Multiomics Based Association Mapping in Wheat Reveals Genetic Architecture of Quality and Allergenic Related Proteins
Abstract
1. Introduction
2. Results
3. Discussion
3.1. Major QTLs Identified for 54 Proteins
3.2. QTLs for Important Gluten Proteins
3.3. Possibility to Breed for Low Allergen Content
4. Materials and Methods
4.1. Plant Material and Field Experiments
4.2. LFQ Proteomic
4.3. Statistical Analysis of Phenotypic Data
4.4. Genomic Data and Genotyping
4.5. Genome-Wide Association Study (GWAS)
4.6. Verification of Two Major QTLs
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shewry, P.R.; Hey, S. The contribution of wheat to human diet and health. Food Energy Secur. 2015, 4, 178–202. [Google Scholar] [CrossRef] [PubMed]
- Goel, S.; Yadav, M.; Singh, K.; Jaat, R.S.; Singh, N.K. Exploring diverse wheat germplasm for novel alleles in HMW-GS for bread quality improvement. J. Food Sci. Technol. 2018, 55, 3257–3262. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Halford, N.G.; Belton, P.S.; Tatham, A.S. The structure and properties of gluten: An elastic protein from wheat grain. Philos. Trans. R. Soc. Lond B Biol. Sci. 2002, 357, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Payne, P.I.; Holt, L.M.; Worland, A.J.; Law, C.N. Structural and genetical studies on the high-molecular-weight subunits of wheat. Part 3. Telocentric mapping of the subunit genes on the long arms of homoeologous group 1 chromosomes. Theor. Appl. Genet. 1982, 63, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Kasarda, D.D. Glutenin polymers: The in vitro to in vivo transition. Cereal Foods World 1999, 44, 566–571. [Google Scholar]
- Payne, P.I.; CorWeld, K.G.; Holt, L.M.; Blackman, J.A. Correlations between the inheritance of certain high molecular weight subunits of glutenin and bread-making quality in progenies of 6 crosses of bread wheat. J. Sci. Food Agric. 1981, 32, 51–60. [Google Scholar] [CrossRef]
- Popineau, Y.; Cornec, M.; Lefebvre, J.; Marchylo, B. Influence of high M(r) glutenin subunits on glutenin polymers and rheological properties of glutens and gluten subfractions of near-isogenic lines of wheat Sicco. J. Cereal Sci. 1994, 19, 231–241. [Google Scholar] [CrossRef]
- Pirozi, M.; Margiotta, B.; Latiandra, D.; MacRitchie, F. Composition of polymeric proteins and bread-making quality of wheat lines with allelic HMW-GS differing in number of cysteines. J. Cereal Sci. 2008, 48, 117–122. [Google Scholar] [CrossRef]
