A Vicious NGF-p75NTR Positive Feedback Loop Exacerbates the Toxic Effects of Oxidative Damage in the Human Retinal Epithelial Cell Line ARPE-19
Abstract
:1. Introduction
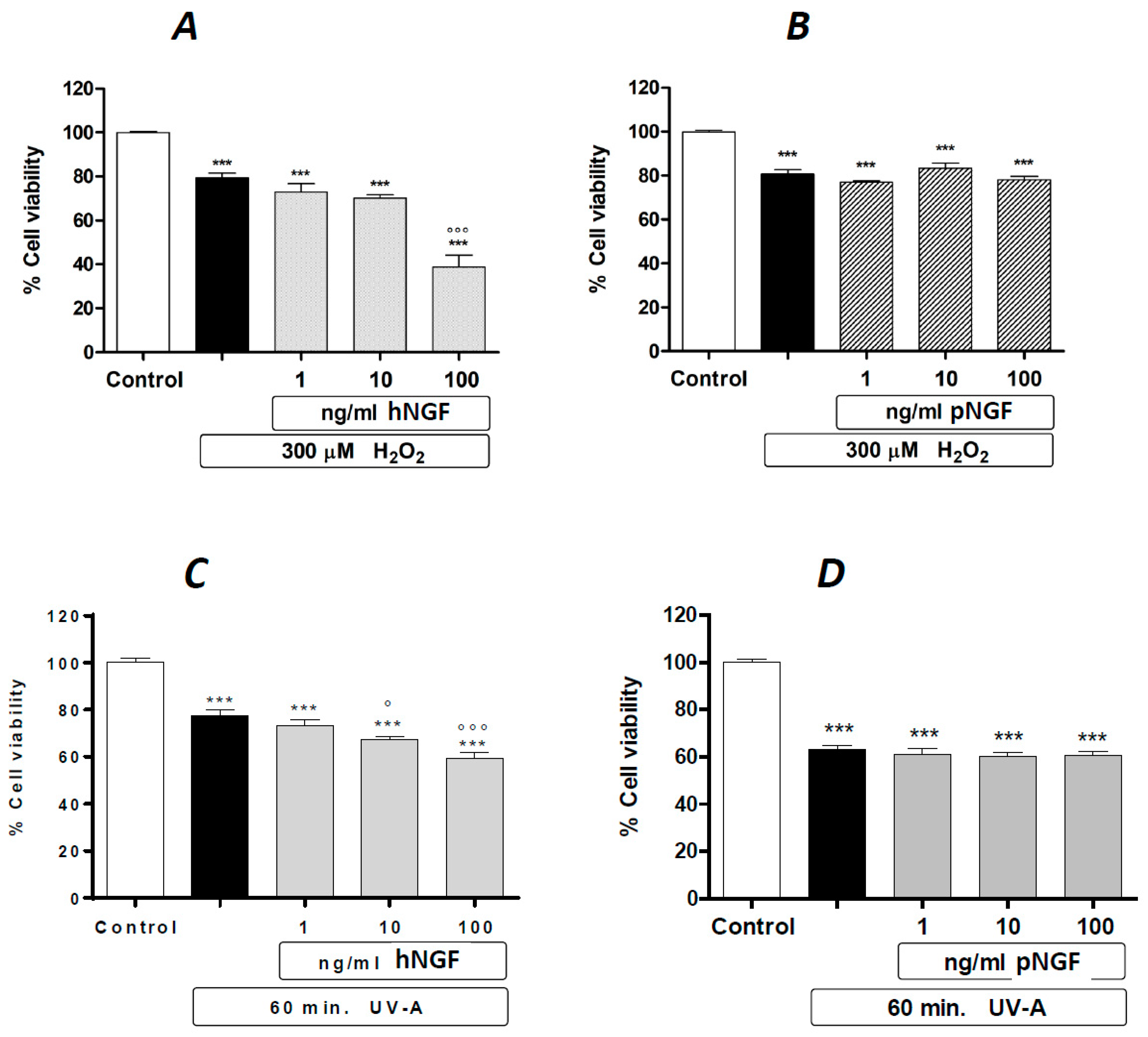
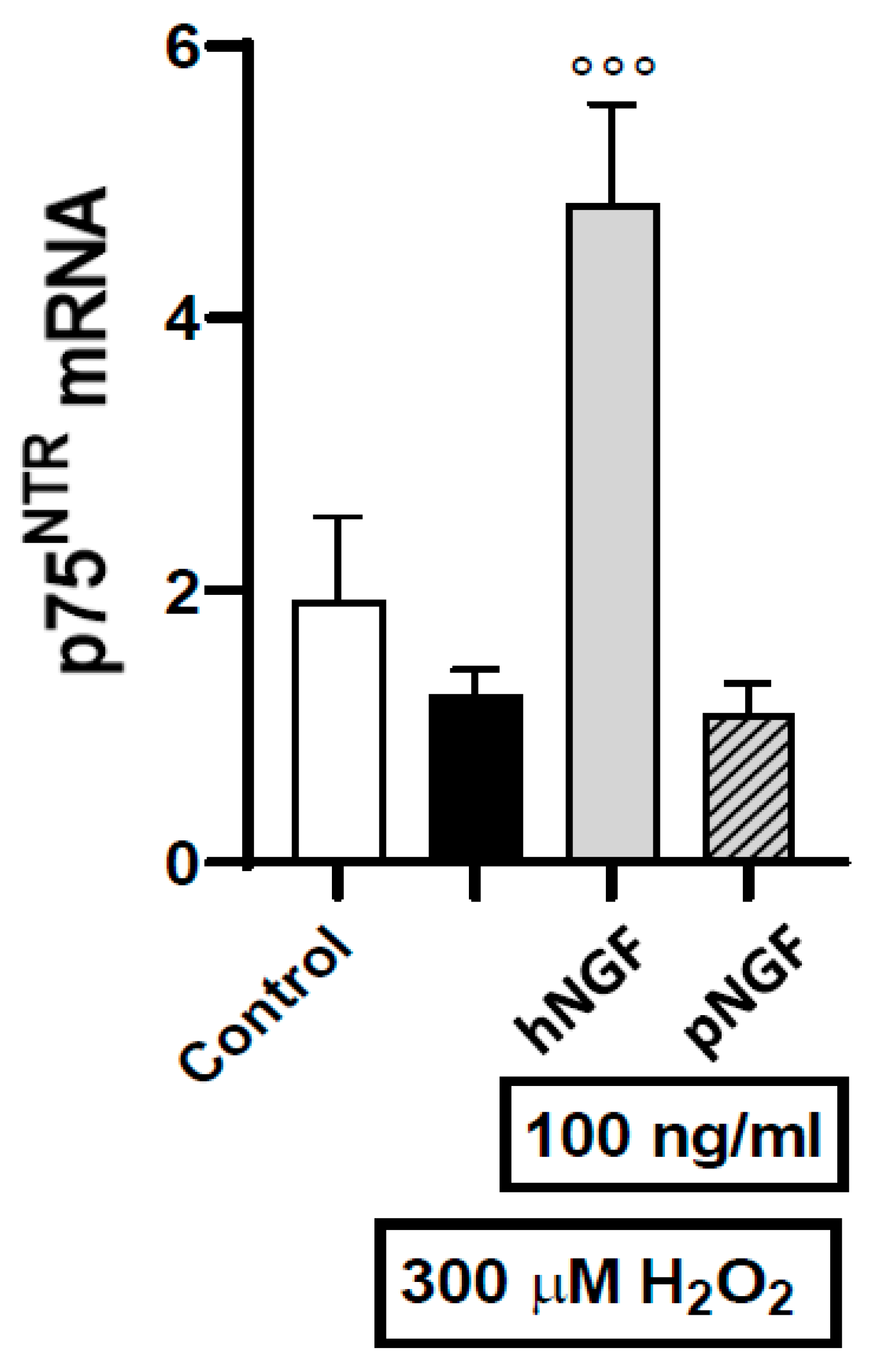
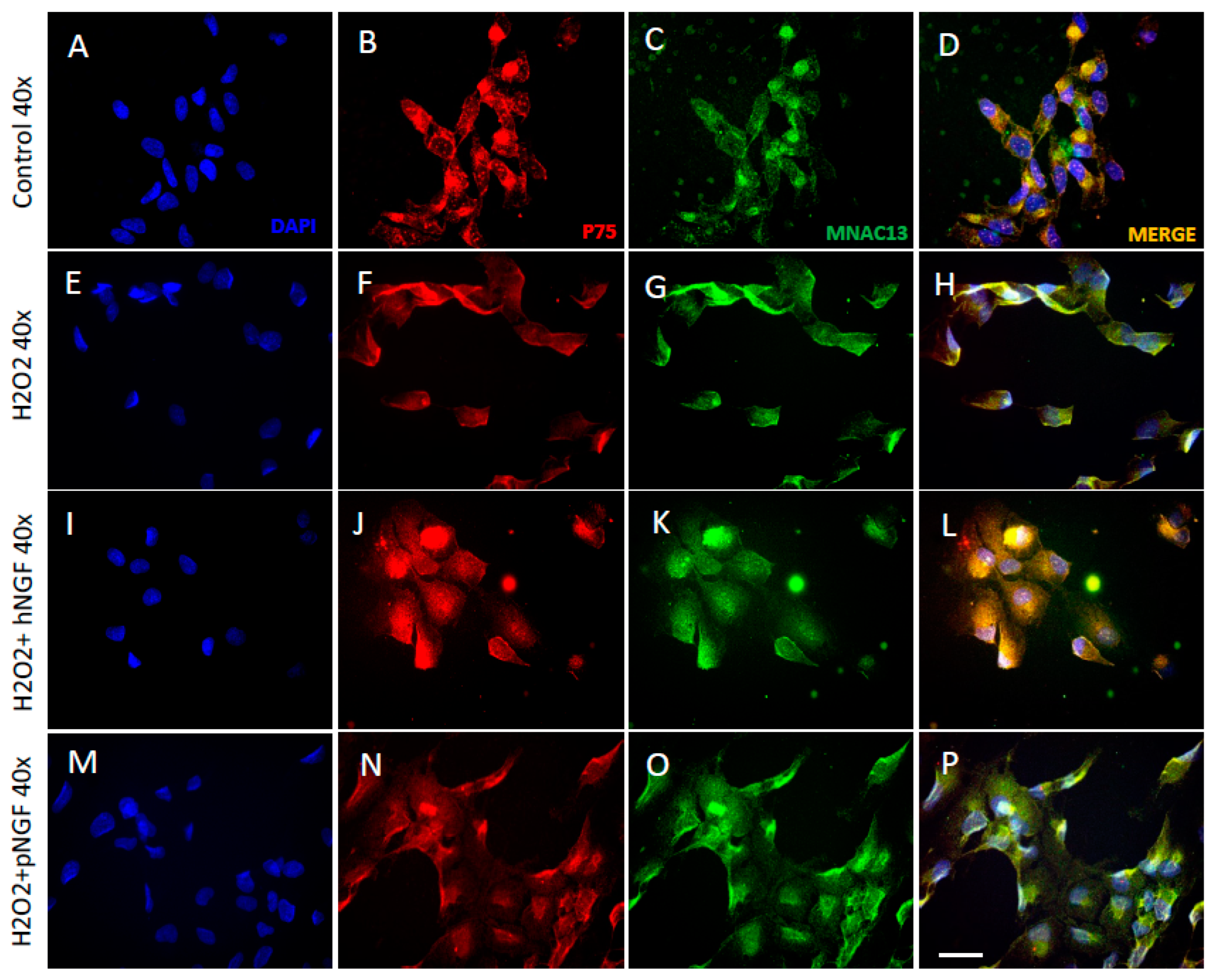
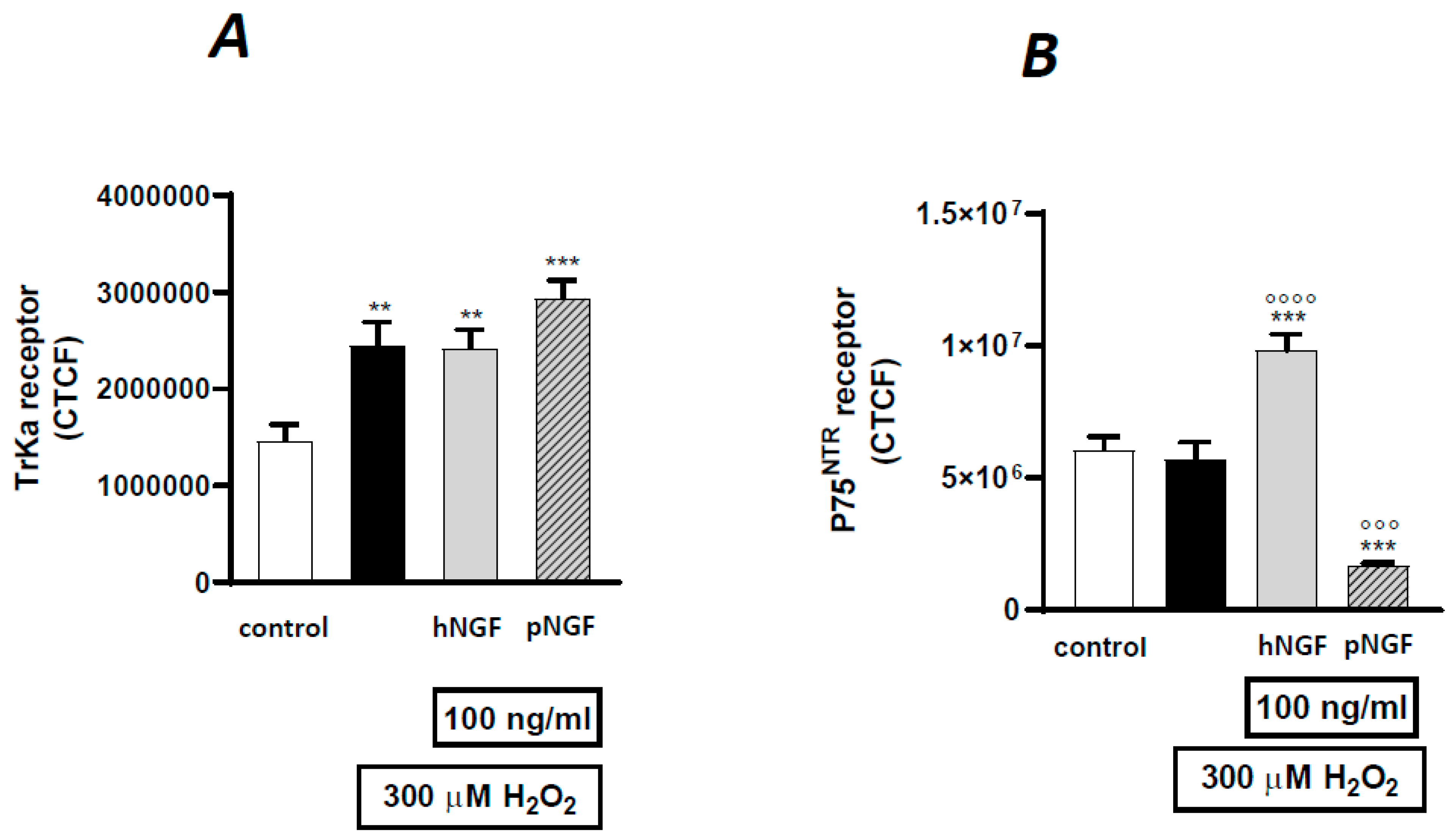
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Cultures and Treatments
4.3. Assessment of Cell Viability
4.4. Real-Time Quantitative RT-PCR (qRT-PCR) Analysis
4.5. Immunofluorescence
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AMD | Age-related macular degeneration |
| ANOVA | Analysis of variance |
| ARPE-19 | Adult retinal pigment epithelial-19 (cell line) |
| DMEM | Dulbecco’s modified Eagle’s medium |
| EC50 | Effective concentration 50 (half-maximal) |
| FBS | Fetal bovine serum |
| hNGF | Human nerve growth factor |
| HSAN V | Hereditary sensory and autonomic neuropathy type V |
| IRD | Inherited retinal degeneration |
| mNGF | Mouse nerve growth factor |
| MTS | (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium) inner salt |
| NGF | Nerve growth factor |
