Dual Role of B Cells in Multiple Sclerosis
Abstract
1. Introduction
2. Dual Role of B Cells in MS
2.1. Inflammatory Subsets of B Cells
2.2. Anti-Inflammatory Subsets of B Cells
3. Therapies That Restrict Inflammatory B Cells and Augment Anti-Inflammatory B Cells in MS
3.1. Anti-CD20 Therapies
3.2. Interferons
3.3. Dimethyl Fumarate
3.4. Fingolimod
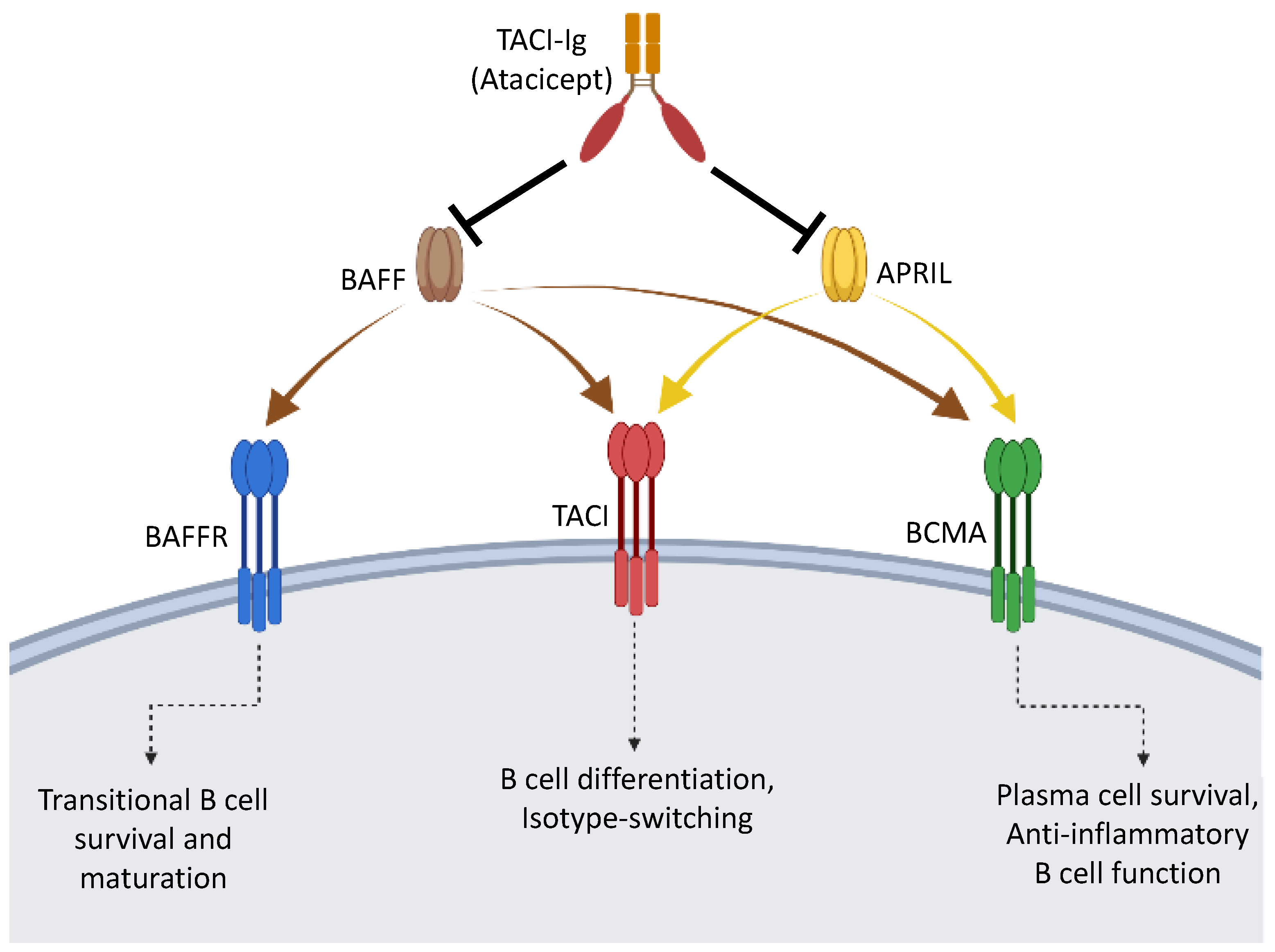
4. Anti-Inflammatory Role of BAFF and APRIL on B Cells
4.1. Blockade of BAFF and APRIL Exacerbates MS
4.2. Effects of BAFF and APRIL Signaling in Neuro-Inflammation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frohman, E.M.; Racke, M.K.; Raine, C.S. Multiple sclerosis—The plaque and its pathogenesis. N. Engl. J. Med. 2006, 354, 942–955. [Google Scholar] [CrossRef]
- Koch-Henriksen, N.; Sorensen, P.S. The changing demographic pattern of multiple sclerosis epidemiology. Lancet Neurol. 2010, 9, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Hafler, D.A. Multiple sclerosis. J. Clin. Investig. 2004, 113, 788–794. [Google Scholar] [CrossRef]
- Lowenthal, A.; Vansande, M.; Karcher, D. The differential diagnosis of neurological diseases by fractionating electrophoretically the CSF gamma-globulins. J. Neurochem. 1960, 6, 51–56. [Google Scholar] [CrossRef] [PubMed]
- von Budingen, H.C.; Bar-Or, A.; Zamvil, S.S. B cells in multiple sclerosis: Connecting the dots. Curr. Opin. Immunol. 2011, 23, 713–720. [Google Scholar] [CrossRef]
- Hauser, S.L.; Waubant, E.; Arnold, D.L.; Vollmer, T.; Antel, J.; Fox, R.J.; Bar-Or, A.; Panzara, M.; Sarkar, N.; Agarwal, S.; et al. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. N. Engl. J. Med. 2008, 358, 676–688. [Google Scholar] [CrossRef] [PubMed]
- Kappos, L.; Li, D.; Calabresi, P.A.; O’Connor, P.; Bar-Or, A.; Barkhof, F.; Yin, M.; Leppert, D.; Glanzman, R.; Tinbergen, J.; et al. Ocrelizumab in relapsing-remitting multiple sclerosis: A phase 2, randomised, placebo-controlled, multicentre trial. Lancet 2011, 378, 1779–1787. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S.D.; Dittel, B.N.; Hardardottir, F.; Janeway, C.A., Jr. Experimental autoimmune encephalomyelitis induction in genetically B cell-deficient mice. J. Exp. Med. 1996, 184, 2271–2278. [Google Scholar] [CrossRef]
- Matsushita, T.; Fujimoto, M.; Hasegawa, M.; Komura, K.; Takehara, K.; Tedder, T.F.; Sato, S. Inhibitory role of CD19 in the progression of experimental autoimmune encephalomyelitis by regulating cytokine response. Am. J. Pathol. 2006, 168, 812–821. [Google Scholar] [CrossRef] [PubMed]
- Kappos, L.; Hartung, H.P.; Freedman, M.S.; Boyko, A.; Radu, E.W.; Mikol, D.D.; Lamarine, M.; Hyvert, Y.; Freudensprung, U.; Plitz, T.; et al. Atacicept in multiple sclerosis (ATAMS): A randomised, placebo-controlled, double-blind, phase 2 trial. Lancet Neurol. 2014, 13, 353–363. [Google Scholar] [CrossRef]
