Nanomaterials and Their Impact on the Immune System
Abstract
1. Introduction
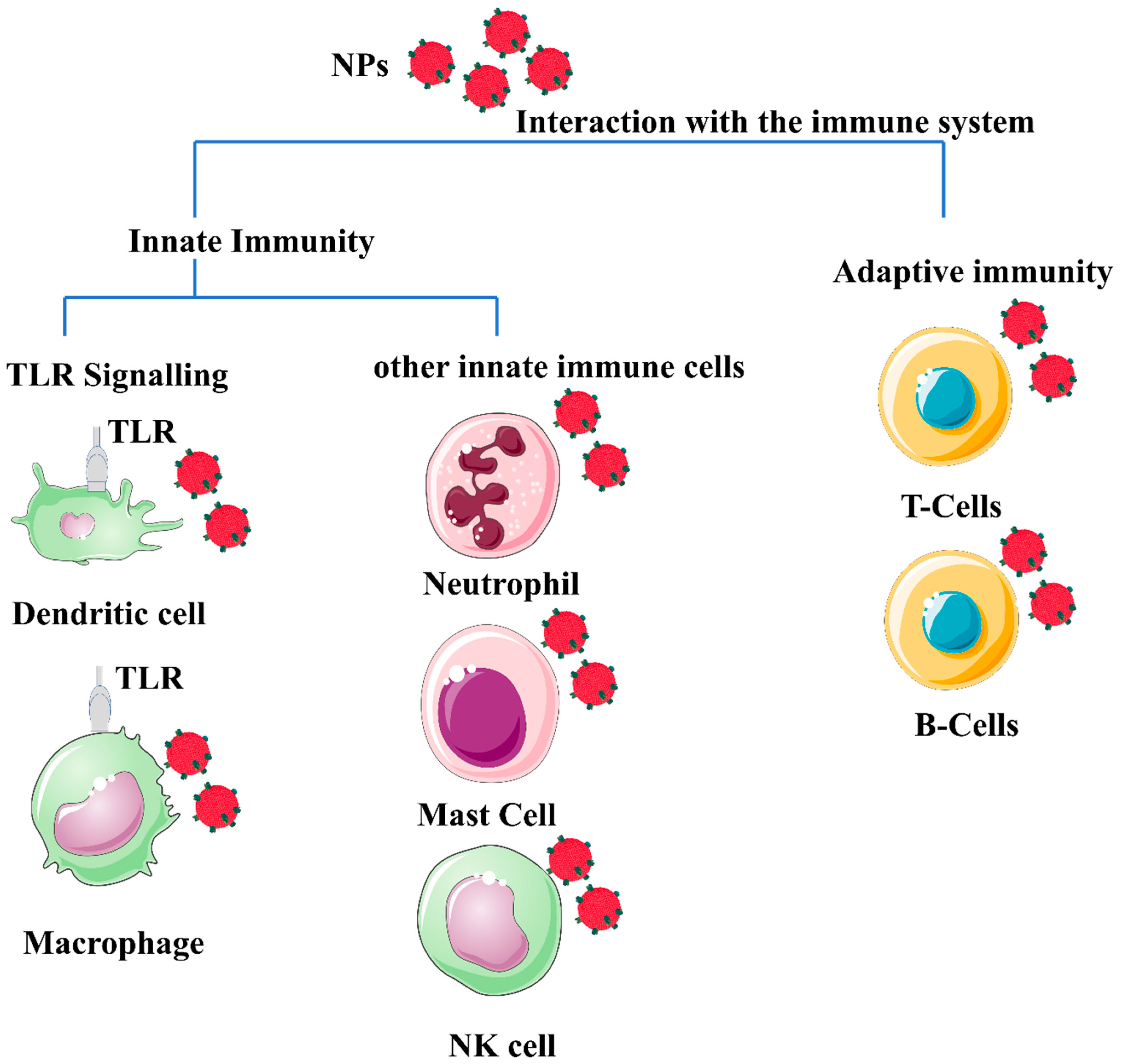
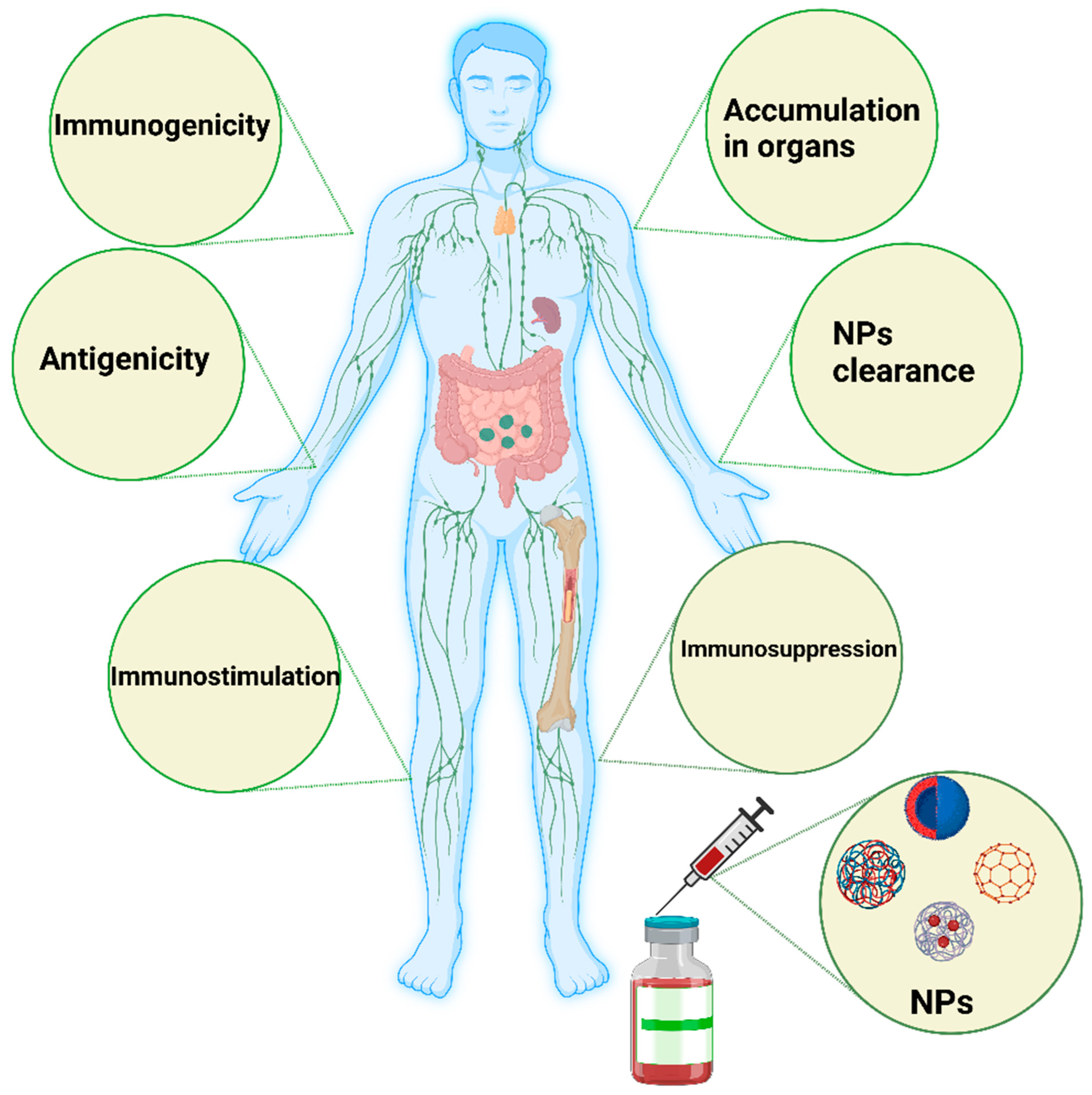
2. NPs and the Immune System
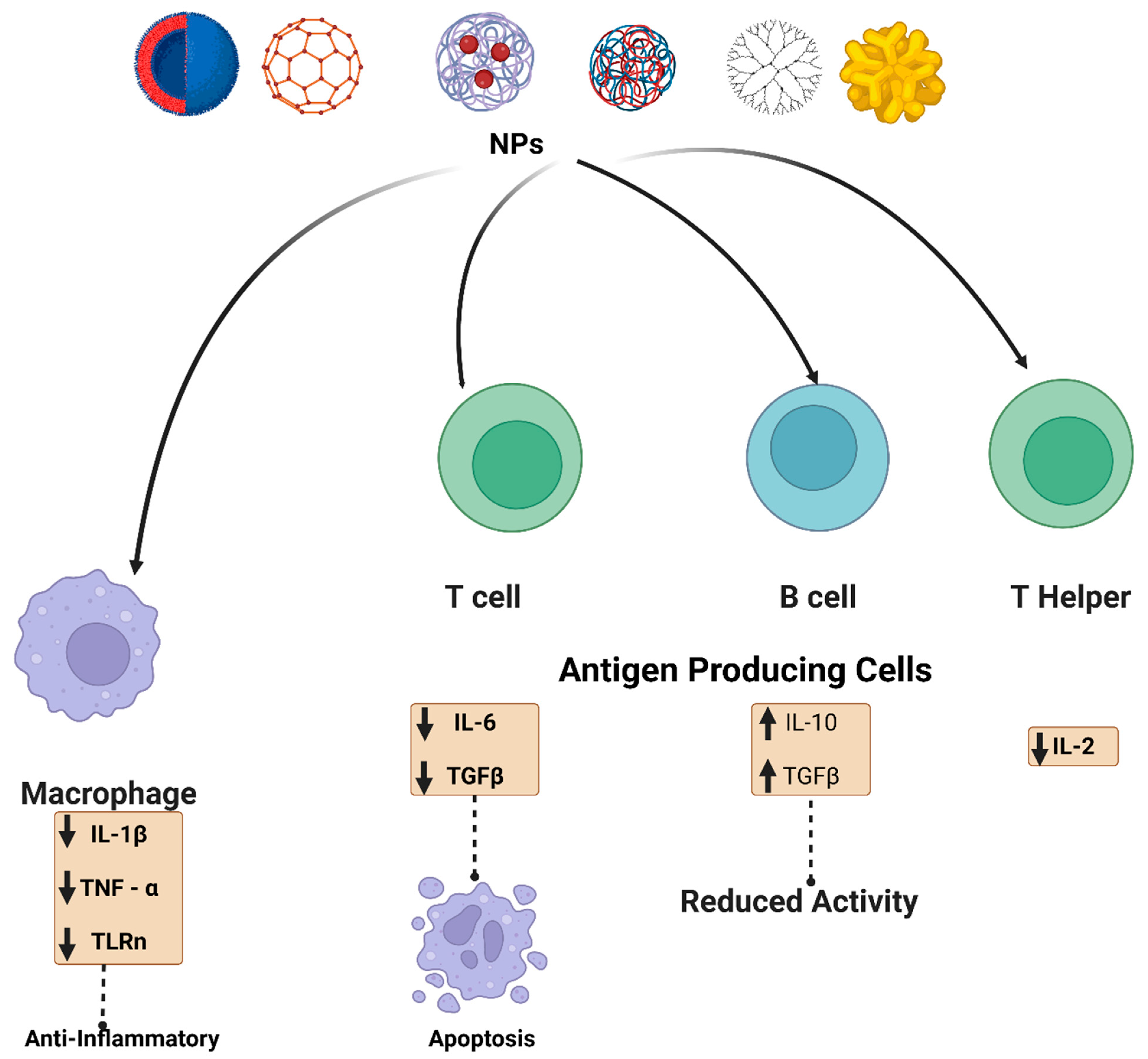
3. Nonspecific Immunomodulation
3.1. Nanomaterials and Immune System Modulations
3.2. Immunological Effects of Nanomaterials (Immunotoxicology)
3.3. Mechanism of Nanomaterials Toxicity
3.4. Nanomaterials Affect Cell Functions
3.5. Immunosuppression
3.6. Immunostimulant
| Type of NPs | Study Outcomes | References |
|---|---|---|
| Liposomes decorated with synthetic long peptides antigen | The immunological response was mediated by antigen-specific CD8+ T cells that were induced | [74] |
| Liposomes | Induction of antigen-specific response | [75] |
| Liposomes loaded with cytosine-phosphate-guanine and 3,5- didodecyloxybenzamidine | DCs were stimulated to release cytokines, co-stimulatory molecules were expressed, and an antigen-specific immune response was enhanced | [76] |
| AuNPs | Macrophage activation | [77] |
| AuNPs loaded with BSA antigen | Anti-BSA antibodies were detected in greater concentrations in the blood serum of mice inoculated with BSA–AuNPs and cytosine-phosphate-guanine –AuNPs conjugates | [78] |
| MWCNTs | Inducing strong CD4+ T and CD8+ T- cells mediated immune response | [79] |
| MWCNTs loaded with anti-CD40 Ig | In subcutaneous or lung pseudo-metastatic tumor models, it increased entrapped ovalbumin-specific T cell responses and suppressed the development of entrapped ovalbumin–expressing B16F10 melanoma cells. | [80] |
| MWCNTs loaded with ovalbumin | Elicited a strong anti-tumor immune response | [81] |
| MWCNTs loaded with cytosine-phosphate-guanine | Elicited a strong cellular and humoral immune response | [82] |
| Iron oxide (Fe3O4) NPs | In vitro, it induced a significant adaptive immune response by stimulating DCs and macrophages, and it reduced tumor development and prevented tumor formation in vivo | [83] |
| Iron oxide (Fe3O4) NPs | Enhanced T cell activation and elevated stimulation of anti-tumor activity | [84] |
| Micelles loaded with cytosine-phosphate-guanine and Trp2 | In tumor-bearing mice, it produced antigen-specific cytotoxic CD8+ T cell-mediated immunity, as well as a robust anti-cancer immune response | [85] |
| Micelles loaded with cytosine-phosphate-guanine and Trp2 | Trp2/PHM10/ cytosine-phosphate-guanine nanoformulation dramatically increased CD8+ T cellular activities while improving anti-tumor effectiveness. | [86] |
| Micelles loaded with ovalbumin and CL264 agonist | A robust antigen-specific cellular and humoral immune response was elicited | [87] |
| Dendrimers loaded with ovalbumin and cytosine-phosphate-guanine | Elicited a much greater T-cell-mediated immunological response | [88,89] |
| Dendrimers loaded with cytosine-phosphate-guanine | Cytosine-phosphate-guanine delivered effectively into DCs triggered an adaptive cellular immune response | [90] |
| Protein NP loaded with melanoma-associated gp100 epitope and cytosine-phosphate-guanine | Antigen-specific antitumor immune response substantially increased | [91] |
| NP protein loaded with peptide epitope cytosine-phosphate-guanine | Elevated CD8+ T cell activation and antigen cross-presentation | [92] |
3.7. Are Nanomaterials Immunogenic?
3.8. NPs and the Effect of Toll-like Receptors
4. NPs and Allergy
5. Interaction with the Immune System
6. Nanomaterials and Inflammation
Immune Evasion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sompayrac, L.M. How the Immune System Works; John Wiley & Sons: New York, NY, USA, 2019. [Google Scholar]
- Schrand, A.M.; Rahman, M.F.; Hussain, S.M.; Schlager, J.J.; Smith, D.A.; Syed, A.F. Metal-based nanoparticles and their toxicity assessment. WIREs Nanomed. Nanobiotechnol. 2010, 2, 544–568. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Wang, J.; Shi, J.; Li, H.; Lu, M.; Fan, Z.; Gu, Z.; Cheng, H. Tailoring the physicochemical properties of nanomaterials for immunomodulation. Adv. Drug Deliv. Rev. 2021, 180, 114039. [Google Scholar] [CrossRef] [PubMed]
- Aljabali, A.A.A.; Obeid, M.A. Inorganic-organic nanomaterials for therapeutics and molecular imaging applications. Nanosci. Nanotechnol. Asia 2020, 10, 748–765. [Google Scholar] [CrossRef]
- Anderson, R.M.; May, R.M. Directly Transmitted Infections Diseases: Control by Vaccination. Science 1982, 215, 1053–1060. [Google Scholar] [CrossRef]
- Mogensen, T.H. Pathogen Recognition and Inflammatory Signaling in Innate Immune Defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef]
- Guermonprez, P.; Valladeau, J.; Zitvogel, L.; Théry, C.; Amigorena, S. Antigen presentation and T cell stimulation by dendritic cells. Annu. Rev. Immunol. 2002, 20, 621–667. [Google Scholar] [CrossRef]
- Roche, P.A.; Furuta, K. The ins and outs of MHC class II-mediated antigen processing and presentation. Nat. Rev. Immunol. 2015, 15, 203–216. [Google Scholar] [CrossRef]
- Joffre, O.; Segura, E.; Savina, A.; Amigorena, S. Cross-presentation by dendritic cells. Nat. Rev. Immunol. 2012, 12, 557–569. [Google Scholar] [CrossRef]
- Farrera, C.; Fadeel, B. It takes two to tango: Understanding the interactions between engineered nanomaterials and the immune system. Eur. J. Pharm. Biopharm. 2015, 95, 3–12. [Google Scholar] [CrossRef]
- Hicks, A.M.; Willingham, M.C.; Du, W.; Pang, C.S.; Old, L.J.; Cui, Z. Effector mechanisms of the anti-cancer immune responses of macrophages in SR/CR mice. Cancer Immun. 2006, 6, 11. [Google Scholar]
- Smith, J.M.; Nemeth, T.L.; McDonald, R.A. Current immunosuppressive agents in pediatric renal transplantation: Efficacy, side-effects and utilization. Pediatr. Transplant. 2004, 8, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Boraschi, D.; Costantino, L.; Italiani, P. Interaction of nanoparticles with immunocompetent cells: Nanosafety considerations. Nanomedicine 2012, 7, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Johnston, H.J.; Hutchison, G.; Christensen, F.M.; Peters, S.; Hankin, S.; Stone, V. A review of the in vivo and in vitro toxicity of silver and gold particulates: Particle attributes and biological mechanisms responsible for the observed toxicity. Crit. Rev. Toxicol. 2010, 40, 328–346. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.P.; Priyadarshini, A.; Gupta, A.; Vibhuti, A.; Leal, E.; Sengupta, U.; Katoch, V.M.; Sharma, P.; Moore, C.E.; Raj, V.S. Exploring the modulation of immune response and oxidative stress of intracellular pathogens using nanoparticles encapsulating drugs. Preprints 2020, 10, 580. [Google Scholar]
- Zhao, C.; Deng, H.; Chen, X. Harnessing immune response using reactive oxygen Species-Generating/Eliminating inorganic biomaterials for disease treatment. Adv. Drug Deliv. Rev. 2022, 188, 114456. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.A.; Hannan, F.; Islam, F.; Ayyaz, A.; Zhang, N.; Chen, W.; Zhang, K.; Huang, Q.; Xu, L.; Zhou, W. The potential of nanomaterials for sustainable modern agriculture: Present findings and future perspectives. Environ. Sci. Nano 2022, 9, 1926–1951. [Google Scholar] [CrossRef]
- Saliani, M.; Jalal, R.; Goharshadi, E.K. Mechanism of oxidative stress involved in the toxicity of ZnO nanoparticles against eukaryotic cells. Nanomed. J. 2016, 3, 1–14. [Google Scholar]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. Biomed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef]