| p75NTR | p75 neurotrophin receptor |
| PBS | Phosphate-buffered saline |
| RNA | Ribonucleic acid |
| RPE | Retinal pigment epithelium |
| RT-PCR | Reverse transcriptase polymerase chain reaction |
| SEM | Standard error of the mean |
| TrkA | Tyrosine kinase A |
| UV | Ultraviolet |
References
- Levi-Montalcini, R. The nerve growth factor 35 years later. Science 1987, 237, 1154–1162. [Google Scholar] [CrossRef]
- Levi-Montalcini, R.; Skaper, S.D.; Dal Toso, R.; Petrelli, L.; Leon, A. Nerve growth factor: From neurotrophin to neurokine. Trends Neurosci. 1996, 19, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Gostynska, N.; Pannella, M.; Rocco, M.L.; Giardino, L.; Aloe, L.; Calzà, L. The pleiotropic molecule NGF regulates the in vitro properties of fibroblasts; keratinocytes; and endothelial cells: Implications for wound healing. Am. J. Physiol.-Cell Physiol. 2020, 318, C360–C371. [Google Scholar] [CrossRef]
- Cattaneo, A.; Capsoni, S. Painless nerve growth factor: A TrkA biased agonist mediating a broad neuroprotection via its actions on microglia cells. Pharmacol. Res. 2019, 139, 17–25. [Google Scholar] [CrossRef]
