Degradation of Diclofenac by Loaded Solid Superbase-Activated Persulfate
Abstract
:1. Introduction
2. Results and Discussion
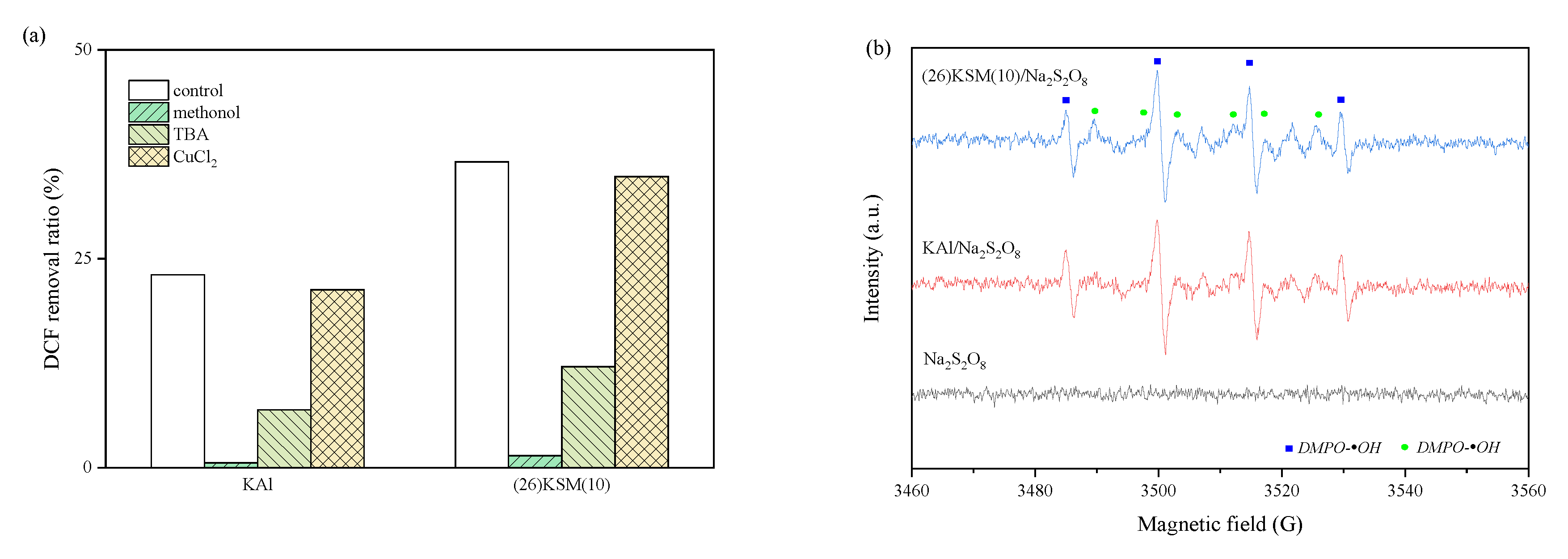
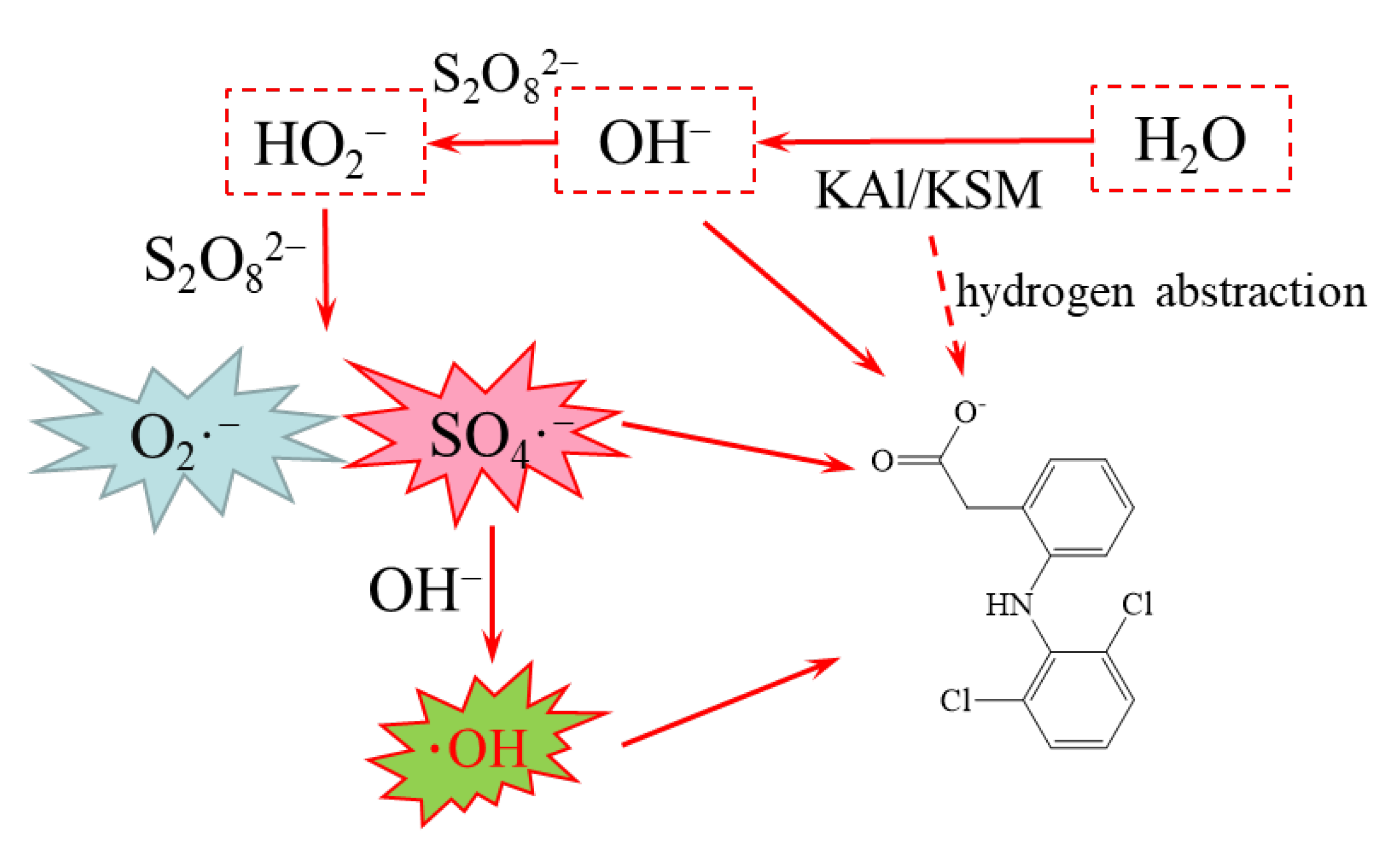
2.1. Identification of Free Radical Species
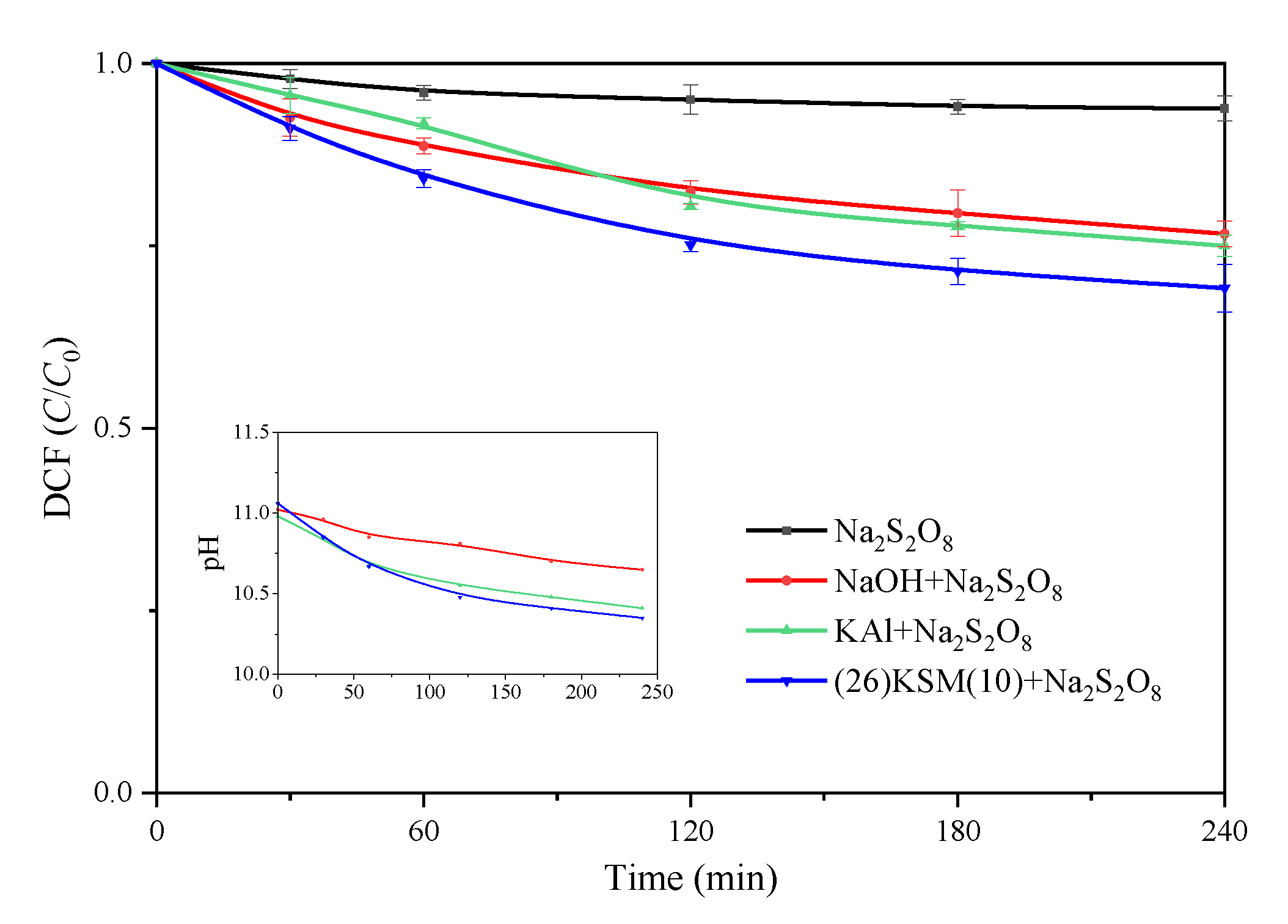
2.2. DCF Removal Effect by Different Bases Activated Persulfate
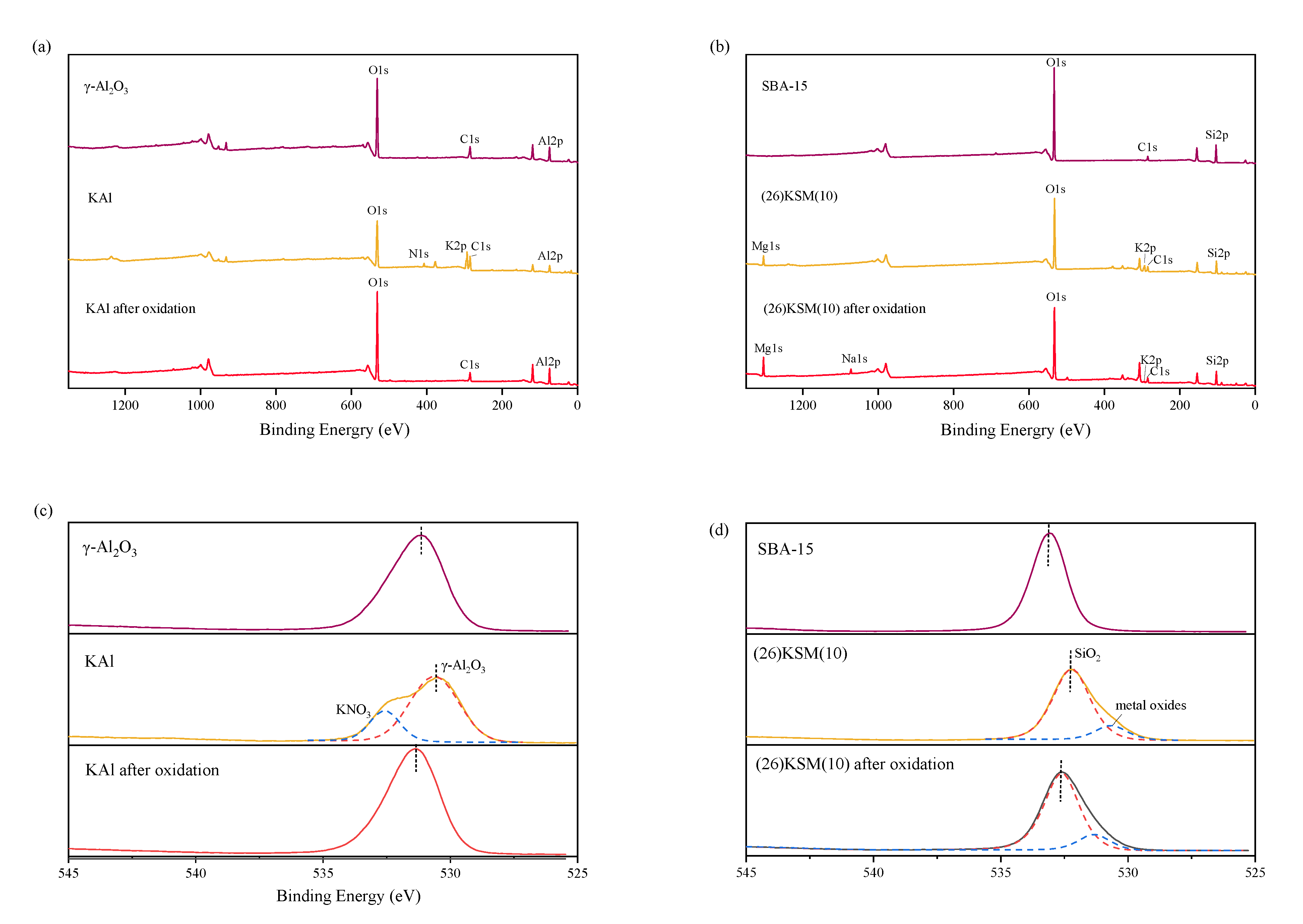
2.3. XPS Analysis
2.4. Influence of MgO and Alkali Loading Ratio on KSM-Activating Effect
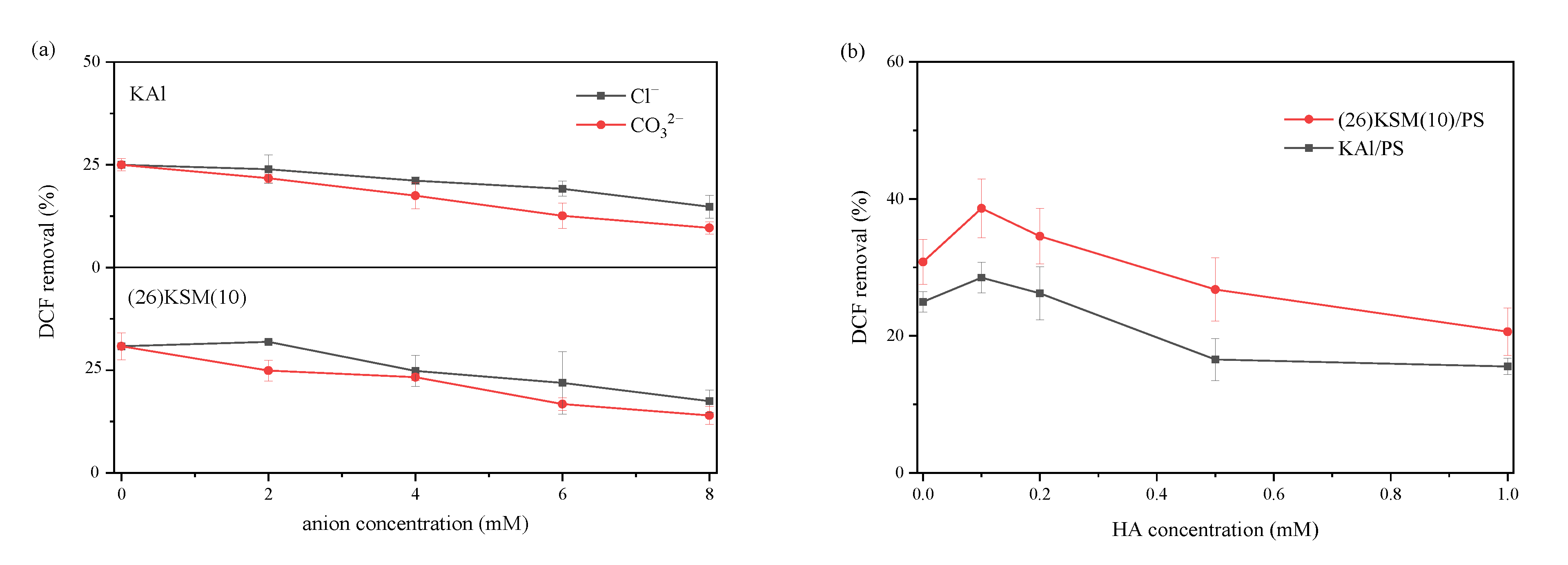
2.5. The Effect of Inorganic Anions and Humic Acid
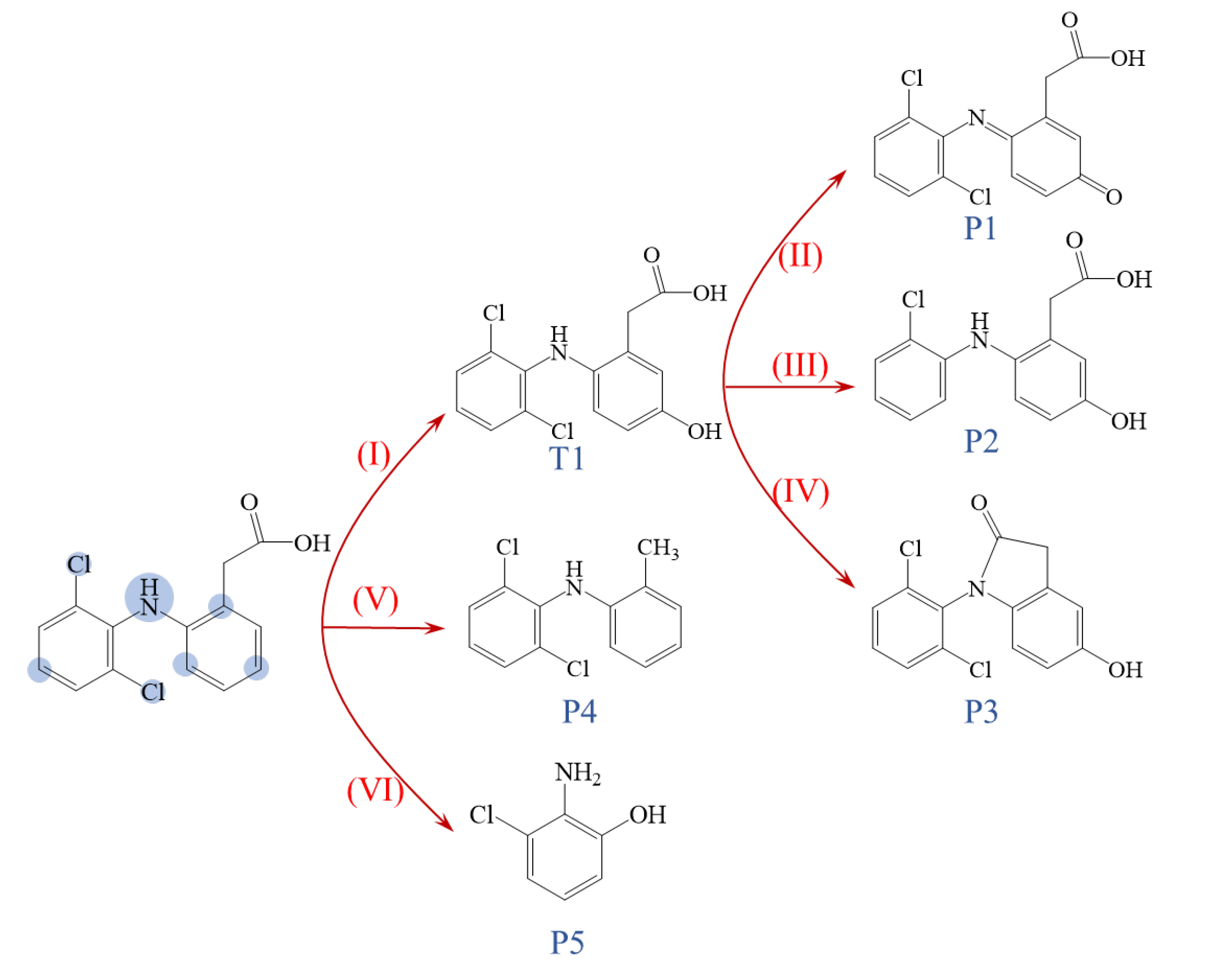
2.6. Proposed Degradation Pathways and Toxicity Assessment of Products
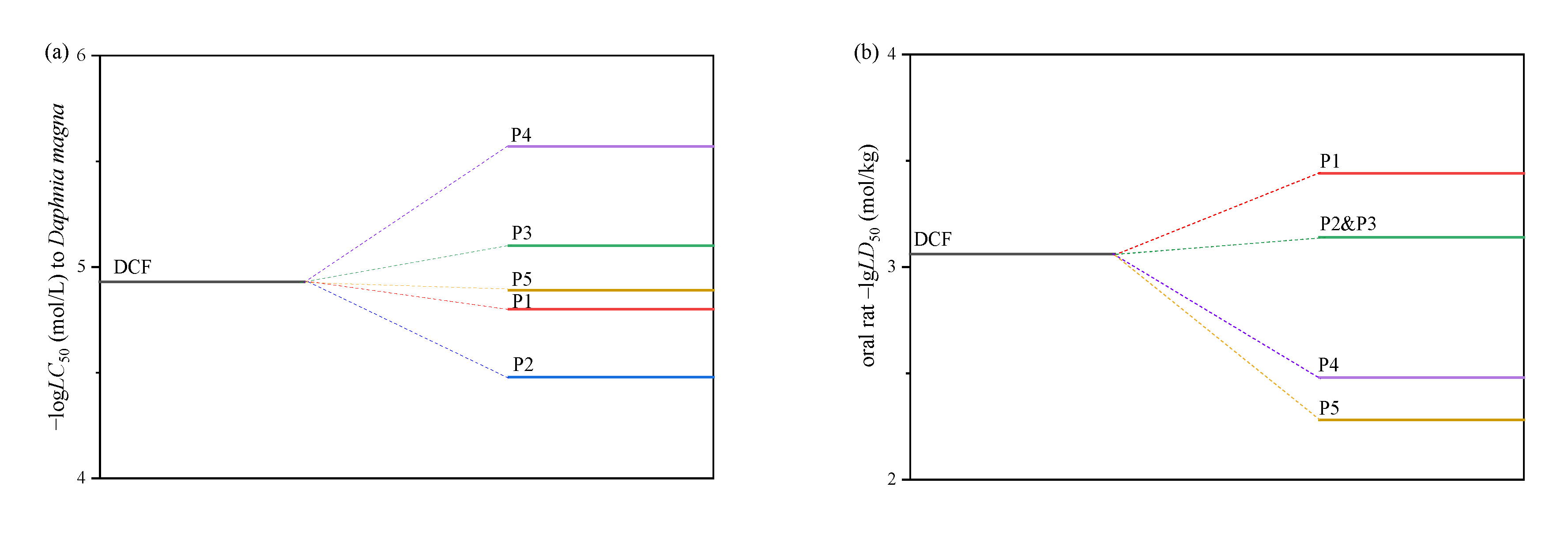
2.7. Toxicity Assessments
3. Materials and Methods
3.1. Chemical Reagents and Equipment
3.2. Preparation of Solid Superbases
3.3. Characterization of Solid Superbase
3.4. Degradation Experiment
3.5. Analytical Methods
3.6. Toxicity Prediction
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Di Lorenzo, T.; Cifoni, M.; Baratti, M.; Pieraccini, G.; Di Marzio, W.D.; Galassi, D.M.P. Four scenarios of environmental risk of diclofenac in European groundwater ecosystems. Environ. Pollut. 2021, 287, 117315. [Google Scholar] [CrossRef] [PubMed]