- Castillo-Trivino, T.; Braithwaite, D.; Bacchetti, P.; Waubant, E. Rituximab in relapsing and progressive forms of multiple sclerosis: A systematic review. PLoS ONE 2013, 8, e66308. [Google Scholar] [CrossRef] [PubMed]
- Sanz, I.; Wei, C.; Jenks, S.A.; Cashman, K.S.; Tipton, C.; Woodruff, M.C.; Hom, J.; Lee, F.E. Challenges and Opportunities for Consistent Classification of Human B Cell and Plasma Cell Populations. Front. Immunol. 2019, 10, 2458. [Google Scholar] [CrossRef] [PubMed]
- Schubert, R.D.; Hu, Y.; Kumar, G.; Szeto, S.; Abraham, P.; Winderl, J.; Guthridge, J.M.; Pardo, G.; Dunn, J.; Steinman, L.; et al. IFN-beta treatment requires B cells for efficacy in neuroautoimmunity. J. Immunol. 2015, 194, 2110–2116. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Rezk, A.; Miyazaki, Y.; Hilgenberg, E.; Touil, H.; Shen, P.; Moore, C.S.; Michel, L.; Althekair, F.; Rajasekharan, S.; et al. Proinflammatory GM-CSF-producing B cells in multiple sclerosis and B cell depletion therapy. Sci. Transl. Med. 2015, 7, 310ra166. [Google Scholar] [CrossRef] [PubMed]
- Bourdette, D.; Yadav, V. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. Curr. Neurol. Neurosci. Rep. 2008, 8, 417–418. [Google Scholar] [CrossRef]
- Mathias, A.; Perriard, G.; Canales, M.; Soneson, C.; Delorenzi, M.; Schluep, M.; Du Pasquier, R.A. Increased ex vivo antigen presentation profile of B cells in multiple sclerosis. Mult. Scler. 2017, 23, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Parker Harp, C.R.; Archambault, A.S.; Sim, J.; Ferris, S.T.; Mikesell, R.J.; Koni, P.A.; Shimoda, M.; Linington, C.; Russell, J.H.; Wu, G.F. B cell antigen presentation is sufficient to drive neuroinflammation in an animal model of multiple sclerosis. J. Immunol. 2015, 194, 5077–5084. [Google Scholar] [CrossRef]
- Molnarfi, N.; Schulze-Topphoff, U.; Weber, M.S.; Patarroyo, J.C.; Prod’homme, T.; Varrin-Doyer, M.; Shetty, A.; Linington, C.; Slavin, A.J.; Hidalgo, J.; et al. MHC class II-dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies. J. Exp. Med. 2013, 210, 2921–2937. [Google Scholar] [CrossRef] [PubMed]
- Barr, T.A.; Shen, P.; Brown, S.; Lampropoulou, V.; Roch, T.; Lawrie, S.; Fan, B.; O’Connor, R.A.; Anderton, S.M.; Bar-Or, A.; et al. B cell depletion therapy ameliorates autoimmune disease through ablation of IL-6-producing B cells. J. Exp. Med. 2012, 209, 1001–1010. [Google Scholar] [CrossRef]
- Li, R.; Rezk, A.; Healy, L.M.; Muirhead, G.; Prat, A.; Gommerman, J.L.; Bar-Or, A.; Team, M.C.B.c.i.M. Cytokine-Defined B Cell Responses as Therapeutic Targets in Multiple Sclerosis. Front. Immunol. 2015, 6, 626. [Google Scholar] [CrossRef] [PubMed]
- Villar, L.M.; Masjuan, J.; Sadaba, M.C.; Gonzalez-Porque, P.; Plaza, J.; Bootello, A.; Alvarez-Cermeno, J.C. Early differential diagnosis of multiple sclerosis using a new oligoclonal band test. Arch. Neurol. 2005, 62, 574–577. [Google Scholar] [CrossRef] [PubMed]
