Transplacental Therapeutic Drug Monitoring in Pregnant Women with Fetal Tachyarrhythmia Using HPLC-MS/MS
Abstract
1. Introduction
2. Results
2.1. Comparative Clinical Data between Pregnant Persons and Controls
2.2. Efficacy of Transplacental Fetal Treatment
2.3. The 3435C > T Polymorphism of the ABCB1 Gene in Pregnant Women and Newborns
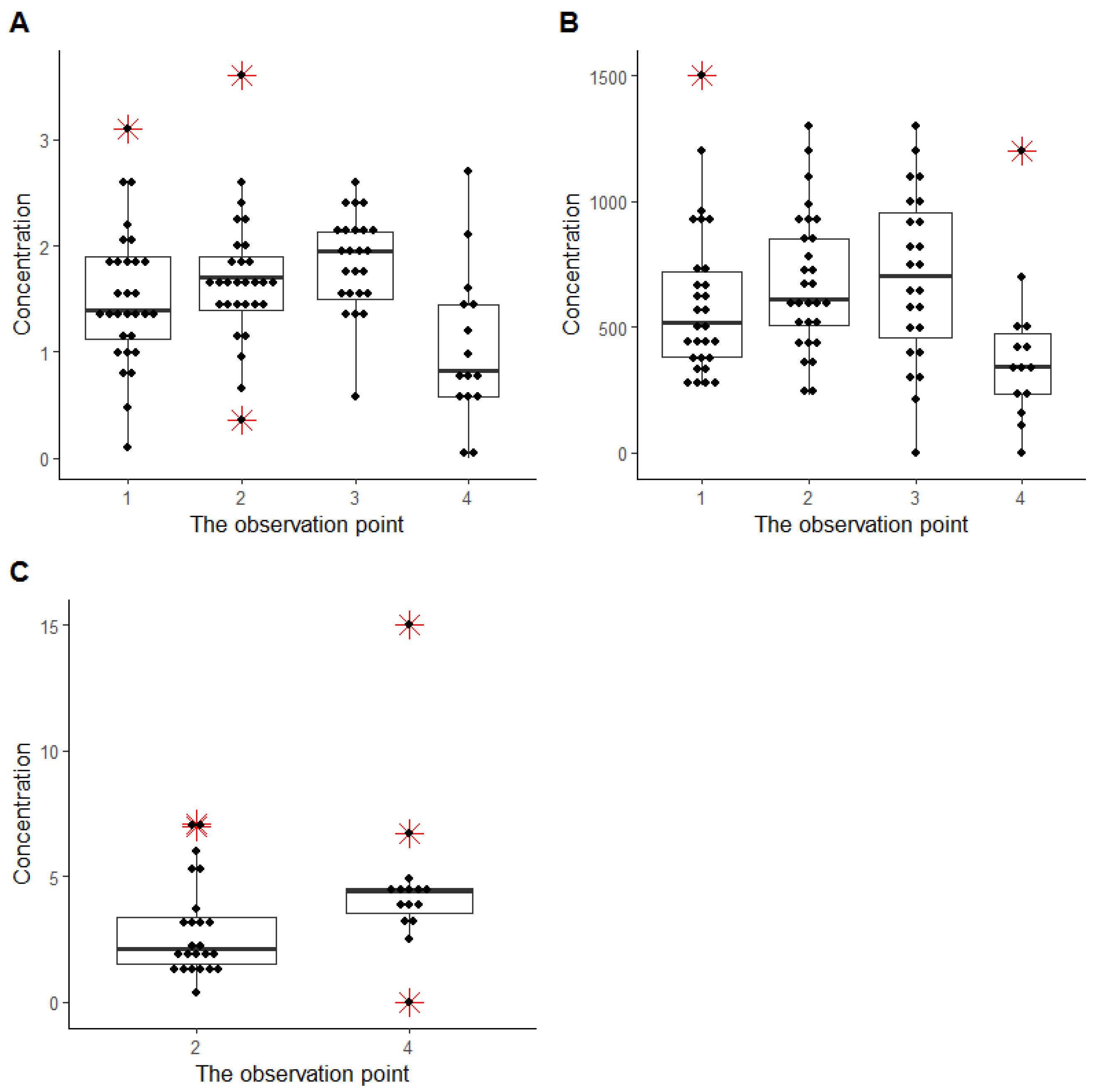
2.4. HPLC-MS/MS for Digoxin and Sotalol Measurement Optimization and Validation
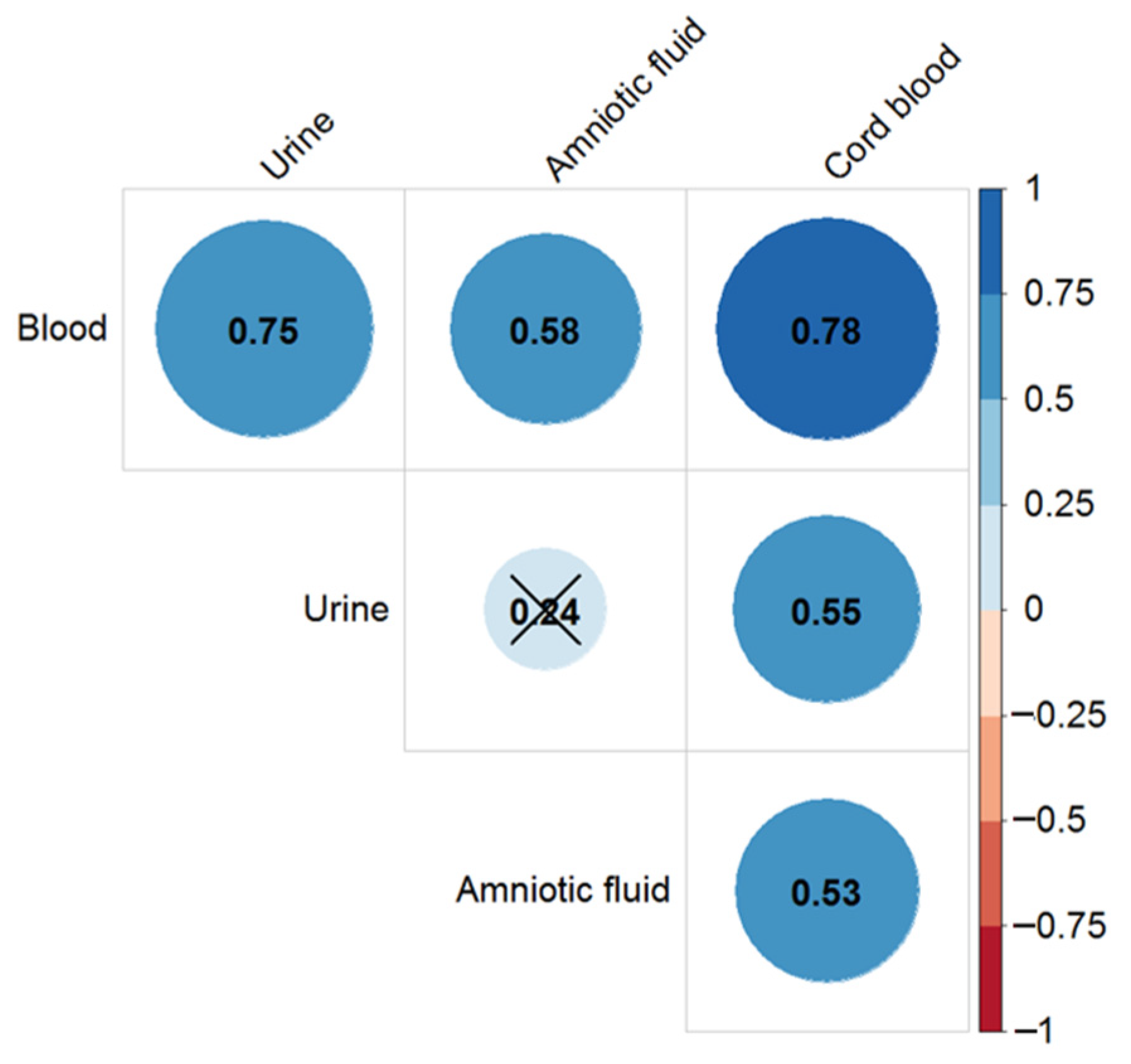
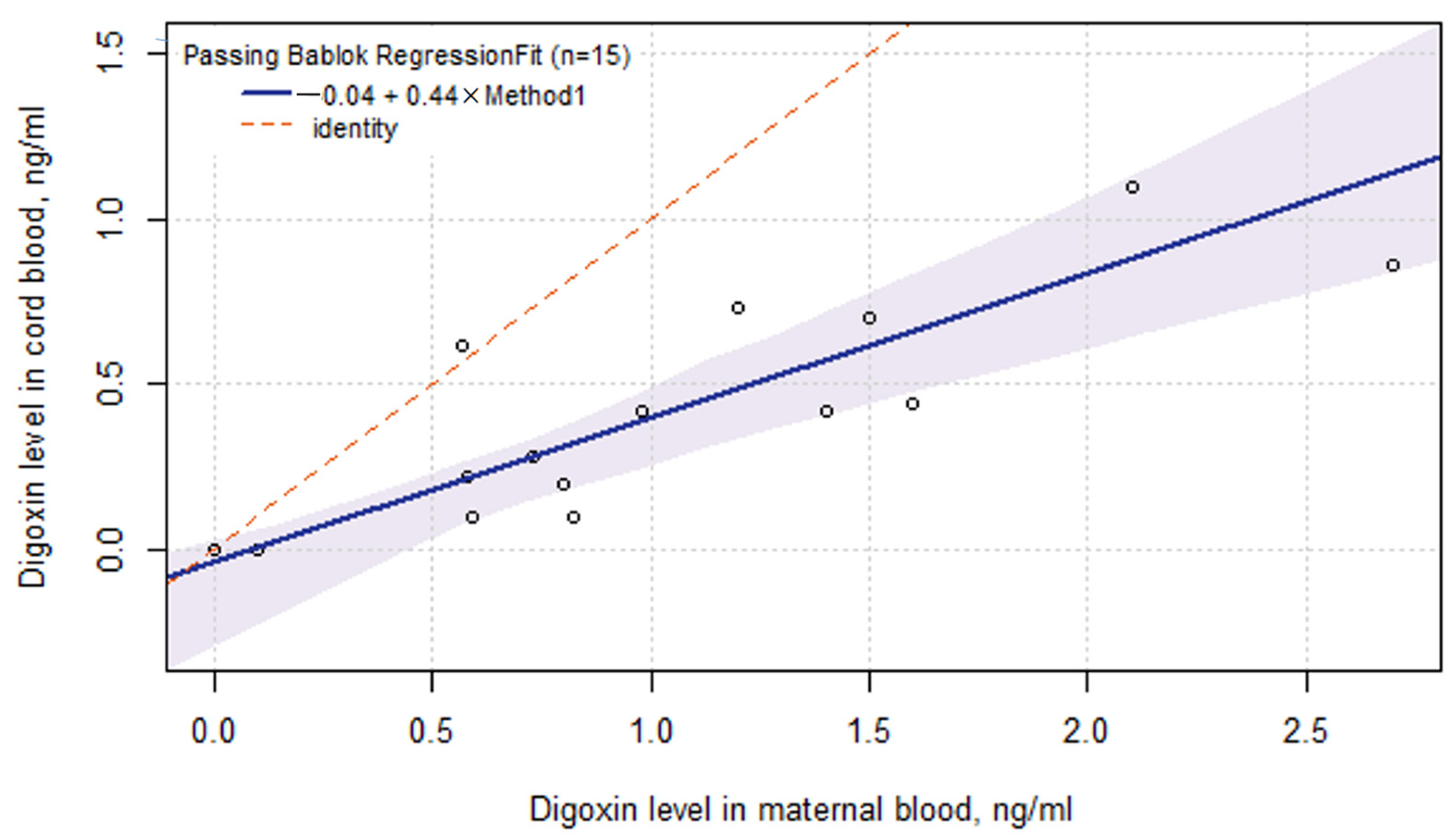
2.5. Digoxin Pharmacokinetics in ART
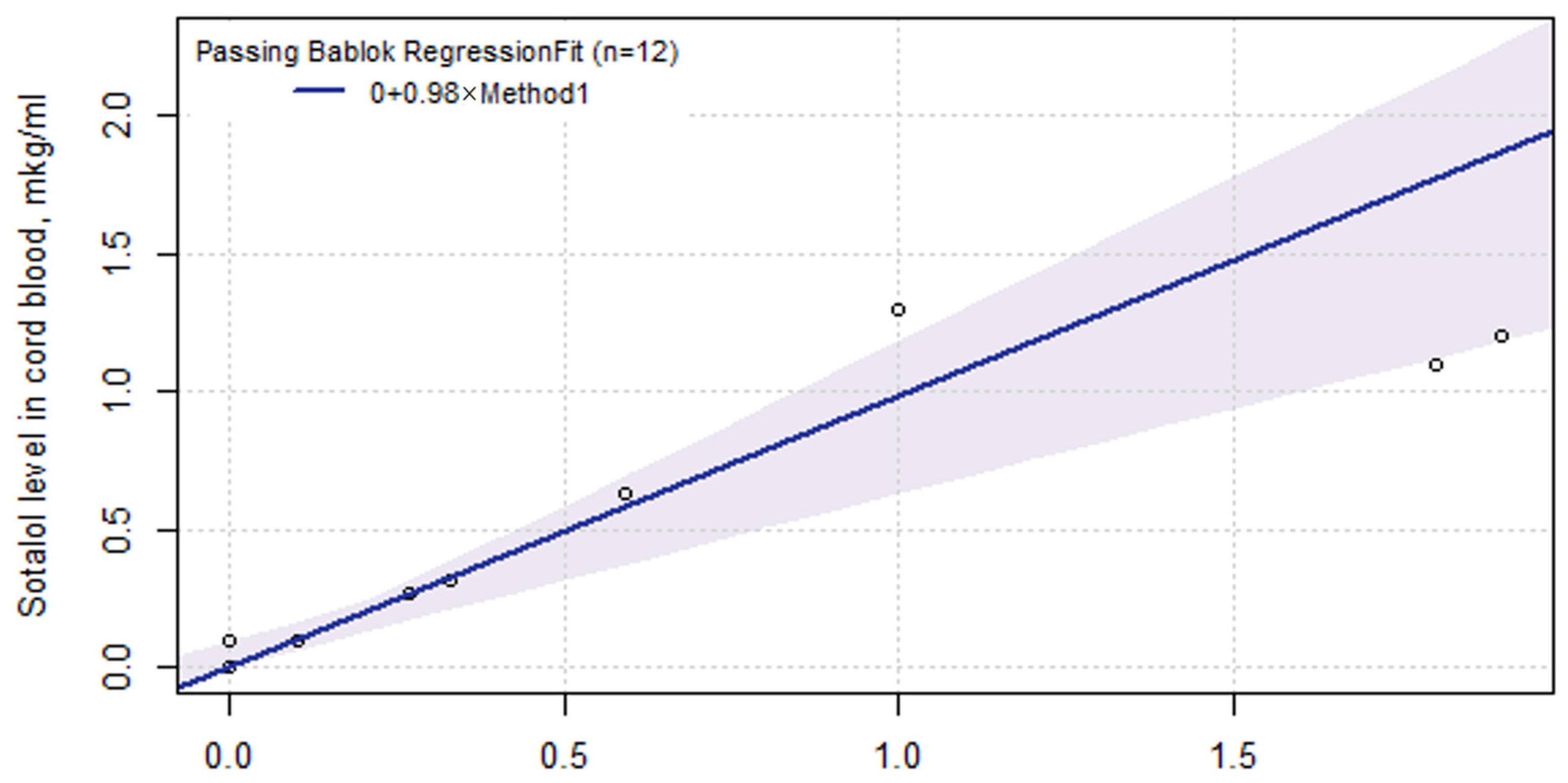
2.6. Sotalol Pharmacokinetics in ART
3. Discussion
4. Material and Methods
4.1. Study Design
4.2. Sample Collection
4.3. Sample Preparation for HPLC-MS/MS
4.4. HPLC-MS/MS Parameters
4.5. HPLC-MS/MS MRM Methods Validation
4.6. ABCB1 Gene Polymorphism Study
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wacker-Gussmann, A.; Strasburger, J.F.; Cuneo, B.F.; Wakai, R.T. Diagnosis and Treatment of Fetal Arrhythmia. Am. J. Perinatol. 2014, 31, 617–629. [Google Scholar] [CrossRef]
- Alsaied, T.; Baskar, S.; Fares, M.; Alahdab, F.; Czosek, R.J.; Murad, M.H.; Prokop, L.J.; Divanovic, A.A. First-Line Antiarrhythmic Transplacental Treatment for Fetal Tachyarrhythmia: A Systematic Review and Meta-Analysis. J. Am. Heart Assoc. 2017, 6, e007164. [Google Scholar] [CrossRef]
- Yuan, S. Fetal arrhythmias: Surveillance and management. Hell. J. Cardiol. 2019, 60, 72–81. [Google Scholar] [CrossRef]
- O’Leary, E.T.; Alexander, M.E.; Bezzerides, V.J.; Drogosz, M.; Economy, K.E.; Friedman, K.G.; Pickard, S.S.; Tworetzky, W.; Mah, D.Y. Low mortality in fetal supraventricular tachycardia: Outcomes in a 30-year single-institution experience. J. Cardiovasc. Electrophysiol. 2020, 31, 1105–1113. [Google Scholar] [CrossRef]
- Srinivasan, S.; Strasburger, J. Overview of fetal tachycardia: Outcomes in a 30-year single-institution experience. Curr. Opin. Pediatr. 2008, 20, 522–531. [Google Scholar] [CrossRef]
- van der Heijden, L.B.; Oudijk, M.A.; Manten, G.T.R.; ter Heide, H.; Pistorius, L.; Freund, M.W. Sotalol as first-line treatment for fetal tachycardia and neonatal follow-up. Ultrasound Obstet. Gynecol. 2013, 42, 285–293. [Google Scholar] [CrossRef]
- Shah, A.; Moon-Grady, A.; Bhogal, N.; Collins, K.K.; Tacy, T.; Brook, M.; Hornberger, L.K. Effectiveness of sotalol as first-line therapy for fetal supraventricular tachyarrhythmias. Am. J. Cardiol. 2012, 109, 1614–1618. [Google Scholar] [CrossRef] [PubMed]
- Strasburger, J.F. Re: Sotalol as first-line treatment for fetal tachycardia and neonatal follow-up. L. B. van der Heijden, M. A. Oudijk, G. Manten, H. ter Heide, L. Pistorius and M. W. Freund. Ultrasound Obstet Gynecol. 2013, 42, 285–293. Ultrasound Obstet. Gynecol. 2013, 42, 254–255. [Google Scholar] [CrossRef] [PubMed]
- Strizek, B.; Berg, C.; Gottschalk, I.; Herberg, U.; Geipel, A.; Gembruch, U. High-dose flecainide is the most effective treatment of fetal supraventricular tachycardia. Heart Rhythm 2016, 13, 1283–1288. [Google Scholar] [CrossRef]
