.W.; Siegfried, K.R. Dmrt1 is necessary for male sexual development in zebrafish. Dev. Biol. 2017, 422, 33–46. [Google Scholar] [CrossRef]
- Liu, X.; Dai, S.; Wu, J.; Wei, X.; Zhou, X.; Chen, M.; Tan, D.; Pu, D.; Li, M.; Wang, D. Roles of anti-Müllerian hormone and its duplicates in sex determination and germ cell proliferation of Nile tilapia. Genetics 2022, 220, iyab237. [Google Scholar] [CrossRef]
- Wang, D.S.; Zhou, L.Y.; Kobayashi, T.; Matsuda, M.; Shibata, Y.; Sakai, F.; Nagahama, Y. Doublesex- and mab-3-related transcription factor-1 repression of aromatase transcription, a possible mechanism favoring the male pathway in tilapia. Endocrinology 2010, 151, 1331–1340. [Google Scholar] [CrossRef]
- Dai, S.; Qi, S.; Wei, X.; Liu, X.; Li, Y.; Zhou, X.; Xiao, H.; Lu, B.; Wang, D.; Li, M. Germline sexual fate is determined by the antagonistic action of dmrt1 and foxl3 /foxl2 in tilapia. Development 2021, 148, dev199380. [Google Scholar] [CrossRef]
- Kim, S.Y.; Weiss, J.; Tong, M.; Laronda, M.M.; Lee, E.J.; Jameson, J.L. Foxl2, a forkhead transcription factor, modulates nonclassical activity of the estrogen receptor-α. Endocrinology 2009, 150, 5085–5093. [Google Scholar] [CrossRef]
- Georges, A.; L’Hôte, D.; Todeschini, A.L.; Auguste, A.; Legois, B.; Zider, A.; Veitia, R.A. The transcription factor FOXL2 mobilizes estrogen signaling to maintain the identity of ovarian granulosa cells. Elife 2014, 3, 1–19. [Google Scholar] [CrossRef]
- Tang, Y.; Li, X.; Xiao, H.; Li, M.; Li, Y.; Wang, D.; Wei, L. Transcription of the sox30 Gene Is Positively Regulated by dmrt1 in Nile Tilapia. Int. J. Mol. Sci. 2019, 20, 5487. [Google Scholar] [CrossRef]
- Sacchi, S.; D’Ippolito, G.; Sena, P.; Marsella, T.; Tagliasacchi, D.; Maggi, E.; Argento, C.; Tirelli, A.; Giulini, S.; la Marca, A. The anti-Müllerian hormone (AMH) acts as a gatekeeper of ovarian steroidogenesis inhibiting the granulosa cell response to both FSH and LH. J. Assist. Reprod. Genet. 2016, 33, 95–100. [Google Scholar] [CrossRef]
- Qiang, J.; Cao, Z.-M.; Zhu, H.-J.; Tao, Y.-F.; He, J.; Xu, P. Knock-down of amh transcription by antisense RNA reduces FSH and increases follicular atresia in female Oreochromis niloticus. Gene 2022, 842, 146792. [Google Scholar] [CrossRef]
- Yan, H.; Ijiri, S.; Wu, Q.; Kobayashi, T.; Li, S.; Nakaseko, T.; Adachi, S.; Nagahama, Y. Expression patterns of gonadotropin hormones and their receptors during early sexual differentiation in nile tilapia Oreochromis niloticus. Biol. Reprod. 2012, 87, 116–117. [Google Scholar] [CrossRef]
- Qiu, Y.; Sun, S.; Charkraborty, T.; Wu, L.; Sun, L.; Wei, J.; Nagahama, Y.; Wang, D.; Zhou, L. Figla favors ovarian differentiation by antagonizing spermatogenesis in a teleosts, Nile Tilapia (Oreochromis niloticus). PLoS ONE 2015, 10, e0123900. [Google Scholar] [CrossRef]
- Zhou, L.; Qiu, Y. Functional studies of Figla in the ovarian differentiation and maintenance in Nile tilapia (Orechromis niloticus). J. Fish China 2016, 40, 665–672. [Google Scholar]
- Li, H.; Xu, W.; Zhang, N.; Shao, C.; Zhu, Y.; Dong, Z.; Wang, N.; Jia, X.; Xu, H.; Chen, S. Two Figla homologues have disparate functions during sex differentiation in half-smooth tongue sole (Cynoglossus semilaevis). Sci. Rep. 2016, 6, 28219. [Google Scholar] [CrossRef]