- Bjornevik, K.; Cortese, M.; Healy, B.C.; Kuhle, J.; Mina, M.J.; Leng, Y.; Elledge, S.J.; Niebuhr, D.W.; Scher, A.I.; Munger, K.L.; et al. Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Science 2022, 375, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Jog, N.R.; McClain, M.T.; Heinlen, L.D.; Gross, T.; Towner, R.; Guthridge, J.M.; Axtell, R.C.; Pardo, G.; Harley, J.B.; James, J.A. Epstein Barr virus nuclear antigen 1 (EBNA-1) peptides recognized by adult multiple sclerosis patient sera induce neurologic symptoms in a murine model. J. Autoimmun. 2020, 106, 102332. [Google Scholar] [CrossRef]
- Tengvall, K.; Huang, J.; Hellstrom, C.; Kammer, P.; Bistrom, M.; Ayoglu, B.; Lima Bomfim, I.; Stridh, P.; Butt, J.; Brenner, N.; et al. Molecular mimicry between Anoctamin 2 and Epstein-Barr virus nuclear antigen 1 associates with multiple sclerosis risk. Proc. Natl. Acad. Sci. USA 2019, 116, 16955–16960. [Google Scholar] [CrossRef] [PubMed]
- Lanz, T.V.; Brewer, R.C.; Ho, P.P.; Moon, J.S.; Jude, K.M.; Fernandez, D.; Fernandes, R.A.; Gomez, A.M.; Nadj, G.S.; Bartley, C.M.; et al. Clonally expanded B cells in multiple sclerosis bind EBV EBNA1 and GlialCAM. Nature 2022, 603, 321–327. [Google Scholar] [CrossRef]
- Blair, P.A.; Norena, L.Y.; Flores-Borja, F.; Rawlings, D.J.; Isenberg, D.A.; Ehrenstein, M.R.; Mauri, C. CD19(+)CD24(hi)CD38(hi) B cells exhibit regulatory capacity in healthy individuals but are functionally impaired in systemic Lupus Erythematosus patients. Immunity 2010, 32, 129–140. [Google Scholar] [CrossRef]
- Bouaziz, J.D.; Yanaba, K.; Tedder, T.F. Regulatory B cells as inhibitors of immune responses and inflammation. Immunol. Rev. 2008, 224, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Flores-Borja, F.; Bosma, A.; Ng, D.; Reddy, V.; Ehrenstein, M.R.; Isenberg, D.A.; Mauri, C. CD19+CD24hiCD38hi B cells maintain regulatory T cells while limiting TH1 and TH17 differentiation. Sci. Transl. Med. 2013, 5, 173ra123. [Google Scholar] [CrossRef]
- Kalampokis, I.; Yoshizaki, A.; Tedder, T.F. IL-10-producing regulatory B cells (B10 cells) in autoimmune disease. Arthritis Res. Ther. 2013, 15 (Suppl. S1), S1. [Google Scholar] [CrossRef] [PubMed]
- Lundy, S.K.; Wu, Q.; Wang, Q.; Dowling, C.A.; Taitano, S.H.; Mao, G.; Mao-Draayer, Y. Dimethyl fumarate treatment of relapsing-remitting multiple sclerosis influences B-cell subsets. Neurol. Neuroimmunol. Neuroinflamm. 2016, 3, e211. [Google Scholar] [CrossRef]
- Dooley, J.; Pauwels, I.; Franckaert, D.; Smets, I.; Garcia-Perez, J.E.; Hilven, K.; Danso-Abeam, D.; Joanne Terbeek, J.; Nguyen, A.T.L.; De Muynck, L.; et al. Immunologic profiles of multiple sclerosis treatments reveal shared early B cell alterations. Neurol. (R) Neuroimmunol. Neuroinflamm. 2016, 3, e240. [Google Scholar] [CrossRef]