- Risom, L.; Møller, P.; Loft, S. Oxidative stress-induced DNA damage by particulate air pollution. Mutat. Res. Mol. Mech. Mutagen. 2005, 592, 119–137. [Google Scholar] [CrossRef]
- Buzea, C.; Pacheco, I.I.; Robbie, K. Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases 2007, 2, MR17–MR71. [Google Scholar] [CrossRef]
- Fubini, B.; Hubbard, A. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) generation by silica in inflammation and fibrosis. Free. Radic. Biol. Med. 2003, 34, 1507–1516. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Young, S.-H.; Schwegler-Berry, D.; Chisholm, W.P.; Fernback, J.E.; Ma, Q. Multiwalled Carbon Nanotubes Induce a Fibrogenic Response by Stimulating Reactive Oxygen Species Production, Activating NF-κB Signaling, and Promoting Fibroblast-to-Myofibroblast Transformation. Chem. Res. Toxicol. 2011, 24, 2237–2248. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, S.; Costa-Almeida, R.; Santos, S.G.; Magalhães, F.D.; Pinto, A.M. Advances in carbon nanomaterials for immunotherapy. Appl. Mater. Today 2022, 27, 101397. [Google Scholar] [CrossRef]
- Holmannova, D.; Borsky, P.; Svadlakova, T.; Borska, L.; Fiala, Z. Carbon Nanoparticles and Their Biomedical Applications. Appl. Sci. 2022, 12, 7865. [Google Scholar] [CrossRef]
- Yuan, X.; Zhang, X.; Sun, L.; Wei, Y.; Wei, X. Cellular Toxicity and Immunological Effects of Carbon-based Nanomaterials. Part. Fibre Toxicol. 2019, 16, 18. [Google Scholar] [CrossRef]
- Zolnik, B.S.; González-Fernández, A.; Sadrieh, N.; Dobrovolskaia, M.A. Nanoparticles and the Immune System. Endocrinology 2010, 151, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Baldim, I.; Oliveira, W.P.; Rao, R.; Raghuvir, S.; Mahant, S.; Gama, F.M.; Souto, E.B. Biofate and cellular interactions of lipid nanoparticles. In Nanoparticle Therapeutics; Academic Press: Cambridge, MA, USA, 2021; pp. 211–246. [Google Scholar] [CrossRef]
- Lai, C.; Chadban, S.J.; Loh, Y.W.; Kwan, T.K.-T.; Wang, C.; Singer, J.; Niewold, P.; Ling, Z.; Spiteri, A.; Getts, D.; et al. Targeting inflammatory monocytes by immune-modifying nanoparticles prevents acute kidney allograft rejection. Kidney Int. 2022, 102, 1090–1102. [Google Scholar] [CrossRef]
- Nguyen, T.L.; Kim, J. Nanoparticle-Based Tolerogenic Vaccines for the Treatment of Autoimmune Diseases: A Review. ACS Appl. Nano Mater. 2022, 5, 6013–6028. [Google Scholar] [CrossRef]
- Liu, J.; Liu, Z.; Pang, Y.; Zhou, H. The interaction between nanoparticles and immune system: Application in the treatment of inflammatory diseases. J. Nanobiotechnol. 2022, 20, 127. [Google Scholar] [CrossRef]
- Jain, P.; Darji, P.; Thakur, B.S.; Jain, A.; Jain, P.K.; Khare, B. Immunostimulants: Concepts, Types and Functions. Asian J. Dent. Health Sci. 2022, 2, 26–34. [Google Scholar] [CrossRef]
- Dobrovolskaia, M.A.; Aggarwal, P.; Hall, J.B.; McNeil, S.E. Preclinical Studies To Understand Nanoparticle Interaction with the Immune System and Its Potential Effects on Nanoparticle Biodistribution. Mol. Pharm. 2008, 5, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X. Gold Nanoparticles: Recent Advances in the Biomedical Applications. Cell Biochem. Biophys. 2015, 72, 771–775. [Google Scholar] [CrossRef] [PubMed]
- Sur, A.; Pradhan, B.; Banerjee, A.; Aich, P. Immune Activation Efficacy of Indolicidin Is Enhanced upon Conjugation with Carbon Nanotubes and Gold Nanoparticles. PLoS ONE 2015, 10, e0123905. [Google Scholar] [CrossRef]
- Wang, S.; Chen, R.; Yu, Q.; Huang, W.; Lai, P.; Tang, J.; Nie, L. Near-Infrared Plasmon-Boosted Heat/Oxygen Enrichment for Reversing Rheumatoid Arthritis with Metal/Semiconductor Composites. ACS Appl. Mater. Interfaces 2020, 12, 45796–45806. [Google Scholar] [CrossRef] [PubMed]
- Thakor, A.S.; Jokerst, J.; Zavaleta, C.; Massoud, T.F.; Gambhir, S.S. Gold Nanoparticles: A Revival in Precious Metal Administration to Patients. Nano Lett. 2011, 11, 4029–4036. [Google Scholar] [CrossRef]
- Chen, H.; Dorrigan, A.; Saad, S.; Hare, D.J.; Cortie, M.B.; Valenzuela, S.M. In Vivo Study of Spherical Gold Nanoparticles: Inflammatory Effects and Distribution in Mice. PLoS ONE 2013, 8, e58208. [Google Scholar] [CrossRef]
- Goodman, C.M.; McCusker, C.D.; Yilmaz, T.; Rotello, V.M. Toxicity of Gold Nanoparticles Functionalized with Cationic and Anionic Side Chains. Bioconjugate Chem. 2004, 15, 897–900. [Google Scholar] [CrossRef]
- Greulich, C.; Diendorf, J.; Geßmann, J.; Simon, T.; Habijan, T.; Eggeler, G.; Schildhauer, T.; Epple, M.; Köller, M. Cell type-specific responses of peripheral blood mononuclear cells to silver nanoparticles. Acta Biomater. 2011, 7, 3505–3514. [Google Scholar] [CrossRef]
- Wu, T.; Tang, M. The inflammatory response to silver and titanium dioxide nanoparticles in the central nervous system. Nanomedicine 2018, 13, 233–249. [Google Scholar] [CrossRef]
- Jan, T.-R.; Shen, C.-C.; Liang, H.-J.; Wang, C.-C.; Liao, M.-H. Iron oxide nanoparticles suppressed T helper 1 cell-mediated immunity in a murine model of delayed-type hypersensitivity. Int. J. Nanomed. 2012, 7, 2729–2737. [Google Scholar] [CrossRef]
- Liao, M.-H.; Liu, S.-S.; Peng, I.-C.; Tsai, F.-J.; Huang, H.H. The stimulatory effects of alpha1-adrenergic receptors on TGF-beta1, IGF-1 and hyaluronan production in human skin fibroblasts. Cell Tissue Res. 2014, 357, 681–693. [Google Scholar] [CrossRef] [PubMed]
- Jin, K.; Luo, Z.; Zhang, B.; Pang, Z. Biomimetic nanoparticles for inflammation targeting. Acta Pharm. Sin. B 2017, 8, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-Y.; Chung, M.-C.; Wang, C.-C.; Huang, C.-H.; Liang, H.-J.; Jan, T.-R. Iron oxide nanoparticles suppress the production of IL-1beta via the secretory lysosomal pathway in murine microglial cells. Part. Fibre Toxicol. 2013, 10, 46. [Google Scholar] [CrossRef]
- Hirst, S.M.; Karakoti, A.S.; Tyler, R.D.; Sriranganathan, N.; Seal, S.; Reilly, C.M. Anti-inflammatory Properties of Cerium Oxide Nanoparticles. Small 2009, 5, 2848–2856. [Google Scholar] [CrossRef] [PubMed]
- Karakoti, A.; Singh, S.; Dowding, J.M.; Seal, S.; Self, W.T. Redox-active radical scavenging nanomaterials. Chem. Soc. Rev. 2010, 39, 4422–4432. [Google Scholar] [CrossRef]
- Liu, H.; Qu, X.; Tan, H.; Song, J.; Lei, M.; Kim, E.; Payne, G.F.; Liu, C. Role of polydopamine’s redox-activity on its pro-oxidant, radical-scavenging, and antimicrobial activities. Acta Biomater. 2019, 88, 181–196. [Google Scholar] [CrossRef]
- Zogovic, N.S.; Nikolic, N.S.; Vranjes-Djuric, S.D.; Harhaji, L.M.; Vucicevic, L.M.; Janjetovic, K.D.; Misirkic, M.S.; Todorovic-Markovic, B.M.; Markovic, Z.M.; Milonjic, S.K.; et al. Opposite effects of nanocrystalline fullerene (C60) on tumour cell growth in vitro and in vivo and a possible role of immunosupression in the cancer-promoting activity of C60. Biomaterials 2009, 30, 6940–6946. [Google Scholar] [CrossRef]
- Cheng-Chieh, Y.; Hwang, K.C.; Yen, C.-C.; Lai, Y.-L. Fullerene derivatives protect against oxidative stress in RAW 264.7 cells and ischemia-reperfused lungs. Am. J. Physiol. Integr. Comp. Physiol. 2004, 287, R21–R26. [Google Scholar] [CrossRef]