- Margalit, A.; Segura-Totten, M.; Gruenbaum, Y.; Wilson, K.L. Barrier-to-autointegration factor is required to segregate and enclose chromosomes within the nuclear envelope and assemble the nuclear lamina. Proc. Natl. Acad. Sci. USA 2005, 102, 3290–3295. [Google Scholar] [CrossRef] [PubMed]
- Tifft, K.E.; Segura-Totten, M.; Lee, K.K.; Wilson, K.L. Barrier-to-autointegration factor-like (BAF-L): A proposed regulator of BAF. Exp. Cell Res. 2006, 312, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Penrad-Mobayed, M.; Perrin, C.; Herman, L.; Todeschini, A.; Nigon, F.; Cosson, B.; Caburet, S.; Veitia, R.A. Conventional and unconventional interactions of the transcription factor FOXL2 uncovered by a proteome-wide analysis. FASEB J. 2020, 34, 571–587. [Google Scholar] [CrossRef] [PubMed]
- L’Hôte, D.; Georges, A.; Todeschini, A.L.; Kim, J.H.; Benayoun, B.A.; Bae, J.; Veitia, R.A. Discovery of novel protein partners of the transcription factor FOXL2 provides insights into its physiopathological roles. Hum. Mol. Genet. 2012, 21, 3264–3274. [Google Scholar] [CrossRef] [PubMed]
- Ron, M.; Weller, J.I. From QTL to QTN identification in livestock—Winning by points rather than knock-out: A review. Anim. Genet. 2007, 38, 429–439. [Google Scholar] [CrossRef]
- Mackay, T.F.C. The genetic architecture of quantitative traits. Annu. Rev. Genet. 2001, 35, 303–339. [Google Scholar] [CrossRef]
- Dor, L.; Shirak, A.; Curzon, A.Y.; Rosenfeld, H.; Ashkenazi, I.M.; Nixon, O.; Seroussi, E.; Weller, J.I.; Ron, M. Preferential mapping of sex-biased differentially-expressed genes of larvae to the sex-determining region of Flathead Grey Mullet (Mugil cephalus). Front. Genet. 2020, 11, 839. [Google Scholar] [CrossRef]
- Kuhl, H.; Guiguen, Y.; Höhne, C.; Kreuz, E.; Du, K.; Klopp, C.; Lopez-Roques, C.; Yebra-Pimentel, E.S.; Ciorpac, M.; Gessner, J.; et al. A 180 Myr-old female-specific genome region in sturgeon reveals the oldest known vertebrate sex determining system with undifferentiated sex chromosomes. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2021, 376, 20200089. [Google Scholar] [CrossRef]
- Curzon, A.Y.; Shirak, A.; Meerson, A.; Degani, G.; Hurvitz, A.; Ben-Naim, N.; Domovitz, R.; Ron, M.; Seroussi, E. Cross-species conservation of a transposase-linked element enables genetic sexing of commercial populations of Russian sturgeon (Acipenser gueldenstaedtii). Anim. Genet. 2022, 53, 441–446. [Google Scholar] [CrossRef]
- Bao, L.; Tian, C.; Liu, S.; Zhang, Y.; Elaswad, A.; Yuan, Z.; Khalil, K.; Sun, F.; Yang, Y.; Zhou, T.; et al. The Y chromosome sequence of the channel catfish suggests novel sex determination mechanisms in teleost fish. BMC Biol. 2019, 17, 6. [Google Scholar] [CrossRef]
- Ventura, T.; Sagi, A. The insulin-like androgenic gland hormone in crustaceans: From a single gene silencing to a wide array of sexual manipulation-based biotechnologies. Biotechnol. Adv. 2012, 30, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Gonen, N.; Futtner, C.R.; Wood, S.; Alexandra Garcia-Moreno, S.; Salamone, I.M.; Samson, S.C.; Sekido, R.; Poulat, F.; Maatouk, D.M.; Lovell-Badge, R. Sex reversal following deletion of a single distal enhancer of Sox9. Science 2018, 360, 1469–1471. [Google Scholar] [CrossRef]