- Sathishkumar, P.; Meena, R.A.A.; Palanisami, T.; Ashokkumar, V.; Palvannan, T.; Gu, F.L. Occurrence, interactive effects and ecological risk of diclofenac in environmental compartments and biota—A review. Sci. Total Environ. 2020, 698, 134057. [Google Scholar] [CrossRef] [PubMed]
- Machuca-Martinez, F.; Madera-Parra, C.A.; Jiménez-Bambague, E.M. The Occurrence of Emerging Compounds in Real Urban Wastewater Before and After the COVID-19 Pandemic in Cali, Colombia. Curr. Opin. Environ. Sci. Health 2023, 33, 100457. [Google Scholar]
- Duan, L.; Zhang, Y.; Wang, B.; Yu, G.; Gao, J.; Cagnetta, G.; Huang, C.; Zhai, N. Wastewater surveillance for 168 pharmaceuticals and metabolites in a WWTP: Occurrence, temporal variations and feasibility of metabolic biomarkers for intake estimation. Water Res. 2022, 216, 118321. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Hara, H.; Watanabe, Y. Elimination of Selected Acidic Pharmaceuticals from Municipal Wastewater by an Activated Sludge System and Membrane Bioreactors. Environ. Sci. Technol. 2007, 41, 3708–3714. [Google Scholar] [CrossRef] [PubMed]
