Opposing MMP-9 Expression in Mesenchymal Stromal Cells and Head and Neck Tumor Cells after Direct 2D and 3D Co-Culture
Abstract
1. Introduction
2. Results
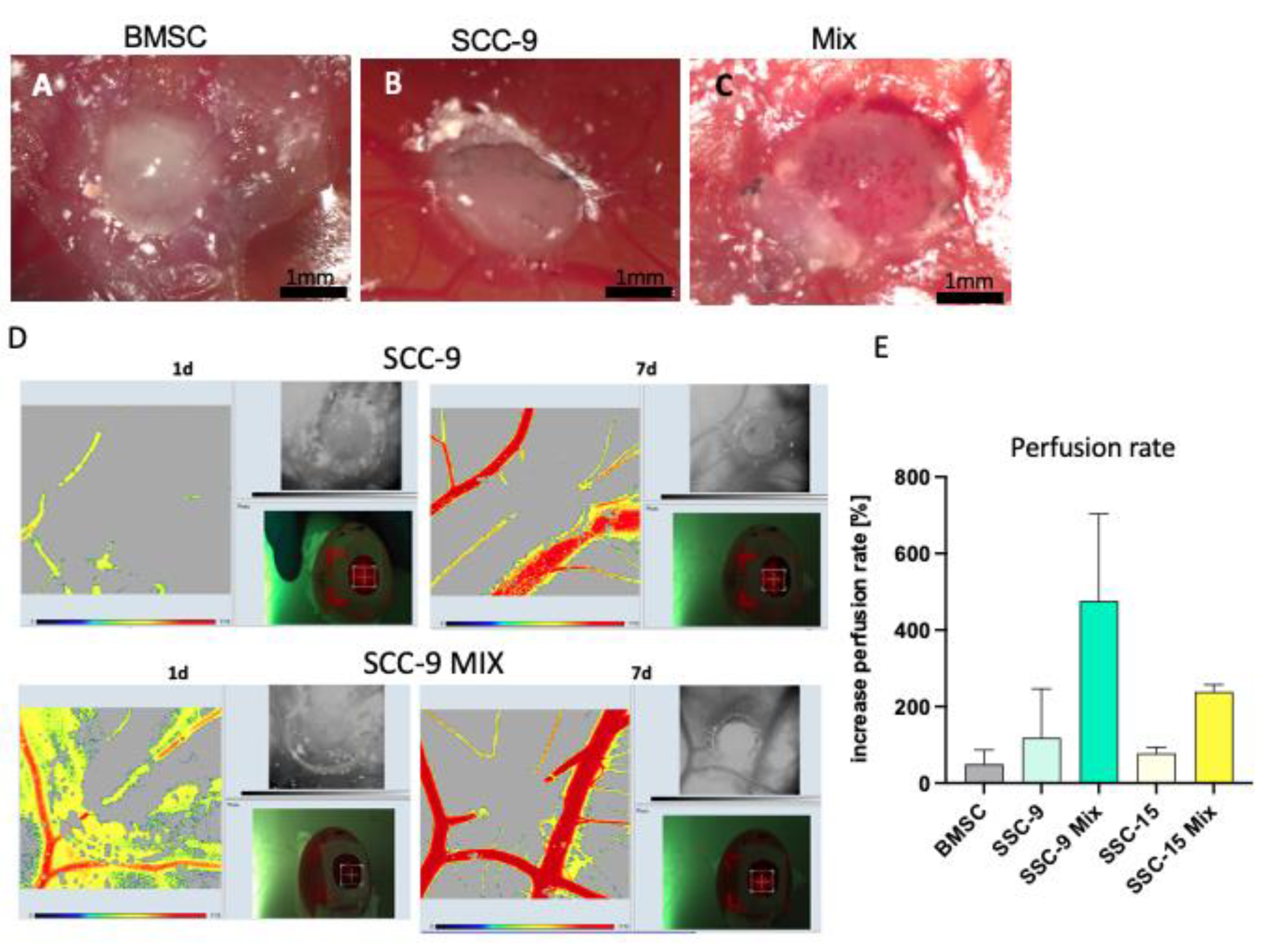
2.1. BMSC/HNSCC Co-Culture Increases Neoangiogenesis and Increased Perfusion in the CAM Model
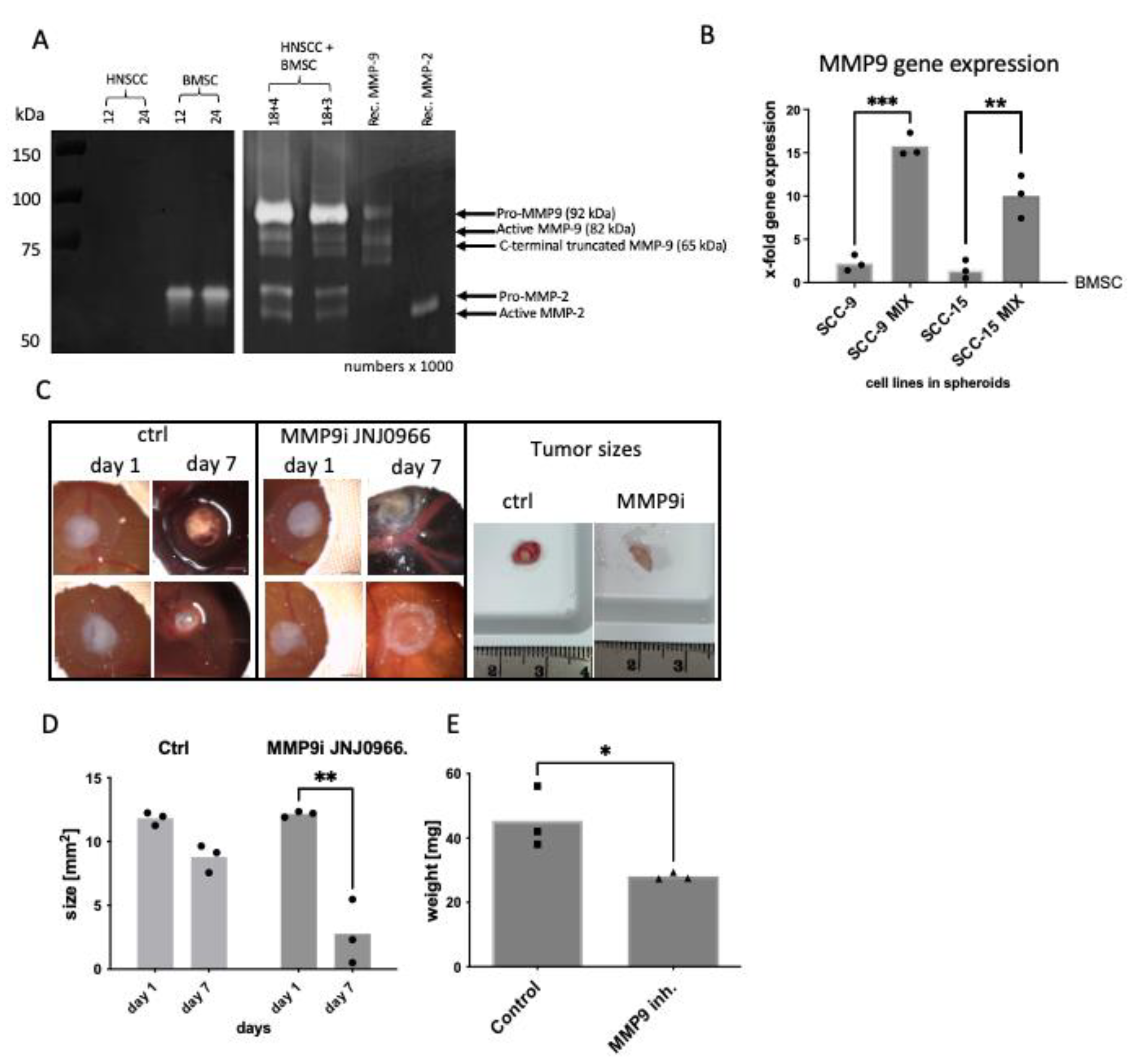
2.2. BMSC/HNSCC Co-Culture Increases Expression and Secretion of MMP-9
2.3. MMP-9 Secretion and Activation Are Responsible for Hemorrhage in the CAM Model
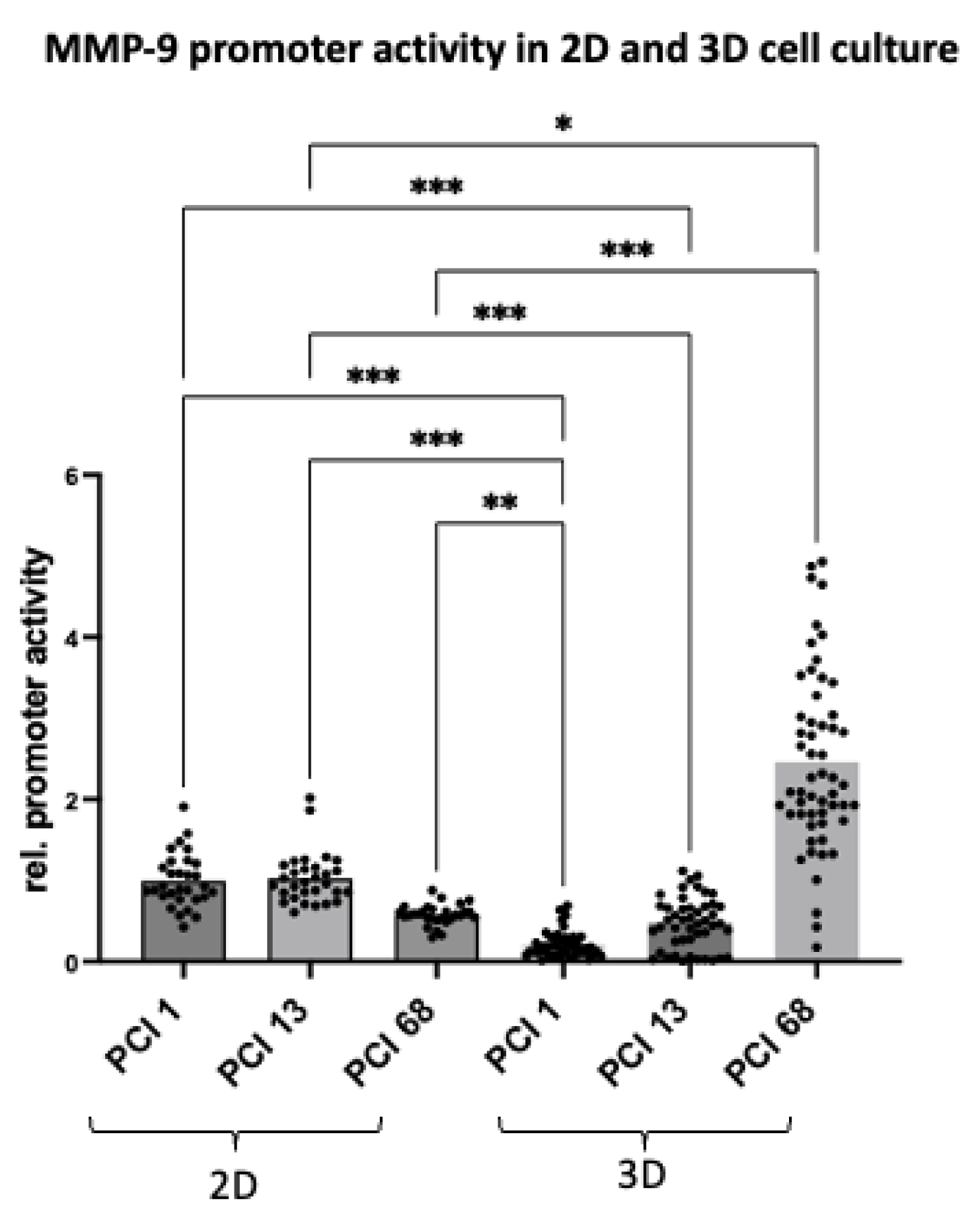
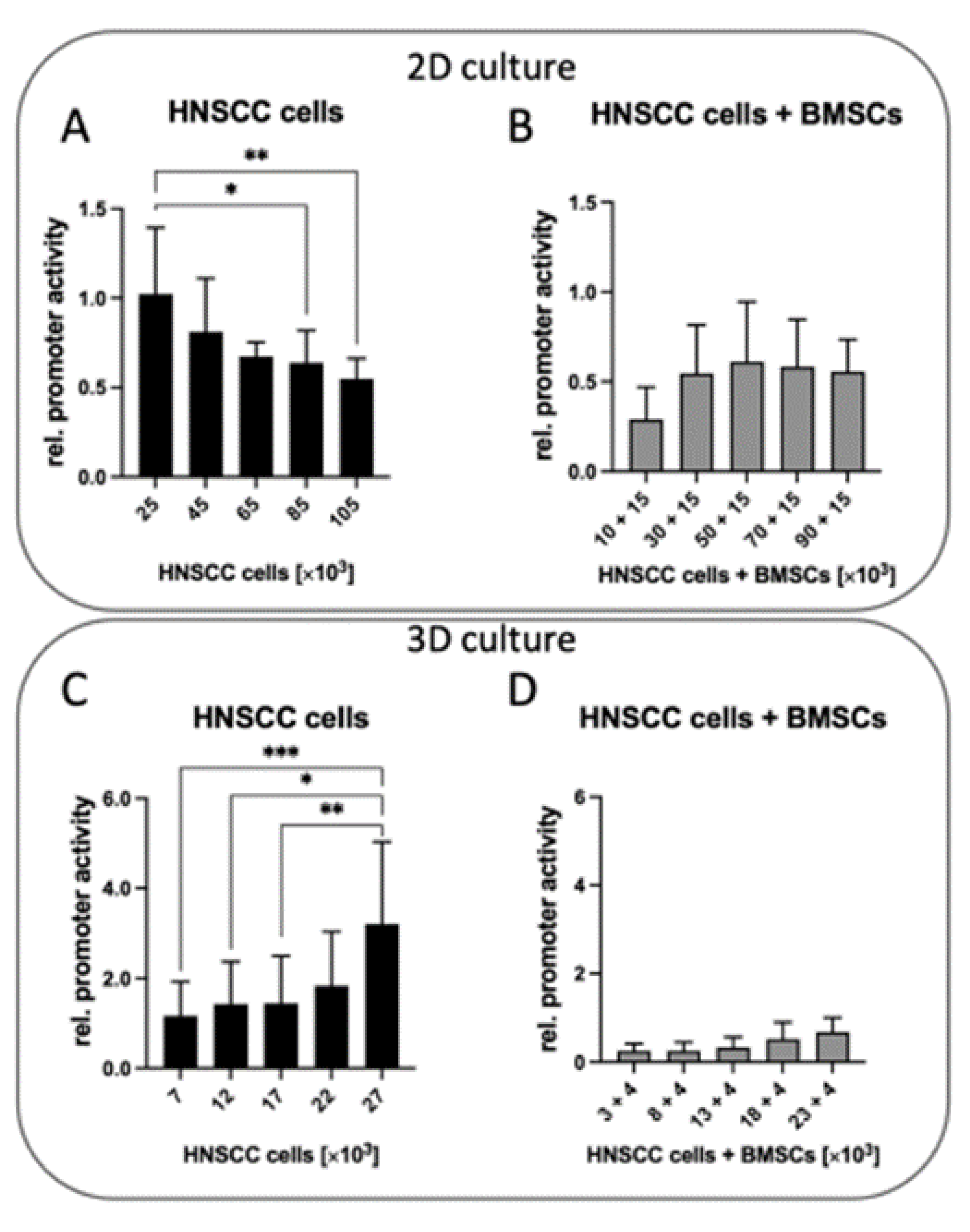
2.4. MMP-9 Promotor Activity in HNSCC Cells Differs Significantly in 2D and 3D Cell Culture
2.5. Decrease of MMP-9 Promoter Activity in HNSCC Cells When Co-Cultured with BMSCs in 2D and 3D
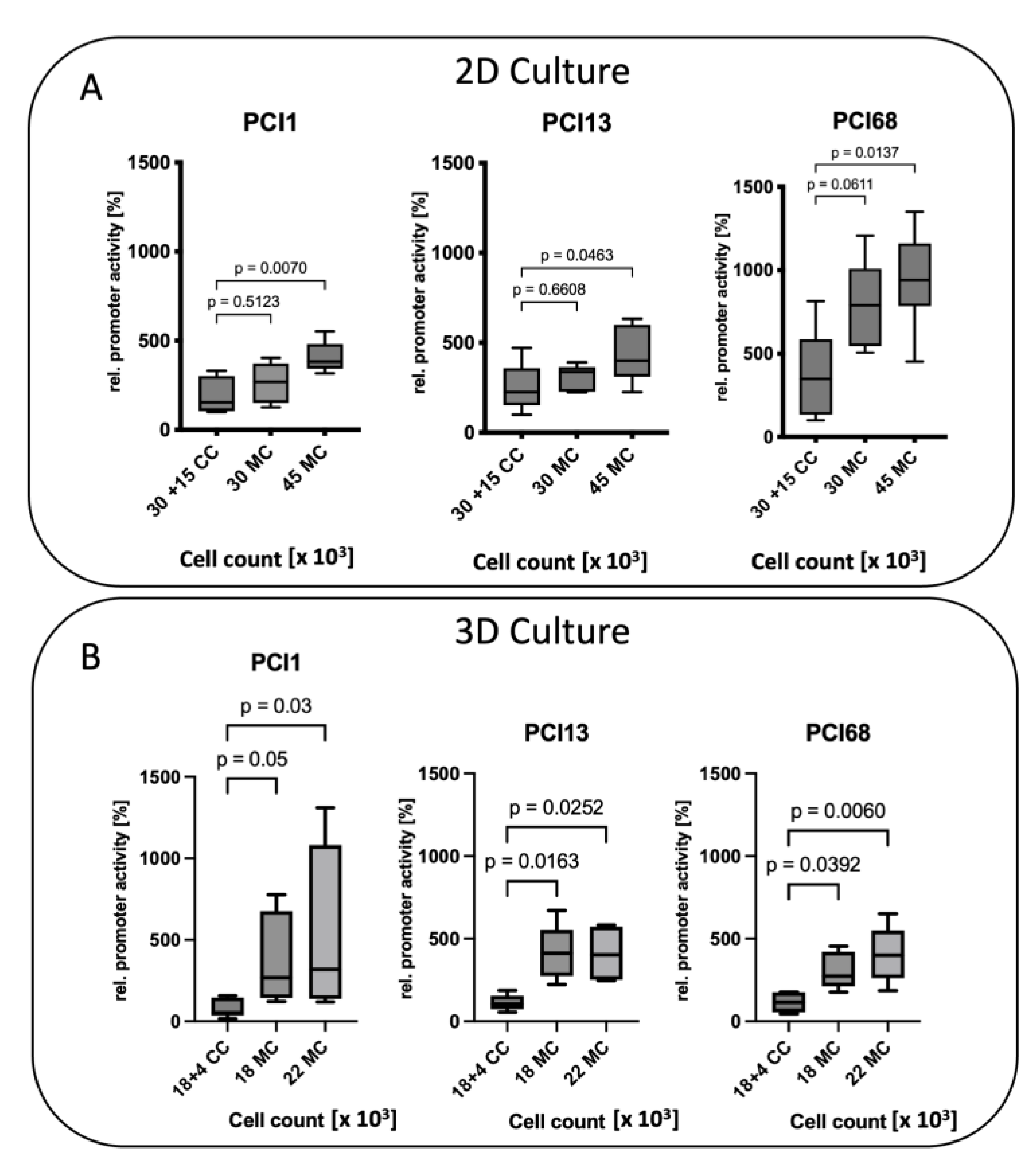
2.6. Influence of Cell Density on MMP-9 Promoter Activity in 2D and 3D
3. Discussion
4. Materials and Methods
4.1. Cells and Culture Conditions
4.1.1. HNSCC Cells
4.1.2. BMSCs
4.1.3. 2D Adherent Cell Cultivatio
4.1.4. 3D Tumor Spheroids Formation
4.2. Chorioallantoic Membrane (CAM) Model
4.3. Assays and Methods
4.3.1. Luciferase Activity Assay
4.3.2. Gelatin Zymography
4.3.3. Transient MMP-9 siRNA Knockdown
4.3.4. Tube Formation Assay
4.3.5. Angiogenesis Measurement via Laser Speckle Contrast Imaging (LSCI)
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Argiris, A.; Karamouzis, M.V.; Raben, D.; Ferris, R.L. Head and Neck Cancer. Lancet 2008, 371, 1695–1709. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.E.; Burtness, B.; Leemans, C.R.; Lui, V.W.Y.; Bauman, J.E.; Grandis, J.R. Head and Neck Squamous Cell Carcinoma. Nat. Rev. Dis. Prim. 2020, 6, 92. [Google Scholar] [CrossRef]
- Powell, S.F.; Vu, L.; Spanos, W.C.; Pyeon, D. The Key Differences between Human Papillomavirus-Positive and -Negative Head and Neck Cancers: Biological and Clinical Implications. Cancers 2021, 13, 5206. [Google Scholar] [CrossRef] [PubMed]
- Marron, M.; Boffetta, P.; Zhang, Z.-F.; Zaridze, D.; Wünsch-Filho, V.; Winn, D.M.; Wei, Q.; Talamini, R.; Szeszenia-Dabrowska, N.; Sturgis, E.M.; et al. Cessation of Alcohol Drinking, Tobacco Smoking and the Reversal of Head and Neck Cancer Risk. Int. J. Epidemiol. 2010, 39, 182–196. [Google Scholar] [CrossRef] [PubMed]
- Rivera, C.; Venegas, B. Histological and Molecular Aspects of Oral Squamous Cell Carcinoma (Review). Oncol. Lett. 2014, 8, 7–11. [Google Scholar] [CrossRef]
- Maulina, T.; Iskandarsyah, A.; Hardianto, A.; Sjamsudin, E.; Nandini, M.; Kasim, A.; Yusuf, H.Y. The Incidence of Oral Squamous Cell Carcinoma (OSCC) and Its Relationship with Orofacial Pain in Oral Cancer Patients in West Java Province, Indonesia. J. Oral Maxillofac. Surg. Med. Pathol. 2017, 29, 29–32. [Google Scholar] [CrossRef]
- Spoerl, S.; Gerken, M.; Fischer, R.; Mamilos, A.; Spoerl, S.; Wolf, S.; Pohl, F.; Klingelhöffer, C.; Ettl, T.; Reichert, T.E.; et al. Lymphatic and Vascular Invasion in Oral Squamous Cell Carcinoma: Implications for Recurrence and Survival in a Population-Based Cohort Study. Oral Oncol. 2020, 111, 105009. [Google Scholar] [CrossRef]
- Brana, I.; Siu, L.L. Locally Advanced Head and Neck Squamous Cell Cancer: Treatment Choice Based on Risk Factors and Optimizing Drug Prescription. Ann. Oncol. 2012, 23 (Suppl. 10), x178–x185. [Google Scholar] [CrossRef]