- Eshel, O.; Shirak, A.; Weller, J.I.; Slossman, T.; Hulata, G.; Cnaani, A.; Ron, M. Fine-mapping of a locus on linkage group 23 for sex determination in Nile tilapia (Oreochromis niloticus). Anim. Genet. 2011, 42, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Eshel, O.; Shirak, A.; Weller, J.I.; Hulata, G.; Ron, M. Linkage and physical mapping of sex region on LG23 of Nile tilapia (Oreochromis niloticus). G3 Genes Genomes Genet. 2012, 2, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Loehlin, D.W.; Carroll, S.B. Expression of tandem gene duplicates is often greater than twofold. Proc. Natl. Acad. Sci. USA 2016, 113, 5988–5992. [Google Scholar] [CrossRef]
- Loehlin, D.W.; Kim, J.Y.; Paster, C.O. A tandem duplication in Drosophila melanogaster shows enhanced expression beyond the gene copy number. Genetics 2022, 220, iyab231. [Google Scholar] [CrossRef]
- Ridnik, M.; Schoenfelder, S.; Gonen, N. Cis-regulatory control of mammalian sex determination. Sex Dev. 2021, 15, 317–334. [Google Scholar] [CrossRef]
- Triay, C.; Conte, M.A.; Baroiller, J.F.; Bezault, E.; Clark, F.E.; Penman, D.J.; Kocher, T.D.; D’cotta, H. Structure and sequence of the sex determining locus in two wild populations of Nile tilapia. Genes 2020, 11, 1017. [Google Scholar] [CrossRef]
- Triay, C.; Courcelle, M.; Caminade, P.; Bezault, E.; Baroiller, J.-F.; Kocher, T.D.; D’Cotta, H. Polymorphism of sex determination amongst wild populations suggests its rapid turnover within the Nile tilapia species. Front. Genet. 2022, 13, 345. [Google Scholar] [CrossRef]
- Landi, M.; Dimech, M.; Arculeo, M.; Biondo, G.; Martins, R.; Carneiro, M.; Carvalho, G.R.; Lo Brutto, S.; Costa, F.O. DNA Barcoding for species assignment: The case of Mediterranean marine fishes. PLoS ONE 2014, 9, e106135. [Google Scholar] [CrossRef]
- Lowenstein, J.H.; Amato, G.; Kolokotronis, S.O. The real maccoyii: Identifying Tuna Sushi with DNA barcodes—Contrasting characteristic attributes and genetic distances. PLoS ONE 2009, 4, e7866. [Google Scholar] [CrossRef]
- Shirak, A.; Cohen-Zinder, M.; Barroso, R.M.; Seroussi, E.; Ron, M.; Hulata, G. DNA barcoding of Israeli indigenous and introduced cichlids. Isr. J. Aquac. 2009, 61, 83–88. [Google Scholar]
- Zhang, J.; Hanner, R. Molecular approach to the identification of fish in the South China sea. PLoS ONE 2012, 7, e30621. [Google Scholar] [CrossRef]
- Shirak, A.; Dor, L.; Seroussi, E.; Ron, M.; Hulata, G.; Golani, D. DNA barcoding of fish species from the Mediterranean coast of Israel. Mediterr. Mar. Sci. 2016, 17, 459–466. [Google Scholar] [CrossRef]
- Keskİn, E.; Atar, H.H. DNA barcoding commercially important fish species of Turkey. Mol. Ecol. Resour. 2013, 13, 788–797. [Google Scholar] [CrossRef] [PubMed]
- Curzon, A.Y.; Shirak, A.; Dor, L.; Zak, T.; Benet-Perelberg, A.; Seroussi, E.; Ron, M. Hybrid origin of the Thai-Chitralada tilapia strain using DNA barcoding and microsatellite analysis. Isr. J. Aquac. 2019, 71, 20988. [Google Scholar] [CrossRef]