- Payne, P.I.; Corfield, K.G.; Blackman, J.A. Identification of a high-molecular-weight subunit of glutenin whose presence correlates with bread-making quality in wheats of related pedigree. Theor. Appl. Genet. 1979, 55, 153–159. [Google Scholar] [CrossRef]
- Sapone, A.; Bai, J.C.; Ciacci, C.; Dolinsek, J.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Rostami, K.; Sanders, D.S.; Schumann, M.; et al. Spectrum of gluten-related disorders: Consensus on new nomenclature and classification. BMC Med. 2012, 10, 13. [Google Scholar] [CrossRef]
- Schuppan, D.; Pickert, G.; Ashfaq-Khan, M.; Zevallos, V. Non-celiac wheat sensitivity: Differential diagnosis, triggers and implications. Best Pract. Res. Clin. Gastroenterol. 2015, 29, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Zevallos, V.F.; Raker, V.; Tenzer, S.; Jimenez-Calvente, C.; Ashfaq-Khan, M.; Russel, N.; Pickert, G.; Schild, H.; Steinbrink, K.; Schuppan, D. Nutritional wheat amylase-trypsin inhibitors promote intestinal inflammation via activation of myeloid cells. Gastroenterology 2017, 152, 1100–1113.e1112. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Hassall, K.L.; Grausgruber, H.; Andersson, A.A.M.; Lampi, A.M.; Piironen, V.; Rakszegi, M.; Ward, J.L.; Lovegrove, A. Do modern types of wheat have lower quality for human health? Nutr. Bull. 2020, 45, 362–373. [Google Scholar] [CrossRef] [PubMed]
- EL Hassouni, K.; Sielaff, M.; Leiser, W.; Würschum, T.; Schuppan, D.; Tenzer, S.; Longin, C.F.H. Genetic Architecture Underlying the Expression of Eight Alpha-Amylase Trypsin Inhibitors. Theor. Appl. Genet. 2021, 134, 3427–3441. [Google Scholar] [CrossRef] [PubMed]
- Jackson, E.A.; Holt, L.M.; Payne, P.I. Characterisation of high molecular weight gliadin and low-molecular-weight glutenin subunits of wheat endosperm by two-dimensional electrophoresis and the chromosomal localisation of their controlling genes. Theor. Appl. Genet. 1983, 66, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Canovas, F.M.; Dumas-Gaudot, E.; Recorbet, G.; Jorrin, J.; Mock, H.P.; Rossignol, M. Plant proteome analysis. Proteomics 2004, 4, 285–298. [Google Scholar] [CrossRef]