- Shaw, R.J.; Brown, J.S.; Woolgar, J.A.; Lowe, D.; Rogers, S.N.; Vaughan, E.D. The Influence of the Pattern of Mandibular Invasion on Recurrence and Survival in Oral Squamous Cell Carcinoma. Head Neck 2004, 26, 861–869. [Google Scholar] [CrossRef]
- Bissell, M.J.; Radisky, D. Putting Tumours in Context. Nat. Rev. Cancer 2001, 1, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.M.; Fusenig, N.E. Friends or Foes—Bipolar Effects of the Tumour Stroma in Cancer. Nat. Rev. Cancer 2004, 4, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage Potential of Adult Human Mesenchymal Stem Cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef] [PubMed]
- De Boeck, A.; Narine, K.; De Neve, W.; Mareel, M.; Bracke, M.; De Wever, O. Resident and Bone Marrow-Derived Mesenchymal Stem Cells in Head and Neck Squamous Cell Carcinoma. Oral Oncol. 2010, 46, 336–342. [Google Scholar] [CrossRef]
- Li, H.-C.; Stoicov, C.; Rogers, A.B.; Houghton, J. Stem Cells and Cancer: Evidence for Bone Marrow Stem Cells in Epithelial Cancers. World J. Gastroenterol. 2006, 12, 363–371. [Google Scholar] [CrossRef]
- Koontongkaew, S. The Tumor Microenvironment Contribution to Development, Growth, Invasion and Metastasis of Head and Neck Squamous Cell Carcinomas. J. Cancer 2013, 4, 66–83. [Google Scholar] [CrossRef]
- Uccelli, A.; Moretta, L.; Pistoia, V. Mesenchymal Stem Cells in Health and Disease. Nat. Rev. Immunol. 2008, 8, 726–736. [Google Scholar] [CrossRef]
- Hutchinson, L.; Stenstrom, B.; Chen, D.; Piperdi, B.; Levey, S.; Lyle, S.; Wang, T.C.; Houghton, J. Human Barrett’s Adenocarcinoma of the Esophagus, Associated Myofibroblasts, and Endothelium Can Arise from Bone Marrow-Derived Cells after Allogeneic Stem Cell Transplant. Stem Cells Dev. 2011, 20, 11–17. [Google Scholar] [CrossRef]
- Galland, S.; Stamenkovic, I. Mesenchymal Stromal Cells in Cancer: A Review of Their Immunomodulatory Functions and Dual Effects on Tumor Progression. J. Pathol. 2020, 250, 555–572. [Google Scholar] [CrossRef]
- Klopp, A.H.; Gupta, A.; Spaeth, E.; Andreeff, M.; Marini, F. Concise Review: Dissecting a Discrepancy in the Literature: Do Mesenchymal Stem Cells Support or Suppress Tumor Growth? Stem Cells 2011, 29, 11–19. [Google Scholar] [CrossRef]
- Liu, C.; Feng, X.; Wang, B.; Wang, X.; Wang, C.; Yu, M.; Cao, G.; Wang, H. Bone Marrow Mesenchymal Stem Cells Promote Head and Neck Cancer Progression through Periostin-Mediated Phosphoinositide 3-Kinase/Akt/Mammalian Target of Rapamycin. Cancer Sci. 2018, 109, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, W.; Wang, T.; Chen, X.; Wang, J.; Zhang, X.; Cai, C.; Zhong, B.; Wu, J.; Chen, Z.; et al. Mesenchymal Stromal Cells-Derived Β2-Microglobulin Promotes Epithelial-Mesenchymal Transition of Esophageal Squamous Cell Carcinoma Cells. Sci. Rep. 2018, 8, 5422. [Google Scholar] [CrossRef] [PubMed]
- Rowan, B.G.; Lacayo, E.A.; Sheng, M.; Anbalagan, M.; Gimble, J.M.; Jones, R.K.; Joseph, W.J.; Friedlander, P.L.; Chiu, E.S. Human Adipose Tissue-Derived Stromal/Stem Cells Promote Migration and Early Metastasis of Head and Neck Cancer Xenografts. Aesthet. Surg. J. 2016, 36, 93–104. [Google Scholar] [CrossRef]