- Geraerts, M.; Vangestel, C.; Artois, T.; de Oliveira Fernandes, J.M.; Jorissen, M.W.P.; Chocha Manda, A.; Danadu Mizani, C.; Smeets, K.; Snoeks, J.; Sonet, G.; et al. Population genomics of introduced Nile tilapia Oreochromis niloticus (Linnaeus, 1758) in the Democratic Republic of the Congo: Repeated introductions since colonial times with multiple sources. Mol. Ecol. 2022, 31, 3304–3322. [Google Scholar] [CrossRef]
- Blackwell, T.; Ford, A.G.P.; Ciezarek, A.G.; Bradbeer, S.J.; Gracida Juarez, C.A.; Smith, A.M.; Ngatunga, B.P.; Shechonge, A.; Tamatamah, R.; Etherington, G.; et al. Newly discovered cichlid fish biodiversity threatened by hybridization with non-native species. Mol. Ecol. 2021, 30, 895–911. [Google Scholar] [CrossRef]
- Gammerdinger, W.J.; Conte, M.A.; Sandkam, B.A.; Penman, D.J.; Kocher, T.D. Characterization of sex chromosomes in three deeply diverged species of Pseudocrenilabrinae (Teleostei: Cichlidae). Hydrobiologia 2019, 832, 397–408. [Google Scholar] [CrossRef]
- Zhu, H.; Liu, Z.; Lu, M.; Gao, F.; Ke, X.; Ma, D.; Huang, Z.; Cao, J.; Wang, M. Screening and identification of a microsatellite marker associated with sex in Wami tilapia, Oreochromis urolepis hornorum. J. Genet. 2016, 95, 283–289. [Google Scholar] [CrossRef]
- Wu, X.; Zhao, L.; Fan, Z.; Lu, B.; Chen, J.; Tan, D.; Jiang, D.; Tao, W.; Wang, D. Screening and characterization of sex-linked DNA markers and marker-assisted selection in blue tilapia (Oreochromis aureus). Aquaculture 2021, 530, 735934. [Google Scholar] [CrossRef]
- Mtaki, K.; Limbu, S.M.; Mmochi, A.J.; Mtolera, M.S.P. Hybrids production as a potential method to control prolific breeding in tilapia and adaptation to aquaculture climate-induced drought. Aquac. Fish. 2022, 7, 647–652. [Google Scholar] [CrossRef]
- Geletu, T.T.; Zhao, J. Genetic resources of Nile tilapia (Oreochromis niloticus Linnaeus, 1758) in its native range and aquaculture. Hydrobiologia 2022, 1–21. [Google Scholar] [CrossRef]
- Nwachi, O.; Irabor, A. Diallel Hybridization of Oreochromis niloticus (Nile Tilapia) to Oreochromis aureus (Blue Tilapia). Yuzuncu Yıl Univ. J. Agric. Sci. 2022, 32, 280–285. [Google Scholar] [CrossRef]
- Stauffer, J.R.; Chirwa, E.R.; Jere, W.; Konings, A.F.; Tweddle, D.; Weyl, O. Nile Tilapia, Oreochromis niloticus (Teleostei: Cichlidae): A threat to native fishes of Lake Malawi? Biol. Invasions 2022, 24, 1585–1597. [Google Scholar] [CrossRef]
- Brown, K.H. Fish mitochondrial genomics: Sequence, inheritance and functional variation. J. Fish Biol. 2008, 72, 355–374. [Google Scholar] [CrossRef]
- Anderson, L.; Camus, M.F.; Monteith, K.M.; Salminen, T.S.; Vale, P.F. Variation in mitochondrial DNA affects locomotor activity and sleep in Drosophila melanogaster. Heredity 2022, 129, 225–232. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, X.; Xu, Q.; Yan, J.; Yu, F.; Wang, F.; Xiao, J.; Luo, Y.; Zhong, H. Nonadditive and allele-specific expression of insulin-like growth factor 1 in Nile tilapia (Oreochromis niloticus, ♀) × blue tilapia (O. aureus, ♂) hybrids. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2019, 232, 93–100. [Google Scholar] [CrossRef]