- Afzal, M.; Pfannstiel, J.; Zimmermann, J.; Bischoff, S.C.; Würschum, T.; Longin, C.F.H. High-resolution proteomics reveals differences in the proteome of spelt and bread wheat flour representing targets for research on wheat sensitivities. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Afzal, M.; Sielaff, M.; Curella, V.; Neerukonda, M.; El Hassouni, K.; Schuppan, D.; Tenzer, S.; Longin, C.F.H. Characterization of 150 wheat cultivars by LC-MS-based label-free quantitative proteomics unravels possibilities to design wheat better for baking quality and human health. Plants 2021, 10, 424. [Google Scholar] [CrossRef]
- Mackay, T.F.C. The genetic architecture of quantitative traits. Annu. Rev. Genet. 2001, 35, 303–339. [Google Scholar] [CrossRef]
- Holland, J.B. Genetic architecture of complex traits in plants. Curr. Opin. Plant Biol. 2007, 10, 156–161. [Google Scholar] [CrossRef]
- Cooper, M.; van Eeuwijk, F.A.; Hammer, G.L.; Podlich, D.W.; Messina, C. Modeling QTL for complex traits: Detection and context for plant breeding. Curr. Opin. Plant Biol. 2009, 12, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Naz, A.A.; Kunert, A.; Lind, V.; Pillen, K.; Léon, J. AB-QTL analysis in winter wheat: II. Genetic analysis of seedling and field resistance against leaf rust in a wheat advanced backcross population. Theor. Appl. Genet. 2008, 116, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Lillemo, M.; Asalf, B.; Singh, R.P.; Huerta-Espino, J.; Chen, X.M.; He, Z.H.; Bjørnstad, A. The adult plant rust resistance loci Lr34/Yr18 and Lr46/Yr29 are important determinants of partial resistance to powdery mildew in bread wheat line Saar. Theor. Appl. Genet. 2008, 116, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Naz, A.A.; Dadshani, S.; Ballvora, A.; Pillen, K.; Léon, J. Genetic Analysis and Transfer of Favorable Exotic QTL Alleles for Grain Yield Across D Genome Using Two Advanced Backcross Wheat Populations. Front. Plant Sci. 2019, 10, 711. [Google Scholar] [CrossRef] [PubMed]
- Chao, S.; Dubcovsky, J.; Dvorak, J.; Luo, M.-C.; Baenziger, S.P.; Matnyazov, R.; Clark, D.R.; E Talbert, L.; A Anderson, J.; Dreisigacker, S.; et al. Population- and genome-specific patterns of linkage disequilibrium and SNP variation in spring and winter wheat (Triticum aestivum L.). BMC Genom. 2010, 11, 727. [Google Scholar] [CrossRef]