- Wu, Y.-L.; Li, H.-Y.; Zhao, X.-P.; Jiao, J.-Y.; Tang, D.-X.; Yan, L.-J.; Wan, Q.; Pan, C.-B. Mesenchymal Stem Cell-Derived CCN2 Promotes the Proliferation, Migration and Invasion of Human Tongue Squamous Cell Carcinoma Cells. Cancer Sci. 2017, 108, 897–909. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, X.; Fang, J.; Song, J.; Ma, D.; Luo, L.; He, B.; Xia, J.; Lui, V.W.Y.; Cheng, B.; et al. Mesenchymal Stem Cells Participate in Oral Mucosa Carcinogenesis by Regulating T Cell Proliferation. Clin. Immunol. 2019, 198, 46–53. [Google Scholar] [CrossRef]
- Liu, C.; Billet, S.; Choudhury, D.; Cheng, R.; Haldar, S.; Fernandez, A.; Biondi, S.; Liu, Z.; Zhou, H.; Bhowmick, N.A. Bone Marrow Mesenchymal Stem Cells Interact with Head and Neck Squamous Cell Carcinoma Cells to Promote Cancer Progression and Drug Resistance. Neoplasia 2021, 23, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Kansy, B.A.; Dißmann, P.A.; Hemeda, H.; Bruderek, K.; Westerkamp, A.M.; Jagalski, V.; Schuler, P.; Kansy, K.; Lang, S.; Dumitru, C.A.; et al. The Bidirectional Tumor--Mesenchymal Stromal Cell Interaction Promotes the Progression of Head and Neck Cancer. Stem Cell Res. Ther. 2014, 5, 95. [Google Scholar] [CrossRef]
- Zielske, S.P.; Livant, D.L.; Lawrence, T.S. Radiation Increases Invasion of Gene-Modified Mesenchymal Stem Cells into Tumors. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 843–853. [Google Scholar] [CrossRef]
- Tan, T.T.; Lai, R.C.; Padmanabhan, J.; Sim, W.K.; Choo, A.B.H.; Lim, S.K. Assessment of Tumorigenic Potential in Mesenchymal-Stem/Stromal-Cell-Derived Small Extracellular Vesicles (MSC-SEV). Pharmaceuticals 2021, 14, 345. [Google Scholar] [CrossRef]
- Bruna, F.; Arango-Rodríguez, M.; Plaza, A.; Espinoza, I.; Conget, P. The Administration of Multipotent Stromal Cells at Precancerous Stage Precludes Tumor Growth and Epithelial Dedifferentiation of Oral Squamous Cell Carcinoma. Stem Cell Res. 2017, 18, 5–13. [Google Scholar] [CrossRef]
- Bruna, F.; Plaza, A.; Arango, M.; Espinoza, I.; Conget, P. Systemically Administered Allogeneic Mesenchymal Stem Cells Do Not Aggravate the Progression of Precancerous Lesions: A New Biosafety Insight. Stem Cell Res. Ther. 2018, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Fan, H.; Zhou, B.; Ju, Z.; Yu, L.; Guo, L.; Han, J.; Lu, S. Fusion of Human Umbilical Cord Mesenchymal Stem Cells with Esophageal Carcinoma Cells Inhibits the Tumorigenicity of Esophageal Carcinoma Cells. Int. J. Oncol. 2012, 40, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Zhang, Z.; Han, Y.; Song, J.; Xu, X.; Jin, J.; Su, S.; Mu, D.; Liu, X.; Xu, S.; et al. Mesenchymal Stem Cells Derived from Normal Gingival Tissue Inhibit the Proliferation of Oral Cancer Cells In Vitro and In Vivo. Int. J. Oncol. 2016, 49, 2011–2022. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Adhikari, N.; Banerjee, S.; Amin, S.A.; Jha, T. Matrix Metalloproteinase-9 (MMP-9) and Its Inhibitors in Cancer: A Minireview. Eur. J. Med. Chem. 2020, 194, 112260. [Google Scholar] [CrossRef] [PubMed]
- Chandolia, B.; Basu, S.K.; Kumar, M. Can MMP-9 Be a Prognosticator Marker for Oral Squamous Cell Carcinoma? J. Clin. Diagn. Res. 2016, 10, ZC09–ZC13. [Google Scholar] [CrossRef] [PubMed]