- Fouron, J.-C.; Fournier, A.; Proulx, F.; Lamarche, J.; Bigras, J.L.; Boutin, C.; Brassard, M.; Gamache, S. Management of fetal tachyarrhythmia based on superior vena cava/aorta Doppler flow recordings. Heart 2003, 89, 1211–1216. [Google Scholar] [CrossRef]
- Yuan, S.M.; Xu, Z.Y. Fetal arrhythmias: Prenatal evaluation and intrauterine therapeutics. Ital. J. Pediatr. 2020, 46, 21. [Google Scholar] [CrossRef] [PubMed]
- Suarez, A.M.; Sanchez-Hernandez, J.G.; Barajas, F.M.; Pérez-Blanco, J.S.; Lanao, J.M.; Alvarez, L.G.-C.; Calvo, M.V. Pharmacokinetics and dosing requirements of digoxin in pregnant women treated for fetal supraventricular tachycardia. Expert Rev. Clin. Pharmacol. 2017, 10, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Hill, G.D.; Kovach, J.R.; Saudek, D.E.; Singh, A.K.; Wehrheim, K.; Frommelt, M.A. Transplacental treatment of fetal tachycardia: A systematic review and meta-analysis. Prenat. Diagn. 2017, 37, 1076–1083. [Google Scholar] [CrossRef] [PubMed]
- Hansmann, M.; Gembruch, U.; Bald, R.; Manz, M.; Redel, D.A. Fetal tachyarrhythmias: Transplacental and direct treatment of the fetus-a report of 60 cases. Ultrasound Obstet. Gynecol. 1991, 1, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Hebert, M.F.; Easterling, T.R.; Kirby, B.; Carr, D.B.; Buchanan, M.L.; Rutherford, T.; Thummel, K.E.; Fishbein, D.P.; Unadkat, J.D. Effects of Pregnancy on CYP3A and P-glycoprotein Activities as Measured by Disposition of Midazolam and Digoxin: A University of Washington Specialized Center of Research Study. Clin. Pharmacol. Ther. 2008, 84, 248–253. [Google Scholar] [CrossRef]
- Tanabe, M.; Ieiri, I.; Nagata, N.; Inoue, K.; Ito, S.; Kanamori, Y.; Takahashi, M.; Kurata, Y.; Kigawa, J.; Higuchi, S.; et al. Expression of P-glycoprotein in human placenta: Relation to genetic polymorphism of the multidrug resistance (MDR)-1 gene. J. Pharmacol. Exp. Ther. 2001, 297, 1137–1143. [Google Scholar]
- Sun, M.; Kingdom, J.; Baczyk, D.; Lye, S.J.; Matthews, S.G.; Gibb, W. Expression of the Multidrug Resistance P-Glycoprotein, (ABCB1 glycoprotein) in the Human Placenta Decreases with Advancing Gestation. Placenta 2006, 27, 602–609. [Google Scholar] [CrossRef]
- Han, L.W.; Gao, C.; Mao, Q. An update on expression and function of P-gp/ABCB1 and BCRP/ABCG2 in the placenta and fetus. Expert Opin. Drug Metab. Toxicol. 2018, 14, 817–829. [Google Scholar] [CrossRef]
- Ho, G.T.; Moodie, F.M.; Satsangi, J. Multidrug resistance 1 gene (P-glycoprotein 170): An important determinant in gastrointestinal disease? Gut 2003, 52, 759–766. [Google Scholar] [CrossRef]
- Hoffmeyer, S.; Burk, O.; von Richter, O.; Arnold, H.P.; Brockmöller, J.; Johne, A.; Cascorbi, I.; Gerloff, T.; Roots, I.; Eichelbaum, M.; et al. Functional polymorphisms of the human multidrug-resistance gene: Multiple sequence variations and correlation of one allele with P-glycoprotein expression and activity in vivo. Proc. Natl. Acad. Sci. USA 2000, 97, 3473–3478. [Google Scholar] [CrossRef]
