Oral Pyogenic Granuloma: A Narrative Review
Abstract
1. Introduction
2. Epidemiology
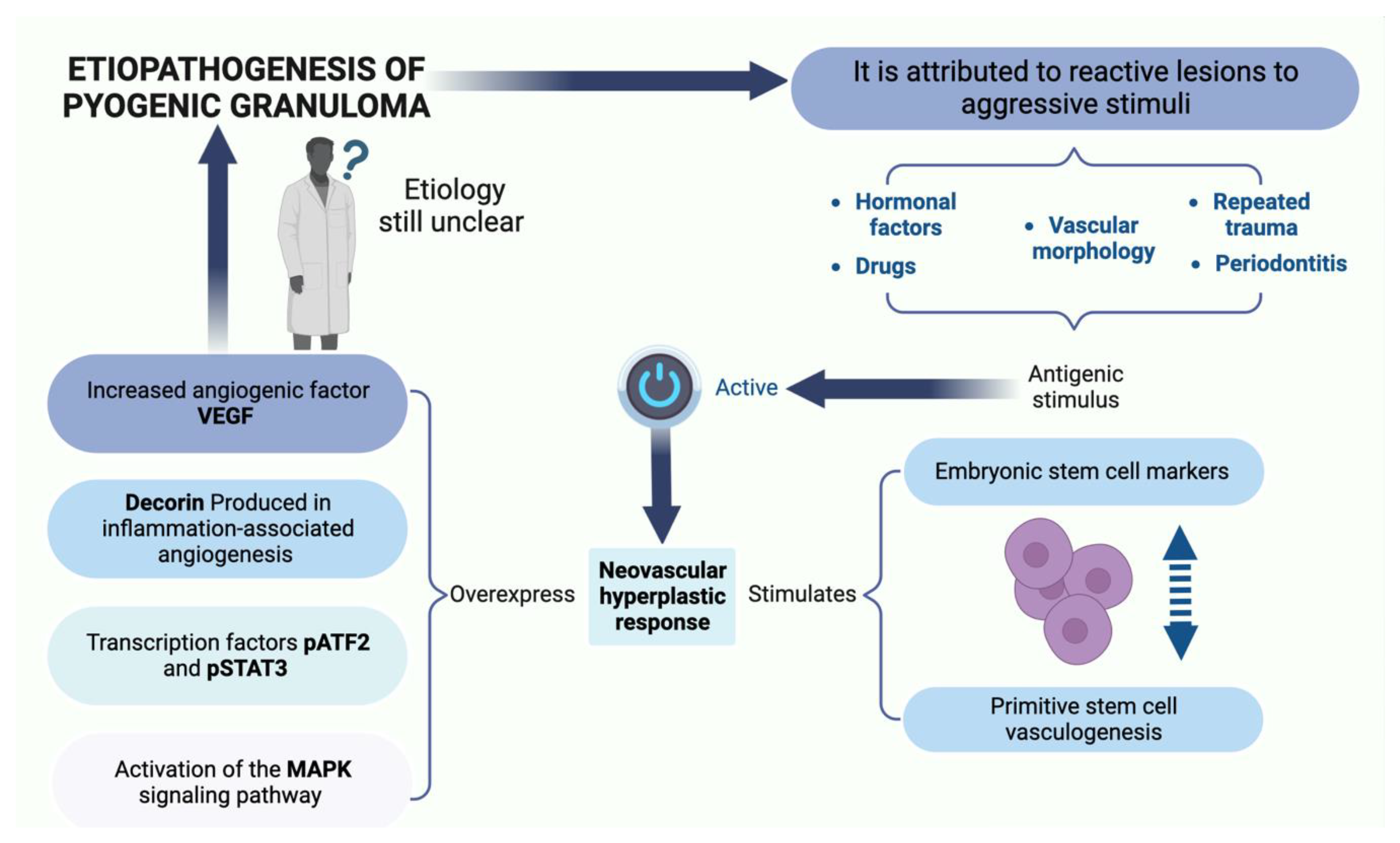
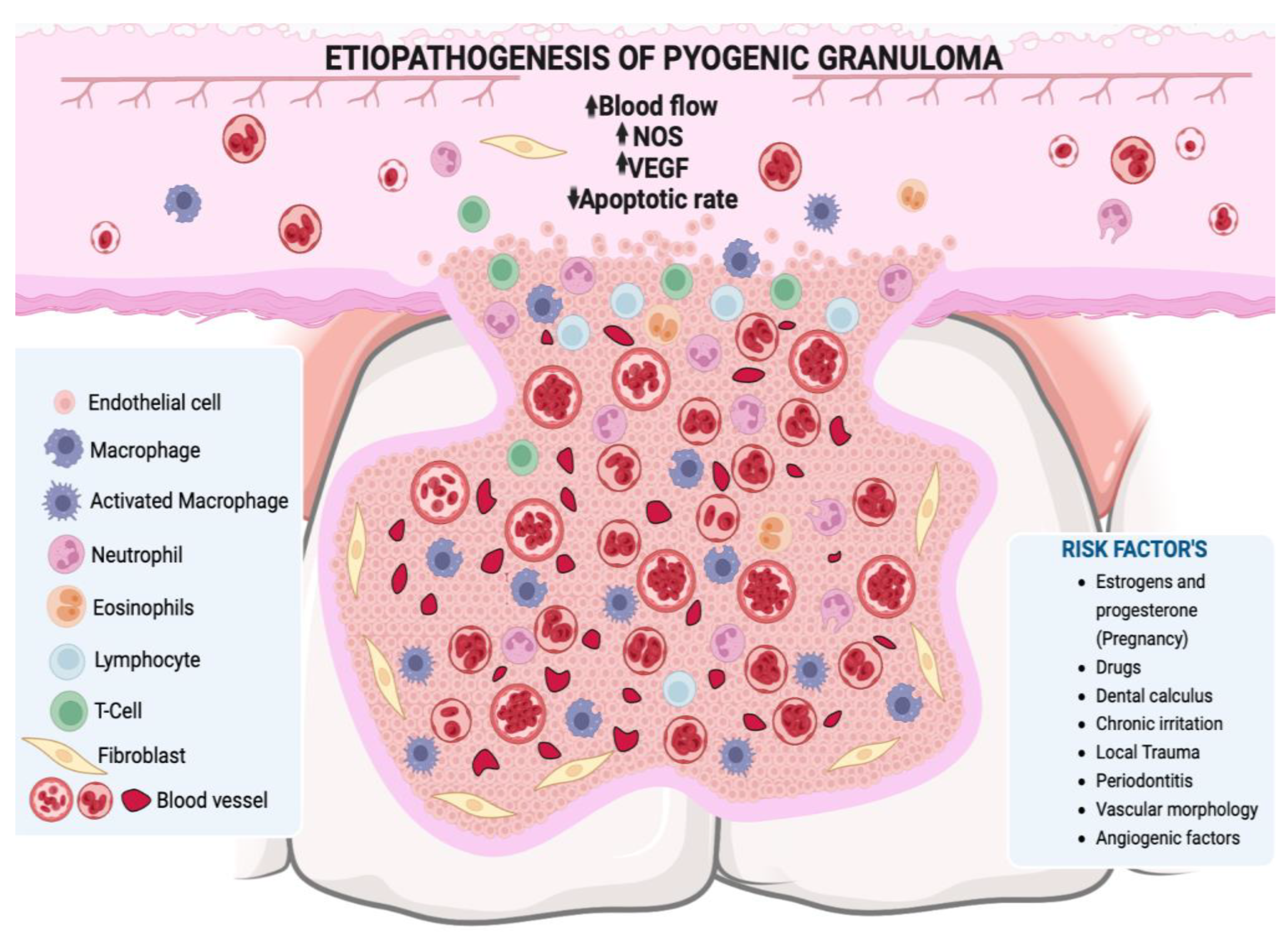
3. Etiopathogenesis
4. Clinical Features
| Year | Author | Country | Age (Years) | Gender | Location | Consistency | Comorbidity | Radiographic | Lesion Size | Diagnosis | Treatment |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Features | |||||||||||
| 2001 | Akyol et al., 2001 [72] | Turkey | 4 m | B | Tongue | Soft | No | NA | 1 × 0.8 × 0.8 cm | H | Surgical excision |
| 2002 | Aguilo L. 2002 [81] | Spain | 19 m | B | Gingiva | Soft | No | Fracture of the crown of 61 | NA | H | Surgical excision |
| 2006 | Parisi E et al., 2006 [6] | USA | 33 | F | Gingiva | NA | NA | NA | 0.5 × 0.5 cm | H | Intralesional corticosteroids |
| 2006 | Patil et al., 2006 [86] | India | 50 | F | Lower Lip | NA | No | NA | 1 × 0.5 cm | H | Surgical excision |
| 2006 | Shenoy S. 2006 [87] | India | 8 | G | Gingiva | NA | NA | loss of alveolar crestal bone | 2.0 × 1.0 × 1.0 cm | H | Surgical excision |
| 2008 | Amirchaghmaghi et al., 2008 [88] | Iran | 16 | M | Hard palate | Firm | No | NA | 0.7 cm | H | Surgical excision |
| 2009 | Goncalves 2009 [79] | Brazil | 12 | F | Upper lip | NA | NA | NA | 1.0 cm | H | Surgical excision |
| 2009 | Olmedo D et al., 2009 [89] | Argentina | 75 | F | Peri-implant mucosa | NA | No | No abnormalities | 1.0 × 1.0 × 0.6 cm | H | Surgical excision |
| Argentina | 64 | F | Peri-implant mucosa | Firm | No | Bone loss | 0.6 × 0.5 × 0.4 cm | H | Surgical excision | ||
| 2010 | Gondivkar et al., 2010 [90] | India | 25 | F | Gingiva | Soft | Pregnancy | Alveolar bone loss | 3 × 7 cm | H | Surgical excision |
| 2010 | Rizwanulla et al., 2010 [91] | Nepal | 13 | F | Gingiva | Firm | No | NA | 2.0 × 1.0 × 1.0 cm | H | Surgical excision |
| 2011 | Behl et al., 2011 [85] | India | 60 | F | Gingiva | Firm | No | Vertical bone loss | 3.2 × 3.4 cm | H | Surgical excision |
| 2011 | Mubben et al., 2011 [76] | India | 63 | F | Gingiva | Soft | No | NA | 5.0 × 3.5 cm | H | Surgical excision |
| 2011 | Penseriya et al. 2011 [82] | India | 30 | M | Gingiva | NA | No | Interdental bone loss | 2.0 × 1.5 cm | H | Surgical excision |
| 2011 | Shivaswamy et al., 2011 [92] | India | 19 | M | Gingiva and palate | Soft | NA | Horizontal bone loss | 4.0 × 5.0 mm | H | Surgical excision |
| 2012 | Chandrashekar 2012 [93] | India | 28 | F | Gingiva | Soft | No | NA | NA | H | Surgical excision |
| 2012 | Panjwani et al., 2012 [75] | India | 69 | M | Tongue | Firm | No | NA | 2.0 × 3.0 cm | H | Surgical excision |
| 2012 | Ravi et al., 2012 [94] | India | 33 | M | Lower lip | Firm | NA | NA | 3.0 × 2.0 cm | H | Surgical excision |
| 2012 | Piscoya et al., 2012 [95] | Brazil | 44 | M | Lower lip | NA | NA | NA | NA | H | Surgical excision |
| 2012 | Verma et al., 2012 [96] | India | 30 | F | Gingiva | NA | No | Alveolar bone loss | 1.5 × 1.0 cm | H | Surgical excision |
| 2013 | Adusumilli et al., 2013 [97] | India | 24 | F | Gingiva | Firm | NA | NA | 2 × 3.5 cm | H | Surgical excision |