- Cui, N.; Hu, M.; Khalil, R.A. Biochemical and Biological Attributes of Matrix Metalloproteinases. Prog. Mol. Biol. Transl. Sci. 2017, 147, 1–73. [Google Scholar] [CrossRef]
- Jackson, B.C.; Nebert, D.W.; Vasiliou, V. Update of Human and Mouse Matrix Metalloproteinase Families. Hum. Genom. 2010, 4, 194–201. [Google Scholar] [CrossRef]
- Nagase, H.; Woessner, J.F. Matrix Metalloproteinases. J. Biol. Chem. 1999, 274, 21491–21494. [Google Scholar] [CrossRef]
- Chaudhary, A.K.; Singh, M.; Bharti, A.C.; Asotra, K.; Sundaram, S.; Mehrotra, R. Genetic Polymorphisms of Matrix Metalloproteinases and Their Inhibitors in Potentially Malignant and Malignant Lesions of the Head and Neck. J. Biomed. Sci. 2010, 17, 10. [Google Scholar] [CrossRef]
- Patel, B.P.; Shah, P.M.; Rawal, U.M.; Desai, A.A.; Shah, S.V.; Rawal, R.M.; Patel, P.S. Activation of MMP-2 and MMP-9 in Patients with Oral Squamous Cell Carcinoma. J. Surg. Oncol. 2005, 90, 81–88. [Google Scholar] [CrossRef]
- Gobin, E.; Bagwell, K.; Wagner, J.; Mysona, D.; Sandirasegarane, S.; Smith, N.; Bai, S.; Sharma, A.; Schleifer, R.; She, J.-X. A Pan-Cancer Perspective of Matrix Metalloproteases (MMP) Gene Expression Profile and Their Diagnostic/Prognostic Potential. BMC Cancer 2019, 19, 581. [Google Scholar] [CrossRef] [PubMed]
- Wessely, A.; Waltera, A.; Reichert, T.E.; Stöckl, S.; Grässel, S.; Bauer, R.J. Induction of ALP and MMP9 Activity Facilitates Invasive Behavior in Heterogeneous Human BMSC and HNSCC 3D Spheroids. FASEB J. 2019, 33, 11884–11893. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, A.S.; Maijenburg, M.W.; Nolte, M.A.; Voermans, C. Organ-Specific Migration of Mesenchymal Stromal Cells: Who, When, Where and Why? Immunol. Lett. 2015, 168, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Stappenbeck, T.S.; Miyoshi, H. The Role of Stromal Stem Cells in Tissue Regeneration and Wound Repair. Science 2009, 324, 1666–1669. [Google Scholar] [CrossRef]
- Egeblad, M.; Nakasone, E.S.; Werb, Z. Tumors as Organs: Complex Tissues That Interface with the Entire Organism. Dev. Cell 2010, 18, 884–901. [Google Scholar] [CrossRef]
- Karnoub, A.E.; Dash, A.B.; Vo, A.P.; Sullivan, A.; Brooks, M.W.; Bell, G.W.; Richardson, A.L.; Polyak, K.; Tubo, R.; Weinberg, R.A. Mesenchymal Stem Cells within Tumour Stroma Promote Breast Cancer Metastasis. Nature 2007, 449, 557–563. [Google Scholar] [CrossRef]
- Vu, B.T.; Shahin, S.A.; Croissant, J.; Fatieiev, Y.; Matsumoto, K.; Le-Hoang Doan, T.; Yik, T.; Simargi, S.; Conteras, A.; Ratliff, L.; et al. Chick Chorioallantoic Membrane Assay as an In Vivo Model to Study the Effect of Nanoparticle-Based Anticancer Drugs in Ovarian Cancer. Sci. Rep. 2018, 8, 8524. [Google Scholar] [CrossRef]
- DeBord, L.C.; Pathak, R.R.; Villaneuva, M.; Liu, H.-C.; Harrington, D.A.; Yu, W.; Lewis, M.T.; Sikora, A.G. The Chick Chorioallantoic Membrane (CAM) as a Versatile Patient-Derived Xenograft (PDX) Platform for Precision Medicine and Preclinical Research. Am. J. Cancer Res. 2018, 8, 1642–1660. [Google Scholar]
- Nowak-Sliwinska, P.; Segura, T.; Iruela-Arispe, M.L. The Chicken Chorioallantoic Membrane Model in Biology, Medicine and Bioengineering. Angiogenesis 2014, 17, 779–804. [Google Scholar] [CrossRef]
