Effect of Epstein–Barr Virus Infection on Selected Immunological Parameters in Children with Type 1 Diabetes
Abstract
1. Introduction
2. Results
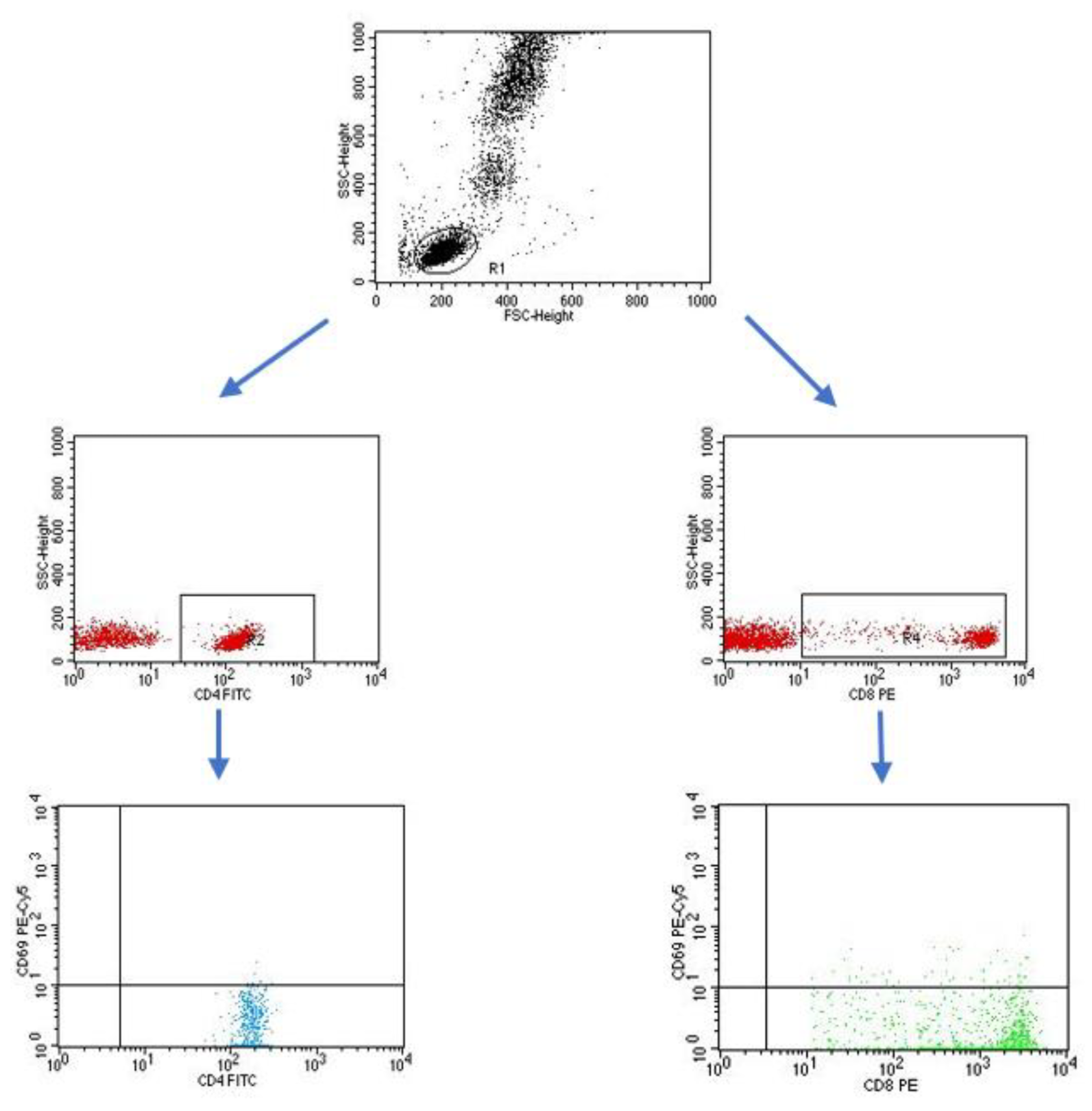
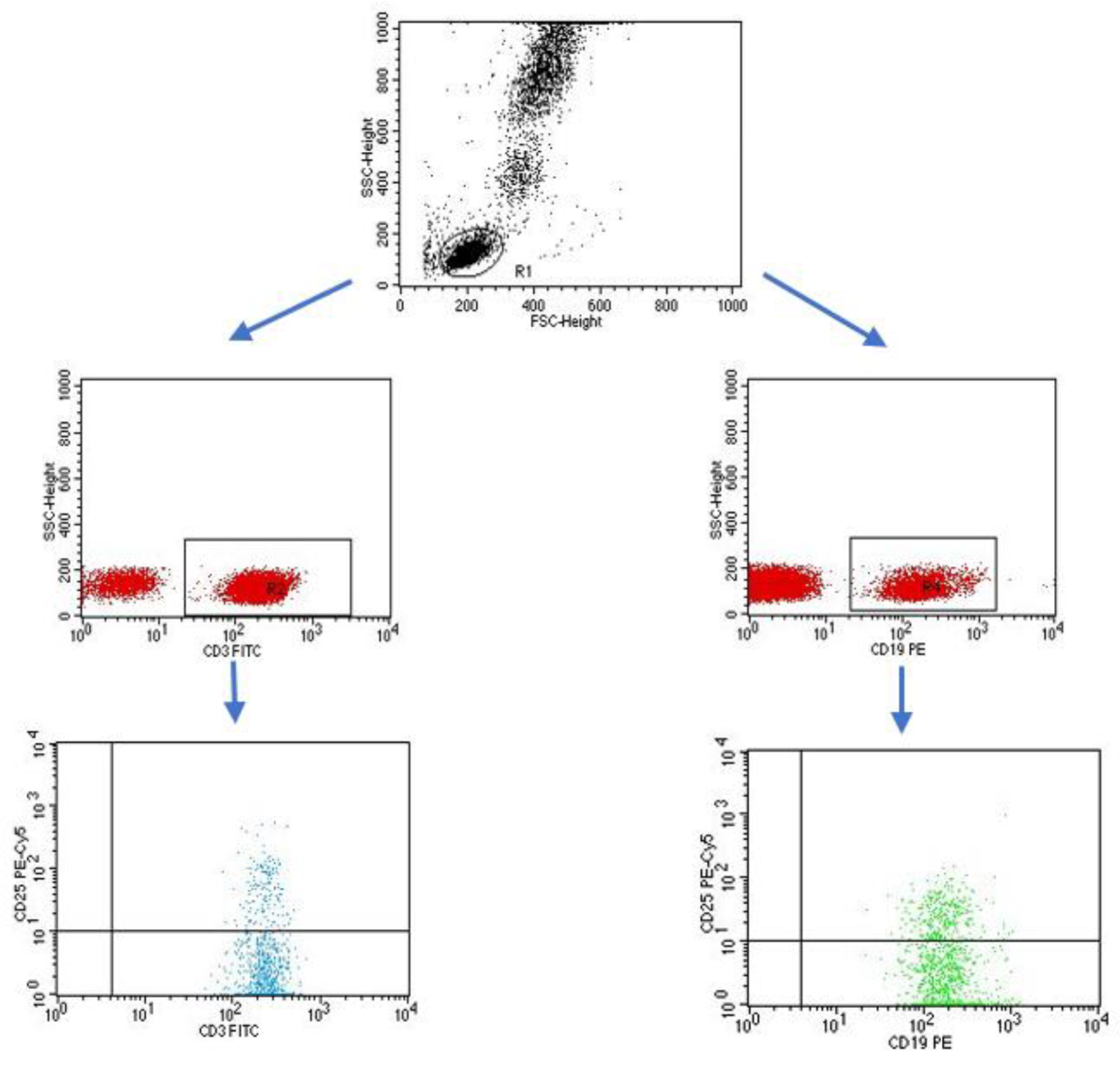
2.1. Evaluation of Selected Subpopulations of Lymphocytes
2.2. Evaluation of the Percentage of Lymphocytes Expressing Selected Activation Markers on T and B Lymphocytes
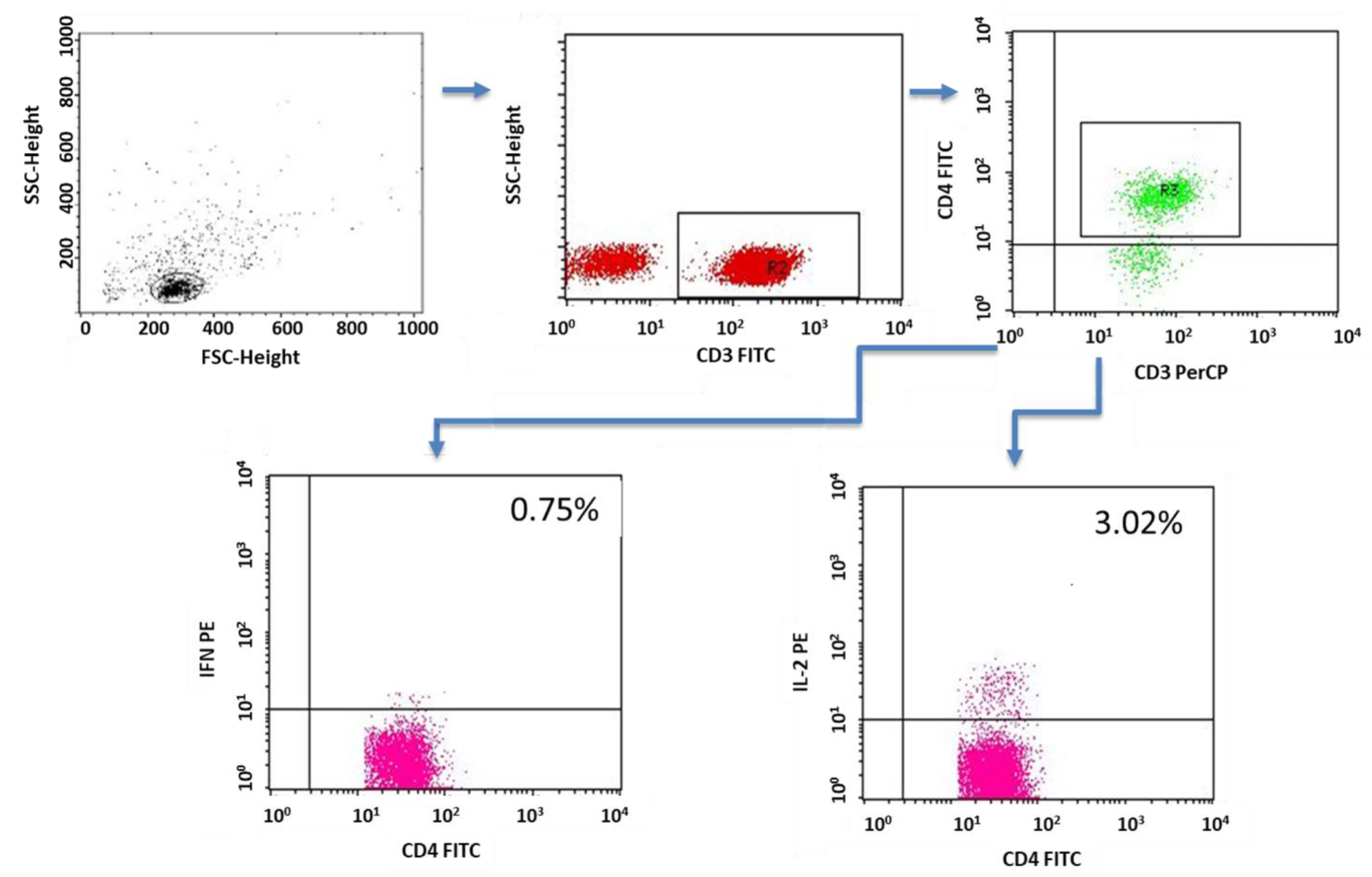
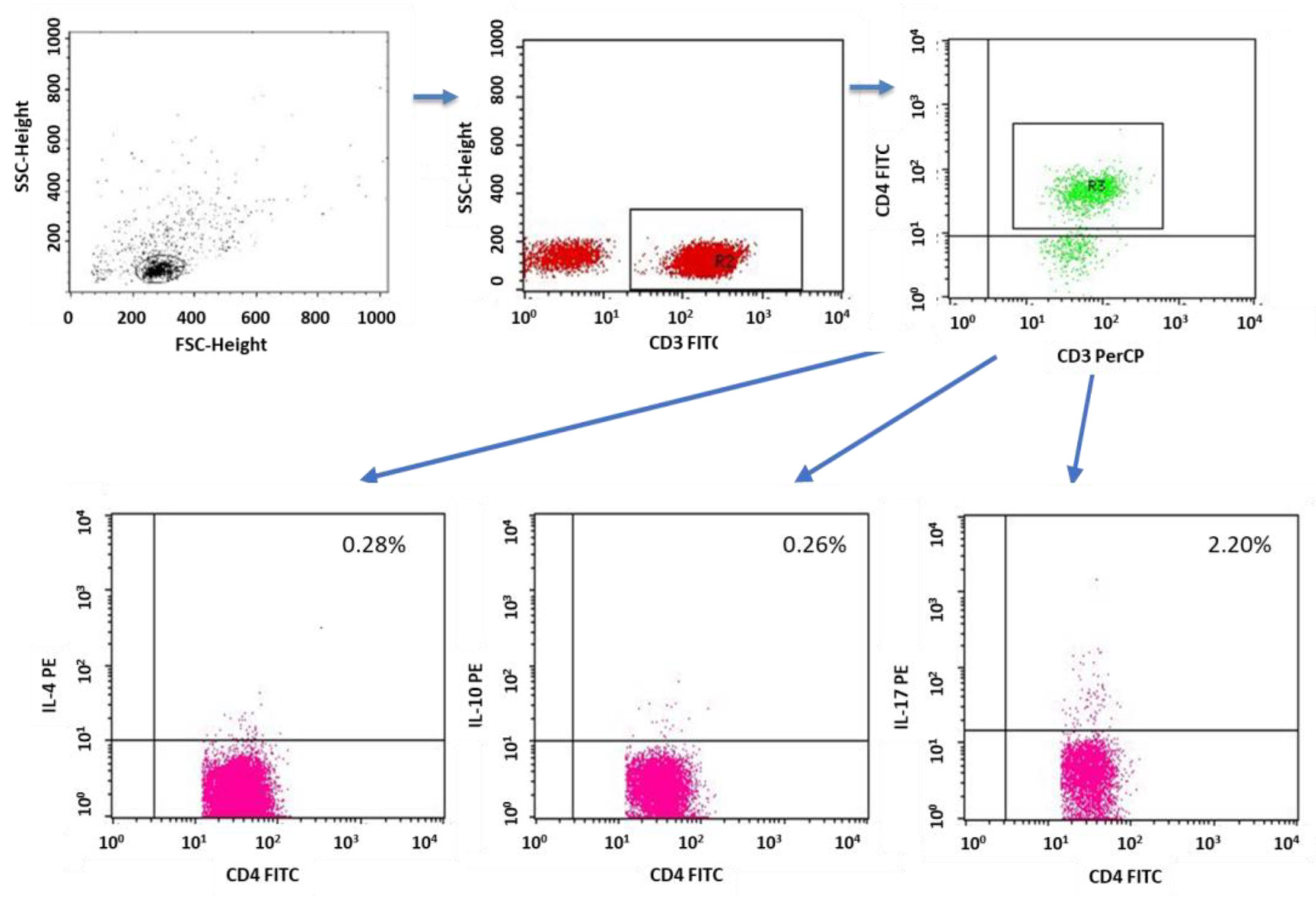
2.3. Evaluation of Lymphocytes with Intracellular Expression of Selected Cytokines
2.4. Evaluation of Antibodies against EBV Antigens
2.5. Evaluation of Selected Subpopulations of Lymphocytes in Patients with Type 1 Diabetes in Relation to the Presence of Antibodies against Antigens of the EBV Virus
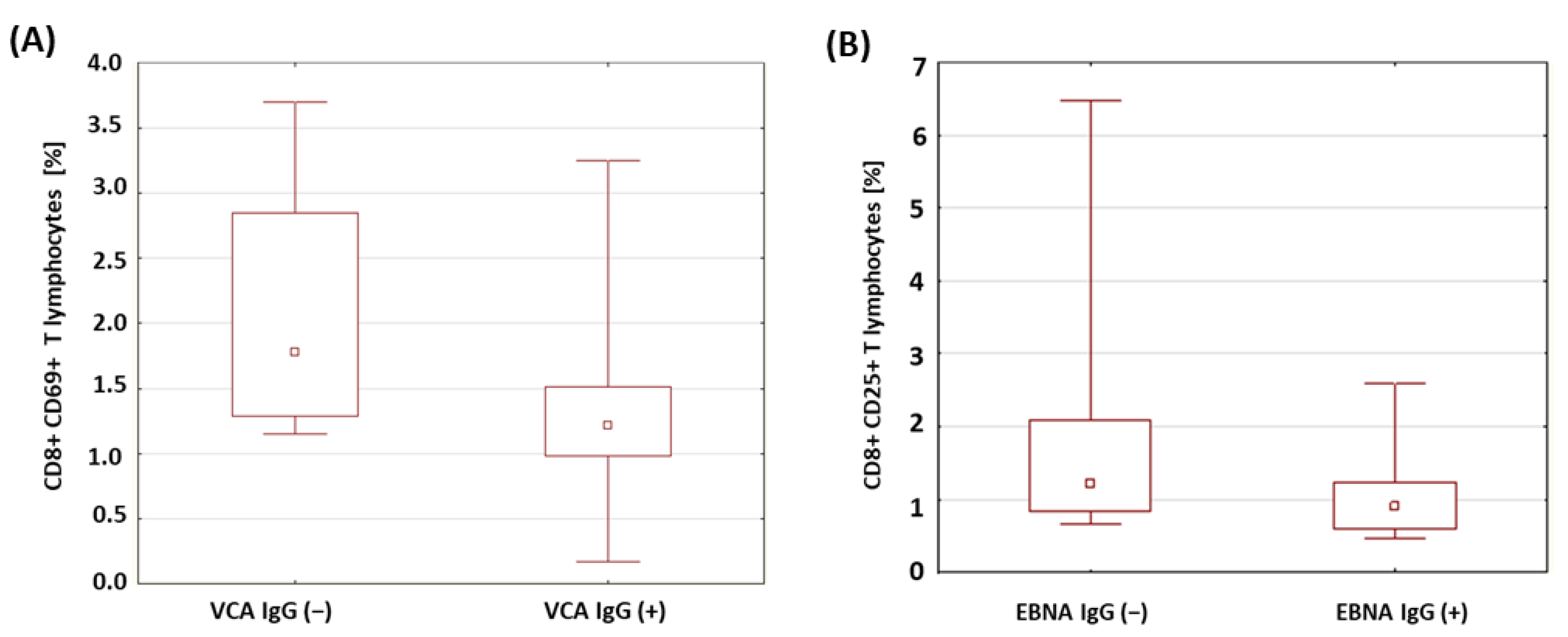
2.6. Evaluation of the Percentage of Lymphocytes Expressing Selected Activation Markers in Patients with Type 1 Diabetesin Relation to the Presence of Antibodies against EBV Antigens
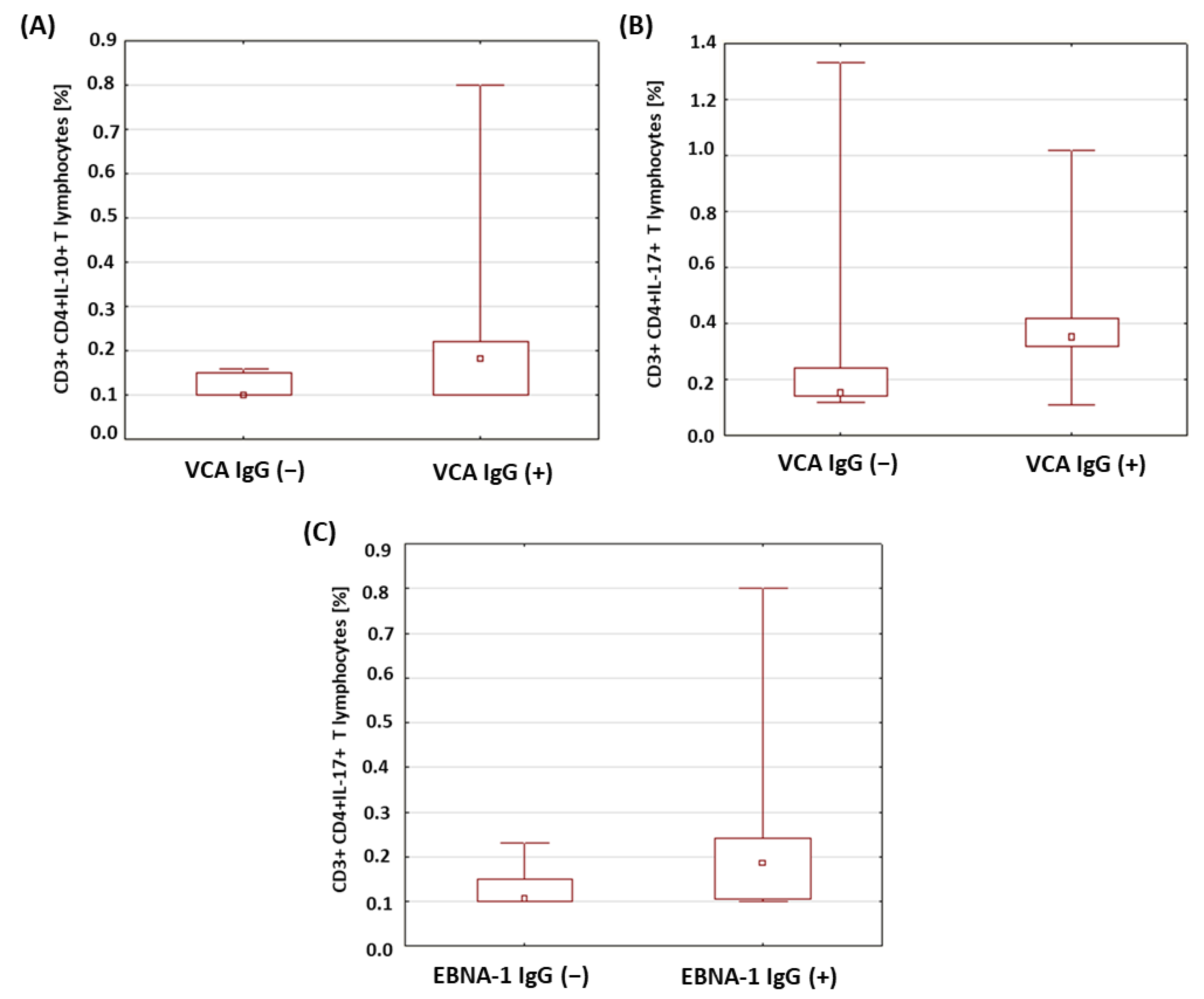
2.7. Evaluation of the Percentage of Lymphocytes with Intracellular Expression of Selected Cytokines in Patients with Type 1 Diabetes in Relation to the Presence of Antibodies against Antigens of the EBV Virus
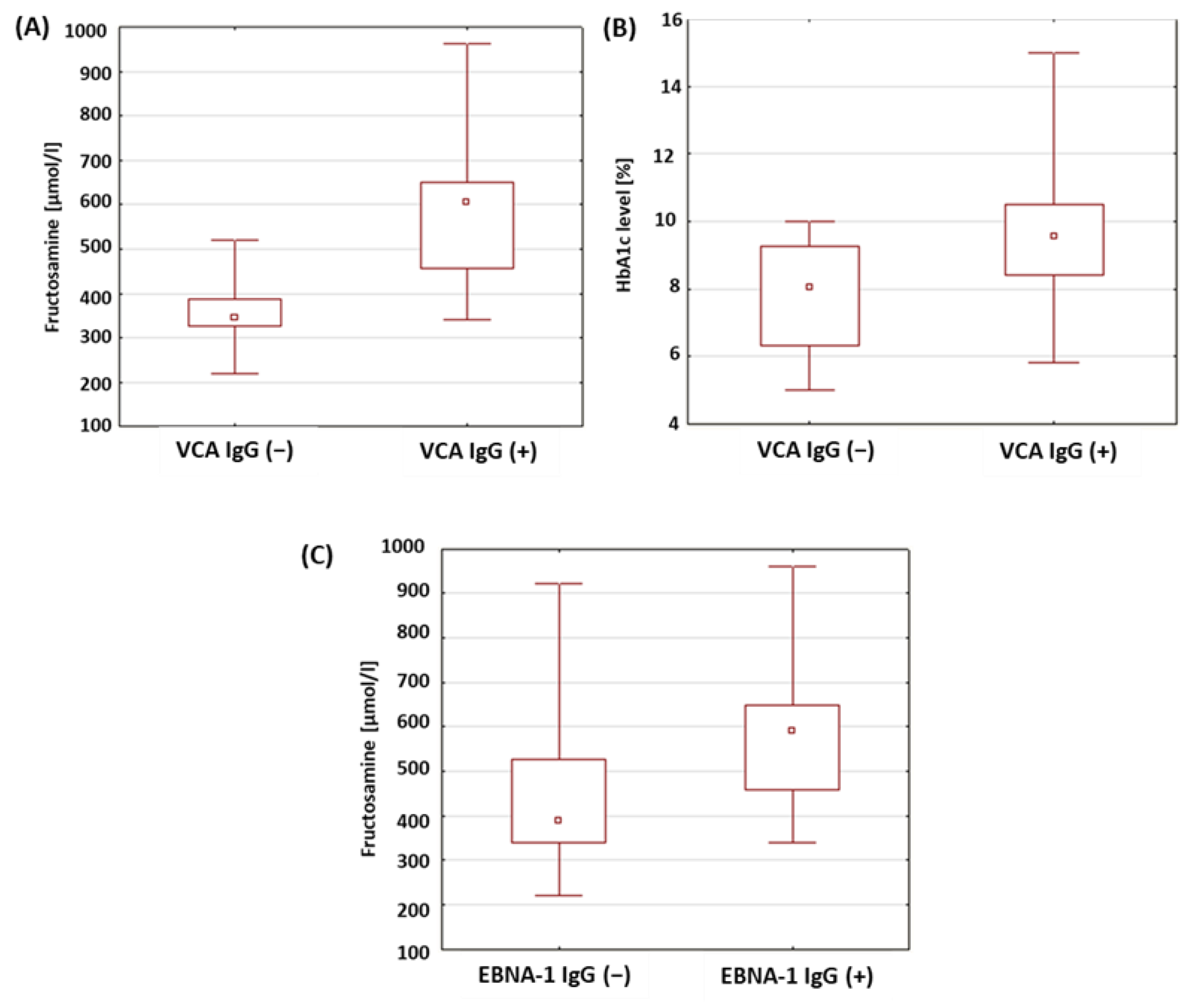
2.8. Evaluation of Selected Parameters of Carbohydrate Balance in Patients with Type 1 Diabetes in Relation to the Presence of Antibodies against Antigens of the EBV Virus
3. Discussion
4. Materials and Methods
4.1. Characteristics of Patients from the Research and Control Groups
4.2. Material for Research
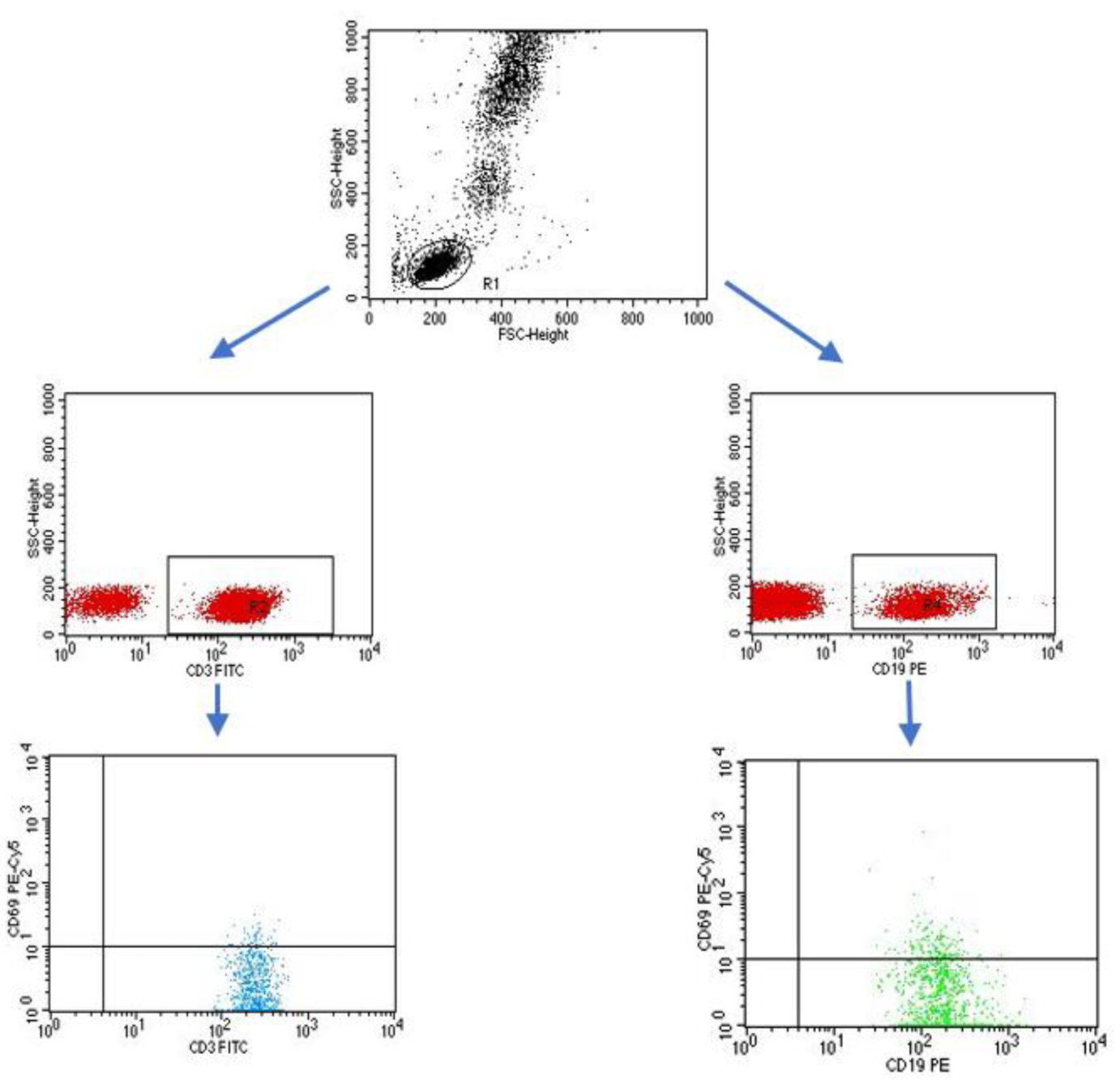
4.3. Evaluation of the Subpopulation of Peripheral-Blood Lymphocytes by Flow Cytometry
4.4. Isolation of Peripheral-Blood Mononuclear Cells
4.5. Cytokine Labeling
4.6. Evaluation of the Concentrations of Antibodies against Epstein–Barr Virus Antigens in the Serum
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| APC | antigen-resenting cells |
| CD | cluster of differentiation |
| CMV | cytomegalovirus |
| DM1 | diabetes mellitus type 1 |
| DM1A | diabetes mellitus type 1A |
| DM1B | diabetes mellitus type 1B |
| EBNA | Epstein–Barr nuclear antigen |
| EBV | Epstein–Barr virus |
| EDTA | ethylenediaminetetraacetate |
| FITC | fluorescein isothiocyanate |
| FSC | forward-scatter channel |
| HbA1c | glycated hemoglobin; hemoglobin A1c |
| IFN-γ | interferon-γ |
| IL | interleukin |
| NK | natural killer cells |
| PBMC | peripheral mononuclear blood cells |
| PBS | phosphate-buffered saline |
| SSC | side-scatter channel |
| TNF | tumor-necrosis factor |
| Treg | regulatory T cells |
| VCA | viral-capsid antigen complex |
References
- American Diabetes Association. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2019. Diabetes Care 2019, 42 (Suppl. S1), S13–S28. [Google Scholar] [CrossRef] [PubMed]
- Definition and Diagnosis of Diabetes Mellitus and Intermediate Hyperglycaemia. Available online: https://www.who.int/publications-detail-redirect/definition-and-diagnosis-of-diabetes-mellitus-and-intermediate-hyperglycaemia (accessed on 14 December 2022).
- Daneman, D. Type 1 diabetes. Lancet 2006, 367, 847–858. [Google Scholar] [CrossRef] [PubMed]
- Praktyczna, D. Zalecenia kliniczne dotyczące postępowania u chorych na cukrzycę 2018. Stanowisko Polskiego Towarzystwa Diabetologicznego. Diabetol. Prakt. 2018, 4, 1–94. [Google Scholar]
- Araszkiewicz, A.; Bandurska-Stankiewicz, E.; Borys, S.; Budzyński, A.; Cyganek, K.; Cypryk, K.; Czech, A.; Czupryniak, L.; Drzewoski, J.; Dzida, G.; et al. Zalecenia kliniczne dotyczące postępowania u chorych z cukrzycą 2021. Stanowisko Polskiego Towarzystwa Diabetologicznego. Diabetol. Prakt. 2021, 7, 1–121. [Google Scholar]
- Ozen, G.; Zanfardino, A.; Confetto, S.; Piscopo, A.; Casaburo, F.; Tinto, N.; Iafusco, F.; Ozen, G.; del Giudice, E.M.; Tasar, M.A.; et al. The Association of Autoimmune Diseases with Type 1 Diabetes Mellitus in Children Depends Also by the Length of Partial Clinical Remission Phase (Honeymoon). Int. J. Endocrinol. 2020, 2020, 2630827. [Google Scholar] [CrossRef]
- Clark, M.; Kroger, C.J.; Tisch, R.M. Type 1 Diabetes: A Chronic Anti-Self-Inflammatory Response. Front. Immunol. 2017, 8, 1898. [Google Scholar] [CrossRef]
- Uusitalo, U.; Liu, X.; Yang, J.; Aronsson, C.A.; Hummel, S.; Butterworth, M.; Lernmark, A.; Rewers, M.; Hagopian, W.; She, J.-X.; et al. Association of Early Exposure of Probiotics and Islet Autoimmunity in the TEDDY Study. JAMA Pediatr. 2016, 170, 20–28. [Google Scholar] [CrossRef]
- Knip, M.; Siljander, H. The role of the intestinal microbiota in type 1 diabetes mellitus. Nat. Rev. Endocrinol. 2016, 12, 154–167. [Google Scholar] [CrossRef]
- Zheng, S.-J.; Luo, Y.; Xiao, J.-H. The Impact of Intestinal Microorganisms and Their Metabolites on Type 1 Diabetes Mellitus. Diabetes, Metab. Syndr. Obesity Targets Ther. 2022, 15, 1123–1139. [Google Scholar] [CrossRef]