| 27 | F | Gingiva | Firm | NA | NA | 2 × 3.5 cm | Surgical excision | ||||
| 27 | F | Gingiva | Firm | NA | NA | 2 × 3.5 cm | H | Surgical excision | |||
| 32 | F | Gingiva | Firm | NA | NA | 1.5 × 2.5 cm | H | Surgical excision | |||
| 23 | F | Gingiva | Soft | NA | NA | 1.5 × 1.75 cm | H | Surgical excision | |||
| 26 | F | Gingiva | Soft | NA | NA | 2.0 × 2.5 cm | H | Surgical excision | |||
| 28 | F | Gingiva | Soft | NA | NA | 0.5 × 0.8 cm | H | Surgical excision | |||
| 2013 | Deshmukh et al., 2013 [83] | India | 9 | M | Gingiva and mucogingival junction | Soft | No | Horizontal bone loss | NA | H | Surgical excision |
| 2013 | Gomes et al., 2013 [2] | India | 22 | M | Gingiva | NA | NA | No bone loss | 2.1 × 4.4 cm | H | Surgical excision |
| 2013 | Kamala 2013 [3] | India | 30 | F | Upper lip | Firm | No | NA | 0.8 cm | H | Surgical excision |
| 2013 | Mahabob et al., 2013 [98] | India | 22 | F | Palate | Firm | Pregnancy | NA | 2 × 2 cm | H | Surgical excision |
| 2013 | Moraes, et al., 2013 [99] | Brazil | 65 | M | Gingiva | NA | Diabetes and high blood pressure | No abnormalities | NA | H | Surgical excision |
| 2013 | Sangamesh et al., 2013 [100] | India | 40 | F | Buccal mucosa | Firm | NA | NA | 1.5 × 1.5 cm | H | Surgical excision |
| 2014 | Asha et al., 2014 [84] | India | 54 | M | Lower lip | Firm | No | NA | 3.0 × 3.0 cm | H | Surgical excision |
| 2014 | Fekrazad et al., 2014 [96] | Iran | 24 | F | Gingiva | Soft | NA | No abnormalities | 1.4 × 0.8 mm | H | Laser excision |
| 2014 | Ghalayani et al., 2014 [5] | Iran | 45 | M | Tongue | NA | Epilepsy beginning | NA | 4.0 × 3.0 × 1.0 cm | H | Surgical excision |
| 2014 | Kejriwal et al., 2014 [101] | India | 59 | M | Gingiva | Firm | No | No abnormalities | 1.5 × 2.0 × 1.0 cm | H | Surgical excision |
| 2014 | Mastammanavar et al., 2014 [77] | India | 44 | F | Gingiva | Soft | NA | NA | 1.5 × 3.0 cm | H | Surgical excision |
| 2014 | Sun et al., 2014 [102] | China | 22 | F | Gingiva | NA | Pregnancy | NA | 3 cm | C | No |
| 2015 | Asnaashari et al., 2015 [103] | Iran | 6 | M | Gingiva | Soft | No | No abnormalities | 1.1 × 1.3 cm | H | Laser excision |
| 2015 | Bugshan et al., 2015 [104] | USA | 51 | F | Gingiva and palatal | NA | NA | NA | 0.9 × 0.6 cm buccal and 0.8 × 0.7 cm palatal. | H | Intralesional injections |
| 2015 | De Carvalho et al., 2015 [105] | Brazil | 11 | B | Upper Lip | NA | No | NA | 4.5 cm | H | Surgical |
| 2015 | Ganesan A. 2015 [80] | India | 49 | F | Gingiva | Firm | No | No bone loss | 4.0 × 3.0 cm | H | Surgical excision |
| 2015 | Sachdeva 2015 [106] | India | 45 | F | Buccal mucosa | Firm | No | NA | 2 × 1 cm | H | Surgical excision |
| 2015 | Tripathi et al., 2015 [107] | India | 55 | M | Gingiva | NA | No | Alveolar bone loss | 3 × 3 cm | H | Surgical excision |
| 2016 | Agarwal N et al., 2016 [108] | India | 8 d | B | Gingiva | Soft | No | NA | 0.5 × 0.8 cm | H | Surgical excision |
| 2016 | Al-Mohaya et al., 2016 [22] | Saudí arabia | 51 | F | Gingiva | Firm | Uncontrolled type II diabetes mellitus | NA | 2.0 × 1.5 cm | H | Laser excision |
| 2016 | Marla et al., 2016 [109] | Nepal | 40 | F | Gingiva | NA | NA | NA | 1.0–2.0 cm | H | Surgical excision |