- Lisi, L.; Marinelli, S.; Ciotti, G.M.P.; Pizzoferrato, M.; Palmerio, F.; Chiavari, M.; Cattaneo, A.; Navarra, P. The effects of painless nerve growth factor on human microglia polarization. Front. Cell. Neurosci. 2022, 16, 969058. [Google Scholar] [CrossRef] [PubMed]
- Apfel, S.C. Nerve growth factor for the treatment of diabetic neuropathy: What went wrong; what went right; and what does the future hold? Int. Rev. Neurobiol. 2002, 50, 393–413. [Google Scholar] [PubMed]
- Pezet, S.; McMahon, S.B. Neurotrophins: Mediators and modulators of pain. Ann. Rev. Neurosci. 2006, 29, 507–538. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.ema.europa.eu/en/documents/product-information/oxervate-epar-product-information_en.pdf (accessed on 15 September 2023).
- ClinicalTrials.gov Identifier: NCT05133180. Available online: https://clinicaltrials.gov/ (accessed on 15 September 2023).
- ClinicalTrials.gov Identifier: NCT05136170. Available online: https://clinicaltrials.gov/ (accessed on 15 September 2023).
- ClinicalTrials.gov Identifier: NCT03019627. Available online: https://clinicaltrials.gov/ (accessed on 15 September 2023).