- Żur, J.; Piński, A.; Wojcieszyńska, D.; Smułek, W.; Guzik, U. Diclofenac Degradation—Enzymes, Genetic Background and Cellular Alterations Triggered in Diclofenac-Metabolizing Strain Pseudomonas moorei KB4. Int. J. Mol. Sci. 2020, 21, 6786. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Shao, Y.; Gao, N.; Chen, J.; Zhang, Y.; Xiang, H.; Guo, Y. Degradation of diclofenac by UV-activated persulfate process: Kinetic studies, degradation pathways and toxicity assessments. Ecotoxicol. Environ. Saf. 2017, 141, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, H.; Liu, G.; Xie, Y.; Gao, S. Oxidation of diclofenac by potassium ferrate (VI): Reaction kinetics and toxicity evaluation. Sci. Total Environ. 2015, 506–507, 252–258. [Google Scholar] [CrossRef]
- Gao, Y.; Rao, Y.; Ning, H.; Chen, J.; Zeng, Q.; Tian, F.; Gao, N. Comparative investigation of diclofenac degradation by Fe2+/chlorine and Fe2+/PMS processes. Sep. Purif. Technol. 2022, 297, 121555. [Google Scholar] [CrossRef]
- Qu, H.; Chen, L.; Yang, F.; Zhu, J.; Qi, C.; Peng, G. Synthesis of an Environmentally Friendly Modified Mulberry Branch-Derived Biochar Composite: High Degradation Efficiency of BPA and Mitigation of Toxicity in Silkworm Larvae. Int. J. Mol. Sci. 2023, 24, 3609. [Google Scholar] [CrossRef]