| Nepal | 40 | F | Buccal mucosa | NA | NA | NA | 1.0–2.0 cm | H | Surgical excision | ||
| Nepal | 9 | M | Buccal mucosa | NA | NA | NA | <1.0 cm | H | Surgical excision | ||
| Nepal | 23 | F | Gingiva | NA | NA | NA | 1.0–2.0 cm | H | Surgical excision | ||
| 2016 | Cheney et al., 2016 [21] | USA | 16 | M | Tongue, Buccal mucosa, | Soft | Acute lymphoblastic leukemia | N/A | N/A | Surgical excision | |
| USA | 14 | G | Tongue | N/A | Fanconi anemia | No | 0.5 × 0.2cm 1.0 × 0.3 cm 0.3 × 0.2 | H | Surgical excision | ||
| USA | 11 | G | Tongue | N/A | Fanconi anemia | No | 2.0 × 2.5 cm | H | Surgical excision | ||
| USA | 15 | Buccal mucosa | Stage IIIB nodular sclerosing Hodgkin lymphoma. | No | 1 cm 2.0 × 4.0 cm | H | CO2 laser | ||||
| USA | 6 | B | Tongue | N/A | Junctional epidermolysis bullosa | No | 1.0 × 0.5 cm | H | Surgical excision | ||
| 2017 | Rosa et al., 2017 [73] | México | 34 | F | Gingiva | Firm | No | Absence of interproximal contact | 1.5 × 0.9 cm | H | Surgical excision |
| 2018 | Tenore et al., 2018 [110] | Italy | 28 | F | Retromolar region | Soft | No | No abnormalities | NA | H | CO2 laser |
| 58 | M | Gingiva | Soft | No | No abnormalities | H | CO2 laser | ||||
| 19 | M | Retromolar region | Soft | No | No abnormalities | NA | H | CO2 laser | |||
| 21 | M | Retromolar region | Soft | No | No abnormalities | NA | H | CO2 laser | |||
| 2018 | Canivell et al., 2018 [111] | Spain | 32 | F | Lower lip | NA | Pregnancy | NA | 0.5 × 1.0 cm | H | Surgical excision |
| 2018 | Parajuli et al., 2018 [74] | Nepal | 26 | F | Tongue | Soft | No | NA | 2.5 × 2.0 cm | H | Surgical excision |
| Nepal | 15 | G | Upper lip | NA | NA | NA | 0.5 × 0.5 cm | C | Surgical excision | ||
| 2019 | Poudel et al., 2019 [112] | Nepal | 49 | M | Upper lip | Firm | NA | NA | 0.6 × 0.8 cm | H | Surgical excision |
| 2020 | Gutierrez 2020 [24] | Perú | 51 | F | Alveolar ridge | NA | Hypothyroidism erythrodermic psoriasis | Alveolar bone loss | 2.5 × 2.5 cm | H | Surgical excision |
| 2020 | Banjar et al., 2020 [113] | Saudí arabia | 15 | B | Lower lip | Soft | No | NA | 1.2 × 0.8 × 0.6 cm | H | Surgical excision |
| 2021 | Aragaki et al., 2021 [27] | Japan | 55 | M | Tongue | Soft | Gastric Cancer | No abnormalities | 0.6cm | H | Surgical excision |
| 67 | M | Upper lip | Soft | Gastric Cancer | No abnormalities | 0.5 mm | H | Surgical excision | |||
| 2021 | Pisano et al., 2021 [114] | Italy | 11 | G | Lower lip | Soft | No | N/A | 1.5 cm | H | Diode Laser |
| 2023 | Lomelí et al., 2023 [115] | México | 32 | F | Hard palate | Soft | No | Alveolar bone loss | 25 × 12 mm | H | Surgical excision |
| México | 42 | F | Gingiva | Soft | No | Alveolar bone loss | 16 × 10 mm | H | Surgical excision | ||
| 38 | F | Gingiva | 20 × 15 mm |
5. Radiographic Features
6. Microscopic Features
7. Differential Diagnosis of Oral PG
8. Treatment of Oral PG
9. Future Perspective
10. Conclusions
Funding
Conflicts of Interest
References
- Sarwal, P.; Lapumnuaypol, K. Pyogenic Granuloma. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA.
