Characterization and Preliminary Application of a Novel Lytic Vibrio parahaemolyticus Bacteriophage vB_VpaP_SJSY21
Abstract
:1. Introduction
2. Results
2.1. Isolation, Purification, and General Characterization of Phages
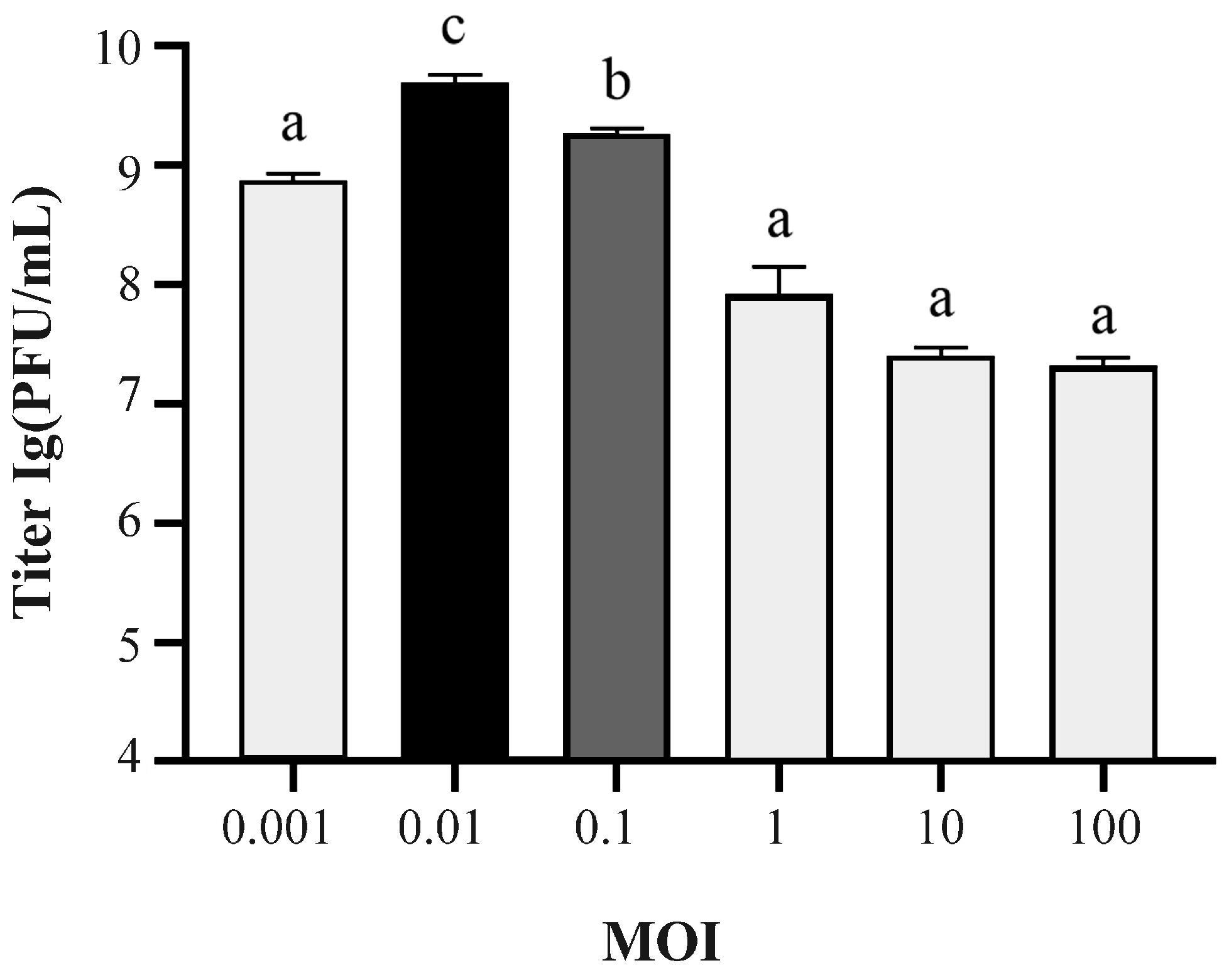
2.2. MOI and One-Step Growth
2.3. Results of Lysis and Temperature Stability Assay
2.4. Characteristics of Phage Genome
2.5. Protein Function Prediction of Bacteriophages
2.6. Analysis of α Diversity and β Diversity
2.7. Analysis of Community Composition Structure and Differential Species
3. Discussion
4. Materials and Methods
4.1. Isolation, Identification, and Purification of Phages
4.2. Morphological Characterization
4.3. Determination of Phage Titer
4.4. Determination of Multiplicity of Infection (MOI) and One-Step Growth
4.5. Lysis Assay and Temperature Stability Assay
4.6. Whole-Genome Sequencing and Analysis
4.7. Experimental Animals and Immune Stimulation
4.8. Extraction and Sequencing of Gut Microbiota Genome
4.9. Analysis of Gut Microbiota Community Structure of L. vannamei
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Panini, R.L.; Freitas, L.E.L.; Guimarães, A.M.; Rios, C.; da Silva, M.F.O.; Vieira, F.N.; Fracalossi, D.M.; Samuels, R.I.; Prudêncio, E.S.; Silva, C.P.; et al. Potential Use of Mealworms as an Alternative Protein Source for Pacific White Shrimp: Digestibility and Performance. Aquaculture 2017, 473, 115–120. [Google Scholar] [CrossRef]
- Han, J.E.; Mohney, L.L.; Tang, K.F.J.; Pantoja, C.R.; Lightner, D.V. Plasmid Mediated Tetracycline Resistance of Vibrio Parahaemolyticus Associated with Acute Hepatopancreatic Necrosis Disease (AHPND) in Shrimps. Aquac. Rep. 2015, 2, 17–21. [Google Scholar] [CrossRef]
- Han, J.E.; Tang, K.F.J.; Tran, L.H.; Lightner, D.V. Photorhabdus Insect-Related (Pir) Toxin-like Genes in a Plasmid of Vibrio Parahaemolyticus, the Causative Agent of Acute Hepatopancreatic Necrosis Disease (AHPND) of Shrimp. Dis. Aquat. Org. 2015, 113, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Nunan, L.; Lightner, D.; Pantoja, C.; Gomez-Jimenez, S. Detection of Acute Hepatopancreatic Necrosis Disease (AHPND) in Mexico. Dis. Aquat. Org. 2014, 111, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Roy, S.; Behera, B.K.; Bossier, P.; Das, B.K. Acute Hepatopancreatic Necrosis Disease (Ahpnd): Virulence, Pathogenesis and Mitigation Strategies in Shrimp Aquaculture. Toxins 2021, 13, 524. [Google Scholar] [CrossRef] [PubMed]