- Gaikovitch, E.A.; Cascorbi, I.; Mrozikiewicz, P.M.; Brockmöller, J.; Frötschl, R.; Köpke, K.; Gerloff, T.; Chernov, J.N.; Roots, I. Polymorphisms of drug-metabolizing enzymes CYP2C9, CYP2C19, CYP2D6, CYP1A1, NAT2 and of P-glycoprotein in a Russian population. Eur. J. Clin. Pharmacol. 2003, 59, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Sychev, D.A.; Shuev, G.N.; Suleymanov, S.S.; Ryzhikova KA Mirzaev, K.B.; Grishina, E.A.; Snalina, N.E.; Sozaeva, Z.A.; Grabuzdov, A.M.; Matsneva, I. A Comparison of CYP2C9, CYP2C19, CYP2D6, frequency in Russian and Nanai populations. Pharmgenomics. Pers. Med. 2017, 10, 93–99. [Google Scholar]
- Chimenea, Á.; García-Díaz, L.; Méndez, A.; Antiñolo, G. Maternal effects induced by oral digoxin during treatment of fetal tachyarrhythmia: Case series and literature review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 256, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.F.; Butler, J.; Patterson, J.H.; Gattis Stough, W.; Bauman, J.L.; van Veldhuisen, D.J.; Schwartz, T.A.; Sabbah, H.; Mackowiak, J.I.; Ventura, H.O.; et al. Dose response characterization of the association of serum digoxin concentration with mortality outcomes in the Digitalis Investigation Group trial. Eur. J. Heart Fail. 2016, 18, 1072–1081. [Google Scholar] [CrossRef]
- Miyoshi, T.; Maeno, Y.; Hamasaki, T.; Inamura, N.; Yasukochi, S.; Kawataki, M.; Horigome, H.; Yoda, H.; Taketazu, M.; Nii, M.; et al. Antenatal Therapy for Fetal Supraventricular Tachyarrhythmias: Multicenter Trial. J. Am. Coll. Cardiol. 2019, 74, 874–885. [Google Scholar] [CrossRef] [PubMed]
- Ebara, H.; Suzuki, S.; Nagashima, K.; Koizumi, T.; Nishida, A.; Kanbe, Y.; Kuroume, T. Digoxin-and digitoxin-like immunoreactive substances in amniotic fluid, cord blood, and serum of neonates. Pediatr. Res. 1986, 20, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Oudijk, M.A.; Ruskamp, J.M.; Ververs, F.; Ambachtsheer, E.; Stoutenbeek, P.; Visser, G.H.; Meijboom, E.J. Treatment of fetal tachycardia with sotalol: Transplacental pharmacokinetics and pharmacodynamics. J. Am. Coll. Cardiol. 2003, 42, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Pashazadeh-Panahi, P.; Hasanzadeh, M. Digoxin as a glycosylated steroid-like therapeutic drug: Recent advances in the clinical pharmacology and bioassays of pharmaceutical compounds. Biomed. Pharmacother. 2020, 123, 109813. [Google Scholar] [CrossRef]
- Simonson, P.D.; Kim, K.H.; Winston-McPherson, G.; Parakh, R.S.; Yamaguchi, D.; Merrill, A.E.; Dickerson, J.A.; Greene, D.N. Characterization of bilirubin interference in three commonly used digoxin assays. Clin. Biochem. 2019, 63, 102–105. [Google Scholar] [CrossRef]
- Dasgupta, A. Therapeutic drug monitoring of digoxin: Impact of endogenous and exogenous digoxin-like immunoreactive substances. Toxicol. Rev. 2006, 25, 273–281. [Google Scholar] [CrossRef]
- Dasgupta, A. Herbal supplements and therapeutic drug monitoring: Focus on digoxin immunoassays and interactions with St. John’s wort. Ther. Drug Monit. 2008, 30, 212–217. [Google Scholar] [CrossRef]