- Jones, H.; Gosman, N.; Horsnell, R.; Rose, G.A.; Everest, L.A.; Bentley, A.R. Strategy for exploiting exotic germplasm using genetic, morphological, and environmental diversity: The Aegilops tauschii Coss. example. Theor. Appl. Genet. 2013, 126, 1793–1808. [Google Scholar] [CrossRef]
- Ogbonnaya, F.C.; Abdalla, O.; Mujeeb-Kazi, A.; Kazi, A.G.; Xu, S.S.; Gosman, N.; Lagudah, E.S.; Bonnett, D.; Sorrells, M.E.; Tsujimoto, H. Synthetic hexaploids: Harnessing species of the primary gene pool for wheat improvement. Plant Breed. Rev. 2013, 37, 35–122. [Google Scholar]
- Rasheed, A.; Wen, W.; Gao, F.; Zhai, S.; Jin, H.; Liu, J.; Guo, Q.; Zhang, Y.; Dreisigacker, S.; Xia, X.; et al. Development and validation of KASP assays for genes underpinning key economic traits in bread wheat. Theor. Appl. Genet. 2016, 129, 1843–1860. [Google Scholar] [CrossRef]
- Rasheed, A.; Jin, H.; Xiao, Y.; Zhang, Y.; Hao, Y.; Zhang, Y.; Hickey, L.; Morgounov, A.; Xia, X.; He, Z. Allelic effects and variations for key bread-making quality genes in bread wheat using high-throughput molecular markers. J. Cereal Sci. 2019, 85, 305–309. [Google Scholar] [CrossRef]
- Liu, S.; Chao, S.; Anderson, J.A. New DNA markers for high molecular weight glutenin subunits in wheat. Theor. Appl. Genet. 2008, 118, 177–183. [Google Scholar] [CrossRef]
- Schwarz Schwarz, G.; Sift, A.; Wenzel, G.; Mohler, V. DHPLC scoring of a SNP between promoter sequences of HMW glutenin x-type alleles at the Glu-D1 locus in wheat. J. Agric. Food Chem. 2003, 51, 4263–4267. [Google Scholar] [CrossRef] [PubMed]
- Dreisigacker, S.; Xiao, Y.; Sehgal, D.; Guzman, C.; He, Z.; Xia, X.; Peña, R.J. SNP markers for low molecular glutenin subunits (LMW-GSs) at the Glu-A3 and Glu-B3 loci in bread wheat. PLoS ONE 2020, 15, e0233056. [Google Scholar] [CrossRef] [PubMed]
- Bonafede, M.D.; Tranquilli, G.; Pflüger, L.A.; Peña, R.J.; Dubcovsky, J. Effect of allelic variation at the Glu-3/Gli-1 loci on breadmaking quality parameters in hexaploid wheat (Triticum aestivum L.). J. Cereal Sci. 2015, 62, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Afzal, M.; Sielaff, M.; Distler, U.; Schuppan, D.; Tenzer, S.; Longin, C.F.H. Reference proteomes of five wheat species as starting point for future design of cultivars with lower allergenic potential. NPJ Sci. Food 2022. submitted. [Google Scholar] [CrossRef]