- Maseda, D.; Candando, K.M.; Smith, S.H.; Kalampokis, I.; Weaver, C.T.; Plevy, S.E.; Poe, J.C.; Tedder, T.F. Peritoneal cavity regulatory B cells (B10 cells) modulate IFN-gamma+CD4+ T cell numbers during colitis development in mice. J. Immunol. 2013, 191, 2780–2795. [Google Scholar] [CrossRef]
- Matsushita, T.; Yanaba, K.; Bouaziz, J.D.; Fujimoto, M.; Tedder, T.F. Regulatory B cells inhibit EAE initiation in mice while other B cells promote disease progression. J. Clin. Investig. 2008, 118, 3420–3430. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Dieter, J.A.; Rothaeusler, K.; Luo, Z.; Baumgarth, N. B-1 cells in the bone marrow are a significant source of natural IgM. Eur. J. Immunol. 2012, 42, 120–129. [Google Scholar] [CrossRef]
- Griffin, D.O.; Rothstein, T.L. Human “orchestrator” CD11b(+) B1 cells spontaneously secrete interleukin-10 and regulate T-cell activity. Mol. Med. 2012, 18, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Bod, L.; Pochet, N.; Kota, S.B.; Hu, D.; Madi, A.; Kilpatrick, J.; Shi, J.; Ho, A.; Zhang, H.; et al. Checkpoint Receptor TIGIT Expressed on Tim-1(+) B Cells Regulates Tissue Inflammation. Cell Rep. 2020, 32, 107892. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.M.; Stott, R.T.; Zhao, G.; SooHoo, J.; Xiong, W.; Lian, M.M.; Fitzgerald, L.; Shi, S.; Akrawi, E.; Lei, J.; et al. TGF-beta-producing regulatory B cells induce regulatory T cells and promote transplantation tolerance. Eur. J. Immunol. 2014, 44, 1728–1736. [Google Scholar] [CrossRef]
- Shen, P.; Roch, T.; Lampropoulou, V.; O’Connor, R.A.; Stervbo, U.; Hilgenberg, E.; Ries, S.; Dang, V.D.; Jaimes, Y.; Daridon, C.; et al. IL-35-producing B cells are critical regulators of immunity during autoimmune and infectious diseases. Nature 2014, 507, 366–370. [Google Scholar] [CrossRef]
- Sorensen, P.S.; Lisby, S.; Grove, R.; Derosier, F.; Shackelford, S.; Havrdova, E.; Drulovic, J.; Filippi, M. Safety and efficacy of ofatumumab in relapsing-remitting multiple sclerosis: A phase 2 study. Neurology 2014, 82, 573–581. [Google Scholar] [CrossRef]
- Herbst, R.; Wang, Y.; Gallagher, S.; Mittereder, N.; Kuta, E.; Damschroder, M.; Woods, R.; Rowe, D.C.; Cheng, L.; Cook, K.; et al. B-cell depletion in vitro and in vivo with an afucosylated anti-CD19 antibody. J. Pharmacol. Exp. Ther. 2010, 335, 213–222. [Google Scholar] [CrossRef]
- Mackay, F.; Schneider, P.; Rennert, P.; Browning, J. BAFF AND APRIL: A tutorial on B cell survival. Annu. Rev. Immunol. 2003, 21, 231–264. [Google Scholar] [CrossRef] [PubMed]
- Landolt-Marticorena, C.; Wither, R.; Reich, H.; Herzenberg, A.; Scholey, J.; Gladman, D.D.; Urowitz, M.B.; Fortin, P.R.; Wither, J. Increased expression of B cell activation factor supports the abnormal expansion of transitional B cells in systemic lupus erythematosus. J. Rheumatol. 2011, 38, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Krumbholz, M.; Derfuss, T.; Hohlfeld, R.; Meinl, E. B cells and antibodies in multiple sclerosis pathogenesis and therapy. Nat. Rev. Neurol. 2012, 8, 613–623. [Google Scholar] [CrossRef] [PubMed]