- Dasgupta, A.; Wu, S.; Actor, J.; Olsen, M.; Wells, A.; Datta, P. Effect of Asian and Siberian ginseng on serum digoxin measurement by five digoxin immunoassays. Significant variation in digoxin-like immunoreactivity among commercial ginsengs. Am. J. Clin. Pathol. 2003, 119, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, N.R.; Ramesh, M. Digoxin—A therapeutic agent and mechanistic probe: Review of liquid chromatographic mass spectrometric methods and recent nuances in the clinical pharmacology attributes of digoxin. Bioanalysis 2009, 1, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Josephs, R.D.; Daireaux, A.; Westwood, S.; Wielgosz, R.I. Simultaneous determination of various cardiac glycosides by liquid chromatography–hybrid mass spectrometry for the purity assessment of the therapeutic monitored drug digoxin. J. Chromatogr. A 2010, 1217, 4535–4543. [Google Scholar] [CrossRef]
- Mitamura, K.; Horikawa, A.; Yamane, Y.; Ikeda, Y.; Fujii, Y.; Shimada, K. Determination of digoxin in human serum using stable isotope dilution liquid chromatography/electrospray ionization-tandem mass spectrometry. Biol. Pharm. Bull. 2007, 30, 1653–1656. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Shibakawa, K.; Nakade, S.; Miyata, Y.J. Validation and application of a 96-well format solid-phase extraction and liquid chromatography–tandem mass spectrometry method for the quantitation of digoxin in human plasma. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2008, 869, 126–132. [Google Scholar] [CrossRef]
- Li, S.; Liu, G.; Jia, J.; Miao, Y.; Gu, S.; Miao, P.; Shi, X.; Wang, Y.; Yu, C. Therapeutic monitoring of serum digoxin for patients with heart failure using a rapid LC-MS/MS method. Clin. Biochem. 2010, 43, 307–313. [Google Scholar] [CrossRef]
- Tracqui, A.; Kintz, P.; Ludes, B.; Mangin, P. High-performance liquid chromatography–ionspray mass spectrometry for the specific determination of digoxin and some related cardiac glycosides in human plasma. J. Chromatogr. B Biomed. Sci. Appl. 1997, 692, 101–109. [Google Scholar] [CrossRef]
- Ballotari, M.; Taus, F.; Tolle, G.; Danese, E.; Dorizzi, R.M.; Tagliaro, F.; Gottardo, R. Development of a new ultra-high-performance liquid chromatography–tandem mass spectrometry method for the determination of digoxin and digitoxin in plasma: Comparison with a clinical immunoassay. Electrophoresis 2022, 43, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Cragan, J.D.; Friedman, J.M.; Holmes, L.B.; Uhl, K.; Green, N.S.; Riley, L. Ensuring the Safe and Effective use of Medications During Pregnancy: Planning and Prevention Through Preconception Care. Matern. Child Health J. 2006, 10 (Suppl. 1), 129–135. [Google Scholar] [CrossRef]