- Ryan, J.J.; Bateman, H.R.; Stover, A.; Gomez, G.; Norton, S.K.; Zhao, W.; Schwartz, L.B.; Lenk, R.; Kepley, C.L. Fullerene Nanomaterials Inhibit the Allergic Response. J. Immunol. 2007, 179, 665–672. [Google Scholar] [CrossRef]
- Mitchell, L.A.; Lauer, F.T.; Burchiel, S.W.; McDonald, J.D. Mechanisms for how inhaled multiwalled carbon nanotubes suppress systemic immune function in mice. Nat. Nanotechnol. 2009, 4, 451–456. [Google Scholar] [CrossRef]
- Tkach, A.V.; Shurin, G.V.; Shurin, M.R.; Kisin, E.R.; Murray, A.R.; Young, S.-H.; Star, A.; Fadeel, B.; Kagan, V.E.; Shvedova, A.A. Direct Effects of Carbon Nanotubes on Dendritic Cells Induce Immune Suppression Upon Pulmonary Exposure. ACS Nano 2011, 5, 5755–5762. [Google Scholar] [CrossRef] [PubMed]
- Leghrib, R. Design, Fabrication and Characterisation of Gas Sensors Based on Nanohybrid Materials; Universitat Rovira i Virgili: Tarragona, Spain, 2010. [Google Scholar]
- Dey, R.S.; Purkait, T.; Kamboj, N.; Das, M. Carbonaceous Materials and Future Energy: Clean and Renewable Energy Sources; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Frick, S.U.; Bacher, N.; Baier, G.; Mailänder, V.; Landfester, K.; Steinbrink, K. Functionalized Polystyrene Nanoparticles Trigger Human Dendritic Cell Maturation Resulting in Enhanced CD4+T Cell Activation. Macromol. Biosci. 2012, 12, 1637–1647. [Google Scholar] [CrossRef] [PubMed]
- Getts, D.R.; Martin, A.J.; McCarthy, D.P.; Terry, R.L.; Hunter, Z.N.; Yap, W.T.; Getts, M.T.; Pleiss, M.; Luo, X.; King, N.J.; et al. Microparticles bearing encephalitogenic peptides induce T-cell tolerance and ameliorate experimental autoimmune encephalomyelitis. Nat. Biotechnol. 2012, 30, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Boridy, S.; Soliman, G.M.; Maysinger, D. Modulation of inflammatory signaling and cytokine release from microglia by celastrol incorporated into dendrimer nanocarriers. Nanomedicine 2012, 7, 1149–1165. [Google Scholar] [CrossRef] [PubMed]
- Hayder, M.; Poupot, M.; Baron, M.; Nigon, D.; Turrin, C.-O.; Caminade, A.-M.; Majoral, J.-P.; Eisenberg, R.A.; Fournié, J.-J.; Cantagrel, A.; et al. A Phosphorus-Based Dendrimer Targets Inflammation and Osteoclastogenesis in Experimental Arthritis. Sci. Transl. Med. 2011, 3, 81ra35. [Google Scholar] [CrossRef]
- Teo, I.; Toms, S.M.; Marteyn, B.; Barata, T.S.; Simpson, P.; Johnston, K.A.; Schnupf, P.; Puhar, A.; Bell, T.; Tang, C.; et al. Preventing acute gut wall damage in infectious diarrhoeas with glycosylated dendrimers. EMBO Mol. Med. 2012, 4, 866–881. [Google Scholar] [CrossRef]
- Shukla, N.M.; Salunke, D.; Balakrishna, R.; Mutz, C.A.; Malladi, S.S.; David, S.A. Potent Adjuvanticity of a Pure TLR7-Agonistic Imidazoquinoline Dendrimer. PLoS ONE 2012, 7, e43612. [Google Scholar] [CrossRef]
- Hong, Y.S.; Laks, H.; Cui, G.; Chong, T.; Sen, L. Localized immunosuppression in the cardiac allograft induced by a new liposome-mediated IL-10 gene therapy. J. Heart Lung Transplant. 2002, 21, 1188–1200. [Google Scholar] [CrossRef]
- Hong, S.-T.; Lin, H.; Wang, C.-S.; Chang, C.-H.; Lin, A.M.-Y.; Yang, J.C.-H.; Lo, Y.-L. Improving the anticancer effect of afatinib and microRNA by using lipid polymeric nanoparticles conjugated with dual pH-responsive and targeting peptides. J. Nanobiotechnol. 2019, 17, 89. [Google Scholar] [CrossRef]
- Yu, Z.; Reynaud, F.; Lorscheider, M.; Tsapis, N.; Fattal, E. Nanomedicines for the delivery of glucocorticoids and nucleic acids as potential alternatives in the treatment of rheumatoid arthritis. WIREs Nanomed. Nanobiotechnol. 2020, 12, e1630. [Google Scholar] [CrossRef]
- Metselaar, J.M.; Wauben, M.H.M.; Wagenaar-Hilbers, J.P.A.; Boerman, O.C.; Storm, G. Complete remission of experimental arthritis by joint targeting of glucocorticoids with long-circulating liposomes. Arthritis Rheum. 2003, 48, 2059–2066. [Google Scholar] [CrossRef] [PubMed]
- Szebeni, J.; Bedöcs, P.; Dezsi, L.; Urbanics, R. A porcine model of complement activation-related pseudoallergy to nano-pharmaceuticals: Pros and cons of translation to a preclinical safety test. Precis. Nanomed. 2018, 1, 63–73. [Google Scholar] [CrossRef]
- Muhammad, Q.; Jang, Y.; Kang, S.H.; Moon, J.; Kim, W.J.; Park, H. Modulation of immune responses with nanoparticles and reduction of their immunotoxicity. Biomater. Sci. 2020, 8, 1490–1501. [Google Scholar] [CrossRef] [PubMed]
- Nygaard, U.C.; Hansen, J.S.; Samuelsen, M.; Alberg, T.; Marioara, C.D.; Løvik, M. Single-Walled and Multi-Walled Carbon Nanotubes Promote Allergic Immune Responses in Mice. Toxicol. Sci. 2009, 109, 113–123. [Google Scholar] [CrossRef]
- Hawkins, M.J.; Soon-Shiong, P.; Desai, N. Protein nanoparticles as drug carriers in clinical medicine. Adv. Drug Deliv. Rev. 2008, 60, 876–885. [Google Scholar] [CrossRef]
- Schöler, N.; Hahn, H.; Müller, R.H.; Liesenfeld, O. Effect of lipid matrix and size of solid lipid nanoparticles (SLN) on the viability and cytokine production of macrophages. Int. J. Pharm. 2002, 231, 167–176. [Google Scholar] [CrossRef]
- Shvedova, A.A.; Kisin, E.R.; Mercer, R.; Murray, A.R.; Johnson, V.J.; Potapovich, A.I.; Tyurina, Y.; Gorelik, O.; Arepalli, S.; Schwegler-Berry, D.; et al. Unusual inflammatory and fibrogenic pulmonary responses to single-walled carbon nanotubes in mice. Am. J. Physiol. Cell. Mol. Physiol. 2005, 289, L698–L708. [Google Scholar] [CrossRef]
- Vallhov, H.; Qin, J.; Johansson, S.M.; Ahlborg, N.; Muhammed, M.A.; Scheynius, A.; Gabrielsson, S. The Importance of an Endotoxin-Free Environment during the Production of Nanoparticles Used in Medical Applications. Nano Lett. 2006, 6, 1682–1686. [Google Scholar] [CrossRef]
- Sato, Y.; Yokoyama, A.; Shibata, K.-I.; Akimoto, Y.; Ogino, S.-I.; Nodasaka, Y.; Kohgo, T.; Tamura, K.; Akasaka, T.; Uo, M.; et al. Influence of length on cytotoxicity of multi-walled carbon nanotubes against human acute monocytic leukemia cell line THP-1 in vitro and subcutaneous tissue of rats in vivo. Mol. Biosyst. 2005, 1, 176–182. [Google Scholar] [CrossRef]
- Heuts, J.; Varypataki, E.M.; van der Maaden, K.; Romeijn, S.; Drijfhout, J.W.; van Scheltinga, A.T.; Ossendorp, F.; Jiskoot, W. Cationic Liposomes: A Flexible Vaccine Delivery System for Physicochemically Diverse Antigenic Peptides. Pharm. Res. 2018, 35, 207. [Google Scholar] [CrossRef]
- Yuba, E.; Tajima, N.; Yoshizaki, Y.; Harada, A.; Hayashi, H.; Kono, K. Dextran derivative-based pH-sensitive liposomes for cancer immunotherapy. Biomaterials 2014, 35, 3091–3101. [Google Scholar] [CrossRef] [PubMed]
- Yoshizaki, Y.; Yuba, E.; Sakaguchi, N.; Koiwai, K.; Harada, A.; Kono, K. pH-sensitive polymer-modified liposome-based immunity-inducing system: Effects of inclusion of cationic lipid and CpG-DNA. Biomaterials 2017, 141, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Fallarini, S.; Paoletti, T.; Battaglini, C.O.; Ronchi, P.; Lay, L.; Bonomi, R.; Jha, S.; Mancin, F.; Scrimin, P.; Lombardi, G. Factors affecting T cell responses induced by fully synthetic glyco-gold-nanoparticles. Nanoscale 2012, 5, 390–400. [Google Scholar] [CrossRef]
- Dykman, L.A.; Staroverov, S.; Fomin, A.S.; Khanadeev, V.A.; Khlebtsov, B.; Bogatyrev, V.A. Gold nanoparticles as an adjuvant: Influence of size, shape, and technique of combination with CpG on antibody production. Int. Immunopharmacol. 2018, 54, 163–168. [Google Scholar] [CrossRef] [PubMed]