- Gomes, S.R.; Shakir, Q.J.; Thaker, P.V.; Tavadia, J.K. Pyogenic granuloma of the gingiva: A misnomer?—A case report and review of literature. J. Indian Soc. Periodontol. 2013, 17, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Kamal, R.; Dahiya, P.; Puri, A. Oral pyogenic granuloma: Various concepts of etiopathogenesis. J. Oral Maxillofac. Pathol. 2012, 16, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Chandra, S.; Gupta, S.; Srivastava, S. Heterogeneous conceptualization of etiopathogenesis: Oral pyogenic granuloma. Natl. J. Maxillofac. Surg. 2019, 10, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Ghalayani, P.; Hajisadeghi, S.; Babadi, F. Extragingival pyogenic granuloma associated with medication: Report of an unusual case. Dent. Res. J. 2014, 11, 400–404. [Google Scholar]
- Parisi, E.; Glick, P.H.; Glick, M. Recurrent intraoral pyogenic granuloma with satellitosis treated with corticosteroids. Oral. Dis. 2006, 12, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Al-Noaman, A.S. Pyogenic granuloma: Clinicopathological and treatment scenario. J. Indian Soc. Periodontol. 2020, 24, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Poncet, A.; Dor, L. Botyromycose humaine. Rev. Chir. 1897, 18, 996. [Google Scholar]
- Greenberger, S.; Boscolo, E.; Adini, I.; Mulliken, J.B.; Bischoff, J. Corticosteroid suppression of VEGF-A in infantile hemangio-ma-derived stem cells. N. Engl. J. Med. 2010, 362, 1005–1013. [Google Scholar] [CrossRef]
- Jafarzadeh, H.; Sanatkhani, M.; Mohtasham, N. Oral pyogenic granuloma: A review. J. Oral. Sci. 2006, 48, 167–175. [Google Scholar] [CrossRef]
- Wiener, R.C.; Wiener-Pla, R. Literacy, pregnancy and potential oral health changes: The Internet and readability levels. Matern. Child Health J. 2014, 18, 657–662. [Google Scholar] [CrossRef]
- Montazer Lotf-Elahi, M.S.; Farzinnia, G.; Jaafari-Ashkavandi, Z. Clinicopathological study of 1000 biopsied gingival lesions among dental outpatients: A 22-year retrospective study. BMC Oral Health 2022, 22, 154. [Google Scholar] [CrossRef] [PubMed]
- Krishnapillai, R.; Punnoose, K.; Angadi, P.V.; Koneru, A. Oral pyogenic granuloma—A review of 215 cases in a South Indian Teaching Hospital, Karnataka, over a period of 20 years. Oral Maxillofac. Surg. 2012, 16, 305–309. [Google Scholar] [CrossRef]
- Shamim, T.; Varghese, V.I.; Shameena, P.M.; Sudha, S. A retrospective analysis of gingival biopsied lesions in South Indian population: 2001–2006. Med. Oral. Patol. Oral Cir. Bucal 2008, 13, E414–E418. [Google Scholar]
- Nejad, E.S.; BigomTaheri, J.; Azimi, S. Frequency of gingival pregnancy tumor in Iran (confirmed by biopsy). J. Int. Oral Health 2014, 6, 72–76. [Google Scholar] [PubMed]
- Chamani, G.; Navvabi, N.; Abdollahzadeh, S.H. The frequency of pregnancy tumor in pregnant mothers. Dent. J. Shiraz Med. Sci. Univ. 2009, 10, 79–82. [Google Scholar]
- Lee, J.; Lynde, C. Pyogenic granuloma: Pyogenic again? Association between pyogenic granuloma and Bartonella. J. Cutan. Med. Surg. 2001, 5, 467–470. [Google Scholar] [CrossRef]
- Kerr, D.A. Granuloma pyogenicum. Oral Surg. Oral Med. Oral Pathol. 1951, 4, 158–176. [Google Scholar] [CrossRef]
- Bhaskar, S.N.; Jacoway, J.R. Pyogenic granuloma—Clinical features, incidence, histology, and result of treatment: Report of 242 cases. J. Oral Surg. 1966, 24, 391–398. [Google Scholar]
- Piraccini, B.M.; Bellavista, S.; Misciali, C.; Tosti, A.; de Berker, D.; Richert, B. Periungual and subungual pyogenic granuloma. Br. J. Dermatol. 2010, 163, 941–953. [Google Scholar] [CrossRef]
- Cheney-Peters, D.; Lund, T.C. Oral Pyogenic Granuloma After Bone Marrow Transplant in the Pediatric/Adolescent Population: Report of 5 Cases. J. Pediatr. Hematol. Oncol. 2016, 38, 570–573. [Google Scholar] [CrossRef]
- Al-Mohaya, M.; Treister, N.; Al-Khadra, O.; Lehmann, L.; Padwa, B.; Woo, S.B. Calcineurin inhibitor-associated oral inflammatory polyps after transplantation. J. Oral Pathol. Med. 2007, 36, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Palmero, M.L.; Pope, E. Eruptive pyogenic granulomas developing after drug hypersensitivity reaction. J. Am. Acad. Dermatol. 2009, 60, 855–857. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez Patiño-Paul, A.A. Un inusual caso de granuloma piogeno oral multiple,¿ asociado a tratamiento con levotiroxina? Rev. Estomatol. Hered. 2020, 30, 294–301. [Google Scholar] [CrossRef]
- Tosti, A.; Piraccini, B.M.; D’Antuono, A.; Marzaduri, S.; Bettoli, V. Paronychia associated with antiretroviral therapy. Br. J. Dermatol. 1999, 140, 1165–1168. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.K.C.; Barankin, B.; Hon, K.L. Pyogenic Granuloma. Clin. Mother. Child. Health 2014, 11, e106. [Google Scholar] [CrossRef]
- Aragaki, T.; Tomomatsu, N.; Michi, Y.; Hosaka, H.; Fukai, Y.; Iijima, M.; Yoda, T. Ramucirumab-related Oral Pyogenic Granuloma: A Report of Two Cases. Intern. Med. 2021, 60, 2601–2605. [Google Scholar] [CrossRef] [PubMed]
- Uzel, M.I.; Kantarci, A.; Hong, H.H.; Uygur, C.; Sheff, M.C.; Firatli, E.; Trackman, P.C. Connective tissue growth factor in drug-induced gingival overgrowth. J. Periodontol. 2001, 72, 921–931. [Google Scholar] [CrossRef]
- Baran, R. Etretinate and the nails (study of 130 cases) possible mechanisms of some side-effects. Clin. Exp. Dermatol. 1986, 11, 148–152. [Google Scholar] [CrossRef]