- Zimmermann, J.; Hubel, P.; Pfannstiel, J.; Afzal, M.; Longin, C.F.H.; Hitzmann, B. Comprehensive proteome analysis of bread deciphering the allergenic potential of bread wheat, spelt and rye. J. Proteomics. 2021, 247, 104318. [Google Scholar] [CrossRef]
- Juhász, A.; Belova, T.; Florides, C.G.; Maulis, C.; Fischer, I.; Gell, G.; Birinyi, Z.; Ong, J.; Keeble-Gagnère, G.; Maharajan, A.; et al. Genome mapping of seed-borne allergens and immunoresponsive proteins in wheat. Sci. Adv. 2018, 4, eaar8602. [Google Scholar] [CrossRef]
- Mari, A.; Rasi, C.; Palazzo, P.; Scala, E. Allergen databases: Current status and perspectives. Curr. Allergy Asthma Rep. 2009, 9, 376–383. [Google Scholar] [CrossRef]
- Bariana, H.S.; McIntosh, R.A. Characterisation and origin of rust and powdery mildew resistance genes in VPM1 wheat. Euphytica 1994, 76, 53–61. [Google Scholar] [CrossRef]
- Seah, S.; Bariana, H.; Jahier, J.; Sivasithamparam, K.; Lagudah, E.S. The introgressed segment carrying rust resistance genes Yr17, Lr37 and Sr38 in wheat can be assayed by a cloned disease resistance gene-like sequence. Theor. Appl. Genet. 2001, 102, 600–605. [Google Scholar] [CrossRef]
- Helguera, M.; Khan, I.A.; Kolmer, J.; Lijavetzky, D.; Zhong-qi, L.; Dubcovsky, J. PCR Assays for the Lr37-Yr17-Sr38 Cluster of Rust Resistance Genes and Their Use to Develop Isogenic Hard Red Spring Wheat Lines. Crop. Sci. 2003, 43, 1839–1847. [Google Scholar] [CrossRef]
- Mondaini, A.; Rosyara, U.; Sehgal, D.; Dreisigacker, S. Selection signatures in the CIMMYT International Elite Spring and Semi-arid Wheat Yield Trials. Plant Genome 2021, 15, 1. [Google Scholar] [CrossRef] [PubMed]
- Cruz, C.D.; Peterson, G.L.; Bockus, W.W.; Kankanala, P.; Dubcovsky, J.; Jordan, K.W.; Akhunov, E.; Chumley, F.; Baldelomar, F.D.; Valent, B. The 2NS translocation from Aegilops ventricosa confers resistance to the Triticum pathotype of Magnaporthe oryzae. Crop. Sci. 2016, 56, 990–1000. [Google Scholar] [CrossRef] [PubMed]
- Rapp, M.; Schwadorf, K.; Leiser, W.L.; Würschum, T.; Longin, C.F.H. Assessing the variation and genetic architecture of asparagine content in wheat: What can plant breeding contribute to a reduction in the acrylamide precursor? Theor. Appl. Genet. 2018, 131, 2427–2437. [Google Scholar] [CrossRef]
- Deutsch, E.W.; Bandeira, N.; Sharma, V.; Perez-Riverol, Y.; Carver, J.J.; Kundu, D.J.; García-Seisdedos, D.; Jarnuczak, A.F.; Hewapathirana, S.; Pullman, B.S.; et al. The ProteomeXchange Consortium in 2020: Enabling ‘Big Data’ Approaches in Proteomics. Nucleic Acids Res. 2020, 48, D1145–D1152. [Google Scholar] [CrossRef] [PubMed]