- Furman, O.S.; Teel, A.L.; Watts, R.J. Mechanism of Base Activation of Persulfate. Environ. Sci. Technol. 2010, 44, 6423–6428. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Chen, X.; Meng, W.; Zhu, M. Research progress on supported solid superbase and its catalytic application. Chem. Pap. 2021, 75, 4445–4463. [Google Scholar] [CrossRef]
- Chen, L.; Zhao, J.; Yin, S.; Au, C. A mini-review on solid superbase catalysts developed in the past two decades. RSC Adv. 2013, 3, 3799. [Google Scholar] [CrossRef]
- Crimi, M.L.; Taylor, J. Experimental Evaluation of Catalyzed Hydrogen Peroxide and Sodium Persulfate for Destruction of BTEX Contaminants. Soil Sediment Contam. Int. J. 2007, 16, 29–45. [Google Scholar] [CrossRef]
- Zhang, Y.; Qian, W.; Zhou, P.; Liu, Y.; Lei, X.; Li, B.; Ning, P. Research on red mud-limestone modified desulfurization mechanism and engineering application. Sep. Purif. Technol. 2021, 272, 118867. [Google Scholar] [CrossRef]
- Zhao, J.; Xie, J.; Au, C.; Yin, S. Novel and versatile solid superbases derived from magnesium–zirconium composite oxide and their catalytic applications. RSC Adv. 2014, 4, 6159–6164. [Google Scholar] [CrossRef]