- Williams, L.H.; Fleckman, P. Painless periungual pyogenic granulomata associated with reverse transcriptase inhibitor therapy in a patient with human immunodeficiency virus infection. Br. J. Dermatol. 2007, 156, 163–164. [Google Scholar] [CrossRef]
- Silva de Araujo Figueiredo, C.; Gonçalves Carvalho Rosalem, C.; Costa Cantanhede, A.L.; Abreu Fonseca Thomaz, É.B.; Fontoura Nogueira da Cruz, M.C. Systemic alterations and their oral manifestations in pregnant women. J. Obstet. Gynaecol. Res. 2017, 43, 16–22. [Google Scholar] [CrossRef]
- Figuero-Ruiz, E.; Prieto Prieto, I.; Bascones-Martínez, A. Cambios hormonales asociados al embarazo. Afectación gingivo-periodontal. Av. Periodoncia Implantol. Oral 2006, 18, 101–113. [Google Scholar]
- Cardoso, J.A.; Spanemberg, J.C.; Cherubini, K.; Figueiredo, M.A.; Salum, F.G. Oral granuloma gravidarum: A retrospective study of 41 cases in Southern Brazil. J. Appl. Oral. Sci. 2013, 21, 215–218. [Google Scholar] [CrossRef]
- Jang, H.; Patoine, A.; Wu, T.T.; Castillo, D.A.; Xiao, J. Oral microflora and pregnancy: A systematic review and meta-analysis. Sci. Rep. 2021, 11, 16870. [Google Scholar] [CrossRef] [PubMed]
- Borgo, P.V.; Rodrigues, V.A.; Feitosa, A.C.; Xavier, K.C.; Avila-Campos, M.J. Association between periodontal condition and sub-gingival microbiota in women during pregnancy: A longitudinal study. J. Appl. Oral Sci. 2014, 22, 528–533. [Google Scholar] [CrossRef]
- Ojanotko-Harri, A.O.; Harri, M.P.; Hurttia, H.M.; Sewon, L.A. Altered tissue metabolism of progesterone in pregnancy gingivitis and granuloma. J. Clin. Periodontol. 1991, 18, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Jin, Y.T.; Lin, M.T. Expression of Tie-2, angiopoietin-1, angiopoietin-2, ephrinB2 and EphB4 in pyogenic granuloma of human gingiva implicates their roles in inflammatory angiogenesis. J. Periodontol. Res. 2000, 35, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Jin, Y.T.; Lin, M.T. The detection and comparison of angiogenesis-associated factors in pyogenic granuloma by im-munohistochemistry. J. Periodontol. 2000, 71, 701–709. [Google Scholar] [CrossRef]
- Shetty, S.J.; Hallikeri, K.; Anehosur, V.; Desai, A. An aggressive pyogenic granuloma masquerading as a vascular neoplasm. J. Indian. Soc. Periodontol. 2020, 24, 276–279. [Google Scholar] [CrossRef]
- Nelimarkka, L.; Salminen, H.; Kuopio, T.; Nikkari, S.; Ekfors, T.; Laine, J.; Pelliniemi, L.; Järveläinen, H. Decorin is produced by capillary endothelial cells in inflammation-associated angiogenesis. Am. J. Pathol. 2001, 158, 345–353. [Google Scholar] [CrossRef]
- Cooper, P.H.; McAllister, H.A.; Helwig, E.B. Intravenous pyogenic granuloma. A study of 18 cases. Am. J. Surg. Pathol. 1979, 3, 221–228. [Google Scholar] [CrossRef]
- Fakoory, J.M.W.; Smith, A.L.; Depeiza, N. Intravascular lobular capillary hemangioma of the temporal artery. J. Vasc. Surg. Cases Innov. Tech. 2022, 8, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Wen, X.; Zhang, M.; Zhou, C.; Huang, J.; He, Y. Ultrasonographic features of an intravascular lobular capillary hemangioma originating in the basilic vein: Case report and literature review. J. Clin. Ultrasound. 2021, 49, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.H.; Kuo, H.W.; Chiu, Y.K.; Huang, P.H. Intravascular pyogenic granuloma arising in an acquired arteriovenous malformation: Report of a case and review of the literature. Dermatol. Surg. 2004, 30, 1050–1053. [Google Scholar] [CrossRef] [PubMed]
- Mahapatra, Q.S.; Sahai, K.; Malik, A.; Mani, N.S. Intravascular papillary endothelial hyperplasia: An unusual histopathological entity. Indian Dermatol. Online J. 2015, 6, 277–279. [Google Scholar] [PubMed]
- Díaz-Flores, L.; Gutiérrez, R.; González-Gómez, M.; García, M.P.; Carrasco, J.L.; Díaz-Flores, L., Jr.; Madrid, J.F.; Álvarez-Argüelles, H. Participation of intussusceptive angiogenesis in the morphogenesis of lobular capillary hemangioma. Sci. Rep. 2020, 10, 4987. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda, A.; Buchanan, E.P. Vascular tumors. Semin. Plast. Surg. 2014, 28, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Blaya, E.; Vento, V.; Kuntz, S.; Bruyns, L.; Ohana, M.; Weingertner, N.; Lejay, A.; Chakfé, N. Intravenous lobular capillary haemangioma (pyogenic granuloma) of the superior vena cava: Case report and literature review. EJVES Vasc. Forum 2021, 50, 32–36. [Google Scholar] [CrossRef]
- Naderi, N.J.; Eshghyar, N.; Esfehanian, H. Reactive lesions of the oral cavity: A retrospective study on 2068 cases. Dent. Res. J. 2012, 9, 251–255. [Google Scholar]
- Wynn, T.A. Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J. Clin. Investig. 2007, 117, 524–529. [Google Scholar] [CrossRef]
- Kapoor, P.; Deshmukh, R.S. VEGF: A critical driver for angiogenesis and subsequent tumor growth: An IHC study. J. Oral Maxillofac. Pathol. 2012, 16, 330–337. [Google Scholar] [CrossRef]
- Aghbali, A.; Akbarzadeh, A.; Kouhsoltani, M. The role of macrophages and eosinophils in reactive lesions of the oral cavity. J. Oral Maxillofac. Pathol. 2018, 22, 147. [Google Scholar] [CrossRef] [PubMed]
- Sheethal, H.S.; Kn, H.; Smitha, T.; Chauhan, K. Role of mast cells in inflammatory and reactive pathologies of pulp, periapical area and periodontium. J. Oral Maxillofac. Pathol. 2018, 22, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Shekar, S.; Angadi, P.V. Quantification of mast cells in reactive oral lesions—A clue to the morphologic diversity. Indian. J. Health Sci. Biomed. Res. 2019, 12, 123–126. [Google Scholar]