- Distler, U.; Łącki, M.K.; Schumann, S.; Wanninger, M.; Tenzer, S. Enhancing Sensitivity of Microflow-Based Bottom-Up Proteomics through Postcolumn Solvent Addition. Anal. Chem. 2019, 91, 7510–7515. [Google Scholar] [CrossRef] [PubMed]
- Bateman, R.H.; Carruthers, R.; Hoyes, J.B.; Jones, C.; Langridge, J.I.; Millar, A.; Vissers, J.P.C. A novel precursor ion discovery method on a hybrid quadrupole orthogonal acceleration time-of-flight (Q-TOF) mass spectrometer for studying protein phosphorylation. J. Am. Soc. Mass Spectrom. 2002, 13, 792–803. [Google Scholar] [CrossRef]
- Distler, U.; Kuharev, J.; Navarro, P.; Levin, Y.; Schild, H.; Tenzer, S. Drift time-specific collision energies enable deep-coverage data-independent acquisition proteomics. Nat. Methods 2013, 11, 167–170. [Google Scholar] [CrossRef]
- Silva, J.C.; Gorenstein, M.V.; Li, G.-Z.; Vissers, J.P.C.; Geromanos, S.J. Absolute Quantification of Proteins by LCMSE. Mol. Cell. Proteomics. 2006, 5, 144–156. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Gilmour, A.R.; Gogel, B.J.; Cullis, B.R.; Thompson, R. ASReml User Guide Release 3.0; VSN International Ltd.: Hemel Hempstead, UK, 2009. [Google Scholar]
- Money, D.; Gardner, K.; Migicovsky, Z.; Schwaninger, H.; Zhong, G.Y.; Myles, S. LinkImpute: Fast and accurate genotype imputation for non-model organisms. G3 Genes Genom. Genet. 2015, 5, 2383–2390. [Google Scholar]
- Zhu, T.; Wang, L.; Rimbert, H.; Rodriguez, J.C.; Deal, K.R.; De Oliveira, R.; Choulet, F.; Keeble-Gagnère, G.; Tibbits, J.; Rogers, J.; et al. Optical maps refine the bread wheat Triticum aestivum cv Chinese Spring genome assembly. Plant J. 2021, 107, 303–314. [Google Scholar] [CrossRef]
- Van Rossum, G.; Drake, F.L. Python 3 Reference Manual; CreateSpace: Scotts Valley, CA, USA, 2009. [Google Scholar]
- Li, H.; Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 2010, 26, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Li, H. A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinformatics 2011, 27, 2987–2993. [Google Scholar] [CrossRef] [PubMed]