- de Faria, P.C.B.; dos Santos, L.I.; Coelho, J.P.; Ribeiro, H.B.; Pimenta, M.A.; Ladeira, L.O.; Gomes, D.A.; Furtado, C.A.; Gazzinelli, R.T. Oxidized Multiwalled Carbon Nanotubes as Antigen Delivery System to Promote Superior CD8+ T Cell Response and Protection against Cancer. Nano Lett. 2014, 14, 5458–5470. [Google Scholar] [CrossRef]
- Hassan, H.A.; Smyth, L.; Wang, J.T.-W.; Costa, P.M.; Ratnasothy, K.; Diebold, S.S.; Lombardi, G.; Al-Jamal, K.T. Dual stimulation of antigen presenting cells using carbon nanotube-based vaccine delivery system for cancer immunotherapy. Biomaterials 2016, 104, 310–322. [Google Scholar] [CrossRef]
- Dong, Z.; Wang, Q.; Huo, M.; Zhang, N.; Li, B.; Li, H.; Xu, Y.; Chen, M.; Hong, H.; Wang, Y. Mannose-Modified Multi-Walled Carbon Nanotubes as a Delivery Nanovector Optimizing the Antigen Presentation of Dendritic Cells. ChemistryOpen 2019, 8, 915–921. [Google Scholar] [CrossRef]
- Xia, Q.; Gong, C.; Gu, F.; Wang, Z.; Hu, C.; Zhang, L.; Qiang, L.; Ding, X.; Gao, S.; Gao, Y. Functionalized Multi-Walled Carbon Nanotubes for Targeting Delivery of Immunostimulatory CpG Oligonucleotides Against Prostate Cancer. J. Biomed. Nanotechnol. 2018, 14, 1613–1626. [Google Scholar] [CrossRef]
- Luo, L.; Iqbal, M.Z.; Liu, C.; Xing, J.; Akakuru, O.U.; Fang, Q.; Li, Z.; Dai, Y.; Li, A.; Guan, Y.; et al. Engineered nano-immunopotentiators efficiently promote cancer immunotherapy for inhibiting and preventing lung metastasis of melanoma. Biomaterials 2019, 223, 119464. [Google Scholar] [CrossRef]
- Perica, K.; Tu, A.; Richter, A.; Bieler, J.G.; Edidin, M.; Schneck, J.P. Magnetic Field-Induced T Cell Receptor Clustering by Nanoparticles Enhances T Cell Activation and Stimulates Antitumor Activity. ACS Nano 2014, 8, 2252–2260. [Google Scholar] [CrossRef]
- Zeng, Q.; Li, H.; Jiang, H.; Yu, J.; Wang, Y.; Ke, H.; Gong, T.; Zhang, Z.; Sun, X. Tailoring polymeric hybrid micelles with lymph node targeting ability to improve the potency of cancer vaccines. Biomaterials 2017, 122, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Feng, X.; Wang, J.; He, L.; Wang, C.; Ding, J.; Chen, X. Polymer nanoparticles as adjuvants in cancer immunotherapy. Nano Res. 2018, 11, 5769–5786. [Google Scholar] [CrossRef]
- Li, C.; Zhang, X.; Chen, Q.; Zhang, J.; Li, W.; Hu, H.; Zhao, X.; Qiao, M.; Chen, D. Synthetic Polymeric Mixed Micelles Targeting Lymph Nodes Trigger Enhanced Cellular and Humoral Immune Responses. ACS Appl. Mater. Interfaces 2018, 10, 2874–2889. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Chen, Q.; Wu, J.-P.; Kirk, T.B.; Xu, J.; Liu, Z.; Xue, W. Reduction-Responsive Codelivery System Based on a Metal–Organic Framework for Eliciting Potent Cellular Immune Response. ACS Appl. Mater. Interfaces 2018, 10, 12463–12473. [Google Scholar] [CrossRef]
- Chen, J.; Fang, H.; Hu, Y.; Wu, J.; Zhang, S.; Feng, Y.; Lin, L.; Tian, H.; Chen, X. Combining mannose receptor mediated nanovaccines and gene regulated PD-L1 blockade for boosting cancer immunotherapy. Bioact. Mater. 2021, 7, 167–180. [Google Scholar] [CrossRef]
- Chen, H.; Fan, Y.; Hao, X.; Yang, C.; Peng, Y.; Guo, R.; Shi, X.; Cao, X. Adoptive cellular immunotherapy of tumors via effective CpG delivery to dendritic cells using dendrimer-entrapped gold nanoparticles as a gene vector. J. Mater. Chem. B 2020, 8, 5052–5063. [Google Scholar] [CrossRef]
- Molino, N.M.; Neek, M.; Tucker, J.A.; Nelson, E.L.; Wang, S.-W. Viral-mimicking protein nanoparticle vaccine for eliciting anti-tumor responses. Biomaterials 2016, 86, 83–91. [Google Scholar] [CrossRef]
- Molino, N.M.; Anderson, A.K.L.; Nelson, E.L.; Wang, S.-W. Biomimetic Protein Nanoparticles Facilitate Enhanced Dendritic Cell Activation and Cross-Presentation. ACS Nano 2013, 7, 9743–9752. [Google Scholar] [CrossRef]
- Saadati, R.; Dadashzadeh, S.; Abbasian, Z.; Soleimanjahi, H. Accelerated Blood Clearance of PEGylated PLGA Nanoparticles Following Repeated Injections: Effects of Polymer Dose, PEG Coating, and Encapsulated Anticancer Drug. Pharm. Res. 2012, 30, 985–995. [Google Scholar] [CrossRef]
- Andreev, S.M.; Babakhin, A.A.; Petrukhina, A.O.; Romanova, V.S.; Parnes, Z.N.; Petrov, R.V. Immunogenic and allergenic properties of fulleren conjugates with aminoacids and proteins. Dokl. Biochem. Proc. Acad. Sci. USSR Biochem. Sect. 2002, 370, 4–7. [Google Scholar]
- Fard, J.K.; Jafari, S.; Eghbal, M.A. A Review of Molecular Mechanisms Involved in Toxicity of Nanoparticles. Adv. Pharm. Bull. 2015, 5, 447–454. [Google Scholar] [CrossRef]
- Hendrickson, O.; Fedyunina, N.; Zherdev, A.; Solopova, O.; Sveshnikov, P.; Dzantiev, B. Production of monoclonal antibodies against fullerene C60and development of a fullerene enzyme immunoassay. Analyst 2011, 137, 98–105. [Google Scholar] [CrossRef]
- Eap, S.; Ferrand, A.; Schiavi, J.; Keller, L.; Kokten, T.; Fioretti, F.; Mainard, D.; Ladam, G.; Benkirane-Jessel, N. Collagen implants equipped with ‘fish scale’-like nanoreservoirs of growth factors for bone regeneration. Nanomedicine 2014, 9, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Keller, L.; Eap, S.; Schiavi, J.; Huck, O.; Jacomine, L.; Gauthier, C.; Sebastian, V.; Schwinté, P.; Jessel, N.; Fioretti, F. A living thick nanofibrous implant bifunctionalized with active growth factor and stem cells for bone regeneration. Int. J. Nanomed. 2015, 10, 1061–1075. [Google Scholar] [CrossRef] [PubMed]
- Kossovich, L.; Salkovskiy, Y.; Kirillova, I. Electrospun chitosan nanofiber materials as burn dressing. In Proceedings of the 6th World Congress of Biomechanics (WCB 2010), Singapore, 1–6 August 2010; pp. 1212–1214. [Google Scholar]
- Lin, H.-Y.; Chen, S.-H.; Chang, S.-H.; Huang, S.-T. Tri-layered chitosan scaffold as a potential skin substitute. J. Biomater. Sci. Polym. Ed. 2015, 26, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Diogenes, A.; Kishen, A. Temporal-controlled Dexamethasone Releasing Chitosan Nanoparticle System Enhances Odontogenic Differentiation of Stem Cells from Apical Papilla. J. Endod. 2015, 41, 1253–1258. [Google Scholar] [CrossRef]
- Razavi, S.; Zarkesh-Esfahani, H.; Morshed, M.; Vaezifar, S.; Karbasi, S.; Golozar, M.A. Nanobiocomposite of poly(lactide-co-glycolide)/chitosan electrospun scaffold can promote proliferation and transdifferentiation of Schwann-like cells from human adipose-derived stem cells. J. Biomed. Mater. Res. Part A 2015, 103, 2628–2634. [Google Scholar] [CrossRef]
- Virlan, M.J.R.; Miricescu, D.; Radulescu, R.; Sabliov, C.M.; Totan, A.; Calenic, B.; Greabu, M. Organic Nanomaterials and Their Applications in the Treatment of Oral Diseases. Molecules 2016, 21, 207. [Google Scholar] [CrossRef]
- Rodríguez-Lozano, F.J.; García-Bernal, D.; Aznar-Cervantes, S.; Ros-Roca, M.A.; Algueró, M.C.; Atucha, N.M.; Lozano-García, A.A.; Moraleda, J.M.; Cenis, J.L. Effects of composite films of silk fibroin and graphene oxide on the proliferation, cell viability and mesenchymal phenotype of periodontal ligament stem cells. J. Mater. Sci. Mater. Med. 2014, 25, 2731–2741. [Google Scholar] [CrossRef]
- Xu, W.-P.; Zhang, W.; Asrican, R.; Kim, H.-J.; Kaplan, D.L.; Yelick, P.C. Accurately Shaped Tooth Bud Cell–Derived Mineralized Tissue Formation on Silk Scaffolds. Tissue Eng. Part A 2008, 14, 549–557. [Google Scholar] [CrossRef]