- Feghali, M.; Venkataramanan, R.; Caritis, S. Pharmacokinetics of drugs in pregnancy. Semin. Perinatol. 2015, 39, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Vahakangas, K.; Myllynen, P. Drug transporters in the human blood-placental barrier. Br. J. Pharmacol. 2009, 158, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Brice-Bennett, S.; D’Souza, S.W. Antiepileptic medication during pregnancy: Does fetal genotype affect outcome? Pediatr. Res. 2007, 62, 120–127. [Google Scholar] [CrossRef]
- Lankas, G.R.; Wise, L.D.; Cartwright, M.E. Placental P-glycoprotein deficiency enhances susceptibility to chemically induced birth defects in mice. Reprod. Toxicol. 1998, 12, 457–463. [Google Scholar] [CrossRef]
- Lankas, G.R.; Wise, L.; E Cartwright, M.; Pippert, T.; Umbenhauer, D.R. Determination of Digoxin in the Blood of Pregnant Women, Fetuses and Neonates before and during Anti-arrhythmic Therapy, Using Four Immunochemical Methods. Clin. Chem. Lab. Med. 1991, 29, 57–66. [Google Scholar] [CrossRef]
- Fromm, M.F.; Kim, R.B.; Stein, C.M.; Wilkinson, G.R.; Roden, D.M. Inhibition of P-Glycoprotein-mediated Drug Transport. Unifying mechanism to Explain the interaction Between Digoxin and Quinidine. Circulation 1999, 99, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.-S.; Lee, M.-H. Overview of Therapeutic Drug Monitoring. Korean J. Intern. Med. 2009, 24, 1–10. [Google Scholar] [CrossRef]
- Moatassim, S.; Touleimat, S.; Hazelzet, T.; Brasseur, M.D.; Diguet, A.; Durand, I.; Verspyck, E. Maternal complications induced by digoxin treatment of fetal tachycardia: A retrospective series of 18 cases. J. Gynecol. Obstet. Hum. Reprod. 2018, 47, 35–38. [Google Scholar] [CrossRef]
- Verstuyft, C.; Schwab, M.; Schaeffeler, E.; Kerb, R.; Brinkmann, U.; Jaillon, P.; Funck-Brentano, C.; Becquemont, L. Digoxin pharmacokinetics and MDR1 genetic polymorphisms. Eur. J. Clin. Pharmacol. 2003, 58, 809–812. [Google Scholar] [CrossRef]
- Sae-Lim, O.; Doungngern, T.; Jaisue, S.; Cheewatanakornkul, S.; Arunmanakul, P.; Anutrakulchai, S.; Kanjanavanit, R.; Wongpoowarak, W. Prediction of serum digoxin concentration using estimated glomerular filtration rate in Thai population. Int. J. Gen. Med. 2019, 12, 455–463. [Google Scholar] [CrossRef]
- Bochud, M.; Eap, C.B.; Maillard, M.; Johnson, T.; Vollenweider, P.; Bovet, P.; Elston, R.C.; Bergmann, S.; Beckmann, J.S.; Waterworth, D.M.; et al. Association of ABCB1genetic variants with renal function in Africans and in Caucasians. BMC Med. Genom. 2008, 1, 21. [Google Scholar] [CrossRef] [PubMed]
- Donofrio, M.T.; Moon-Grady, A.J.; Hornberger, L.K.; Copel, J.A.; Sklansky, M.S.; Abuhamad, A.; Cuneo, B.F.; Huhta, J.C.; Jonas, R.A.; Krishnan, A.; et al. Diagnosis and treatment of fetal cardiac disease: A scientific statement from the american heart association. Circulation 2014, 129, 2183–2242. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, J.S. Fetal dysrhythmias. In Best Practice and Research: Clinical Obstetrics and Gynaecology; Bailliere Tindall Ltd.: London, UK, 2019; Volume 58, pp. 28–41. [Google Scholar]
- Qin, J.; Deng, Z.; Tang, C.; Zhang, Y.; Hu, R.; Li, J.; Hua, Y.; Li, Y. Efficacy and Safety of Various First-Line Therapeutic Strategies for Fetal Tachycardias: A Network Meta-Analysis and Systematic Review. Front. Pharmacol. 2022, 13, 935455. [Google Scholar] [CrossRef] [PubMed]