- Hedegaard, C.J.; Sellebjerg, F.; Krakauer, M.; Hesse, D.; Bendtzen, K.; Nielsen, C.H. Interferon-beta increases systemic BAFF levels in multiple sclerosis without increasing autoantibody production. Mult. Scler. 2010, 17, 567–577. [Google Scholar] [CrossRef]
- Krumbholz, M.; Faber, H.; Steinmeyer, F.; Hoffmann, L.A.; Kumpfel, T.; Pellkofer, H.; Derfuss, T.; Ionescu, C.; Starck, M.; Hafner, C.; et al. Interferon-beta increases BAFF levels in multiple sclerosis: Implications for B cell autoimmunity. Brain 2008, 131, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Mimpen, M.; Damoiseaux, J.; van Doorn, W.; Rolf, L.; Muris, A.H.; Hupperts, R.; van Luijn, M.M.; Gerlach, O.; Smolders, J. Proportions of circulating transitional B cells associate with MRI activity in interferon beta-treated multiple sclerosis patients. J. Neuroimmunol. 2021, 358, 577664. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, F.; Giacomini, E.; Mechelli, R.; Buscarinu, M.C.; Salvetti, M.; Severa, M.; Coccia, E.M. Interferon-beta therapy specifically reduces pathogenic memory B cells in multiple sclerosis patients by inducing a FAS-mediated apoptosis. Immunol. Cell Biol. 2016, 94, 886–894. [Google Scholar] [CrossRef]
- Gold, R.; Kappos, L.; Arnold, D.L.; Bar-Or, A.; Giovannoni, G.; Selmaj, K.; Tornatore, C.; Sweetser, M.T.; Yang, M.; Sheikh, S.I.; et al. Placebo-controlled phase 3 study of oral BG-12 for relapsing multiple sclerosis. N. Engl. J. Med. 2012, 367, 1098–1107. [Google Scholar] [CrossRef]
- Kappos, L.; Gold, R.; Miller, D.H.; Macmanus, D.G.; Havrdova, E.; Limmroth, V.; Polman, C.H.; Schmierer, K.; Yousry, T.A.; Yang, M.; et al. Efficacy and safety of oral fumarate in patients with relapsing-remitting multiple sclerosis: A multicentre, randomised, double-blind, placebo-controlled phase IIb study. Lancet 2008, 372, 1463–1472. [Google Scholar] [CrossRef]
- Li, R.; Rezk, A.; Ghadiri, M.; Luessi, F.; Zipp, F.; Li, H.; Giacomini, P.S.; Antel, J.; Bar-Or, A. Dimethyl Fumarate Treatment Mediates an Anti-Inflammatory Shift in B Cell Subsets of Patients with Multiple Sclerosis. J. Immunol. 2017, 198, 691–698. [Google Scholar] [CrossRef]
- Matloubian, M.; Lo, C.G.; Cinamon, G.; Lesneski, M.J.; Xu, Y.; Brinkmann, V.; Allende, M.L.; Proia, R.L.; Cyster, J.G. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 2004, 427, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Josefowicz, S.Z.; Lu, L.F.; Rudensky, A.Y. Regulatory T cells: Mechanisms of differentiation and function. Annu. Rev. Immunol. 2012, 30, 531–564. [Google Scholar] [CrossRef]