- Lide, D.R. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 2004; Volume 85. [Google Scholar]
- Zhang, S.; Wei, Y.; Yin, S.; Luo, S.; Au, C. Superbasic sodium stannate as catalyst for dehydrogenation, Michael addition and transesterification reactions. Appl. Catal. A Gen. 2011, 406, 113–118. [Google Scholar] [CrossRef]
- Wu, Z.Y.; Jiang, Q.; Wang, Y.M.; Wang, H.J.; Sun, L.B.; Shi, L.Y.; Xu, J.H.; Wang, Y.; Chun, Y.; Zhu, J.H. Generating Superbasic Sites on Mesoporous Silica SBA-15. Chem. Mater. 2006, 18, 4600–4608. [Google Scholar] [CrossRef]
- Tan, C.; Gao, N.; Deng, Y.; Zhang, Y.; Sui, M.; Deng, J.; Zhou, S. Degradation of antipyrine by UV, UV/H2O2 and UV/PS. J. Hazard. Mater. 2013, 260, 1008–1016. [Google Scholar] [CrossRef]
- Ahmed, N.; Vione, D.; Rivoira, L.; Carena, L.; Castiglioni, M.; Bruzzoniti, M.C. A Review on the Degradation of Pollutants by Fenton-Like Systems Based on Zero-Valent Iron and Persulfate: Effects of Reduction Potentials, pH, and Anions Occurring in Waste Waters. Molecules 2021, 26, 4584. [Google Scholar] [CrossRef]
- Ahmad, M.; Teel, A.L.; Watts, R.J. Mechanism of Persulfate Activation by Phenols. Environ. Sci. Technol. 2013, 47, 5864–5871. [Google Scholar] [CrossRef] [PubMed]