- Jaeggi, E.; Öhman, A. Fetal and Neonatal Arrhythmias. Clin. Perinatol. 2016, 43, 99–112. [Google Scholar] [CrossRef]
- Bravo-Valenzuela, N.J.; Rocha, L.A.; Nardozza, L.M.M.; Júnior, E.A. Fetal cardiac arrhythmias: Current evidence. Ann. Pediatr. Cardiol. 2018, 11, 148–163. [Google Scholar]
- Jaeggi, E.T.; Carvalho, J.S.; De Groot, E.; Api, O.; Clur, S.-A.B.; Rammeloo, L.; McCrindle, B.W.; Ryan, G.; Manlhiot, C.; Blom, N.A. Comparison of transplacental treatment of fetal supraventricular tachyarrhythmias with digoxin, flecainide, and sotalol: Results of a nonrandomized multicenter study. Circulation 2011, 124, 1747–1754. [Google Scholar] [CrossRef]
- Bonnin, A.; Tassin, M.; Vauloup-Fellous, C.; Letamendia, E.; Stos, B.; Bonnet, D.; Gajdos, V.; Mabille, M.; Benachi, A. Case of a healthy infant born following antenatal enterovirus myocarditis and hydrops. J. Clin. Virol. 2014, 61, 459–462. [Google Scholar] [CrossRef]
- Takahashi, H.; Tsukamoto, K.; Takahashi, S.; Nakamura, T.; Ito, Y.; Kaneko, M.; Sago, H. Reversible Atrioventricular Block and Junctional Ectopic Tachycardia in Coxsackievirus B3-Induced Fetal–Neonatal Myocarditis without Left Ventricular Dysfunction. Am. J. Perinatol. Rep. 2011, 1, 37–42. [Google Scholar] [CrossRef][Green Version]
- Chan, S.H.-S.; Lun, K. Ventricular aneurysm complicating neonatal Coxsackie B4 myocarditis. Pediatr. Cardiol. 2001, 22, 247–249. [Google Scholar] [CrossRef]
- Savarese, I.; De Carolis, M.P.; Costa, S.; De Rosa, G.; De Carolis, S.; Lacerenza, S.; Romagnoli, C. Atypical manifestations of congenital parvovirus B19 infection. Eur. J. Pediatr. 2008, 167, 1463–1466. [Google Scholar] [CrossRef]
- DeJong, S.; Salmanian, B.; A Shamshirsaz, A.; Ruano, R. Perinatal management of fetal supraventricular tachycardia complicated by maternal pertussis. BMJ Case Rep. 2015, 2015, 2–4. [Google Scholar] [CrossRef]
- Bockerija, E.L. Perinatal cardiology: The present and the future. Part II: Cardiac arrhythmias and conduction. Ross. Vestn. Perinatol. Pediatr. 2019, 64, 6–10. [Google Scholar] [CrossRef]
- Maeno, Y.; Hirose, A.; Kanbe, T.; Hori, D. Fetal arrhythmia: Prenatal diagnosis and perinatal management. J. Obstet. Gynaecol. Res. 2009, 35, 623–629. [Google Scholar] [CrossRef]
- Juares-Olguin, H.; Buendia-Soto, E.; Lares-Asseff, I. Pharmacology for the fetus and the newborn. Gac. Med. Mex. 2015, 151, 361–368. [Google Scholar]
- Kirby, B.J.; Kalhorn, T.; Hebert, M.; Easterling, T.; Unadkat, J.D. Sensitive and specific LC-MS assay for quantification of digoxin in human plasma and urine. Biomed. Chromatogr. 2008, 22, 712–718. [Google Scholar] [CrossRef]
- Keane, S.; Wallace, G.; Munday, C.; Wright, M. Sensitive and robust LC-MS/MS analysis of digoxin in human plasma through optimization of in-source adduct formation. Bioanalysis 2018, 10, 1401–1411. [Google Scholar] [CrossRef]
