Intra-Articular Injection of Botulinum Toxin for the Treatment of Knee Osteoarthritis: A Systematic Review of Randomized Controlled Trials
Abstract
1. Introduction
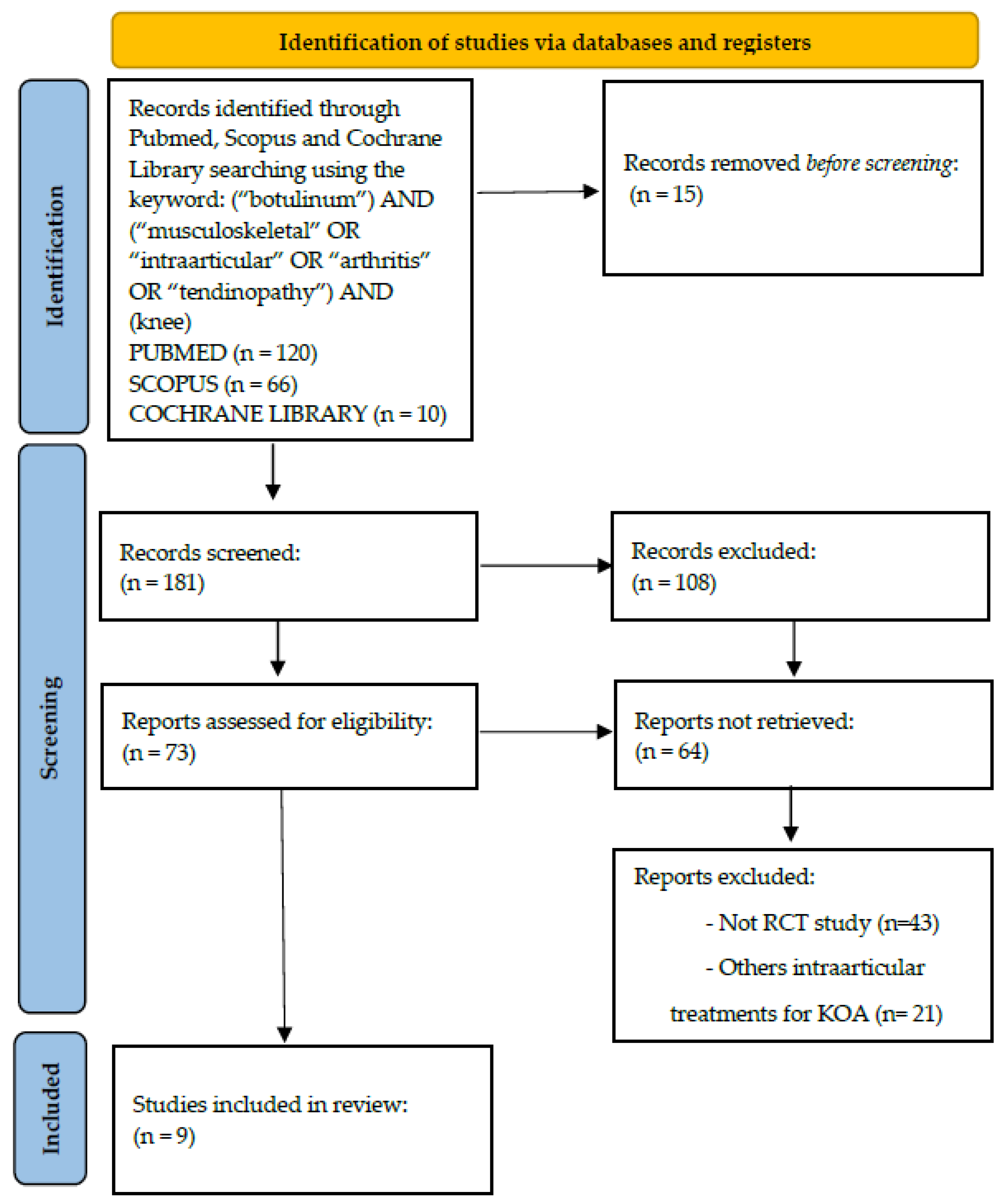
2. Methods
3. Results and Discussion
3.1. Results
3.1.1. Study Design and Quality
| Publication | Study Design | Pathology | Score | Patients Features | BoNT-A Preparation Method | Therapeutic Protocol and F-up | Results | Overall Performance of BTX |
|---|---|---|---|---|---|---|---|---|
| Rezasoltani et al., 2021 [28] | Single-blind RCT (BTX injection vs. physical therapy [PT]) | knee osteoarthritis | VAS, KOOS | 50 (25 vs. 25) Age: 77.7 ± 7.3 y 63.0 ± 8.0 y Sex: F 73%: F 80% | V & Conc: 100 IU of BTX (250 units from disport brand) in 5 mL of SS | F-up at 1, 3, and 6 mo | At 1 mo F-up, VAS score and all KOOS subscales were improved in the BTX group in comparison to the PT group. The use of BTX can reduce pain and improve the function and quality of life in patients with KOA. | BTX+ |
| Rezasoltani et al., 2020 [5] | RCT (Physical therapy vs. BTX injection vs. Hyaluronic acid vs. Dextrose prolotherapy) | knee osteoarthritis | VAS, Persian version of KOOS | 120 (30 vs. 30 vs. 30 vs. 30) Age: 70 (±6.3) y 67.7 (±7.3) y 66.1 (±9.1) y 64.8 (±5.8) y Sex: 12 M:18 F 8 M:22 F 14 M:16 F 11 M:19 F | V & Conc: 250 units of Dysport, equivalent to 100 units of BoNT/A (Dysport, Abobotulinumtoxin A), diluted with 5 mL of SS | F-up at baseline, and in 1 wk, 4 wks, and 3 mo | An IA injection of BTX or dextrose prolotherapy is effective first-line treatments. In the next place stands physical therapy particularly if the patient is not willing to continue regular exercise programs. The study was not very supportive of IA injection of hyaluronic acid as an effective treatment of KOA | BTX+ |
| Mendes et al., 2019 [20] | Double-blind RCT (BTX injection vs. TH injection vs. placebo) | knee osteoarthritis | VASm, VASr, WOMAC, 6-min walk test, TUG, SF-36, ROM of knee and US measurement of synovial hypertrophy. | 105 (35 vs. 35 vs. 35) Age: (64.2 ± 6.9 y) Sex: 9 M:96 F | V & Conc: 100 IU of BTX in 2 mL of SS 0.9% | F-up at baseline and at 4, 8, and 12 wks | IA injection with TH in primary KOA had a higher effectiveness than that with BTX or SS in the short-term assessment (4 wks) for VASm, WOMAC, and US measurement of synovial hypertrophy | BTX- |
| McAlindon et al., 2018 [11] | Double-blind RCT (BTX injection vs. placebo) | knee osteoarthritis | NRS, WOMAC pain and physical function scores, PGIC | 176 (44 vs. 43 vs. 89) Age: 60.7± 8.3 y 60.2 ± 8.4 y 61.1± 7.8 y Sex: 30 (68.2%) F 26 (60.5 %) F 51 (57.3%) F | V & Conc: 400 or 200 IU of BTX in a total volume of 2 mL | F-up at at wks 1 and 4 and every 4 wks thereafter to wk 24 were undertaken | There were no significant differences between IA BTX and placebo in reducing WOMAC pain and physical function scores at wk 8 compared with baseline, in patients with KOA and nociceptive pain | BTX= |