- Claes, N.; Dhaeze, T.; Fraussen, J.; Broux, B.; Van Wijmeersch, B.; Stinissen, P.; Hupperts, R.; Hellings, N.; Somers, V. Compositional changes of B and T cell subtypes during fingolimod treatment in multiple sclerosis patients: A 12-month follow-up study. PLoS ONE 2014, 9, e111115. [Google Scholar] [CrossRef]
- Grutzke, B.; Hucke, S.; Gross, C.C.; Herold, M.V.; Posevitz-Fejfar, A.; Wildemann, B.T.; Kieseier, B.C.; Dehmel, T.; Wiendl, H.; Klotz, L. Fingolimod treatment promotes regulatory phenotype and function of B cells. Ann. Clin. Transl. Neurol. 2015, 2, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Hartung, H.P.; Kieseier, B.C. Atacicept: Targeting B cells in multiple sclerosis. Ther. Adv. Neurol. Disord. 2010, 3, 205–216. [Google Scholar] [CrossRef]
- Schneider, P.; MacKay, F.; Steiner, V.; Hofmann, K.; Bodmer, J.L.; Holler, N.; Ambrose, C.; Lawton, P.; Bixler, S.; Acha-Orbea, H.; et al. BAFF, a novel ligand of the tumor necrosis factor family, stimulates B cell growth. J. Exp. Med. 1999, 189, 1747–1756. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.V.; Lopez-Fraga, M.; Elustondo, F.A.; Carvalho-Pinto, C.E.; Rodriguez, D.; Gomez-Caro, R.; De Jong, J.; Martinez, A.C.; Medema, J.P.; Hahne, M. APRIL modulates B and T cell immunity. J. Clin. Investig. 2002, 109, 1587–1598. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.A.; Johnston, J.; Mudri, S.; Enselman, R.; Dillon, S.R.; Madden, K.; Xu, W.; Parrish-Novak, J.; Foster, D.; Lofton-Day, C.; et al. TACI and BCMA are receptors for a TNF homologue implicated in B-cell autoimmune disease. Nature 2000, 404, 995–999. [Google Scholar] [CrossRef]
- Thompson, J.S.; Bixler, S.A.; Qian, F.; Vora, K.; Scott, M.L.; Cachero, T.G.; Hession, C.; Schneider, P.; Sizing, I.D.; Mullen, C.; et al. BAFF-R, a newly identified TNF receptor that specifically interacts with BAFF. Science 2001, 293, 2108–2111. [Google Scholar] [CrossRef]
- Marsters, S.A.; Yan, M.; Pitti, R.M.; Haas, P.E.; Dixit, V.M.; Ashkenazi, A. Interaction of the TNF homologues BLyS and APRIL with the TNF receptor homologues BCMA and TACI. Curr. Biol. 2000, 10, 785–788. [Google Scholar] [CrossRef] [PubMed]
- Rennert, P.; Schneider, P.; Cachero, T.G.; Thompson, J.; Trabach, L.; Hertig, S.; Holler, N.; Qian, F.; Mullen, C.; Strauch, K.; et al. A soluble form of B cell maturation antigen, a receptor for the tumor necrosis factor family member APRIL, inhibits tumor cell growth. J. Exp. Med. 2000, 192, 1677–1684. [Google Scholar] [CrossRef] [PubMed]
- Shu, H.B.; Johnson, H. B cell maturation protein is a receptor for the tumor necrosis factor family member TALL-1. Proc. Natl. Acad. Sci. USA 2000, 97, 9156–9161. [Google Scholar] [CrossRef]
- Thompson, J.S.; Schneider, P.; Kalled, S.L.; Wang, L.; Lefevre, E.A.; Cachero, T.G.; MacKay, F.; Bixler, S.A.; Zafari, M.; Liu, Z.Y.; et al. BAFF binds to the tumor necrosis factor receptor-like molecule B cell maturation antigen and is important for maintaining the peripheral B cell population. J. Exp. Med. 2000, 192, 129–135. [Google Scholar] [CrossRef]