- Fang, G.; Gao, J.; Dionysiou, D.D.; Liu, C.; Zhou, D. Activation of Persulfate by Quinones: Free Radical Reactions and Implication for the Degradation of PCBs. Environ. Sci. Technol. 2013, 47, 4605–4611. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Long, T.; Zhou, Y.; Wang, L.; Jiang, C.; Pan, D.; Zhu, X. Efficiency and Quantitative Structure-Activity Relationship of Monoaromatics Oxidation by Quinone-Activated Persulfate. Front. Chem. 2021, 9, 580643. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Zhou, G.; Liu, Y.; Fu, Y.; Wang, H.; Wu, P. Kinetics and pathways of diclofenac degradation by heat-activated persulfate. RSC Adv. 2019, 9, 31370–31377. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; He, S.; Zhu, M.; Wei, C. The effect of peroxymonosulfate in WS2 nanosheets for the removal of diclofenac: Information exposure and degradation pathway. Chemosphere 2020, 245, 125678. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Jiang, J.; Chen, Q.; Wang, L.; Nian, K.; Long, T. Production of higher toxic intermediates of organic pollutants during chemical oxidation processes: A review. Arab. J. Chem. 2023, 16, 104856. [Google Scholar] [CrossRef]
- Sun, L.B.; Yang, J.; Kou, J.H.; Gu, F.N.; Chun, Y.; Wang, Y.; Zhu, J.H.; Zou, Z.G. One-pot synthesis of potassium-functionalized mesoporous gamma-alumina: A solid superbase. Angew. Chem. Int. Ed. 2008, 47, 3418–3421. [Google Scholar] [CrossRef] [PubMed]
- Mozaffari, N.; Ţălu, Ş.; Mozaffari, N.; Sillanpää, M.; Vambol, V.; Vambol, S. Pharmaceuticals Measurements and Estimation Methods; IWA Publishing: London, UK, 2022. [Google Scholar]
- Zhao, J.; Zhang, Y.; Quan, X.; Chen, S. Enhanced oxidation of 4-chlorophenol using sulfate radicals generated from zero-valent iron and peroxydisulfate at ambient temperature. Sep. Purif. Technol. 2010, 71, 302–307. [Google Scholar] [CrossRef]
- Wu, G.; Katsumura, Y.; Chu, G. Photolytic and radiolytic studies of SO4•− in neat organic solvents. Phys. Chem. Chem. Phys. 2000, 2, 5602–5605. [Google Scholar] [CrossRef]
- Dang, Y.; Cheng, Y.; Zhou, Y.; Huang, Y.; Wang, K. Nano-PAA-CuCl2 Composite as Fenton-Like Reusable Catalyst to Enhanced Degrade Organic Pollutant MB/MO. Catalysts 2021, 11, 10. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, W.; Chun, Y.; Xia, J.; Zhu, J. Dispersion of potassium nitrate and the resulting strong basicity on zirconia. Chem. Mat. 2001, 13, 670–677. [Google Scholar] [CrossRef]