- Komi, D.; Khomtchouk, K.; Maria, P. A review of the contribution of mast cells in wound healing: Involved molecular and cellular mechanisms. Clin. Rev. Allergy Immunol. 2020, 58, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.; Xavier, F.; Freitas, M.; Nunes, F.; Gurgel, C.; Cangussu, M. Involvement of mast cells and microvessels density in reactive lesions of oral cavity: A comparative immunohistochemical study. Pathol. Res. Pract. 2016, 212, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Kouhsoltani, M.; Khiavi, M.M.; Tahamtan, S. Evaluation of mast cell counts and microvessel density in reactive lesions of the oral cavity. J. Dent. Res. Dent. Clin. Dent. Prospects. 2016, 10, 241–246. [Google Scholar] [CrossRef]
- Puxeddu, I.; Berkman, N.; Nissim Ben Efraim, A.H.; Davies, D.E.; Ribatti, D.; Gleich, G.J.; Levi-Schaffer, F. The role of eosinophil major basic protein in angiogenesis. Allergy 2009, 64, 368–374. [Google Scholar] [CrossRef]
- Nissim Ben Efraim, A.H.; Levi-Schaffer, F. Roles of eosinophils in the modulation of angiogenesis. Chem. Immunol. Allergy 2014, 99, 138–154. [Google Scholar] [CrossRef]
- Nakamura, F.; Fifita, S.F.; Kuyama, K. A study of oral irritation fibroma with special reference to clinicopathological and immunohistochemical features of stromal spindle cells. Int. J. Oral Med. Sci. 2005, 4, 83–91. [Google Scholar] [CrossRef][Green Version]
- Vasanthi, V.; Divya, B.; Ramadoss, R.; Deena, P.; Annasamy, R.K.; Rajkumar, K. Quantification of inflammatory, angiogenic and fibrous components of reactive oral lesions with an insight into pathogenesis. J. Oral Maxillofac. Pathol. 2022, 26, 600. [Google Scholar]
- Hunasgi, S.; Koneru, A.; Vanishree, M.; Manvikar, V. Assessment of reactive gingival lesions of oral cavity: A histopathological study. J. Oral Maxillofac. Pathol. 2017, 21, 180. [Google Scholar] [CrossRef] [PubMed]
- Vara, J.T.; Gurudu, V.S.; Ananthaneni, A.; Bagalad, B.S.; Kuberappa, P.H.; Ponnapalli, H.P. Correlation of Vascular and Inflammatory Index in Oral Pyogenic Granuloma and Periapical Granuloma—An Insight into Pathogenesis. J. Clin. Diagn. Res. 2017, 11, ZC25–ZC28. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, M.H.; Shaik, A.S.; Vaali-Mohammed, M.A.; Al Khayal, K.A.; Traiki, T.B.; Zubaidi, A.M.; Al-Johani, T.; Shakoor, Z.; Al-Obeed, O.A. Expression of VEGF, EGF and HGF in early- and late-stage colorectal cancer. Mol. Clin. Oncol. 2021, 15, 251. [Google Scholar] [CrossRef] [PubMed]
- Blackwell, M.G.; Itinteang, T.; Chibnall, A.M.; Davis, P.F.; Tan, S.T. Expression of embryonic stem cell markers in pyogenic granu-loma. J. Cutan. Pathol. 2016, 43, 1096–1101. [Google Scholar] [CrossRef]
- Pereira, T.D.S.F.; de Amorim, L.S.D.; Pereira, N.B.; Vitório, J.G.; Duarte-Andrade, F.F.; Guimarães, L.M.; Diniz, M.G.; Gomes, C.C.; Gomez, R.S. Oral pyogenic granulomas show MAPK/ERK signaling pathway activation, which occurs independently of BRAF, KRAS, HRAS, NRAS, GNA11, and GNA14 mutations. J. Oral Pathol. Med. 2019, 48, 906–910. [Google Scholar] [CrossRef]
- Gapizov, A.; Sidhu, H.K.; Affaf, M.; Safdar, S.; Irfan, R.; Ekhator, C.; Saddique, M.N.; Devi, M.; Cabana, E.O.S.; Kamran, M.; et al. Unraveling Intravascular Lobular Capillary Hemangioma: A Comprehensive Scoping Review. Cureus 2023, 15, e45142. [Google Scholar] [CrossRef]
- Lim, Y.H.; Douglas, S.R.; Ko, C.J.; Antaya, R.J.; McNiff, J.M.; Zhou, J.; Choate, K.A.; Narayan, D. Somatic activating RAS mutations cause vascular tumors including pyogenic granuloma. J. Investig. Dermatol. 2015, 135, 1698–1700. [Google Scholar] [CrossRef]
- Groesser, L.; Peterhof, E.; Evert, M.; Landthaler, M.; Berneburg, M.; Hafner, C. BRAF and RAS mutations in sporadic and secondary pyogenic granuloma. J. Investig. Dermatol. 2016, 136, 481–486. [Google Scholar] [CrossRef]
- Lim, Y.H.; Bacchiocchi, A.; Qiu, J.; Straub, R.; Bruckner, A.; Bercovitch, L.; Narayan, D.; McNiff, J.; Ko, C.; Robinson-Bostom, L.; et al. GNA14 somatic mutation causes congenital and sporadic vascular tumors by MAPK activation. Am. J. Hum. Genet. 2016, 99, 443–450. [Google Scholar] [CrossRef]
- Yao, T.; Nagai, E.; Utsunomiya, T.; Tsuneyoshi, M. An intestinal counterpart of pyogenic granuloma of the skin: A newly proposed entity. Am. J. Surg. Pathol. 1995, 19, 1054–1060. [Google Scholar] [CrossRef]
- Akyol, M.U.; Yalçiner, E.G.; Doğan, A.I. Pyogenic granuloma (lobular capillary hemangioma) of the tongue. Int. J. Pediatr. Otorhinolaryngol. 2001, 58, 239–241. [Google Scholar] [CrossRef] [PubMed]
- Rosa, G.; Cartagena, C.; Andrea, L.; La Torre, C. Oral pyogenic granuloma diagnosis and treatment: A series of cases. Rev. Odontol. Mex. 2017, 21, 253–261. Available online: http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1870-199X2017000400253&lng=es&tlng=en (accessed on 4 September 2023). [CrossRef]
- Parajuli, R.; Maharjan, S. Unusual presentation of oral pyogenic granulomas: A review of two cases. Clin. Case Rep. 2018, 6, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Panjwani, S.; Issrani, R.; Keluskar, V.; Naik, Z. An unusual presentation of pyogenic granuloma. Indian J. Dent. 2012, 3, 178–181. [Google Scholar] [CrossRef]
- Mubeen, K.; Vijayalakshmi, K.R.; Abhishek, R.P. Oral pyogenic granuloma with mandible involvement: An unusual presentation. J. Dent. Oral Hyg. 2011, 3, 6–9. [Google Scholar]