- Mackay, F.; Schneider, P. TACI, an enigmatic BAFF/APRIL receptor, with new unappreciated biochemical and biological properties. Cytokine Growth Factor Rev. 2008, 19, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Rickert, R.C.; Jellusova, J.; Miletic, A.V. Signaling by the tumor necrosis factor receptor superfamily in B-cell biology and disease. Immunol. Rev. 2011, 244, 115–133. [Google Scholar] [CrossRef]
- Ou, X.; Xu, S.; Lam, K.P. Deficiency in TNFRSF13B (TACI) expands T-follicular helper and germinal center B cells via increased ICOS-ligand expression but impairs plasma cell survival. Proc. Natl. Acad. Sci. USA 2012, 109, 15401–15406. [Google Scholar] [CrossRef]
- O’Connor, B.P.; Raman, V.S.; Erickson, L.D.; Cook, W.J.; Weaver, L.K.; Ahonen, C.; Lin, L.L.; Mantchev, G.T.; Bram, R.J.; Noelle, R.J. BCMA is essential for the survival of long-lived bone marrow plasma cells. J. Exp. Med. 2004, 199, 91–98. [Google Scholar] [CrossRef]
- Xu, S.; Lam, K.P. B-cell maturation protein, which binds the tumor necrosis factor family members BAFF and APRIL, is dispensable for humoral immune responses. Mol. Cell Biol. 2001, 21, 4067–4074. [Google Scholar] [CrossRef]
- Yan, M.; Brady, J.R.; Chan, B.; Lee, W.P.; Hsu, B.; Harless, S.; Cancro, M.; Grewal, I.S.; Dixit, V.M. Identification of a novel receptor for B lymphocyte stimulator that is mutated in a mouse strain with severe B cell deficiency. Curr. Biol. 2001, 11, 1547–1552. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Richman, D.P.; Zamvil, S.S.; Agius, M.A. Accelerated central nervous system autoimmunity in BAFF-receptor-deficient mice. J. Neurol. Sci. 2011, 306, 9–15. [Google Scholar] [CrossRef]
- Carrillo-Ballesteros, F.J.; Oregon-Romero, E.; Franco-Topete, R.A.; Govea-Camacho, L.H.; Cruz, A.; Munoz-Valle, J.F.; Bustos-Rodriguez, F.J.; Pereira-Suarez, A.L.; Palafox-Sanchez, C.A. B-cell activating factor receptor expression is associated with germinal center B-cell maintenance. Exp. Ther. Med. 2019, 17, 2053–2060. [Google Scholar] [CrossRef] [PubMed]
- Krumbholz, M.; Theil, D.; Derfuss, T.; Rosenwald, A.; Schrader, F.; Monoranu, C.M.; Kalled, S.L.; Hess, D.M.; Serafini, B.; Aloisi, F.; et al. BAFF is produced by astrocytes and up-regulated in multiple sclerosis lesions and primary central nervous system lymphoma. J. Exp. Med. 2005, 201, 195–200. [Google Scholar] [CrossRef]