- Bielka, W.; Przezak, A.; Pawlik, A. The Role of the Gut Microbiota in the Pathogenesis of Diabetes. Int. J. Mol. Sci. 2022, 23, 480. [Google Scholar] [CrossRef]
- Han, H.; Li, Y.; Fang, J.; Liu, G.; Yin, J.; Li, T.; Yin, Y. Gut Microbiota and Type 1 Diabetes. Int. J. Mol. Sci. 2018, 19, 995. [Google Scholar] [CrossRef] [PubMed]
- Imagawa, A.; Hanafusa, T. Fulminant type 1 diabetes-an important subtype in East Asia. Diabetes/Metabolism Res. Rev. 2011, 27, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Aida, K.; Nishida, Y.; Kobayashi, T. Pathophysiological mechanisms involving aggressive islet cell destruction in fulminant type 1 diabetes [Review]. Endocr. J. 2013, 60, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Imagawa, A.; Hanafusa, T.; Awata, T.; Ikegami, H.; Uchigata, Y.; Osawa, H.; Kawasaki, E.; Kawabata, Y.; Kobayashi, T.; Shimada, A.; et al. Report of the Committee of the Japan Diabetes Society on the Research of Fulminant and Acute-onset Type 1 Diabetes Mellitus: New diagnostic criteria of fulminant type 1 diabetes mellitus. J. Diabetes Investig. 2012, 3, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Winter, W.E.; Schatz, D.A.; Sacks, D.B.; Nathan, D.M.; Lachin, J.M. Autoimmune Markers in Diabetes. Clin. Chem. 2011, 57, 168–175. [Google Scholar] [CrossRef]
- Jin, Y.; She, J.-X. Novel Biomarkers in Type 1 Diabetes. Rev. Diabet. Stud. 2012, 9, 224–235. [Google Scholar] [CrossRef][Green Version]
- Hummel, M.; Bonifacio, E.; Schmid, S.; Walter, M.; Knopff, A.; Ziegler, A.-G. Brief Communication: Early Appearance of Islet Autoantibodies Predicts Childhood Type 1 Diabetes in Offspring of Diabetic Parents. Ann. Intern. Med. 2004, 140, 882–886. [Google Scholar] [CrossRef]
- Ziegler, A.-G.; Pflueger, M.; Winkler, C.; Achenbach, P.; Akolkar, B.; Krischer, J.P.; Bonifacio, E. Accelerated progression from islet autoimmunity to diabetes is causing the escalating incidence of type 1 diabetes in young children. J. Autoimmun. 2011, 37, 3–7. [Google Scholar] [CrossRef]
- Roche, E.F.; Menon, A.; Gill, D.; Hoey, H. Clinical presentation of type 1 diabetes. Pediatr. Diabetes 2005, 6, 75–78. [Google Scholar] [CrossRef]
- Honesta, K.; Edna, M.; Benjamin, K.; Lucy, M. Clinical Presentation and Factors Associated with Diabetic Ketoacidosis at the Onset of Type-1 Diabetes Mellitus in Children and Adolescent at Muhimbili National Hospital, Tanzania: A Cross Section Study. Int. J. Diabetes Clin. Res. 2020, 7, 1410126. [Google Scholar] [CrossRef]
- Houen, G.; Trier, N.H. Epstein–Barr Virus and Systemic Autoimmune Diseases. Front. Immunol. 2021, 11, 587380. [Google Scholar] [CrossRef]
- França, S.A.D.S.; Viana, J.B.G.d.O.; Góes, H.C.A.; Fonseca, R.R.D.S.; Laurentino, R.V.; Costa, I.B.; Oliveira-Filho, A.B.; Machado, L.F.A. Epidemiology of the Epstein–Barr Virus in Autoimmune Inflammatory Rheumatic Diseases in Northern Brazil. Viruses 2022, 14, 694. [Google Scholar] [CrossRef]
- Kuri, A.; Jacobs, B.M.; Vickaryous, N.; Pakpoor, J.; Middeldorp, J.; Giovannoni, G.; Dobson, R. Epidemiology of Epstein–Barr virus infection and infectious mononucleosis in the United Kingdom. BMC Public Health 2020, 20, 912. [Google Scholar] [CrossRef]
- Smatti, M.K.; Al-Sadeq, D.W.; Ali, N.H.; Pintus, G.; Abou-Saleh, H.; Nasrallah, G.K. Epstein–Barr Virus Epidemiology, Serology, and Genetic Variability of LMP-1 Oncogene Among Healthy Population: An Update. Front. Oncol. 2018, 8, 211. [Google Scholar] [CrossRef]
- Dugan, J.P.; Coleman, C.B.; Haverkos, B. Opportunities to Target the Life Cycle of Epstein–Barr Virus (EBV) in EBV-Associated Lymphoproliferative Disorders. Front. Oncol. 2019, 9, 127. [Google Scholar] [CrossRef]
- Toussirot, E.; Roudier, J. Epstein–Barr virus in autoimmune diseases. Best Pract. Res. Clin. Rheumatol. 2008, 22, 883–896. [Google Scholar] [CrossRef]
- Janegova, A.; Janega, P.; Rychly, B.; Kuracinova, K.; Babal, P. Rola infekcji wirusem Epstein–Barr’a w rozwoju autoimmunologicznych chorób tarczycy. Endokrynol. Polska 2015, 66, 132–136. [Google Scholar] [CrossRef]
- Pyzik, A.; Grywalska, E.; Matyjaszek-Matuszek, B.; Ludian, J.; Kiszczak-Bochyńska, E.; Smoleń, A.; Roliński, J.; Pyzik, D. Does the Epstein–Barr Virus Play a Role in the Pathogenesis of Graves’ Disease? Int. J. Mol. Sci. 2019, 20, 3145. [Google Scholar] [CrossRef]
- Dreyfus, D.H.; Farina, A.; Farina, G.A. Molecular mimicry, genetic homology, and gene sharing proteomic “molecular fingerprints” using an EBV (Epstein–Barr virus)-derived microarray as a potential diagnostic method in autoimmune disease. Immunol. Res. 2018, 66, 686–695. [Google Scholar] [CrossRef]
- Santiago, O.; Gutiérrez-Fernández, J.; Sorlozano, A.; de Dios Luna, J.; Villegas, E.; Fernandez, O. Relation between Epstein–Barr virus and multiple sclerosis: Analytic study of scientific production. Eur. J. Clin. Microbiol. Infect. Dis. 2010, 29, 857–866. [Google Scholar] [CrossRef]