- ClinicalTrials.gov Identifier: NCT03982368. Available online: https://clinicaltrials.gov/ (accessed on 15 September 2023).
- ClinicalTrials.gov Identifier: NCT02855450. Available online: https://clinicaltrials.gov/ (accessed on 15 September 2023).
- Lambiase, A.; Rama, P.; Bonini, S.; Caprioglio, G.; Aloe, L. Topical treatment with nerve growth factor for corneal neurotrophic ulcers. N. Engl. J. Med. 1998, 338, 1174–1180. [Google Scholar] [CrossRef]
- Generini, S.; Tuveri, M.A.; Matucci Cerinic, M.; Mastinu, F.; Manni, L.; Aloe, L. Topical application of nerve growth factor in human diabetic foot ulcers. A study of three cases. Exp. Clin. Endocrinol. Diabetes 2004, 112, 542–544. [Google Scholar] [CrossRef]
- Falsini, B.; Chiaretti, A.; Rizzo, D.; Piccardi, M.; Ruggiero, A.; Manni, L.; Soligo, M.; Dickmann, A.; Federici, M.; Salerni, A.; et al. Nerve growth factor improves visual loss in childhood optic gliomas: A randomized; double-blind; phase II clinical trial. Brain 2016, 139, 404–414. [Google Scholar] [CrossRef]