- Stohl, W.; Banfalvi, A. B cell-independent contribution of BAFF to murine autoimmune disease. Clin. Immunol. 2016, 172, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Hase, H.; Legarda-Addison, D.; Varughese, L.; Seed, B.; Ting, A.T. B cell maturation antigen, the receptor for a proliferation-inducing ligand and B cell-activating factor of the TNF family, induces antigen presentation in B cells. J. Immunol. 2005, 175, 2814–2824. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Maria, Z.; Kohli, U.; Agasing, A.; Quinn, J.L.; Ko, R.M.; Zamvil, S.S.; Axtell, R.C. CNS Autoimmune Responses in BCMA-Deficient Mice Provide Insight for the Failure of Atacicept in MS. Neurol. Neuroimmunol. Neuroinflamm. 2021, 8. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.A.; Hoffmann, F.S.; Kuhn, P.H.; Cheng, Q.; Chu, Y.; Schmidt-Supprian, M.; Hauck, S.M.; Schuh, E.; Krumbholz, M.; Rubsamen, H.; et al. gamma-Secretase directly sheds the survival receptor BCMA from plasma cells. Nat. Commun. 2015, 6, 7333. [Google Scholar] [CrossRef] [PubMed]

| Inflammatory B Cells | Source | Functions |
|---|---|---|
| Memory B cells | MS patients | Antigen presentation |
| Pro-inflammatory cytokine production | ||
| EAE mice | Exacerbates EAE | |
| Produces IL-6 | ||
| IgG + plasma cells | CNS Lesions in MS patients | Oligoclonal bands |
| Auto-antibody production | ||
| EAE mice | Facilitate CNS damage | |
| Increases disease severity | ||
| Regulatory B Cells | ||
| Naïve B cells | MS patients | High levels of IL-10 |
| Bregs/Transitional B cells | MS patients | Produce IL-10, IL-35, TGF-β |
| Suppresses TNF production by monocytes. | ||
| EAE mice | Produce IL-10 | |
| Inhibits TH1 and TH17 cells | ||
| IgA + plasma cells | EAE mice | Attenuates EAE |
| Produces IL-10 |
| MS Therapies | Mechanism of Action |
|---|---|
| Anti-CD20 | Removes all B cell subsets except pro-B cells and plasma cells |
| Depletes IL-6 producing B cells | |
| Interferon | Elevates serum BAFF levels |
| Increases IL-10 producing transitional B cells | |
| Decreases memory B cells | |
| Dimethyl fumarate | Reduces memory B cells |
| Spares transitional B cells | |
| Decreases pro-inflammatory cytokines GM-CSF, IL-6 and TNF-α | |
| Fingolimod | Blocks egression of inflammatory cells from secondary lymphoid tissues. |
| Reduced circulating lymphocytes | |
| Reduced CNS infiltration of inflammatory cells | |
| Reduces memory B cells | |
| Increases number and CNS infiltration of regulatory B cells |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, G.; Axtell, R.C. Dual Role of B Cells in Multiple Sclerosis. Int. J. Mol. Sci. 2023, 24, 2336. https://doi.org/10.3390/ijms24032336
Kumar G, Axtell RC. Dual Role of B Cells in Multiple Sclerosis. International Journal of Molecular Sciences. 2023; 24(3):2336. https://doi.org/10.3390/ijms24032336
Chicago/Turabian StyleKumar, Gaurav, and Robert C. Axtell. 2023. "Dual Role of B Cells in Multiple Sclerosis" International Journal of Molecular Sciences 24, no. 3: 2336. https://doi.org/10.3390/ijms24032336
APA StyleKumar, G., & Axtell, R. C. (2023). Dual Role of B Cells in Multiple Sclerosis. International Journal of Molecular Sciences, 24(3), 2336. https://doi.org/10.3390/ijms24032336