- Dittfeld, A.; Gwizdek, K.; Michalski, M.; Wojnicz, R. A possible link between the Epstein–Barr virus infection and autoimmune thyroid disorders. Central Eur. J. Immunol. 2016, 3, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Chougule, D.; Nadkar, M.; Rajadhyaksha, A.; Pandit-Shende, P.; Surve, P.; Dawkar, N.; Khadilkar, P.; Patwardhan, M.; Kaveri, S.; Ghosh, K.; et al. Association of clinical and serological parameters of systemic lupus erythematosus patients with Epstein–Barr virus antibody profile. J. Med. Virol. 2017, 90, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Jog, N.; Young, K.A.; Munroe, M.E.; Harmon, M.; Guthridge, J.M.; Kelly, J.; Kamen, D.L.; Gilkeson, G.S.; Weisman, M.H.; Karp, D.R.; et al. Association of Epstein–Barr virus serological reactivation with transitioning to systemic lupus erythematosus in at-risk individuals. Ann. Rheum. Dis. 2019, 78, 1235–1241. [Google Scholar] [CrossRef] [PubMed]
- Principi, N.; Berioli, M.G.; Bianchini, S.; Esposito, S. Type 1 diabetes and viral infections: What is the relationship? J. Clin. Virol. 2017, 96, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.-L.; Vaziri-Sani, F.; Broberg, P.; Elfaitouri, A.; Pipkorn, R.; Blomberg, J.; Ivarsson, S.-A.; Larsson, H.E.; Lernmark, A. Serological evaluation of possible exposure to Ljungan virus and related parechovirus in autoimmune (type 1) diabetes in children. J. Med. Virol. 2015, 87, 1130–1140. [Google Scholar] [CrossRef]
- Honeyman, M.C.; Laine, D.; Zhan, Y.; Londrigan, S.; Kirkwood, C.; Harrison, L.C. Rotavirus Infection Induces Transient Pancreatic Involution and Hyperglycemia in Weanling Mice. PLoS ONE 2014, 9, e106560. [Google Scholar] [CrossRef]
- Kondrashova, A.; Hyöty, H. Role of Viruses and Other Microbes in the Pathogenesis of Type 1 Diabetes. Int. Rev. Immunol. 2014, 33, 284–295. [Google Scholar] [CrossRef]
- Isaacs, S.; Foskett, D.; Maxwell, A.; Ward, E.; Faulkner, C.; Luo, J.; Rawlinson, W.; Craig, M.; Kim, K. Viruses and Type 1 Diabetes: From Enteroviruses to the Virome. Microorganisms 2021, 9, 1519. [Google Scholar] [CrossRef]
- Jun, H.-S.; Yoon, J.-W. A new look at viruses in type 1 diabetes. Diabetes/Metabolism Res. Rev. 2003, 19, 8–31. [Google Scholar] [CrossRef]
- Hara, N.; Alkanani, A.K.; Dinarello, C.A.; Zipris, D. Modulation of virus-induced innate immunity and type 1 diabetes by IL-1 blockade. J. Endotoxin Res. 2013, 20, 574–584. [Google Scholar] [CrossRef]
- Rasmussen, T.; Witsø, E.; Tapia, G.; Stene, L.C.; Rønningen, K.S. Self-reported lower respiratory tract infections and development of islet autoimmunity in children with the type 1 diabetes high-risk HLA genotype: The MIDIA study. Diabetes/Metabolism Res. Rev. 2011, 27, 834–837. [Google Scholar] [CrossRef]
- Blanter, M.; Sork, H.; Tuomela, S.; Flodström-Tullberg, M. Genetic and Environmental Interaction in Type 1 Diabetes: A Relationship Between Genetic Risk Alleles and Molecular Traits of Enterovirus Infection? Curr. Diabetes Rep. 2019, 19, 82. [Google Scholar] [CrossRef]
- Beyerlein, A.; Wehweck, F.; Ziegler, A.-G.; Pflueger, M. Respiratory Infections in Early Life and the Development of Islet Autoimmunity in Children at Increased Type 1 Diabetes Risk: Evidence from the BABYDIET Study. JAMA Pediatr. 2013, 167, 800–807. [Google Scholar] [CrossRef]
- Filippi, C.M.; von Herrath, M.G. Viral Trigger for Type 1 Diabetes. Diabetes 2008, 57, 2863–2871. [Google Scholar] [CrossRef]
- Reddy, M.; Eirikis, E.; Davis, C.; Davis, H.M.; Prabhakar, U. Comparative analysis of lymphocyte activation marker expression and cytokine secretion profile in stimulated human peripheral blood mononuclear cell cultures: An in vitro model to monitor cellular immune function. J. Immunol. Methods 2004, 293, 127–142. [Google Scholar] [CrossRef]
- Wieland, E. Chapter 10—Markers of Lymphocyte Activation and Proliferation. In Personalized Immunosuppression in Trans-Plantation; Oellerich, M., Dasgupta, A., Eds.; Elsevier: San Diego, CA, USA, 2016; pp. 227–257. ISBN 978-0-12-800885-0. [Google Scholar]
- Shipkova, M.; Wieland, E. Surface markers of lymphocyte activation and markers of cell proliferation. Clin. Chim. Acta 2012, 413, 1338–1349. [Google Scholar] [CrossRef]
- Rudnicka, K.; Matusiak, A.; Chmiela, M. CD25 (IL-2R) expression correlates with the target cell induced cytotoxic activity and cytokine secretion in human natural killer cells. Acta Biochim. Pol. 2015, 62, 885–894. [Google Scholar] [CrossRef]
- Clark, M.; Kroger, C.J.; Ke, Q.; Tisch, R.M. The Role of T Cell Receptor Signaling in the Development of Type 1 Diabetes. Front. Immunol. 2021, 11, 615371. [Google Scholar] [CrossRef]
- Pugliese, A. Autoreactive T cells in type 1 diabetes. J. Clin. Investig. 2017, 127, 2881–2891. [Google Scholar] [CrossRef]