- Santos, H.M.; Tsai, C.Y.; Maquiling, K.R.A.; Tayo, L.L.; Mariatulqabtiah, A.R.; Lee, C.W.; Chuang, K.P. Diagnosis and Potential Treatments for Acute Hepatopancreatic Necrosis Disease (AHPND): A Review. Aquac. Int. 2020, 28, 169–185. [Google Scholar] [CrossRef] [PubMed]
- Flegel, T.W. Historic Emergence, Impact and Current Status of Shrimp Pathogens in Asia. J. Invertebr. Pathol. 2012, 110, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Kongrueng, J.; Yingkajorn, M.; Bunpa, S.; Sermwittayawong, N.; Singkhamanan, K.; Vuddhakul, V. Characterization of Vibrio Parahaemolyticus Causing Acute Hepatopancreatic Necrosis Disease in Southern Thailand. J. Fish Dis. 2015, 38, 957–966. [Google Scholar] [CrossRef]
- Dong, X.; Bi, D.; Wang, H.; Zou, P.; Xie, G.; Wan, X.; Yang, Q.; Zhu, Y.; Chen, M.; Guo, C.; et al. PirABvp-Bearing Vibrio Parahaemolyticus and Vibrio Campbellii Pathogens Isolated from the Same AHPND-Affected Pond Possess Highly Similar Pathogenic Plasmids. Front. Microbiol. 2017, 8, 1859. [Google Scholar] [CrossRef]
- Pinoargote, G.; Flores, G.; Cooper, K.; Ravishankar, S. Effects on Survival and Bacterial Community Composition of the Aquaculture Water and Gastrointestinal Tract of Shrimp (Litopenaeus vannamei) Exposed to Probiotic Treatments after an Induced Infection of Acute Hepatopancreatic Necrosis Disease. Aquac. Res. 2018, 49, 3270–3288. [Google Scholar] [CrossRef]
- Aslam, S.; Lampley, E.; Wooten, D.; Karris, M.; Benson, C.; Strathdee, S.; Schooley, R.T. Lessons Learned from the First 10 Consecutive Cases of Intravenous Bacteriophage Therapy to Treat Multidrug-Resistant Bacterial Infections at a Single Center in the United States. Open Forum Infect. Dis. 2020, 7, ofaa389. [Google Scholar] [CrossRef] [PubMed]
- Soto-Rodriguez, S.A.; Lozano-Olvera, R.; Montfort, G.R.C.; Zenteno, E.; Sánchez-Salgado, J.L.; Vibanco-Pérez, N.; Aguilar Rendón, K.G. New Insights into the Mechanism of Action of PirAB from Vibrio Parahaemolyticus. Toxins 2022, 14, 243. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.R.; Shin, J.; Guevarra, R.B.; Lee, J.H.; Kim, D.W.; Seol, K.H.; Lee, J.H.; Kim, H.B.; Isaacson, R.E. Indices for a Better Understanding of MicrDeciphering Diversityobial Communities. J. Microbiol. Biotechnol. 2017, 27, 2089–2093. [Google Scholar] [CrossRef] [PubMed]
- Fortier, L.C.; Sekulovic, O. Importance of Prophages to Evolution and Virulence of Bacterial Pathogens. Virulence 2013, 4, 354–365. [Google Scholar] [CrossRef]