- Paoletti, F.; Malerba, F.; Bruni Ercole, B.; Lamba, D.; Cattaneo, A. A comparative analysis of the structural; functional and biological differences between Mouse and Human Nerve Growth Factor. Biochim. Biophys. Acta 2015, 1854, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Covaceuszach, S.; Capsoni, S.; Marinelli, S.; Pavone, F.; Ceci, M.; Ugolini, G.; Vignone, D.; Amato, G.; Paoletti, F.; Lamba, D.; et al. In vitro receptor binding properties of a “painless” NGF mutein; linked to hereditary sensory autonomic neuropathy type V. Biochem. Biophys. Res. Commun. 2010, 391, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Capsoni, S. From genes to pain: Nerve growth factor and hereditary sensory and autonomic neuropathy type V. Eur. J. Neurosci. 2014, 39, 392–400. [Google Scholar] [CrossRef]
- Capsoni, S.; Covaceuszach, S.; Marinelli, S.; Ceci, M.; Bernardo, A.; Minghetti, L.; Ugolini, G.; Pavone, F.; Cattaneo, A. Taking pain out of NGF: A “painless” NGF mutant; linked to hereditary sensory autonomic neuropathy type V; with full neurotrophic activity. PLoS ONE 2011, 6, e17321. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov Identifier: NCT05733572. Available online: https://clinicaltrials.gov/ (accessed on 15 September 2023).