- Giordano, C.; De Maria, R.; Todaro, M.; Stassi, G.; Mattina, A.; Richiusa, P.; Galluzzo, G.; Panto, F.; Galluzzo, A. Study of T-cell activation in Type I diabetic patients and pre-Type I diabetic subjects by cytometric analysis: Antigen expression defectin vitro. J. Clin. Immunol. 1993, 13, 68–78. [Google Scholar] [CrossRef]
- Beyerlein, A.; Donnachie, E.; Jergens, S.; Ziegler, A.-G. Infections in Early Life and Development of Type 1 Diabetes. JAMA 2016, 315, 1899–1901. [Google Scholar] [CrossRef] [PubMed]
- Tracy, S.; Drescher, K.M. Coxsackievirus Infections and NOD Mice: Relevant Models of Protection from, and Induction of, Type 1 Diabetes. Ann. N. Y. Acad. Sci. 2007, 1103, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Tovo, P.-A.; Rabbone, I.; Tinti, D.; Galliano, I.; Trada, M.; Daprà, V.; Cerutti, F.; Bergallo, M. Enhanced expression of human endogenous retroviruses in new-onset type 1 diabetes: Potential pathogenetic and therapeutic implications. Autoimmunity 2020, 53, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Berg, A.-K.; Korsgren, O.; Frisk, G. Induction of the chemokine interferon-γ-inducible protein-10 in human pancreatic islets during enterovirus infection. Diabetologia 2006, 49, 2697–2703. [Google Scholar] [CrossRef]
- Zanone, M.M.; Favaro, E.; Ferioli, E.; Huang, G.C.; Klein, N.J.; Perin, P.C.; Peakman, M.; Conaldi, P.G.; Camussi, G. Human pancreatic islet endothelial cells express coxsackievirus and adenovirus receptor and are activated by coxsackie B virus infection. FASEB J. 2007, 21, 3308–3317. [Google Scholar] [CrossRef] [PubMed]
- Faustman, D.L.; Davis, M. TNF Receptor 2 and Disease: Autoimmunity and Regenerative Medicine. Front. Immunol. 2013, 4, 478. [Google Scholar] [CrossRef] [PubMed]
- Christen, U.; Wolfe, T.; Möhrle, U.; Hughes, A.C.; Rodrigo, E.; Green, E.A.; Flavell, R.A.; Von Herrath, M.G. A Dual Role for TNF-α in Type 1 Diabetes: Islet-Specific Expression Abrogates the Ongoing Autoimmune Process When Induced Late but Not Early During Pathogenesis. J. Immunol. 2001, 166, 7023–7032. [Google Scholar] [CrossRef]
- Luczyński, W.; Stasiak-Barmuta, A.; Urban, R.; Urban, M.; Florys, B.; Hryszko, M. Lower percentages of T regulatory cells in children with type 1 diabetes—Preliminary report. Pediatr. Endocrinol. Diabetes Metab. 2009, 15, 34–38. [Google Scholar]
- Szypowska, A.; Stelmaszczyk-Emmel, A.; Demkow, U.; Łuczyński, W. Low Frequency of Regulatory T Cells in the Peripheral Blood of Children with Type 1 Diabetes Diagnosed under the Age of Five. Arch. Immunol. Ther. Exp. 2012, 60, 307–313. [Google Scholar] [CrossRef]
- Metwalley, K.; Zahran, A.; Elsayh, K. Regulatory T cells in children with recently diagnosed type 1 diabetes. Indian J. Endocrinol. Metab. 2012, 16, 952–957. [Google Scholar] [CrossRef]
- Zhang, A.; Ning, B.; Sun, N.; Wei, J.; Ju, X. Indirubin Increases CD4+CD25+Foxp3+ Regulatory T Cells to Prevent Immune Thrombocytopenia in Mice. PLoS ONE 2015, 10, e0142634. [Google Scholar] [CrossRef]
- Salomon, B.; Lenschow, D.J.; Rhee, L.; Ashourian, N.; Singh, B.; Sharpe, A.; A Bluestone, J. B7/CD28 Costimulation Is Essential for the Homeostasis of the CD4+CD25+ Immunoregulatory T Cells that Control Autoimmune Diabetes. Immunity 2000, 12, 431–440. [Google Scholar] [CrossRef]
- Dendrou, C.A.; Wicker, L.S. The IL-2/CD25 Pathway Determines Susceptibility to T1D in Humans and NOD Mice. J. Clin. Immunol. 2008, 28, 685–696. [Google Scholar] [CrossRef]
- Long, S.A.; Cerosaletti, K.; Bollyky, P.L.; Tatum, M.; Shilling, H.; Zhang, S.; Zhang, Z.-Y.; Pihoker, C.; Sanda, S.; Greenbaum, C.; et al. Defects in IL-2R Signaling Contribute to Diminished Maintenance of FOXP3 Expression in CD4+CD25+ Regulatory T-Cells of Type 1 Diabetic Subjects. Diabetes 2009, 59, 407–415. [Google Scholar] [CrossRef]
- Rabinovitch, A.; Suarez-Pinzon, W.L. Roles of cytokines in the pathogenesis and therapy of type 1 diabetes. Cell Biochem. Biophys. 2007, 48, 159–163. [Google Scholar] [CrossRef]
- Long, S.A.; Rieck, M.; Sanda, S.; Bollyky, J.B.; Samuels, P.L.; Goland, R.; Ahmann, A.; Rabinovitch, A.; Aggarwal, S.; Phippard, D.; et al. Rapamycin/IL-2 Combination Therapy in Patients With Type 1 Diabetes Augments Tregs yet Transiently Impairs β-Cell Function. Diabetes 2012, 61, 2340–2348. [Google Scholar] [CrossRef]
- Pérol, L.; Lindner, J.M.; Caudana, P.; Nunez, N.G.; Baeyens, A.; Valle, A.; Sedlik, C.; Loirat, D.; Boyer, O.; Créange, A.; et al. Loss of immune tolerance to IL-2 in type 1 diabetes. Nat. Commun. 2016, 7, 13027. [Google Scholar] [CrossRef]
- Lindley, S.; Dayan, C.M.; Bishop, A.; Roep, B.O.; Peakman, M.; Tree, T.I. Defective Suppressor Function in CD4+CD25+ T-Cells From Patients With Type 1 Diabetes. Diabetes 2005, 54, 92–99. [Google Scholar] [CrossRef]
- El-Masry CD4 and CD25 Cells in Children under the Age of 5 Diagnosed with Type I Diabetes Mellitus. Available online: https://www.tdj.eg.net/article.asp?issn=1110-1415;year=2018;volume=46;issue=1;spage=67;epage=76;aulast=El-Masry (accessed on 14 December 2022).