- Dion, M.B.; Oechslin, F.; Moineau, S. Phage Diversity, Genomics and Phylogeny. Nat. Rev. Microbiol. 2020, 18, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Chen, H.; Huang, Q.; Huang, S.; He, J.; Zhang, J.; Wu, Q.; Li, X.; Hu, W.; Yang, M. Characterization and Genomic Analysis of Novel Vibrio Parahaemolyticus Phage VB_VpaP_DE10. Viruses 2022, 14, 1609. [Google Scholar] [CrossRef] [PubMed]
- Chaturongakul, S.; Ounjai, P. Phage-Host Interplay: Examples from Tailed Phages and Gram-Negative Bacterial Pathogens. Front. Microbiol. 2014, 5, 442. [Google Scholar] [CrossRef]
- Thanki, A.M.; Brown, N.; Millard, A.D.; Clokie, M.R.J. Genomic Characterization of Jumbo Salmonella Phages That Effectively Target United Kingdom Pig-Associated Salmonella Serotypes. Front. Microbiol. 2019, 10, 1491. [Google Scholar] [CrossRef]
- Lim, A.N.W.; Yen, M.; Seed, K.D.; Lazinski, D.W.; Camilli, A. A Tail Fiber Protein and a Receptor-Binding Protein Mediate ICP2 Bacteriophage Interactions with Vibrio Cholerae OmpU. J. Bacteriol. 2021, 203, 1128. [Google Scholar] [CrossRef]
- Sun, Z.; Huang, S.; Zhu, P.; Tzehau, L.; Zhao, H.; Lv, J.; Zhang, R.; Zhou, L.; Niu, Q.; Wang, X.; et al. Species-Resolved Sequencing of Low-Biomass or Degraded Microbiomes Using 2bRAD-M. Genome Biol. 2022, 23, 36. [Google Scholar] [CrossRef]
- Wang, S.; Meyer, E.; Mckay, J.K.; Matz, M.V. 2b-RAD: A Simple and Flexible Method for Genome-Wide Genotyping. Nat. Methods 2012, 9, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Y.; Ng, T.H.; Wu, J.H.; Chen, J.W.; Wang, H.C. Microbiome Dynamics in a Shrimp Grow-out Pond with Possible Outbreak of Acute Hepatopancreatic Necrosis Disease. Sci. Rep. 2017, 7, 9395. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.H. Bacteriophages; Interscience Publishers Inc.: New York, NY, USA, 1959. [Google Scholar]
- Ackermann, H.-W.; Heldal, M. Basic Electron Microscopy of Aquatic Viruses. In Manual of Aquatic Viral Ecology; American Society of Limnology and Oceanography: Waco, TX, USA, 2010; pp. 182–192. [Google Scholar]
- Verma, V.; Harjai, K.; Chhibber, S. Characterization of a T7-Like Lytic Bacteriophage of Klebsiella Pneumoniae B5055: A Potential Therapeutic Agent. Curr. Microbiol. 2009, 59, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Hyman, P. Phages for Phage Therapy: Isolation, Characterization, and Host Range Breadth. Pharmaceuticals 2019, 12, 35. [Google Scholar] [CrossRef]