- Quinlan, A.R.; Hall, I.M. BEDTools: A flexible suite of utilities for comparing genomic features. Bioinformatics 2010, 26, 841–842. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Z. GAPIT Version 3: Boosting Power and Accuracy for Genomic Association and Prediction. Genom. Proteom. Bioinform. 2021, 19, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Turner, S. qqman: An R package for visualizing GWAS results using Q-Q and manhattan plots. J. Open Source Softw. 2018, 3, 731. [Google Scholar] [CrossRef]
- Utz, H.F.; Melchinger, A.E.; Schön, C.C. Bias and sampling error of the estimated proportion of genotypic variance explained by quantitative trait loci determined from experimental data in maize using cross validation and validation with independent samples. Genetics 2000, 154, 1839–1849. [Google Scholar] [CrossRef]
- Würschum, T.; Leiser, W.L.; Kazman, E.; Longin, C.F.H. Genetic control of protein content and sedimentation volume in European winter wheat cultivars. Theor. Appl. Genet. 2016, 129, 1685–1696. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2009; pp. 115–137. [Google Scholar]
- Singh, N.K.; Shepherd, K.W.; Cornish, G.B. A simplified SDS-PAGE procedure for separating LMW subunits of glutenin. J. Cereal Sci. 1991, 14, 203–208. [Google Scholar] [CrossRef]
- Payne, P.I.; Lawrence, G.J. Catalogue of alleles for the complex gene loci, Glu-A1, Glu-B1, and Glu-D1 which code for high-molecular-weight subunits of glutenin in hexaploid wheat. Cereal Res. Commun. 1983, 11, 29–35. [Google Scholar]
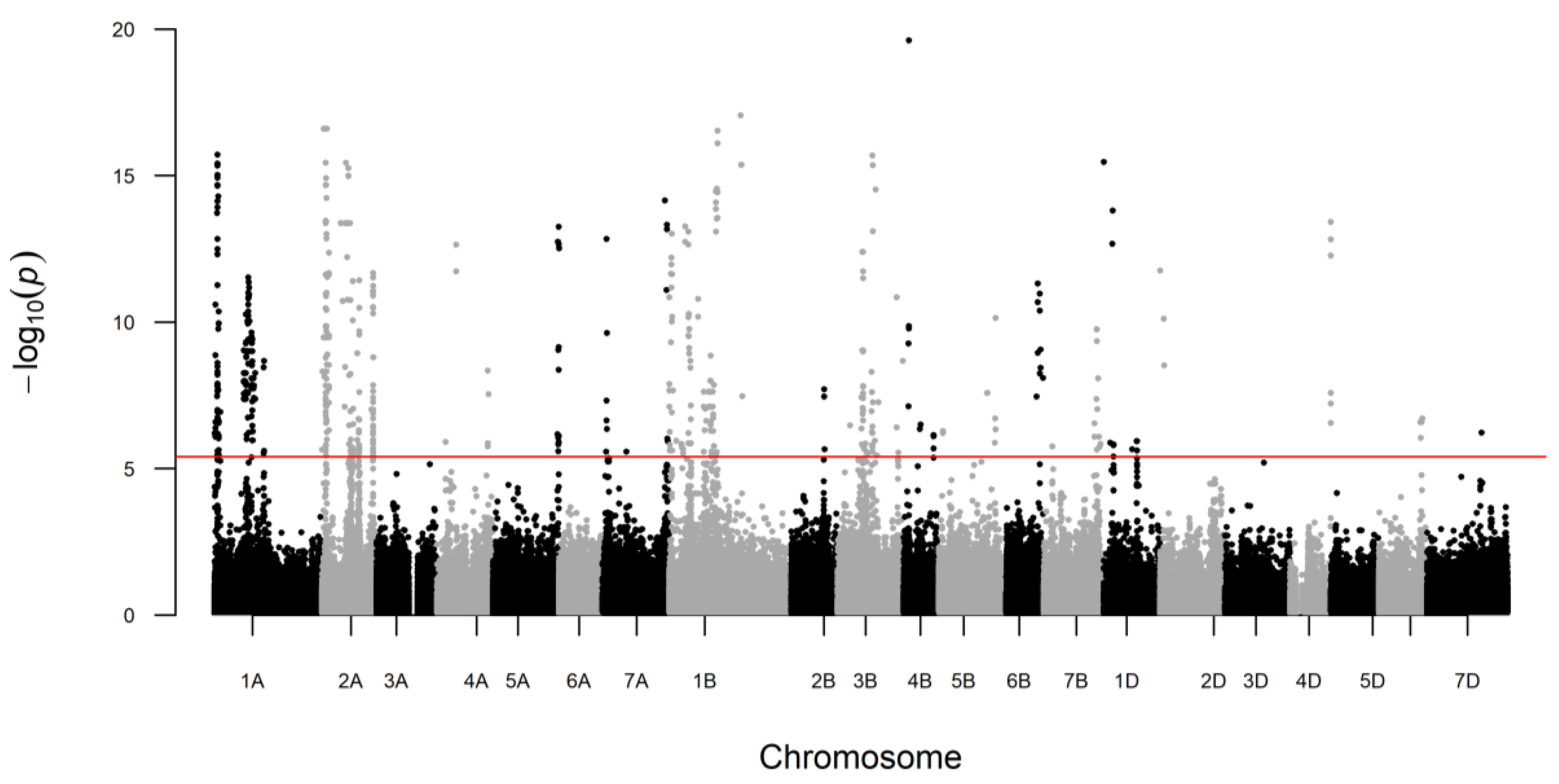
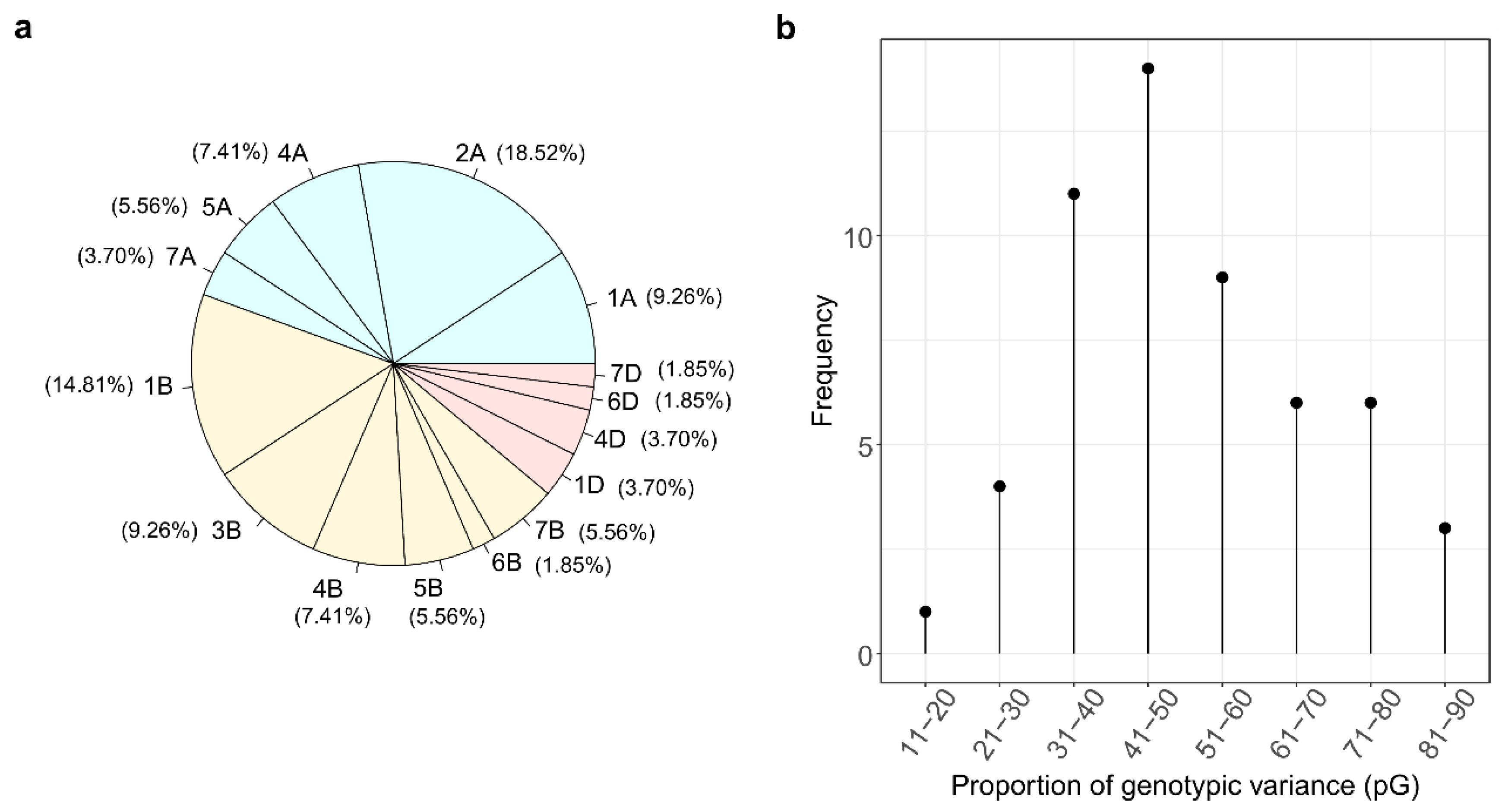
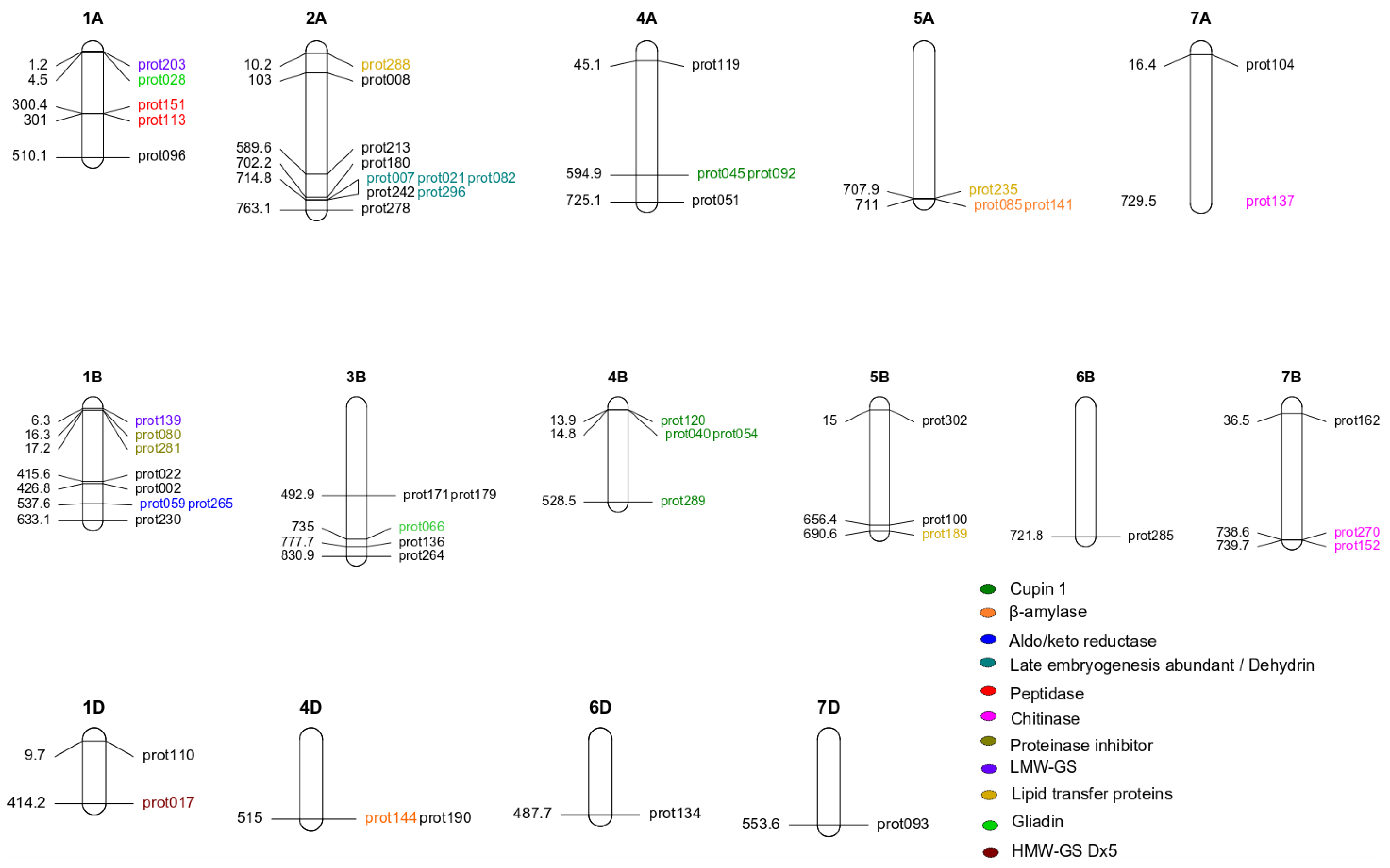
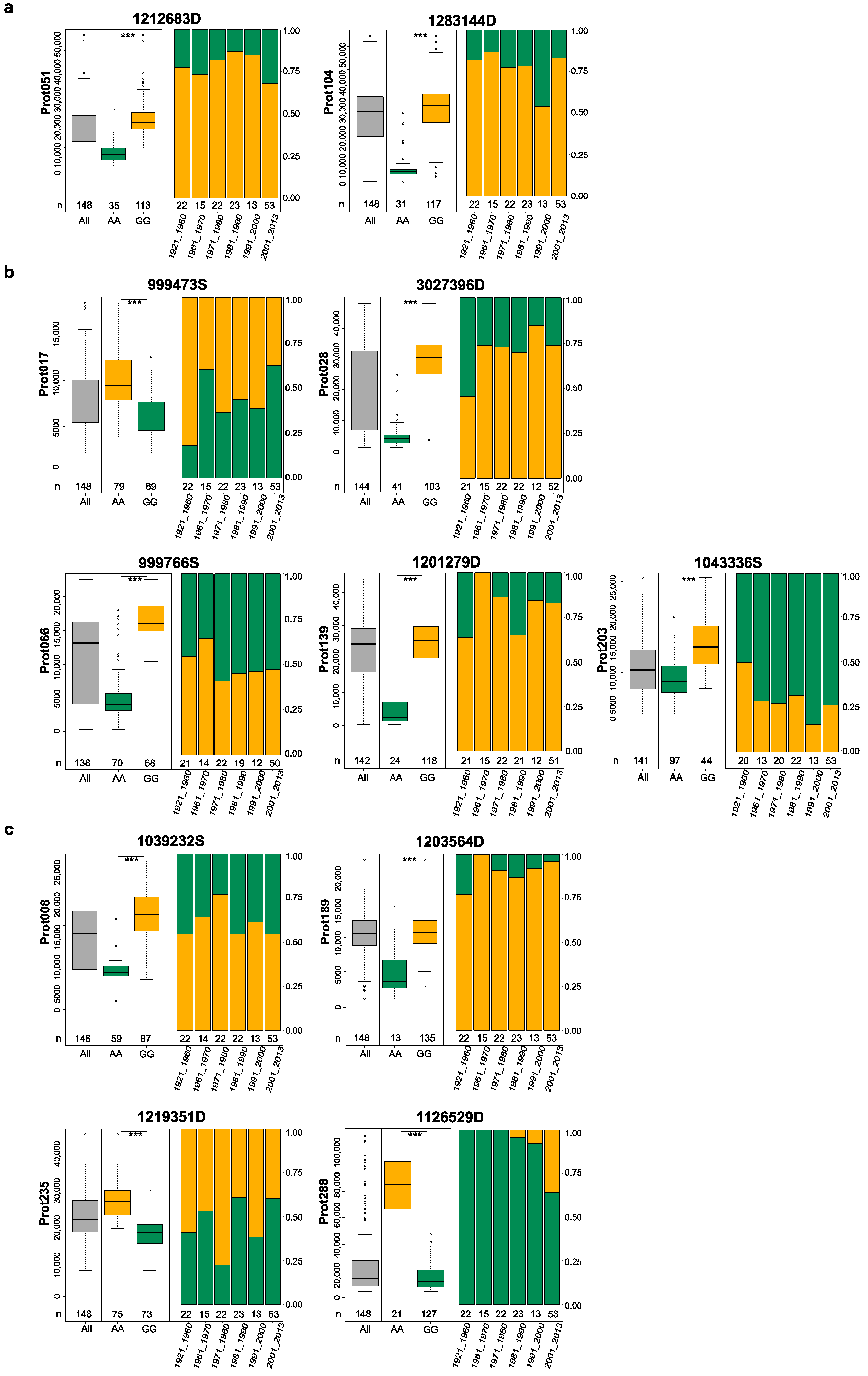
| Trait | UniProt ID | UniProt Name | Marker | Chr. | p-Value | LOD | Gen Pos. (cM) | Phy Pos. Start (bp) | Phy Pos. Stop (bp) | PG | α-Effect | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gluten | prot051 | A0A3B6I2R2 | 1212683D | 4A | 4.65 × 10−9 | 8.33 | 2,310,811 | 725,135,595 | 725,135,664 | 45.53 | 7006.50 | |
| prot104 | A0A3B6RB62 | 1283144D | 7A | 1.48 × 10−13 | 12.83 | 211,140 | 16,051,783 | 16,366,772 | 54.70 | 13,071.91 | ||
| Gluten and allergen | prot017 | G1E6K7 | HMW-Dx5 | 999473S | 1D | 1.18 × 10−6 | 5.93 | 1,492,346 | 414,170,821 | 414,170,890 | 42.73 | −1947.51 |
| prot028 | Q94G97 | γ-Gliadin | 3027396D | 1A | 1.93 × 10−16 | 15.72 | 150,899 | 4,462,173 | 4,462,215 | 82.41 | 12,546.17 | |
| prot066 | D2KFH0 | Gliadin/avenin | 999766S | 3B | 2.07 × 10−16 | 15.68 | 1,607,210 | 734,966,174 | 734,966,228 | 82.01 | 5516.69 | |