- Mastammanavar, D.; Hunasgi, S.; Koneru, A.; Vanishree, M.R.; Surekha, R.; Vardendra, M. Aggressive Pyogenic Granuloma: A Case Report. Int. J. Oral Maxillofac. Pathol. 2014, 5, 29–32. [Google Scholar]
- Gonçales, E.S.; Damante, J.H.; Fischer Rubira, C.M.; Taveira, L.A. Pyogenic granuloma on the upper lip: An unusual location. J. Appl. Oral Sci. 2010, 18, 538–541. [Google Scholar] [CrossRef]
- Fekrazad, R.; Nokhbatolfoghahaei, H.; Khoei, F.; Kalhori, K.A. Pyogenic Granuloma: Surgical Treatment with Er:YAG Laser. J. Lasers Med. Sci. 2014, 5, 199–205. [Google Scholar]
- Ganesan, A.; Kumar, N.G.; Azariah, E.; Asokan, G.S. Oral pyogenic granuloma: A case report and a comprehensive review. SRM J. Res. Dent. Sci. 2015, 6, 257–260. Available online: https://www.srmjrds.in/text.asp?2015/6/4/257/170284 (accessed on 17 July 2023). [CrossRef]
- Aguilo, L. Pyogenic granuloma subsequent to injury of a primary tooth—A case report. Int. J. Paediatr. Dent. 2002, 12, 438–441. [Google Scholar] [CrossRef]
- Panseriya, B.J.; Hungund, S. Pyogenic granuloma associated with periodontal abscess and bone loss—A rare case report. Contemp. Clin. Dent. 2011, 2, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh JKulkarni, V.K.; Katti, G.; Deshpande, S. Pyogenic Granuloma, An Unusual Presentation in Pediatric Patient—A Case Report. Indian J. Dent. Sci. 2013, 5, 90–93. [Google Scholar]
- Asha, V.; Dhanya, M.; Patil, B.A.; Revanna, G. An unusual presentation of pyogenic granuloma of the lower lip. Contemp. Clin. Dent. 2014, 5, 524–526. [Google Scholar] [CrossRef] [PubMed]
- Behl, A.B.; Bali, V.; Bali, R. Pyogenic Granuloma—A case report and review of literature. Int. J. Stomatol. Occlusion Med. 2011, 4, 166–170. [Google Scholar] [CrossRef]
- Patil, K.; Mahima, V.G.; Lahari, K. Extragingival pyogenic granuloma. Indian J. Dent. Res. 2006, 17, 199–202, Erratum in Indian J. Dent. Res. 2007, 18, 45. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, S.S.; Dinkar, A.D. Pyogenic granuloma associated with bone loss in an eight year old child: A case report. J. Indian. Soc. Pedod. Prev. Dent. 2006, 24, 201–203. [Google Scholar] [CrossRef]
- Amirchaghmaghi, M.; Falaki, F.; Mohtasham, N.; Mozafari, P.M. Extragingival pyogenic granuloma: A case report. Cases J. 2008, 1, 371. [Google Scholar] [CrossRef]
- Olmedo, D.G.; Paparella, M.L.; Brandizzi, D.; Cabrini, R.L. Reactive lesions of peri-implant mucosa associated with titanium dental implants: A report of 2 cases. Int. J. Oral Maxillofac. Surg. 2010, 39, 503–507. [Google Scholar] [CrossRef]
- Gondivkar, S.M.; Gadbail, A.; Chole, R. Oral pregnancy tumor. Contemp. Clin. Dent. 2010, 1, 190–192. [Google Scholar] [CrossRef]
- Rizwanulla, M.D.; Koirala, B.; Sharma, S.; Adhikari, L.; Pradhan, A. Pyogenic Granuloma: A Case Report. Health Renaiss. 2011, 8, 196–198. [Google Scholar] [CrossRef]
- Shivaswamy, S.; Siddiqui, N.; Jain, S.A.; Koshy, A.; Tambwekar, S.; Shankar, A. A rare case of generalized pyogenic granuloma: A case report. Quintessence Int. 2011, 42, 493–499. [Google Scholar] [PubMed]
- Chandrashekar, B. Minimally invasive approach to eliminate pyogenic granuloma: A case report. Case Rep. Dent. 2012, 2012, 909780. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ravi, V.; Jacob, M.; Sivakumar, A.; Saravanan, S.; Priya, K. Pyogenic granuloma of labial mucosa: A misnomer in an anomolous site. J. Pharm. Bioallied Sci. 2012, 4 (Suppl. 2), S194–S196. [Google Scholar] [CrossRef] [PubMed]
- de Vasconcellos Piscoya MD, B.; de Alencar Ximenes, R.A.; Silva, G.M.; Jamelli, S.R.; Coutinho, S.B. Periodontitis-associated risk factors in pregnant women. Clinics 2012, 67, 27–33. [Google Scholar] [CrossRef]
- Verma, P.K.; Srivastava, R.; Baranwal, H.C.; Chaturvedi, T.P.; Gautam, A.; Singh, A. Pyogenic granuloma—Hyperplastic lesion of the gingiva: Case reports. Open Dent. J. 2012, 6, 153–156, Erratum in Open Dent. J. 2012, 6, 188. [Google Scholar] [CrossRef][Green Version]
- Adusumilli, S.; Yalamanchili, P.S.; Manthena, S. Pyogenic granuloma near the midline of the oral cavity: A series of case reports. J. Indian Soc. Periodontol. 2014, 18, 236–239. [Google Scholar] [CrossRef]
- Mahabob, N.; Kumar, S.; Raja, S. Palatal pyogenic granulomaa. J. Pharm. Bioallied Sci. 2013, 5 (Suppl. 2), S179–S181. [Google Scholar] [CrossRef]
- Moraes, S.H.; Moraes, G.F.; Durski, J.; Viero, F.L.; Da Silva Meira, D.D.; Caron, M.E. Granuloma Piogênico: Relato de Caso Clínico. Rev. Gestão Saúde 2013, 9, 12–19. [Google Scholar]
- Sangamesh, N.C.; Poornima, B.; Vidya, K.C.; Sakri, S.B. Extragingival pyogenic granuloma: A rare case report. J. Sci. Soc. 2013, 40, 49–51. [Google Scholar] [CrossRef]
- Kejriwal, S.; Bhandary, R.; Thomas, B. Oral pyogenic granuloma: A case report. Nitte Univ. J. Health Sci. 2014, 4, 123–125. [Google Scholar] [CrossRef]
- Sun, W.L.; Lei, L.; Zhong, L.; Yu, S.; Zhou, J.W. Multiple gingival pregnancy tumors with rapid growth. J. Dent. Sci. 2013, 9, 289–293. [Google Scholar] [CrossRef]
- Asnaashari, M.; Mehdipour, M.; MoradiAbbasabadi, F.; Azari-Marhabi, S. Expedited removal of pyogenic granuloma by diode laser in a pediatric patient. J. Lasers Med. Sci. 2015, 6, 40–44. [Google Scholar]
- Bugshan, A.; Patel, H.; Garber, K.; Meiller, T.F. Alternative Therapeutic Approach in the Treatment of Oral Pyogenic Granuloma. Case Rep. Oncol. 2015, 8, 493–497. [Google Scholar] [CrossRef]
- de Carvalho, F.K.; Pinheiro, T.N.; Arid, J.; de Queiroz, A.M.; de Rossi, A.; Nelson-Filho, P. Trauma-Induced Giant Pyogenic Granuloma in the Upper Lip. J. Dent. Child 2015, 82, 168–170. [Google Scholar]