- Sys, G.; Van Bockstal, M.; Forsyth, R.; Balke, M.; Poffyn, B.; Uyttendaele, D.; Bracke, M.; De Wever, O. Tumor Grafts Derived from Sarcoma Patients Retain Tumor Morphology, Viability, and Invasion Potential and Indicate Disease Outcomes in the Chick Chorioallantoic Membrane Model. Cancer Lett. 2012, 326, 69–78. [Google Scholar] [CrossRef]
- Drexler, K.; Schwertner, B.; Haerteis, S.; Aung, T.; Berneburg, M.; Geissler, E.K.; Mycielska, M.E.; Haferkamp, S. The Role of Citrate Homeostasis in Merkel Cell Carcinoma Pathogenesis. Cancers 2022, 14, 3425. [Google Scholar] [CrossRef]
- Hu, J.; Ishihara, M.; Chin, A.I.; Wu, L. Establishment of Xenografts of Urological Cancers on Chicken Chorioallantoic Membrane (CAM) to Study Metastasis. Precis. Clin. Med. 2019, 2, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Kuri, P.M.; Pion, E.; Mahl, L.; Kainz, P.; Schwarz, S.; Brochhausen, C.; Aung, T.; Haerteis, S. Deep Learning-Based Image Analysis for the Quantification of Tumor-Induced Angiogenesis in the 3D In Vivo Tumor Model-Establishment and Addition to Laser Speckle Contrast Imaging (LSCI). Cells 2022, 11, 2321. [Google Scholar] [CrossRef] [PubMed]
- Morelli, C.; Campioni, K.; Parolin, C.; Palù, G.; Tognon, M. Activity of the Matrix Metalloproteinase-9 Promoter in Human Normal and Tumor Cells. J. Cell Physiol. 2004, 199, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Gialeli, C.; Theocharis, A.D.; Karamanos, N.K. Roles of Matrix Metalloproteinases in Cancer Progression and Their Pharmacological Targeting. FEBS J. 2011, 278, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Y.; Xiao, Q.; Yin, L.; Yang, L.; He, W. Potent Delivery of an MMP Inhibitor to the Tumor Microenvironment with Thermosensitive Liposomes for the Suppression of Metastasis and Angiogenesis. Sig. Transduct. Target. Ther. 2019, 4, 1–9. [Google Scholar] [CrossRef]
- Quintero-Fabián, S.; Arreola, R.; Becerril-Villanueva, E.; Torres-Romero, J.C.; Arana-Argáez, V.; Lara-Riegos, J.; Ramírez-Camacho, M.A.; Alvarez-Sánchez, M.E. Role of Matrix Metalloproteinases in Angiogenesis and Cancer. Front. Oncol. 2019, 9, 1370. [Google Scholar] [CrossRef] [PubMed]
- Hamano, Y.; Zeisberg, M.; Sugimoto, H.; Lively, J.C.; Maeshima, Y.; Yang, C.; Hynes, R.O.; Werb, Z.; Sudhakar, A.; Kalluri, R. Physiological Levels of Tumstatin, a Fragment of Collagen IV Alpha3 Chain, Are Generated by MMP-9 Proteolysis and Suppress Angiogenesis via AlphaV Beta3 Integrin. Cancer Cell 2003, 3, 589–601. [Google Scholar] [CrossRef]
- Bergers, G.; Brekken, R.; McMahon, G.; Vu, T.H.; Itoh, T.; Tamaki, K.; Tanzawa, K.; Thorpe, P.; Itohara, S.; Werb, Z.; et al. Matrix Metalloproteinase-9 Triggers the Angiogenic Switch during Carcinogenesis. Nat. Cell Biol. 2000, 2, 737–744. [Google Scholar] [CrossRef]
- Bedal, K.B.; Grässel, S.; Oefner, P.J.; Reinders, J.; Reichert, T.E.; Bauer, R. Collagen XVI Induces Expression of MMP9 via Modulation of AP-1 Transcription Factors and Facilitates Invasion of Oral Squamous Cell Carcinoma. PLoS ONE 2014, 9, e86777. [Google Scholar] [CrossRef]
- Hu, X.; Li, D.; Zhang, W.; Zhou, J.; Tang, B.; Li, L. Matrix Metalloproteinase-9 Expression Correlates with Prognosis and Involved in Ovarian Cancer Cell Invasion. Arch. Gynecol. Obstet. 2012, 286, 1537–1543. [Google Scholar] [CrossRef] [PubMed]