- Yin, S.; Xu, B.; Wang, S.; Au, C. Nanosized Ru on high-surface-area superbasic ZrO2-KOH for efficient generation of hydrogen via ammonia decomposition. Appl. Catal. A General. 2006, 301, 202–210. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Wang, L.; Gao, S.; Huang, J.; Yang, H.; Lu, H.; Cao, S. Degradation of Diclofenac by Loaded Solid Superbase-Activated Persulfate. Int. J. Mol. Sci. 2023, 24, 14313. https://doi.org/10.3390/ijms241814313
Shi J, Wang L, Gao S, Huang J, Yang H, Lu H, Cao S. Degradation of Diclofenac by Loaded Solid Superbase-Activated Persulfate. International Journal of Molecular Sciences. 2023; 24(18):14313. https://doi.org/10.3390/ijms241814313
Chicago/Turabian StyleShi, Jiaqi, Lei Wang, Shang Gao, Jianbo Huang, Hao Yang, Hao Lu, and Shaohua Cao. 2023. "Degradation of Diclofenac by Loaded Solid Superbase-Activated Persulfate" International Journal of Molecular Sciences 24, no. 18: 14313. https://doi.org/10.3390/ijms241814313
APA StyleShi, J., Wang, L., Gao, S., Huang, J., Yang, H., Lu, H., & Cao, S. (2023). Degradation of Diclofenac by Loaded Solid Superbase-Activated Persulfate. International Journal of Molecular Sciences, 24(18), 14313. https://doi.org/10.3390/ijms241814313