- Avella, M.; Berhaut, J.; Bornancin, M. Salinity tolerance of two tropical fishes, Oreochromis aureus and O. niloticus. I. Biochemical and morphological changes in the gill epithelium. J. Fish Biol. 1993, 42, 243–254. [Google Scholar] [CrossRef]
- Shiozaki, T.; Itoh, F.; Hirose, Y.; Onodera, J.; Kuwata, A.; Harada, N. A DNA metabarcoding approach for recovering plankton communities from archived samples fixed in formalin. PLoS ONE 2021, 16, e0245936. [Google Scholar] [CrossRef]
- Uller, T.; Pen, I.; Wapstra, E.; Beukeboom, L.W.; Komdeur, J. The evolution of sex ratios and sex-determining systems. Trends Ecol. Evol. 2007, 22, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.A.; High, S.K.; McCluskey, B.M.; Amores, A.; Yan, Y.L.; Titus, T.A.; Anderson, J.L.; Batzel, P.; Carvan, M.J.; Schartl, M.; et al. Wild sex in zebrafish: Loss of the natural sex determinant in domesticated strains. Genetics 2014, 198, 1291–1308. [Google Scholar] [CrossRef] [PubMed]
- Panthum, T.; Jaisamut, K.; Singchat, W.; Ahmad, S.F.; Kongkaew, L.; Wongloet, W.; Dokkaew, S.; Kraichak, E.; Muangmai, N.; Duengkae, P.; et al. Something fishy about Siamese fighting fish (Betta splendens) sex: Polygenic sex determination or a newly emerged sex-determining region? Cells 2022, 11, 1764. [Google Scholar] [CrossRef] [PubMed]
- Fraser, B.A.; Whiting, J.R.; Paris, J.R.; Weadick, C.J.; Parsons, P.J.; Charlesworth, D.; Bergero, R.; Bemm, F.; Hoffmann, M.; Kottler, V.A.; et al. Improved reference genome uncovers novel sex-linked regions in the guppy (Poecilia reticulata). Genome Biol. Evol. 2020, 12, 1789–1805. [Google Scholar] [CrossRef]
- Franchini, P.; Jones, J.C.; Xiong, P.; Kneitz, S.; Gompert, Z.; Warren, W.C.; Walter, R.B.; Meyer, A.; Schartl, M. Long-term experimental hybridisation results in the evolution of a new sex chromosome in swordtail fish. Nat. Commun. 2018, 9, 5136. [Google Scholar] [CrossRef]
- Moore, E.C.; Ciccotto, P.J.; Peterson, E.N.; Lamm, M.S.; Albertson, R.C.; Roberts, R.B. Polygenic sex determination produces modular sex polymorphism in an African cichlid fish. Proc. Natl. Acad. Sci. USA 2022, 119, e2118574119. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Curzon, A.Y.; Shirak, A.; Ron, M.; Seroussi, E. Master-Key Regulators of Sex Determination in Fish and Other Vertebrates—A Review. Int. J. Mol. Sci. 2023, 24, 2468. https://doi.org/10.3390/ijms24032468
Curzon AY, Shirak A, Ron M, Seroussi E. Master-Key Regulators of Sex Determination in Fish and Other Vertebrates—A Review. International Journal of Molecular Sciences. 2023; 24(3):2468. https://doi.org/10.3390/ijms24032468
Chicago/Turabian StyleCurzon, Arie Yehuda, Andrey Shirak, Micha Ron, and Eyal Seroussi. 2023. "Master-Key Regulators of Sex Determination in Fish and Other Vertebrates—A Review" International Journal of Molecular Sciences 24, no. 3: 2468. https://doi.org/10.3390/ijms24032468
APA StyleCurzon, A. Y., Shirak, A., Ron, M., & Seroussi, E. (2023). Master-Key Regulators of Sex Determination in Fish and Other Vertebrates—A Review. International Journal of Molecular Sciences, 24(3), 2468. https://doi.org/10.3390/ijms24032468