| Bao et al., 2018 [30] | Single-blind RCT (BTX injection + therapeutic exercises vs. Hyaluronate injection + therapeutic exercises vs. placebo + therapeutic exercises) | knee osteoarthritis | VAS, WOMAC and SF-36 | 60 (20 vs. 20 vs. 20) Age: 66.4 ± 3.49 y 66.0 ± 2.09 y 65.3 ± 3.52 y Sex: 10 M:10 F 13 M:7 F 9 M:11 F | V & Conc: 100 IU BTX (Botox; Allergan Inc., Irvine, KY, USA) diluted with 2.5 mL preservative- free 0.9% SS | F-up at baseline, and at the end of the 4th and 8th wks | At the end of the 4th and 8th wks, WOMAC and VAS scores were higher in the CG. Therapeutic exercise plus BTX or hyaluronate injection can significantly reduce pain and improve knee functioning in patients with KOA. BTX plus therapeutic exercise appears to be more effective. | BTX+ |
| Hsieh et al., 2016 [29] | Single-blind RCT (BTX injection vs. education only for arthritis care) | knee osteoarthritis | VAS, LEQUESNE and WOMAC indexes | 46 (21 vs. 20) Age: 67.82 ± 9.06 y 68.06 ± 4.53 y Sex: 32: F (52.5%) 30: F (50.0%) | V & Conc: 100 IU of BTX (Botox, Allergan Inc., Parsippany, NJ, USA) diluted with 2 mL of preservative- free 0.9% SS | F-up at 1 wk and 6 mo | The pain VAS score in the BTX group significantly decreased at 1 wk and at 6 mo post treatment but not in the CG. Significant differences for the between-group comparison were observed in WOMAC and Lequesne indexes at 6 mo f-up. The IA injection of BTX provided pain relief and improved functional abilities in patients with KOA in both the short- and long-term f-up | BTX+ |
| Arendt-Nielsen et al., 2016 [25] | Double-blind RCT (BTX injection vs. placebo) | knee osteoarthritis | NRS, WOMAC, ADP, GIC | 121 (61 vs. 60) Age: 62.5 ± 8.6 y 62.1 ± 8.6 y Sex: 23:M 15:F 23:M 14:F | V & Conc: 200 IU of BTX containing 2 mL of 0.9% SS | F-up at baseline and weeks 4, 8, and 12 | The nociceptive group showed significant improvement after IA BTX at wk 8 for all WOMAC outcomes, ADP at wks 9 and 10, and patient GIC at wk 12. IA BTX given to patients with nociceptive KOA reduced pain sensitization together with improvement in pain and function | BTX= |
| Boon et al., 2010 [27] | Double-blind RCT (Low-dose of BTX injection vs. High-dose of BTX injection vs. CS injection) | knee osteoarthritis | VAS, WOMAC, SF-36, PGA, 40-m timed walk | 60 (20 vs. 20 vs. 20) Age: 64.1 ± 13.4 y 61.2 ± 9.4 y 60.8 ± 10.1 y Sex: 9/11: M (45%) 9/11: M (45%) 7/13: M (35%) | V & Conc: 100 IU of BTX, 200 IU of BTX | F-up at baseline, 4, 8, 12, and 26 wks | At 8 wks, VAS score decreased within each group but only reached statistical significance in the low-dose BTX group. All groups showed statistically significant improvements in all WOMAC scores at 8 wks. Possible role for BTX as a treatment option for moderate pain and functional impairment secondary to KOA. | Low dose of BTX+ |