- Clementi, M.E.; Pizzoferrato, M.; Bianchetti, G.; Brancato, A.; Sampaolese, B.; Maulucci, G.; Tringali, G. Cytoprotective Effect of Idebenone through Modulation of the Intrinsic Mitochondrial Pathway of Apoptosis in Human Retinal Pigment Epithelial Cells Exposed to Oxidative Stress Induced by Hydrogen Peroxide. Biomedicines 2022, 10, 503. [Google Scholar] [CrossRef]
- Chao, M.V.; Hempstead, B.L. p75 and Trk: A two-receptor system. Trends Neurosci. 1995, 18, 321–326. [Google Scholar] [CrossRef]
- Kaplan, D.R.; Miller, F.D. Neurotrophin signal transduction in the nervous system. Curr. Opin. Neurobiol. 2000, 10, 381–391. [Google Scholar] [CrossRef]
- Dechant, G.; Barde, Y.A. The neurotrophin receptor p75(NTR): Novel functions and implications for diseases of the nervous system. Nat. Neurosci. 2002, 5, 1131–1136. [Google Scholar] [CrossRef]
- Chao, M.V. Neurotrophins and their receptors: A convergence point for many signalling pathways. Nat. Rev. Neurosci. 2003, 4, 299–309. [Google Scholar] [CrossRef]
- Gupta, S.; Lytvynchuk, L.; Ardan, T.; Studenovska, H.; Faura, G.; Eide, L.; Znaor, L.; Erceg, S.; Stieger, K.; Motlik, J.; et al. Retinal Pigment Epithelium Cell Development: Extrapolating Basic Biology to Stem Cell Research. Biomedicines 2023, 11, 310. [Google Scholar] [CrossRef]
- Manley, A.; Meshkat, B.I.; Jablonski, M.M.; Hollingsworth, T.J. Cellular and Molecular Mechanisms of Pathogenesis Underlying Inherited Retinal Dystrophies. Biomolecules 2023, 13, 271. [Google Scholar] [CrossRef] [PubMed]
- Maurya, M.; Bora, K.; Blomfield, A.K.; Pavlovich, M.C.; Huang, S.; Liu, C.H.; Chen, J. Oxidative stress in retinal pigment epithelium degeneration: From pathogenesis to therapeutic targets in dry age-related macular degeneration. Neural Regen. Res. 2023, 18, 2173–2181. [Google Scholar] [PubMed]
- Pedrotti, E.; Bonacci, E.; Chierego, C.; De Gregorio, A.; Cozzini, T.; Brighenti, T.; Caldarella, G.; Pastore, G.; Fasolo, A.; Marchini, G. Eight months follow-up of corneal nerves and sensitivity after treatment with cenegermin for neurotrophic keratopathy. Orphanet J. Rare Dis. 2022, 17, 63. [Google Scholar] [CrossRef] [PubMed]
- Coassin, M.; Lambiase, A.; Sposato, V.; Micera, A.; Bonini, S.; Aloe, L. Retinal p75 and bax overexpression is associated with retinal ganglion cells apoptosis in a rat model of glaucoma. Graefe’s Arch. Clin. Exp. Ophthalmol. 2008, 246, 1743–1749. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Dergham, P.; Nedev, H.; Xu, J.; Galan, A.; Rivera, J.C.; Zhi Hua, S.; Mehta, H.M.; Woo, S.B.; Sarunic, M.V.; et al. Chronic and acute models of retinal neurodegeneration TrkA activity are neuroprotective whereas p75NTR activity is neurotoxic through a paracrine mechanism. J. Biol. Chem. 2010, 285, 39392–39400. [Google Scholar] [CrossRef]
- Kraemer, B.R.; Snow, J.P.; Vollbrecht, P.; Pathak, A.; Valentine, W.M.; Deutch, A.Y.; Carter, B.D. A role for the p75 neurotrophin receptor in axonal degeneration and apoptosis induced by oxidative stress. J. Biol. Chem. 2014, 289, 21205–21216. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Sima, A.; Lee, J.; Brachet, P.; Dicou, E. Nerve growth factor (NGF); proNGF and NGF receptor-like immunoreactivity in BB rat retina. Brain Res. 1990, 523, 11–15. [Google Scholar] [CrossRef]