- Heikkilä, P.; Suojanen, J.; Pirilä, E.; Väänänen, A.; Koivunen, E.; Sorsa, T.; Salo, T. Human tongue carcinoma growth is inhibited by selective antigelatinolytic peptides. Int. J. Cancer 2006, 118, 2202–2209. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Yan, C.; Gieling, R.G.; Kida, Y.; Garner, W.; Li, W.; Han, Y.-P. Tumor Necrosis Factor-Alpha Induced Expression of Matrix Metalloproteinase-9 through P21-Activated Kinase-1. BMC Immunol. 2009, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Ratzinger, S.; Grassel, S.; Dowejko, A.; Reichert, T.E.; Bauer, R.J. Induction of Type XVI Collagen Expression Facilitates Proliferation of Oral Cancer Cells. Matrix Biol. 2011, 30, 118–125. [Google Scholar] [CrossRef]
- Hey, S.; Ratt, A.; Linder, S. There and Back Again: Intracellular Trafficking, Release and Recycling of Matrix Metalloproteinases. Biochim. Biophys. Acta Mol. Cell Res. 2022, 1869, 119189. [Google Scholar] [CrossRef]
- Roderfeld, M.; Graf, J.; Giese, B.; Salguero-Palacios, R.; Tschuschner, A.; Müller-Newen, G.; Roeb, E. Latent MMP-9 Is Bound to TIMP-1 before Secretion. Biol. Chem. 2007, 388, 1227–1234. [Google Scholar] [CrossRef]
- Ruokolainen, H.; Pääkkö, P.; Turpeenniemi-Hujanen, T. Expression of Matrix Metalloproteinase-9 in Head and Neck Squamous Cell Carcinoma: A Potential Marker for Prognosis. Clin. Cancer Res. 2004, 10, 3110–3116. [Google Scholar] [CrossRef]
- Ogbureke, K.U.E.; Weinberger, P.M.; Looney, S.W.; Li, L.; Fisher, L.W. Expressions of Matrix Metalloproteinase-9 (MMP-9), Dentin Sialophosphoprotein (DSPP), and Osteopontin (OPN) at Histologically Negative Surgical Margins May Predict Recurrence of Oral Squamous Cell Carcinoma. Oncotarget 2012, 3, 286–298. [Google Scholar] [CrossRef]
- Bhoopathi, P.; Chetty, C.; Gujrati, M.; Dinh, D.H.; Rao, J.S.; Lakka, S.S. The Role of MMP-9 in the Anti-Angiogenic Effect of Secreted Protein Acidic and Rich in Cysteine. Br. J. Cancer 2010, 102, 530–540. [Google Scholar] [CrossRef]
- Masson, V.; de la Ballina, L.R.; Munaut, C.; Wielockx, B.; Jost, M.; Maillard, C.; Blacher, S.; Bajou, K.; Itoh, T.; Itohara, S.; et al. Contribution of Host MMP-2 and MMP-9 to Promote Tumor Vascularization and Invasion of Malignant Keratinocytes. FASEB J. 2005, 19, 234–236. [Google Scholar] [CrossRef]
- Yu, Q.; Stamenkovic, I. Cell Surface-Localized Matrix Metalloproteinase-9 Proteolytically Activates TGF-Beta and Promotes Tumor Invasion and Angiogenesis. Genes Dev. 2000, 14, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Power, C.; Henry, S.; Del Bigio, M.R.; Larsen, P.H.; Corbett, D.; Imai, Y.; Yong, V.W.; Peeling, J. Intracerebral Hemorrhage Induces Macrophage Activation and Matrix Metalloproteinases. Ann. Neurol. 2003, 53, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.E.A.; Biernaskie, J.; Szymanska, A.; Larsen, P.H.; Yong, V.W.; Corbett, D. Matrix Metalloproteinase (MMP)-12 Expression Has a Negative Impact on Sensorimotor Function Following Intracerebral Haemorrhage in Mice. Eur. J. Neurosci. 2005, 21, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tsirka, S.E. Neuroprotection by Inhibition of Matrix Metalloproteinases in a Mouse Model of Intracerebral Haemorrhage. Brain 2005, 128, 1622–1633. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Hollenberg, M.D.; Yong, V.W. Combination of Thrombin and Matrix Metalloproteinase-9 Exacerbates Neurotoxicity in Cell Culture and Intracerebral Hemorrhage in Mice. J. Neurosci. 2006, 26, 10281–10291. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, G.A.; Navratil, M. Metalloproteinase Inhibition Blocks Edema in Intracerebral Hemorrhage in the Rat. Neurology 1997, 48, 921–926. [Google Scholar] [CrossRef]
- Gu, J.J.; Hoj, J.; Rouse, C.; Pendergast, A.M. Mesenchymal Stem Cells Promote Metastasis through Activation of an ABL-MMP9 Signaling Axis in Lung Cancer Cells. PLoS ONE 2020, 15, e0241423. [Google Scholar] [CrossRef]
- Ho, I.A.W.; Yulyana, Y.; Sia, K.C.; Newman, J.P.; Guo, C.M.; Hui, K.M.; Lam, P.Y.P. Matrix Metalloproteinase-1-Mediated Mesenchymal Stem Cell Tumor Tropism Is Dependent on Crosstalk with Stromal Derived Growth Factor 1/C-X-C Chemokine Receptor 4 Axis. FASEB J. 2014, 28, 4359–4368. [Google Scholar] [CrossRef]
- Krstic, J.; Trivanovic, D.; Jaukovic, A.; Santibanez, J.F.; Bugarski, D. Metabolic Plasticity of Stem Cells and Macrophages in Cancer. Front. Immunol. 2017, 8, 939. [Google Scholar] [CrossRef]
- Fiaschi, T.; Marini, A.; Giannoni, E.; Taddei, M.L.; Gandellini, P.; De Donatis, A.; Lanciotti, M.; Serni, S.; Cirri, P.; Chiarugi, P. Reciprocal Metabolic Reprogramming through Lactate Shuttle Coordinately Influences Tumor-Stroma Interplay. Cancer Res. 2012, 72, 5130–5140. [Google Scholar] [CrossRef]
- Van Kilsdonk, J.W.J.; Bergers, M.; Van Kempen, L.C.L.T.; Schalkwijk, J.; Swart, G.W.M. Keratinocytes Drive Melanoma Invasion in a Reconstructed Skin Model. Melanoma Res. 2010, 20, 372. [Google Scholar] [CrossRef] [PubMed]
- Van Valckenborgh, E.; Bakkus, M.; Munaut, C.; Noël, A.; St. Pierre, Y.; Asosingh, K.; Van Riet, I.; Van Camp, B.; Vanderkerken, K. Upregulation of Matrix Metalloproteinase-9 in Murine 5T33 Multiple Myeloma Cells by Interaction with Bone Marrow Endothelial Cells. Int. J. Cancer 2002, 101, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, S.; Matsuyama, Y.; Nitta, A.; Sakai, Y.; Ishiguro, N. Highly Efficient Transfection of Human Marrow Stromal Cells by Nucleofection. Transpl. Proc. 2005, 37, 2290–2292. [Google Scholar] [CrossRef] [PubMed]
- Lesueur, L.L.; Mir, L.M.; André, F.M. Overcoming the Specific Toxicity of Large Plasmids Electrotransfer in Primary Cells In Vitro. Mol. Ther. Nucleic Acids 2016, 5, e291. [Google Scholar] [CrossRef]
- Rufaihah, A.J.; Cheyyatraivendran, S.; Mazlan, M.D.M.; Lim, K.; Chong, M.S.K.; Mattar, C.N.Z.; Chan, J.K.Y.; Kofidis, T.; Seliktar, D. The Effect of Scaffold Modulus on the Morphology and Remodeling of Fetal Mesenchymal Stem Cells. Front. Physiol. 2018, 9, 1555. [Google Scholar] [CrossRef]
- Bauman, E.; Feijão, T.; Carvalho, D.T.O.; Granja, P.L.; Barrias, C.C. Xeno-Free Pre-Vascularized Spheroids for Therapeutic Applications. Sci. Rep. 2018, 8, 230. [Google Scholar] [CrossRef]
- Lin, C.J.; Grandis, J.R.; Carey, T.E.; Gollin, S.M.; Whiteside, T.L.; Koch, W.M.; Ferris, R.L.; Lai, S.Y. Head and Neck Squamous Cell Carcinoma Cell Lines: Established Models and Rationale for Selection. Head Neck 2007, 29, 163–188. [Google Scholar] [CrossRef]