| Mahowald et al., 2009 [26] | RCT (BTX+ Lidocaine injection vs. saline placebo + Lidocaine) | shoulder and knee osteoarthritis | VAS, WOMAC, SF-MPQ, SF-36 | 78 (36 vs. 42) (Shoulder study vs. Knee study) Age: NA Sex: NA | V & Conc: 25–100 IU of BTX with 2 cc of 2% Bupivacaine | F-up at baseline, at 1 and 3 mo | In the shoulder study, IA-BTX produced a significant decrease in shoulder pain severity at 1 mo that was also significantly better than the non-significant change after IA-Saline placebo. In the knee study IA-BTX produced a significant 48% decrease in SF-MPQ at 1 mo that was still significant at 3 mo after injection. There was a strong placebo response in 1/3 of those but the decrease in pain severity was not significant. | BTX= |
3.1.2. Patients and Evaluation Methods
3.1.3. Treatment
3.1.4. Complications
3.1.5. Reported Clinical Outcome
3.2. Discussion
Limitations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Neogi, T. The epidemiology and impact of pain in osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.E. Osteoarthritis year in review 2017: Clinical. Osteoarthr. Cartilage. 2018, 26, 319–325. [Google Scholar] [CrossRef] [PubMed]
- De Sire, A.; Marotta, N.; Ferrillo, M.; Agostini, F.; Sconza, C.; Lippi, L.; Respizzi, S.; Giudice, A.; Invernizzi, M.; Ammendolia, A. Oxygen-Ozone Therapy for Reducing Pro-Inflammatory Cytokines Serum Levels in Musculoskeletal and Temporomandibular Disorders: A Comprehensive Review. Int. J. Mol. Sci. 2022, 23, 2528. [Google Scholar] [CrossRef] [PubMed]
- Sconza, C.; Respizzi, S.; Virelli, L.; Vandenbulcke, F.; Iacono, F.; Kon, E.; Di Matteo, B. Oxygen-Ozone Therapy for the Treatment of Knee Osteoarthritis: A Systematic Review of Randomized Controlled Trials. Arthroscopy 2020, 36, 277–286. [Google Scholar] [CrossRef]
- Rezasoltani, Z.; Azizi, S.; Najafi, S.; Sanati, E.; Dadarkhah, A.; Abdorrazaghi, F. Physical therapy, intra-articular dextrose prolotherapy, botulinum neurotoxin, and hyaluronic acid for knee osteoarthritis: Randomized clinical trial. Int. J. Rehabil. Res. 2020, 43, 219–227. [Google Scholar] [CrossRef]
- Oliveria, S.A.; Felson, D.T.; Reed, J.I.; Cirillo, P.A.; Walker, A.M. Incidence of symptomatic hand, hip, and knee osteoarthritis among patients in a health maintenance organization. Arthritis Rheum. 1995, 38, 1134–1141. [Google Scholar] [CrossRef]