- Gianak, O.; Kyzas, G.Z.; Samanidou, V.F.; Deliyanni, E.A. A Review for the Synthesis of Silk Fibroin Nanoparticles with Different Techniques and Their Ability to be Used for Drug Delivery. Curr. Anal. Chem. 2019, 15, 339–348. [Google Scholar] [CrossRef]
- Mottaghitalab, F.; Farokhi, M.; Shokrgozar, M.A.; Atyabi, F.; Hosseinkhani, H. Silk fibroin nanoparticle as a novel drug delivery system. J. Control. Release 2015, 206, 161–176. [Google Scholar] [CrossRef]
- Kundu, J.; Chung, Y.-I.; Kim, Y.H.; Tae, G.; Kundu, S.C. Silk fibroin nanoparticles for cellular uptake and control release. Int. J. Pharm. 2010, 388, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Zhang, S.; Liu, Z. Graphene based gene transfection. Nanoscale 2011, 3, 1252–1257. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, F.; Yang, H.; Huang, X.; Liu, H.; Zhang, J.; Guo, S. Graphene Oxide as a Matrix for Enzyme Immobilization. Langmuir 2010, 26, 6083–6085. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, Z.; Wang, J.; Li, J.; Lin, Y. Graphene and graphene oxide: Biofunctionalization and applications in biotechnology. Trends Biotechnol. 2011, 29, 205–212. [Google Scholar] [CrossRef]
- Itoo, A.M.; Vemula, S.L.; Gupta, M.T.; Giram, M.V.; Kumar, S.A.; Ghosh, B.; Biswas, S. Multifunctional graphene oxide nanoparticles for drug delivery in cancer. J. Control. Release 2022, 350, 26–59. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Soroshnia, S.; Hashemi, S.A.; Babapoor, A.; Ghasemi, Y.; Savardashtaki, A.; Amani, A.M. Graphene nano-ribbon based high potential and efficiency for DNA, cancer therapy and drug delivery applications. Drug Metab. Rev. 2019, 51, 91–104. [Google Scholar] [CrossRef]
- Iannazzo, D.; Pistone, A.; Celesti, C.; Triolo, C.; Patané, S.; Giofré, S.V.; Romeo, R.; Ziccarelli, I.; Mancuso, R.; Gabriele, B.; et al. A Smart Nanovector for Cancer Targeted Drug Delivery Based on Graphene Quantum Dots. Nanomaterials 2019, 9, 282. [Google Scholar] [CrossRef]
- Basu, A.; Upadhyay, P.; Ghosh, A.; Bose, A.; Gupta, P.; Chattopadhyay, S.; Chattopadhyay, D.; Adhikary, A. Hyaluronic acid engrafted metformin loaded graphene oxide nanoparticle as CD44 targeted anti-cancer therapy for triple negative breast cancer. Biochim. Et Biophys. Acta (BBA)-Gen. Subj. 2021, 1865, 129841. [Google Scholar] [CrossRef]
- Maleki, M.; Zarezadeh, R.; Nouri, M.; Sadigh, A.R.; Pouremamali, F.; Asemi, Z.; Kafil, H.S.; Alemi, F.; Yousefi, B. Graphene Oxide: A Promising Material for Regenerative Medicine and Tissue Engineering. Biomol. Concepts 2020, 11, 182–200. [Google Scholar] [CrossRef] [PubMed]
- Grebinyk, A.; Prylutska, S.; Buchelnikov, A.; Tverdokhleb, N.; Grebinyk, S.; Evstigneev, M.; Matyshevska, O.; Cherepanov, V.; Prylutskyy, Y.; Yashchuk, V.; et al. C60 Fullerene as an Effective Nanoplatform of Alkaloid Berberine Delivery into Leukemic Cells. Pharmaceutics 2019, 11, 586. [Google Scholar] [CrossRef] [PubMed]
- Bilobrov, V.; Sokolova, V.; Prylutska, S.; Panchuk, R.; Litsis, O.; Osetskyi, V.; Evstigneev, M.; Prylutskyy, Y.; Epple, M.; Ritter, U.; et al. A Novel Nanoconjugate of Landomycin A with C60 Fullerene for Cancer Targeted Therapy: In Vitro Studies. Cell. Mol. Bioeng. 2018, 12, 41–51. [Google Scholar] [CrossRef]
- Borowik, A.; Prylutskyy, Y.; Kawelski, Ł.; Kyzyma, O.; Bulavin, L.; Ivankov, O.; Cherepanov, V.; Wyrzykowski, D.; Kaźmierkiewicz, R.; Gołuński, G.; et al. Does C60 fullerene act as a transporter of small aromatic molecules? Colloids Surfaces B: Biointerfaces 2018, 164, 134–143. [Google Scholar] [CrossRef]
- Prylutskyy, Y.I.; Evstigneev, M.P.; Cherepanov, V.V.; Kyzyma, O.A.; Bulavin, L.A.; Davidenko, N.A.; Scharff, P. Structural organization of C60 fullerene, doxorubicin, and their complex in physiological solution as promising antitumor agents. J. Nanoparticle Res. 2015, 17, 45. [Google Scholar] [CrossRef]
- Wang, I.C.; Tai, L.A.; Lee, D.D.; Kanakamma, P.P.; Shen, C.K.-F.; Luh, T.-Y.; Cheng, C.H.; Hwang, K.C. C60 and Water-Soluble Fullerene Derivatives as Antioxidants Against Radical-Initiated Lipid Peroxidation. J. Med. Chem. 1999, 42, 4614–4620. [Google Scholar] [CrossRef] [PubMed]
- Horie, M.; Fukuhara, A.; Saito, Y.; Yoshida, Y.; Sato, H.; Ohi, H.; Obata, M.; Mikata, Y.; Yano, S.; Niki, E. Antioxidant action of sugar-pendant C60 fullerenes. Bioorganic Med. Chem. Lett. 2009, 19, 5902–5904. [Google Scholar] [CrossRef]
- Engin, A.B.; Engin, A.J. Nanoantibiotics: A novel rational approach to antibiotic resistant infections. Curr. Drug. Metab. 2019, 20, 720–741. [Google Scholar] [CrossRef]
- Sethi, S. Approaches to Anti Infective Therapies. Bentham Sci. 2021, 5, 120. [Google Scholar]
- Xu, P.-Y.; Li, X.-Q.; Chen, W.-G.; Deng, L.-L.; Tan, Y.-Z.; Zhang, Q.; Xie, S.-Y.; Zheng, L.-S. Progress in Antiviral Fullerene Research. Nanomaterials 2022, 12, 2547. [Google Scholar] [CrossRef]
- Marchesan, S.; Da Ros, T.; Spalluto, G.; Balzarini, J.; Prato, M. Anti-HIV properties of cationic fullerene derivatives. Bioorganic Med. Chem. Lett. 2005, 15, 3615–3618. [Google Scholar] [CrossRef]
- Heredia, D.A.; Durantini, A.M.; Durantini, J.E.; Durantini, E.N. Fullerene C60 derivatives as antimicrobial photodynamic agents. J. Photochem. Photobiol. C Photochem. Rev. 2021, 51, 100471. [Google Scholar] [CrossRef]
- Gaur, M.; Misra, C.; Yadav, A.B.; Swaroop, S.; Maolmhuaidh, F.; Bechelany, M.; Barhoum, A. Biomedical Applications of Carbon Nanomaterials: Fullerenes, Quantum Dots, Nanotubes, Nanofibers, and Graphene. Materials 2021, 14, 5978. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lee, S.; Koo, K.-I.; Kim, K. Nanowire-Based Sensors for Biological and Medical Applications. IEEE Trans. NanoBiosci. 2016, 15, 186–199. [Google Scholar] [CrossRef] [PubMed]
- Bian, Z.; Wang, R.J.; Wang, W.H.; Zhang, T.; Inoue, A. Carbon-Nanotube-Reinforced Zr-Based Bulk Metallic Glass Composites and Their Properties. Adv. Funct. Mater. 2004, 14, 55–63. [Google Scholar] [CrossRef]
- Patolsky, F.; Zheng, G.; Lieber, C.M. Nanowire sensors for medicine and the life sciences. Nanomedicine 2006, 1, 51–65. [Google Scholar] [CrossRef]
- Eatemadi, A.; Daraee, H.; Karimkhanloo, H.; Kouhi, M.; Zarghami, N.; Akbarzadeh, A.; Abasi, M.; Hanifehpour, Y.; Joo, S.W. Carbon nanotubes: Properties, synthesis, purification, and medical applications. Nanoscale Res. Lett. 2014, 9, 393. [Google Scholar] [CrossRef]
- Ren, G.; Oxford, J.S.; Reip, P.W.; Lambkin-Williams, R.; Mann, A. Anti-viral formulations nanomaterials and nanoparticles. US13/691,099, 18 April 2013. [Google Scholar]
- Bankier, C.; Matharu, R.K.; Cheong, Y.K.; Ren, G.G.; Cloutman-Green, E.; Ciric, L. Synergistic Antibacterial Effects of Metallic Nanoparticle Combinations. Sci. Rep. 2019, 9, 16074. [Google Scholar] [CrossRef]
- Slavin, Y.N.; Asnis, J.; Häfeli, U.O.; Bach, H. Metal nanoparticles: Understanding the mechanisms behind antibacterial activity. J. Nanobiotechnol. 2017, 15, 65. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmad, A.; Oves, M.; Ismail, I.M.I.; Qari, H.A.; Umar, K.; Ibrahim, M.N.M. Recent Advances in Metal Decorated Nanomaterials and Their Various Biological Applications: A Review. Front. Chem. 2020, 8, 341. [Google Scholar] [CrossRef]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial activity of the metals and metal oxide nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Khezerlou, A.; Alizadeh-Sani, M.; Azizi-Lalabadi, M.; Ehsani, A. Nanoparticles and their antimicrobial properties against pathogens including bacteria, fungi, parasites and viruses. Microb. Pathog. 2018, 123, 505–526. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Feng, X. Gold nanoparticles for skin drug delivery. Int. J. Pharm. 2022, 625, 122122. [Google Scholar] [CrossRef]