- Cinek, O.; Stene, L.C.; Kramna, L.; Tapia, G.; Oikarinen, S.; Witsø, E.; Rasmussen, T.; Torjesen, P.A.; Hyöty, H.; Rønningen, K.S. Enterovirus RNA in longitudinal blood samples and risk of islet autoimmunity in children with a high genetic risk of type 1 diabetes: The MIDIA study. Diabetologia 2014, 57, 2193–2200. [Google Scholar] [CrossRef]
- Frederiksen, B.; Kroehl, M.; Lamb, M.M.; Seifert, J.; Barriga, K.; Eisenbarth, G.S.; Rewers, M.; Norris, J.M. Infant Exposures and Development of Type 1 Diabetes Mellitus. JAMA Pediatr. 2013, 167, 808–815. [Google Scholar] [CrossRef]
- Chen, T.; Hudnall, S.D. Anatomical mapping of human herpesvirus reservoirs of infection. Mod. Pathol. 2006, 19, 726–737. [Google Scholar] [CrossRef] [PubMed]
- Osame, K.; Takahashi, Y.; Takasawa, H.; Watanabe, S.; Kishimoto, M.; Yasuda, K.; Kaburagi, Y.; Nakanishi, K.; Kajio, H.; Noda, M. Rapid-Onset Type 1 Diabetes Associated with Cytomegalovirus Infection and Islet Autoantibody Synthesis. Intern. Med. 2007, 46, 873–877. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hyöoty, H.; Räasäanen, L.; Hiltunen, M.; Lehtinen, M.; Huupponen, T.; Leinikki, P. Decreased antibody reactivity to Epstein–Barr virus capsid antigen in type 1 (insulin-dependent) diabetes mellitus. Apmis 1991, 99, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Draborg, A.H.; Jacobsen, S.; Westergaard, M.; Mortensen, S.; Larsen, J.L.; Houen, G.; Duus, K. Reduced response to Epstein–Barr virus antigens by T-cells in systemic lupus erythematosus patients. Lupus Sci. Med. 2014, 1, e000015. [Google Scholar] [CrossRef] [PubMed]
- Kang, I.; Quan, T.; Nolasco, H.; Park, S.-H.; Hong, M.S.; Crouch, J.; Pamer, E.G.; Howe, J.G.; Craft, J. Defective Control of Latent Epstein–Barr Virus Infection in Systemic Lupus Erythematosus. J. Immunol. 2004, 172, 1287–1294. [Google Scholar] [CrossRef]
- Márquez, A.C.; Horwitz, M.S. The Role of Latently Infected B Cells in CNS Autoimmunity. Front. Immunol. 2015, 6, 544. [Google Scholar] [CrossRef]
- Pender, M.P. Infection of autoreactive B lymphocytes with EBV, causing chronic autoimmune diseases. Trends Immunol. 2003, 24, 584–588. [Google Scholar] [CrossRef]
- Grimm, T.; Schneider, S.; Naschberger, E.; Huber, J.; Guenzi, E.; Kieser, A.; Reitmeir, P.; Schulz, T.F.; Morris, C.A.; Stürzl, M. EBV latent membrane protein-1 protects B cells from apoptosis by inhibition of BAX. Blood 2005, 105, 3263–3269. [Google Scholar] [CrossRef]
- Accardi, R.; Fathallah, I.; Gruffat, H.; Mariggiò, G.; Le Calvez-Kelm, F.; Voegele, C.; Bartosch, B.; Hernandez-Vargas, H.; McKay, J.; Sylla, B.S.; et al. Epstein–Barr Virus Transforming Protein LMP-1 Alters B Cells Gene Expression by Promoting Accumulation of the Oncoprotein ΔNp73α. PLoS Pathog. 2013, 9, e1003186. [Google Scholar] [CrossRef]
- Marshall, N.A.; Culligan, D.J.; Johnston, P.W.; Millar, C.; Barker, R.N.; Vickers, M. CD4+T-cell responses to Epstein–Barr virus (EBV) latent membrane protein 1 in infectious mononucleosis and EBV-associated non-Hodgkin lymphoma: Th1 in active disease but Tr1 in remission. Br. J. Haematol. 2007, 139, 81–89. [Google Scholar] [CrossRef]
- Anders, H.-J.; Zecher, D.; Pawar, R.D.; Patole, P.S. Molecular mechanisms of autoimmunity triggered by microbial infection. Thromb. Haemost. 2005, 7, 215–224. [Google Scholar] [CrossRef]
- Al Shamsi, M.; Shahin, A.; Iwakura, Y.; Lukic, M.L.; Mensah-Brown, E.P. Pam3CSK4 enhanced beta cell loss and diabetogenesis: The roles of IFN-gamma and IL-17. Clin. Immunol. 2013, 149, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, H.; Lou, X.; Qu, X.; Gao, L.; Liu, X.; Li, M.; Guo, H.; Jiang, Y. Decreased percentage of NKG2D+NK cells in patients with incident onset of Type 1 Diabetes. Clin. Exp. Pharmacol. Physiol. 2017, 44, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Latif, M.; Abdel-Moneim, A.A.; El-Hefnawy, M.H.; Khalil, R.G. Comparative and Correlative Assessments of Cytokine, Complement and Antibody Patterns in Paediatric Type 1 Diabetes. Clin. Exp. Immunol. 2017, 190, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Zareian, P.; Dizgah, I.M. Serum Interleukin 17 in Type 2 Diabetes Mellitus. J. Arch. Mil. Med. 2014, 2, e24689. [Google Scholar] [CrossRef]
- Russell, M.A.; Morgan, N.G. The impact of anti-inflammatory cytokines on the pancreatic β-cell. Islets 2014, 6, e950547. [Google Scholar] [CrossRef]
- Shih, Y.-L.; Ho, K.-T.; Tsao, C.-H.; Chang, Y.-H.; Shiau, M.-Y.; Huang, C.-N.; Yang, S.-C. Role of Cyotkines in Metabolism and Type 2 Diabetes Mellitus. Int. J. Biomed. Lab. 2013, 2, 1–16. [Google Scholar]
- Kaminski, A.; Kaminski, E.R.; Morgan, N.G. Pre-incubation with interleukin-4 mediates a direct protective effect against the loss of pancreatic β-cell viability induced by proinflammatory cytokines. Clin. Exp. Immunol. 2007, 148, 583–588. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Ho, K.-T.; Lu, S.-H.; Huang, C.-N.; Shiau, M.-Y. Regulation of glucose/lipid metabolism and insulin sensitivity by interleukin-4. Int. J. Obes. 2011, 36, 993–998. [Google Scholar] [CrossRef]
- Müller, S.; Martin, S.; Koenig, W.; Hanifi-Moghaddam, P.; Rathmann, W.; Haastert, B.; Giani, G.; Illig, T.; Thorand, B.; Kolb, H. Impaired glucose tolerance is associated with increased serum concentrations of interleukin 6 and co-regulated acute-phase proteins but not TNF-α or its receptors. Diabetologia 2002, 45, 805–812. [Google Scholar] [CrossRef]

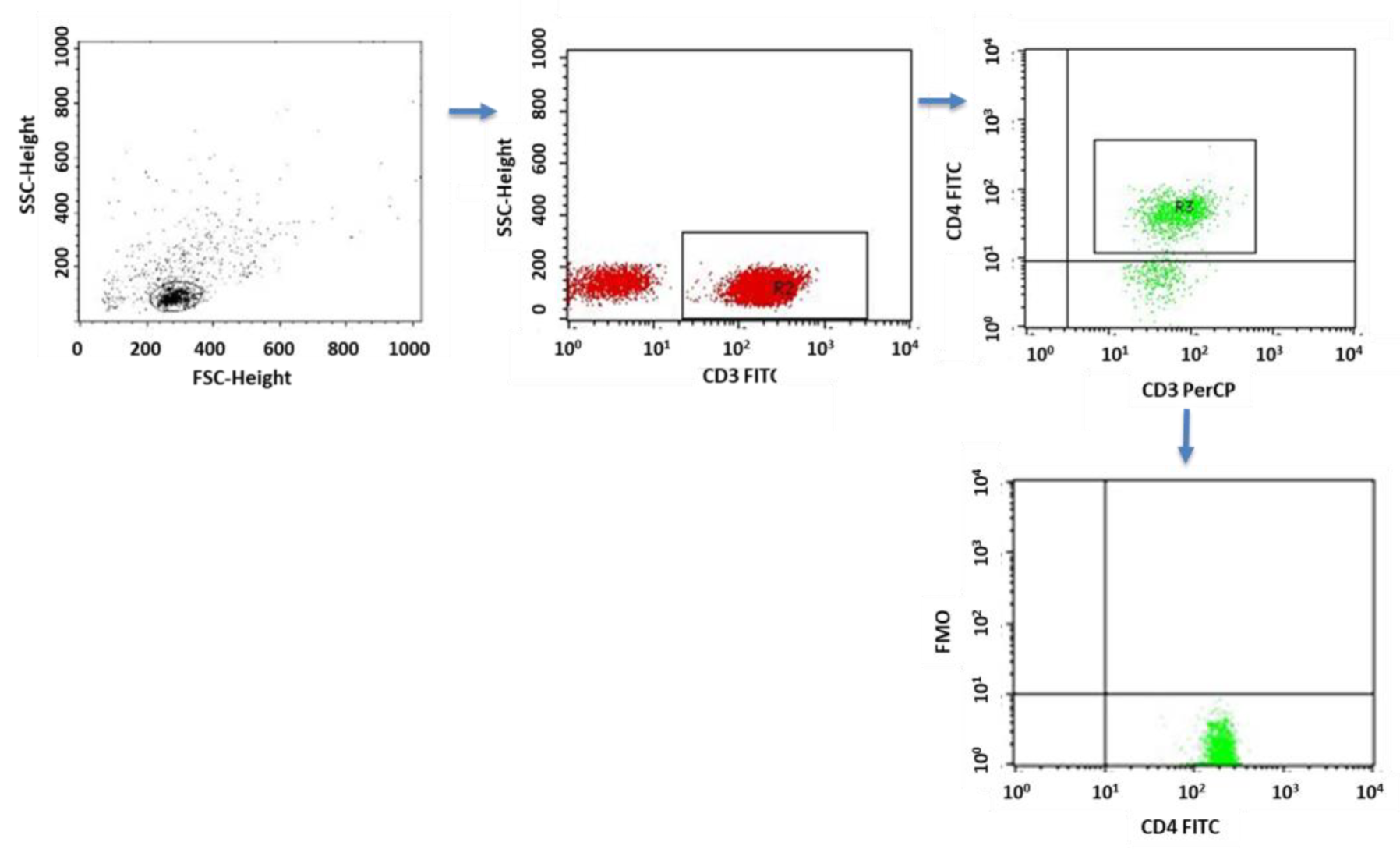
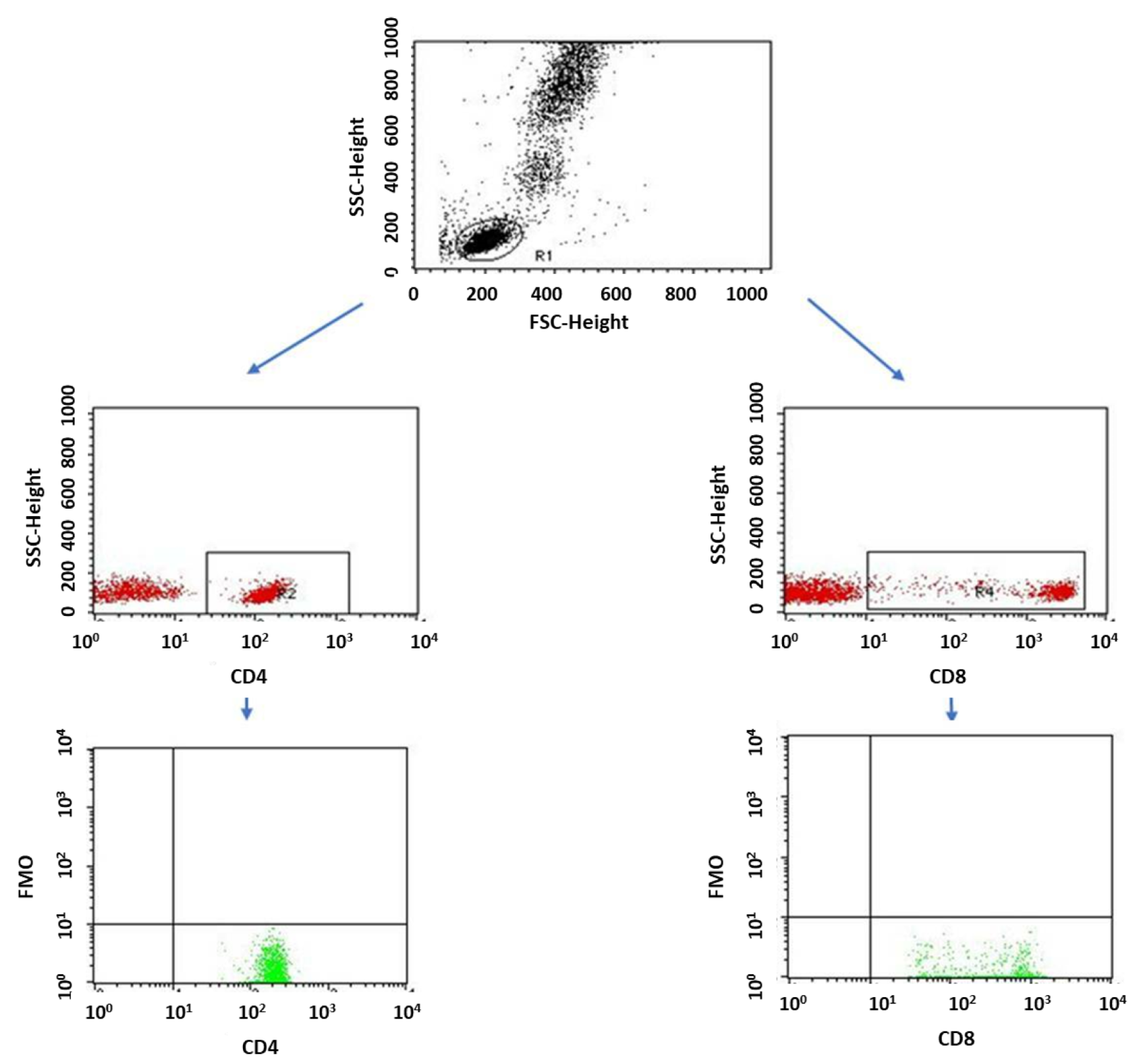
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| CD3+ T lymphocytes (%) | Study group | 71.93 | 71.18 | 60.69 | 83.83 | 6.24 | 0.74 |
| Control group | 70.22 | 68.43 | 58.64 | 85.21 | 7.72 | ||
| CD3+ T lymphocytes (103/mm3) | Study group | 1.53 | 1.44 | 0.38 | 2.71 | 0.45 | 0.52 |