- Tang, L.; Jia, P.; Zhao, L.; Kang, D.; Luo, Y.; Liu, J.; Li, L.; Zheng, H.; Li, Y.; Li, N.; et al. Acupuncture treatment for knee osteoarthritis with sensitive points: Protocol for a multicentre randomised controlled trial. BMJ Open 2018, 8, e023838. [Google Scholar] [CrossRef]
- Dantas, L.O.; Salvini, T.F.; McAlindon, T.E. Knee osteoarthritis: Key treatments and implications for physical therapy. Braz. J. Phys. Ther. 2021, 25, 135–146. [Google Scholar] [CrossRef]
- Letizia Mauro, G.; Scaturro, D.; Gimigliano, F.; Paoletta, M.; Liguori, S.; Toro, G.; Iolascon, G.; Moretti, A. Physical Agent Modalities in Early Osteoarthritis: A Scoping Review. Medicina 2021, 57, 1165. [Google Scholar] [CrossRef] [PubMed]
- Coxib and Traditional NSAID Trialists’ (CNT) Collaboration; Bhala, N.; Emberson, J.; Merhi, A.; Abramson, S.; Arber, N.; Baron, J.A.; Bombardier, C.; Cannon, C.; Farkouh, M.E.; et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet 2013, 382, 769–779. [Google Scholar] [CrossRef]
- McAlindon, T.E.; Schmidt, U.; Bugarin, D.; Abrams, S.; Geib, T.; DeGryse, R.E.; Kim, K.; Schnitzer, T.J. Efficacy and safety of single-dose onabotulinumtoxinA in the treatment of symptoms of osteoarthritis of the knee: Results of a placebo-controlled, double-blind study. Osteoarthr. Cartil. 2018, 26, 1291–1299. [Google Scholar] [CrossRef]
- Scaturro, D.; Vitagliani, F.; Terrana, P.; Cuntrera, D.; Falco, V.; Tomasello, S.; Letizia Mauro, G. Intra-Articular Hybrid Hyaluronic Acid Injection Treatment in Overweight Patients with Knee Osteoarthritis: A Single-Center, Open-Label, Prospective Study. Appl. Sci. 2021, 11, 8711. [Google Scholar] [CrossRef]
- Fusco, G.; Gambaro, F.M.; Di Matteo, B.; Kon, E. Injections in the osteoarthritic knee: A review of current treatment options. EFORT Open Rev. 2021, 6, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Di Matteo, B.; Polignano, A.; Onorato, F.; La Porta, A.; Iacono, F.; Bonanzinga, T.; Raspugli, G.; Marcacci, M.; Kon, E. Knee Intraosseous Injections: A Systematic Review of Clinical Evidence of Different Treatment Alternatives. Cartilage 2021, 13 (Suppl. S1), 1165S–1177S. [Google Scholar] [CrossRef] [PubMed]
- Kon, E.; Di Matteo, B. Editorial Commentary: Minimally Invasive Strategies for Osteoarthritis: From Platelets to Mesenchymal Stem Cells. Arthroscopy 2021, 37, 2258–2261. [Google Scholar] [CrossRef] [PubMed]
- Eisele, K.H.; Fink, K.; Vey, M.; Taylor, H.V. Studies on the dissociation of botulinum neurotoxin type A complexes. Toxicon 2011, 57, 555–565. [Google Scholar] [CrossRef]
- Intiso, D. Therapeutic use of botulinum toxin in neurorehabilitation. J. Toxicol. 2012, 2012, 802893. [Google Scholar] [CrossRef]