- Khoshnevisan, K.; Daneshpour, M.; Barkhi, M.; Gholami, M.; Samadian, H.; Maleki, H. The promising potentials of capped gold nanoparticles for drug delivery systems. J. Drug Target. 2017, 26, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Desai, N.; Momin, M.; Khan, T.; Gharat, S.; Ningthoujam, R.S.; Omri, A. Metallic nanoparticles as drug delivery system for the treatment of cancer. Expert Opin. Drug Deliv. 2021, 18, 1261–1290. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Lv, H.; Zhao, B.; Zhou, L.; Wang, S.; Luo, J.; Liu, J.; Shang, P. Iron and leukemia: New insights for future treatments. J. Exp. Clin. Cancer Res. 2019, 38, 406. [Google Scholar] [CrossRef] [PubMed]
- Hassanen, E.I.; Korany, R.M.S.; Bakeer, A.M. Cisplatin-conjugated gold nanoparticles-based drug delivery system for targeting hepatic tumors. J. Biochem. Mol. Toxicol. 2021, 35, e22722. [Google Scholar] [CrossRef]
- Shevtsov, M.; Zhou, Y.; Khachatryan, W.; Multhoff, G.; Gao, H. Recent Advances in Gold Nanoformulations for Cancer Therapy. Curr. Drug Metab. 2018, 19, 768–780. [Google Scholar] [CrossRef]
- Singh, P.; Pandit, S.; Mokkapati, V.; Garg, A.; Ravikumar, V.; Mijakovic, I. Gold Nanoparticles in Diagnostics and Therapeutics for Human Cancer. Int. J. Mol. Sci. 2018, 19, 1979. [Google Scholar] [CrossRef]
- Sacks, D.; Baxter, B.; Campbell, B.C.V.; Carpenter, J.S.; Cognard, C.; Dippel, D.; Eesa, M.; Fischer, U.; Hausegger, K.; Hirsch, J.A.; et al. Multisociety Consensus Quality Improvement Revised Consensus Statement for Endovascular Therapy of Acute Ischemic Stroke. Int. J. Stroke 2018, 13, 612–632. [Google Scholar] [CrossRef]
- Allahverdiyev, A.M.; Abamor, E.S.; Bagirova, M.; Rafailovich, M. Antimicrobial effects of TiO2 and Ag2O nanoparticles against drug-resistant bacteria and leishmania parasites. Futur. Microbiol. 2011, 6, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, S.; Reboul, J.; Diring, S.; Sumida, K.; Kitagawa, S. Structuring of metal–organic frameworks at the mesoscopic/macroscopic scale. Chem. Soc. Rev. 2014, 43, 5700–5734. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Lu, K.; Poon, C.; Lin, W. Metal–Organic Frameworks as Sensory Materials and Imaging Agents. Inorg. Chem. 2013, 53, 1916–1924. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.M.; Salgado, A.; Sousa, N.; Mano, J.; Reis, R.L. Dendrimers and derivatives as a potential therapeutic tool in regenerative medicine strategies—A review. Prog. Polym. Sci. 2010, 35, 1163–1194. [Google Scholar] [CrossRef]
- Shaikh, A.; Kesharwani, P.; Gajbhiye, V. Dendrimer as a momentous tool in tissue engineering and regenerative medicine. J. Control. Release 2022, 346, 328–354. [Google Scholar] [CrossRef]
- Zhu, L. The Synthesis of Nano-Doxorubicin and its Anticancer Effect. Anti-Cancer Agents Med. Chem. 2021, 21, 2466–2477. [Google Scholar] [CrossRef] [PubMed]
- Mehdipoor, E.; Adeli, M.; Bavadi, M.; Sasanpour, P.; Rashidian, B. A possible anticancer drug delivery system based on carbon nanotube–dendrimer hybrid nanomaterials. J. Mater. Chem. 2011, 21, 15456–15463. [Google Scholar] [CrossRef]
- Medina, S.H.; El-Sayed, M.E.H. Dendrimers as Carriers for Delivery of Chemotherapeutic Agents. Chem. Rev. 2009, 109, 3141–3157. [Google Scholar] [CrossRef]
- Wrońska, N.; Majoral, J.P.; Appelhans, D.; Bryszewska, M.; Lisowska, K. Synergistic Effects of Anionic/Cationic Dendrimers and Levofloxacin on Antibacterial Activities. Molecules 2019, 24, 2894. [Google Scholar] [CrossRef]
- Winnicka, K.; Wroblewska, M.; Wieczorek, P.; Sacha, P.T.; Tryniszewska, E.A. The Effect of PAMAM Dendrimers on the Antibacterial Activity of Antibiotics with Different Water Solubility. Molecules 2013, 18, 8607–8617. [Google Scholar] [CrossRef]
- Chen, C.Z.; Beck-Tan, N.C.; Dhurjati, P.; van Dyk, T.K.; LaRossa, R.A.; Cooper, S.L. Quaternary Ammonium Functionalized Poly(propylene imine) Dendrimers as Effective Antimicrobials: Structure−Activity Studies. Biomacromolecules 2000, 1, 473–480. [Google Scholar] [CrossRef]
- Reynolds, J.M.; Martinez, G.J.; Chung, Y.; Dong, C. Toll-like receptor 4 signaling in T cells promotes autoimmune inflammation. Proc. Natl. Acad. Sci. USA 2012, 109, 13064–13069. [Google Scholar] [CrossRef]
- Cui, Y.; Liu, H.; Zhou, M.; Duan, Y.; Li, N.; Gong, X.; Hu, R.; Hong, M.; Hong, F. Signaling pathway of inflammatory responses in the mouse liver caused by TiO2 nanoparticles. J. Biomed. Mater. Res. Part A 2010, 96A, 221–229. [Google Scholar] [CrossRef]
- Yuan, C.; Liu, Y.; Wang, T.; Sun, M.; Chen, X. Nanomaterials as Smart Immunomodulator Delivery System for Enhanced Cancer Therapy. ACS Biomater. Sci. Eng. 2020, 6, 4774–4798. [Google Scholar] [CrossRef]
- Dugershaw, B.B.; Aengenheister, L.; Hansen, S.S.K.; Hougaard, K.S.; Buerki-Thurnherr, T. Recent insights on indirect mechanisms in developmental toxicity of nanomaterials. Part. Fibre Toxicol. 2020, 17, 31. [Google Scholar] [CrossRef]
- Vasilichin, V.A.; Tsymbal, S.A.; Fakhardo, A.F.; Anastasova, E.I.; Marchenko, A.S.; Shtil, A.A.; Vinogradov, V.V.; Koshel, E.I. Effects of Metal Oxide Nanoparticles on Toll-Like Receptor mRNAs in Human Monocytes. Nanomaterials 2020, 10, 127. [Google Scholar] [CrossRef]
- Fischer, S.; Schlosser, E.; Mueller, M.; Csaba, N.; Merkle, H.P.; Groettrup, M.; Gander, B. Concomitant delivery of a CTL-restricted peptide antigen and CpG ODN by PLGA microparticles induces cellular immune response. J. Drug Target. 2009, 17, 652–661. [Google Scholar] [CrossRef]
- Heit, A.; Schmitz, F.; Haas, T.; Busch, D.H.; Wagner, H. Antigen co-encapsulated with adjuvants efficiently drive protective T cell immunity. Eur. J. Immunol. 2007, 37, 2063–2074. [Google Scholar] [CrossRef]
- Schlosser, E.; Mueller, M.; Fischer, S.; Basta, S.; Busch, D.H.; Gander, B.; Groettrup, M. TLR ligands and antigen need to be coencapsulated into the same biodegradable microsphere for the generation of potent cytotoxic T lymphocyte responses. Vaccine 2008, 26, 1626–1637. [Google Scholar] [CrossRef]
- Kasturi, S.P.; Skountzou, I.; Albrecht, R.A.; Koutsonanos, D.; Hua, T.; Nakaya, H.I.; Ravindran, R.; Stewart, S.; Alam, M.; Kwissa, M. Programming the Magnitude and Persistence of Antibody Responses with Innate Immunity. Nature 2011, 470, 543–547. [Google Scholar] [CrossRef]
- Bal, S.M.; Slütter, B.; Verheul, R.; Bouwstra, J.A.; Jiskoot, W. Adjuvanted, antigen loaded N-trimethyl chitosan nanoparticles for nasal and intradermal vaccination: Adjuvant- and site-dependent immunogenicity in mice. Eur. J. Pharm. Sci. 2012, 45, 475–481. [Google Scholar] [CrossRef]
- Kim, H.; Niu, L.; Larson, P.; Kucaba, T.A.; Murphy, K.A.; James, B.R.; Ferguson, D.M.; Griffith, T.S.; Panyam, J. Polymeric nanoparticles encapsulating novel TLR7/8 agonists as immunostimulatory adjuvants for enhanced cancer immunotherapy. Biomaterials 2018, 164, 38–53. [Google Scholar] [CrossRef]