- Sachdeva, S.K. Extragingival Pyogenic Granuloma: An Unusual Clinical Presentation. J. Dent. 2015, 16 (Suppl. 3), 282–285. [Google Scholar]
- Tripathi, A.K.; Upadhaya, V.; Kumar, V.; Saimbi, C.S. Hyperactive lesions of gingiva associated with severe alveolar bone loss: A rare finding. Contemp. Clin. Dent. 2015, 6, 223–225. [Google Scholar] [CrossRef]
- Agarwal, N.; Kumar, D.; Vaish, A.; Anand, A. A Rare Case of Pyogenic Granuloma with a Natal Tooth. J. Clin. Diagn. Res. 2016, 10, ZD28–ZD29. [Google Scholar] [CrossRef]
- Marla, V.; Shrestha, A.; Goel, K.; Shrestha, S. The Histopathological Spectrum of Pyogenic Granuloma: A Case Series. Case Rep. Dent. 2016, 2016, 1323798. [Google Scholar] [CrossRef]
- Tenore, G.; Mohsen, A.; Pompa, G.; Brauner, E.; Cassoni, A.; Valentini, V.; Polimeni, A.; Romeo, U. Gingival Reactive Lesions in Orally Rehabilitated Patients by Free Revascularized Flap. Case Rep. Dent. 2018, 2018, 2474706. [Google Scholar] [CrossRef]
- Canivell-Zabaleta, M.; Martin-Lozano, G.; Olmos-Juarez, E.; Fontillon-Alberdi, M.; Infante-Cossio, P. Extragingival Pregnancy Pyogenic Granuloma on the Lip. J. Craniofac. Surg. 2018, 29, e49–e50. [Google Scholar] [CrossRef]
- Poudel, P.; Chaurasia, N.; Marla, V.; Srii, R. Pyogenic granuloma of the upper lip: A common lesion in an uncommon location. J. Taibah Univ. Med. Sci. 2018, 14, 95–98. [Google Scholar] [CrossRef]
- Banjar, A.; Abdrabuh, A.; Al-Habshi, M.; Parambil, M.; Bastos, P.; Abed, H. Labial pyogenic granuloma related to trauma: A case report and mini-review. Dent. Traumatol. 2020, 36, 446–451. [Google Scholar] [CrossRef]
- Pisano, M.; Sammartino, P.; Di Vittorio, L.; Iandolo, A.; Caggiano, M.; Roghi, M.; Bizzoca, M.E.; Lo Muzio, L. Use of Diode Laser for Surgical Removal of Pyogenic Granuloma of the Lower Lip in a Pediatric Patient: A Case Report. Am. J. Case Rep. 2021, 22, e929690. [Google Scholar] [CrossRef]
- Lomelí Martínez, S.M.; Bocanegra Morando, D.; Mercado González, A.E.; Gómez Sandoval, J.R. Unusual clinical presentation of oral pyogenic granuloma with severe alveolar bone loss: A case report and review of literature. World J. Clin. Cases. 2023, 11, 3907–3914. [Google Scholar] [CrossRef]
- Ma, Y.; Nicolet, J. Specificity models in MAPK cascade signaling. FEBS Open Biol. 2023, 13, 1177–1192. [Google Scholar] [CrossRef]
- Rodríguez-Carballo, E.; Gámez, B.; Ventura, F. p38 MAPK Signaling in Osteoblast Differentiation. Front. Cell Dev. Biol. 2016, 4, 40. [Google Scholar] [CrossRef]
- Al-Mohaya, M.A.; Al-Malik, A.M. Excision of oral pyogenic granuloma in a diabetic patient with 940 nm diode laser. Saudi Med. J. 2016, 37, 1395–1400. [Google Scholar] [CrossRef]
- Mirshams, M.; Daneshpazhooh, M.; Mirshekari, A.; Taheri, A.; Mansoori, P.; Hekmat, S. Cryotherapy in the treatment of pyogenic granuloma. J. Eur. Acad. Dermatol. Venereol. 2006, 20, 788–790. [Google Scholar] [CrossRef]
- Moon, S.E.; Hwang, E.J.; Cho, K.H. Treatment of pyogenic granuloma by sodium tetradecyl sulfate sclerotherapy. Arch. Dermatol. 2005, 141, 644–646. [Google Scholar] [CrossRef]
- Román-Quesada, N.; González-Navarro, B.; Izquierdo-Gómez, K.; Jané-Salas, E.; Marí-Roig, A.; Estrugo-Devesa, A.; López-López, J. An analysis of the prevalence of peripheral giant cell granuloma and pyogenic granuloma in relation to a dental implant. BMC Oral Health 2021, 21, 204. [Google Scholar] [CrossRef]
- Baesso, R.C.P.; de Lima Jacy Monteiro Barki, M.C.; de Souza Azevedo, R.; da Costa Fontes, K.B.F.; Pereira, D.L.; Tucci, R.; Pires, F.R.; Picciani, B.L.S. Peripheral giant cell granuloma associated with a dental implant. BMC Oral Health 2019, 19, 283. [Google Scholar] [CrossRef] [PubMed]
- Frumkin, N.; Nashef, R.; Shapira, L.; Wilensky, A. Nonsurgical treatment of recurrent gingival pyogenic granuloma: A case report. Quintessence Int. 2015, 46, 539–544. [Google Scholar] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lomeli Martinez, S.M.; Carrillo Contreras, N.G.; Gómez Sandoval, J.R.; Zepeda Nuño, J.S.; Gomez Mireles, J.C.; Varela Hernández, J.J.; Mercado-González, A.E.; Bayardo González, R.A.; Gutiérrez-Maldonado, A.F. Oral Pyogenic Granuloma: A Narrative Review. Int. J. Mol. Sci. 2023, 24, 16885. https://doi.org/10.3390/ijms242316885
Lomeli Martinez SM, Carrillo Contreras NG, Gómez Sandoval JR, Zepeda Nuño JS, Gomez Mireles JC, Varela Hernández JJ, Mercado-González AE, Bayardo González RA, Gutiérrez-Maldonado AF. Oral Pyogenic Granuloma: A Narrative Review. International Journal of Molecular Sciences. 2023; 24(23):16885. https://doi.org/10.3390/ijms242316885
Chicago/Turabian StyleLomeli Martinez, Sarah Monserrat, Nadia Guadalupe Carrillo Contreras, Juan Ramón Gómez Sandoval, José Sergio Zepeda Nuño, Juan Carlos Gomez Mireles, Juan José Varela Hernández, Ana Esther Mercado-González, Rubén Alberto Bayardo González, and Adrián Fernando Gutiérrez-Maldonado. 2023. "Oral Pyogenic Granuloma: A Narrative Review" International Journal of Molecular Sciences 24, no. 23: 16885. https://doi.org/10.3390/ijms242316885
APA StyleLomeli Martinez, S. M., Carrillo Contreras, N. G., Gómez Sandoval, J. R., Zepeda Nuño, J. S., Gomez Mireles, J. C., Varela Hernández, J. J., Mercado-González, A. E., Bayardo González, R. A., & Gutiérrez-Maldonado, A. F. (2023). Oral Pyogenic Granuloma: A Narrative Review. International Journal of Molecular Sciences, 24(23), 16885. https://doi.org/10.3390/ijms242316885