- Poulain, B. La neurotoxine botulinique [Botulinum neurotoxin]. Rev. Neurol. 2010, 166, 7–20. [Google Scholar] [CrossRef]
- Kerscher, M.; Wanitphakdeedecha, R.; Trindade de Almeida, A.; Maas, C.; Frevert, J. IncobotulinumtoxinA: A Highly Purified and Precisely Manufactured Botulinum Neurotoxin Type A. J. Drugs Dermatol. 2019, 18, 52–57. [Google Scholar]
- Mendes, J.G.; Natour, J.; Nunes-Tamashiro, J.C.; Toffolo, S.R.; Rosenfeld, A.; Furtado, R.N.V. Comparison between intra-articular Botulinum toxin type A, corticosteroid, and saline in knee osteoarthritis: A randomized controlled trial. Clin. Rehabil. 2019, 33, 1015–1026. [Google Scholar] [CrossRef]
- Aoki, K.R. Review of a proposed mechanism for the antinociceptive action of botulinum toxin type A. Neurotoxicology 2005, 26, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.C.; Wakita, M.; Xie, D.J.; Yamaga, T.; Iwata, S.; Torii, Y.; Harakawa, T.; Ginnaga, A.; Kozaki, S.; Akaike, N. Inhibition of membrane Na+ channels by A type botulinum toxin at femtomolar concentrations in central and peripheral neurons. J. Pharmacol. Sci. 2012, 118, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.Y.; Kim, M.J.; Ju, J.S.; Park, S.K.; Lee, C.G.; Kim, S.T.; Bae, Y.C.; Ahn, D.K. Antinociceptive Effects of Botulinum Toxin Type A on Trigeminal Neuropathic Pain. J. Dent. Res. 2016, 95, 1183–1190. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.A.; Mahowald, M.L.; Noorbaloochi, S. Intraarticular botulinum toxin A for refractory painful total knee arthroplasty: A randomized controlled trial. J. Rheumatol. 2010, 37, 2377–2386. [Google Scholar] [CrossRef]
- Arendt-Nielsen, L.; Jiang, G.L.; DeGryse, R.; Turkel, C.C. Intra-articular onabotulinumtoxinA in osteoarthritis knee pain: Effect on human mechanistic pain biomarkers and clinical pain. Scand. J. Rheumatol. 2017, 46, 303–316. [Google Scholar] [CrossRef]
- Mahowald, M.L.; Krug, H.E.; Singh, J.A.; Dykstra, D. Intra-articular Botulinum Toxin Type A: A new approach to treat arthritis joint pain. Toxicon 2009, 54, 658–667. [Google Scholar] [CrossRef]
- Boon, A.J.; Smith, J.; Dahm, D.L.; Sorenson, E.J.; Larson, D.R.; Fitz-Gibbon, P.D.; Dykstra, D.D.; Singh, J.A. Efficacy of intra-articular botulinum toxin type A in painful knee osteoarthritis: A pilot study. PM R 2010, 2, 268–276. [Google Scholar] [CrossRef]
- Rezasoltani, Z.; Dadarkhah, A.; Tabatabaee, S.M.; Abdorrazaghi, F.; Morteza, M.K.; Mofrad, R.K. Therapeutic Effects of Intra-articular Botulinum Neurotoxin Versus Physical Therapy in Knee Osteoarthritis. Anesth. Pain Med. 2021, 11, e112789. [Google Scholar] [CrossRef]
- Hsieh, L.F.; Wu, C.W.; Chou, C.C.; Yang, S.W.; Wu, S.H.; Lin, Y.J.; Hsu, W.C. Effects of Botulinum Toxin Landmark-Guided Intra-articular Injection in Subjects With Knee Osteoarthritis. PM R 2016, 8, 1127–1135. [Google Scholar] [CrossRef]