- Ilyinskii, P.O.; Roy, C.; O’Neil, C.P.; Browning, E.A.; Pittet, L.A.; Altreuter, D.H.; Alexis, F.; Tonti, E.; Shi, J.; Basto, P.; et al. Adjuvant-carrying synthetic vaccine particles augment the immune response to encapsulated antigen and exhibit strong local immune activation without inducing systemic cytokine release. Vaccine 2014, 32, 2882–2895. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-Y.; Kim, S.; Kim, J.-E.; Lee, S.N.; Shin, I.W.; Shin, H.S.; Jin, S.M.; Noh, Y.-W.; Kang, Y.J.; Kim, Y.S.; et al. Lyophilizable and multifaceted toll-like receptor 7/8 agonist-loaded nanoemulsion for the reprogramming of tumor microenvironments and enhanced cancer immunotherapy. ACS Nano 2019, 13, 12671–12686. [Google Scholar] [CrossRef]
- Silva, D.-A.; Yu, S.; Ulge, U.Y.; Spangler, J.B.; Jude, K.M.; Labão-Almeida, C.; Ali, L.R.; Quijano-Rubio, A.; Ruterbusch, M.; Leung, I.; et al. De novo design of potent and selective mimics of IL-2 and IL-15. Nature 2019, 565, 186–191. [Google Scholar] [CrossRef]
- Park, Y.-H.; Jeong, S.H.; Yi, S.M.; Choi, B.H.; Kim, Y.-R.; Kim, I.-K.; Kim, M.-K.; Son, S.W. Analysis for the potential of polystyrene and TiO2 nanoparticles to induce skin irritation, phototoxicity, and sensitization. Toxicol. Vitr. 2011, 25, 1863–1869. [Google Scholar] [CrossRef]
- Lee, S.; Yun, H.-S.; Kim, S.-H. The comparative effects of mesoporous silica nanoparticles and colloidal silica on inflammation and apoptosis. Biomaterials 2011, 32, 9434–9443. [Google Scholar] [CrossRef]
- Thyssen, J.P.; Menné, T. Metal Allergy—A Review on Exposures, Penetration, Genetics, Prevalence, and Clinical Implications. Chem. Res. Toxicol. 2009, 23, 309–318. [Google Scholar] [CrossRef]
- Swinnen, I.; Goossens, A. An update on airborne contact dermatitis: 2007-2011. Contact Dermat. 2013, 68, 232–238. [Google Scholar] [CrossRef]
- Auttachoat, W.; McLoughlin, C.E.; White, K.L.J.; Smith, M.J. Route-dependent systemic and local immune effects following exposure to solutions prepared from titanium dioxide nanoparticles. J. Immunotoxicol. 2014, 11, 273–282. [Google Scholar] [CrossRef]
- Fischer, H.C.; Chan, W.C. Nanotoxicity: The growing need for in vivo study. Curr. Opin. Biotechnol. 2007, 18, 565–571. [Google Scholar] [CrossRef]
- Alyamani, H.; Obeid, M.A.; Tate, R.J.; Ferro, V.A. Exosomes: Fighting cancer with cancer. Ther. Deliv. 2019, 10, 37–61. [Google Scholar] [CrossRef] [PubMed]
- Al Qaraghuli, M.M.; Alzahrani, A.R.; Niwasabutra, K.; Obeid, M.A.; Ferro, V.A. Where traditional drug discovery meets modern technology in the quest for new drugs. Ann. Pharmacol. Pharm. 2017, 2, 1061. [Google Scholar]
- Gu, H.; Xu, K.; Yang, Z.; Chang, C.K.; Xu, B. Synthesis and cellular uptake of porphyrin decorated iron oxide nanoparticles—A potential candidate for bimodal anticancer therapy. Chem. Commun. 2005, 4270–4272. [Google Scholar] [CrossRef] [PubMed]
- Tomić, S.; Đokić, J.; Vasilijić, S.; Ogrinc, N.; Rudolf, R.; Pelicon, P.; Vučević, D.; Milosavljević, P.; Janković, S.; Anžel, I.; et al. Size-dependent effects of gold nanoparticles uptake on maturation and antitumor functions of human dendritic cells In Vitro. PloS ONE 2014, 9, e96584. [Google Scholar] [CrossRef]
- Rothen-Rutishauser, B.; Kuhn, D.A.; Ali, Z.; Gasser, M.; Amin, F.; Parak, W.J.; Vanhecke, D.; Fink, A.; Gehr, P.; Brandenberger, C. Quantification of gold nanoparticle cell uptake under controlled biological conditions and adequate resolution. Nanomedicine 2014, 9, 607–621. [Google Scholar] [CrossRef]
- Rothen-Rutishauser, B.M.; Schürch, S.; Haenni, B.; Kapp, N.; Gehr, P. Interaction of Fine Particles and Nanoparticles with Red Blood Cells Visualized with Advanced Microscopic Techniques. Environ. Sci. Technol. 2006, 40, 4353–4359. [Google Scholar] [CrossRef] [PubMed]
- Oh, W.-K.; Kim, S.; Choi, M.; Kim, C.; Jeong, Y.S.; Cho, B.-R.; Hahn, J.-S.; Jang, J. Cellular uptake, cytotoxicity, and innate immune response of silica− titania hollow nanoparticles based on size and surface functionality. ACS Nano 2010, 4, 5301–5313. [Google Scholar] [CrossRef]
- Liu, Y.; Yin, Y.; Wang, L.; Zhang, W.; Chen, X.; Yang, X.; Xu, J.; Ma, G. Surface hydrophobicity of microparticles modulates adjuvanticity. J. Mater. Chem. B 2013, 1, 3888–3896. [Google Scholar] [CrossRef]
- Moyano, D.F.; Goldsmith, M.; Solfiell, D.J.; Landesman-Milo, D.; Miranda, O.R.; Peer, D.; Rotello, V.M. Nanoparticle Hydrophobicity Dictates Immune Response. J. Am. Chem. Soc. 2012, 134, 3965–3967. [Google Scholar] [CrossRef]
- Saha, K.; Rahimi, M.; Yazdani, M.; Kim, S.T.; Moyano, D.F.; Hou, S.; Das, R.; Mout, R.; Rezaee, F.; Mahmoudi, M.; et al. Regulation of Macrophage Recognition through the Interplay of Nanoparticle Surface Functionality and Protein Corona. ACS Nano 2016, 10, 4421–4430. [Google Scholar] [CrossRef] [PubMed]
- Obeid, M.A.; Gany, S.A.S.; Gray, A.I.; Young, L.; Igoli, J.O.; Ferro, V.A. Niosome-encapsulated balanocarpol: Compound isolation, characterisation, and cytotoxicity evaluation against human breast and ovarian cancer cell lines. Nanotechnology 2020, 31, 195101. [Google Scholar] [CrossRef] [PubMed]
- Dadashzadeh, S.; Mirahmadi, N.; Babaei, M.; Vali, A. Peritoneal retention of liposomes: Effects of lipid composition, PEG coating and liposome charge. J. Control. Release 2010, 148, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Meyer, O.; Kirpotin, D.; Hong, K.; Sternberg, B.; Park, J.W.; Woodle, M.C.; Papahadjopoulos, D. Cationic Liposomes Coated with Polyethylene Glycol As Carriers for Oligonucleotides. J. Biol. Chem. 1998, 273, 15621–15627. [Google Scholar] [CrossRef]
- Kawai, N.; Ito, A.; Nakahara, Y.; Futakuchi, M.; Shirai, T.; Honda, H.; Kobayashi, T.; Kohri, K. Anticancer effect of hyperthermia on prostate cancer mediated by magnetite cationic liposomes and immune-response induction in transplanted syngeneic rats. Prostate 2005, 64, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.; Kisin, E.; Leonard, S.; Young, S.; Kommineni, C.; Kagan, V.; Castranova, V.; Shvedova, A. Oxidative stress and inflammatory response in dermal toxicity of single-walled carbon nanotubes. Toxicology 2009, 257, 161–171. [Google Scholar] [CrossRef]
- Katwa, P.; Wang, X.; Urankar, R.N.; Podila, R.; Hilderbrand, S.C.; Fick, R.B.; Rao, A.M.; Ke, P.C.; Wingard, C.J.; Brown, J.M. A Carbon Nanotube Toxicity Paradigm Driven by Mast Cells and the IL-33/ST2 Axis. Small 2012, 8, 2904–2912. [Google Scholar] [CrossRef]
- Niikura, K.; Matsunaga, T.; Suzuki, T.; Kobayashi, S.; Yamaguchi, H.; Orba, Y.; Kawaguchi, A.; Hasegawa, H.; Kajino, K.; Ninomiya, T.; et al. Gold Nanoparticles as a Vaccine Platform: Influence of Size and Shape on Immunological Responses In Vitro and In Vivo. ACS Nano 2013, 7, 3926–3938. [Google Scholar] [CrossRef]
- Heidegger, S.; Gößl, D.; Schmidt, A.; Niedermayer, S.; Argyo, C.; Endres, S.; Bein, T.; Bourquin, C. Immune response to functionalized mesoporous silica nanoparticles for targeted drug delivery. Nanoscale 2015, 8, 938–948. [Google Scholar] [CrossRef]
- Deng, Z.J.; Liang, M.; Monteiro, M.; Toth, I.; Minchin, R.F. Nanoparticle-induced unfolding of fibrinogen promotes Mac-1 receptor activation and inflammation. Nat. Nanotechnol. 2010, 6, 39–44. [Google Scholar] [CrossRef]