| Read Number | Total Bases (bp) | N Rate (%) | GC Content (%) | Q20 Rate (%) | Q30 Rate (%) |
|---|---|---|---|---|---|
| 20,297,674 | 3,044,540,950 | 0.000779 | 40.8 | 98.52 | 94.98 |
| Size (bp) | GC Content (%) | A (%) | G (%) | C (%) | T (%) |
|---|---|---|---|---|---|
| 110,776 | 40.91 | 29.48 | 19.37 | 21.54 | 29.61 |
| ORF Number | EggNOG Number | Prediction of Protein Functions |
|---|---|---|
| ORF 1 | ENOG411EP44 | DNA polymerase |
| ORF 17 | ENOG411EPEK | PhoH-like protein |
| ORF 20 | ENOG411EP1T | Deoxyribonucleoside diphosphate metabolic process |
| ORF 21 | ENOG411EPH2 | Ribonucleotide reductase, barrel domain |
| ORF 35 | ENOG411EPBV | Flavin adenine dinucleotide binding |
| ORF 47 | ENOG411EPAI | NAD biosynthetic process |
| ORF 53 | ENOG411EP4J | Macro domain |
| ORF 55 | ENOG411EPQT | Magnesium ion binding |
| ORF 57 | ENOG411EP64 | DNA helicase |
| ORF 71 | ENOG411EP4Y | - |
| ORF 73 | ENOG411EPJD | Glutamine amidotransferase domain |
| ORF 76 | ENOG411EPAU | Phage phiEco32-like COOH.NH2 ligase-type 2 |
| ORF 152 | ENOG411EP64 | DNA helicase |
| ORF 153 | ENOG411EPA3 | - |
| ORF 156 | ENOG411EPGN | - |
| ORF 167 | ENOG411EP2N | - |
| ORF 175 | ENOG411EP0H | T4 recombination endonuclease VII |
| ORF 185 | ENOG411EP9Z | Exonuclease activity |
| ORF 194 | ENOG411EP4C | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Y.; Sun, J.; Hu, J.; Bao, Z.; Wang, M. Characterization and Preliminary Application of a Novel Lytic Vibrio parahaemolyticus Bacteriophage vB_VpaP_SJSY21. Int. J. Mol. Sci. 2023, 24, 17529. https://doi.org/10.3390/ijms242417529
Xu Y, Sun J, Hu J, Bao Z, Wang M. Characterization and Preliminary Application of a Novel Lytic Vibrio parahaemolyticus Bacteriophage vB_VpaP_SJSY21. International Journal of Molecular Sciences. 2023; 24(24):17529. https://doi.org/10.3390/ijms242417529
Chicago/Turabian StyleXu, Yajin, Jun Sun, Jingjie Hu, Zhenmin Bao, and Mengqiang Wang. 2023. "Characterization and Preliminary Application of a Novel Lytic Vibrio parahaemolyticus Bacteriophage vB_VpaP_SJSY21" International Journal of Molecular Sciences 24, no. 24: 17529. https://doi.org/10.3390/ijms242417529
APA StyleXu, Y., Sun, J., Hu, J., Bao, Z., & Wang, M. (2023). Characterization and Preliminary Application of a Novel Lytic Vibrio parahaemolyticus Bacteriophage vB_VpaP_SJSY21. International Journal of Molecular Sciences, 24(24), 17529. https://doi.org/10.3390/ijms242417529