- Bylda, C.; Thiele, R.; Kobold, U.; Volmer, D.A. Rapid quantification of digitoxin and its metabolites using differential ion mobility spectrometry-tandem mass spectrometry. Anal. Chem. 2015, 87, 2121–2128. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, Y.; Zhou, Q.; Yu, Y.; Chen, L.; Zheng, J. A sensitive method for digoxin determination using formate-adduct ion based on the effect of ionization enhancement in liquid chromatograph-mass spectrometer. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015, 978–979, 138–144. [Google Scholar] [CrossRef]
- Salvador, A.; Sagan, C.; Denouel, J. Rapid Quantitation of Digoxin in Human Plasma and Urine Using Isotope Dilution Liquid Chromatography-Tandem Mass Spectrometry. J. Liq. Chromatogr. Relat. Technol. 2006, 29, 1917–1932. [Google Scholar] [CrossRef]
- Carlier, J.; Guitton, J.; Romeuf, L.; Bévalot, F.; Boyer, B.; Fanton, L.; Gaillard, Y. Screening approach by ultra-high performance liquid chromatography–tandem mass spectrometry for the blood quantification of thirty-four toxic principles of plant origin. Application to forensic toxicology. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015, 975, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Øiestad, E.L.; Johansen, U.; Opdal, M.S.; Bergan, S.; Christophersen, A.S. Determination of digoxin and digitoxin in whole blood. J. Anal. Toxicol. 2009, 33, 372–378. [Google Scholar] [CrossRef] [PubMed]
- O’Hare, M.F.; Murnaghan, G.A.; Russell, C.J.; Leahey, W.J.; Varma, M.P.S.; McDevitt, D.G. Sotalol As a Hypotensive Agent in Pregnancy. BJOG Int. J. Obstet. Gynaecol. 1980, 87, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Hitzl, M.; Schaeffeler, E.; Hocher, B.; Slowinski, T.; Halle, H.; Eichelbaum, M.; Kaufmann, P.; Fritz, P.; Fromm, M.F.; Schwab, M. Variable expression of P-glycoprotein in the human placenta and its association with mutations of the multidrug resistance 1 gene (MDR1, ABCB1). Pharmacogenetics 2004, 14, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Le, J. Overview of Pharmacokinetics; Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California: San Diego, CA, USA, 2022. [Google Scholar]
- Wessler, J.D.; Grip, L.T.; Mendell, J.; Giugliano, R.P. The P-Glycoprotein Transport System and Cardiovascular Drugs. J. Am. Coll. Cardiol. 2013, 61, 2495–2502. [Google Scholar] [CrossRef]
- Pauwels, S.; Allegaert, K. Therapeutic drug monitoring in neonates. Arch. Dis. Child. 2016, 101, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Schinkel, A.; Mol, C.; Wagenaar, E.; van Deemter, L.; Smit, J.; Borst, P. Multidrug resistance and the role of P-glycoprotein knockout mice. Eur. J. Cancer 1995, 31, 1295e8. [Google Scholar] [CrossRef] [PubMed]
- M10 Bioanalytical Method Validation and Study Sample Analysis; International Council for Harmonisation; Guidance for Industry; Availability//Food and Drug Administration, HHS, Current Step 5, Version 2022. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/ich-guideline-m10-bioanalytical-method-validation-step-5_en.pdf (accessed on 25 July 2022).