- Dailey, L.A.; Jekel, N.; Fink, L.; Gessler, T.; Schmehl, T.; Wittmar, M.; Kissel, T.; Seeger, W. Investigation of the proinflammatory potential of biodegradable nanoparticle drug delivery systems in the lung. Toxicol. Appl. Pharmacol. 2006, 215, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Albanese, A.; Tang, P.S.; Chan, W.C. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu. Rev. Biomed. Eng. 2012, 14, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Trindade, R.; Albrektsson, T.; Tengvall, P.; Wennerberg, A. Foreign Body Reaction to Biomaterials: On Mechanisms for Buildup and Breakdown of Osseointegration. Clin. Implant. Dent. Relat. Res. 2014, 18, 192–203. [Google Scholar] [CrossRef]
- Dvir, T.; Timko, B.P.; Kohane, D.S.; Langer, R. Nanotechnological strategies for engineering complex tissues. Nat. Nanotechnol. 2011, 6, 13–22. [Google Scholar] [CrossRef]
- Demento, S.L.; Eisenbarth, S.C.; Foellmer, H.G.; Platt, C.; Caplan, M.J.; Saltzman, W.M.; Mellman, I.; Ledizet, M.; Fikrig, E.; Flavell, R.A.; et al. Inflammasome-activating nanoparticles as modular systems for optimizing vaccine efficacy. Vaccine 2009, 27, 3013–3021. [Google Scholar] [CrossRef]
- Malik, A.F.; Hoque, R.; Ouyang, X.; Ghani, A.; Hong, E.; Khan, K.; Moore, L.B.; Ng, G.; Munro, F.; Flavell, R.A.; et al. Inflammasome components Asc and caspase-1 mediate biomaterial-induced inflammation and foreign body response. Proc. Natl. Acad. Sci. USA 2011, 108, 20095–20100. [Google Scholar] [CrossRef]
- Chapekar, M.S. Tissue engineering: Challenges and opportunities. J. Biomed. Mater. Res. 2000, 53, 617–620. [Google Scholar] [CrossRef]
- Rubin, J.P.; Gurtner, G.C.; Liu, W.; March, K.L.; Seppänen-Kaijansinkko, R.; Yaszemski, M.J.; Yoo, J.J. Tissue Engineering Clinical Council Roundtable Discussion: Opportunities and Challenges in Clinical Translation. Tissue Eng. Part A 2020, 26, 113–119. [Google Scholar] [CrossRef]
- Boraschi, D.; Italiani, P.; Palomba, R.; Decuzzi, P.; Duschl, A.; Fadeel, B.; Moghimi, S.M. Nanoparticles and innate immunity: New perspectives on host defence. Semin. Immunol. 2017, 34, 33–51. [Google Scholar] [CrossRef]
- Verhoef, J.J.F.; Carpenter, J.F.; Anchordoquy, T.J.; Schellekens, H. Potential induction of anti-PEG antibodies and complement activation toward PEGylated therapeutics. Drug Discov. Today 2014, 19, 1945–1952. [Google Scholar] [CrossRef] [PubMed]
- Fadeel, B. Hide and Seek: Nanomaterial Interactions With the Immune System. Front. Immunol. 2019, 10, 133. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Z.-Y.; Zhao, W.-J.; Gao, Y.-J.; Cong, Y.; Zhao, L.; Hu, Z.; Wang, H. Reconfigurable Peptide Nanotherapeutics at Tumor Microenvironmental pH. ACS Appl. Mater. Interfaces 2017, 9, 30426–30436. [Google Scholar] [CrossRef] [PubMed]
- Oldenborg, P.-A.; Zheleznyak, A.; Fang, Y.-F.; Lagenaur, C.F.; Gresham, H.D.; Lindberg, F.P. Role of CD47 as a Marker of Self on Red Blood Cells. Science 2000, 288, 2051–2054. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, S.; Jamieson, C.H.; Pang, W.W.; Park, C.Y.; Chao, M.P.; Majeti, R.; Traver, D.; van Rooijen, N.; Weissman, I.L. CD47 Is Upregulated on Circulating Hematopoietic Stem Cells and Leukemia Cells to Avoid Phagocytosis. Cell 2009, 138, 271–285. [Google Scholar] [CrossRef]
- Rodriguez, P.L.; Harada, T.; Christian, D.A.; Pantano, D.A.; Tsai, R.K.; Discher, D.E. Minimal “Self” Peptides That Inhibit Phagocytic Clearance and Enhance Delivery of Nanoparticles. Science 2013, 339, 971–975. [Google Scholar] [CrossRef]
- Zhang, K.-L.; Zhou, J.; Zhou, H.; Wu, Y.; Liu, R.; Wang, L.-L.; Lin, W.-W.; Huang, G.; Yang, H.-H. Bioinspired “Active” Stealth Magneto-Nanomicelles for Theranostics Combining Efficient MRI and Enhanced Drug Delivery. ACS Appl. Mater. Interfaces 2017, 9, 30502–30509. [Google Scholar] [CrossRef]
| NPs Effect | Immunosuppression | Immunostimulant | References |
|---|---|---|---|
| Desirable | Reducing allergic response | Used as cancer immunotherapy | [27,28] |
| Organ and tissue transplant rejection | Enhanced vaccine efficacy | [29] | |
| Very useful for autoimmune disorders and inflammation treatment | Antitumorigenic | [30] | |
| Undesirable | Severe suppression of the immune system to a level affecting the recognition | Development of inflammation | [31] |
| Myelosuppression (bone marrow and thymus gland) | Development of hypersensitivity | [32] | |
| Development of anaphylaxis | [27] |
| Nanomaterial | Medical Use | References | |
|---|---|---|---|
| Organic | Chitosan nanosystems | Dental medicine applications | [97,98,99,100,101] |
| Wound dressing, tissue regeneration | [48,99,102] | ||
| Silk fibroin | Dental medicine applications | [103,104,105] | |
| Drug delivery | [106,107,108] | ||
| Graphene- and Graphene oxide- based nanosystems | Gene and small molecular drug delivery Biofunctionalization of proteins Anticancer therapy Antimicrobial agent for bone and teeth implantation Regenerative medicine | [109,110,111] [112,113,114,115,116] | |
| Fullerenes | Anticancer-targeted drug delivery Antioxidant Anti-infective agents Medical implants, tissue engineering, wound healing, biosensing, bioimaging, vaccination, and photodynamic therapy | [117,118,119,120,121,122,123,124,125,126,127,128] | |
| Carbon nanowires and nantotubes | Medical implants, tissue engineering, wound healing, chemosensing, biosensing, bioimaging, vaccination, and photodynamic therapy | [128,129,130,131,132] | |
| Inorganic | Metals-based nanosystems | Antimicrobial agents (Ag, Cu, WC, Au, Fe nanosystems) | [133,134,135,136,137,138] |
| Targeted drug delivery systems | [139,140,141,142] | ||
| Diagnosis and anticancer therapy (Au, Ag, Zn, Ti) | [143,144,145,146] | ||
| Metal-oxide nanosystems | Antimicrobial agents (TiO2, ZnO, NO, Ag2O, CaO, MgO, CuO) | [135,137,138,147] | |
| Biosensing and bioimaging | [148,149] | ||
| Dendrimers | Regenerative medicine | [150,151] | |
| Drug delivery | [146,152,153,154] | ||
| Antimicrobial agents | [155,156,157] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aljabali, A.A.; Obeid, M.A.; Bashatwah, R.M.; Serrano-Aroca, Á.; Mishra, V.; Mishra, Y.; El-Tanani, M.; Hromić-Jahjefendić, A.; Kapoor, D.N.; Goyal, R.; et al. Nanomaterials and Their Impact on the Immune System. Int. J. Mol. Sci. 2023, 24, 2008. https://doi.org/10.3390/ijms24032008
Aljabali AA, Obeid MA, Bashatwah RM, Serrano-Aroca Á, Mishra V, Mishra Y, El-Tanani M, Hromić-Jahjefendić A, Kapoor DN, Goyal R, et al. Nanomaterials and Their Impact on the Immune System. International Journal of Molecular Sciences. 2023; 24(3):2008. https://doi.org/10.3390/ijms24032008
Chicago/Turabian StyleAljabali, Alaa A., Mohammad A. Obeid, Rasha M. Bashatwah, Ángel Serrano-Aroca, Vijay Mishra, Yachana Mishra, Mohamed El-Tanani, Altijana Hromić-Jahjefendić, Deepak N. Kapoor, Rohit Goyal, and et al. 2023. "Nanomaterials and Their Impact on the Immune System" International Journal of Molecular Sciences 24, no. 3: 2008. https://doi.org/10.3390/ijms24032008
APA StyleAljabali, A. A., Obeid, M. A., Bashatwah, R. M., Serrano-Aroca, Á., Mishra, V., Mishra, Y., El-Tanani, M., Hromić-Jahjefendić, A., Kapoor, D. N., Goyal, R., Naikoo, G. A., & Tambuwala, M. M. (2023). Nanomaterials and Their Impact on the Immune System. International Journal of Molecular Sciences, 24(3), 2008. https://doi.org/10.3390/ijms24032008