- Bao, X.; Tan, J.W.; Flyzik, M.; Ma, X.C.; Liu, H.; Liu, H.Y. Effect of therapeutic exercise on knee osteoarthritis after intra-articular injection of botulinum toxin type A, hyaluronate or saline: A randomized controlled trial. J. Rehabil. Med. 2018, 50, 534–541. [Google Scholar] [CrossRef]
- Mahowald, M.L.; Singh, J.A.; Dykstra, D. Long term effects of intra-articular botulinum toxin A for refractory joint pain. Neurotox. Res. 2006, 9, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.A.; Mahowald, M.L.; Kushnaryov, A.; Goelz, E.; Dykstra, D. Repeat injections of intra-articular botulinum toxin a for the treatment of chronic arthritis joint pain. J. Clin. Rheumatol. 2009, 15, 35–38. [Google Scholar] [CrossRef]
- Guo, G.X.; Zhu, Q.G.; Chen, Z.Y.; Gu, Y.J.; Yao, F.; Fang, M. Complex mechanisms of chronic pain in knee osteoarthritis identified by neuroimaging technology. Sheng Li Xue Bao 2021, 73, 423–432. (In Chinese) [Google Scholar]
- Woolf, C.J.; Salter, M.W. Neuronal plasticity: Increasing the gain in pain. Science 2000, 288, 1765–1769. [Google Scholar] [CrossRef] [PubMed]
- Saito, T. Neurogenic inflammation in osteoarthritis of the knee. Mod. Rheumatol. 2003, 13, 301–304. [Google Scholar] [CrossRef]
- Sconza, C.; Leonardi, G.; Kon, E.; Respizzi, S.; Massazza, G.; Marcacci, M.; Di Matteo, B. Oxygen-ozone therapy for the treatment of low back pain: A systematic review of randomized controlled trials. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 6034–6046. [Google Scholar] [CrossRef] [PubMed]
- Sconza, C.; Braghetto, G.; Respizzi, S.; Morenghi, E.; Kon, E.; Di Matteo, B. Ultrasound-guided periradicular oxygen-ozone injections as a treatment option for low back pain associated with sciatica. Int. Orthop. 2021, 45, 1239–1246. [Google Scholar] [CrossRef]
- Aoki, K.R. Evidence for antinociceptive activity of botulinum toxin type A in pain management. Headache 2003, 43 (Suppl. S1), S9–S15. [Google Scholar] [CrossRef] [PubMed]
- Aoki, K.R.; Francis, J. Updates on the antinociceptive mechanism hypothesis of botulinum toxin A. Park. Relat. Disord. 2011, 17 (Suppl. S1), S28–S33. [Google Scholar] [CrossRef] [PubMed]
- Deveza, L.A.; Melo, L.; Yamato, T.P.; Mills, K.; Ravi, V.; Hunter, D.J. Knee osteoarthritis phenotypes and their relevance for outcomes: A systematic review. Osteoarthr. Cartil. 2017, 25, 1926–1941. [Google Scholar] [CrossRef]
- Monticone, M.; Sconza, C.; Portoghese, I.; Nishigami, T.; Wand, B.M.; Sorrentino, G.; Lemorini, G.; Respizzi, S.; Giordano, A.; Franchignoni, F. Cross-cultural adaptation, reliability and validity of the Fremantle Knee Awareness Questionnaire in Italian subjects with painful knee osteoarthritis. Health Qual Life Outcomes 2021, 19, 114. [Google Scholar] [CrossRef] [PubMed]