| prot139 | I1XB56 | LMW-GS | 1201279D | 1B | 9.60 × 10−14 | 13.02 | 182,700 | 6,301,587 | 6,301,656 | 73.82 | 10,631.02 | |
| prot203 | C3VN75 | LMW-GluA3 | 1043336S | 1A | 2.51 × 10−9 | 8.60 | 149,830 | 1,235,911 | 1,235,980 | 49.85 | −12,552.71 | |
| Non-gluten allergen | prot008 | Q6W8Q2 | Peroxiredoxin | 1039232S | 2A | 5.61 × 10−16 | 15.25 | 1,198,150 | 98,916,927 | 102,957,935 | 75.2 | 6937.00 |
| prot189 | Q2PCC3 | LTP | 1203564D | 5B | 7.33 × 10−11 | 10.13 | 2,583,420 | 690,570,279 | 690,570,348 | 32.32 | 3056.54 | |
| prot235 | Q2PCC5 | LTP | 1219351D | 5A | 5.60 × 10−14 | 13.25 | 2,978,100 | 707,857,285 | 707,857,336 | 56.53 | −47,278.73 | |
| prot288 | A0A1D5UB33 | 1126529D | 2A | 2.51 × 10−17 | 16.60 | 80,770 | 5,395,135 | 10,230,260 | 84.51 | −34,444.07 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Hassouni, K.; Afzal, M.; Steige, K.A.; Sielaff, M.; Curella, V.; Neerukonda, M.; Tenzer, S.; Schuppan, D.; Longin, C.F.H.; Thorwarth, P. Multiomics Based Association Mapping in Wheat Reveals Genetic Architecture of Quality and Allergenic Related Proteins. Int. J. Mol. Sci. 2023, 24, 1485. https://doi.org/10.3390/ijms24021485
El Hassouni K, Afzal M, Steige KA, Sielaff M, Curella V, Neerukonda M, Tenzer S, Schuppan D, Longin CFH, Thorwarth P. Multiomics Based Association Mapping in Wheat Reveals Genetic Architecture of Quality and Allergenic Related Proteins. International Journal of Molecular Sciences. 2023; 24(2):1485. https://doi.org/10.3390/ijms24021485
Chicago/Turabian StyleEl Hassouni, Khaoula, Muhammad Afzal, Kim A. Steige, Malte Sielaff, Valentina Curella, Manjusha Neerukonda, Stefan Tenzer, Detlef Schuppan, Carl Friedrich Horst Longin, and Patrick Thorwarth. 2023. "Multiomics Based Association Mapping in Wheat Reveals Genetic Architecture of Quality and Allergenic Related Proteins" International Journal of Molecular Sciences 24, no. 2: 1485. https://doi.org/10.3390/ijms24021485
APA StyleEl Hassouni, K., Afzal, M., Steige, K. A., Sielaff, M., Curella, V., Neerukonda, M., Tenzer, S., Schuppan, D., Longin, C. F. H., & Thorwarth, P. (2023). Multiomics Based Association Mapping in Wheat Reveals Genetic Architecture of Quality and Allergenic Related Proteins. International Journal of Molecular Sciences, 24(2), 1485. https://doi.org/10.3390/ijms24021485