- Heo, D.S.; Snyderman, C.; Gollin, S.M.; Pan, S.; Walker, E.; Deka, R.; Barnes, E.L.; Johnson, J.T.; Herberman, R.B.; Whiteside, T.L. Biology, Cytogenetics, and Sensitivity to Immunological Effector Cells of New Head and Neck Squamous Cell Carcinoma Lines. Cancer Res. 1989, 49, 5167–5175. [Google Scholar]
- Leyh, M.; Seitz, A.; Dürselen, L.; Schaumburger, J.; Ignatius, A.; Grifka, J.; Grässel, S. Subchondral Bone Influences Chondrogenic Differentiation and Collagen Production of Human Bone Marrow-Derived Mesenchymal Stem Cells and Articular Chondrocytes. Arthritis Res. Ther. 2014, 16, 453. [Google Scholar] [CrossRef]
- Chitturi Suryaprakash, R.T.; Kujan, O.; Shearston, K.; Farah, C.S. Three-Dimensional Cell Culture Models to Investigate Oral Carcinogenesis: A Scoping Review. Int. J. Mol. Sci. 2020, 21, 9520. [Google Scholar] [CrossRef]
- Kohl, C.; Aung, T.; Haerteis, S.; Papathemelis, T. Assessment of Breast Cancer Primary Tumor Material in a 3D in Vivo Model. Clin. Hemorheol. Microcirc. 2021, 79, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Sherf, B.; Navarro, S.L.; Hannah, R.; Wood, K. Dual-Luciferase TM Reporter Assay: An Advanced Co-Reporter Technology Integrating Firefly and Renilla Luciferase Assays. Promega Notes 1996, 57, 2–8. [Google Scholar]
| Cell Line | Origin | TNM Status | Type of Tumor | Age, Sex |
|---|---|---|---|---|
| SCC-9 | tongue | n.a. | primary tumor | 25, m |
| SCC-15 | tongue | n.a. | primary tumor | 55, m |
| PCI 1 | larynx | pT2N0M0G2 | primary tumor recurrence | 65, m |
| PCI 13 | retromolar area | pT4N1M0G3 | primary tumor | 50, m |
| PCI 68 | tongue | pT4N0M0G1 | n.a. | n.a. |
| Donor Number | Age, Sex | Origin |
|---|---|---|
| 706 | 67, m | Acetabulum |
| 821 | 60, f | Acetabulum |
| 822 | 84, f | Acetabulum |
| 866 | 63, f | Caput femoris |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waltera, A.; Schulz, D.; Schaefer, N.; Stoeckl, S.; Pion, E.; Haerteis, S.; Reichert, T.E.; Ettl, T.; Bauer, R.J. Opposing MMP-9 Expression in Mesenchymal Stromal Cells and Head and Neck Tumor Cells after Direct 2D and 3D Co-Culture. Int. J. Mol. Sci. 2023, 24, 1293. https://doi.org/10.3390/ijms24021293
Waltera A, Schulz D, Schaefer N, Stoeckl S, Pion E, Haerteis S, Reichert TE, Ettl T, Bauer RJ. Opposing MMP-9 Expression in Mesenchymal Stromal Cells and Head and Neck Tumor Cells after Direct 2D and 3D Co-Culture. International Journal of Molecular Sciences. 2023; 24(2):1293. https://doi.org/10.3390/ijms24021293
Chicago/Turabian StyleWaltera, Anna, Daniela Schulz, Nicole Schaefer, Sabine Stoeckl, Eric Pion, Silke Haerteis, Torsten E. Reichert, Tobias Ettl, and Richard J. Bauer. 2023. "Opposing MMP-9 Expression in Mesenchymal Stromal Cells and Head and Neck Tumor Cells after Direct 2D and 3D Co-Culture" International Journal of Molecular Sciences 24, no. 2: 1293. https://doi.org/10.3390/ijms24021293
APA StyleWaltera, A., Schulz, D., Schaefer, N., Stoeckl, S., Pion, E., Haerteis, S., Reichert, T. E., Ettl, T., & Bauer, R. J. (2023). Opposing MMP-9 Expression in Mesenchymal Stromal Cells and Head and Neck Tumor Cells after Direct 2D and 3D Co-Culture. International Journal of Molecular Sciences, 24(2), 1293. https://doi.org/10.3390/ijms24021293