| Control group | 1.79 | 1.63 | 1.07 | 2.73 | 0.50 | ||
| CD19+ B lymphocytes (%) | Study group | 13.39 | 13.38 | 7.12 | 20.35 | 3.47 | 0.31 |
| Control group | 11.92 | 12.38 | 5.82 | 15.73 | 2.80 | ||
| CD19+ B lymphocytes (103/mm3) | Study group | 0.28 | 0.30 | 0.06 | 0.47 | 0.09 | 0.43 |
| Control group | 0.32 | 0.33 | 0.16 | 0.53 | 0.11 | ||
| CD3+CD4+ T lymphocytes (%) | Study group | 39.30 | 39.79 | 20.90 | 50.36 | 6.31 | 0.29 |
| Control group | 36.91 | 36.78 | 26.88 | 44.85 | 5.28 | ||
| CD3+CD4+ T lymphocytes (103/mm3) | Study group | 0.85 | 0.81 | 0.21 | 1.58 | 0.28 | 0.38 |
| Control group | 0.95 | 0.82 | 0.53 | 1.52 | 0.34 | ||
| CD3+CD8+ T lymphocytes (%) | Study group | 26.68 | 26.23 | 16.08 | 55.79 | 7.05 | 0.47 |
| Control group | 26.17 | 25.13 | 17.40 | 36.55 | 5.22 | ||
| CD3+CD8+ T lymphocytes (103/mm3) | Study group | 0.58 | 0.54 | 0.17 | 1.13 | 0.23 | 0.79 |
| Control group | 0.67 | 0.68 | 0.41 | 0.99 | 0.18 | ||
| CD3+CD4+/ CD3+CD8+ ratio | Study group | 1.61 | 1.52 | 0.37 | 3.04 | 0.52 | 0.40 |
| Control group | 1.47 | 1.39 | 0.83 | 2.40 | 0.39 | ||
| NK CD3-CD16+CD56+ (%) | Study group | 12.21 | 10.42 | 2.10 | 25.19 | 5.57 | 0.71 |
| Control group | 14.18 | 13.08 | 6.43 | 25.15 | 6.65 | ||
| NK CD3-CD16+CD56+ (103/mm3) | Study group | 0.27 | 0.26 | 0.04 | 0.60 | 0.14 | 0.63 |
| Control group | 0.36 | 0.30 | 0.14 | 0.97 | 0.21 |
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| CD3+ CD69+ T lymphocytes (%) | Study group | 3.72 | 3.15 | 1.25 | 9.23 | 1.86 | 0.123 |
| Control group | 3.01 | 2.72 | 1.12 | 5.58 | 1.65 | ||
| CD19+ CD69+ B lymphocytes (%) | Study group | 12.43 | 9.87 | 1.52 | 48.77 | 10.47 | 0.245 |
| Control group | 9.79 | 7.53 | 2.49 | 20.96 | 6.28 | ||
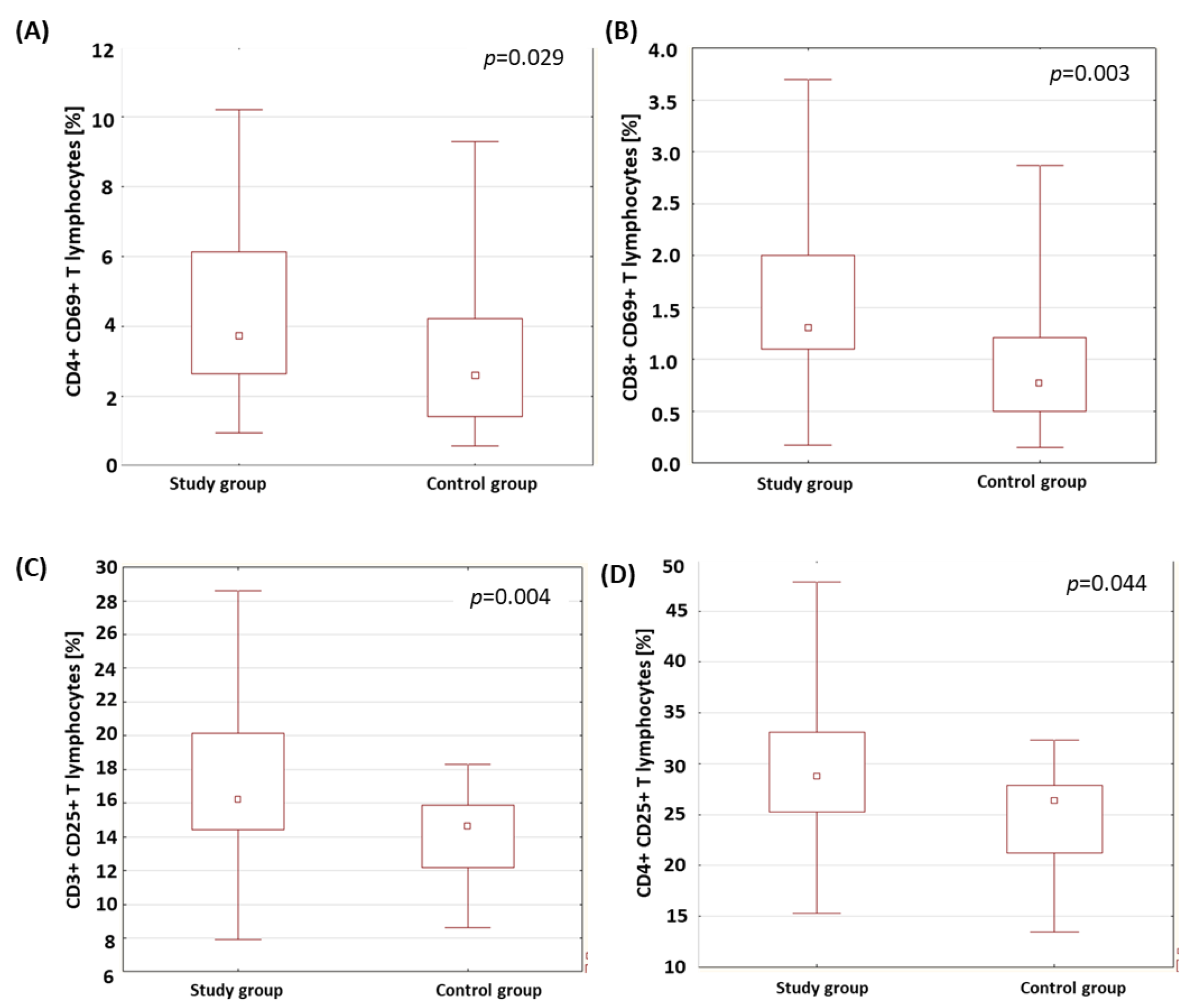
| CD4+ CD69+ T lymphocytes (%) | Study group | 4.53 | 3.72 | 0.95 | 10.20 | 2.55 | 0.029 * |
| Control group | 3.09 | 2.60 | 0.56 | 9.30 | 2.31 | ||
| CD8+ CD69+ T lymphocytes (%) | Study group | 1.58 | 1.30 | 0.17 | 3.70 | 0.86 | 0.003 * |
| Control group | 0.96 | 0.77 | 0.15 | 2.87 | 0.73 | ||
| CD3+ CD25+ T lymphocytes (%) | Study group | 17.60 | 16.22 | 7.93 | 28.60 | 4.91 | 0.004 * |
| Control group | 14.12 | 14.63 | 8.60 | 18.31 | 2.65 | ||
| CD19+ CD25+ B lymphocytes (%) | Study group | 20.38 | 19.18 | 7.85 | 38.16 | 6.88 | 0.219 |
| Control group | 19.87 | 19.77 | 11.24 | 31.29 | 5.22 | ||
| CD4+ CD25+ T lymphocytes (%) | Study group | 29.26 | 28.73 | 15.29 | 47.83 | 7.40 | 0.044 * |
| Control group | 24.80 | 26.33 | 13.45 | 32.30 | 5.79 | ||
| CD8+ CD25+ T lymphocytes (%) | Study group | 1.40 | 1.03 | 0.46 | 6.49 | 1.15 | 0.139 |
| Control group | 1.05 | 0.92 | 0.40 | 2.19 | 0.55 |
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| CD3+ CD4+IFN-γ+ T lymphocytes (%) | Study group | 1.38 | 0.94 | 0.12 | 7.26 | 1.54 | 0.236 |
| Control group | 1.76 | 1.30 | 0.12 | 4.19 | 1.29 | ||
| CD3+ CD4+IL-2+ T lymphocytes (%) | Study group | 2.99 | 2.32 | 0.11 | 11.83 | 2.80 | 0.187 |
| Control group | 2.93 | 2.71 | 0.29 | 5.38 | 1.50 | ||
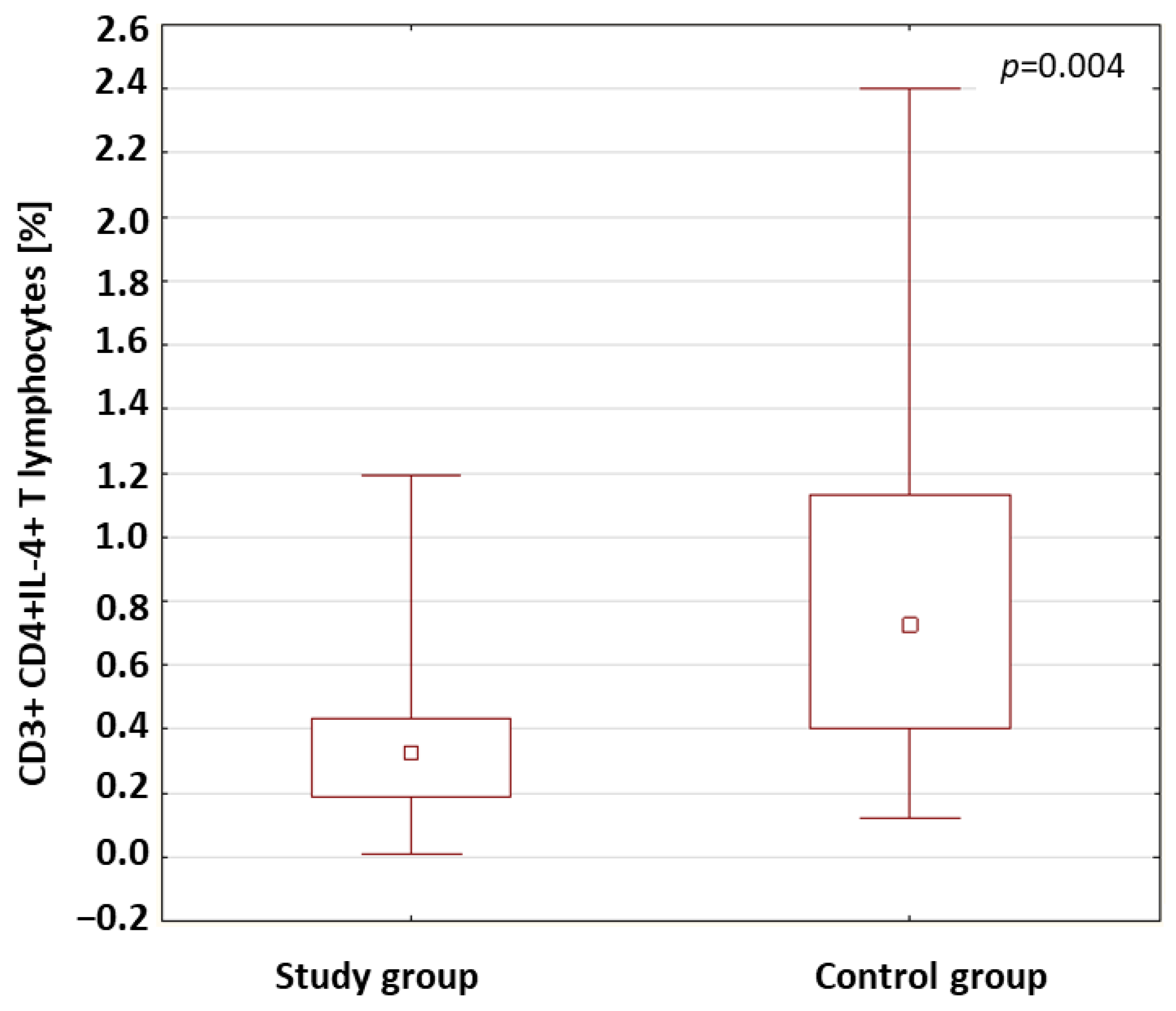
| CD3+ CD4+IL-4+ T lymphocytes (%) | Study group | 0.40 | 0.36 | 0.11 | 1.19 | 0.24 | 0.004 * |
| Control group | 0.84 | 0.72 | 0.12 | 2.40 | 0.64 | ||
| CD3+ CD4+IL-10+ T lymphocytes (%) | Study group | 0.17 | 0.13 | 0.10 | 0.80 | 0.12 | 0.312 |
| Control group | 0.19 | 0.15 | 0.10 | 0.51 | 0.12 | ||
| CD3+ CD4+IL-17+ T lymphocytes (%) | Study group | 0.37 | 0.33 | 0.11 | 1.33 | 0.28 | 0.411 |
| Control group | 0.46 | 0.29 | 0.14 | 2.20 | 0.54 |
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| VCA IgM (U/mL) | Study group | 20.63 | 18.51 | 13.21 | 37.18 | 6.68 | - |
| Control group | – | – | – | – | – | ||
| VCA IgG (U/mL) | Study group | 65.97 | 51.49 | 16.88 | 252.76 | 53.29 | 0.364 |
| Control group | 38.51 | 32.70 | 14.82 | 84.97 | 19.90 | ||
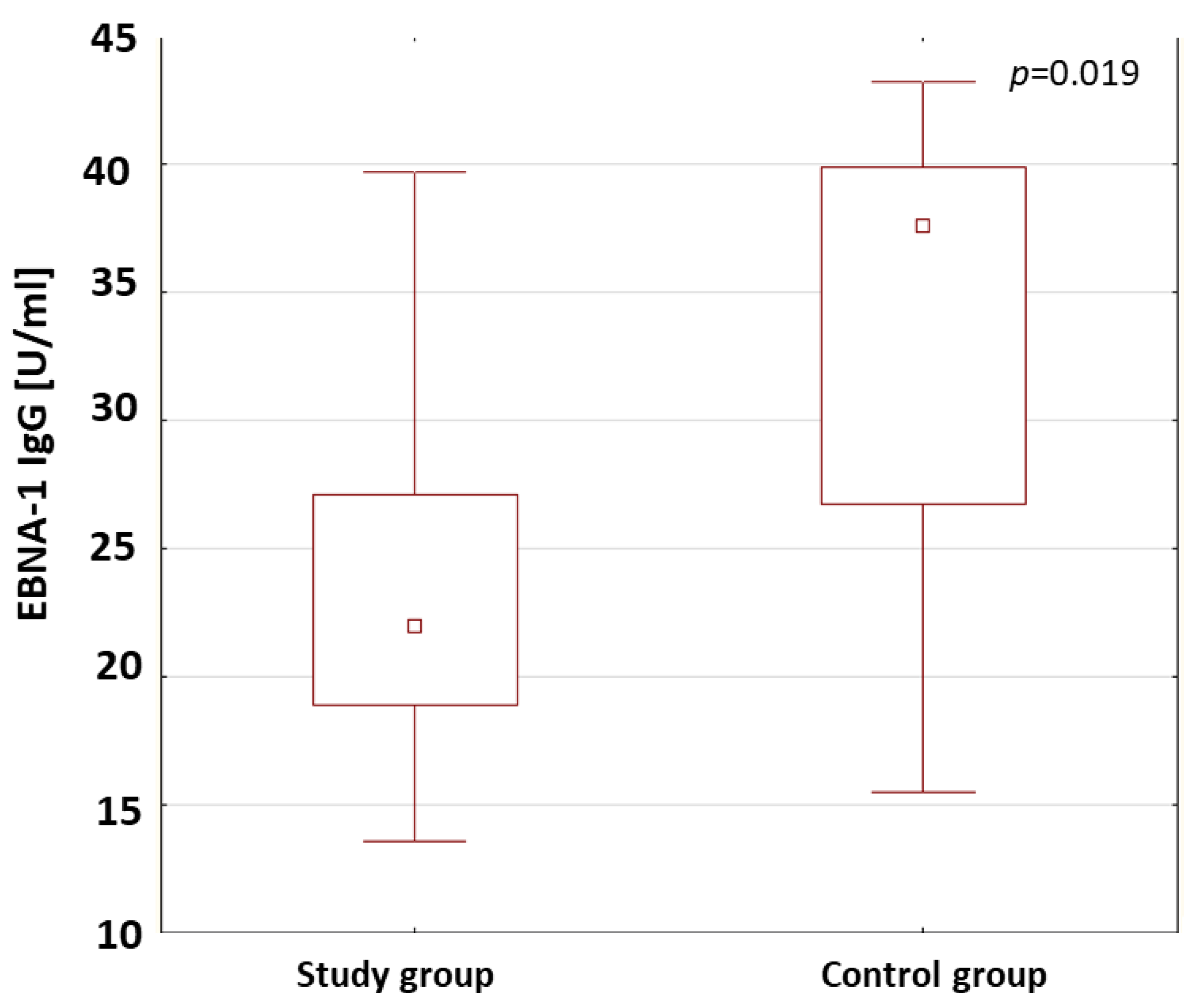
| EBNA-1 IgG (U/mL) | Study group | 23.94 | 21.94 | 13.60 | 39.68 | 7.69 | 0.019 * |
| Control group | 34.16 | 37.58 | 15.49 | 43.20 | 9.72 |
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| CD3+ T lymphocytes (%) | VCA IgM (+) | 71.79 | 71.17 | 60.69 | 83.83 | 7.23 | 0.109 |
| VCA IgM (−) | 72.40 | 71.38 | 64.02 | 81.91 | 4.43 | ||
| VCA IgG (+) | 72.45 | 72.96 | 60.69 | 83.83 | 6.43 | 0.297 | |
| VCA IgG (−) | 71.13 | 70.41 | 62.31 | 82.84 | 5.83 | ||
| EBNA-1 IgG (+) | 71.03 | 70.78 | 60.69 | 83.83 | 7.01 | 0.147 | |
| EBNA-1 IgG (−) | 72.99 | 73.34 | 62.31 | 82.84 | 5.31 | ||
| CD3+ T lymphocytes (103/mm3) | VCA IgM (+) | 1.60 | 1.47 | 0.96 | 2.71 | 0.47 | 0.264 |
| VCA IgM (−) | 1.41 | 1.48 | 0.38 | 2.02 | 0.39 | ||
| VCA IgG (+) | 1.58 | 1.52 | 0.38 | 2.71 | 0.47 | 0.144 | |
| VCA IgG (−) | 1.38 | 1.26 | 1.07 | 2.02 | 0.33 | ||
| EBNA-1 IgG (+) | 1.54 | 1.51 | 0.38 | 2.71 | 0.48 | 0.321 | |
| EBNA-1 IgG (−) | 1.56 | 1.38 | 1.07 | 2.58 | 0.41 | ||
| CD19+ B lymphocytes (%) | VCA IgM (+) | 13.47 | 13.32 | 7.26 | 20.35 | 3.64 | 0.122 |
| VCA IgM (−) | 13.18 | 13.38 | 7.12 | 18.66 | 3.20 | ||
| VCA IgG (+) | 13.01 | 13.10 | 7.12 | 20.35 | 3.41 | 0.302 | |
| VCA IgG (−) | 14.11 | 14.07 | 8.34 | 19.72 | 3.49 | ||
| EBNA-1 IgG (+) | 13.70 | 14.01 | 7.78 | 20.35 | 3.19 | 0.104 | |
| EBNA-1 IgG (−) | 13.02 | 13.32 | 7.12 | 19.72 | 3.70 | ||
| CD19+ B lymphocytes (103/mm3) | VCA IgM (+) | 0.29 | 0.30 | 0.13 | 0.47 | 0.08 | 0.210 |
| VCA IgM (−) | 0.26 | 0.27 | 0.06 | 0.40 | 0.10 | ||
| VCA IgG (+) | 0.28 | 0.29 | 0.06 | 0.47 | 0.09 | 0.410 | |
| VCA IgG (−) | 0.28 | 0.29 | 0.13 | 0.40 | 0.09 | ||
| EBNA-1 IgG (+) | 0.30 | 0.30 | 0.06 | 0.47 | 0.09 | 0.108 | |
| EBNA-1 IgG (−) | 0.25 | 0.24 | 0.13 | 0.04 | 0.09 | ||
| CD3+CD4+ T lymphocytes (%) | VCA IgM (+) | 39.18 | 39.92 | 20.90 | 50.36 | 7.44 | 0.212 |
| VCA IgM (−) | 39.52 | 39.79 | 32.89 | 47.73 | 3.92 | ||