- Kociok, N.; Heppekausen, H.; Schraermeyer, U.; Esser, P.; Thumann, G.; Grisanti, S.; Heimann, K. The mRNA expression of cytokines and their receptors in cultured iris pigment epithelial cells: A comparison with retinal pigment epithelial cells. Exp. Eye Res. 1998, 67, 237–250. [Google Scholar] [CrossRef]
- Sacchetti, M.; Mantelli, F.; Rocco, M.L.; Micera, A.; Brandolini, L.; Focareta, L.; Pisano, C.; Aloe, L.; Lambiase, A. Recombinant Human Nerve Growth Factor Treatment Promotes Photoreceptor Survival in the Retinas of Rats with Retinitis Pigmentosa. Curr. Eye Res. 2017, 42, 1064–1068. [Google Scholar] [CrossRef]
- Rocco, M.L.; Balzamino, B.O.; Petrocchi Passeri, P.; Micera, A.; Aloe, L. Effect of Purified Murine NGF on Isolated Photoreceptors of a Rodent Developing Retinitis Pigmentosa. PLoS ONE 2015, 10, e0124810. [Google Scholar] [CrossRef]
- Cocchiaro, P.; Di Donato, V.; Rubbini, D.; Mastropasqua, R.; Allegretti, M.; Mantelli, F.; Aramini, A.; Brandolini, L. Intravitreal Administration of rhNGF Enhances Regenerative Processes in a Zebrafish Model of Retinal Degeneration. Front. Pharmacol. 2022, 13, 822359. [Google Scholar] [CrossRef]
- Campochiaro, P.A.; Iftikhar, M.; Hafiz, G.; Akhlaq, A.; Tsai, G.; Wehling, D.; Lu, L.; Wall, G.M.; Singh, M.S.; Kong, X. Oral N-acetylcysteine improves cone function in retinitis pigmentosa patients in phase I trial. J. Clin. Investig. 2020, 130, 1527–1541. [Google Scholar] [CrossRef] [PubMed]
- Kularatne, R.N.; Bulumulla, C.; Catchpole, T.; Takacs, A.; Christie, A.; Stefan, M.C.; Csaky, K.G. Protection of human retinal pigment epithelial cells from oxidative damage using cysteine prodrugs. Free. Radic. Biol. Med. 2020, 152, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, B.A.; Fliesler, S.J. Reassessing the suitability of ARPE-19 cells as a valid model of native RPE biology. Exp. Eye Res. 2022, 219, 109046. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tringali, G.; Pizzoferrato, M.; Lisi, L.; Marinelli, S.; Buccarello, L.; Falsini, B.; Cattaneo, A.; Navarra, P. A Vicious NGF-p75NTR Positive Feedback Loop Exacerbates the Toxic Effects of Oxidative Damage in the Human Retinal Epithelial Cell Line ARPE-19. Int. J. Mol. Sci. 2023, 24, 16237. https://doi.org/10.3390/ijms242216237
Tringali G, Pizzoferrato M, Lisi L, Marinelli S, Buccarello L, Falsini B, Cattaneo A, Navarra P. A Vicious NGF-p75NTR Positive Feedback Loop Exacerbates the Toxic Effects of Oxidative Damage in the Human Retinal Epithelial Cell Line ARPE-19. International Journal of Molecular Sciences. 2023; 24(22):16237. https://doi.org/10.3390/ijms242216237
Chicago/Turabian StyleTringali, Giuseppe, Michela Pizzoferrato, Lucia Lisi, Silvia Marinelli, Lucia Buccarello, Benedetto Falsini, Antonino Cattaneo, and Pierluigi Navarra. 2023. "A Vicious NGF-p75NTR Positive Feedback Loop Exacerbates the Toxic Effects of Oxidative Damage in the Human Retinal Epithelial Cell Line ARPE-19" International Journal of Molecular Sciences 24, no. 22: 16237. https://doi.org/10.3390/ijms242216237
APA StyleTringali, G., Pizzoferrato, M., Lisi, L., Marinelli, S., Buccarello, L., Falsini, B., Cattaneo, A., & Navarra, P. (2023). A Vicious NGF-p75NTR Positive Feedback Loop Exacerbates the Toxic Effects of Oxidative Damage in the Human Retinal Epithelial Cell Line ARPE-19. International Journal of Molecular Sciences, 24(22), 16237. https://doi.org/10.3390/ijms242216237