- Lyon, E. Mutation Detection Using Fluorescent Hybridization Probes and Melting Curve Analysis. Exp. Rev. Mol. Diagn. 2001, 1, 92–101. [Google Scholar] [CrossRef]


| Fetal Arrhythmia Group (n = 89) | Control Group (n = 50) | p-Value | |
|---|---|---|---|
| Age (year) | 31 (28–34) | 31 (27–34) | 0.31 |
| Growth (m) | 1.65 (1.62–1.68) | 1.64 (1.60–1.69) | 0.79 |
| Weight (kg) | 71 (64–80) | 70 (64–77) | 0.75 |
| Pregnancy weight gain (kg) | 11 (8–13) | 12 (10–15) | 0.19 |
| Parity | 2 (1–3) | 2 (1–2) | 0.99 |
| Chronic diseases | 77 (86.5) | 25 (50) | <0.001 |
| Toxicosis | 19 (21.4) | 10 (20) | 0.85 |
| Early pregnancy termination threat | 12 (13.5) | 8 (16) | 0.69 |
| Threat of preterm delivery in the third trimester | 35 (39.3) | 1 (2) | <0.001 |
| Gestation age at delivery (weeks) | 386 (373–392) | 390 (384–400) | 0.015 |
| Newborn weight (g) | 3240 (2860–3600) | 3346 (3020–3565) | 0.33 |
| SVT (n = 49, 55.1%) | PAC (n = 26, 29.2%) | AFL (n = 14, 15.7%) | p-Value | |
|---|---|---|---|---|
| Sustained FA (n = 28) | 19 (38.8) | 0 | 9 (64.3) | <0.001 |
| Nonimmune fetal hydrops (n = 10) | 9 (18.4) | 0 | 1 (7.1) | 0.049 |
| Ascites (n = 12) | 11 (22.5) | 0 | 1 (7.1) | 0.018 |
| Fetal ART (n = 64) | 38 (77.6) | 12 (47.2) | 14 (100) | <0.001 |
| Dose reduction in digoxin (n = 36) | 25 (65.8) | 3 (25) | 8 (57.1) | 0.046 |
| Sotalol (n = 43) | 32 (84.2) | 3 (25) | 8 (57.1) | <0.001 |
| Dose reduction in sotalol (n = 30) | 23 (60.5) | 1 (8.3) | 6 (42.9) | 0.004 |
| Rhythm recovery episodes (n = 57) | 34 (91.9) | 11 (91.7) | 12 (85.7) | 0.838 |
| Complete rhythm recovery (n = 50) | 31 (63.3) | 9 (34.6) | 10 (71.4) | 0.027 |
| Rhythm recovery time | 5 (2–8) | 2 (1–6) | 6 (1–6) | 0.486 |
| Absence of rhythm disruption on ATR | 23 (60.5) | 10 (83.3) | 9 (64.3) | 0.304 |
| Disruption of the rhythm on a drug dosage decrease | 13 (34.2) | 0 | 1 (7.14) | 0.015 |
| Gestation age at delivery | 38.5 (37.32–39.1) | 39.05 (38.42–39.62) | 38.2 (37.62–39) | 0.084 |
| Q1, Da | Q3, Da | Time, ms | ID | DP, V | EP, V | CE, V | CXP, V |
|---|---|---|---|---|---|---|---|
| 273.0 | 255.0 | 120.0 | Sotalol1 | 41.0 | 10.0 | 17.0 | 22.0 |
| 273.0 | 213.000 | 120.0 | Sotalol2 | 41.0 | 10.0 | 25.0 | 22.0 |
| 273.0 | 133.100 | 120.0 | Sotalol3 | 41.0 | 10.0 | 35.0 | 14.0 |
| 267.1 | 145.1 | 120.0 | Atenolol1(IS) | 136.0 | 10.0 | 35.0 | 12.0 |
| 267.1 | 190.000 | 120.0 | Atenolol2(IS) | 136.0 | 10.0 | 25.0 | 16.0 |
| 267.1 | 74.100 | 120.0 | Atenolol3(IS) | 136.0 | 10.0 | 29.0 | 8.0 |
| 798.4 | 651.4 | 80.0 | Digoxin1 | 41.0 | 10.0 | 19.0 | 12.0 |
| 798.4 | 781.5 | 80.0 | Digoxin2 | 41.0 | 10.0 | 13.0 | 16.0 |
| 798.4 | 97.1 | 80.0 | Digoxin3 | 41.0 | 10.0 | 51.0 | 8.0 |
| 782.3 | 97.1 | 80.0 | Digytoxin1(IS) | 76.0 | 10.0 | 33.0 | 12.0 |
| 782.3 | 635.5 | 80.0 | Digytoxin2(IS) | 76.0 | 10.0 | 17.0 | 20.0 |
| 782.3 | 243.1 | 80.0 | Digytoxin3(IS) | 76.0 | 10.0 | 21.0 | 22.0 |
| Maternal Blood (ng/mL) | Fetus Blood (ng/mL) | |
|---|---|---|
| First rhythm recovery episode | 1.4 (1.1, 1.9) | 0.58 (0.46, 0.80) |
| Delivery day | 0.82 (0.58, 1.5) | 0.42 (0.20, 0.66) |
| Newborn 3435C > T ABCB1 Genotype | CC | TC and TT | p-Value |
|---|---|---|---|
| Pregnant women blood | 0.34 (0.075, 1.1) | 0.98 (0.76, 1.40) | 0.1773 |
| Cord blood | 0.31 (0, 0.68) | 0.42 (0.21, 0.57) | 0.6468 |
| Amniotic fluid | 2.9 (1.9, 3.5) | 4.5 (4.2, 4.8) | 0.01076 * |
| Pregnant Women Venous Blood | Pregnant Women Urine | Amniotic Fluid | Cord Blood | |
|---|---|---|---|---|
| First observation point | + | + | - | - |
| Second observation point | + | + | + | - |
| Third observation point | + | + | - | - |
| Delivery | + (with continued therapy) | + (with continued therapy) | + | + |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Starodubtseva, N.; Kindysheva, S.; Potapova, A.; Kukaev, E.; Khodzhaeva, Z.; Bockeria, E.; Chagovets, V.; Frankevich, V.; Sukhikh, G. Transplacental Therapeutic Drug Monitoring in Pregnant Women with Fetal Tachyarrhythmia Using HPLC-MS/MS. Int. J. Mol. Sci. 2023, 24, 1848. https://doi.org/10.3390/ijms24031848
Starodubtseva N, Kindysheva S, Potapova A, Kukaev E, Khodzhaeva Z, Bockeria E, Chagovets V, Frankevich V, Sukhikh G. Transplacental Therapeutic Drug Monitoring in Pregnant Women with Fetal Tachyarrhythmia Using HPLC-MS/MS. International Journal of Molecular Sciences. 2023; 24(3):1848. https://doi.org/10.3390/ijms24031848
Chicago/Turabian StyleStarodubtseva, Natalia, Svetlana Kindysheva, Alyona Potapova, Evgenii Kukaev, Zulfiya Khodzhaeva, Ekaterina Bockeria, Vitaliy Chagovets, Vladimir Frankevich, and Gennady Sukhikh. 2023. "Transplacental Therapeutic Drug Monitoring in Pregnant Women with Fetal Tachyarrhythmia Using HPLC-MS/MS" International Journal of Molecular Sciences 24, no. 3: 1848. https://doi.org/10.3390/ijms24031848
APA StyleStarodubtseva, N., Kindysheva, S., Potapova, A., Kukaev, E., Khodzhaeva, Z., Bockeria, E., Chagovets, V., Frankevich, V., & Sukhikh, G. (2023). Transplacental Therapeutic Drug Monitoring in Pregnant Women with Fetal Tachyarrhythmia Using HPLC-MS/MS. International Journal of Molecular Sciences, 24(3), 1848. https://doi.org/10.3390/ijms24031848