| VCA IgG (+) | 39.05 | 39.32 | 20.90 | 48.64 | 5.92 | 0.134 | |
| VCA IgG (−) | 40.05 | 39.92 | 30.78 | 50.36 | 7.30 | ||
| EBNA-1 IgG (+) | 39.84 | 39.92 | 30.25 | 48.64 | 4.97 | 0.120 | |
| EBNA-1 IgG (−) | 38.67 | 38.25 | 20.90 | 50.36 | 7.62 | ||
| CD3+CD4+ T lymphocytes (103/mm3) | VCA IgM (+) | 0.89 | 0.79 | 0.40 | 1.58 | 0.30 | 0.142 |
| VCA IgM (−) | 0.78 | 0.81 | 0.21 | 1.07 | 0.22 | ||
| VCA IgG (+) | 0.87 | 0.81 | 0.21 | 1.58 | 0.28 | 0.219 | |
| VCA IgG (−) | 0.79 | 0.75 | 0.49 | 1.37 | 0.26 | ||
| EBNA-1 IgG (+) | 0.87 | 0.83 | 0.21 | 1.58 | 0.28 | 0.106 | |
| EBNA-1 IgG (−) | 0.82 | 0.76 | 0.40 | 1.37 | 0.26 | ||
| CD3+CD8+ T lymphocytes (%) | VCA IgM (+) | 26.36 | 26.44 | 16.08 | 55.79 | 8.06 | 0.117 |
| VCA IgM (−) | 26.57 | 25.84 | 16.34 | 38.32 | 5.68 | ||
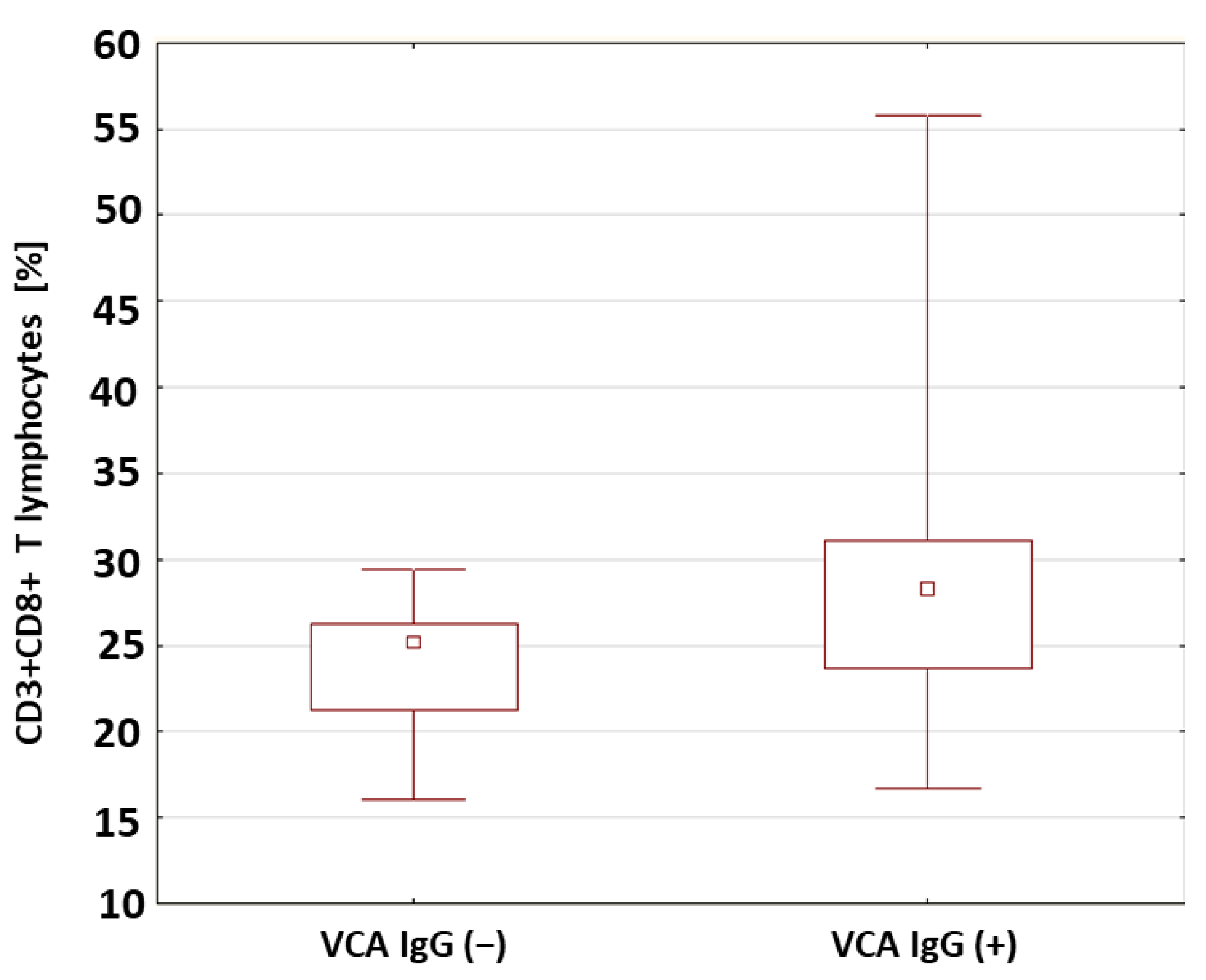
| VCA IgG (+) | 27.90 | 28.21 | 16.34 | 55.79 | 7.63 | 0.033 * | |
| VCA IgG (−) | 23.06 | 24.44 | 16.08 | 29.42 | 4.47 | ||
| EBNA-1 IgG (+) | 25.82 | 27.09 | 16.34 | 37.54 | 5.47 | 0.202 | |
| EBNA-1 IgG (−) | 27.04 | 26.05 | 16.08 | 55.79 | 8.53 | ||
| CD3+CD8+ T lymphocytes (103/mm3) | VCA IgM (+) | 0.61 | 0.55 | 0.31 | 1.13 | 0.24 | 0.114 |
| VCA IgM (−) | 0.52 | 0.54 | 0.17 | 0.85 | 0.20 | ||
| VCA IgG (+) | 0.62 | 0.59 | 0.17 | 1.13 | 0.24 | 0.041 * | |
| VCA IgG (−) | 0.45 | 0.45 | 0.31 | 0.78 | 0.14 | ||
| EBNA-1 IgG (+) | 0.57 | 0.55 | 0.17 | 0.94 | 0.21 | 0.147 | |
| EBNA-1 IgG (−) | 0.58 | 0.49 | 0.31 | 1.13 | 0.25 | ||
| CD3+CD4+/CD3+CD8+ ratio | VCA IgM (+) | 1.65 | 1.54 | 0.37 | 3.04 | 0.59 | 0.216 |
| VCA IgM (−) | 1.58 | 1.50 | 0.92 | 2.44 | 0.42 | ||
| VCA IgG (+) | 1.52 | 1.48 | 0.37 | 2.52 | 0.47 | 0.302 | |
| VCA IgG (−) | 1.87 | 1.78 | 1.05 | 3.04 | 0.59 | ||
| EBNA-1 IgG (+) | 1.62 | 1.53 | 1.06 | 2.52 | 0.43 | 0.155 | |
| EBNA-1 IgG (−) | 1.63 | 1.52 | 0.37 | 3.04 | 0.62 | ||
| NK CD3-CD16+CD56+ (%) | VCA IgM (+) | 12.56 | 10.63 | 3.39 | 25.19 | 6.00 | 0.163 |
| VCA IgM (−) | 11.58 | 10.15 | 2.10 | 18.26 | 4.88 | ||
| VCA IgG (+) | 12.12 | 10.21 | 2.10 | 25.19 | 5.89 | 0.143 | |
| VCA IgG (−) | 12.47 | 10.63 | 6.58 | 18.20 | 4.81 | ||
| EBNA-1 IgG (+) | 12.90 | 14.61 | 2.10 | 25.19 | 6.63 | 0.125 | |
| EBNA-1 IgG (−) | 11.42 | 9.89 | 5.93 | 18.26 | 4.16 | ||
| NK CD3-CD16+CD56+ (103/mm3) | VCA IgM (+) | 0.29 | 0.27 | 0.08 | 0.60 | 0.14 | 0.120 |
| VCA IgM (−) | 0.23 | 0.19 | 0.04 | 0.48 | 0.14 | ||
| VCA IgG (+) | 0.26 | 0.25 | 0.04 | 0.60 | 0.15 | 0.169 | |
| VCA IgG (−) | 0.29 | 0.28 | 0.17 | 0.48 | 0.12 | ||
| EBNA-1 IgG (+) | 0.28 | 0.26 | 0.04 | 0.60 | 0.16 | 0.208 | |
| EBNA-1 IgG (−) | 0.25 | 0.26 | 0.11 | 0.48 | 0.11 |
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| CD3+ CD69+ T lymphocytes (%) | VCA IgM (+) | 3.59 | 3.09 | 1.65 | 9.23 | 1.80 | 0.078 |
| VCA IgM (−) | 3.86 | 3.28 | 1.25 | 8.14 | 1.95 | ||
| VCA IgG (+) | 4.03 | 3.68 | 1.65 | 9.23 | 1.95 | 0.093 | |
| VCA IgG (−) | 2.81 | 2.69 | 1.25 | 5.74 | 1.17 | ||
| EBNA-1 IgG (+) | 4.22 | 3.72 | 1.65 | 9.23 | 2.11 | 0.141 | |
| EBNA-1 IgG (−) | 3.06 | 2.82 | 1.25 | 5.74 | 1.22 | ||
| CD19+ CD69+ B lymphocytes (%) | VCA IgM (+) | 12.56 | 8.91 | 1.52 | 48.77 | 10.45 | 0.079 |
| VCA IgM (−) | 11.82 | 9.66 | 1.62 | 42.05 | 10.63 | ||
| VCA IgG (+) | 12.18 | 10.07 | 1.62 | 42.05 | 9.27 | 0.103 | |
| VCA IgG (−) | 12.43 | 6.17 | 1.52 | 48.77 | 13.22 | ||
| EBNA-1 IgG (+) | 13.14 | 11.05 | 2.10 | 42.05 | 9.45 | 0.089 | |
| EBNA-1 IgG (−) | 11.22 | 6.11 | 1.52 | 48.77 | 11.58 | ||
| CD4+ CD69+ T lymphocytes (%) | VCA IgM (+) | 4.72 | 3.72 | 1.41 | 10.20 | 2.82 | 0.116 |
| VCA IgM (−) | 4.13 | 3.50 | 0.95 | 7.96 | 2.14 | ||
| VCA IgG (+) | 4.31 | 3.59 | 0.95 | 10.20 | 2.19 | 0.217 | |
| VCA IgG (−) | 4.85 | 3.64 | 1.41 | 10.13 | 3.36 | ||
| EBNA-1 IgG (+) | 4.65 | 4.51 | 0.95 | 10.20 | 2.25 | 0.269 | |
| EBNA-1 IgG (−) | 4.29 | 3.37 | 1.35 | 10.13 | 2.88 | ||
| CD8+ CD69+ T lymphocytes (%) | VCA IgM (+) | 1.72 | 1.28 | 0.43 | 3.70 | 0.98 | 0.077 |
| VCA IgM (−) | 1.39 | 1.31 | 0.17 | 2.87 | 0.62 | ||
| VCA IgG (+) | 1.42 | 1.23 | 0.17 | 3.25 | 0.78 | 0.029 * | |
| VCA IgG (−) | 1.97 | 1.68 | 1.15 | 3.70 | 0.91 | ||
| EBNA-1 IgG (+) | 1.58 | 1.24 | 0.43 | 3.25 | 0.81 | 0.069 | |
| EBNA-1 IgG (−) | 1.57 | 1.31 | 0.17 | 3.70 | 0.91 | ||
| CD3+ CD25+ T lymphocytes (%) | VCA IgM (+) | 17.09 | 15.47 | 7.93 | 27.02 | 4.57 | 0.122 |
| VCA IgM (−) | 18.19 | 17.40 | 9.39 | 28.60 | 5.32 | ||
| VCA IgG (+) | 17.64 | 15.89 | 9.39 | 28.60 | 4.42 | 0.099 | |
| VCA IgG (−) | 17.35 | 18.70 | 7.93 | 27.57 | 5.97 | ||
| EBNA-1 IgG (+) | 18.39 | 16.77 | 13.29 | 28.60 | 4.45 | 0.178 | |
| EBNA-1 IgG (−) | 16.67 | 15.47 | 7.93 | 27.57 | 5.23 | ||
| CD19+ CD25+ B lymphocytes (%) | VCA IgM (+) | 20.59 | 19.96 | 11.91 | 30.00 | 5.47 | 0.129 |
| VCA IgM (−) | 20.08 | 17.75 | 7.85 | 38.16 | 8.42 | ||
| VCA IgG (+) | 20.70 | 18.64 | 11.35 | 38.16 | 7.05 | 0.143 | |
| VCA IgG (−) | 19.64 | 19.96 | 7.85 | 28.93 | 6.42 | ||
| EBNA-1 IgG (+) | 20.45 | 18.91 | 11.92 | 31.62 | 5.74 | 0.147 | |
| EBNA-1 IgG (−) | 20.29 | 19.96 | 7.85 | 38.16 | 7.96 | ||
| CD4+ CD25+ T lymphocytes (%) | VCA IgM (+) | 28.26 | 28.26 | 15.29 | 44.60 | 6.66 | 0.096 |
| VCA IgM (−) | 30.38 | 29.85 | 18.25 | 47.83 | 8.20 | ||
| VCA IgG (+) | 29.79 | 29.24 | 18.80 | 44.60 | 6.25 | 0.154 | |
| VCA IgG (−) | 27.69 | 26.76 | 15.29 | 47.83 | 9.55 | ||
| EBNA-1 IgG (+) | 30.56 | 29.73 | 21.67 | 44.6 | 6.42 | 0.312 | |
| EBNA-1 IgG (−) | 27.61 | 27.92 | 15.29 | 47.83 | 8.11 | ||
| CD8+ CD25+ T lymphocytes (%) | VCA IgM (+) | 1.42 | 1.11 | 0.46 | 4.09 | 0.91 | 0.088 |
| VCA IgM (−) | 1.39 | 0.96 | 0.56 | 6.49 | 1.43 | ||
| VCA IgG (+) | 1.40 | 1.02 | 0.4 | 6.49 | 1.26 | 0.073 | |
| VCA IgG (−) | 1.42 | 1.21 | 0.65 | 2.85 | 0.76 | ||
| EBNA-1 IgG (+) | 1.07 | 0.91 | 0.46 | 2.60 | 0.56 | 0.042 * | |