| Publication | Random Sequence Generation | Allocation Concealment | Selective Reporting | Other Bias | Blinding of Participants and Personnel | Blinding of Outcome Assessment | Incomplete Outcome Data | AHRQ Standard |
|---|---|---|---|---|---|---|---|---|
| Rezasoltani et al., 2021 [28] | Low | Low | Low | Unclear | High | Low | Low | Fair |
| Rezasoltani et al., 2020 [5] | High | Unclear | Unclear | Unclear | High | High | Low | Poor |
| Mendes et al., 2019 [20] | Low | Low | Low | Low | Low | Low | Low | Good |
| McAlindon et al., 2018 [11] | High | High | Unclear | High | Low | Low | Unclear | Poor |
| Bao et al., 2018 [30] | Low | Unclear | Unclear | Unclear | High | Unclear | Unclear | Poor |
| Hsieh et al., 2016 [29] | Low | Low | Unclear | High | High | Unclear | Low | Fair |
| Arendt-Nielsen et al., 2016 [25] | Low | Low | Unclear | Unclear | Low | Low | Unclear | Fair |
| Boon et al., 2010 [27] | Low | Low | Low | Low | Low | Low | Low | Good |
| Mahowald et al., 2009 [26] | Unclear | Unclear | Unclear | High | Unclear | Unclear | Unclear | Poor |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sconza, C.; Leonardi, G.; Carfì, C.; Kon, E.; Respizzi, S.; Scaturro, D.; Letizia Mauro, G.; Massazza, G.; Di Matteo, B. Intra-Articular Injection of Botulinum Toxin for the Treatment of Knee Osteoarthritis: A Systematic Review of Randomized Controlled Trials. Int. J. Mol. Sci. 2023, 24, 1486. https://doi.org/10.3390/ijms24021486
Sconza C, Leonardi G, Carfì C, Kon E, Respizzi S, Scaturro D, Letizia Mauro G, Massazza G, Di Matteo B. Intra-Articular Injection of Botulinum Toxin for the Treatment of Knee Osteoarthritis: A Systematic Review of Randomized Controlled Trials. International Journal of Molecular Sciences. 2023; 24(2):1486. https://doi.org/10.3390/ijms24021486
Chicago/Turabian StyleSconza, Cristiano, Giulia Leonardi, Carla Carfì, Elizaveta Kon, Stefano Respizzi, Dalila Scaturro, Giulia Letizia Mauro, Giuseppe Massazza, and Berardo Di Matteo. 2023. "Intra-Articular Injection of Botulinum Toxin for the Treatment of Knee Osteoarthritis: A Systematic Review of Randomized Controlled Trials" International Journal of Molecular Sciences 24, no. 2: 1486. https://doi.org/10.3390/ijms24021486
APA StyleSconza, C., Leonardi, G., Carfì, C., Kon, E., Respizzi, S., Scaturro, D., Letizia Mauro, G., Massazza, G., & Di Matteo, B. (2023). Intra-Articular Injection of Botulinum Toxin for the Treatment of Knee Osteoarthritis: A Systematic Review of Randomized Controlled Trials. International Journal of Molecular Sciences, 24(2), 1486. https://doi.org/10.3390/ijms24021486