| EBNA-1 IgG (−) | 1.82 | 1.33 | 0.65 | 6.49 | 1.51 |
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| CD3+ CD4+IFN-γ+ T lymphocytes (%) | VCA IgM (+) | 1.36 | 1.02 | 0.12 | 6.47 | 1.46 | 0.087 |
| VCA IgM (−) | 1.36 | 0.76 | 0.31 | 7.26 | 1.71 | ||
| VCA IgG (+) | 1.34 | 1.08 | 0.12 | 7.26 | 1.39 | 0.302 | |
| VCA IgG (−) | 1.39 | 0.65 | 0.19 | 6.47 | 1.87 | ||
| EBNA-1 IgG (+) | 1.423 | 1.10 | 0.14 | 7.26 | 1.51 | 0.078 | |
| EBNA-1 IgG (−) | 1.29 | 0.75 | 0.12 | 6.47 | 1.59 | ||
| CD3+ CD4+IL-2+ T lymphocytes (%) | VCA IgM (+) | 3.16 | 2.89 | 0.12 | 11.83 | 2.93 | 0.069 |
| VCA IgM (−) | 2.55 | 1.10 | 0.11 | 6.70 | 2.59 | ||
| VCA IgG (+) | 3.19 | 3.11 | 0.11 | 6.70 | 2.54 | 0.110 | |
| VCA IgG (−) | 2.44 | 1.05 | 0.12 | 11.83 | 3.29 | ||
| EBNA-1 IgG (+) | 3.51 | 3.20 | 0.12 | 6.45 | 2.32 | 0.118 | |
| EBNA-1 IgG (−) | 2.43 | 0.96 | 0.11 | 11.83 | 3.12 | ||
| CD3+ CD4+IL-4+ T lymphocytes (%) | VCA IgM (+) | 0.39 | 0.37 | 0.11 | 0.81 | 0.18 | 0.094 |
| VCA IgM (−) | 0.43 | 0.30 | 0.11 | 1.19 | 0.35 | ||
| VCA IgG (+) | 0.42 | 0.39 | 0.11 | 1.19 | 0.26 | 0.103 | |
| VCA IgG (−) | 0.36 | 0.32 | 0.11 | 0.88 | 0.20 | ||
| EBNA-1 IgG (+) | 0.41 | 0.38 | 0.11 | 0.86 | 0.22 | 0.159 | |
| EBNA-1 IgG (−) | 0.39 | 0.33 | 0.11 | 1.19 | 0.27 | ||
| CD3+ CD4+IL-10+ T lymphocytes (%) | VCA IgM (+) | 0.18 | 0.12 | 0.10 | 0.80 | 0.14 | 0.210 |
| VCA IgM (−) | 0.15 | 0.13 | 0.10 | 0.34 | 0.07 | ||
| VCA IgG (+) | 0.19 | 0.18 | 0.10 | 0.80 | 0.13 | 0.011 * | |
| VCA IgG (−) | 0.12 | 0.10 | 0.10 | 0.16 | 0.02 | ||
| EBNA-1 IgG (+) | 0.20 | 0.18 | 0.10 | 0.80 | 0.15 | 0.033 * | |
| EBNA-1 IgG (−) | 0.13 | 0.10 | 0.10 | 0.23 | 0.04 | ||
| CD3+ CD4+IL-17+ T lymphocytes (%) | VCA IgM (+) | 0.38 | 0.33 | 0.11 | 1.33 | 0.30 | 0.113 |
| VCA IgM (−) | 0.36 | 0.32 | 0.12 | 1.02 | 0.24 | ||
| VCA IgG (+) | 0.39 | 0.35 | 0.11 | 1.02 | 0.20 | 0.030 * | |
| VCA IgG (−) | 0.34 | 0.15 | 0.12 | 1.33 | 0.40 | ||
| EBNA-1 IgG (+) | 0.36 | 0.33 | 0.11 | 1.02 | 0.22 | 0.099 | |
| EBNA-1 IgG (−) | 0.38 | 0.32 | 0.12 | 1.33 | 0.33 |
| Parameter | Group | Mean | Median | Minimum | Maximum | SD | p-Value |
|---|---|---|---|---|---|---|---|
| Fructosamine (µmol/L) | VCA IgM (+) | 501.74 | 458.00 | 257.00 | 962.00 | 179.38 | 0.089 |
| VCA IgM (−) | 541.94 | 576.50 | 220.00 | 921.00 | 181.97 | ||
| VCA IgG (+) | 596.37 | 600.00 | 340.00 | 962.00 | 165.98 | <0.001 * | |
| VCA IgG (−) | 370.93 | 359.50 | 220.00 | 520.00 | 88.68 | ||
| EBNA-1 IgG (+) | 588.79 | 580.00 | 340.00 | 962.00 | 173.95 | 0.015 * | |
| EBNA-1 IgG (−) | 459.45 | 415.00 | 220.00 | 921.00 | 165.03 | ||
| HbA1c level (%) | VCA IgM (+) | 8.69 | 8.70 | 5.00 | 13.80 | 2.11 | 0.073 |
| VCA IgM (−) | 9.71 | 9.70 | 6.20 | 15.00 | 2.24 | ||
| VCA IgG (+) | 9.62 | 9.50 | 5.80 | 15.00 | 2.20 | 0.017 * | |
| VCA IgG (−) | 7.80 | 8.10 | 5.00 | 10.00 | 1.60 | ||
| EBNA-1 IgG (+) | 9.42 | 8.90 | 5.80 | 15.00 | 2.42 | 0.066 | |
| EBNA-1 IgG (−) | 8.75 | 8.85 | 5.00 | 13.80 | 1.95 |
| Parameter | Study Group (n = 43) | Control Group (n = 20) | |
|---|---|---|---|
| Sex | Girls | 21 (48.8%) | 8 (40%) |
| Boys | 22 (51.2%) | 12 (60%) | |
| Age | Mean ± SD | 13.59 ± 3.55 | 11.02 ± 2.49 |
| Median (minimum–maximum) | 14 (3.9–18.0) | 10.85 (7.75–14.4) | |
| BMI | Mean ± SD | 20.13 ± 3.63 | 15.83 ± 1.67 |
| Median (minimum–maximum) | 19.94 (11.19–31.31) | 15.9 (12.72–18.5) | |
| Leukocytosis (103/µL) | Mean ± SD | 6.59 ± 2.05 | 6.59 ± 0.99 |
| Median (minimum–maximum) | 5.85 (3.63–12.02) | 6.22 (5.15–8.64) | |
| Lymphocytosis (103/µL) | Mean ± SD | 2.14 ± 0.56 | 2.64 ± 0.74 |
| Median (minimum–maximum) | 2.03 (0.54–3.62) | 2.39 (1.68–4.34) | |
| Monocytosis (103/µL) | Mean ± SD | 0.58 ± 0.38 | 0.57 ± 0.12 |
| Median (minimum–maximum) | 0.52 (0.20–2.63) | 0.55 (0.39–0.80) | |
| Neutrocytosis (103/µL) | Mean ± SD | 3.60 ± 2.01 | 3.16 ± 0.89 |
| Median (minimum–maximum) | 2.93 (0.35–9.62) | 3.16 (1.27–4.60) | |
| Erythrocytes (106/µL) | Mean ± SD | 4.95 ± 0.42 | 4.71 ± 0.36 |
| Median (minimum-maximum) | 4.89 (4.26–6.02) | 6.64 (4.23–5.61) | |
| Hemoglobin (g/dL) | Mean ± SD | 14.26 ± 1.25 | 13.29 ± 0.93 |
| Median (minimum–maximum) | 14.00 (11.90–17.10) | 13.20 (11.90–16.00) | |
| Hematocrit (%) | Mean ± SD | 40.37 ± 3.33 | 37.99 ± 2.73 |
| Median (minimum–maximum) | 40.70 (32.90–48.50) | 38.00 (33.70–46.00) | |
| Platelets (103/µL) | Mean ± SD | 251.13 ± 46.36 | 326.65 ± 58.43 |
| Median (minimum–maximum) | 255.00 (154.00–361.00) | 321.00 (241.00–443.00) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klatka, M.; Rysz, I.; Hymos, A.; Polak, A.; Mertowska, P.; Mertowski, S.; Smolak, K.; Grywalska, E. Effect of Epstein–Barr Virus Infection on Selected Immunological Parameters in Children with Type 1 Diabetes. Int. J. Mol. Sci. 2023, 24, 2392. https://doi.org/10.3390/ijms24032392
Klatka M, Rysz I, Hymos A, Polak A, Mertowska P, Mertowski S, Smolak K, Grywalska E. Effect of Epstein–Barr Virus Infection on Selected Immunological Parameters in Children with Type 1 Diabetes. International Journal of Molecular Sciences. 2023; 24(3):2392. https://doi.org/10.3390/ijms24032392
Chicago/Turabian StyleKlatka, Maria, Izabela Rysz, Anna Hymos, Agnieszka Polak, Paulina Mertowska, Sebastian Mertowski, Konrad Smolak, and Ewelina Grywalska. 2023. "Effect of Epstein–Barr Virus Infection on Selected Immunological Parameters in Children with Type 1 Diabetes" International Journal of Molecular Sciences 24, no. 3: 2392. https://doi.org/10.3390/ijms24032392
APA StyleKlatka, M., Rysz, I., Hymos, A., Polak, A., Mertowska, P., Mertowski, S., Smolak, K., & Grywalska, E. (2023). Effect of Epstein–Barr Virus Infection on Selected Immunological Parameters in Children with Type 1 Diabetes. International Journal of Molecular Sciences, 24(3), 2392. https://doi.org/10.3390/ijms24032392






